Abstract
In this study, the thermally stable poly(amide–imide) (PAI)/cupric oxide (CuO) nanocomposites (NCs) containing 4, 6 and 8 wt% of CuO nanoparticles (NPs) were prepared by ultrasonic technique. At first, the optically active PAI was synthesized from the polymerization of a chiral monomer, N-trimellitylimido-
Keywords
Introduction
Nanocomposite (NC) is an interesting class of composites containing two or more phases, where at least one of the dimensions of the filler materials is in the nanoscale. In the last 20 years, the research on polymeric NCs (PNCs) has been extremely extended. PNCs have an enormous potential for many various applications, such as automobiles, sporting goods, packaging and electrical applications. 1 These materials have exhibited significant improvement in tunable physical properties (e.g., optical, mechanical, electronic and magnetic properties), permeability, thermal stability, flame retardancy and chemical resistance. These superior properties are owing to nanometre size characteristic of fillers, so various kinds of additives are necessary to be added to the pure polymers. 2 –4
Cupric oxide (CuO) nanoparticles (NPs) have been widely studied because of their unique properties such as catalytic and chelating effect, 5 special electrical and optical properties 6 and antioxidant and antimicrobial behavior. 7 This metal oxide has broad applications in the electrochemical cells, gas sensors and lubricant. 8 –11 CuO NPs are added to different materials for the preparation of a wide range of nanostructured composites. 12 –18
The properties of the particle-reinforced PNCs intensely depend upon the dispersion ability of nanomaterials in the polymer matrix, 19 but NPs due to their high surface energy tend to strongly agglomerate. 20 In order to achieve an improved compatibility between the NPs and polymer matrix, the use of different coupling agents containing hydroxyl (OH) and carboxyl groups for surface modification of NPs were suggested. 21 –23 The existence of these organic functional groups on the surface of the NPs leads to the formation of chemical and physical interactions with the polymer matrix. 24
In recent years, the utilization of ionic liquids as solvents in green chemistry has attracted wide attention because of its considerable properties, such as extensive-ranging liquid, high polarity, solvating ability of organic, inorganic and polymer materials, non-flammability, low volatility, exceptional chemical and thermal resistance. 25,26
Through the past decade, the research on biodegradable polymer-based NCs has been particularly increased. 27,28 Biodegradable polymers have been employed in the industrial and biomedical segments. 29 Aromatic polyimides are a kind of polymer materials with high performance. However, their applications are reduced owing to their high melting temperatures and insoluble nature in common organic solvents. To overcome these problems, some structural modifications often become necessary. For example, synthesis of poly(amide–imides) (PAIs) was caused balance between excellent thermal stability and processability. 30 PAIs are widely utilized for different applications due to its high-temperature performance, solvent resistance and special mechanical and thermal properties. 31,32
Fabrication and characterization of PAI-based NCs reinforced with modified NPs have been performed in our previous works. 33 –36 In the present study, first, the biodegradable PAI 37 was prepared by polycondensation reaction. Then, CuO NPs were modified using a chiral diacid (DA) coupling agent to increase the dispersion ability of the NPs into the polymeric matrix. Finally, NCs reinforced with CuO NPs were synthesized and characterized by various techniques, such as Fourier transform infrared spectra (FT-IR), X-ray diffraction (XRD), thermogravimetric analysis (TGA), field emission scanning electron microscopy (FE-SEM) and transmission electron microscopy (TEM). The optical properties of the obtained NCs were studied using ultraviolet–visible (UV–vis) spectroscopy.
Experimental
Materials
Nanosized CuO powder was purchased from Nanosabz Co. (Tehran, Iran), with average particle size <50 nm and specific surface area >80 m2 g−1. Solvents and chemicals were obtained from Aldrich Chemical Co. (Milwaukee, Wisconsin, USA), Fluka Chemical Co. (Buchs, Switzerland), Riedel-deHaen AG (Seelze, Germany) and Merck Chemical Co. (Darmstadt, Germany).
