Abstract
Polypropylene (PP)/nano-calcium carbonate (nCC) composites modified with methacrylic acid (MA) with and without dicumyl peroxide (DCP) were prepared by a twin-screw extruder. The effects of nCC and MA on the mechanical, thermal, and rheological properties of PP/nCC composites were investigated. The mechanical tests indicating that nCC could simultaneously reinforce and toughen PP. In addition, incorporation of MA further increased the mechanical properties of the composites. In the presence of DCP, a small amount of MA could increase markedly the mechanical properties of PP/nCC composites. Differential scanning calorimetry results indicate that the addition of nCC increased the crystallization temperature (T c) as a result of heterogeneous nucleation effect of nCC on PP and can induce β-phase PP. The addition of MA can further increase the T c of PP and the intensity of β-phase PP. The results of rheological property analysis indicated that the viscosity increased with increasing amount of the filler, especially at low shear rates. The addition of MA improved the dispersion of nCC resulting in the increasing of apparent viscosity.
Introduction
As one of the most important commodity polymers, polypropylene (PP) has been studied for many years. PP is a semicrystalline polymer. It has a number of advantages such as high strength, good processing properties, and low cost and is usually used in industries for production of articles for daily use. 1 But its drawbacks such as high shaping shrinkage and bad impact toughness are also outstanding. Therefore, the modification and toughening PP has become one hot point of studies in polymeric materials in recent years. Several methods for toughening PP have been proposed. In order to increase the impact strength of semicrystalline polymers, the incorporation of elastomeric particles has been widely used with great increases in the impact resistance but accompanied of losses in the elastic modulus. 2 An alternative for the incorporation of elastomeric particles is the incorporation of debonding rigid particles that are able to increase the impact resistance with the advantage of increasing the elastic modulus of the matrix. 3 In this context, the incorporation of inorganic rigid nanoparticles have been of much interest in the field of science because of the possibility of low-use contents of fillers with great increases in both impact and tensile mechanical properties.4 –7 In various inorganic rigid nanoparticles, calcium carbonate (CaCO3) is one of the most frequently used fillers for PP and is usually used owing to the characteristics such as high strength, abundant source, and low cost. With the development of nanotechnology, new polymeric composites filled with nanofillers have been rapidly developed.8 –13 The mechanical properties of polymeric materials depend on the crystallization behavior of the polymeric matrix, so there were some reports on the crystallization behavior of PP filled with nanomaterials. The incorporation of fillers into PP can lead to changes in crystallization and melting behavior of PP. According to the nucleation ability of the filler for the crystallization of PP, the fillers can be classified into two kinds, active and inactive. The nucleating effect of CaCO3 on PP is drastically disputed, which relates to the crystal form, surface topography, and dispersion of CaCO3. The addition of CaCO3 with different crystal form and surface topography into PP results in various changes in the crystallization morphology, the proportion of crystallization phase to amorphous phase, and the distribution of these two phases, which accordingly affects the mechanical properties of PP/CaCO3 composites. Therefore, many studies on the crystallization and melting behavior of PP/CaCO3 composites have been reported. There were reports that the addition of CaCO3 markedly improved the nucleation effect and the crystallization temperature (T c) of PP attributed to the heterogeneous nucleation effect of CaCO3. 14,15 The increase in T c was related to the particle size and content of CaCO3: the smaller the size, 16 the larger the amount of CaCO3 17 and the more significant the improvement in crystallization behavior. Nano-CaCO3 (nCC) significantly improved the nucleation of PP, which is different with micro-CaCO3.Wang et al. 18 studied the effect of nCC on the crystallization property of PP. The onset T c of PP was increased slightly with increasing the nCC content, and the heat of fusion initially increased and then fell. For the nCC content below 3.5 wt%, nCC dispersed well in the PP matrix and reinforced the interface interaction between the matrix and the nCC, which significantly induced the heterogeneous nucleation of PP and increased the degree of crystallization. For the nCC content above 3.5 wt%, the heterogeneous nucleation effect and the degree of crystallization of PP decreased because of the severe aggregation of nCC. The heat of fusion of PP in its composites filled with nCC was higher than that of the composites filled with micro-CaCO3 as a result of the high heterogeneous nucleation effect of nCC. The melting behavior of PP/nCC composite was also investigated by Tao et al. 19 For the pure PP, a melting peak of the α-crystal was observed. For PP/nCC, there was not only a melting peak of the α-crystal at 165°C but also a small peak at 150.6°C, which corresponded to the β-crystal. It was suggested that the nCC induced the formation of a few β-crystal of PP. The smaller the nanoparticles, the greater the heterogeneous nucleation effect of β crystallization. However, rheological effects of the incorporation of the methyl acrylate to the PP/nCC blends are a subject that has not been discussed in depth.
Viscoelasticity is a key parameter indicating the use properties and processability of these materials. It is very important to understand the rheological properties of polymer melts for processing and equipment or mold design. Rheological properties of CaCO3-filled polymers have been documented for the past 20 years.20 –23 For polymer blends, the factors affecting the melt shear viscosity are more and complicated, such as compatibility between phases, blending ratio and the viscosity difference between components, and so on.24 –28 One can get more information on macromolecular structure–rheological property during polymer melt flow with shear low rate levels, such as zero-shear viscosity. However, there have been still no reports about using methacrylic acid (MA) as a surface modifier for nCC and on the melt shear viscosity of MA and its blends at low shear rate level. In this study, PP/nCC composites modified with MA were prepared using the melt process. The effects of nCC and MA on the mechanical properties and the crystallization and melting behavior of PP/nCC composites are discussed and also to investigate the flow behavior and its mechanisms of PP and PP/nCC blend melts at low shear rates.
