Abstract
In the last decades, poly(methyl methacrylate) (PMMA) has been widely studied due to its outstanding mechanical, chemical, and physical properties. In this article, the PMMA composites doped with an excited state intramolecular proton transfer (ESIPT) compound 3-hydroxy-2-naphthoic acid (3HNA) are reported. The 3HNA-doped PMMA composites were synthesized by in situ polymerization and characterized by Fourier transform infrared spectra, ultraviolet absorption spectra, fluorescence spectra, and thermogravimetric analysis. The effect of 3HNA and various solvents on the fluorescent properties of the PMMA composites was systematically investigated. The results indicated that the emission spectra of the 3HNA-doped PMMA composites exhibited 3HNA dual emission. A violet emission was observed in 3HNA-doped PMMA composites containing nonpolar solvents or weak polar solvents, whereas the composites containing strong polar solvents exhibited a large Stokes-shifted green fluorescence, which was attributed to the ESIPT of 3HNA.
Keywords
Introduction
Excited state intramolecular proton transfer (ESIPT) is one of the most important photoexcitation processes in nature. It is well known that in an excited state, a molecule can undergo structural changes and assumes a different geometry due to significant charge redistribution, thereby greatly changing the excited state’s properties. In ESIPT compounds, an excited state tautomeric reaction concerning a PT from a proton donor to an acceptor group occurs via an intramolecular H bond or H bonding bridge, which leads to an intrinsic peculiar four-level photophysical cycle (absorption–PT–emission–back to PT) after photoexcitation. 1 The presence of the intramolecular hydrogen bond (IMHB) in the molecule is an essential prerequisite for ESIPT reactions. 2 An asymmetric double well-type potential model was proposed by Weller to describe the feasibility of the PT reaction for the ground and excited states. 3 The resulting PT tautomer or the enol (E) tautomer differed vastly from its corresponding keto (K) tautomer in structure, charge density distribution, dipole moment, and emission properties. Hence, the ESIPT process typically exhibited a large Stokes-shifted fluorescence. Recently, intense experimental 4 –7 and theoretical 8 –10 studies on the ESIPT materials have been executed due to potential applications such as chemical sensors, 11,12 fluorescence imaging, 13 laser dyes, 14 organic light-emitting diodes, 15 and white light generation. 16
3-Hydroxy-2-naphthoic acid (3HNA) with ESIPT property is an important intermediate for dye and organic pigment and has been widely used in printing and dyeing industry, waterproof agent, medicine, and photosensitive materials. Ware et al. reported the normal emission of 3HNA in nonpolar solvents such as toluene and the Stokes-shifted emission in polar solvents such as acetonitrile and pyridine. 17 However, Stokes-shifted emission was also observed when a proton acceptor such as triethylamine (TEA) was added to the nonpolar solvent. Pal et al. studied the enhancement of the ESIPT emission of 3HNA in the presence of poly(N-vinyl-2-pyrrolidone). 18 Mishra et al. studied the spectral and photophysical properties of 3HNA and revealed that 3HNA seemed to form different emitting species in different media. 19
Poly(methyl methacrylate) (PMMA) has been widely studied in the last decades due to its outstanding mechanical and chemicophysical properties. Its wide applications in many technological and productive fields take advantage of the unique combination of excellent optical properties, chemical inertness, good mechanical properties, thermal stability, electrical properties, safety, weather resistance, formability, and moldability. 20 To the best of our knowledge, the ESIPT process of 3HNA was mainly investigated in various solvents and media with different pH values. So far it is rare to study the 3HNA-doped PMMA composites. The composites should show excellent potentials for the combination of ESIPT of 3HNA and the outstanding properties of PMMA.
In this article, a series of PMMA composites containing 3HNA and solvents were synthesized by in situ polymerization. Moreover, the effect of 3HNA and various solvents (especially dimethylformamide (DMF), N, N-dimethylacetamide (DMAC), and TEA) on the fluorescence properties of the composites was systematically investigated in the solid state.
Experimental
For the purpose of this study, 3.00 g methyl methacrylate, 0.003 g (0.1 wt%) 2,2-azobisisobutyronitrile (AIBN), approximately 0.003–0.06 g (approximately 0.1–2.0 wt%) 3HNA, and approximately 0–0.45 g (approximately 0–15.0 wt%) solvent (such as DMF, DMAC, and TEA) were added to a polymerization tube. The mixture was fully oscillated to dissolve 3HNA and AIBN completely. The polymerization tubes were placed in an oven at about 333 K for a week. Then, at 373 K, transparent 3HNA-doped PMMA composites were obtained after 3 h.
Infrared (IR) spectra were recorded on a Fourier transform infrared spectrophotometer (model 380; Nicolet, Madison, Wisconsin, USA) with potassium bromide pellet in the range 4000–400 cm−1. Ultraviolet (UV) absorption spectra were depicted in a Lambda 900 UV/visible (Vis)/near IR spectrophotometer (Perkin Elmer, Waltham, Massachusetts, USA). The fluorescent spectra were measured on an F900 spectrophotometer (Edinburgh, UK) in the solid state at room temperature. Thermogravimetric analysis (TGA) was performed under nitrogen (N2) atmosphere using a Fourier transform IR (FTIR) Mettler TGA/SDTA 851 thermogravimetric analyzer (model 5700; Nicolet) at a heating rate of 5 K min−1 from 303 K to 873 K.
