Abstract
The focus of this study is to investigate the effects of oxidative functionalized carbon nanotubes (f-CNTs) and aminosilanized carbon nanotubes (s-CNTs) on the mechanical and thermal properties of polyamide 6 nanocomposites. Oxidation of nanotube surfaces was conducted with sulfuric acid/nitric acid mixture and then aminosilanization was carried out with γ-aminopropyltriethoxysilane. Nanocomposites were compounded by melt mixing technique and shaped by injection molding. Scanning electron microscopy images revealed that f-CNTs and s-CNTs were dispersed more evenly due to increased interactions with the matrix. Tensile tests indicated that yield strength and Young’s modulus of nanocomposites increased 20 and 23%, respectively, with the addition of only 1 wt% s-CNTs due to very efficient load transfer from the matrix to covalently bonded CNTs. Both dynamic mechanical analysis and thermogravimetric analysis showed that surface-modified CNTs improve all thermal properties due to decreased matrix mobility and physical barrier formation. For example, increases in the storage modulus values were as much as 25%, while the increase in the thermal degradation temperatures were as much as 5°C in the specimens with only 1 wt% s-CNTs.
Introduction
The concept of taking advantage of the extraordinary properties of carbon nanotubes (CNTs) has found considerable academic interest in nanomaterials science. Their unique atomic structure, high thermal conductivity, and unusual mechanical properties make CNTs ideal reinforcement material in the production of high-performance and multifunctional polymer nanocomposites.
Despite their superior properties, it is difficult to obtain uniform dispersion of CNTs throughout polymer matrix. This difficulty is related to the poor interfacial adhesion with polymer matrix and the inevitable bundle and entanglement formation because of strong van der Waals attraction between nanotubes.
Chemical functionalization of CNT surfaces represents an efficient way for the achievement of uniform dispersion and enhancement of interfacial adhesion. Oxidative functionalization using the mixture of nitric acid (HNO3)/sulfuric acid (H2SO4) is one of the most effective routes, which includes the incorporation of oxygen-containing groups, mainly carboxyl and hydroxyl. Silanization of functionalized nanotubes is another preferred method used to enhance the interfacial adhesion between the nanotubes and the matrix.
In the literature, there are several investigations on the functionalization and silanization of CNTs including our previous work, 1 which focuses on the use of various characterization techniques of oxidative functionalization and aminosilanization of CNTs. After functionalization and silanization, what is important is to study the effects of these surface treatments on the properties of polymer/CNT nanocomposites as discussed by several investigations. 2 –15 A few of these studies are summarized below:
Mukherjee et al. 2 investigated effects of vinyltriacetoxy silane-treated CNTs on the properties of polycarbonate (PC)/liquid crystalline polymer (LCP) system. They revealed that incorporation of silane coupling agent enhanced thermal stability and storage modulus of the PC/LCP system as much as 14 and 50%, respectively. They claimed that these results are due to the better dispersion and enhanced interaction of CNTs with the matrix system.
Yuen et al. 3 prepared polyimide nanocomposites with γ-aminopropyltriethoxysilane (APTES)-treated CNTs. They indicated that the glass transition temperature (T g) of polyimide increased from 281°C to 304°C when 4.8 wt% APTES-treated CNTs were used, while tensile strength of polyimide increased 26% with only 1.0 wt% APTES-treated CNTs.
Wu et al. 4 produced bisphenol A diglycidyl ether (DGEBA)-type epoxy matrix nanocomposites with 3-isocyanatopropyltriethoxysilane-treated CNTs. Their scanning electron microscopy (SEM) analysis revealed that silane-treated CNTs were well dispersed and embedded in the matrix, while tensile strength of epoxy increased to 41% with only 1.0 wt% of these CNTs.
Kim et al. 5 compared the effects of acid-treated and APTES-modified CNTs on the flexural properties of CNT/epoxy/basalt composites. They indicated that 1 wt% silane-treated CNTs lead to 10% higher flexural modulus, 14% higher flexural strength, and 40% higher fracture toughness compared to acid-treated CNTs.
Lee et al. 6 produced carbon/CNT/epoxy composites containing 1 wt% unmodified, acid-treated, and APTES-modified CNTs. It was shown that silane-modified CNTs resulted in 8 and 16% higher tensile strength compared to acid-modified and unmodified CNTs, respectively. For the storage modulus values, these increases were 14 and 61%, respectively.
Zhou et al. 7 investigated polypropylene matrix nanocomposites reinforced with 3-methacryloxypropyltrimethoxysilane-treated CNTs, and they concluded that using silane-treated CNTs lead to 4% higher tensile strength compared to untreated CNTs.
Jiang et al. 8 used bis(3-triethoxysilylpropyl) tetrasulfide as a silane coupling agent to modify CNT surfaces. Then, they developed natural rubber/CNT nanocomposites by solvent casting. It was revealed that using 3 wt% silane-modified CNTs resulted in 33% higher storage modulus compared with unmodified CNTs.
Although polyamide 6 (PA6) is one of the most significant thermoplastic polymers, to the best of our knowledge, there appears to be no work investigating the effects of CNT surface treatments on the properties of PA6 matrices. Therefore, the objective of this study is to contribute this field by revealing the influences of oxidative functionalization and aminosilanization of CNTs on the mechanical and thermal properties of PA6.
Experimental
Materials
In this study, the matrix material used was PA6 (EMS GriltexD1894A, Domat/Ems, Switzerland) having a melting range between 220 and 225°C and a density of 1.14 g cm−3, while the nanoreinforcement was multi-walled CNTs (MWCNTs; Nanocyl NC7000, Sambreville, Belgium) produced via catalytic carbon vapor deposition process. Oxidative functionalization was conducted with H2SO4 (97%, Sigma-Aldrich) and HNO3 (67%, Sigma-Aldrich, St Louis, Missouri, USA). For aminosilanization, APTES (99%, Sigma-Aldrich) and ethanol (99.5%, Sigma–Aldrich) were used.
