Abstract
Flexible polymer nanocomposites emerge as promising materials to meet the requirements of strategic sectors such as automotive, packaging, and electronics. In this work, the influence of incorporating multiwalled carbon nanotubes (MWCNTs) at low contents (0.5 and 1 parts per hundred resin, phr) was investigated in a polyamide 6 (PA6)/maleic anhydride-grafted styrene–ethylene–butylene–styrene (SEBS-g-MA) blend. The materials were processed in a twin-screw extruder and molded by injection. The torque reometry properties, impact resistance, tensile strength, X-ray diffraction (XRD), Raman spectroscopy, Fourier-transform infrared spectroscopy (FTIR), differential scanning calorimetry (DSC), and heat deflection temperature (HDT) were evaluated, as well as scanning electron microscopy (SEM). Torque rheometry revealed an increase in torque with the incorporation of SEBS-g-MA into PA6, suggesting enhanced viscosity and compatibility. As a result, tough and flexible nanocomposites were formed at room temperature. FTIR, DSC and XRD analyses indicated the predominance of the α-crystalline phase of PA6 in the nanocomposites, while Raman spectroscopy detected the presence of MWCNTs only at the 1 phr concentration. SEM imaging confirmed increased ductility in the PA6/SEBS-g-MA blend and in the nanocomposites, with good distribution of carbon nanotubes, particularly at 1 phr. Notably, the PA6/SEBS-g-MA/MWCNT (1 phr) nanocomposite exhibited remarkable improvements, 178.9% increase in impact strength, 221.4% increase in elongation at break, while maintaining tensile strength and HDT compared to neat PA6. These findings suggest the potential for producing tailor-made flexible nanocomposites for targeted applications, offering a well-balanced set of properties.
Introduction
Polymeric nanocomposites are materials produced by incorporating nanoparticles into a polymer matrix, allowing for the development of tailored materials for specific applications.1,2 Due to their low weight, enhanced performance, and versatility, these materials have found applications across a wide range of sectors, including packaging, automotive, electronics, and biomedical fields.3,4 The most commonly used nanofillers in polymer nanocomposites include clays, carbon nanotubes, graphene, silica, and magnetic ferrites, each offering distinct characteristics that impart functionalities not typically present in the pure polymer matrix.5–10
The choice of polymeric matrices for nanocomposite production depends largely on the intended application and the desired material properties. Among the most widely used polymers in engineering applications are polyamides (PAs), which are extensively employed in manufacturing industries. 11 Polyamide 6 (PA6), in particular, combines good mechanical strength, durability, versatility, and abrasion resistance. It also offers excellent processability and thermal stability, making it especially attractive for the automotive industry and a candidate for the development of polymeric nanocomposites.12,13 Although PA6 exhibits good overall mechanical performance, it tends to show limited impact strength, particularly in regions with stress concentrators, often resulting in premature failure.14,15 This brittleness becomes more pronounced in dry environments. To address this limitation, PA6 is often modified with additives, such as impact modifiers, especially those functionalized with maleic anhydride, to enhance its performance under more demanding mechanical conditions.16,17 This approach represents a common strategy for developing tough nanocomposites that retain flexibility and durability at room temperature.
Toughened PA6 nanocomposites are being developed to combine lightweight characteristics with high impact and fracture strength, even under harsh conditions. 18 Ghorbankhan et al. 19 fabricated PA6-based nanocomposites using nitrile butadiene rubber (NBR) reinforced with 1% graphene. The incorporation of graphene into the PA6/NBR system resulted in enhanced thermal stability, as well as improved impact performance and tensile strength. PA6/EPDM-g-MA nanocomposites containing multiwalled carbon nanotubes (MWCNTs) were melt-processed using PA6 with different molecular weights, as reported by Esmizadeh et al. 20 The presence of MWCNTs promoted a finer dispersion and refinement of the EPDM-g-MA phase in the high molecular weight PA6 (HPA6), resulting in a more ductile fracture morphology, as observed through SEM analysis. Additionally, the carbon nanotubes were more uniformly dispersed in HPA6 compared to the low molecular weight PA6 (LPA6). Although the incorporation of MWCNTs reduced the overall ductility of the PA6/EPDM-g-MA blend, the nanocomposites based on HPA6 exhibited a better balance between stiffness and elongation at break, leading to enhanced toughness. González et al. 21 investigated the effect of montmorillonite clays on the production of PA6/SEBS-g-MA nanocomposites (at a 70/30 wt% ratio), and reported a synergistic effect with the addition of 3% clay. This modification led to enhanced toughness at room temperature and a 44% increase in the modulus compared to the neat PA6 matrix.