Experimental details
Materials
The grade of PP used in this work was SEETEC Homo polymer PP by LG Chem (Seoul, South Korea). This acts as the matrix. The homopolymer PP has a density of 0.90 g cm−3 and a melt flow rate of 14 g/10 min (2.16 kg at 230°C). The nano-CaCO3 used in this study was kindly supplied by SK Corporation (South Korea); its density was 2.71 g ml−1, and its particle diameter was between 40 and 60 nm. Chemical grade MA, used as the reactive monomer, was purchased from SK Corporation (South Korea). Chemical grade dicumyl peroxide (DCP), used as an initiator for the grafting reaction, was a commercial product of SK Corporation (South Korea).
Methods
Preparation of PP/nCC composites
The composition of nanocomposites is shown in Table 1. Unmodified nano-CaCO3 (nCC) is prepared by drying its water suspension in an oven at 100°C to constant weight. The MA and DCP were added when 10 wt% nCC and 90 wt% PP pellets were blended in a GH-10 high-speed mixing machine (Plastic Machine Factory, Beijing, China). The contents of MA were 2, 3, and 5 wt% and the dosages of DCP were 0.1 wt%. The mixture was then extruded by a corotational twin-screw extruder (model: PLE-331; Brabender Plasticorder, Germany). Figure 1 shows the mixing chamber capacity with 30 ml. The processing temperature, rotor speed, and blending time were set at 180−220°C, 60 r min−1, and 10 min, respectively. The sample weight of each blending was controlled at 60 g. After 10 min, the mixing chamber of the Brabender apparatus was opened and the resulting mixture was taken out. The resultant mixture was compression molded in a hot press at 190°C for 5 min without any applied pressure. After this period, 10 MPa pressure was applied for 5 min, and then the press platelets, containing coils for fluids. Finally, the pressure was released, and the mold was removed from the plates. The resulting sheets were quenched immediately to room temperature in air compression. The samples were cut into standard shapes and sizes for testing mechanical properties.
Composition of PP/nCC composites.
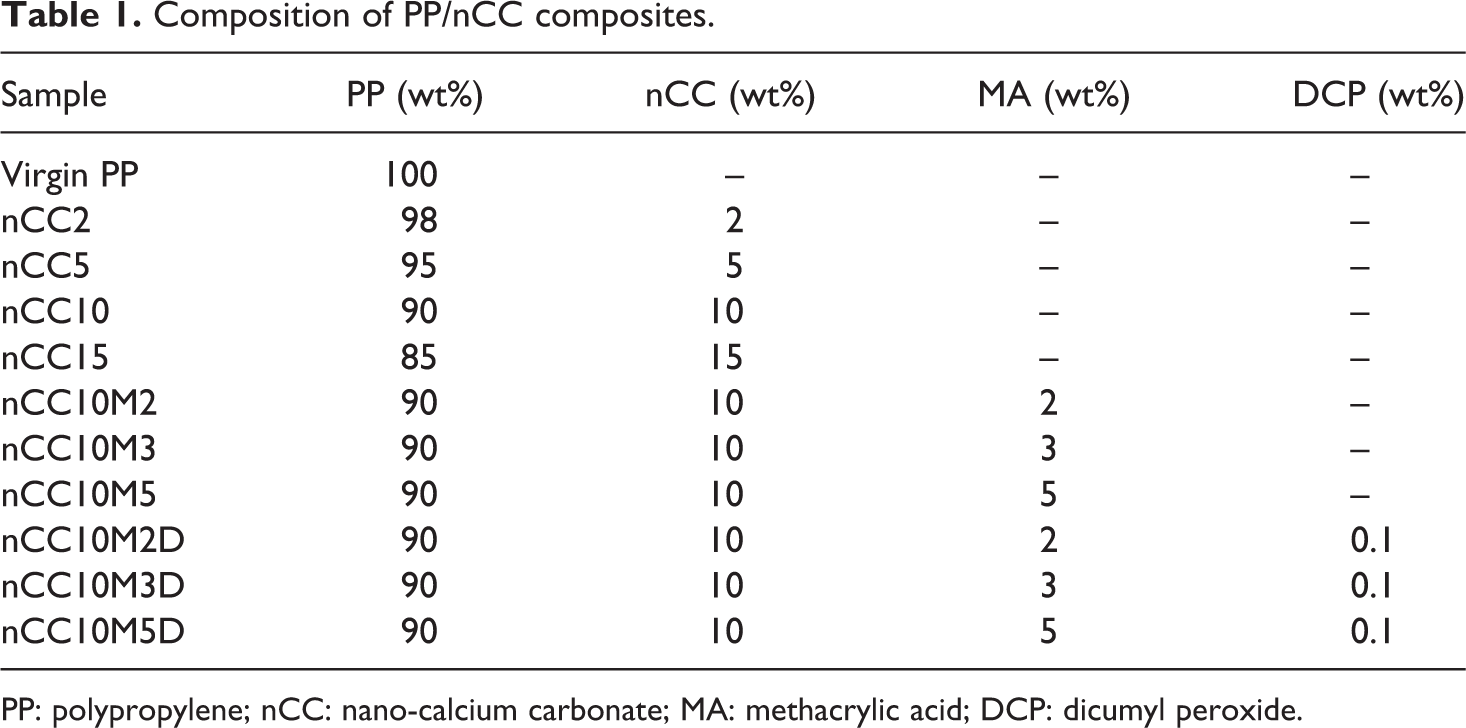
PP: polypropylene; nCC: nano-calcium carbonate; MA: methacrylic acid; DCP: dicumyl peroxide.