Oxidative functionalization and aminosilanization of CNTs
For oxidative functionalization, unmodified (as received) CNTs were dispersed in acid solution (H2SO4/HNO3) and refluxed for 4 h in an ultrasonic bath at 50°C. This sample is named as oxidative functionalized carbon nanotubes (f-CNTs). For aminosilanization, f-CNTs were dispersed in APTES–water–ethanol mixture with ultrasonication. In order to conduct aminosilanization reaction, mixture is refluxed for 4 h at 70°C. This sample is named as oxidative functionalized and then aminosilanized carbon nanotubes (s-CNTs). Details of these procedures are given in our previous study. 1
Characterization of these f-CNTs and s-CNTs were conducted using x-ray photoelectron spectroscopy, Fourier transform infrared spectroscopy, SEM–energy dispersive x-ray spectroscopy, x-ray diffractometer, and thermogravimetric analysis (TGA), and the results were discussed in detail in our earlier study. 1
Compounding and shaping of the specimens
CNTs and PA6 granules were first premixed at 300 r min−1 for 5 min in a high-energy ball mill (RETSCH Pulverisette 6, Haan, Germany). Then, nanocomposites were produced by melt mixing method via laboratory size twin-screw extruder (Rondol Microlab 400, 10 mm, length/diameter ratio of 20, Staffordshire, UK) with a temperature profile of 185-220-230-225-205°C and a screw speed of 68 r min−1.
Prior to shaping via injection molding (DSM Xplore Micro, 12 cc, Geleen, The Netherlands), materials were dried under vacuum for 24 h at 80°C. During shaping, heating time was 4 min, while barrel and mold temperatures were 240°C and 70°C, respectively. PA6 nanocomposites were produced using three different loading contents for each CNT. Designation and composition of all these specimens are given in Table 1.
Designations and compositions (in weight percentage) of the specimens.
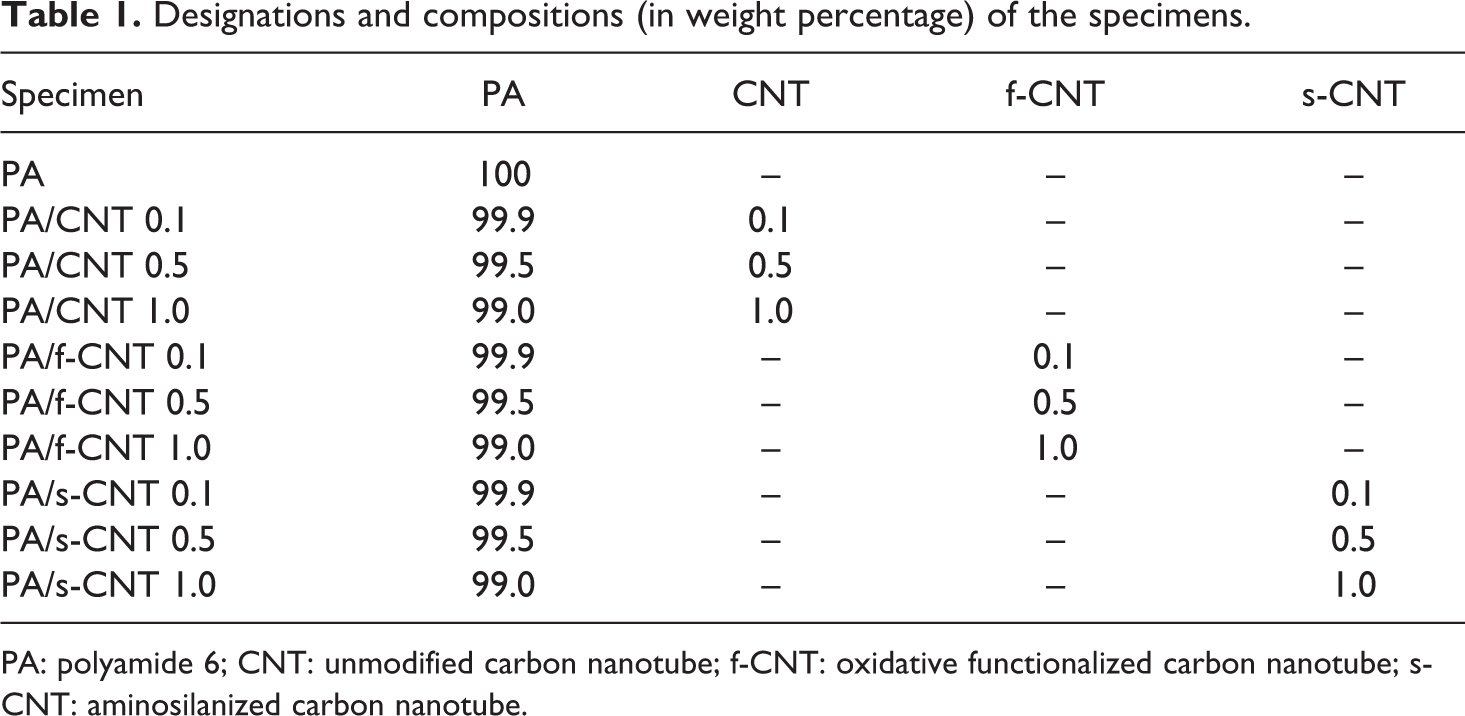
PA: polyamide 6; CNT: unmodified carbon nanotube; f-CNT: oxidative functionalized carbon nanotube; s-CNT: aminosilanized carbon nanotube.
Testing and analyses of the specimens
SEM was carried out in order to observe distribution and morphology of CNTs on the fracture surfaces of nanocomposites using FEI Nova Nano 430 (FEI Company, Hillsboro, OR, USA). The sample surfaces were sputtered with a thin layer of gold to provide a conductive surface and avoid electrostatic charging.