Characterization techniques
FT-IR spectra were recorded on a Jasco-680 spectrophotometer (Tokyo, Japan) at a resolution of 4 cm− 1. The samples were mixed with potassium bromide powder, ground and compressed into a pellet. They were scanned at wave number in the range of 400–4000 cm−1. Proton nuclear magnetic resonance ( 1 H NMR, 500 MHz) spectra were recorded in N,N-dimethylsulfoxide-d 6 solution by a Bruker Avance 500 instrument (Germany). The thermal stability of NCs was examined by thermal analyser (model STA503, BÄHR thermoanalyse GmbH, Hüllhorst, Germany) from 25 to 800°C at a heating rate of 20°C min−1 under nitrogen atmosphere. The XRD pattern of the synthesized samples was studied by an x-ray diffractometer (Philips Xpert MPD, Karlsruhe, Germany), with copper (Cu) Kα radiation (λ = 1.540 Å) over Bragg angles ranging from 10° to 80° at the speed of 0.05° min−1. The operating voltage and current were maintained at 40 kV and 35 mA, respectively. The morphology of the prepared NCs was investigated using a transmission electron microscope (model CM 120; Philips, Germany) under an acceleration voltage of 100 kV and field emission scanning electron microscope (model S-4160; HITACHI, Tokyo, Japan). The preparation of NCs was carried out by MISONIX (Raleigh, North Carolina, USA) ultrasonic liquid processors, XL-2000 SERIES (USA), with frequency wave of 2.25 × 104 Hz. The absorption spectra of prepared NCs were measured using a UV/vis/near infrared spectrophotometer (model V-570; JASCO, Tokyo, Japan), with solid pellets of the samples in the spectral range between 200 and 800 nm.
Synthesis of N-trimellitylimido-l -valine monomer and polymer
N-Trimellitylimido-
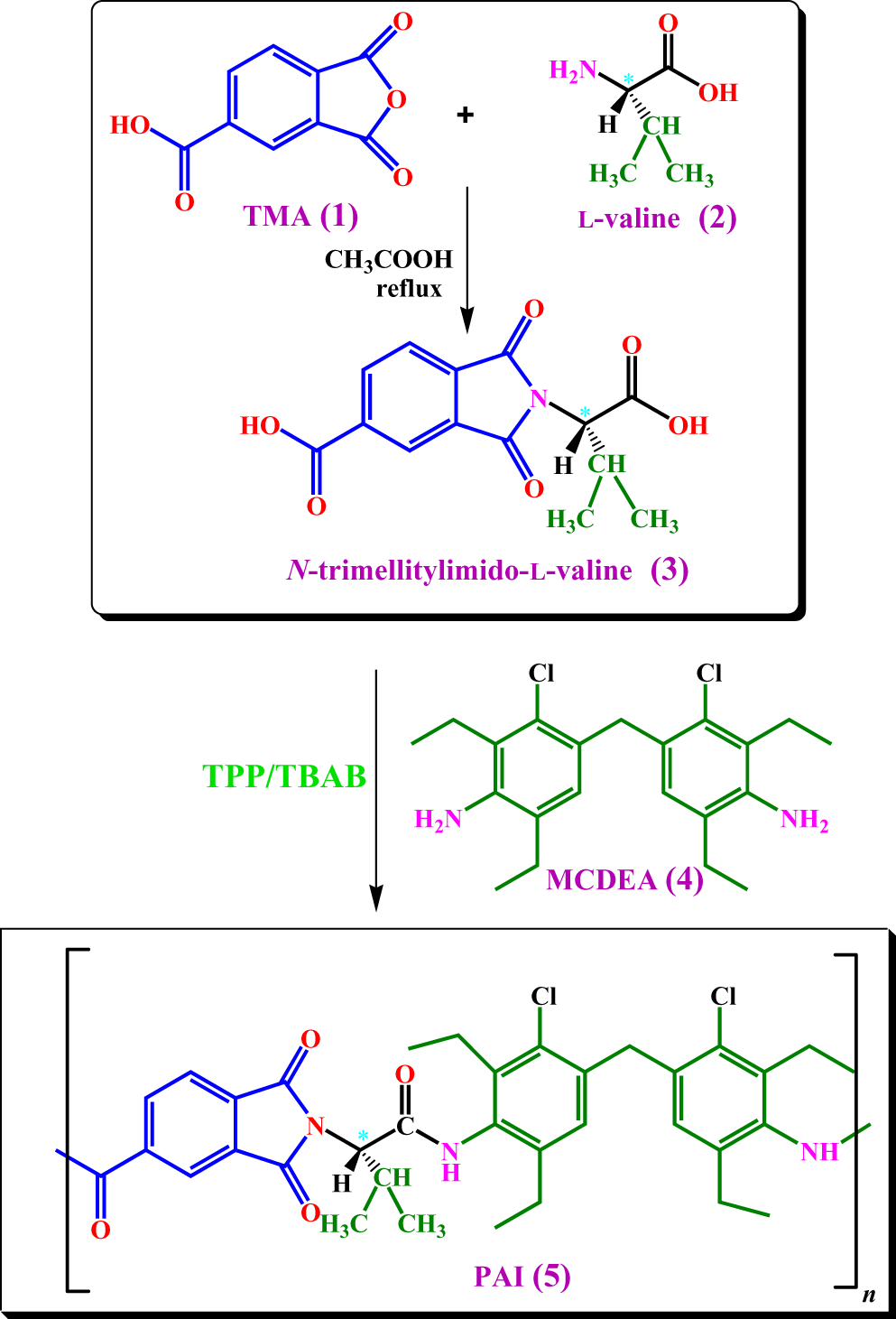
Syntheses of DA and PAI. DA: diacid; PAI: poly(amide–imide).
Surface modification of CuO NPs with N-trimellitylimido-l -valine
In order to modify the surface of NPs, 0.10 g of CuO nanopowder and 15 wt% of N-trimellitylimido-
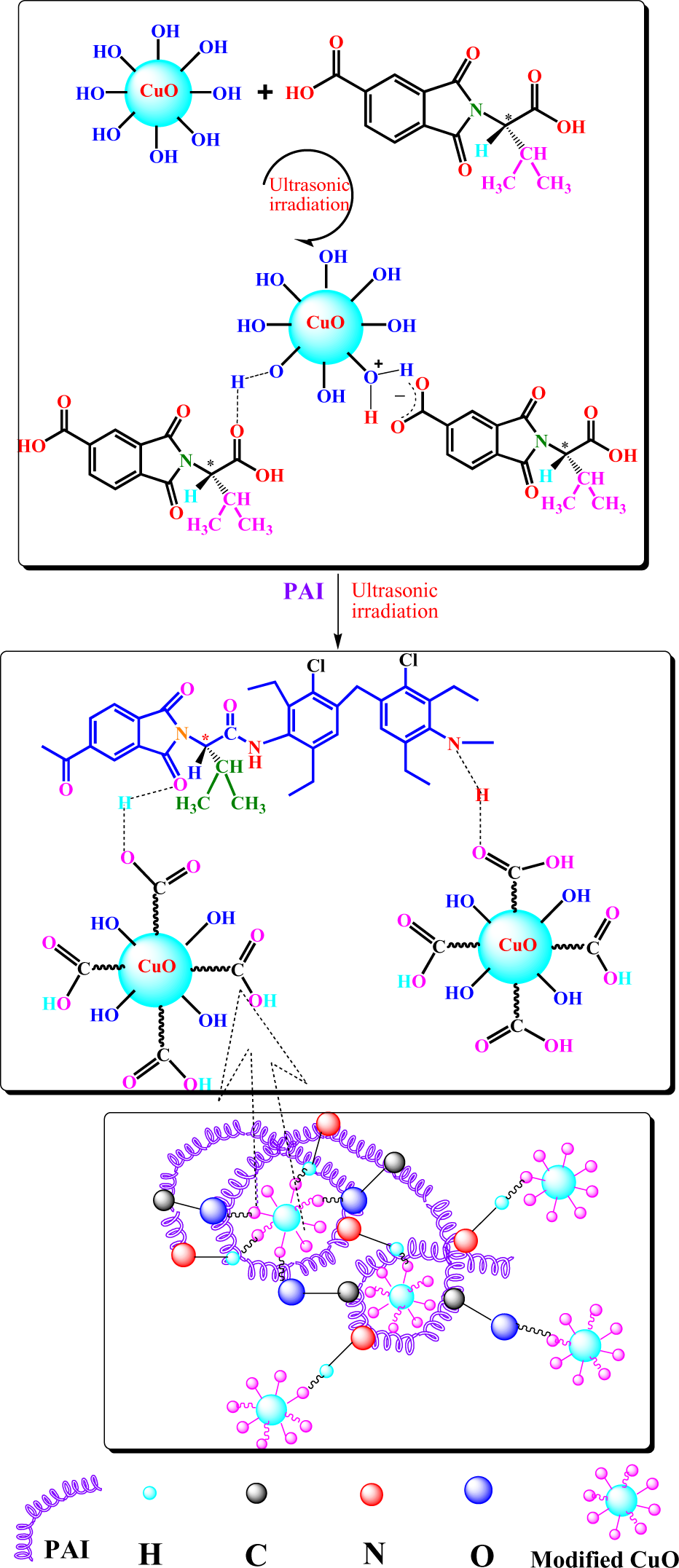
Preparation of PAI/CuO NCs. PAI: poly(amide–imide); CuO: cupric oxide; NCs: nanocomposites.
