Abstract
This study tended to modify polyvinylidene fluoride (PVDF) microfiltration membranes by the addition of nanomagnesium oxide (nano-MgO) particles via fabrication of the phase inversion method. The detailed structure and properties of these composite membranes were characterized. The findings showed that the membrane hydrophilicity was significantly improved with the addition of nano-MgO content increasing from 0% to 1%. The improved hydrophilicity and decrease in roughness of the composite membrane enhanced the antifouling performance during the reclaimed water treatment.
Introduction
Currently, much effort is being devoted to improve the performance of the existing membranes in terms of antifouling properties, high mechanical strength, and good chemical resistance. The emergence of new membrane processes such as membrane contactor, membrane distillation, or membrane reactor has also received great attention from researchers in the recent years. These developments have been motivated by the needs in the industrial sectors to reduce the overall operational costs compared with the conventional separation processes as well as broaden the membrane applications. 1,2 Poly(vinylidene fluoride) (PVDF) has received great attention as a membrane material with regard to its outstanding properties such as high mechanical strength, thermal stability, chemical resistance, and high hydrophobicity compared with other commercialized polymeric materials. PVDF membranes have been extensively applied in ultrafiltration and microfiltration for general separation purposes and are currently being explored as potential candidates in the applications of membrane contactor and membrane distillation. 3–8
PVDF is also advantageous over other membrane materials due to its high mechanical strength and excellent chemical resistance, in which these properties make it suitable for wastewater treatment. By possessing low level of extractables, PVDF can be considered as a pure polymer, which makes it a suitable candidate in biomedical and bioseparation applications. Unlike other crystalline polymers, PVDF exhibits thermodynamic compatibility with other polymers such as poly(methyl methacrylate) (PMMA) over a wide range of blend compositions, 9,10 where this characteristic can be useful in the fabrication of membrane with the desired properties. PVDF can be further chemically modified to obtain some specific functions 11,12 and can be cross-linked when subjected to electron beam radiation or γ radiation. 13
Nanomagnesium oxide (nano-MgO) is a versatile material that has been widely used in various areas, such as catalysis, adsorption, oxidation, and sterilization. Notably, nano-MgO exhibits good bactericidal performance in aqueous environments due to the formation of superoxide anions on its surface. Compared with other kinds of solid bactericides, nano-MgO has the advantages of being prepared from readily available and economical precursors. 14 However, it leads to high pH (more than 10) due to hydrolysis of formed Mg(OH)2. Obviously, other antibacterial factors of nano-MgO besides alkaline effect are still important. 15
In this study, nano-MgO-filled PVDF membrane was prepared and the performance of the membrane was investigated.
Experimental
Materials and specimens
PVDF was purchased from Shanghai 3F New Materials Co. Ltd (Shanghai, China). Blood serum albumin was from Sinopharm Chemical Reagent Co. Ltd (Shanghai, China). Nano-MgO particles were obtained from Star Nano Scientific and Technological development Co. Ltd (Henan, China). Distilled water was used as the nonsolvent for polymer precipitation. The chemical and physical properties of nano-MgO particles are given in Table 1.
Properties of nanomaterials.
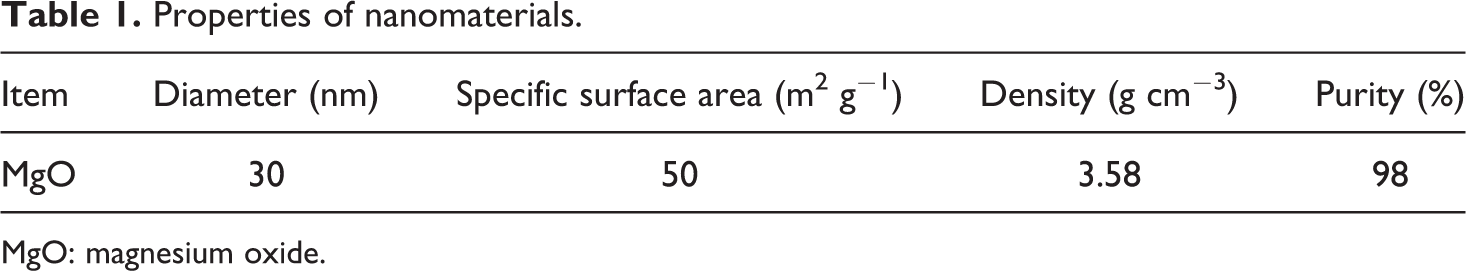
MgO: magnesium oxide.