Tensile testing was conducted in order to reveal effects of oxidative functionalization and aminosilanization of CNTs on the mechanical performance of PA6. Tests were performed via 5 kN universal testing machine (model 5565A; Instron, Norwood, Massachusetts, USA), at a crosshead speed of 1 mm min−1 according to ISO 527-2 standard.
Dynamic mechanical analyses (DMA) were performed in order to investigate the thermomechanical behavior of the specimens. Analyses were performed for the specimens of 40 × 10 × 4 mm3 via DMA 8000 (Perkin Elmer; Waltham, Massachusetts, USA) in three-point bending mode at a frequency of 1 Hz. The temperature program was run from −50 to 150°C at a heating rate of 2°C min−1.
TGA was carried out in order to determine the levels of thermal degradation in the specimens. Analyses were conducted via TGA 4000 (Perkin Elmer) with a heating rate of 10°C min−1 from 30 to 700°C under nitrogen flow.
Results and discussion
Morphology and dispersion analysis by SEM
SEM images of the fractured surfaces of nanocomposites with 1.0 wt% CNTs, f-CNTs, and s-CNTs are shown in Figure 1. This figure shows that CNTs are all oriented with the injection molding axes, so that only pulled out tips of CNTs are seen in the fractographs.

SEM images showing dispersion state (images on the left) and pull-out morphologies (images on the right) of 1.0 wt% unmodified CNT, f-CNT, and s-CNT in PA6 matrix. SEM: scanning electron microscopy; PA: polyamide 6; CNT: unmodified carbon nanotube; f-CNT: oxidative functionalized carbon nanotube; s-CNT: aminosilanized carbon nanotube.
It is seen that unmodified CNTs are unevenly distributed while oxidative functionalization (f-CNTs) lead to more even distribution. Figure 1 shows that the most homogeneous distribution was obtained with aminosilanization (s-CNTs).
It is known that dispersion of CNTs in polymer matrices takes place in two stages: the first stage is wetting of CNTs by the matrix and second stage corresponds to diffusion of individual CNTs into the matrix. Unmodified CNTs cannot be wetted properly due to the poor interfacial interactions between nanotube surfaces and PA molecules. Thus, uniform diffusion of unmodified CNTs into PA matrix becomes very difficult.
Oxidative functionalization enhance hydrophilicity of CNTs resulting in stronger interfacial interactions between CNTs and PA. Thus, this treatment increases wetting of CNTs by PA matrix leading to enhanced dispersion of CNTs. Another reason for the more uniform dispersion of f-CNTs could be due to the decreased length of CNTs with acid treatment, as the degree of formation of CNT bundles will decrease with decreased lengths.
Aminosilanization of CNTs provides the strongest interfacial interactions due to the covalent attachment of aminosilane molecules with surface functional groups of f-CNTs. Thus, s-CNTs can be sufficiently wetted, making uniform diffusion into the PA matrix possible.
Therefore, Figure 1 also shows that lengths of the pulled out tips of unmodified CNTs are much longer, which is attributed to fiber pull out due to their insufficient chemical bonding with the matrix. On the other hand, lengths of the pulled out tips of s-CNTs are very short due to strong covalent attractions between their surfaces and the matrix.
Mechanical properties by tensile testing
In order to investigate the effects of unmodified CNTs, f-CNTs, and s-CNTs on the mechanical performance of PA6, tensile tests were conducted with three different loading levels (0.1, 0.5, and 1.0 wt%) of each. Typical tensile stress–strain curves are given in Figure 2 for PA6 and its nanocomposites only with 1.0 wt% CNTs, while yield strength and Young’s modulus values with respect to CNT loadings are shown in Figure 3. All the tensile properties including tensile strength and percentage strain at break data with SDs are tabulated in Table 2.
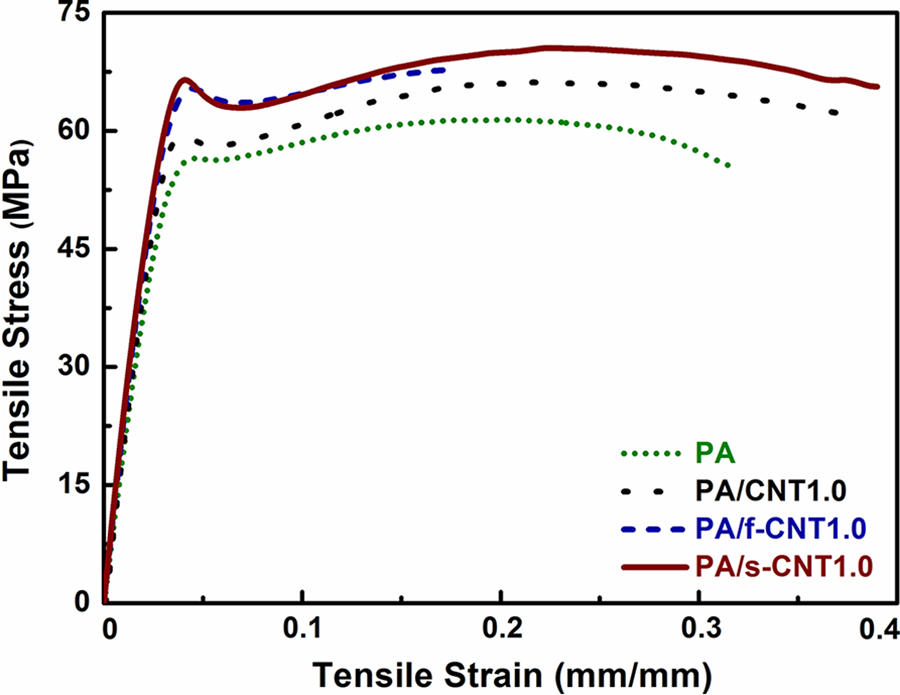
Tensile stress versus tensile strain curves of PA and PA with 1.0 wt% carbon nanotubes. PA: polyamide 6; CNT: unmodified carbon nanotube; f-CNT: oxidative functionalized carbon nanotube; s-CNT: aminosilanized carbon nanotube.