(a) General view of Brabender Plasticorder PLE 331. (b) The mixing chamber and the rotors after the mixing process of the composite.
SEM analyses
Studies on the morphology of impact-fractured surfaces of samples were carried out using a scanning electron microscopy (SEM), model JSM-5900LV (JEOL, Tokyo, Japan). All samples were immersed into liquid nitrogen for 0.5 h. Then, the samples were fractured. The fracture ends of the specimens were mounted on aluminum stubs and sputter coated with a thin layer of gold to avoid electrostatic charging during examination and observed at an accelerating voltage of 10 kV.
Determination of mechanical properties
Mechanical properties of virgin PP and PP/nCC composites were studied in tensile strength, tensile modulus, and impact strength. Standard specimens were sampled from the compression-molded sheet and then conditioned at the temperature of (25 ± 2°C) and the relative humidity of 50 ± 5% for 24 h. Tensile tests were conducted in an Instron materials testing machine (model 4206, USA). The width and thickness of the specimens were 13 mm and 3.20 mm, respectively. The speed of crosshead was 50 mm min−1. To measure the Young’s modulus of the materials, an extensometer was used with a tensile rate of 1 mm min−1. Izod impact tests of notched samples were carried out according to ASTM D256-93a standard, the instruments was Ceast pendulum impact tester (model 6545/000, Italy). The dimension of the specimens (length (L) × width × thickness) was 63.50 × 13 × 3.20 mm3.
Density measurement of nanocomposites
The swelling ratios of both PP and the nanocomposites were determined by immersing the samples in toluene to swelling equilibrium. The swelling ratio (S) was calculated according to equation (1).
where w t is the weight of swelled sample at t hours and w 0 is the weight of nonswelled sample.
The density of PP and the nanocomposites was calculated according to equation (2). The samples were weighed in balance (0.1 mg) in air and in coal oil, respectively.
where ρ is the density of sample; ρ i is the density of coal oil, w a is the weight of sample and filament in air, w f is the weight of filament, and wi is the weight of sample and filament in coal oil.
DSC analyses
The melting and crystalline behaviors of virgin PP and samples were measured with a Perkin Elmer DSC-7 (Perkin Elmer Inc., Wellesley, Massachusetts, USA). First, virgin PP and samples were heated from 25 to 250°C for 10 min to eliminate all of the thermal history in the materials. Then, the samples were cooled to 25°C at a cooling rate (R) of 10°C min−1 to obtain their crystalline characteristics. Finally, the samples were heated to 250°C at a heating rate of 10°C min−1 to obtain their melting characteristics. The temperature and heat capacity scales were calibrated from the melting scans of high-purity indium and zinc samples at the same heating rate. All measurements were carried out under nitrogen atmosphere environment. The sample weight was in the range of 5−8 mg.
Capillary rheological measurement
Capillary rheological properties were investigated with a capillary rheometer (RH2000, Rosand Precision, UK) at 180°C with shear rates varied from 20 to 3000 s−1. The ratio of length to diameter (L/D) and the diameter of the capillary were 35 and 1 mm, respectively. The samples were preheated for 5 min at a test temperature before measuring. The apparent shear viscosity (η) and the apparent shear rate (γ) were calculated from the experimental data. The flow curves were established using the corrected values of shear stress and shear rate based on Bagley and Rabinowitsch methods. 29
Results and discussions
Mechanical properties of PP/nCC composites