PVDF-MgO composite membranes were prepared by the phase inversion method. Casting dopes were prepared by dissolving the PVDF and Nano-MgO particles in the dimethylacetamide at 323.1 K, and the dopes were stirred for 24 h. Then, the casting dopes were maintained in the dark for at least 24 h for removal of air bubbles.
The casting solution containing different amount of Nano-MgO particles was cast uniformly on a glass plate with 2.00 × 10−4 m of casting knife. After exposure to air for 20 s, the glass plate was immersed in a bath filled with distilled water. The membranes were then soaked in distilled water.
Results and discussion
Hydrophilicity
The surface hydrophilicity of membranes could affect the flux and antifouling ability of membranes. In general, the hydrophilicity could be evaluated by water contact angle, and higher hydrophilicity by smaller contact angle. As shown in Figure 1, when the nano-MgO content of dopes increased from 0% to 1%, the contact angle decreased from 82.33° to 70.06°.
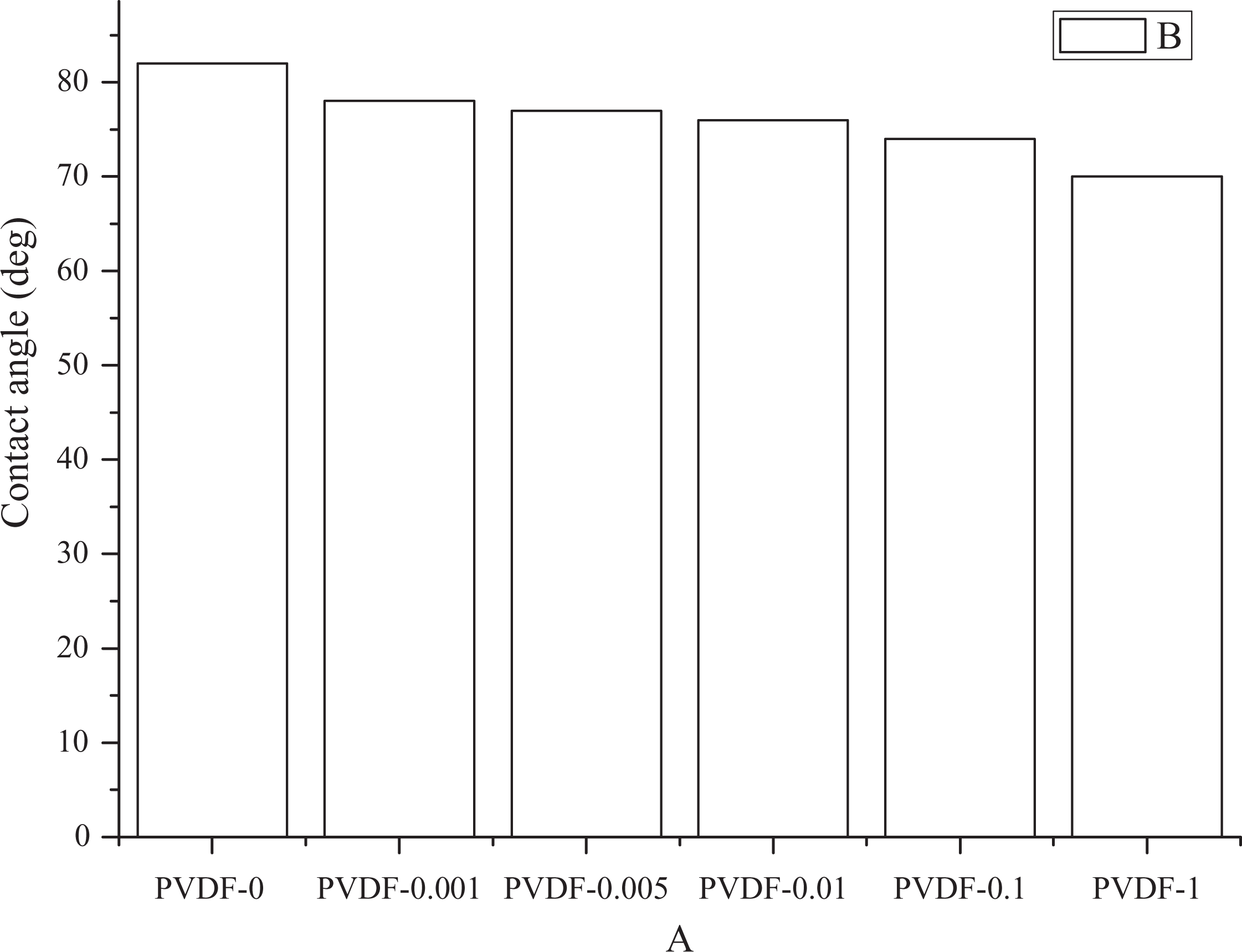
Contact angle of membranes.