Fabrication of PAI/CuO NCs
The preparation process of NCs is as follows. The samples were produced by mixing different amounts of modified CuO NPs (4, 6 and 8 wt%) to the 0.10 g of synthesized PAI in 20 mL of absolute ethanol. Solution was dispersed by stirring for 5 h at room temperature. Then, the NPs/PAI mixture was ultrasonically stirred for 30 min. At last, ethanol was removed and the pale brown solid was dried under vacuum for 2 h at 80°C (Figure 2).
Result and discussion
Syntheses of chiral bioactive DA and biodegradable polymer
N-Trimellitylimido-
Preparation and FT-IR characterization of PAI/CuO NCs
It has been reported that the CuO NPs which were mixed with polymer matrix improved the thermal conductivity, thermal stability and other properties of the obtained NCs. 3,5,13 In order to avoid the formation of agglomerated NPs and to increase their dispersion in the matrix of polymer, it is essential to modify the surface of NPs. 20 Organic molecules containing two or more functional groups, such as dicarboxylic acids are appropriate coupling agents for surface modification of NPs. The modifiers were joined to the surface of the NPs via the coordination bond between the metal ions and the carboxylate group. 38 In this study, the surface of CuO NPs was treated with a bioactive DA using the sonochemical reaction. The modified NPs are attached to the PAI chains through H bonding between –OH groups and amidic–NH. The details of preparation of PAI/CuO NCs are shown in Figure 2.
Figure 3 shows the FT-IR spectra of pure CuO NPs (a), N-trimellitylimido-
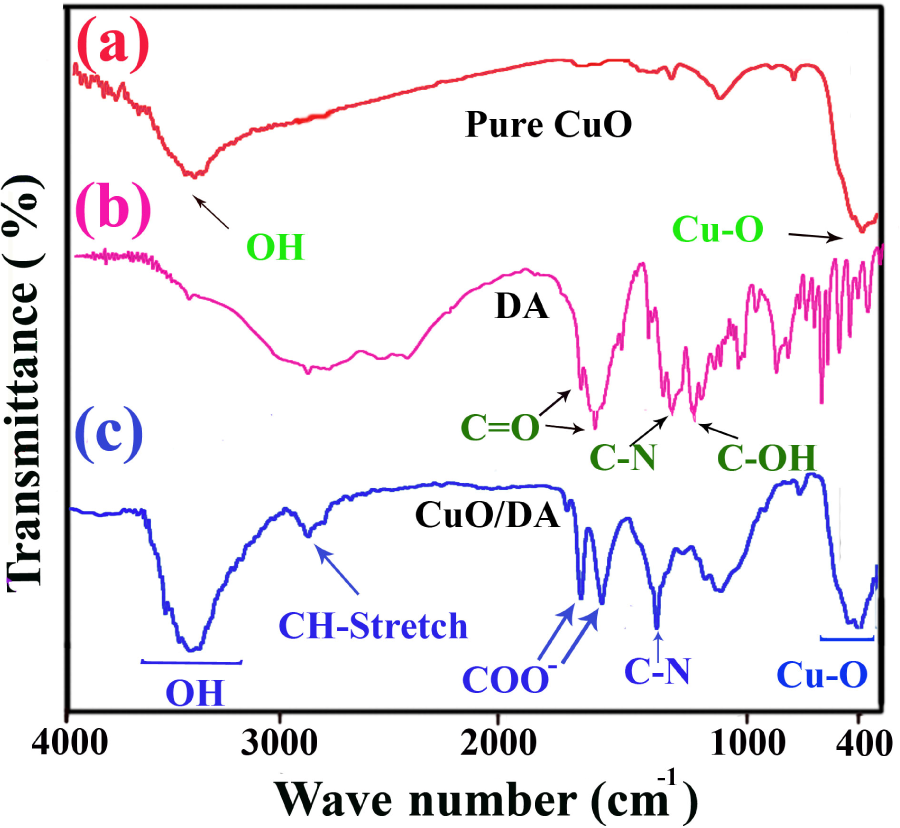
FT-IR spectra of unmodified CuO NPs (a), DA (b) and modified CuO NPs (c). FT-IR: Fourier transform infrared spectra; CuO: cupric oxide; NPs: nanoparticles; DA: diacid.