The combination of polyamide 6 (PA6) with SEBS-g-MA (styrene-ethylene/butylene-styrene grafted with maleic anhydride) offers a promising route to achieve good thermal stability, enhanced moisture resistance, and efficient processability. At the same time, SEBS-g-MA acts as an impact modifier for PA6, significantly increasing its ductility.22,23 Given the potential of PA6/SEBS-g-MA blends, several studies have been conducted to develop nanocomposites, with the literature predominantly focusing on the use of clays, particularly montmorillonite (MMT), as nanofillers.24,25 However, there are virtually no reports on the use of carbon nanotubes (MWCNT) in this specific PA6/SEBS-g-MA system, highlighting the need for investigations that explore the potential to obtain materials that are both flexible and tough. Multiwalled carbon nanotubes (MWCNTs) exhibit high mechanical strength, as well as relevant electrical, thermal, and chemical properties, which has led to growing interest in their use in the preparation of polymer nanocomposites. Although alternatives such as single-walled carbon nanotubes (SWCNTs) and graphene exist, MWCNTs have a lower cost and are widely available on a commercial scale, making them a technically suitable and economically viable option for studies and industrial applications.26–28
The present study aimed to evaluate the effect of low concentrations of multiwalled carbon nanotubes (MWCNTs), specifically 0.5 and 1 parts per hundred resin (phr), in a PA6/SEBS-g-MA (80/20 wt%) blend, focusing on the analysis of rheological, mechanical, thermal, and thermomechanical properties, as well as morphological characteristics.
Materials and Methods
Materials
In this study, the polymeric matrix used was polyamide 6 (PA6), commercially known as B300®, with a density of 1.13 g/cm3. The material was supplied in pellet form by ThaThi Polímeros. As an impact modifier, a maleic anhydride-grafted styrene-ethylene-butylene-styrene block copolymer (SEBS-g-MA) was used, provided by Kraton Polymers under the trade name Kraton FG 1901G®. This copolymer contains 30% styrene and incorporates 1.4–2.0% maleic anhydride groups grafted onto the ethylene-butylene phase, with an MFI of 22 g/10 min (230°C/5 kg). The nanoparticles employed were multiwalled carbon nanotubes (MWCNTs), supplied by Advanced 2D Materials Co., featuring an outer diameter of 8–15 nm, lengths ranging from 3 to 12 µm, and a purity level exceeding 95%.
Methods
Nanocomposites prepared based on PA6/SEBS-g-MA, according to the MWCNT content.
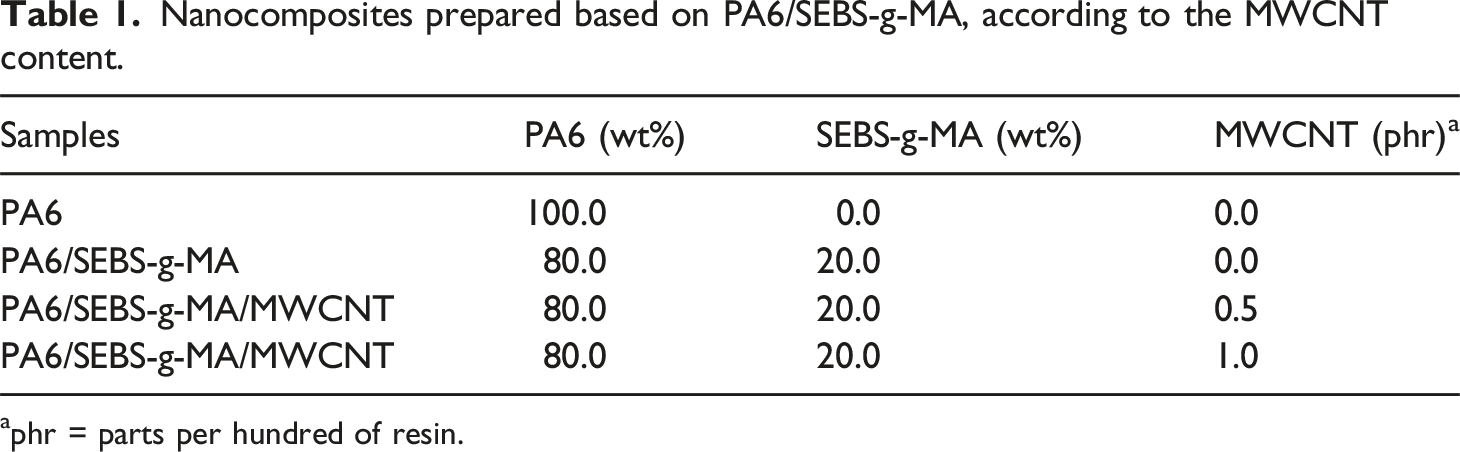
aphr = parts per hundred of resin.