The surface hydrophilicity could be improved with the MgO amount in membrane; however, the contact angle of composite membranes increased when MgO concentration was higher than 3% due to the formation of large-scale network. Since nano-MgO has a higher affinity to water than PVDF, the surface hydrophilicity was significantly improved with the increased amount of nano-MgO content. Furthermore, excessive addition of nano-MgO (PVDF-1) improved the hydrophilicity slightly rather than the increase of contact angle when MgO concentration was higher than 3%.
Scanning electron microscopy
In order to investigate the effect of nano-MgO particles on the microstructure of the membranes, cross sections of PVDF membranes by scanning electron microscopy (SEM) analysis have been obtained. As depicted in Figure 2, all membranes exhibited the typical asymmetric cross-sectional structure. The upper layer of cross sections had a finger-like structure and the sublayer owned a porous structure. Apparently, it could be seen that the finger-like cavities became larger as the nano-MgO content increased from 0% to 0.005%. Formation and growth of macrovoids was related to the kinetics of phase inversion. Instantaneous liquid–liquid demising is thought to provide conditions for macrovoid formation. Since nano-MgO has higher affinity to water than PVDF, penetration velocity of water into nascent membrane increased with nano-MgO content during the phase inversion. In addition, the solvent diffusion velocity from membrane to water could also be increased by adding nano-MgO. As known, pore size and porosity increased with solvent interdiffusion velocity. The increase in water–solvent interdiffusion velocity caused the formation of larger cavities. However, the finger-like cavities became smaller with the nano-MgO content increasing from 0.01% to 1%. It has been shown that increasing viscosity may work as a void-suppressing factor, as it slows the exchange rate of solvent/nonsolvent, and thus shifting the path of phase inversion from instantaneous into delayed liquid–liquid demising. Thus, the increase in the dope viscosity results in the suppression of macrovoids.
