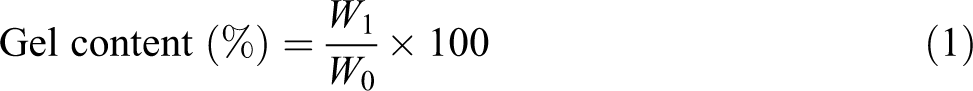Abstract
In an attempt to maximize the beneficial effect of irradiation, the influence of multifunctional acrylates (MFAs) such as trimethylol propane triacrylate, ethylene glycol dimethacrylate, trimethylol propane trimethacrylate, and tripropylene glycol diacrylate on the 90/10 ethylene vinyl acetate/waste tire dust (EVA/WTD) were studied. The 90/10 EVA/WTD and EVA-containing 4 phr MFA prepared using a Haake mixer at 140°C and 50 r min−1 rotor speed. The blends were then irradiated using a 3.0-MeV electron beam machine. Results on gel fraction revealed that EVA/WTD blends were cross-linked by electron beam irradiation. Among the MFA employed in this studies, TPGDA was found to render highest tensile strength with the best retention in elongation at break and increased heat of fusion and crystallinity of blends upon irradiation. The changes in the properties of EVA/WTD blends upon irradiation are attributed to the cross-linking of the EVA matrix. The changes in crystallinity and crystalline melting temperatures of EVA/WTD blends upon irradiation are discussed in detail.
Keywords
Introduction
The large amount of disposable tires presently produced makes imperative the search for alternative procedures for recycling or reuse of tires, since they are not biodegradable. Recycling discarded tires is a challenging process to accomplish due to the highly cross-linked nature of tires. There have been substantial progresses in investigating new technology to utilize the scrap tires. Among them are by using it for cleaning up oil spill, road surface, roofing material, and playground surfaces. To date, recycled rubber has not been used as a replacement for new or synthetic rubber in significant quantities, largely because the desired properties have not been achieved. As chemical processing is most often costly and sometimes aggressive to the environment, a possible solution is the recycling of such material by blending with thermoplastics. Several studies have been carried out by blending recycled waste tire dust (WTD) with thermoplastics such as polypropylene (PP), 1 recycled high-density polyethylene (rHDPE), 2 natural rubber (NR) 3 , and ethylene vinyl acetate (EVA). 4 However, the blends obtained found to exhibit poor compatibility and homogeneity that resulted in poor mechanical properties.
High-energy radiation such as γ and electron beam radiation has been extensively used to improve mechanical properties and enhance compatibilization of polymeric blends. A few studies has employed the use of high-energy radiation to recycled materials such as WTD and found promising future for properties enhancement and improved compatibilization of the blends. Studies on electron beam irradiation of EVA/WTD 5 and γ irradiation of rHDPE/WTD 6 and indicated that an improved adhesion between WTD particles and the surrounding thermoplastic matrix can be achieved by irradiation.
Use of polyfunctional monomers, such as multifunctional acrylates (MFAs) and allelic reactive molecules, with recycle-based polymer blends are rare. Polyfunctional monomers are effective ways to achieve desirable cross-linking at lower irradiation dose. 7 This is very crucial in processing recycled material as it would prevent properties deterioration at higher irradiation doses. In previous work, 5 we have indicated that the irradiation of EVA/WTD blend in the presence of trimethylol propane triacrylate (TMPTA) and tripropylene glycol diacrylate (TPGDA) enhanced the compatibility of the blend. Dynamic mechanical analyses of the EVA/WTD blends were conducted to show the changes in the thermal properties (tan δ curve) in the amorphous phase of EVA. Meanwhile, in this study the effect of various MFA on the tensile, morphology, and differential scanning calorimetry (DSC) of the EVA/WTD blends were reported with a particular attention to changes in melting behavior and crystallinity of EVA phase due to irradiation-induced cross-linking.
Experimental
Materials
EVA (Grade H2020) having 15% vinyl acetate content with melt flow index (MFI), value of 1.5 g/10 min and density of 0.93 g cm−3 was purchased from the Polyolefin Company (Singapore). Rubber dust from recycle tires (40 mesh) used in this study was obtained from Sin Rubtech Consultancy Sdn. Bhd. (Malaysia) The TMPTA, ethylene glycol dimethacrylate (EGDMA), trimethylol propane trimethacrylate (TMPTMA), and TPGDA were products of UCB Asia Pacific. They were used as received.
Blend preparations
The EVA/WTD blends with 90/10 blend ratio containing 4 phr MFA were prepared by melt mixing the EVA, WTD, and the respective MFA in a Haake Rheomix Polydrive R600/610 at 140°C and 50 r min−1 rotor speed for 10 min. The blending was carried out as follows:
When the desired temperature was reached, EVA was charged into the mixing chamber and allowed to melt for 2 min. Then, WTD and MFA were added to the molten EVA, and the mixing was continued for another 10 min. The blends from the Haake Rheomix were then compression molded into 1 mm thick sheets under a pressure of 14.7 MPa at 123°C for 5 min. The sheets were immediately cooled between two plates of a cold press at 25°C. Similar preparation method was followed for the preparation of EVA compounds (0 parts of WTD).
Irradiation
The molded sheets were irradiated in air at room temperature using a 3-MeV electron beam accelerator model NHV EPS-3000 at a dose range of 0 to 200 kGy. The acceleration energy, beam current, and dose rate were 2 MeV, 2 mA, and 50 kGy per pass, respectively.
Gel content
The gel content of the samples was determined by the extraction of samples in boiling xylene for 24 h using Soxhlet apparatus. The extracted samples were dried in oven at 50°C till constant weight is obtained. The gel fraction was calculated as:
Tensile properties
The tensile properties were measured with an Instron universal testing machine 4301 H119 at a crosshead speed of 50 mm min−1. The molded sheets of 1 mm thickness were cut into dumbbell-shaped test pieces using BS6746 die. Five samples were used for tensile test, and average results were taken as the resultant value.
Differential scanning calorimetry
Differential scanning calorimetric (DSC) thermograms were obtained with a Perkin Elmer DSC-7 (USA) at a heating rate of 10°C min−1. The samples were scanned from 0 to 140°C using nitrogen. The result was taken from the first scanning process. The melting temperature (Tm) and heat of fusion of the samples (ΔHf) were determined from the DSC thermograms. The percentage of crystallinity (Xc) of EVA in the blend was calculated as equation (2).
Scanning electron microscopy
Selected samples were manually fractured at liquid–nitrogen temperature. The fractured surfaces were then sputter coated with gold and examined using model Zeiss SUPRA 35VP (Germany). The SEM micrographs were taken at a magnification of 1000×.