Results and discussion
IR spectral analysis
The spectra of the 3HNA and the 3HNA-doped PMMA composites are shown in Figure 1. In the IR spectrum of 3HNA (curve 1), the O–H stretching vibrations of C–OH group and the COOH group appear at 3284 and 2557 cm−1, respectively. A very strong band at 1666 cm−1 correspond to C=O stretching vibrations due to intramolecular and intermolecular hydrogen bonding interaction in 3HNA. The peaks at 1599, 1512, and 1465 cm−1 correspond to naphthalene ring C–C stretching vibrations. 21 The band at 660 cm−1 is assigned to naphthalene ring out-of-plane bending vibration. The peaks at 904 and 1443 cm−1 correspond to out-of-plane and in-plane O–H bending vibration of the COOH group, respectively, and the peaks at 636 and 1352 cm−1 are assigned to out-of-plane and in-plane O–H bending vibration of the C–OH group, respectively. The C–O stretching vibrations of C–OH and COOH groups appear at 1219 and 1283 cm–1, respectively. 22
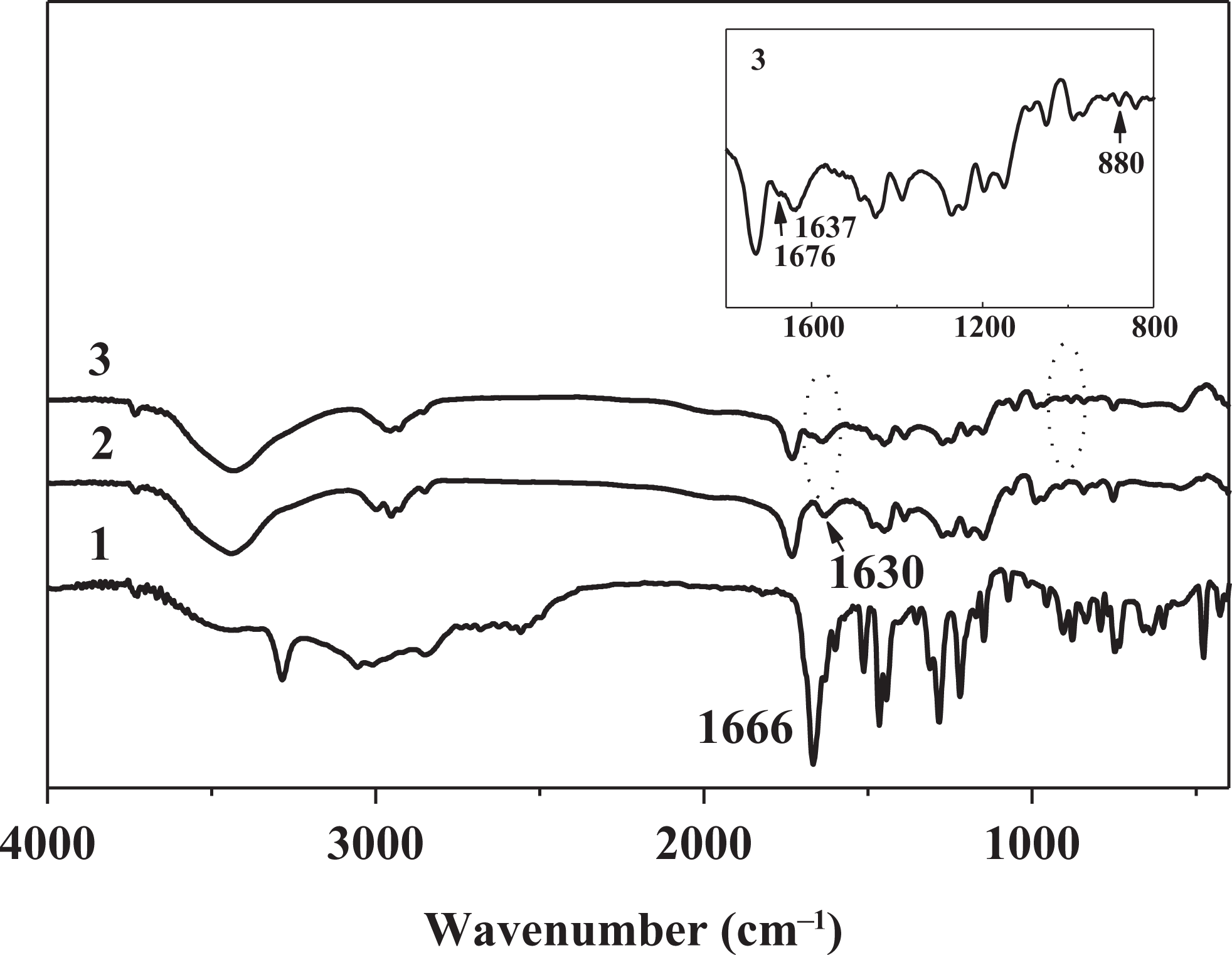
IR spectra of 3HNA (1) and 2 wt% 3HNA-doped PMMA composites: without DMF (2) and with 10 wt% DMF (3). IR: infrared; 3HNA: 3-hydroxy-2-naphthoic acid; PMMA: poly(methyl methacrylate); DMF: dimethylformamide.
A new absorption band that appears at 1630 cm−1 in the IR spectra of the 3HNA-doped PMMA composites without DMF (curve 2) is attributed to the C=O stretching vibrations of 3HNA and it shows bathochromic shift about 36 cm−1 in comparison with the IR spectrum of 3HNA.
When 10 wt% DMF is added to the 3HNA-doped PMMA composites, the new absorption bands at 1676 and 880 cm− 1 appear as shown in Figure 1 (curve 3) and correspond to C=O and C–N stretching vibrations of DMF. 21 The absorption band at 1630 cm− 1 corresponding to C=O stretching vibration of 3HNA shifts to 1637 cm− 1. The result indicates that the hydrogen bond between 3HNA and DMF might be formed in the PMMA composites containing 3HNA and DMF, but it may affect the HNA IMHB. Therefore, the C=O stretching vibration frequency shows a slight red shift. The shape and position of other absorption bands are similar to those of 3HNA-doped PMMA composite without DMF in the FTIR spectra.
TGA-DTG studies
TG-derivative thermogravimetric (DTG) analyses were carried out up to 873 K under N2 atmosphere at a heating rate of 5 K min−1. The TGA–DTG curves of the composites are similar. TGA and DTG curves of the PMMA composite doped with 1 wt% 3HNA and 7 wt% DMF in the polymerization system are depicted in Figure 2. TGA curve of the composite shows the first mass loss between 383 K and 473 K, and the mass loss percentage is 3.5%. At the same time, DTG curve shows a small peak at 399 K. It may be attributed to release the solvent DMF. A followed mass loss (approximately 3.0%) between 473 K and 543 K can be attributed to the release of the residual DMF and bonded DMF from an intramolecular H bond complex, corresponding to a weak peak centered at 528 K in DTG curve. In addition, there is a remarkable peak in the DTG curve at 649 K accompanied with an obvious mass loss in the TGA curve, which is attributed to the decomposition of 3HNA and PMMA. The results prove that the composites have relatively high thermal stability and there is 6.5 wt% DMF in the composite, which is close to the 7.0 wt% doping amount.
