Abstract
Nanocomposites (NCs) of poly(vinyl alcohol) (PVA) with chiral-modified magnesium–aluminum-layered double hydroxides (LDHs) were prepared by solution intercalation method. A novel chiral organomodified LDH was synthesized from the coprecipitation reaction of the aluminum (III) nitrate nonahydrate, magnesium (II) nitrate hexahydrate, and bioactive N-trimellitylimido-
Keywords
Introduction
The interest in nanomaterials has been growing at an extraordinary speed owing to its potential in diverse scientific and technological areas. 1,2 Nanoscience and nanotechnology basically involve developing processes and products on a scale which varies between 1 and 100 nm that may potentially benefit all areas, including biotechnology, electronics, food and nutrition industries, pharmaceuticals, agriculture, biomedical sectors, and several others. 3 –5 Nanocomposites (NCs) are a combination of two or more components in the nanometric dimension presenting an exceptional association of physicochemical, mechanical, and biochemical properties because of their size reduction and large interfacial area offering an enormous opportunity for scientists and materials engineers in different segments of science and technology. 6 –9 NCs based on polymers are among the most promising composite systems. 10
Polymers with broad range of characteristics play an important role in our everyday life. 11 However, with amplified use of polymeric products, there are increased concerns on the impact these polymers have on the environment. These concerns have forced the development of environmentally friendly, sustainable, and biodegradable polymers and composites in recent years. 11 –13 Along with the existing synthetic polymeric materials, poly(vinyl alcohol) (PVA) has been intensively researched in a myriad of studies ranging from conventional to advanced prospective applications. 14 PVA as a thermoplastic and biocompatible petroleum-based polymer is a water-soluble polymer with many hydroxyl groups pendant in the side chains. It has been studied intensively because it has high hydrophilicity, processability, biocompatibility, good physical and mechanical properties, complete biodegradability, excellent chemical resistance, and a favorable capacity to form a film. These properties have led to its broad industrial use in, for example, medical wrapping membranes, drug delivery, adhesive and thickener materials, filtration applications, and gas barrier applications. 15 –23 However, PVA has relatively low strength and thermal stability for some applications. 24
Among the different nanofillers for reinforcing of polymeric NCs, layered plate-like particles have shown very promising results and achieved an adequate dispersion of the lamellar nanoparticles within the polymer matrix. 25,26 Most work in this area was carried out on polymeric NCs derived from layered phyllosilicates. 27 Hydrotalcite-like compounds, also known as layered double hydroxide (LDH) or anionic clays, are now gaining attention for this purpose, as they display specific advantages, such as purity, low cost, anion exchange, adsorption ability, crystallinity, particle size control, tunable chemical composition in association with layer charge density, and easy fictionalization. 28 –30 Also, they show good biocompatibility because some LDH interlayer compositions are envisioned as reservoir for drug delivery and gene reservoir. 31 –33
The general chemical composition of LDH can be represented as
There are several publications associated with the preparation and properties of PVA-LDH hybrid materials, and the results showed that the final properties of the hybrid materials were improved. 40 –44 However, preparation and characteristic studies of PVA and modified chiral LDH NC materials have not been reported. In this article, we prepared a series of NC materials by effectively dispersing the inorganic chiral diacid-modified LDH in an organic PVA matrix under ultrasonic irradiation via a simple film-casting technique. The properties of the hybrid materials were studied by Fourier transform infrared (FTIR), X-ray diffraction (XRD), thermogravimetric analysis (TGA), field-emission scanning electron microscopy (FESEM), and transmission electron microscopy (TEM) techniques.
Experimental
Materials
PVA (99% hydrolysis, weight-average molecular weight = 72,000 g mol−1), aluminum (III) nitrate nonahydrate (Al(NO3)3·9H2O), magnesium (II) nitrate hexahydrate (Mg(NO3)2·6H2O), trimellitic anhydride (TMA),
Equipments
FTIR spectra were recorded on Jasco-680 (Tokyo, Japan) spectrophotometer with 2 cm−1 resolution. The potassium bromide pellet technique was applied for monitoring changes in the FTIR spectra of the samples in the range of 4000–400 cm−1. The vibrational transition frequencies are reported in wave numbers (in per centimeter).