Mechanical property tests were performed on virgin PP and PP/nCC unmodified with nCC loadings varying from 0 to 15 wt% and modified with different MA contents in the presence of DCP. The testing results for the mechanical properties are listed in Table 2. It has been reported 30 that the mechanical properties of composites depend on the characteristics and interaction between the composition components. It can be seen that tensile strength (TS) and tensile modulus (TM) of PP are increased when nCC is added, and their TM values rise but TS values fall with the increase in CaCO3 content. With increasing concentration of untreated nCC, the TS of PP/nCC composites decreased. This is attributed to the poor compatibility between PP and nCC. The nCC particles had polar, hydrophilic surfaces and high surface free energy, which was incompatible with nonpolar, hydrophobic, and low free energy PP; they thus could not be wetted by the molten matrix. The addition of untreated nCC in the PP matrix induced loss of stress concentrators, which led to negative effects on the mechanical properties. Thus, the TS of PP/nCC composites decreased. These results are consistent with the results of the research study carried out by Li et al. 31 and Doufnoune et al. 32 By contrast, outstanding effectiveness of MA-treated nCC increasing the TS of PP composites was demonstrated. As the concentration of the filler increased in the composites filled with MA-treated nCC, the TS was first remarkably increased and then decreased. When the concentration of MA-treated nCC was 2%, the maximum TS obtained was 39.9 MPa, which was 1.42 times greater than that of the virgin PP. It is found that the composite modified with MA in the presence of DCP has slightly decreased TS values. With the addition of untreated and MA-treated nCC, the TM and impact strength (IS) of PP/nCC composites increased, indicating that the nCC could increase the TM and IS of PP. 33 The values of TM and IS of the PP/nCC composite with 2−10 wt% nCC addition are better than those of PP. The TM and IS of the PP/10 wt % nCC composite increased about 36 and 61%, respectively with respect to those of virgin PP. In order to improve the dispersion of the filler in the polymeric matrix and increase the interfacial adhesion between filler and matrix, MA is introduced to modify PP/nCC composites. After PP/nCC composites are modified with MA, the values of TM and IS are better than those of unmodified PP/nCC composites and virgin PP and increase with the increase in MA content. When the incorporation of MA is 5 wt%, TM and IS values of PP/nCC with 10 wt% filler content are increased by 9 and 8%, respectively, and their TM and IS are increased by 48 and 75%, relative to those of virgin PP. The TM of PP/nCC composites modified with MA is also better than those of unmodified PP/nCC and virgin PP, especially at higher filler content. IS values of PP/nCC modified with MA are greater than those of unmodified composites at the same filler content. With the increase in the MA content, the greater the IS of the modified composites, which can be attributed to the synergism of MA and nCC on the PP/nCC composites. It is believed that MA can cover or pack nanoparticles, therefore reduce the surface energy of nanoparticles and improve the dispersion of nCC in PP. On the other hand, as a reactive polar small molecule, MA is prone to diffuse into the agglomerates of nanoparticles and prevent them from agglomeration. With the action of high temperature and shear stress during the process, MA can homopolymerize or graft to PP, consequently improving the interfacial action between the nanoparticles and the matrix, and raising the TM and IS of the composites. In order to promote the reaction ability of MA and PP, the initiator DCP is added. It is noted that the mechanical properties of composites modified with 2 wt% MA in the presence of DCP are best at the same filler content. With the increase in MA content in the presence of DCP, MA tends to homopolymerize and form poly(methyl methacrylate), which perhaps decreases the mechanical properties of the composites.
Mechanical properties of virgin PP and PP/nCC unmodified and modified with different MA contents in the presence of DCP.
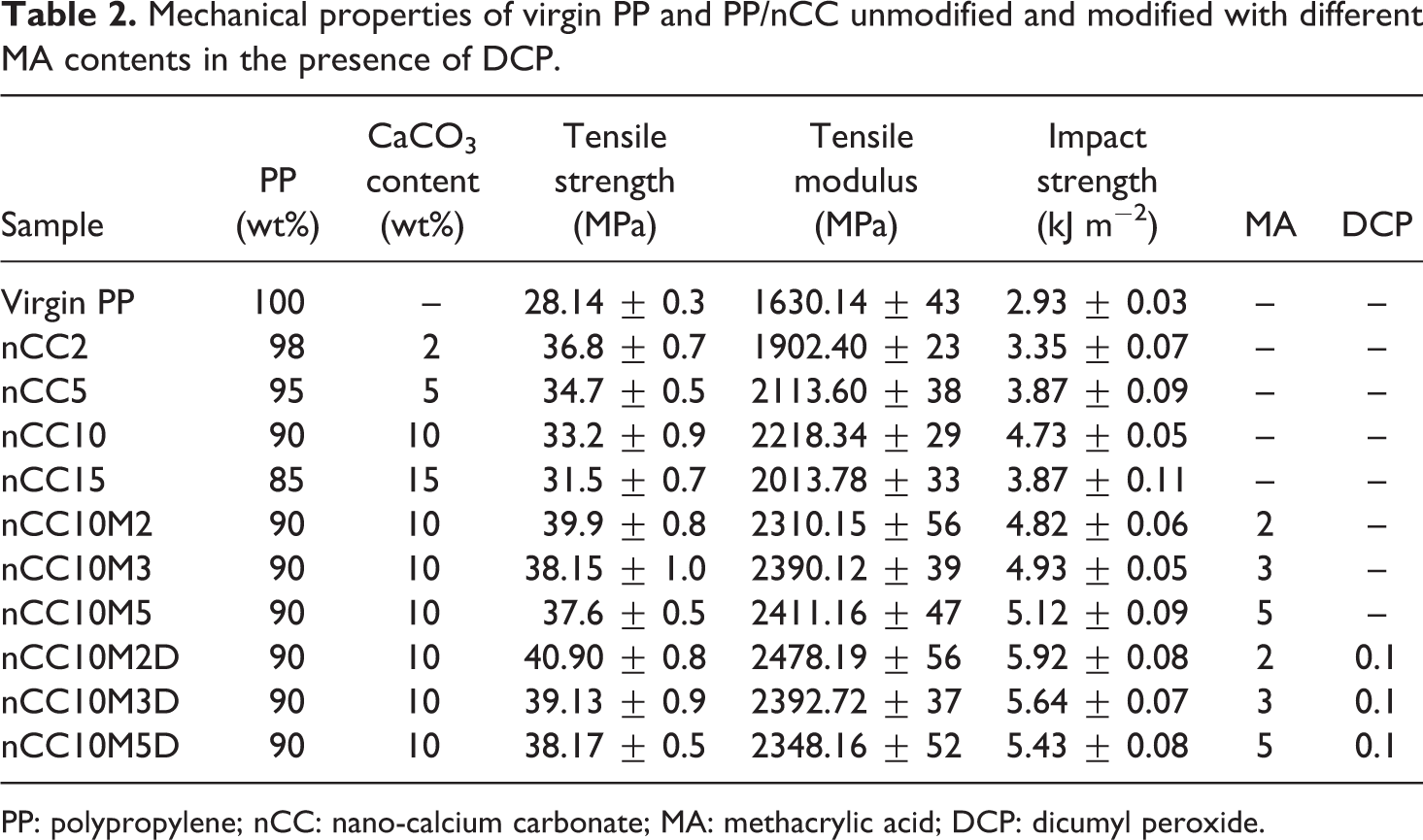
PP: polypropylene; nCC: nano-calcium carbonate; MA: methacrylic acid; DCP: dicumyl peroxide.