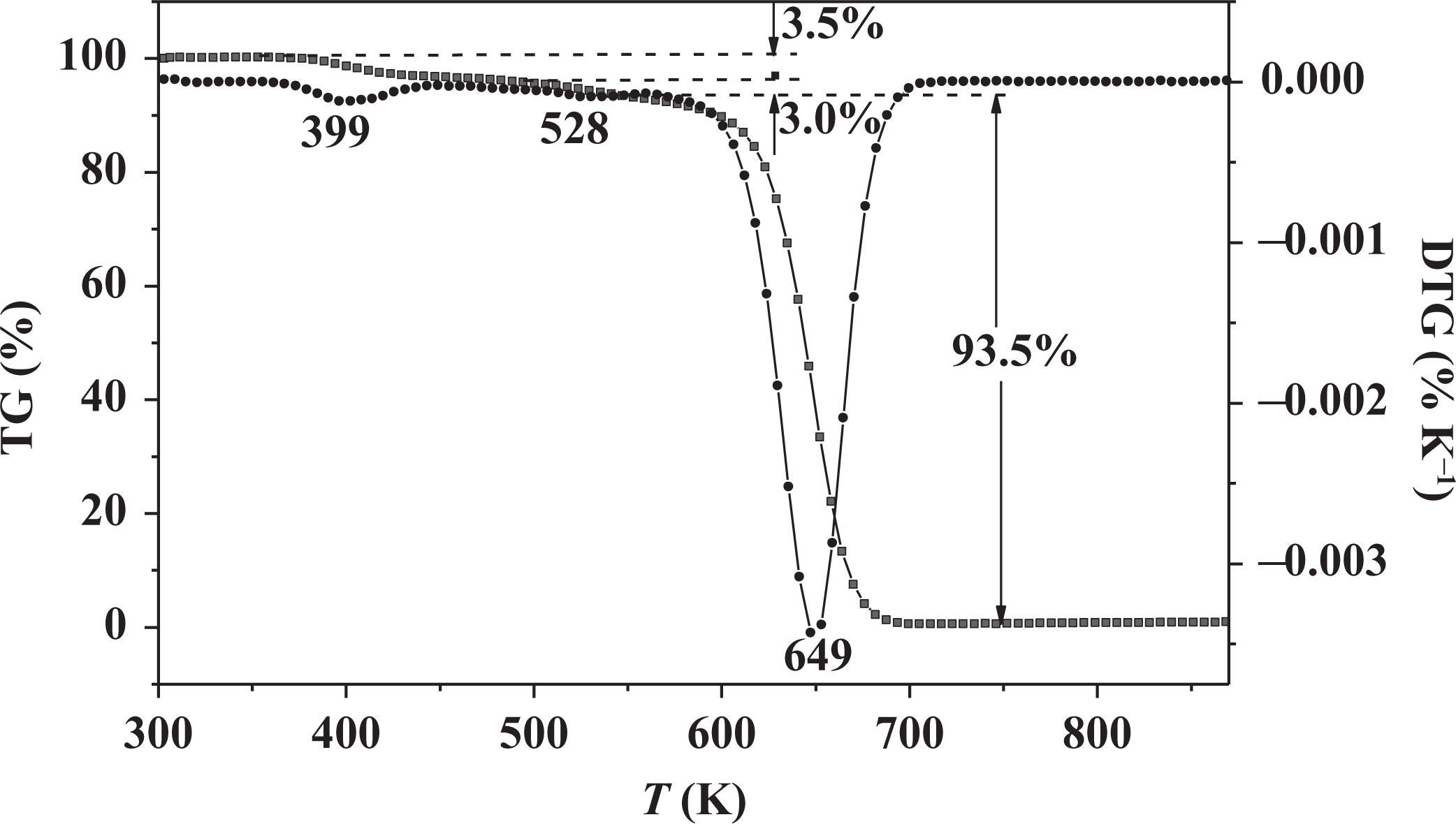
TGA and DTG curves of 1 wt% 3HNA-doped PMMA composite with 7 wt% DMF. TGA: thermogravimetric analysis; DTG: derivative thermogravimetry; 3HNA: 3-hydroxy-2-naphthoic acid; PMMA: poly(methyl methacrylate); DMF: dimethylformamide.
UV spectral analysis
UV-Vis spectra for 0.5 wt% 3HNA-doped PMMA composites without DMF and with 10 wt% DMF were determined in the solid powder state at room temperature and showed broad absorption bands in the near UV range (200–420 nm) as shown in Figure 3. In the UV-Vis absorption spectrum of the composite without DMF, there are four absorption peaks at about <200, 250, 288, and 375 nm, and the intensity of absorption gradually decreases. The absorption peak at 375 nm (R band) is attributed to n → π* transition of C=O group and others are assigned to π → π* transition of naphthalene ring (aromatic ring). The UV-Vis spectrum of the composite with DMF is similar to that of the composite without DMF, but the remarkable differences in the red shift from <200 nm to 206 nm and the blue shift from 375 nm to 365 nm are observed, with relative intensity of absorption in R band obviously increasing in the presence of DMF. The results indicate that the presence of DMF in the composite system obviously enhances absorption in the longer wavelength and the lower energy region.