Results and discussion
Tensile properties
Tensile strength
The effect of radiation on the tensile strength (TS) of EVA/WTD blend and EVA compounds are shown in Figures 1 and 2, respectively. It is obvious from Figures 1 and 2 that property enhancement has occurred upon irradiation of EVA/WTD blend and EVA compounds. Such enhancement in the TS is associated with the occurrence of radiation-induced cross-linking in the EVA/WTD. Similar observation was reported on irradiation modification of polyvinyl chloride (PVC)/epoxidized natural rubber (ENR) blends 9 and EVA/ENR blends. 10 The addition of MFA found to further improve the TS of the EVA/WTD blend and EVA compound upon irradiation. This improvement in TS is attributed to the acceleration of irradiation-induced cross-linking of the polymers by the MFA, akin to those reported by Ratnam and Zaman. 11 It is of interest to note that upon addition of MFA, the EVA/WTD blend and EVA compounds achieve the maximum TS at 50 kGy. Beyond this point, subsequent irradiation yields reduction in TS. This is believed to be associated with the embrittlement of the blends due to high extent of cross-linking. Similar observations were also noted by Thomas et al. 12 while studying γ-irradiated blends of PVC and thermoplastic copolyesters. 12 It is also apparent from Figures 1 and 2 that the compounds containing TPGDA exhibit the highest TS value between 50 and 150 kGy, implying TPGDA is the best choice of radiation sensitizer for EVA-based compounds investigated in this study.
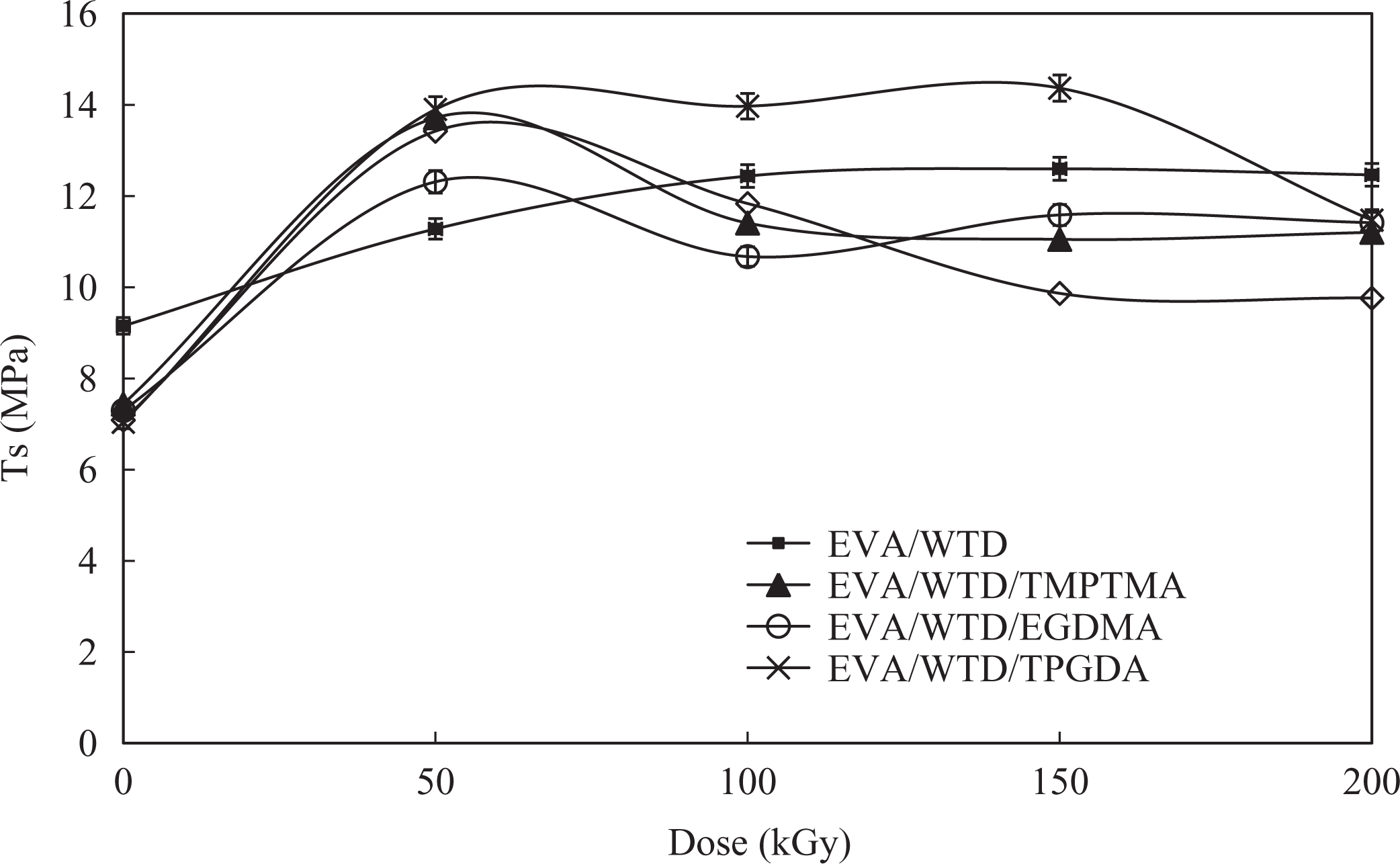
Effect of irradiation on the TS of EVA/WTD blends. TS: tensile strength; EVA: ethylene vinyl acetate; WTD: waste tire dust.