The interlayer spacing of the organoclays was measured by an XRD (Bruker, D8 Avance, Germany) with copper K α radiation (λ = 0.1542 nm) at 45 kV and 100 mA. The diffraction patterns were collected between 2θ of 1.2° and 70° at a scanning rate of 0.05° min−1. Basal spacing were determined from the position of the d(001) reflection. The scanning speed was 0.02 s−1. The d-spacing of the hybrid materials was analyzed using Bragg’s equation (nλ = 2d sin θ). Where n is an integer, λ is the wavelength, θ is the glancing angle of incidence, and d is the interplanar spacing of the crystal.
TGA was performed on a STA503 TA Instrument (Hullhorst, Germany) under nitrogen atmosphere at a heating rate of 10°C min−1 from ambient temperature to 800°C.
The morphology of the nanostructure materials was examined by FESEM (S-4160; Hitachi, Tokyo, Japan). The powdered sample was dispersed in water, and then the sediment was dried at room temperature before gold coating.
The nanostructure morphology of the novel materials was also examined by TEM. The TEM images were obtained from a Philips CM120 (Eindhoven, Netherlands) using an accelerator voltage of 100 kV. The inorganic components appear black/gray colored on the micrographs.
The reaction was carried out on an ultrasonic liquid processor (model XL-2000 series; Misonix, Raleigh, North Carolina, USA). The ultrasonic wave frequency was at 2.25 × 104 Hz with a power of 100 W.
Sono-assisted synthesis of LDH-
For the preparation of LDH-
One-step preparation of CLDH
N-Trimellitylimido-
Synthesis of PVA/CLDH NCs
PVA/modified chiral LDH NCs were synthesized by a solution intercalation method using ultrasound energy: At first, distilled water was mixed with different amount of modified LDH (2, 4, and 8%) to form clay/water suspension of ≤2.5% concentration. The suspension was stirred for 3 h at 40°C and sonicated for 1 h. Then, PVA was added to the stirred suspensions to make the total solid concentration w = 5%. The mixture was then heated to 90°C for 6 h to dissolve PVA. The mixed solution was condensed and the total solid concentration controlled at w = 10%. After being sonicated for 1 h, the final films were made via casting on a petridish in a closed oven at 40°C for 24 h.