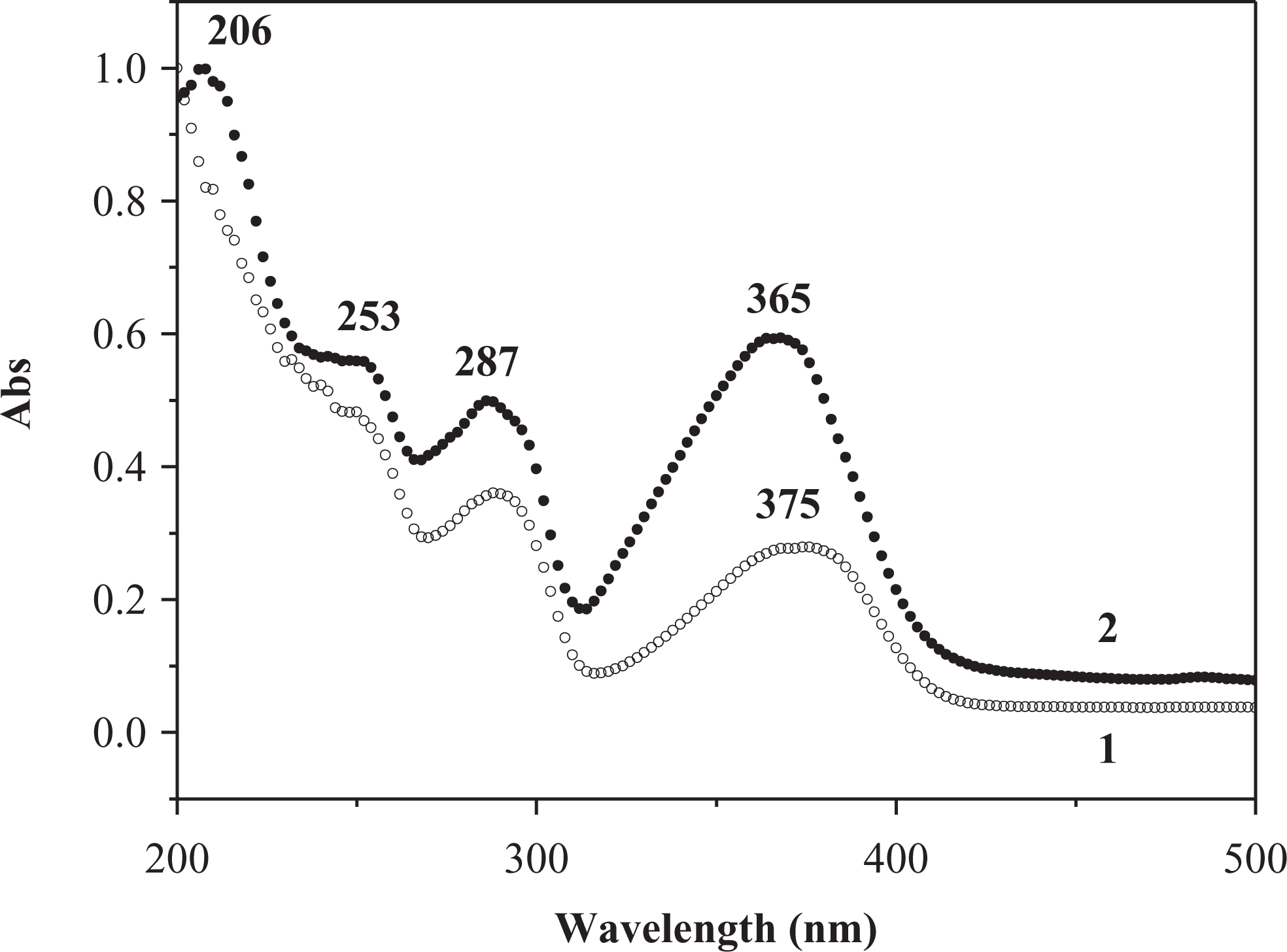
UV-Vis absorption spectra of 0.5 wt% 3HNA-doped PMMA composites: (1) without DMF and (2) with 10 wt% DMF. UV-Vis: ultraviolet–visible; 3HNA: 3-hydroxy-2-naphthoic acid; PMMA: poly(methyl methacrylate); DMF: dimethylformamide.
Fluorescence properties
The excitation and emission spectra of 3HNA in solid state at room temperature are recorded in Figure 4. The excitation spectrum is obtained by monitoring the emission of 3HNA at 510 nm. There is a wide and strong excitation band from 300 nm to 450 nm, which indicates that 3HNA can be excited after absorbing energy in the wider wavelength region. The fluorescence emission spectrum of 3HNA under the excitation of 350 nm shows a strong emission peak at 511 nm, which is green light emission. The green band emission may be attributed to the formation of the cyclic dimer as illustrated in Figure 5, which undergoes ESIPT. 19
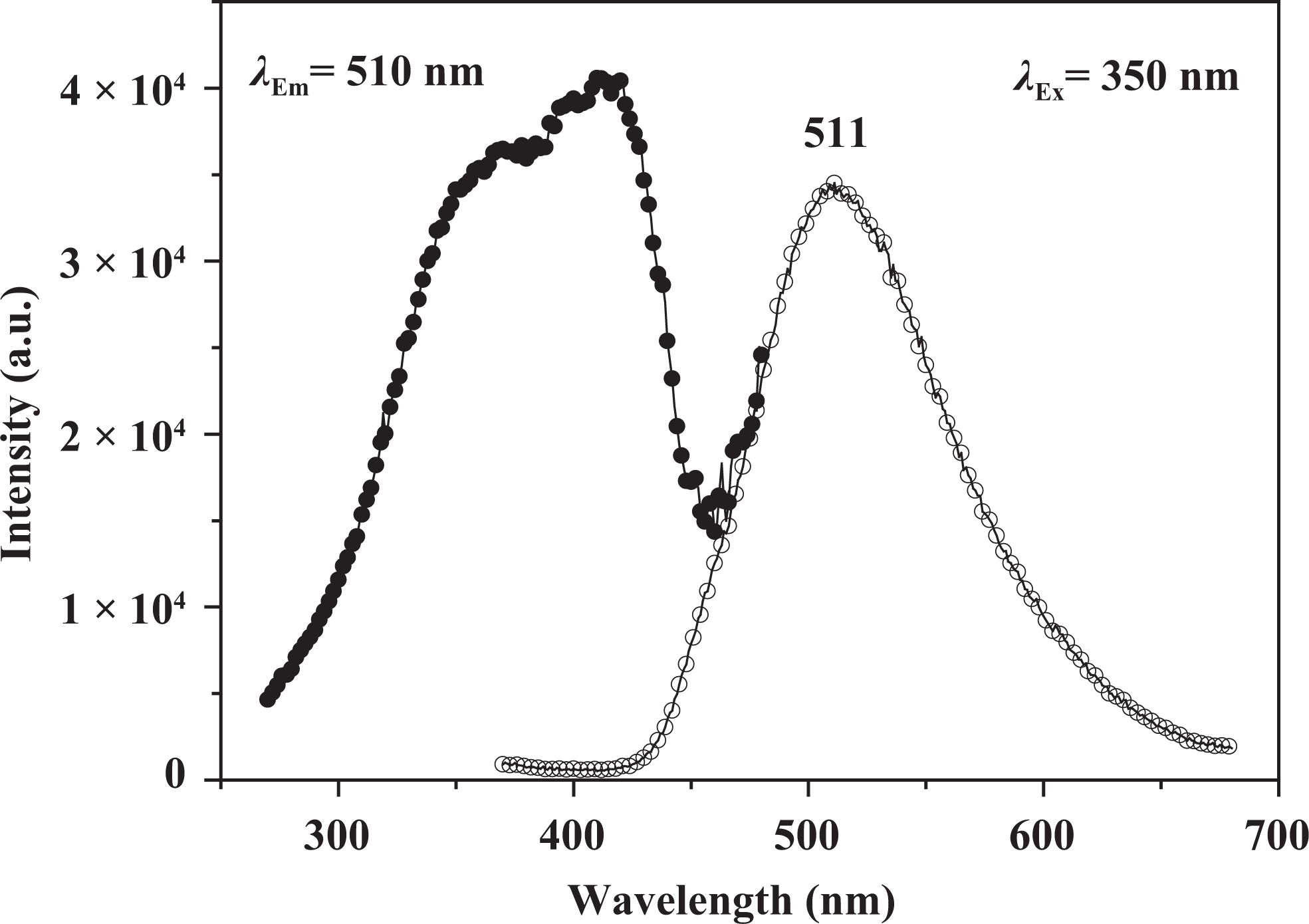
Fluorescence excitation and emission spectra of 3HNA. 3HNA: 3-hydroxy-2-naphthoic acid.