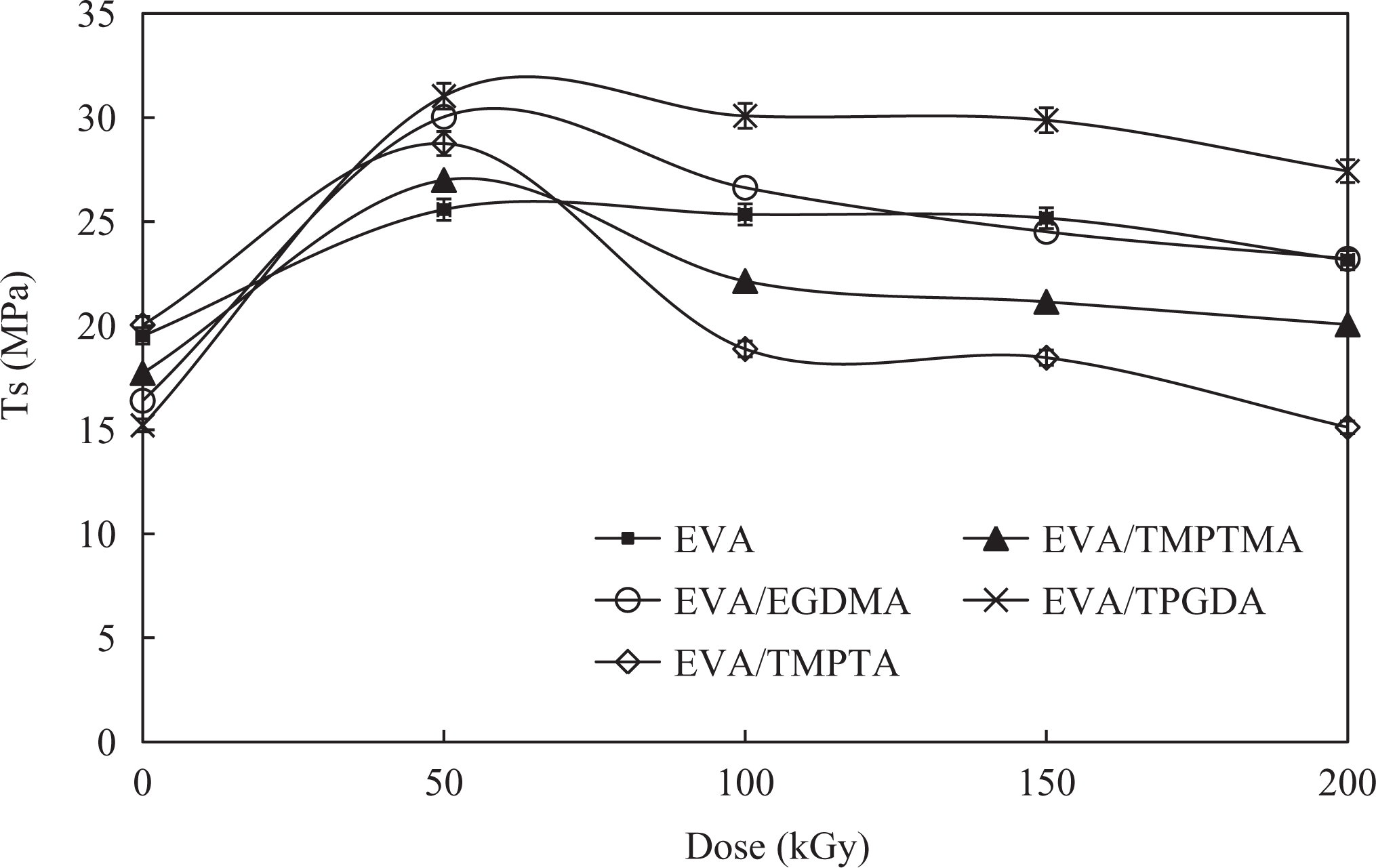
Effect of irradiation on TS of EVA compounds. TS: tensile strength; EVA: ethylene vinyl acetate.
Elongation at break
The effect of MFA on the elongation at break (EB) of the EVA/WTD blend and EVA compound with increasing irradiation dose is illustrated in Figures 3 and 4, respectively. The EB found to show a gradual drop with irradiation dose. Indeed, the addition of TMPTMA, EGDMA, and TMPTA found to cause a more drastic deterioration in EB compared with their control containing no MFA. Such a decline in EB indicates that the compounds losing their elastic properties become increasingly brittle as a consequence of the increase in cross-link density upon irradiation in the presence of MFA. The best retention in EB obtained for the compounds containing TPGDA suggests that the acceleration of radiation-induced cross-linking by the TPGDA is relatively less vigorous compared with TMPTMA, EGDMA, and TMPTA.
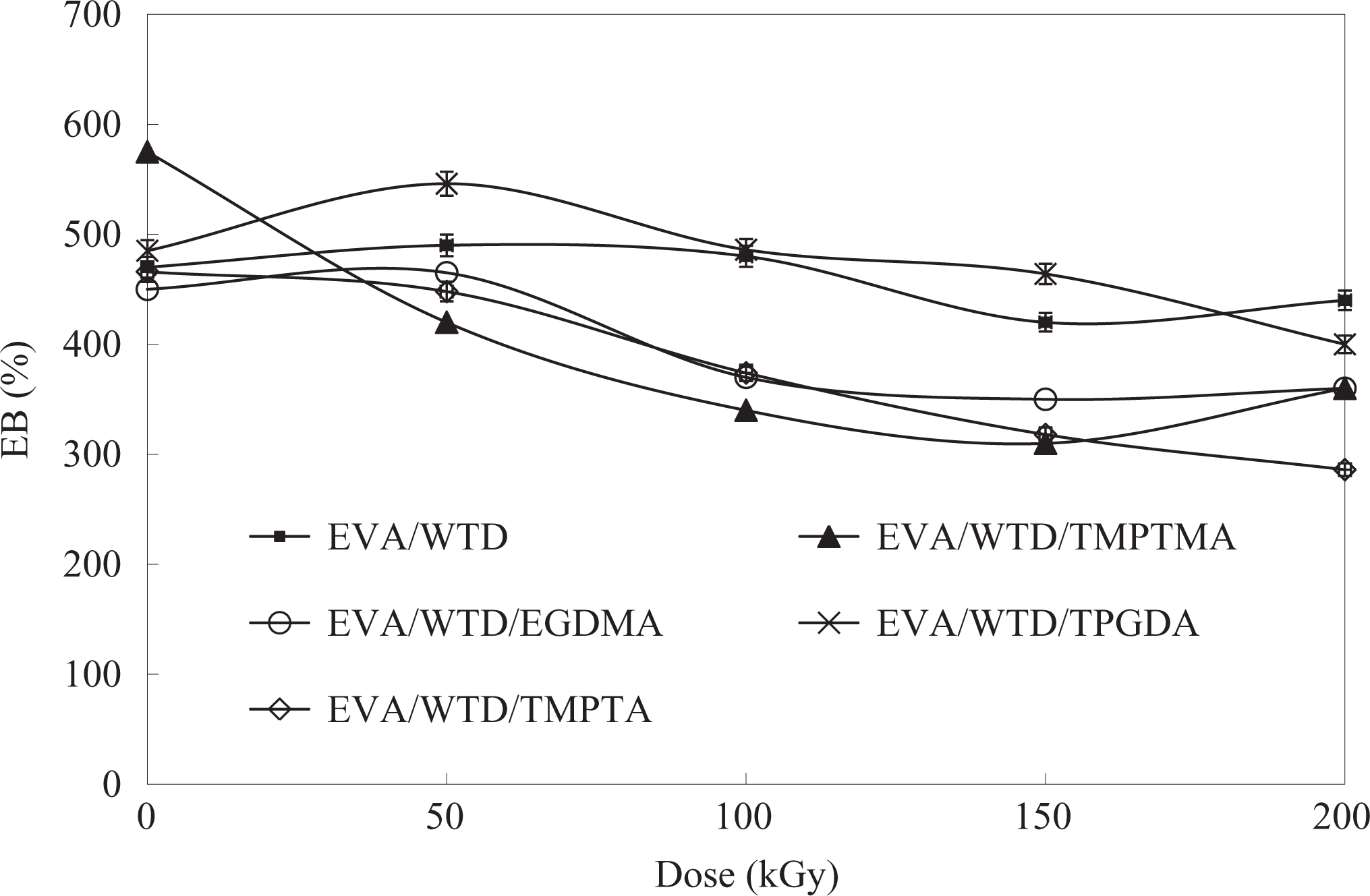
Effect of irradiation on the EB of EVA/WTD blends. EB: elongation at break; EVA: ethylene vinyl acetate; WTD: waste tire dust.