Dispersion of nanoparticles in PP matrix
Figure 2(a) to (c) presents SEM micrographs of the impact-fractured surface of nCC10, nCC10M5, and nCC10M2D composites, respectively. The particles of nCC10 were dispersed in the matrix of PP irregularly, and a lot of particles agglomerate together and lead to bigger cavities around the agglomerates with a particle size >3 μm (Figure 2(a)). By observing these aggregations, we found that they were formed of many primary particles. The nCC10 sample had a large particle size of 3−5 μm. From an analysis of the mechanical properties, we determined that this composite exhibited impact energy higher than that of virgin PP and much lower than that of the nCC10M5D composites. Figure 2(b) and (c) presents photographs of the impact-fractured surface of nCC10M5 and nCC10M2D composites. The results show that the broken surface morphologies of nCC10M5 and nCC10M2D composites were different. The broken surface morphology of nCC10M5 composite was more irregular than that of the nCC10M2D composite, and the average particle size was larger than that of nCC10M2D composite. When the surface of nCC was covered with MA, it could disperse uniformly in the PP matrix for nanoparticles, aggregation could be avoided, and the adhesion between the filler and resin was enhanced by the effect of MA. In addition, the incorporation of nCC10M2D in PP matrix was more homogeneous and the cohesive force between them was stronger. Obviously, MA-treated nCC in the presence of DCP improved the compatibility between the PP matrix and nCC.

SEM photographs of the impact-fractured surfaces of (a) nCC10, (b) nCC10M5 and nCC10M2D composites. SEM: scanning electron microscopy; nCC: nano-calcium carbonate.
Table 3 indicates that the density of nCC10M2D composite is lower than nCC10 composite, suggesting the existence of more cavities in nCC10M2D composite. Results of equilibrium-swelling experiments as shown in Figure 3 are consistent with density determination and SEM observation. The equilibrium-swelling ratio of nCC10M2D composite is higher than that of nCC10 composite, which suggests that the cavitations increased in nCC10M2D composite. These experiments prove that there are more and finer cavities in nCC10M2D composite, which is one of the reasons for improved toughness and other mechanical properties.
Density of PP and its nanocomposites.
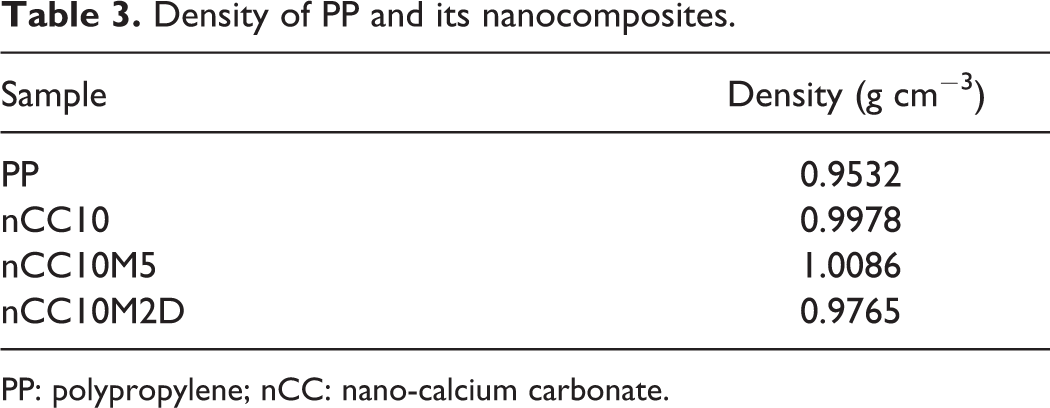
PP: polypropylene; nCC: nano-calcium carbonate.
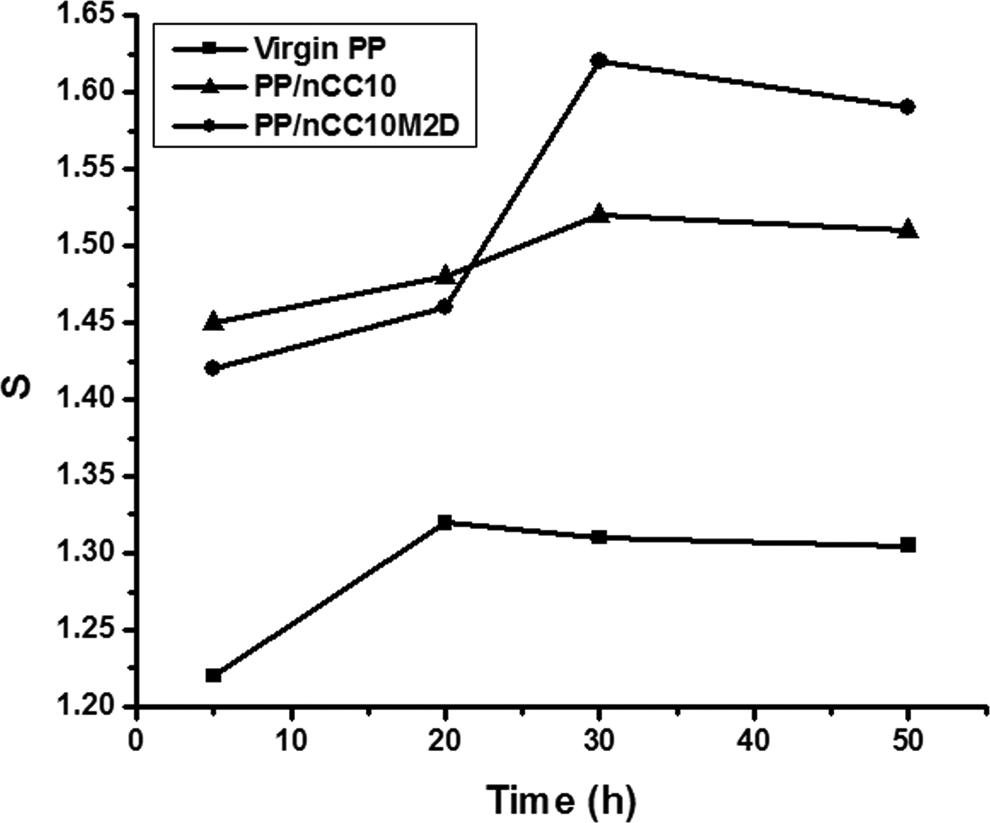
Swelling curves of polypropylene and its composites.