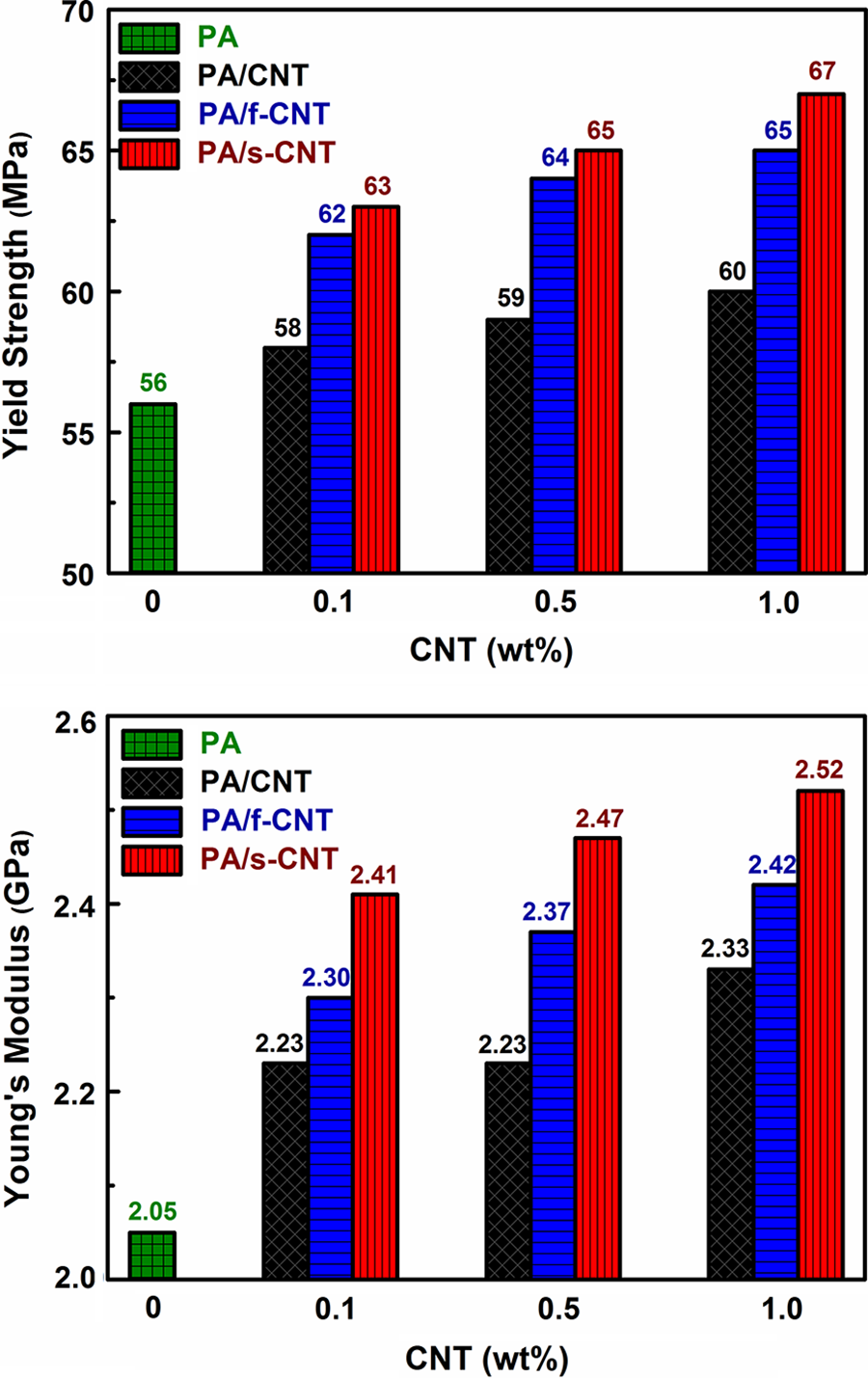
Effects of CNT loading on yield strength and Young’s modulus of PA/CNT nanocomposites. PA: polyamide 6; CNT: unmodified carbon nanotube; f-CNT: oxidative functionalized carbon nanotube; s-CNT: aminosilanized carbon nanotube.
Tensile properties of PA and PA/CNT nanocomposites.