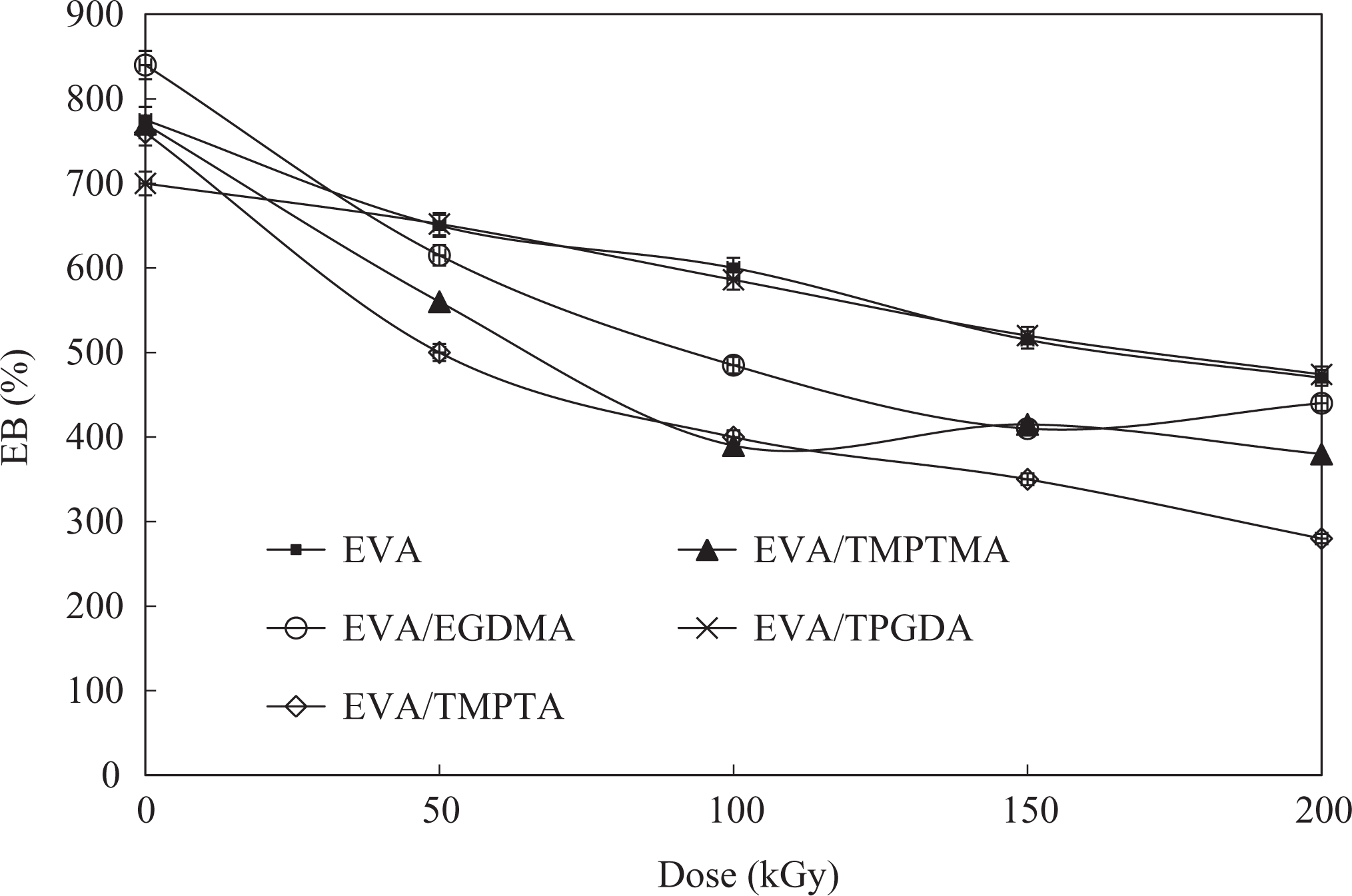
Effect of irradiation on the EB of EVA compounds. EB: elongation at break; EVA: ethylene vinyl acetate.