Results and discussion
Preparation of CLDH and PVA/CLDH NCs
Owing to its layered structure, LDH is an outstanding choice as nanofiller considered for preparation of multifunctional polymer/layered crystal NCs. But, its use as nanofiller is limited by its layers’ high charge density and high content of anionic species and water molecules. To facilitate the intercalation of polymer in the layers of LDH or to achieve a good degree of the layer dispersion in polymer matrices, the interlayer space should be modified with appropriate organic anions with intention of increasing both the interlayer distance and the hydrophobicity of LDH layers. In this investigation, an optically active and biodegradable diacid based on TMA and natural
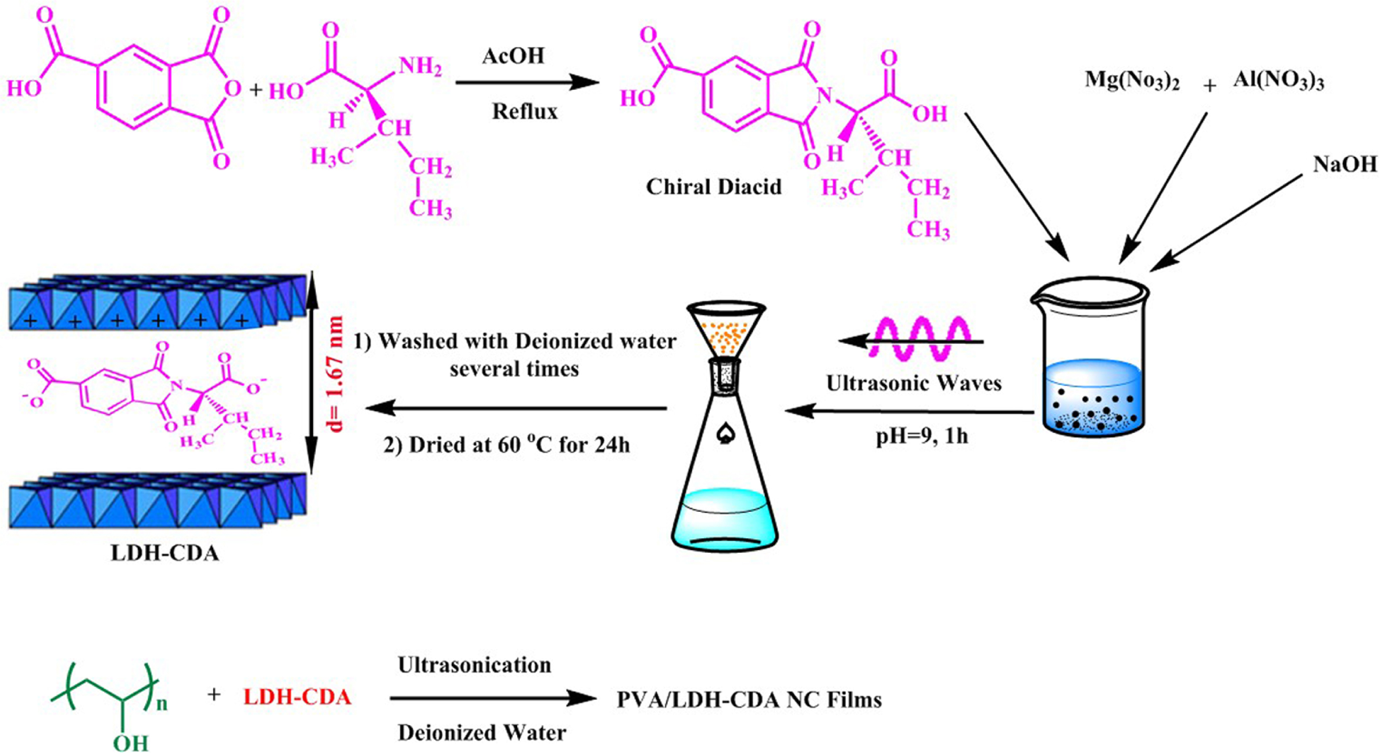
Preparation of chiral diacid. Proposed models for the modification of Mg/Al-LDH and interaction of PVA with CLDH. Mg: magnesium; Al: aluminum; LDH: layered double hydroxide; CLDH: chiral diacid intercalated layered double hydroxide.
Characterizations
XRD pattern
The degree of clay layer separation in the modified LDHs and the clay dispersion in the polymer composite was studied by XRD (Figure 2). Comparison of the XRD patterns of unmodified LDH (Figure 2(a)) containing only carbonate anions with the diacid-immobilized LDH (Figure 2(b)) revealed that diacid anions are successfully intercalated within the LDH layers. In the unmodified carbonate-LDH, the first basal reflection (d 003) appeared at 11.2° corresponding to an interlayer distance of 0.79 nm. In the XRD pattern of CLDH, the d 003 has shifted to 5.28°, corresponding to an interlayer distance of 1.67 nm. This evidence confirms that the chiral diacid is intercalated into the gallery of the LDH crystal structure.
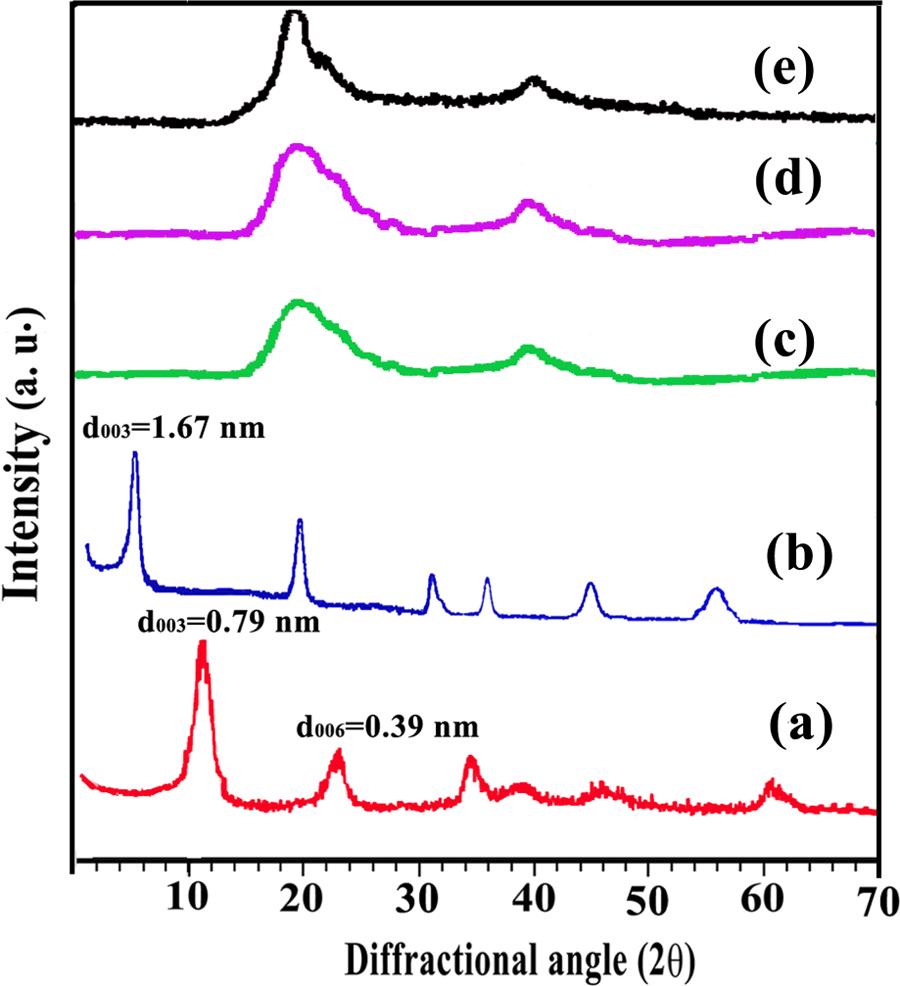
XRD patterns of (a) LDH-