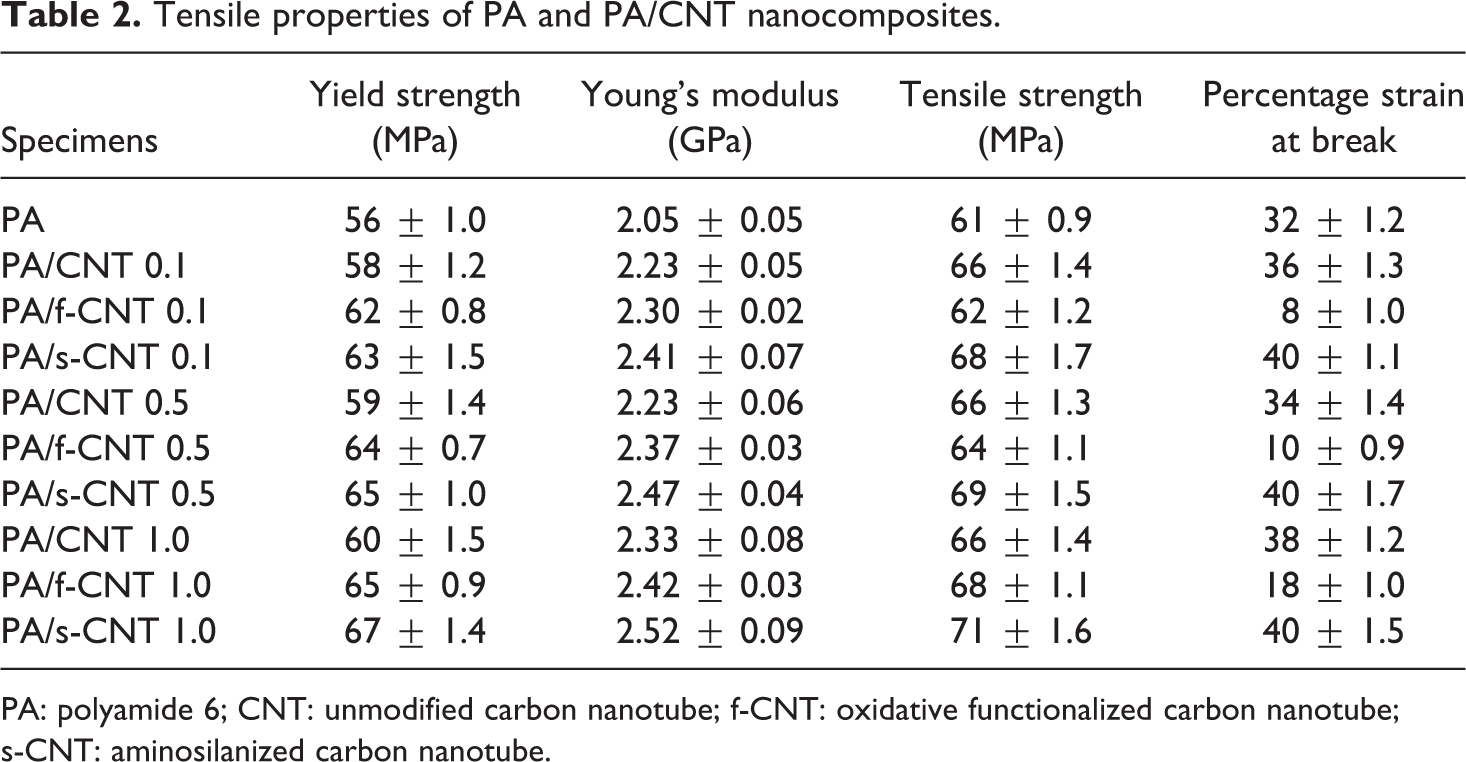
PA: polyamide 6; CNT: unmodified carbon nanotube; f-CNT: oxidative functionalized carbon nanotube; s-CNT: aminosilanized carbon nanotube.
Figure 2 and Table 2 show that even the use of unmodified CNTs increases yield strength and Young’s modulus values to a certain extent. This could be especially due to the decreased mobility of PA6 molecular chains. Therefore, increasing CNT loading should increase these properties.
When CNTs are functionalized by oxidation (f-CNT) and aminosilanization (s-CNT), yield strength and Young’s modulus values increase further. Because, these treatments increase interfacial interactions between the CNT surfaces and PA6 molecules, which leads to a formation of either hydrogen bonding or covalent bonding at the interface. This results in an effective load transfer from the matrix to the CNTs.
In this respect, the highest increases were obtained in the specimen of PA/s-CNT 1.0. Compared to neat PA6, the increased values of yield strength and Young’s modulus were 20 and 23%, respectively.
Table 2 also shows that the use of unmodified CNTs and s-CNTs increases tensile strength and percentage elongation at break values compared to neat PA6. For instance, the increases in the specimens of PA/s-CNT 1.0 are 16% for tensile strength and 25% for percentage elongation at break values.
However, these two mechanical properties of the specimens with f-CNTs were not as enhanced as expected. This might be due to very strong acid treatment leading to decreased lengths of f-CNTs, which makes them insufficient for the efficient load transfer from the matrix to the reinforcement.
Thermomechanical behaviour by DMA
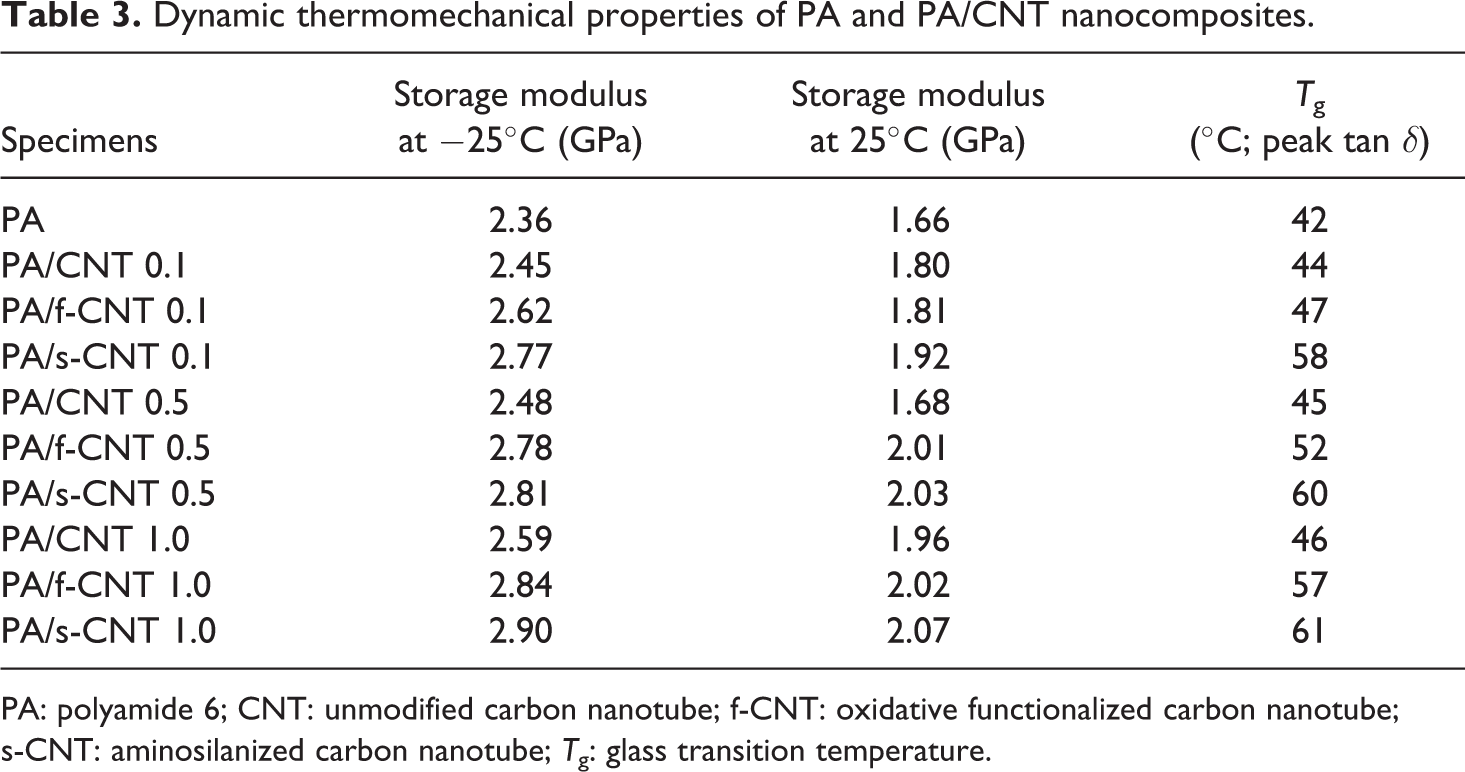
In order to investigate the thermomechanical properties of the specimens, DMA were conducted. Typical storage modulus and tan δ curves are given in Figure 4 only for the specimens with 1 wt% CNTs, while Table 3 gives the data for all specimens. In this table, results are evaluated as two different storage modulus values at −25 and 25°C, including primary transition temperature (T g) taken at peak tan δ values.