Gel fraction
Generally, the polymers which cross-links upon irradiation will form insoluble gel and the gel fraction are often used to indicate the changes in cross-link density of polymers upon irradiation. 13 Figures 5 and 6 illustrate the variation in the gel fraction of EVA/WTD blend and EVA compound upon exposure to EB irradiation in the presence of MFA. The higher gel fraction observed for the EVA/WTD blends and EVA compounds containing MFA provide fresh evident for the acceleration of irradiation-induced cross-linking by the MFA employed in this studies. The compounds incorporated with TPGDA exhibits lower gel fraction as compared to the counterpart-containing TMPTMA, EGDMA, and TMPTA. Such observation is attributed to the functionality of the MFA in which monomers with higher functionality will form more cross-link bridges that in return increases the cross-link density. TMPTMA, EGDMA, and TMPTA are trifunctional monomers that render higher degree of cross-linking as compared to TPGDA, which is a difunctional monomer. However, the TS and retention in EB obtained for EVA/WTD blends and EVA compounds in this study confirm that TPGDA render optimum cross-link density upon EB irradiation. The trends shown by the gel fraction results also imply that the EVA phase undergo cross-linking upon irradiation. 14
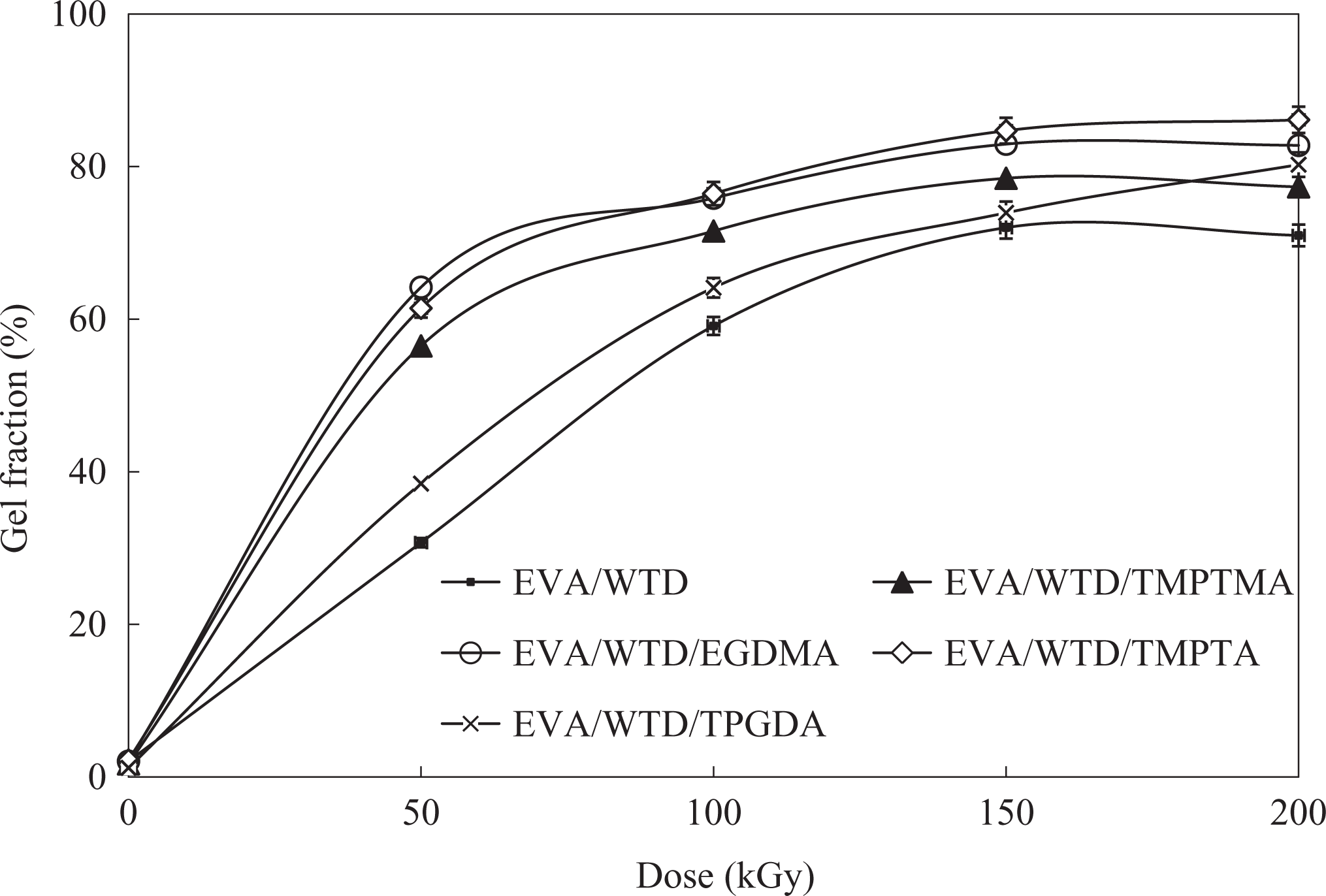
Effect of irradiation on the gel fraction of EVA/WTD blends. EVA: ethylene vinyl acetate; WTD: waste tire dust.
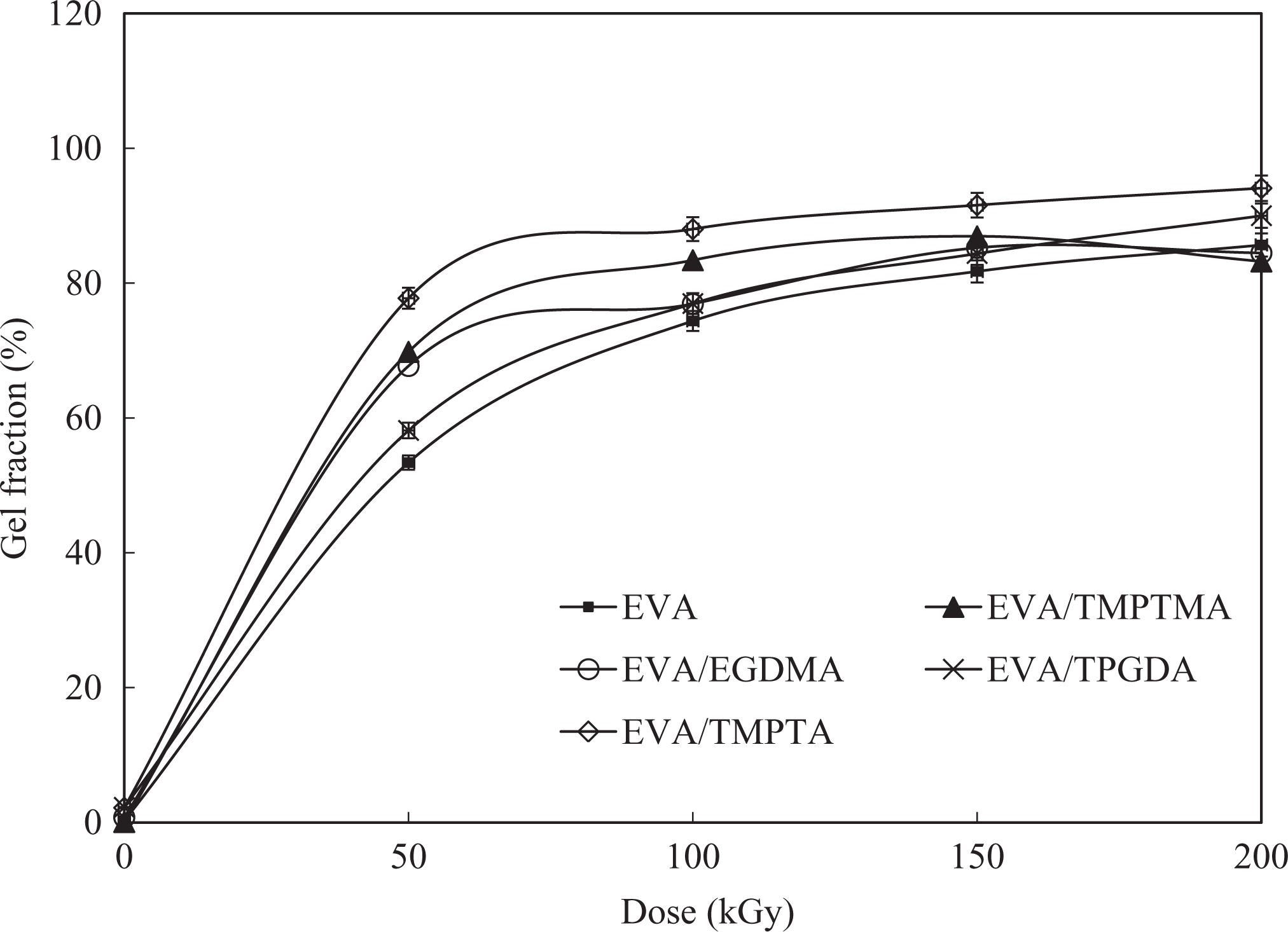
Effect of irradiation on the gel fraction of EVA compounds. EVA: ethylene vinyl acetate.