The XRD patterns of the neat PVA, NC4%, and NC8% are also shown in Figure 1. Native PVA is crystallizable, with a typical sharp and narrow peak in its intensity curve with maximum reflection at 2θ = 19° (Figure 1(e)). The XRD patterns of the PVA/modified LDH NCs (4% and 8%) are characterized by the disappearance of the diffraction peaks corresponding to the LDH irrespective of the variation in LDH content (Figure 1(c) and (d)). This complete disappearance of LDH peaks may be due to the partial exfoliated structure, in which the gallery height of intercalated layers is large enough and the layer correlation is not detected by XRD (Figure 2). Although XRD provides a partial picture about distribution of nanofiller and disappearance of peak corresponding to d spacing does not always confirm the exfoliated binucleated cells (BNCs), a complete characterization of NC morphology requires microscopic investigation.
FTIR study
Figure 3(a) and (b) shows the FTIR spectra of Mg/Al-LDH and CLDH, respectively. The broad absorption band in the region 3200–3600 cm−1 is assigned to the OH stretching vibrations, ν(OH), associated with the basal hydroxyl groups and interlayer water. At 1625 cm−1, the characteristic band of angular deformation in water molecules, δ(H–O–H), is observed. A strong absorption band at 1360 cm−1 is due to the presence of carbonate. The sharp bands around 780, 554, and 440–450 cm−1 are caused by various lattice vibrations associated with metal hydroxide sheets (Figure 3(a)). The FTIR spectrum of modified LDH shows two types of bands: one corresponding to the chiral anionic species intercalated and other corresponding to the host LDH materials (Figure 3(b)). The broad band in the range 3200–3700 cm−1 mainly from O–H groups of the hydroxide layers. The absorption bands at 2930–3100 cm−1 correspond to the ν asym and ν sym (C–H) modes of CH2 group in the chiral dicarboxylate molecules. The other dicarboxylate bands originated from various functional groups are also found at 1700–1740 cm−1 (C=O) and 1000–1300 cm−1 (C–O). The band broadening by intercalation results from the electrostatic interaction between chiral dicarboxylate molecules and hydroxide sheets to suggest their safe stabilization in the interlayer space of CLDH (Figure 3(b)). The CLDH featured sheets with exposed hydroxyl groups. They allow strong hydrogen bonding with the alcohol functional groups present in the PVA.