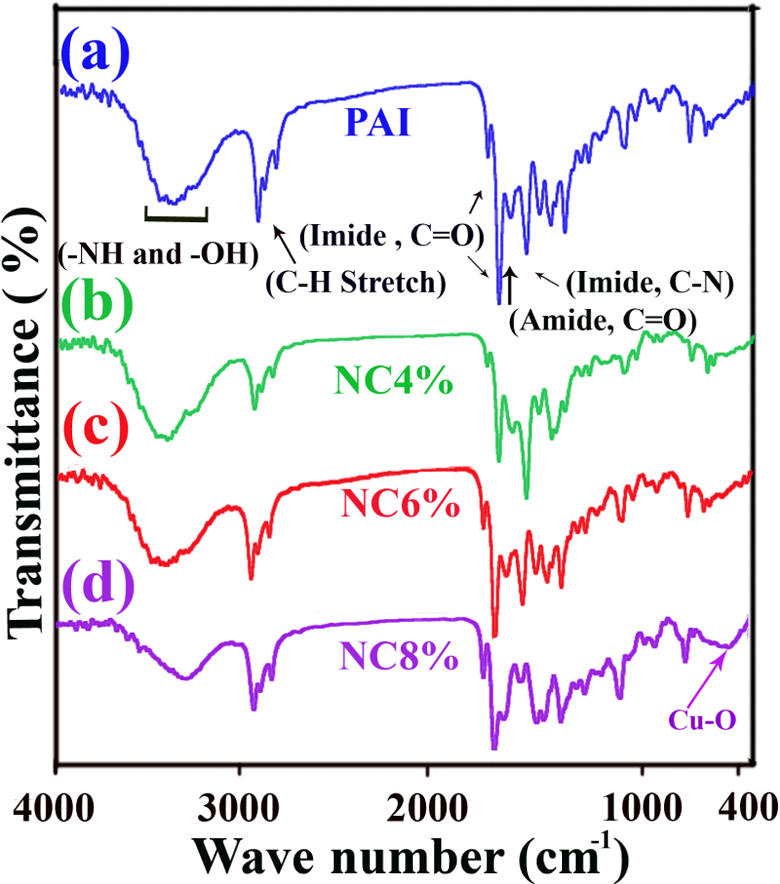
FT-IR spectra of pure PAI (a), 4 wt% PAI/CuO NC (b), 6 wt% PAI/CuO NC (c) and 8 wt% PAI/CuO NC (d). FT-IR: Fourier transform infrared spectra; PAI: poly(amide–imide); CuO: cupric oxide; NC: nanocomposite.