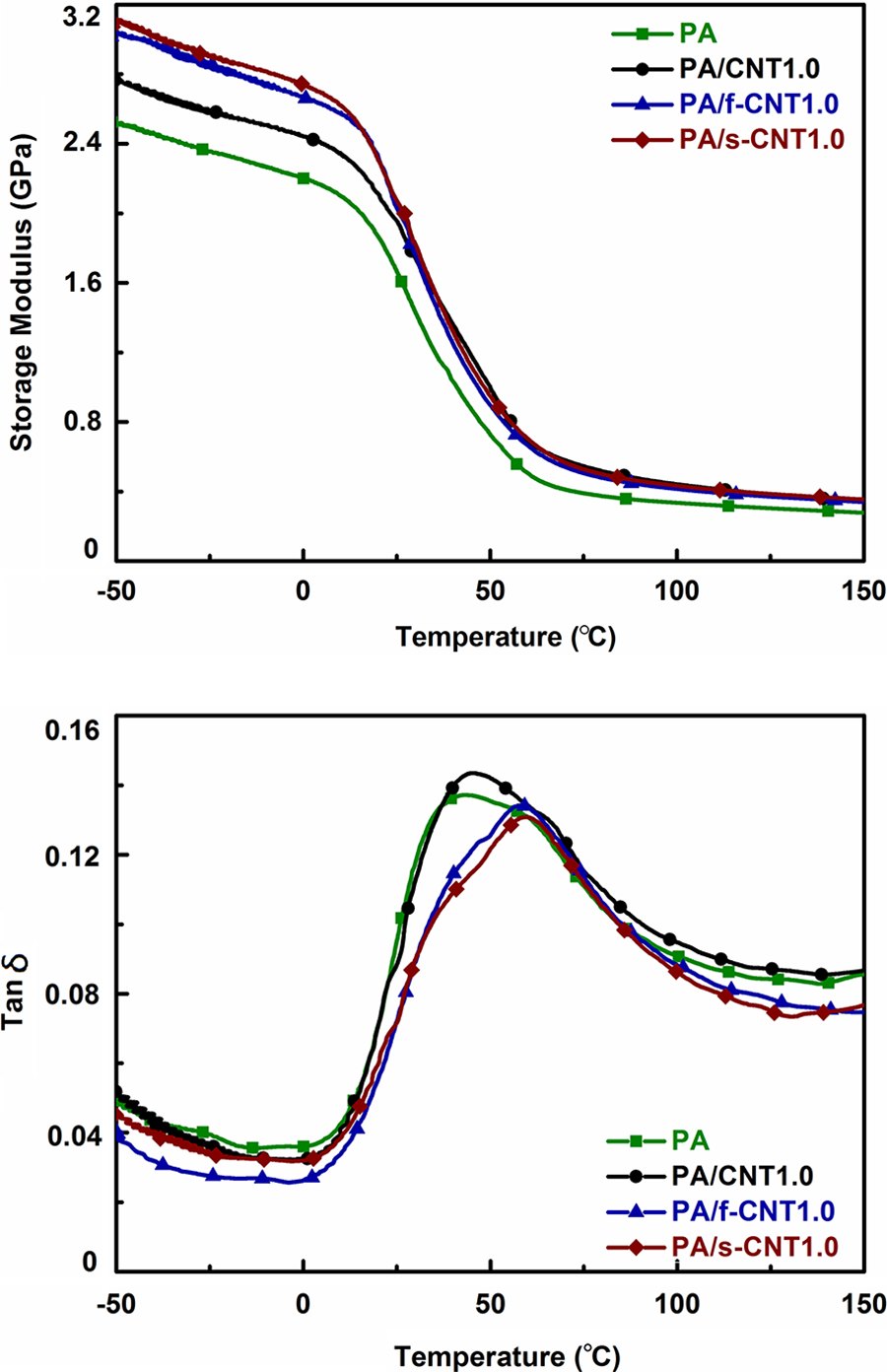
Storage modulus and tan δ curves of PA and PA with 1.0 wt% CNTs. PA: polyamide 6; CNT: unmodified carbon nanotube; f-CNT: oxidative functionalized carbon nanotube; s-CNT: aminosilanized carbon nanotube.