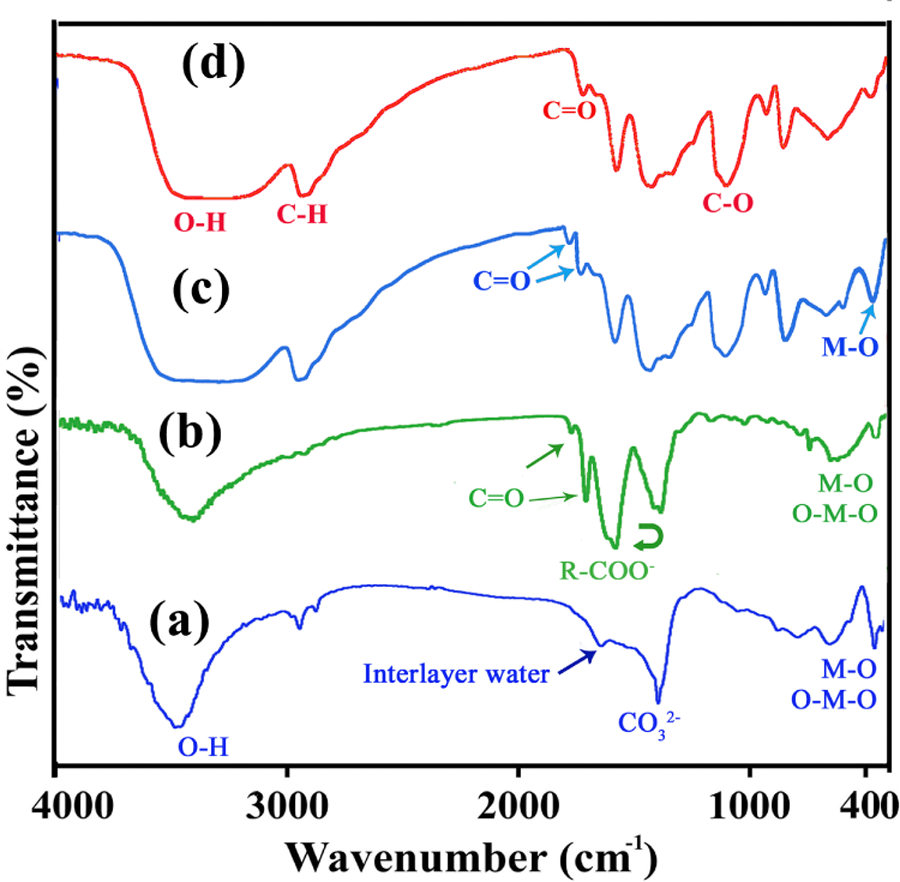
FTIR spectra of (a) LDH-
With the objective to probe the nature of interaction between hydroxyl groups of PVA and CLDH nanoparticles, FTIR spectra were recorded for neat PVA and PVA films containing 4 wt% CLDH in transmittance mode and shown in Figure 3(c) and (d). Since PVA is a hydrolytic derivative of polyvinyl acetate, it shows FTIR peaks characteristics of hydroxyl groups as well as acetate groups of unhydrolyzed poly(vinyl acetate) (Figure 2(d)). Neat PVA shows a broad absorption peak around 3100–3500 cm−1, which is due to the presence of hydroxyl groups of the PVA molecules and adsorbed water. The absorption peak in the region of 2942 cm−1 is due to stretching vibrations of –CH and CH2 groups, respectively. A peak at 1725–1732 cm−1 is due to the residual acetate groups still present in the partially hydrolyzed form of PVA and 1429–1433 cm−1 is corresponding to a C=C group in PVA backbone. A band at 1090–1094 cm−1 corresponds to C–O–C stretching of the acetyl group present on PVA backbone. In the spectrum of NC4%, the formation of new absorption bands in the range of 300–900 cm−1 could be attributed to the M–O stretching. Also, the new absorption bands in the 1735 cm−1 provided the presence of imide carbonyl group of the CLDH. Hence, from FTIR spectroscopy the interaction between PVA and CLDH and their complex formation have been confirmed (Figure 3(c)).