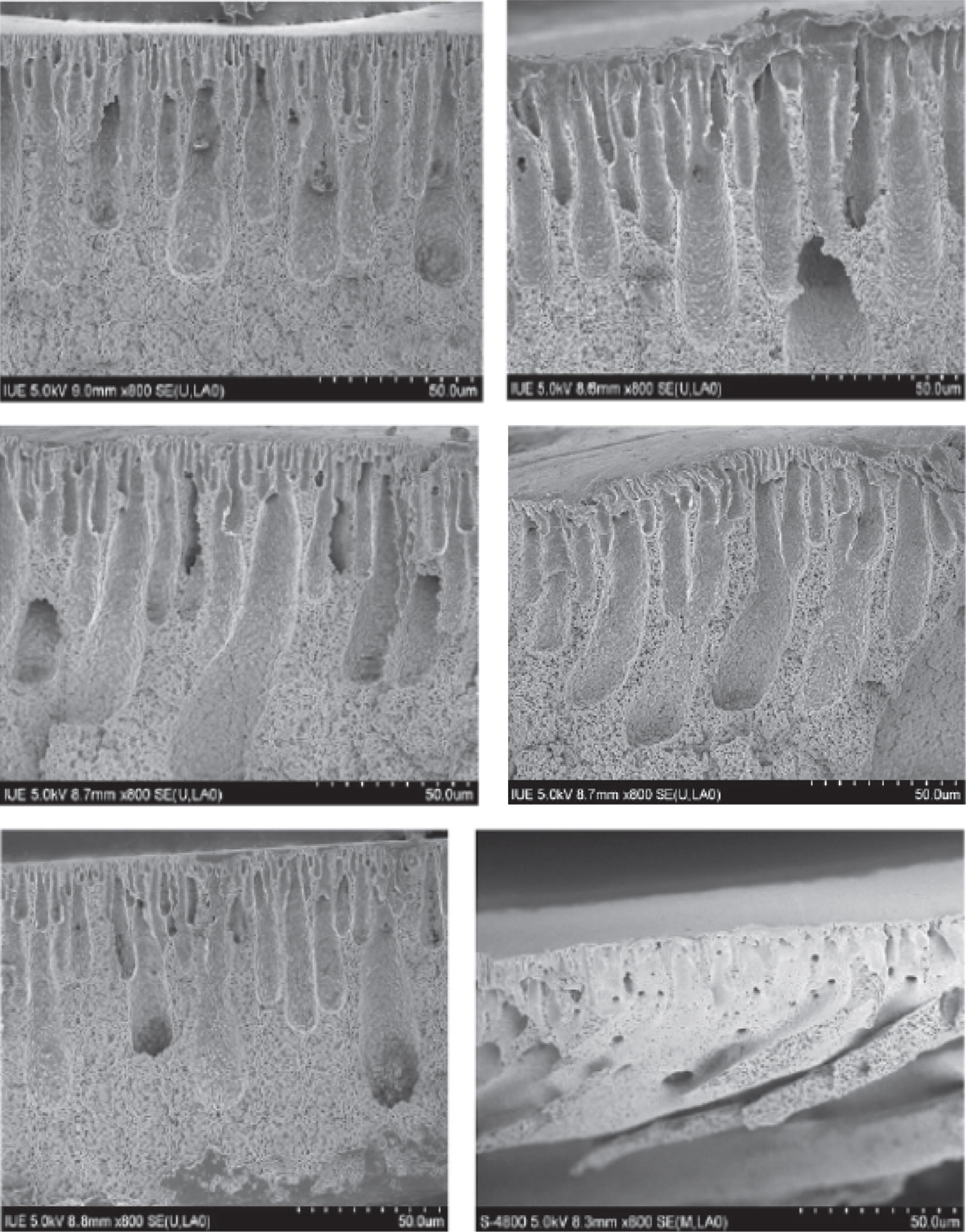
SEM images of PVDF membranes cross-sections. SEM: scanning electron microscopic; PVDF: polyvinylidene fluoride.
The lower amount of added nano-MgO particles (≤ 0.005 wt%) resulted in the formation of larger surface pores compared with PVDF membrane (PVDF-0). This phenomenon could be interpreted as follows: The precipitation rate increased with increasing nano-MgO content from PVDF-0 to PVDF-0.005, which favored the formation of a large porous structure. However, higher amount of nano-MgO particles increased the dope viscosity, thereby slowing down the precipitation rate and leading to the formation of a smaller porous structure.
When the addition amount of nano-MgO particles was 1 wt%, the pore morphologies on the top surface came into an uneven narrow gap, which may be due to the excessive amount of nano-MgO restricting the cross-linking of PVDF molecules.