Dynamic thermomechanical properties of PA and PA/CNT nanocomposites.
PA: polyamide 6; CNT: unmodified carbon nanotube; f-CNT: oxidative functionalized carbon nanotube; s-CNT: aminosilanized carbon nanotube; T g: glass transition temperature.
Figure 4 and Table 3 show that in the glassy plateau region, for example, at −25°C, even the use of unmodified CNTs increase storage modulus value of PA6 due to the decreased mobility of the side groups of PA6 backbone. Increasing the CNT content leads to more improvement in the storage modulus values.
Table 3 also indicates that oxidative functionalization (f-CNT) and aminosilanization (s-CNT) increase the storage modulus values at −25°C even further. For instance, the increases were 20 and 23% for the specimens with 1.0 wt% f-CNTs and s-CNTs, respectively. This is thought to be due to the treatments leading to the formation of stronger chemical bonding making mobility of side groups even harder.
Tan δ curves in Figure 4 show that above 0°C, the main backbone of the PA6 chains start to move over each other significantly, which decreases storage modulus values. However, storage modulus data at 25°C given in Table 3 indicates that the use of unmodified CNTs, f-CNTs, and s-CNTs again increases this value compared to neat PA6. This time, the increases in storage modulus values are 18, 22, and 25% for the 1.0 wt% unmodified CNTs, f-CNTs, and s-CNTs, respectively. This is again due to the uniform dispersion and stronger interfacial interactions between the treated nanotube surfaces and PA6 chains leading to decreased mobility.
It is known that tan δ curves make a peak when the temperature reaches to a critical transition where blocks of polymer chain segments move over each other. This primary transition temperature (at tan δ peak) can be interpreted as the T g of the specimens. Table 3 shows that the T g of neat PA6 increases with the use of all types of CNTs. Again as discussed above, due to more restricted mobility of PA6 chains with functionalized nanotubes surfaces, the increases in T g of neat PA6 were 4, 15, and 19°C for the 1.0 wt% unmodified CNTs, f-CNTs, and s-CNTs, respectively.
Thermal degradation by TGA
In order to investigate the effects of CNT surface treatments on the thermal degradation behavior of PA6, TGA data were evaluated in the form of thermogravimetric (TG) and differential thermogravimetric (DTG) curves for 1 wt% CNTs, as shown in Figure 5. Table 4 also tabulates the two sets of data for all specimens; T 25 wt% and T MLR-max, which are the thermal degradation temperatures at 25 wt% mass loss and maximum mass loss rate peak, respectively.
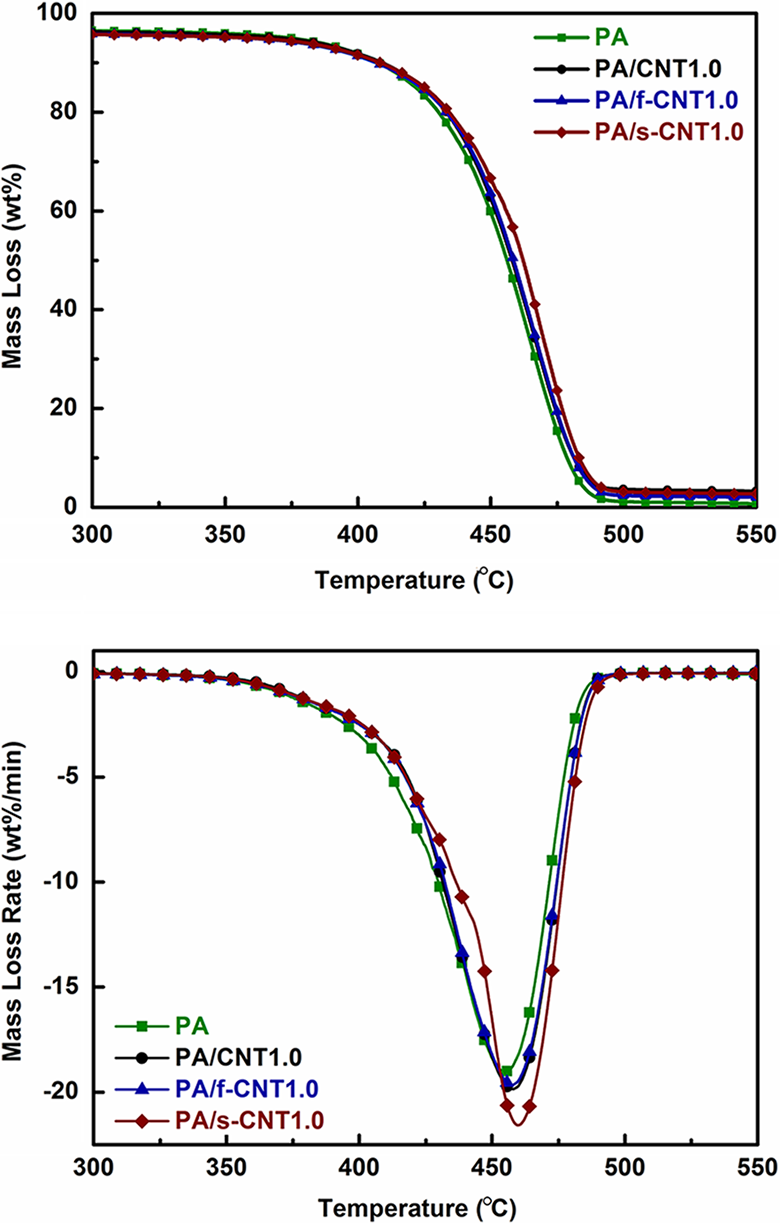
TG and DTG curves of PA and PA with 1.0 wt% carbon nanotubes. PA: polyamide 6; CNT: unmodified carbon nanotube; f-CNT: oxidative functionalized carbon nanotube; s-CNT: aminosilanized carbon nanotube; TG: thermogravimetric; DTG: differential thermogravimetric.