Crystallization and melting behavior of PP/nCC composites
The crystallization behavior of PP significantly influences its mechanical properties. Therefore, investigation of the crystallization behavior is significant both from the theoretical and practical points of view. Table 4 shows the DSC data for virgin PP and the PP/nCC composites modified with MA in the presence of DCP. It can be seen from Table 4 that when the content of nCC is only 2 wt%, T c is increased to 116.5°C, then changes little with increasing nCC content. The heat of crystallization and fusion are also increased when only 2 wt% nCC is incorporated, and then has little change. The greater the fusion heat, the greater the crystallinity, so it is indicated that the incorporation of nCC promotes the crystallization of PP and increases the crystallinity of PP, but in general it does not change the melting temperature (T m). The degree of supercooling (▵T) is related to the rate of crystallization, and the lower ▵T is, the more rapid the rate of crystallization. It is clear that ▵T decreases after nCC is added. The DSC results indicate that nCC has a significant heterogeneous nucleation effect on PP, which is also, confirmed by SEM examination.
DSC crystallization and melting behavior results of virgin PP and PP/nCC composites.
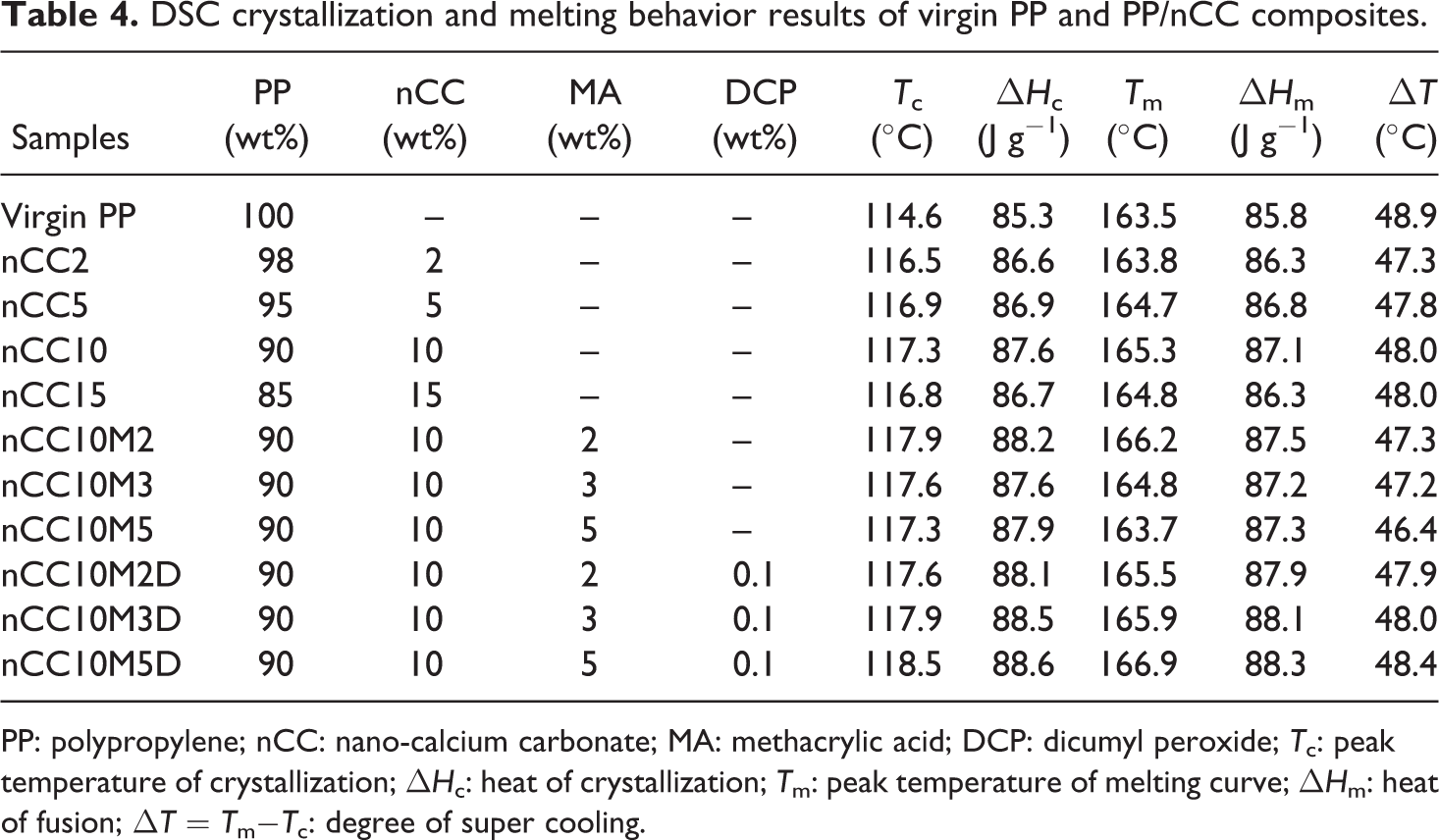
PP: polypropylene; nCC: nano-calcium carbonate; MA: methacrylic acid; DCP: dicumyl peroxide; T c: peak temperature of crystallization; ▵H c: heat of crystallization; T m: peak temperature of melting curve; ▵H m: heat of fusion; ▵T = T m−T c: degree of super cooling.