Prior to processing, PA6, SEBS-g-MA, and MWCNTs were dried in a vacuum oven at 80°C for 24 hours. For each composition, the amount of material processed in the extruder was 600 g. Initially, PA6/MWCNT (0.5 phr: 245 g/3 g; 1 phr: 245 g/6 g) masterbatches were prepared using an internal mixer with a volume of 310 cm3, equipped with roller-type rotors, operating at 220°C, 60 r/min, and with a residence time of 3 minutes. Subsequently, the PA6/MWCNT masterbatches were ground in a knife mill to enable their dilution in the extruder. The processing was carried out in a co-rotating intermeshing twin-screw extruder (ZSK18 K model, Coperion), with an 18 mm screw diameter and an L/D ratio of 40. The screw configuration was designed with elements optimized to promote both dispersion and distribution of the phases. The compositions processed in the extruder were prepared according to Table 1, corresponding to: PA6 (600 wt%), PA6/SEBS-g-MA (480/120 wt%), PA6/SEBS-g-MA/MWCNT (0.5 phr) (235/120 wt% + 245 g/3 g masterbatch), and PA6/SEBS-g-MA/MWCNT (1 phr) (235/120 wt% + 245 g/6 g masterbatch).
During extrusion, a temperature gradient ranging from 200 to 230°C was applied, with a screw speed of 250 r/min and a feed rate of 3 kg/h. The extrudate was subsequently pelletized and dried in a vacuum oven at 80°C for 24 hours. In the next stage, the dried pellets were injection molded using an Arburg Allrounder 207C Golden Edition machine to produce specimens for impact testing (ASTM D256-10), tensile testing (ASTM D638-22), and heat deflection temperature (HDT) testing (ASTM D648-18). All samples were processed under standardized conditions, previously established based on relevant literature.18,34 The injection molding parameters were as follows: injection pressure of 1200 bar, barrel temperatures set at 230, 240, 240, 240, and 245°C, mold temperature maintained at 50°C, cooling time of 25 seconds, and holding pressure of 1000 bar. The experimental procedure for preparing the nanocomposites is illustrated in Figure 1. Processing scheme of nanocomposites.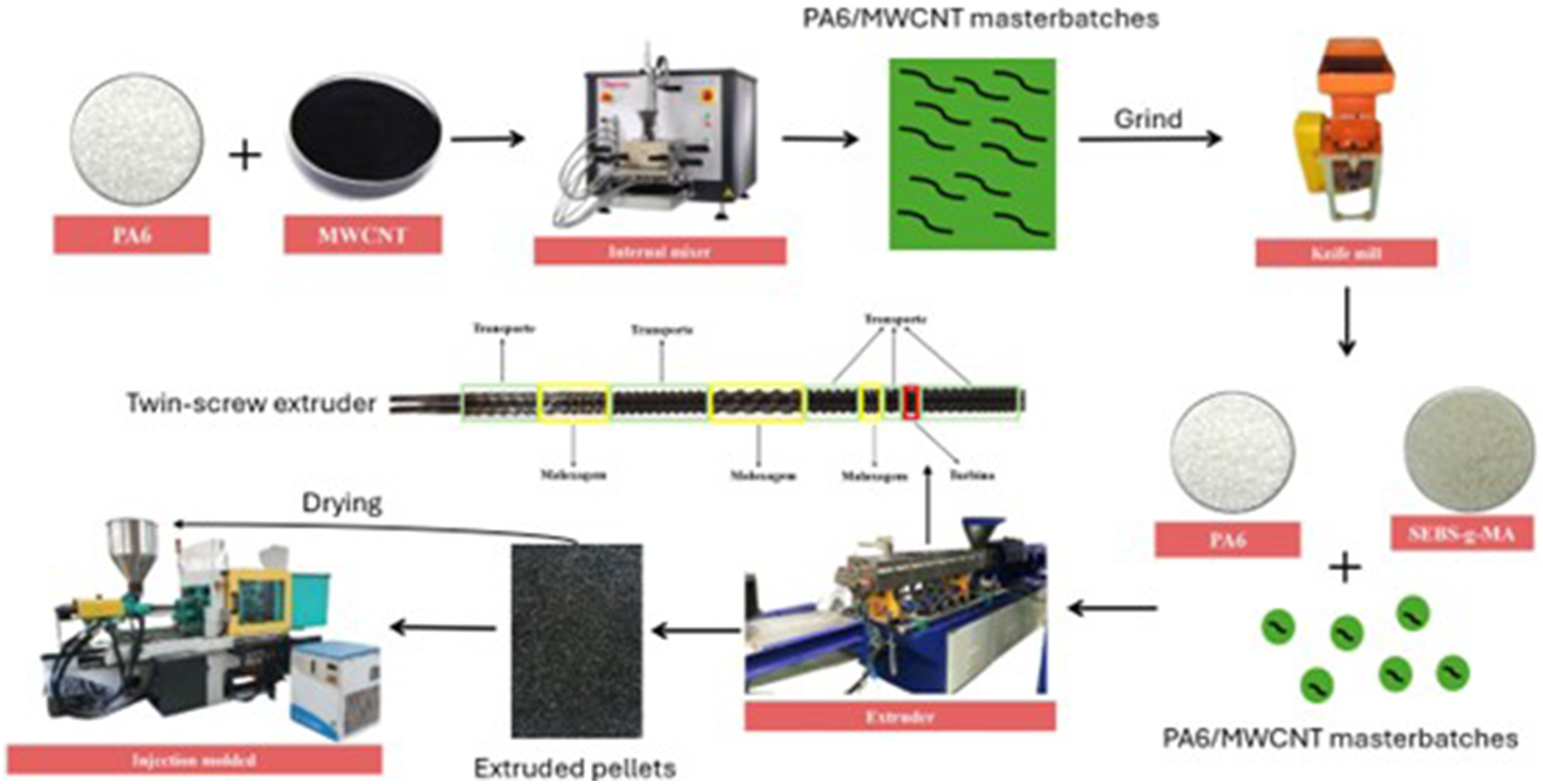
Compound Characterization
The rheological behavior of neat PA6, the PA6/SEBS-g-MA blend, and the corresponding nanocomposites was evaluated using a HAAKE Rheomix 3000 torque rheometer, equipped with roller-type intensive rotors. The tests were carried out at 220°C, with a rotational speed of 60 r/min and a mixing time of 10 minutes.