Differential scanning calorimetry
The thermal properties of 90/10 EVA/WTD blend, irradiated and nonirradiated blends, were analyzed by differential scanning calorimetry (DSC). Tm, ΔHf, and crystallinity of the blends are reported in Table 1 and Figure 7. The endothermic transition obtained for EVA represents the melting of the ethylene crystalline component in the EVA copolymer. As shown in Figure 7, from 45°C to 90°C, two overlapped endothermic peaks are observed in the DSC thermograms of EVA/WTD blends. The presence of two endothermic peaks, corresponding to the melting of two different crystallites present in EVA. The second peak (Tm2)at about 80 to 85°C is widely agreed to be the melting peak of ethylene segment, while the origin of Tm1 centred at about 50°C was attributed to the melting of less-ordered crystalline (imperfect crystals) phase formed in between the primary crystals during ambient storage or slow cooling. 15 The imperfect crystals are the result of incorporation of branching and vinyl acetate comonomers into the polyethylene crystal lattice.
Thermal properties of 90/10 EVA/WTD blends and the components at different irradiation doses.
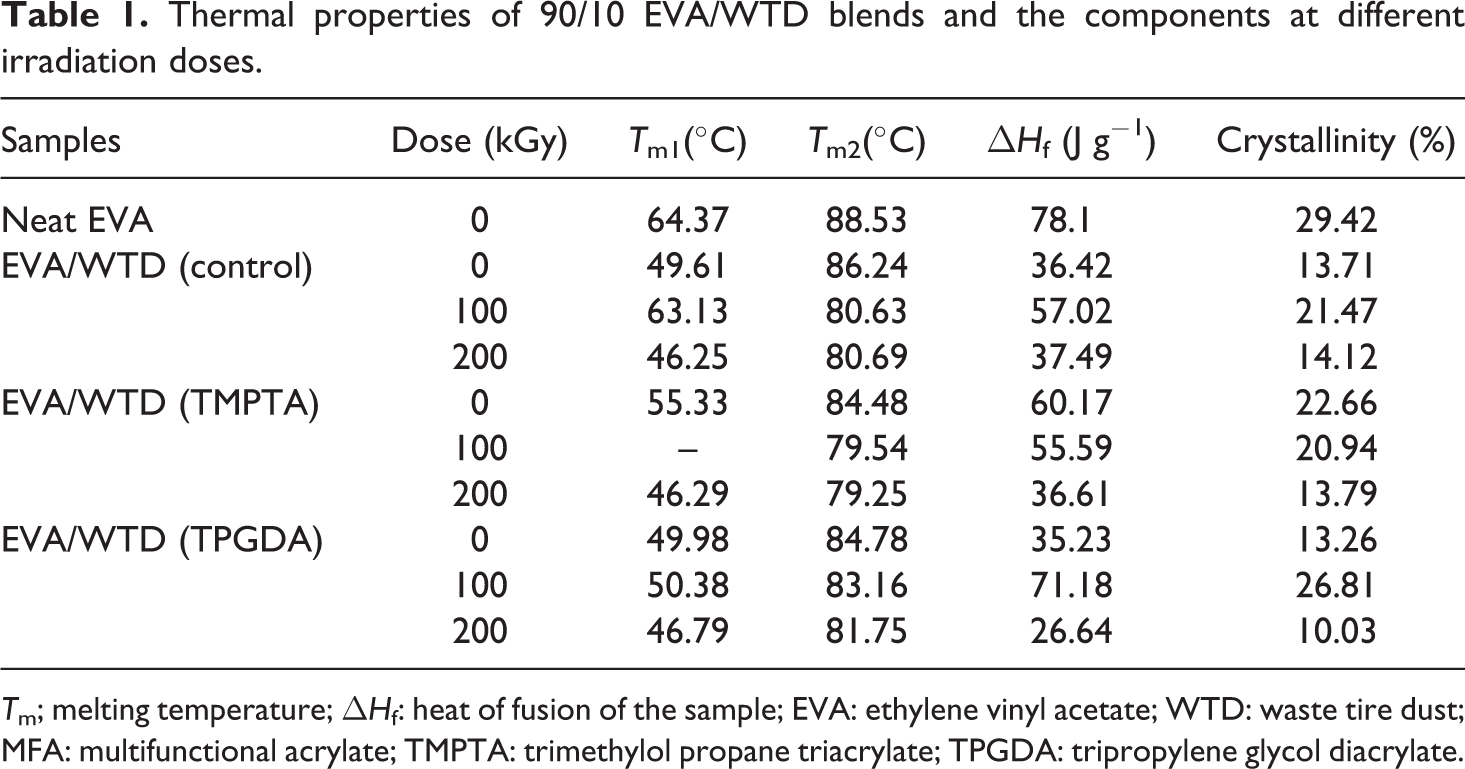
Tm; melting temperature; ΔHf: heat of fusion of the sample; EVA: ethylene vinyl acetate; WTD: waste tire dust; MFA: multifunctional acrylate; TMPTA: trimethylol propane triacrylate; TPGDA: tripropylene glycol diacrylate.