It can be observed from Table 4 that the addition of MA increases the T c of PP in PP/nCC10 composites. The T c of PP in nCC10M2 is approximately 3.3°C higher than that of PP and 0.6°C higher than that in nCC10. Addition of MA slightly increased the T c of PP in PP/nCC10 composite and ▵T is decreased, which suggests that the addition of MA improves the crystallization ability of PP. Many studies had indicated that organic acid could act as a nucleating agent for PP. It is believed that the carboxylic group introduced onto the PP chain exhibits an enhanced heterogeneous nucleation effect. The T c of nCC10M3 is about 3°C higher than that of PP and 0.3°C higher than that in nCC10. It can be seen that the melting behavior of nCC10M2 is close to that of nCC10.
Table 4 shows the crystallization and melting curves of PP/nCC10 modified with different MA contents in presence of DCP. Compared with PP/nCC10 and PP, T c of PP/nCC10 modified with different MA contents in the presence of DCP is higher and ▵T is lower, especially at 2 wt% MA content. These results imply that in the presence of DCP, only a small amount of MA can increase the crystallization ability of PP. It is thought that MA can easily react with PP in the presence of DCP and form more MA-g-PP, which can facilitate the crystallization of PP. In the presence of DCP, the intensity of the peak is increased with the increase in MA, which is perhaps attributed to the increasing formation of β-phase PP with the increase in MA-g-PP.
Capillary rheological properties
The rheological properties of polymer materials are most often studied by capillary rheometer. The rheological behaviors of virgin PP and for the unmodified PP/nCC composites were examined by a capillary rheometer.
Effect of nano-CaCO3 content on viscosity
Figure 4 shows logarithmic plots of melt viscosity versus shear rate at 180°C for virgin PP and for the unmodified PP/nCC composites containing varies nCC contents. From Figure 4, it is observed that the viscosity of the composites decreases with increasing shear rate, and at high shear rates, the curves converge. It is also noted that the apparent viscosity was higher with higher nCC content. nCC granules are undeformational solid granules that have low fluidity. The addition of nCC granules into PP matrix enhanced the rigidity of the melt system and brought down the system energy simultaneously decreasing the fluidity. Meanwhile, as the nCC content increased, the space between granules became smaller and the interaction became stronger, so the agglomeration was easy. As a result, the apparent viscosity rose with higher nCC content. 34 The apparent viscosity decreased gently with increasing shear rate and lower nCC content. It is shown that, as expected, the filler increased the viscosity of the composite owing to an extension effect.
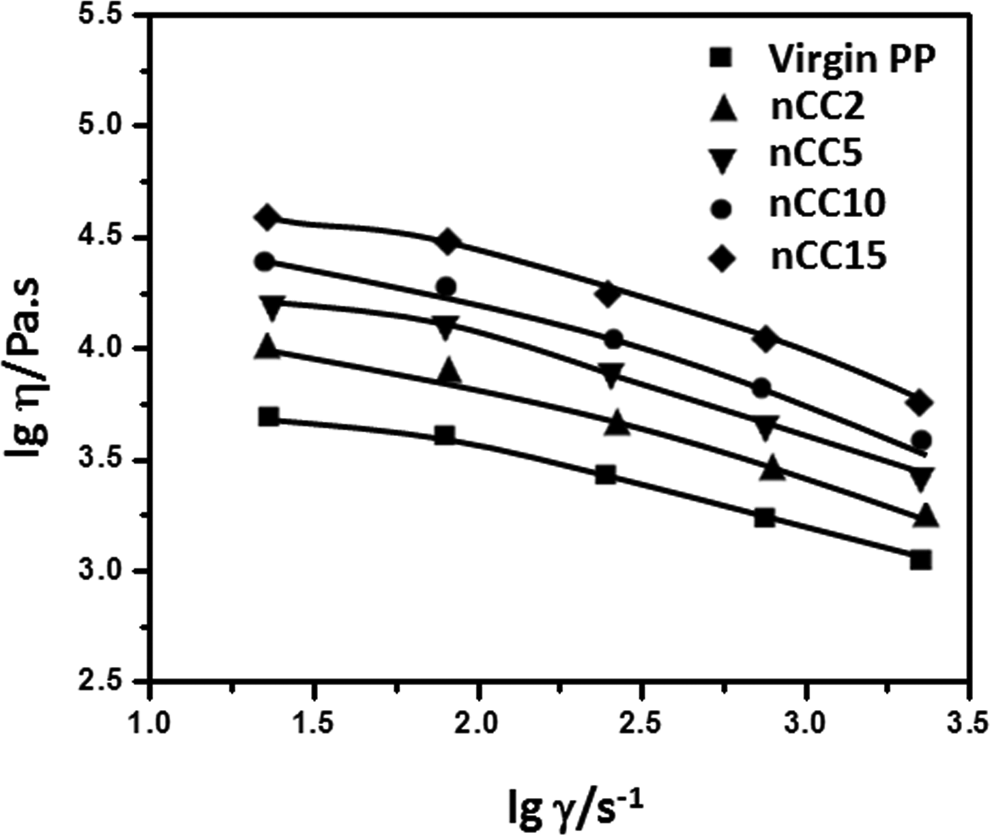
Melt flow curves of virgin PP and PP/nCC composites at 180°C. PP: polypropylene; nCC: nano-calcium carbonate.