TGA
Figure 5 shows the thermal degradation of the pure PAI and PAI with different weight percentage of CuO NPs. The curves of thermal decomposition of the neat PAI and PAI/CuO NCs have two-step degradation processes. In the first step, slight weight loss is associated with the removal of moisture and physisorbed water molecules and the second weight loss is related to the degradation process of PAI chain. However, the thermogravimetric curves of the PAI/CuO NCs are placed at a higher temperature than that of the pure PAI. The improvement in thermal stability after the reinforcement of PAI with modified NPs may proves a strong interaction between the modified NPs and the polymer matrix. Metallic compounds could have catalytic effect on degradation of the polymer matrix. 3 Therefore, the existence of the pure CuO NPs in the NCs may facilitate the thermal decomposition of the PAI by serving as a catalyst. But the presence of a thin interfacial layer from DA inhibits the direct contact of the CuO NPs with the polymer matrix by passivating the particle surface and so can increase the thermal stability of the NCs. Thermal stabilities of the PAI and PAI/CuO NPs were investigated based on 5 and 10% weight loss (T 5 and T 10) and residue at 800°C (CR, char yield). Also the limiting oxygen index (LOI) for materials is calculated based on Van Krevelen and Hoftyzer equation. 39
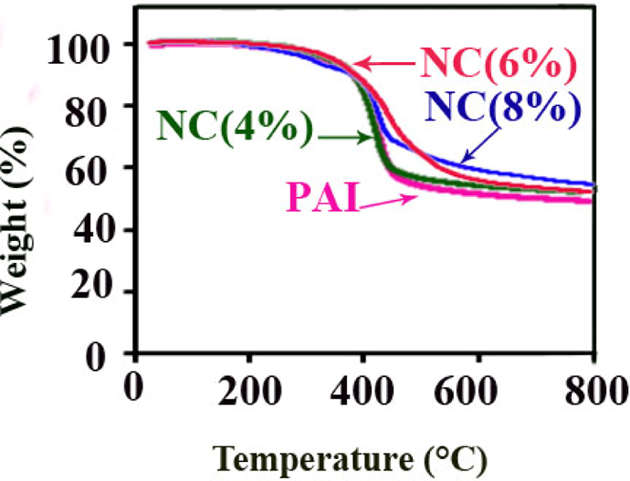
TGA thermograms of PAI and PAI/CuO NCs under argon atmosphere at heating rate of 20°C min−1. TGA: thermogravimetric analysis; PAI: poly(amide–imide); CuO: cupric oxide; NCs: nanocomposites.