Thermal degradation data of PA and PA/CNT nanocomposites.
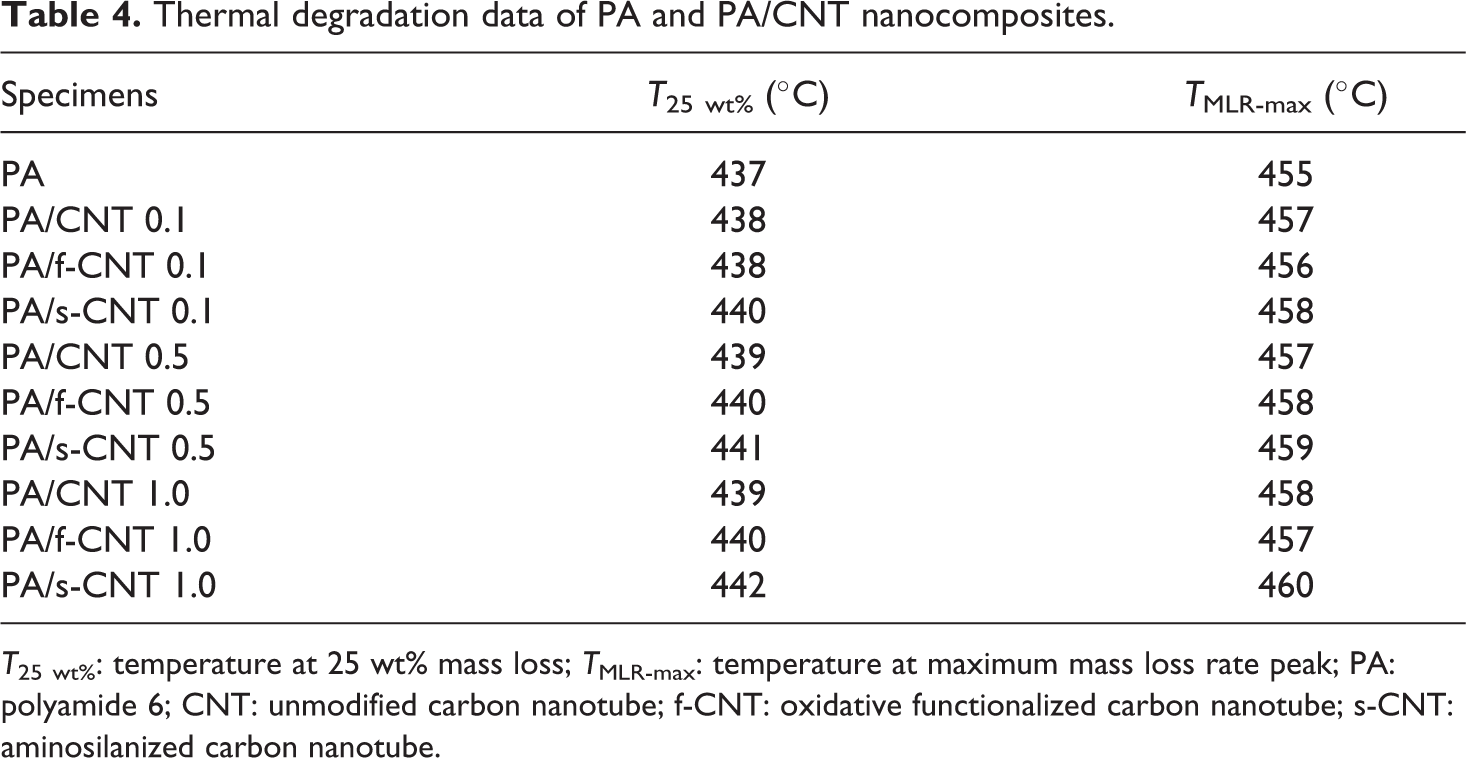
T 25 wt%: temperature at 25 wt% mass loss; T MLR-max: temperature at maximum mass loss rate peak; PA: polyamide 6; CNT: unmodified carbon nanotube; f-CNT: oxidative functionalized carbon nanotube; s-CNT: aminosilanized carbon nanotube.
Figure 5 and Table 4 show that there are increases in the thermal degradation temperatures of neat PA6 when reinforced with CNTs. These improvements should be due to the physical barrier function of CNTs, which prevent the diffusion of volatiles and decomposed products out of the polymer matrix during decomposition.
Improved dispersion and interfacial bonding between the polymer matrix and functionalized CNTs leads to higher thermal degradation temperatures. For example, for the specimens of PA/s-CNT1.0, the increases in T 25 wt% and T MLR-max values are both as much as 5°C.
Conclusions
Basic conclusions drawn from this study investigating the influences of oxidative functionalized (f-CNT) and aminosilanized (s-CNT) carbon nanotubes on the mechanical and thermal properties of PA6 nanocomposites are as follows:
Compared to unmodified CNTs, SEM analysis revealed that f-CNTs and s-CNTs were dispersed more evenly due to the increased interactions between the matrix and CNTs.
Yield strength and Young’s modulus of nanocomposites with surface-modified CNTs were as much as 20 and 23% higher compared to neat PA6, both due to decreased matrix chain mobility and very efficient load transfer from the matrix to chemically bonded CNTs.
Thermomechanical analysis by DMA indicated that surface modification of CNTs decreases mobility of both side groups and main backbone of PA6 chains significantly leading to improved storage modulus values as much as 25% and increased T g (peak tan δ) by 19°C.
TGA analysis also resulted in higher thermal degradation temperatures, as much as by 5°C, with the efficient physical barrier formation of surface-treated CNTs preventing diffusion of volatiles and decomposed products from the matrix.
Footnotes
Acknowledgement
Authors greatly acknowledge the Middle East Technical University Wind Energy Center for performing thermal analyses.
Funding
This research received financial support from TUBITAK, the Scientific and Technological Research Council of Turkey, as part of the project 111M460.