Thermal properties
Thermal decomposition of LDHs normally follows three distinct decomposition steps. 46 –48 The first event is usually assigned to the loss of physisorbed and interlayer water. The onset of this step is at about 50°C and complete by 150°C. The second step is due to a dehydroxylation process and it is immediately followed by an oxidative degradation of the organic anions within the interlayer. The former occurs at about 280°C and the latter above 450°C. Mass loss is effectively complete at 700°C. The thermal decomposition analysis of the unmodified LDH and its modified forms is presented in Figure 4. According to TGA/derivative thermogravimetric thermograms, the low temperature decomposition step in the unmodified LDH lies below 200°C. The high temperature decomposition of the unmodified LDH shows the decomposition peak around 400°C (Figure 4). The organic modification of the LDH changes its the thermal decomposition behavior in comparison with the unmodified sample, especially the second stage of the decomposition process, which results in complete collapse of materials structure. 46 According to the TGA/DTG thermograms, the degradation of interlayer diacids anion took place before dehydroxylation process in the case of modified LDHs and so showed less thermal stability than the original LDH. 46 –48 The presence of a larger weight loss step within 390–560°C for LDH-dicarboxylate anions compared with the LDH confirmed the presence of interlayer surfactant anions in LDH (Figure 4).
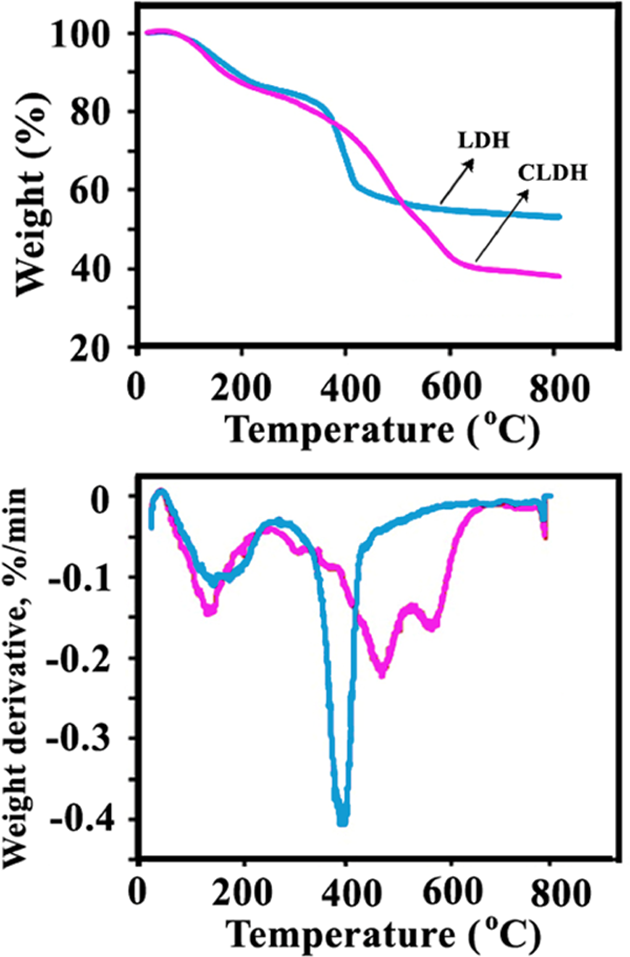
TGA/DTG thermograms of LDH-
The thermal stability of PVA and its composite materials with different amount of CLDH (2, 4, and 8%) was studied by TGA, and the results are demonstrated in Figure 5. The first weight loss process is associated with the loss of absorbed moisture. The second weight loss process corresponds to degradation of the PVA by the dehydration reaction of the polymer chain (side chain). 49 The NC films showed higher resistance toward thermal degradation. In the third weight loss process, the polyene residues are further degraded at approximately 450°C to yield carbon and hydrocarbons. The degradation temperature corresponding to this weight loss process is again independent of the LDH content. This result is in agreement with the near absence of interactions between the residual polyene and the LDH. The neat LDH shows better thermal stability than neat PVA and its composites. The endothermic decomposition of LDHs takes off heat from the surrounding, and the liberated water vapor reduces the concentration of combustible volatile in the vicinity of the polymer surface. As a result, the decomposition temperature of the polymer is increased. 50 As can be seen from Figure 5, PVA film shows 7% residue at 800°C, while the NC films show 10–17% residue at this temperature. It is notable that even small amounts of clay were effective in improving the weight residues of the hybrids.