Mechanical properties
The tensile strength initially increased with the addition of nano-MgO and they reached a maximum value (2.92 × 106 Pa) when the nano-MgO content was 0.01% and then declined as the nano-MgO content was gradually increased. Moreover, the elongation at break value was initially increased from 35.7% to 210.6% when the nano-MgO content increased from 0% to 0.01% and then declined as the nano-MgO content was further increased. As seen in Figure 3 for the magnification of sublayer membranes with different nano-MgO content, all membranes had a sponge-like cross section; however, they presented some differences in compact morphology. For the pure PVDF membrane, cellular pores formed in the cross-section region. When the nano-MgO was 0.01 wt%, the cross section gradually became a little denser. That is, there were interactions between nano-MgO particle and PVDF. Nano-MgO particle could act as a cross-linking point in composite membranes to associate with the polymeric chains and increase the rigidity of polymeric chains. That is, more energy would be needed to break down the bond between nano-MgO and PVDF. Due to this, the mechanical strength of composite membranes was augmented. While the nano-MgO content increased to 1%, the massive amount of nano-MgO particle restricted the space for free movement of PVDF chain, resulting in the decrease of mechanical properties. Apparently, these indicated that supplementation of nano-MgO could improve the mechanical properties of the membrane. However, excessive supplementation of nano-MgO could cause the decline of membrane strengths and its elasticity, thereby leading to decrease in the tensile strength of membranes and elongation at break value.
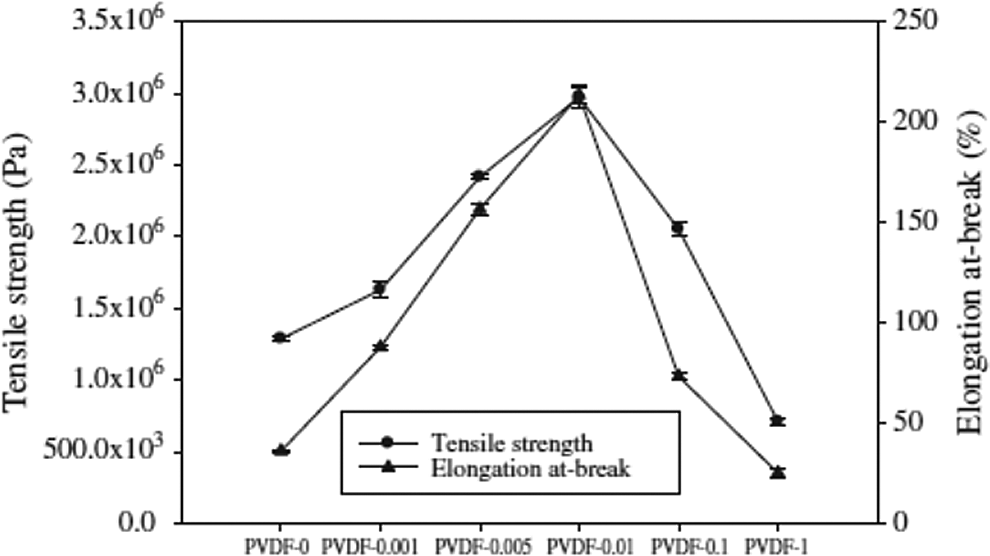
Mechanical properties of membranes.
Differential scanning calorimetry
As shown in Figure 4, the melting temperature of membranes increased from 437.85 K to 439.55 K when the addition of nano-MgO content increased from 0% to 0.1%. The enthalpy of fusion increased from 2.56 × 103 J kg−1 to 2.75 × 103 J kg−1 when the addition of nano-MgO content increased from 0% to 0.01%, then decreased from 2.75 × 103 J kg−1 to 2.60 103 J kg−1 when nano-MgO content increased from 0.01% to 1%. This revealed that nano-MgO significantly influenced upon the thermal stabilities. The presence of nano-MgO in the membrane-forming materials strengthens the network of the composite membranes, thereby leading to a parallel enhancement on the thermal stabilities.