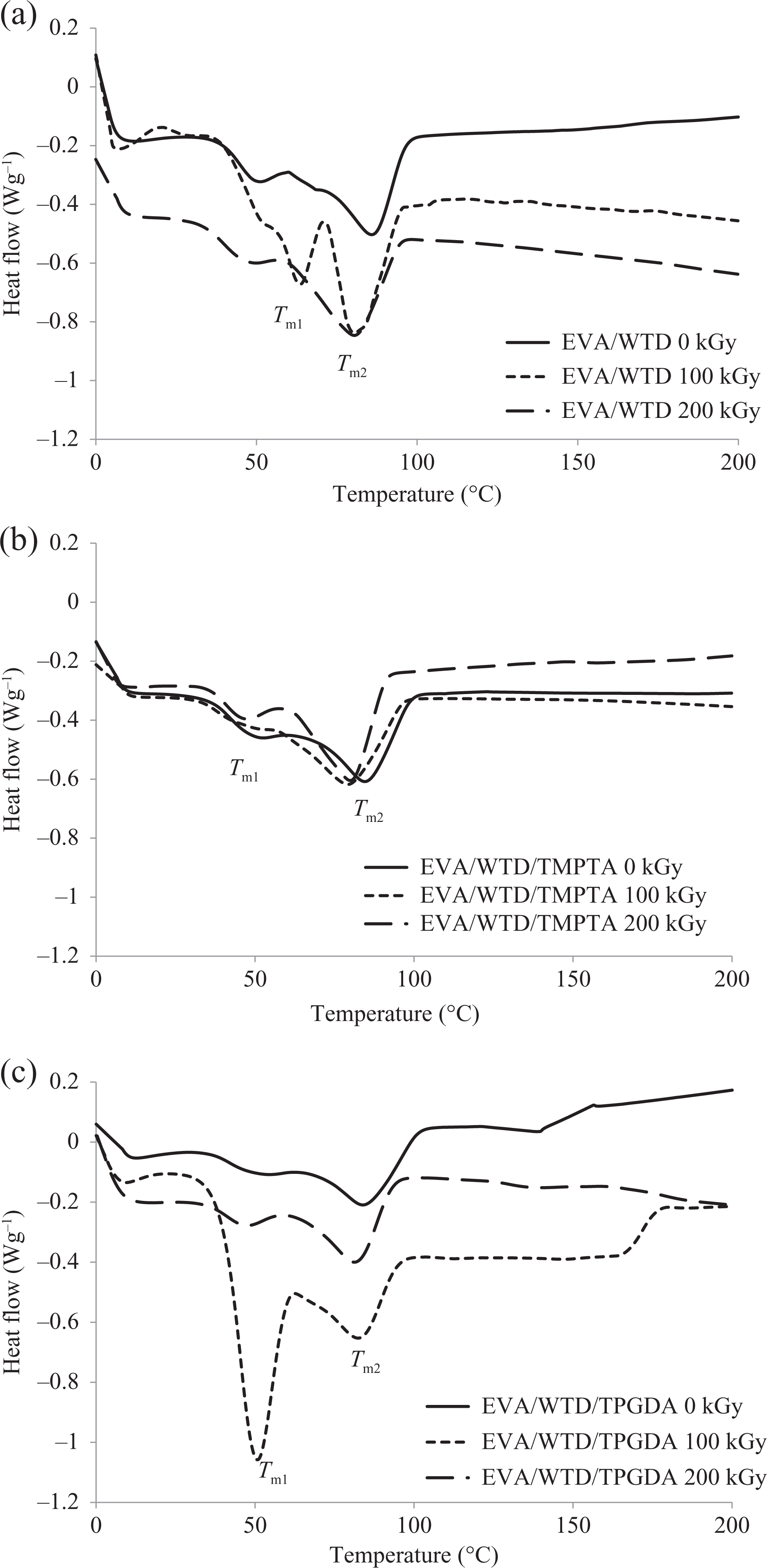
(a) DSC thermograms of EVA/WTD blends at different irradiation doses (without MFA: control). (b) DSC thermograms of EVA/WTD blends at different irradiation doses (with TMPTA). (c) DSC thermograms of EVA/WTD blends at different irradiation doses (with TPGDA). DSC: differential scanning calorimetry; EVA: ethylene vinyl acetate; WTD: waste tire dust; MFA: multifunctional acrylate; TMPTA: trimethylol propane triacrylate; TPGDA: tripropylene glycol diacrylate.
Apparently, Tm2 corresponds to the melting transition of the primary crystals in EVA, and Tm1 corresponds mainly to the melting transition of the secondary crystals formed in between the crystals in the EVA. This is due to the tendency of polyethylene domain to recrystallize. 16 Marcilla et al. 17 reported the similar DSC curve for EVA. The same phenomenon was reported by Przybytniak et al. 18 for PP/EVA.
Table 1 shows that addition of WTD to EVA caused a substantial decline in the crystallinity and crystalline Tms, implying the crystallization of EVA reduced with the presence of WTD. This observation is attributed to the noncrystallisable WTD that retards the crystal growth in EVA, which lead to imperfect crystals. George et al. 19 reported similar decrease in crystallinity of PP with the addition of nitrile rubber. Similarly, Koshy et al. 20 associated the decrease in the crystallinity of EVA/NR blend with increase in NR loading to the migration of NR phase into the interchain space of EVA. Thus, in the present study, it is believed that WTD-dispersed phase had a disrupting role in the arrangement of EVA chain molecules in a crystal lattice when cooling from melt; as a result, crystallinity reduced.
Table 1 shows that Tm2 decreases as the EVA/WTD blends experience irradiation, while the Tm1 and crystallinity show an increase at 100 kGy irradiation before exhibiting adecline at 200 kGy irradiation dose. Such observations indicate that the crystalline phase of EVA/WTD blend exposed to electron beam irradiation contains more defective crystals. Imperfection might be attributed to the occurrence of radiation-induced free radical reactions in the EVA that produce three-dimensional network, chain scissioning, side chain branches, and oxygen-containing groups. Radiation-induced free radical reactions occurs both at crystalline and amorphous region. The melting point temperature, Tm2 is reduced with increasing irradiation dose due to the resulting irregularities in ordered phase which are comprised series of ethylene units. In agreement with this, it had been reported that the crystallinity of rHDPE/WTD blends decrease due to γ6 and peroxide 21 cross-linking of the blends. Indeed, Reyes-Labarta et al. 16 had indicated that the crystalline phase of PP/EVA blends exposed to electron beam irradiation contains more defective crystals due to the presence of functional groups produced upon irradiation. 16
The higher crystallinity and Tm1 achieved at 100 kGy indicate the imperfect crystal growth in EVA. This observation is attributed to that higher mobility of free radicals in amorphous region which increases the recrystallization and rearrangement of short chain and long macromolecules produced by irradiation-induced cross-linking, grafting, scissioning, and oxidation. Besides that, the improvement in the adhesion between WTD and EVA matrix upon irradiation 5 also contribute toward the nucleation for crystallization occurring effectively. Lee and Kim 22 had reported the similar observation for EVA/carbon black blends.