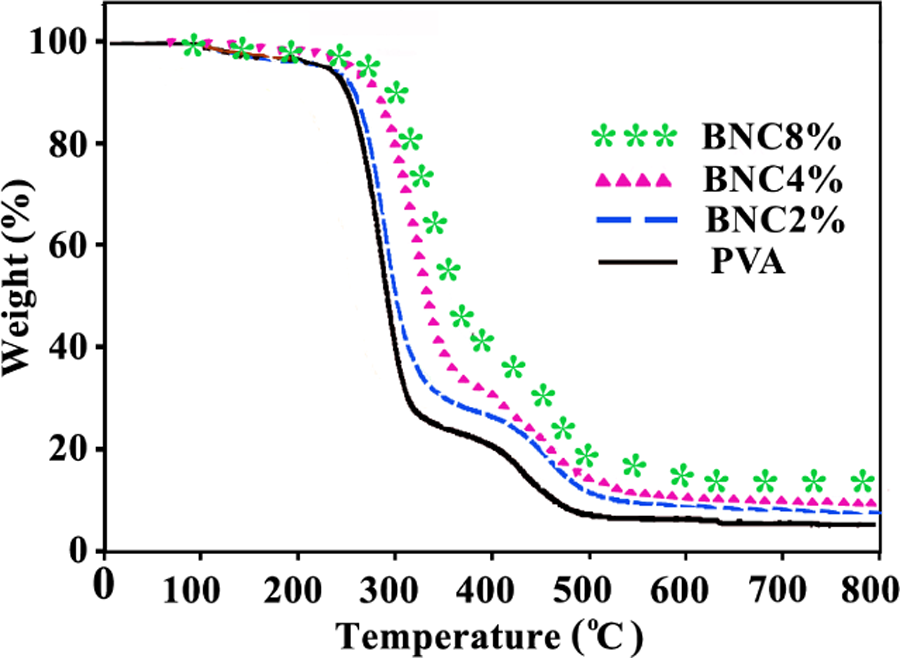
TGA thermograms of neat PVA and different PVA/CLDH NCs. TGA: thermogravimetric analysis; PVA: poly(vinyl alcohol); CLDH: chiral diacid intercalated layered double hydroxide; NCs: nanocomposites.
Morphological study
Figure 6(a) to (d) shows the TEM images of the synthesized

TEM micrographs of (a, b) LDH-
The FESEM micrograph of

FESEM photographs of (a, b) LDH-
The FESEM images of neat PVA and PVA/CLDH NC4% and PVA/CLDH NC8% are shown in Figure 8(a) to (f), respectively. According to these photographs, in the presence of different amounts of CLDH, the morphology of PVA is changed. In the BNCs, the micrograph exhibits good dispersion of CLDH into PVA matrix. These observations suggested that the presence of LDH has a positive effect on PVA morphology. This morphological change can be attributed to the reordered crystalline phase of the PVA matrix in the presence of modified LDH, causing a packed network. It seems that the particles are distributed uniformly in the polymer matrix.

FESEM photographs of (a, b) pure PVA, (c, d) NC4%, and (e, f) NC8%. FESEM: field-emission scanning electron microscopic; PVA: poly(vinyl alcohol); NCs: nanocomposites.
Conclusions
Bioactive amino acids containing dicarboxylic acid was used for the preparation of the novel organomodified CLDH in one step by coprecipitation reaction in aqueous media under green conditions. CLDH was characterized by XRD, FTIR, TGA, FESEM, and TEM techniques. The XRD results of the CLDH show that the diacid is intercalated in the interlayer region of Mg/Al-LDH and enlarge the interlayer distance. Novel PVA/CLDH hybrid NCs were prepared via nanoplatelet-like organic LDHs by solution intercalation method using ultrasound energy. Three polymer/CLDH NCs were prepared by loading different amounts of CLDH (2, 4, and 8%) into PVA matrix. The XRD and TEM observation confirmed the formation of exfoliated microstructure of the polymer-based NCs. The thermal stability was increased compared with the pure polymer. Since both chiral amino acid-based dicarboxylic acid and PVA are biodegradable and biocompatible, the resulting hybrid organic–inorganic PVA/CLDH NCs are expected to be biodegradable. Besides, the organic LDH nanoplatelets can be used with other polymers for preparing exfoliated polymer/LDH NCs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to express their gratitude to the Research Affairs Division, Isfahan University of Technology (IUT), Isfahan, Islamic Republic of Iran, for partial financial support of this research. Also, the present study was financially supported by the National Elite Foundation (NEF), Iran Nanotechnology Initiative Council (INIC), and Center of Excellency in Sensors and Green Chemistry Research (IUT).