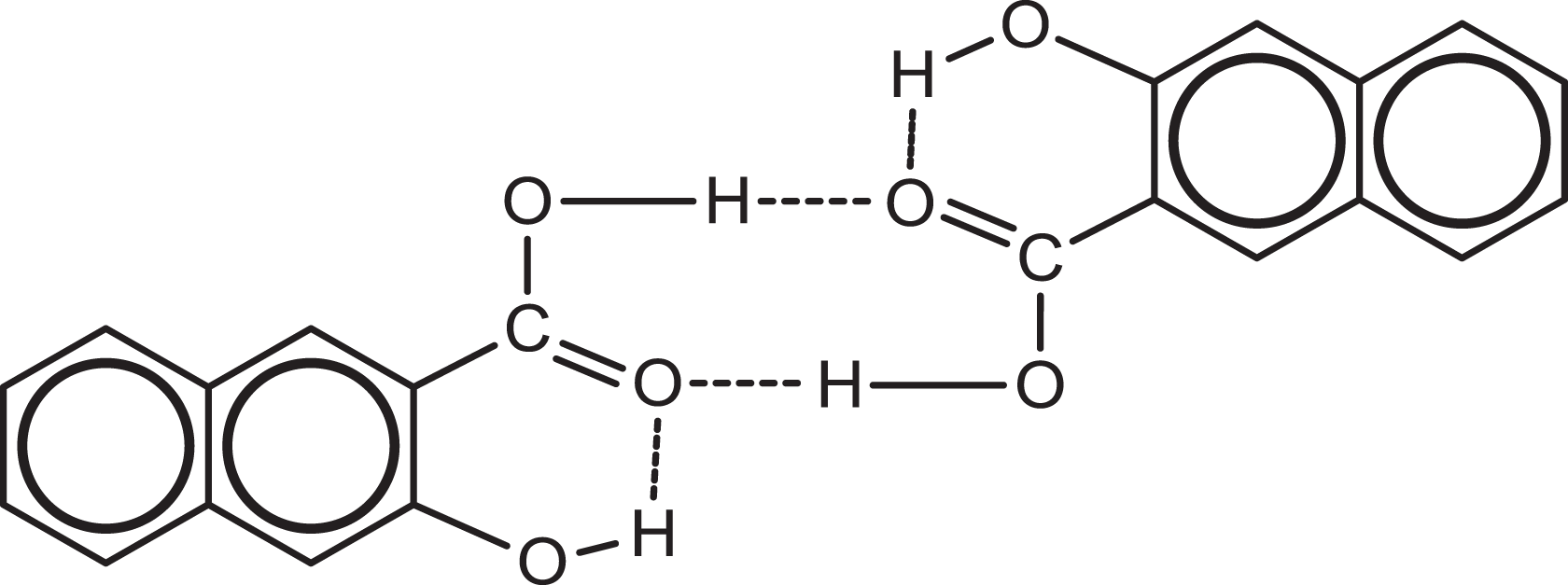
3HNA dimer of P form. 3HNA: 3-hydroxy-2-naphthoic acid.
The emission spectra of 3HNA and the PMMA composites doped with increasing concentration of 3HNA in solid state at room temperature are reproduced in Figure 6. The spectrum (curve 1) of 3HNA displays a strong emission peak at 511 nm. In comparison with the 3HNA, the emission spectra of the 3HNA-doped PMMA composites show 3HNA dual emission. One is the violet emission peak at about 420 nm, while the other is green emission peak at about 520 nm. The violet emission peak is assigned to the normal 1 (π → π*) transition from the naphthalene unit of 3HNA in its K form and the green emission peak is attributed to a 1 (π → π*) transition from an E tautomer generated by the ESIPT from hydroxyl groups to the carbonyl oxygen of the 3HNA as shown in Figure 7. 18,19 As shown in Figure 6, with the increase in the concentration of 3HNA from 0.5% to 2.0%, the intensity of the violet band decreases, while the green band changes little in intensity. The violet band emission peak shifts from 413 nm to 426 nm. The results indicate that the emission characteristic of 3HNA dominates the emission wavelength of the 3HNA-doped PMMA composites without DMF. The emission intensity of the composites is also sensitive to the concentration of 3HNA.
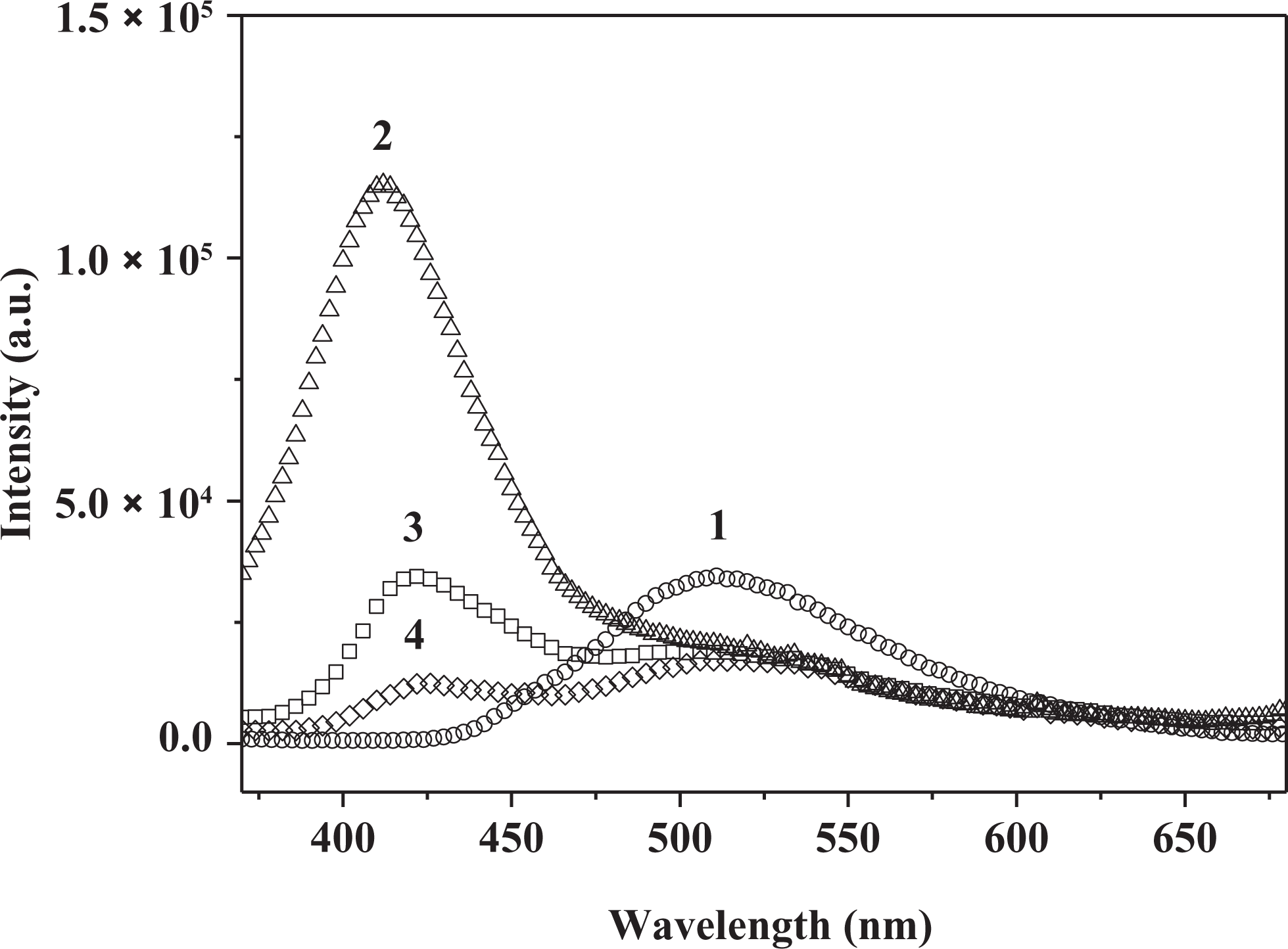
The fluorescence emission spectra of 3HNA (1) and the PMMA composites without DMF with increasing concentration of 3HNA: (2) 0.5 wt%, (3) 1.0 wt%, (4) 2.0 wt% 3HNA, respectively (λex = 350 nm). 3HNA: 3-hydroxy-2-naphthoic acid; PMMA: poly(methyl methacrylate); DMF: dimethylformamide.