Thermal properties of PAI and PAI/CuO NCs.
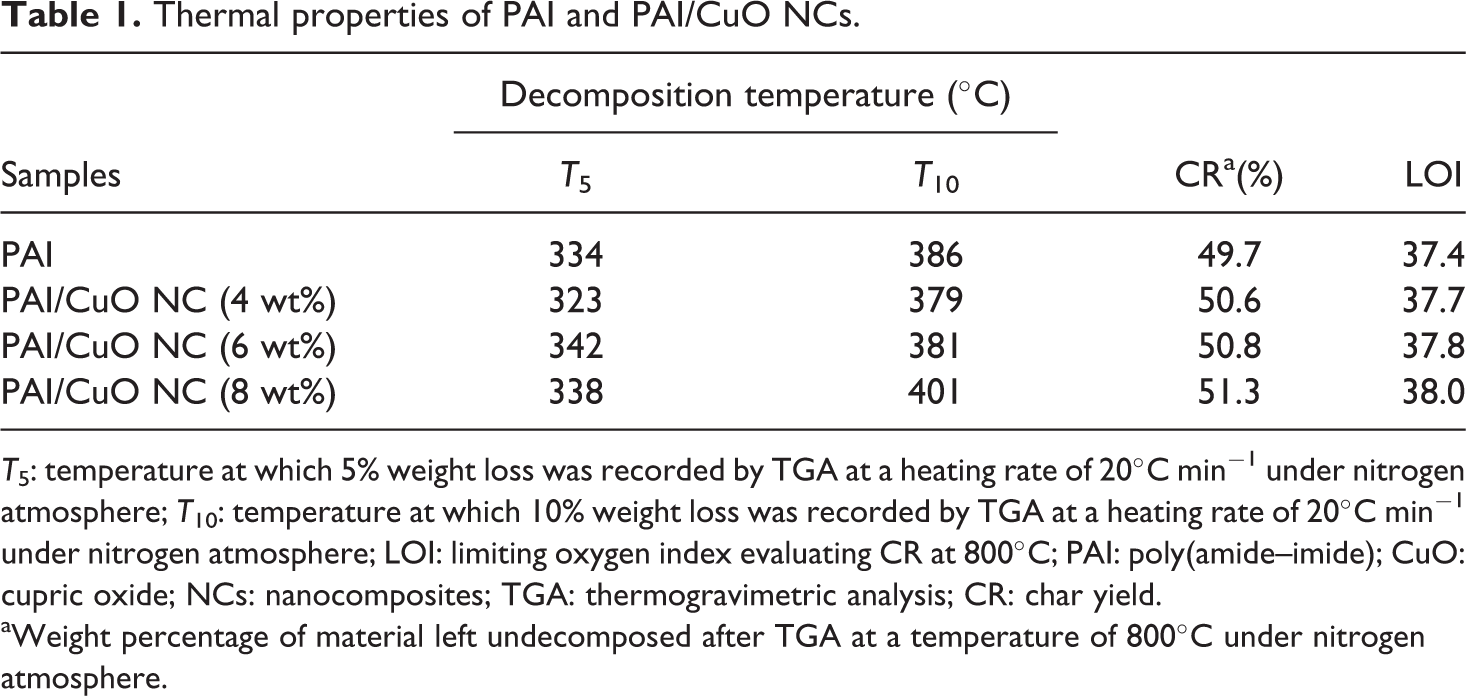
T 5: temperature at which 5% weight loss was recorded by TGA at a heating rate of 20°C min−1 under nitrogen atmosphere; T 10: temperature at which 10% weight loss was recorded by TGA at a heating rate of 20°C min−1 under nitrogen atmosphere; LOI: limiting oxygen index evaluating CR at 800°C; PAI: poly(amide–imide); CuO: cupric oxide; NCs: nanocomposites; TGA: thermogravimetric analysis; CR: char yield.
aWeight percentage of material left undecomposed after TGA at a temperature of 800°C under nitrogen atmosphere.
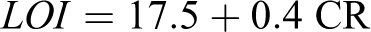
X-ray diffraction analysis
Figure 6 illustrates the XRD results of the neat PAI (a), CuO NPs (b), modified CuO (c), 6 wt% PAI/CuO NC (d) and 8 wt% PAI/CuO NC (e). Figure 6(a) shows the structure of synthesized neat PAI is amorphous. In Figure 6(b), the diffraction peaks can be indexed to a monoclinic CuO NPs. The XRD patterns of NCs (Figure 6(d) and (e)) clearly indicated that the samples are crystalline and consequent, processing of creation of NCs did not change the crystalline phase of CuO NPs. The average crystalline size of the CuO NPs in the composites was estimated to be about 35–50 nm using the Debye–Scherrer formula from the (200) reflection.
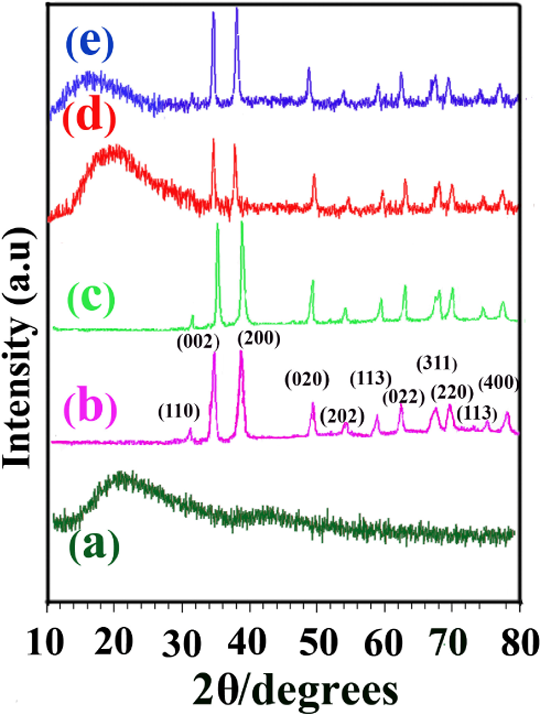
XRD patterns of neat PAI (a), CuO NPs (b), modified CuO NPs (c), 6% NC (d) and 8% NC (e). XRD: x-ray diffraction; PAI: poly(amide–imide); CuO: cupric oxide; NPs: nanoparticles; NC: nanocomposite.