However, at higher irradiation dose (200 kGy) the chain mobility is reduced due to excessive amount of cross-linking networks which are formed in the EVA matrix. This will restrict the chain recrystallization and rearrangement. Moreover, it has been reported that the presence of irradiation-induced cross-linking inhibits the process of crystal growth which changes the crystallinity and size of crystals. 23
The melting temperature and crystallinity for blends containing TPGDA exhibit similar changes with irradiation dose. However, at 100 kGy, higher primary crystal melting temperature, Tm2, and crystallinity are achieved for the blend incorporated with TPGDA compared with the control. The Tm of the imperfect crystal, that is, Tm1 was found to be lower than the control. This melting behaviour is described as the result of melting of larger, more regularly formed crystallites in the EVA/WTD blends upon irradiation in the presence of TPGDA. Such observation is attributed to the new EVA macromolecules that are formed upon irradiation in the presence of TPGDA which have higher chain mobility and flexibility. The increase in chain mobility and flexibility will enable easy arrangement of chain that favor perfect crystal growth. The higher EB obtained following irradiation of EVA/WTD blend with the incorporation of TPGDA compared with its control further support this observation. Similar observation was noted by Oral et al. 24 and Mohamad et al. 25 in their research of α-tocopherol/ultrahigh-molecular-weight polyethylene and ENR/EVA blends, respectively.
The addition of TMPTA to EVA/WTD blends renders somehow different melting behavior. It is interesting to note from Table 1 and Figure 7 that prior to irradiation, the EVA/WTD blend-containing TMPTA exhibit the highest Xc compared with the blend incorporated with TPGDA and control. This observation suggests that the nucleation for the crystallization occurs effectively with the presence of TMPTA as compared to TPGDA. However, the value of Xc of EVA/WTD blend-containing TMPTA found to show progressive drop with irradiation dose. The endothermic double peak between 50 and 90°C (0 kGy) has changed to a broad shoulder upon irradiation at 100 kGy with its maximum at 79.54°C. This change from peaks into a broad shoulder is an indication of a distribution of crystals with reduced thickness as a consequence of irradiation at 100 kGy. Such observation is attributed to the occurrence of irradiation-induced cross-linking in EVA in the presence of TMPTA which has introduced irregularities in ordered phase. 18 TMPTA is a highly reactive trifunctional monomer that renders higher degree of cross-linking as compared to TPGDA, which is a difunctional monomer. The presence of TMPTA favors more radiation-induced cross-linking over degradation or chain scissioning. Radiation-induced degradation in the presence of air produces oxygen-containing functional groups 26 that lead to imperfect crystal growth. Therefore, in the presence of TMPTA, the EVA/WTD blend did not exhibit secondary crystal melting peak as the blend undergoes predominantly cross-linking. The effect of TMPTA in the acceleration of cross-linking in polymer blends was well described by Ratnam and Zaman. 11 On the contrary, at 200 kGy, the melting peak corresponding to imperfect crystal, Tm1, found to appear. This implies that, at 200 kGy, the extensive free radical reactions that occurred in the presence of TMPTA has resulted in similar effect in crystallinity of EVA/WTD blend as observed for the control and TPGDA.
Morphological study
SEM was employed to study the cryogenically fractured surface of EVA/WTD blends. The morphological changes occurred upon irradiation of EVA/WTD blend in the absence and presence of MFAs are shown in Figure 8(a) to (f). All the samples show similar fracture feature; however, one can see that addition of MFA and exposure to irradiation produces modification at the matrix surface. The unirradiated blend shows ductile-type failure with high extent of plastic deformation as evidenced by the presence of irregular discontinues fracture paths. It is apparent that all irradiated EVA/WTD blend had undergone brittle failure compared with their nonirradiated counterpart. The reduction in the formation of irregular fracture paths and smooth fracture surfaces upon irradiation of the EVA/WTD blends indicates brittle failure of the compounds. The brittle failures are due to embrittlement of the material as a result of high extent of cross-linking by electron beam irradiation at high dosage (200 kGy). 27

SEM micrographs of fractured surfaces of EVA/WTD compounds:(a) without MFA, 0 kGy; (b) without MFA, 200 kGy; (c)with TMPTA, 0 kGy; (d) with TMPTA, 200 kGy; (e) with TPGDA, 0 kGy; and (f) with TPGDA, 200 kGy. SEM: scanning electron microscopy; EVA: ethylene vinyl acetate; WTD: waste tire dust; MFA: multifunctional acrylate; TMPTA: trimethylol propane triacrylate; TPGDA: tripropylene glycol diacrylate.
The micrograph of unirradiated EVA/WTD blend show some deep holes left after WTD particles were pulled out of the matrix. The holes proximity indicates that the WTD could not provide an efficient stress transfer from the matrix. Reduction in such holes and a more regular fracture paths observed for the irradiated EVA/WTD blends suggests the improvement in the adhesion between EVA matrix and WTD. It is also evident from Figure 8 that the EVA/WTD-containing TMPTA exhibit very thin waves of fibril and smoother surface compared to EVA/ WTD blend incorporated with TPGDA. Besides that in the presence of TPGDA (Figure 8(f)), EVA/WTD blend show some extent of plastic deformation in between that of Figure 8(b) and (d) indicating that the blend is essentially ductile although TPGDA caused acceleration in irradiation-induced cross-linking. This observation supports the tensile finding where the TPGDA found to render the highest tensile strength with good retention in EB upon irradiation.
Conclusions
Results revealed that among the MFA employed in this studies, TPGDA is found to render the highest TS with the best retention in EB upon irradiation. TPGDA also promoted increase in ΔHf and crystallinity of the blends. The observation on the TS, EB, and gel fraction of EVA indicated the changes in the physical properties of EVA/WTD blends upon irradiation associated with the irradiation-induced cross-linking of the EVA matrix.
Footnotes
Acknowledgements
The authors wish to thank all ALURTRON staff, Mr Wan Ali, and Mr MN Falah of Nuclear Malaysia for the assistance during the irradiation and laboratory facility usage.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present work was financially supported by the Ministry of Higher Education Malaysia (MOHE) under Fundamental Research Grant Scheme (FRGS/1/2012/TK04/IP-ANM/04/1) and Ministry of Science Technology and Innovation Malaysia (MOSTI) under Science Fund (03-03-01-SF0023).