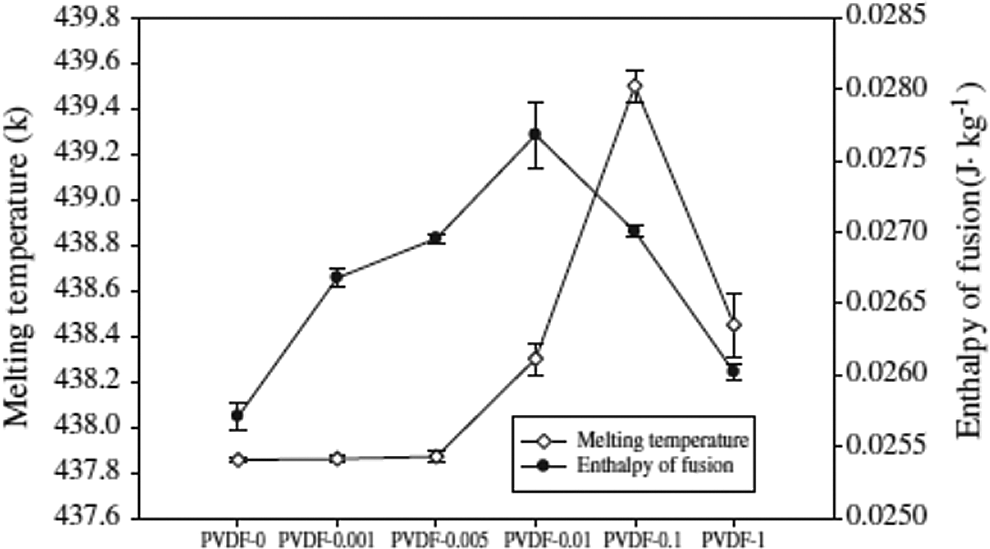
Effects of MgO weight fraction on melting temperature and enthalpy of fusion of membranes. MgO: magnesium oxide.
EDAX analysis
To investigate the mechanism predicted for magnesium (Mg) insertion into the C–S–H nanostructure, energy dispersive X-ray analysis (EDAX) of a composites containing nano-MgO were conducted. The SEM images (back-scattered mode) and typical EDAX analysis for C–S–H in composites containing nano-MgO is presented in Figure 5(a) and (b). Although the formation of M–S–H cannot be concluded from EDAX analysis due to the simultaneous presence of Mg, calcium (Ca) and silicon (Si), the insertion of Mg into C–S–H by analyzing the C–S–H structure was investigated. Based on the test results, the gray areas, which present the mean MgO/calcium oxide ratios to be 0.49 (Zones 1–3 in Figure 5), can be considered modified C–S–H incorporated with Mg.
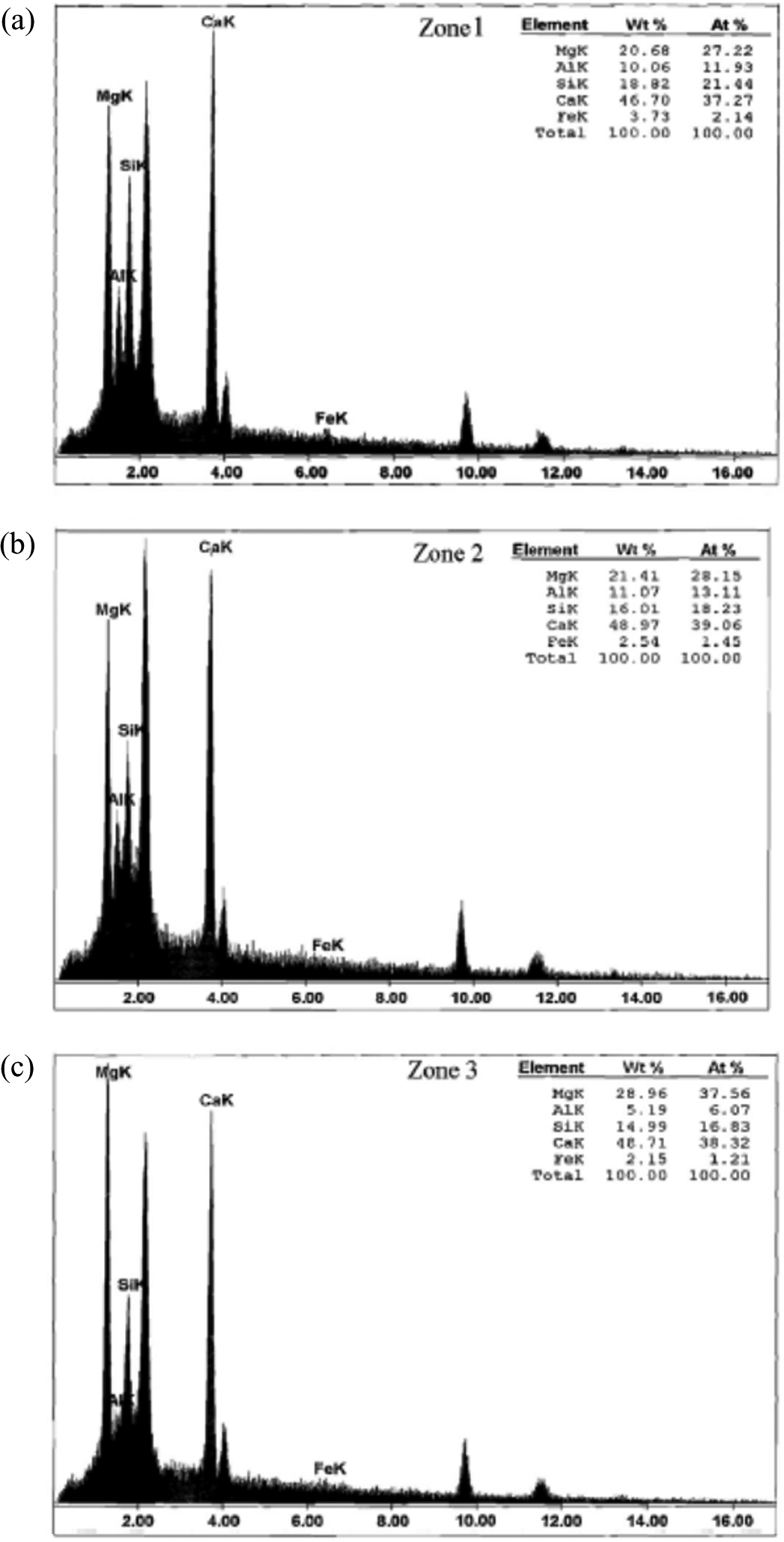
Typical EDAX results for the C–S–H in composites containing nano-MgO. EDAX: energy dispersive X-ray; MgO: magnesium oxide.
In Figure 6, which present the mid-infrared (IR) spectra for plain and MgO composites, the bands at 710 and 910 cm−1 can be attributed to Si–O–Si bending vibrations and Si–O stretching vibrations, respectively, and the vibration band appearing at about 970 cm−1 corresponds to C–S–H. In both the plain and nano-MgO-containing samples, a small shoulder at about 1100cm−1 corresponding to silica gel and a vibration band at about 1605 cm−1 corresponding to water (H2O) bending appear. There are also stretching vibration bands identified at about 3440 and 3650 cm−1, which can be attributed to O–H groups in H2O or hydroxyl. In this research, the mid-IR spectrum for the composites containing nano-MgO displays a sharp stretching located in the range of deformation of Si–O stretching vibrations (902 cm−1), which is slightly shifted to higher frequency, and a large shoulder in the range of Si–O–Si bending vibrations at approximately 710 cm−1, which is not observed in the plain sample. Furthermore, for the composites containing nano-MgO, a small shoulder at approximately 520 cm−1 is observed that can be attributed to the deformation of Si–O tetrahedron reported at 400–500 cm−1, which is shifted slightly to higher frequency. This shift can be the evidence of the possibility of Mg insertion into the structure of C–S–H, and based on the interaction mechanism of Mg in cement hydration and the proposed pattern for Mg2+ ions insertion into the dreierketten pattern of the calcium silicate hydrate gel, the two following possibilities for Mg insertion can be considered. The first possibility is Mg insertion into the gap of Q2p tetrahedral, which may modify the TiO2-SiO2 composite oxide (TO4) deformation vibration of the Si(Q2) tetrahedron and cause the shoulder that is attributed to the deformation of SiO4 tetrahedra shift to higher frequency, and the second possibility is the incorporation of MgO tetrahedra into the gap of the Q2p silicon deformation, which is equilibrated by Ca.