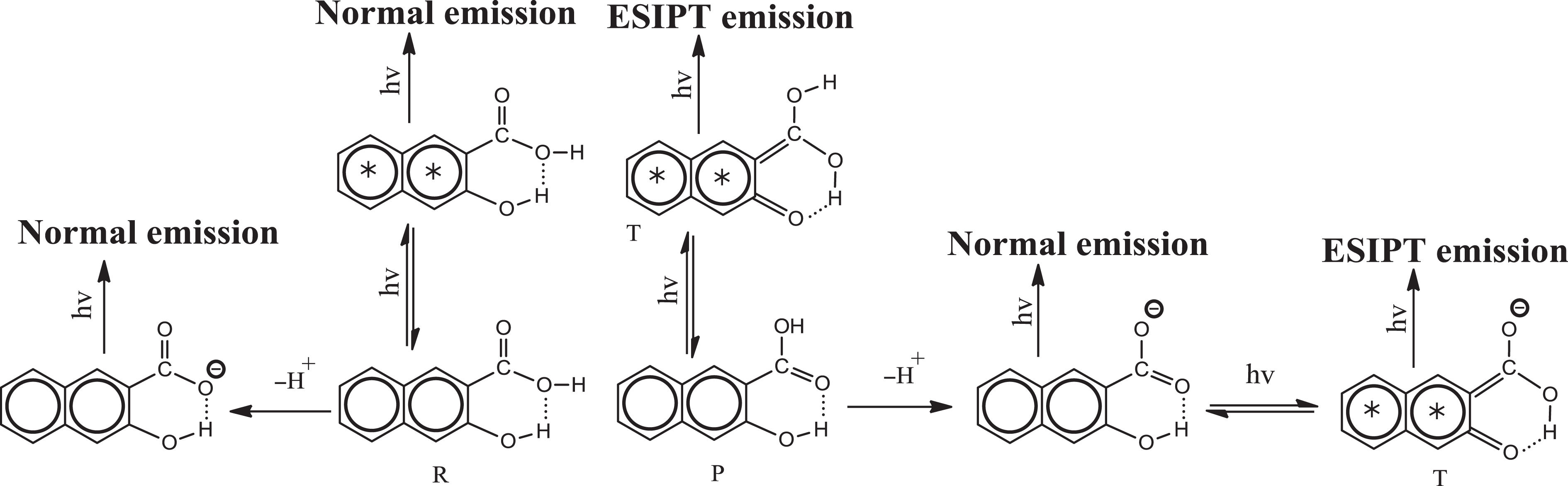
Various emitting species of 3HNA. 3HNA: 3-hydroxy-2-naphthoic acid.
As shown in Figure 8, when the polar solvent DMF is added to the 3HNA-doped PMMA composites, the PMMA composites also exhibit dual emission peaks. When 0.5 wt% DMF is added, the intensity of violet band and green band emission decreases compared with the composites without DMF. At the same time, with increasing concentration of DMF, the intensity of green band is remarkably enhanced with the intensity of violet band keeping almost unchanged. The results indicate that DMF acts as an efficient weak base and that the formation of strong intermolecular hydrogen bond between DMF and the carboxylic group proton, which increases the strength of the IMHB, is expected to lower the transfer potential energy, thus facilitating ESIPT, as shown in Figure 9. 19
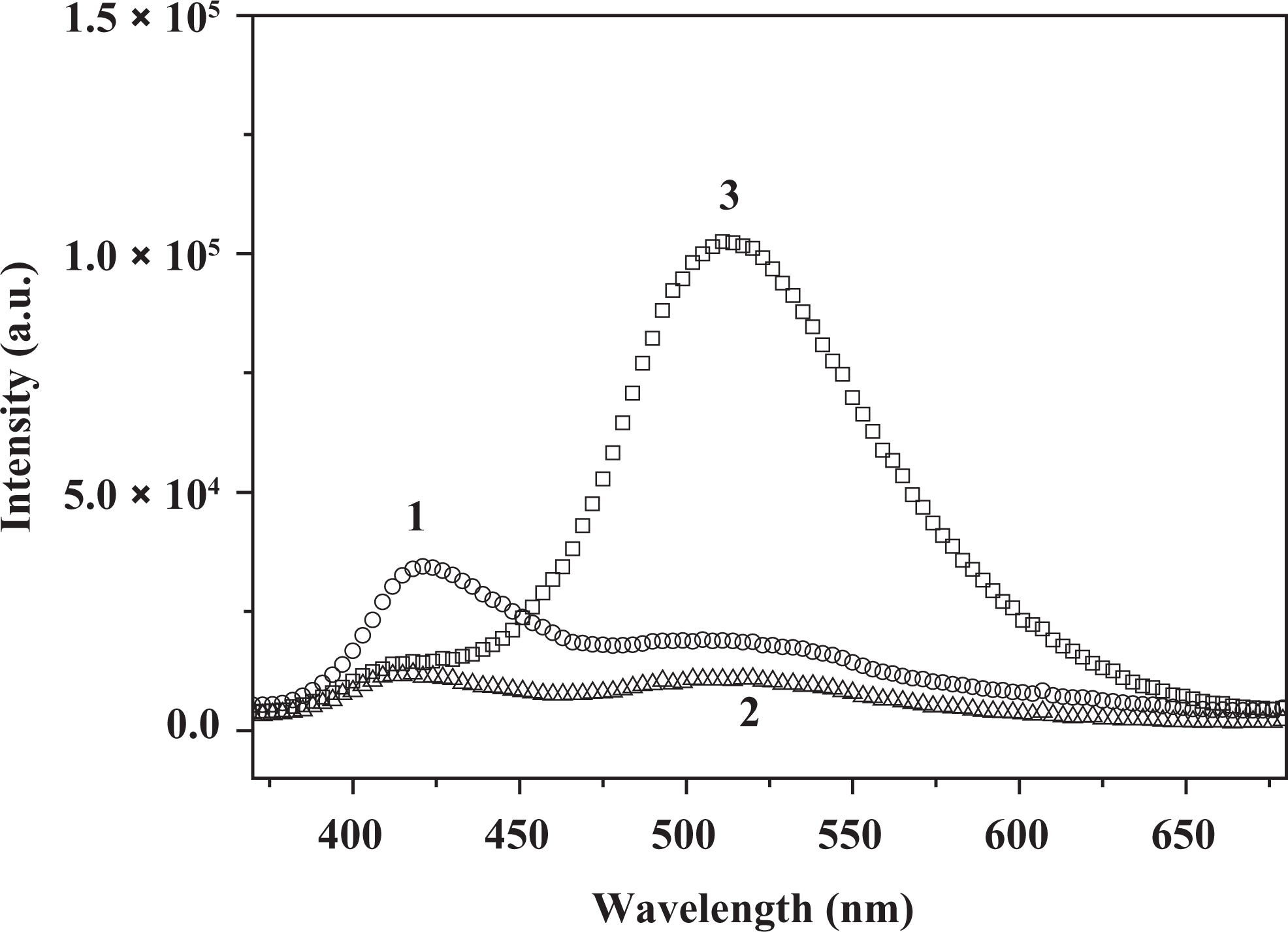
The fluorescence emission spectra of the 1.0 wt% 3HNA-doped PMMA composites with increasing amount of DMF: (1) 0 wt%, (2) 0.5 wt%, (3) 3.0 wt%, respectively (λex = 350 nm). 3HNA: 3-hydroxy-2-naphthoic acid; PMMA: poly(methyl methacrylate); DMF: dimethylformamide.
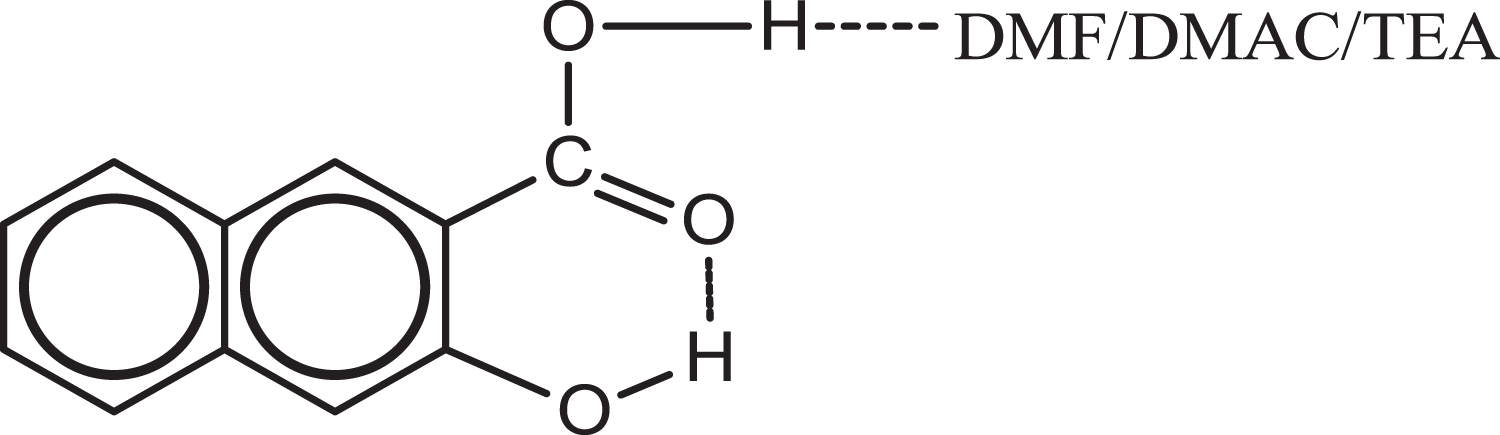
Hydrogen-bonded complex of 3HNA with DMF/DMAC/TEA. DMF: dimethylformamide; DMAC: N, N-dimethylacetamide; TEA: triethylamine.
The influence of different solvents on the green emission intensity of the 3HNA-doped PMMA composites was investigated. As shown in Figure 10, the green emission intensity of the PMMA composites increases in the presence of DMF/DMAC/TEA when compared with the PMMA composites without solvent. When the concentration of 3HNA is equal to 0.1 wt%, DMF and DMAC have almost the same effect on enhancing the green emission intensity of the PMMA composites, whereas TEA remarkably enhances the green emission intensity. When the concentration of 3HNA is equal to 0.5 wt%, the enhancement effect of the PMMA composites with different solvents is TEA > DMF > DMAC. Meanwhile, only 3.0 wt% TEA in the PMMA composite has obviously enhanced the green emission intensity of the system. We propose that the formation of the strong intermolecular hydrogen bond between the TEA and the carboxylic group proton of 3HNA facilitates ESIPT, which is similar to that of DMF and DMAC as shown in Figure 9 19 and the intermolecular hydrogen bonding ability of TEA is better than that of DMF and DMAC. As a result, the effect of TEA is more efficient than that of DMF and DMAC.