Size and morphology of PAI/CuO NCs
The structures, morphologies and sizes of modified CuO NPs, pure PAI and PAI/CuO NCs are demonstrated by means of FE-SEM and TEM (Figures 7 and 8). FE-SEM micrographs indicate that for 4 wt% NCs (Figure 7(e) and (f)), the CuO NPs were uniformly distributed in the polymer matrix, but in the case of 6 and 8 wt% NCs (Figure 7(g) to (j)), some aggregation are recognized with further enhancing NPs concentration. TEM images of pure CuO NPs, modified CuO NPs and NCs with the filler loading 4 wt% of CuO NPs are shown in Figure 8(a) to (f). TEM micrographs proved the presence of rod-like nanosize particle (30–50 nm) in the polymer matrix (Figure 8(d) to (f)). Also these photographs show that the shape of NCs is beehive like (honeybee) and introducing PAI did not change the general shape of CuO NPs. TEM observations proposed a proper dispersion of NPs in the polymer matrix at low concentration.

FE-SEM photographs of modified CuO ((a) and (b)), pure PAI ((c) and (d)), 4% PAI/CuO NC ((e) and (f)), 6% NC ((g) and (h)) and 8% NC ((i) and (j)). FE-SEM: field emission scanning electron microscopy; CuO: cupric oxide; PAI: poly(amide–imide); NC: nanocomposite.

TEM micrographs of pure CuO (a), modified CuO ((b) and (c)) and PAI/CuO NC containing 4 wt% CuO NPs ((d) to (f)). TEM: transmission electron microscopy; CuO: cupric oxide; PAI: poly(amide–imide); NC: nanocomposite; NPs: nanoparticles.
Optical properties of NCs
The UV–vis absorption spectra of the pure PAI, modified CuO and NCs containing different amounts of CuO NPs are illustrated in Figure 9(a) to (e). The modified CuO with N-trimellitylimido-
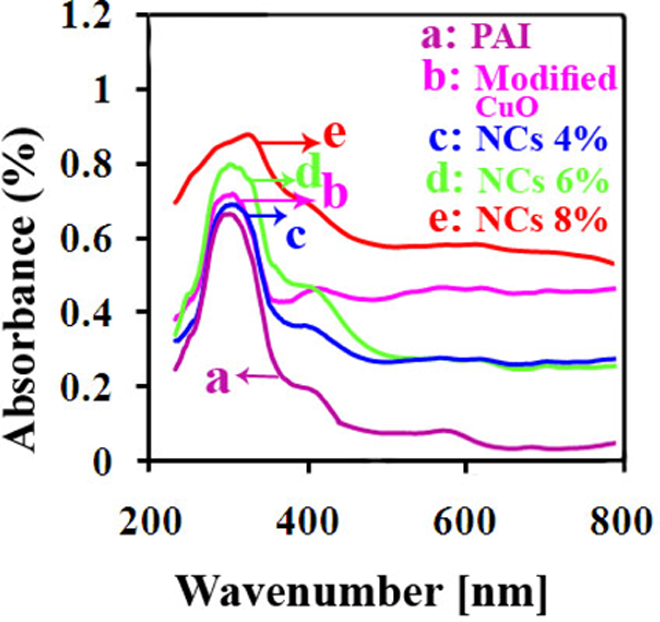
UV absorption spectra of the PAI/CuO NCs. UV: ultraviolet; PAI: poly(amide–imide); CuO: cupric oxide; NCs: nanocomposites.
Conclusions
In the present investigation, the PAI-based NCs reinforced with CuO NPs were prepared. At first, a thermally stable optically active PAI was synthesized via direct polycondensation of DA with MCDEA in the presence of TBAB and TPP. Secondly, in order to improve the dispersion ability of NPs in the polymer matrix, the surface modification of NPs was carried out using N-trimellitylimido-
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to acknowledge the help and financial support of the Research Affairs Division Isfahan University of Technology (IUT). Further financial support from National Elite Foundation (NEF) and Center of Excellency in Sensors and Green Chemistry Research (IUT) Iran Nanotechnology Initiative Council (INIC) is gratefully acknowledged.