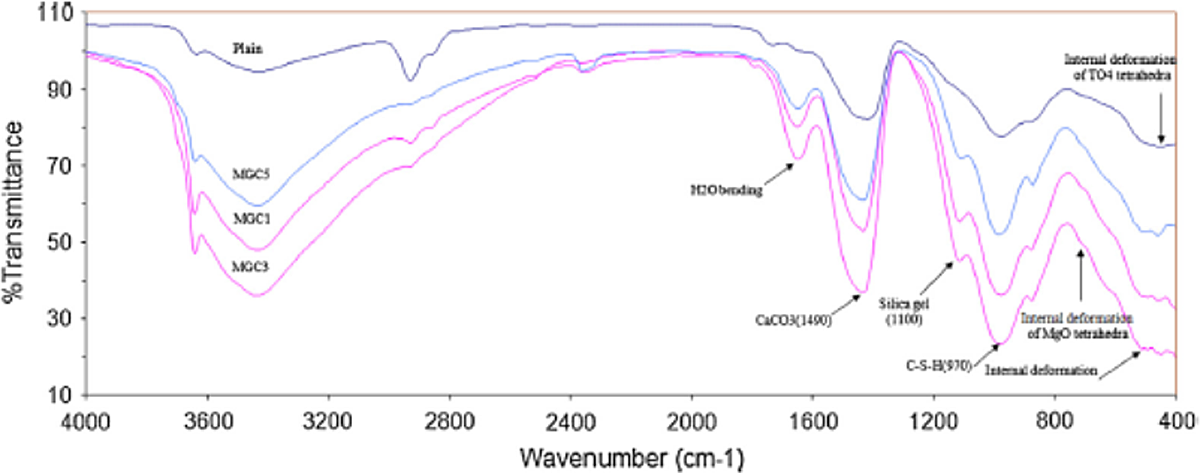
Mid-IR absorption spectrum (in per centimeter) of the plain and composites containing nano-MgO. IR: infrared; MgO: magnesium oxide.
The mid-IR results for the sample containing nano-MgO show better resolution and stretched O–Si–O and Si–O–Si bands compared with those of plain cement samples. This stretching band and large shoulder for the MgO-composite confirm the improved crystallinity of C–S–H in the sample containing nano-MgO with respect to plain sample and prove that the degree of crystallinity, the silicate chain length, and the extent of condensation of the silicate change across the interlayer (cross-linkage) for C–S–H in MgO-composites are all significantly higher in comparison with C–S–H in plain composites. These results show that the addition of a limited amount of MgO into cement-based composites can give rise to improved mechanical properties due to the incorporation of limited amounts of Mg2+ ions into the C–S–H nanostructure and the modification of the C–S–H nanostructure.
This nanostructure modification can be considered one of the reasons for the mechanical strength variation in addition to the known mechanisms of nanoparticles in the improvement of the mechanical properties in cement-based composites.
Conclusions
The present study reveals the performance of nano-sized MgO on PVDF microfiltration membranes. Experimental results indicated that the morphology of surface and cross section were controlled by the difference content of nano-MgO particles. The increase in water–solvent interdiffusion velocity caused the formation of larger pore size when small amounts of MgOs were introduced.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was sponsored by the National Natural Science Foundation of China (No. 31300783, No. 51202144, No. 71201099), Doctoral Fund of the Ministry of Education Jointly Funded Project (No. 20123121120004), the 678 Journal of Thermoplastic Composite Materials 29(5) Shanghai Top Academic Discipline Project-Management Science & Engineering, the Natural Science Foundation of Shanghai (No. 08XD1401900, No. 13ZR1419200), NSFTJ (No.11JCZDJC16900), 973 Program (No.2011CB711000), the Shanghai Municipal Education Commission Project (No.13YZ080), the Ministry of Transport Research Projects (No.2012-329-810-180), the Shanghai Maritime University Research Project (No. 20130474), and Shanghai College Young Teacher Training Program (shhs008), the High-Tech Research and Development Program of China (No. 2013A2041106).