Effect of MA content on viscosity
MA is an excellent reactive monomer and acted as a bridge between PP and nCC. Adding MA to the PP/10 wt% nCC composites can enhance the dispersion of nCC in the PP matrix due to the increase of fluidity. Figure 5 shows the effect of MA content on viscosity of the PP/10 wt% nCC composites. The apparent viscosity as a function of shear rate of the composites containing different contents of MA with 0.1 wt % DCP was tested at 180°C. It is evident that the melt viscosity decreases with an increase of the shear rate, indicating that the melts of all these samples belong to pseudoplastic fluids and have the flow behavior of shear thinning and conform to the equation of power law fluid. It is attributed to shear-induced change in molecular state, which is often described in terms of entanglement 35 among molecular chains. The entanglement density decreases with increasing shear rate, leading to shear thinning of the melt.
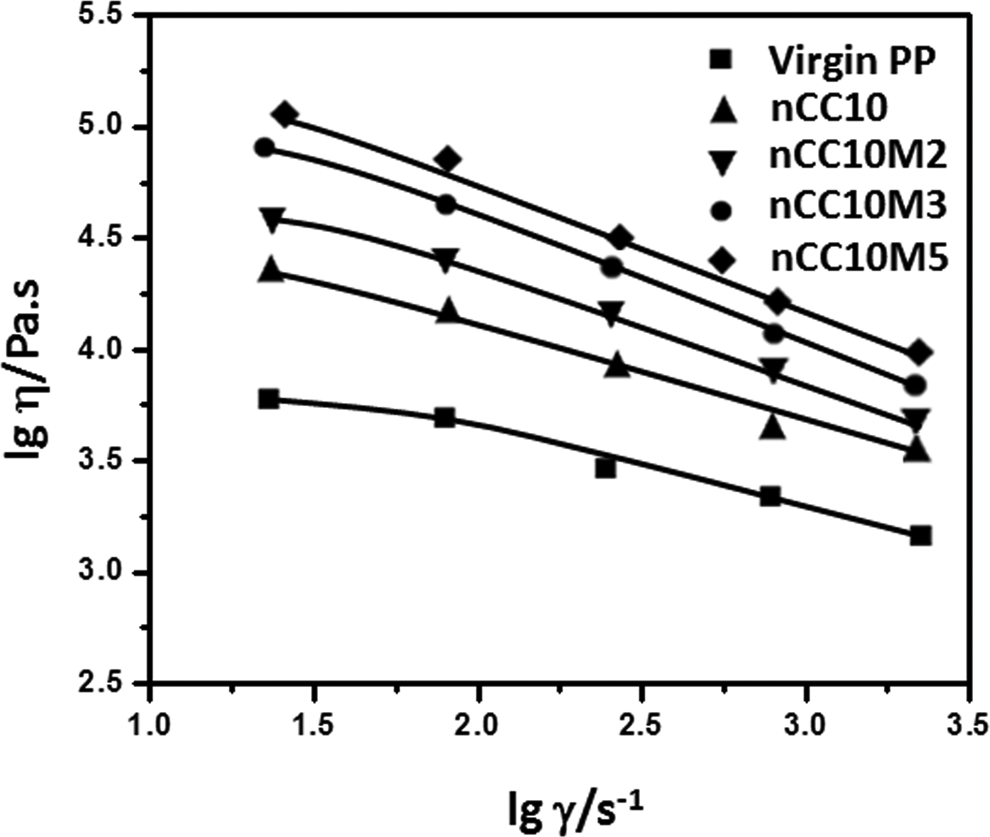
Melt flow curves of virgin PP and PP/10 wt% nCC composites treated with MA at different concentrations at 180°C. PP: polypropylene; nCC: nano-calcium carbonate; MA: methacrylic acid.
Conclusions
MA was used as a surface modifier for nCC, and its effects on the morphology, mechanical properties, crystallization and melting behavior, and rheological properties of PP/nCC composites have been investigated. SEM observation shows the surface treatment of nCC with MA improves the dispersion of the filler in the matrix. The mechanical tests indicate that nCC can simultaneously reinforce and toughen PP. In addition, incorporation of MA further increased the mechanical properties of the composites. In the presence of DCP, a small amount of MA could increase markedly the mechanical properties of PP/nCC composites. DSC results indicate that nCC has a heterogeneous nucleation effect on PP and can induce β-phase PP. The addition of MA can further increase the T c of PP. The capillary rheological test indicated that the melts of the composite behaves peudoplasticity. It was observed that the melt viscosity increased with an increase of the nCC content. The addition of MA improved the dispersion of nCC granule resulting in the increasing of apparent viscosity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