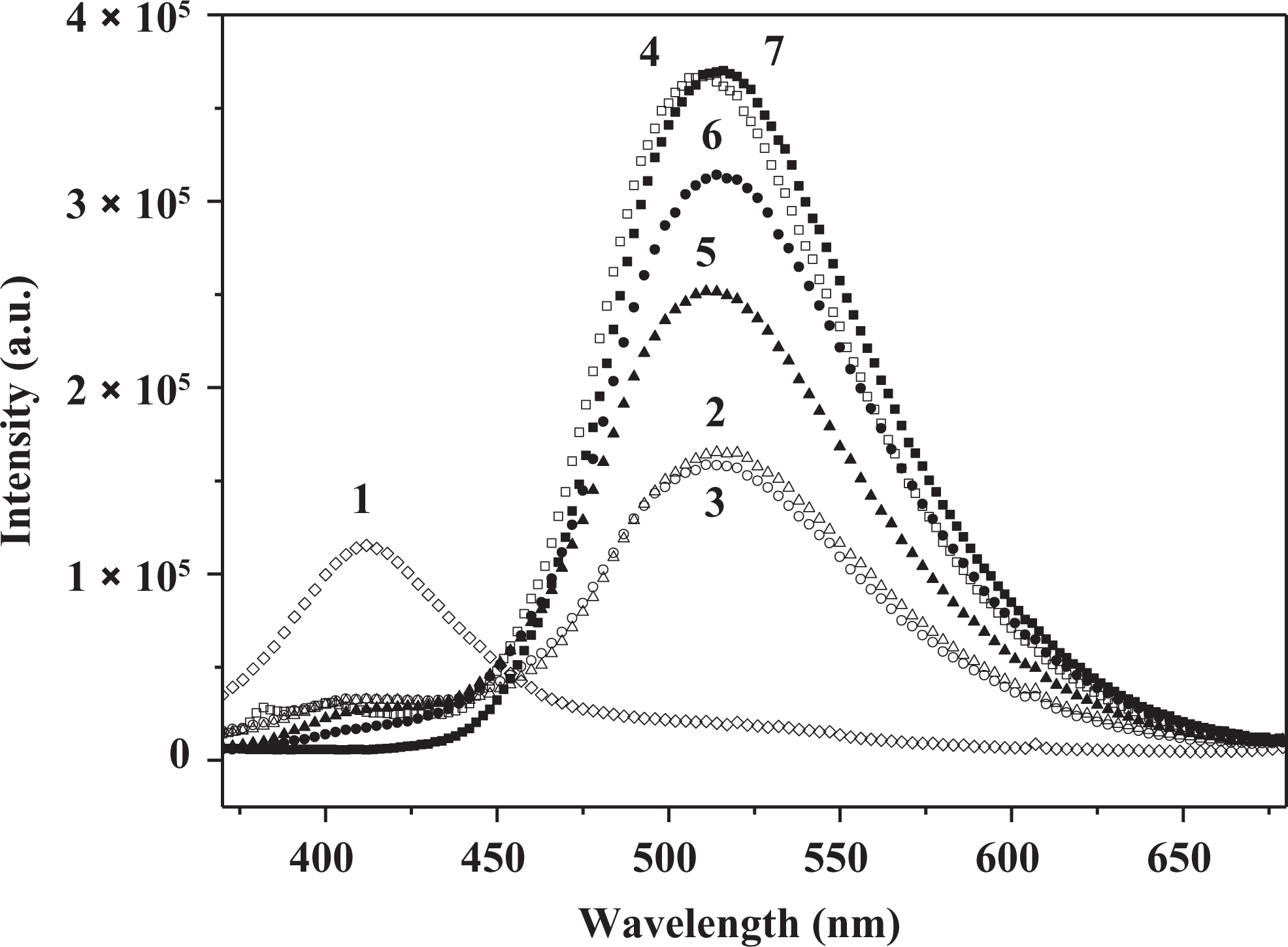
The fluorescence emission spectra of the 3HNA-doped PMMA composites with different amount of solvents: (1) none, (2) 15 wt% DMAC, (3) 15 wt% DMF, (4) 15 wt% TEA, (5) 15 wt% DMAC, (6) 15 wt% DMF, (7) 3.0 wt% TEA, respectively, and in each case concentration of 3HNA is 0.5 wt%, 0.1 wt%, 0.1 wt%, 0.1 wt%, 0.5 wt%, 0.5 wt%, 0.5 wt%, respectively (λex = 350 nm). 3HNA: 3-hydroxy-2-naphthoic acid; PMMA: poly(methyl methacrylate); DMAC: N, N-dimethylacetamide; DMF: dimethylformamide; TEA: triethylamine.
Effect of various solvents on the photoluminescence of the composites
In order to know the effect of various solvents on the photoluminescence of the composites, the 1.0 wt% 3HNA-doped PMMA composites were synthesized by in situ polymerization in the presence of 10 wt% solvents. According to the different polarities, organic solvents (such as dimethyl sulfoxide (DMSO), DMF, DMAC, TEA, 12 tertiary amines, methylene chloride, pyridine, chloroform, tetrahydrofuran, ethyl acetate, toluene, or n-hexane) were added to the 3HNA-MMA polymerization system, respectively. The effects of all kinds of organic solvents on the fluorescence of the composites have been observed with a UV lamp at 365 nm or 254 nm wavelength.
Table 1 shows the results of the light-emitting performance of the PMMA composites. The composites emit green light when strong polar solvents or strong hydrogen bonding regents like pyridine, DMAC, DMF, TEA, 12 tertiary amines, and DMSO are added. At the same time, violet light is observed by adding other solvents. On the exterior, the composites by adding solvents such as methylene chloride, DMSO, TEA, and 12 tertiary amines are white opaque or translucent and the composite by adding pyridine is brown transparent. Whereas the composites by adding solvents such as DMF and DMAC are light color transparent and emit green light, which is the light-emitting color of 3HNA.
The appearance and photoluminescence of the polymer composites.

DMAC: N, N-dimethylacetamide; DMF: dimethylformamide; DMSO: dimethyl sulfoxide; TEA: triethylamine.
Conclusions
In this article, the fluorescence properties of 3HNA-doped PMMA composites were systematically studied. The PMMA composites exhibited violet light emission and green light emission in the presence of nonpolar or weak polar solvents and strong polar solvents or strong hydrogen bonding regents, respectively. The presence of DMF in the composite systems not only obviously enhanced absorption in the longer wavelength and the lower energy region but also increased the ESIPT emission intensity along with the decreased violet emission intensity. Besides, the emission intensity of the PMMA composites was sensitive to the concentration of 3HNA, solvent polarity, and the concentration of solvents. In the meantime, FTIR spectra showed that the hydrogen bond between 3HNA and DMF might be formed in the composite systems in the presence of DMF, which was in accordance with the result that the composites exhibited the ESIPT emission of 3HNA.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Fujian Province of China (no. 2012J01031), Educational Commission of Fujian Province of China (no. JK2012011), and National Natural Science Foundation of China (nos 61250016, 60976019, and 51003016).