X-ray diffraction (XRD) analyses were performed using a Shimadzu XRD-7000 diffractometer, equipped with a Cu-Kα radiation source (λ = 1.5406 Å), operating at 40 kV and 30 mA. Measurements were recorded over a 2θ range of 5° to 50°, with a scanning rate of 2°/min.
Fourier-transform infrared spectroscopy (FTIR) was employed to investigate potential chemical interactions between the components. The analyses were carried out using a Jasco Spectrum 4600 spectrometer, operated via Spectra Manager software. Spectra were collected over the range of 4000 to 500 cm−1, with a resolution of 4 cm−1, and 32 scans were accumulated for each sample.
Raman spectroscopy measurements were performed using a RENISHAW InVia system, equipped with a coupled Raman microscope. A 532 nm laser excitation source was used, and spectra were acquired over the spectral range of 250 to 2500 cm−1.
Notched Izod impact tests were performed at room temperature using a Ceast Resil 5.5 impact tester equipped with a 4 J pendulum, in accordance with ASTM D256-10. The reported values represent the average of 10 specimens tested for each formulation.
Tensile properties were evaluated using a universal testing machine (EMIC DL 500 MF), operating at a crosshead speed of 50 mm/min with a load capacity of 200 kgf, under ambient temperature conditions. Specimens were injection molded in accordance with ASTM D638, and the results represent the average of 10 samples per formulation.
Differential scanning calorimetry (DSC) was conducted on Shimadzu equipment, model DSC-60Plus. During analyses, nitrogen was employed as the carrier gas (50 mL/min), and 3 mg samples were subjected to a heating-cooling-heating program from 50 to 250°C, employing a 10°C/min heating rate.
Heat Deflection Temperature (HDT) tests were carried out using a Ceast HDT 6 VICAT apparatus, in accordance with ASTM D648-18. The tests were performed under a load of 1.82 MPa, with a heating rate of 120°C/h, and the samples were immersed in silicone oil during testing. The HDT was recorded at the point when a deflection of 0.25 mm occurred. Results were based on an average of three injection-molded specimens.
The sample surfaces were examined using a TESCAN MIRA3 scanning electron microscope (SEM), operated under high-vacuum conditions and controlled via Mira TC software. The analyses were performed on the fracture surfaces obtained from the impact tests, after coating the specimens with a thin layer of gold to ensure conductivity.
Results and Discussion
Torque Rheometry
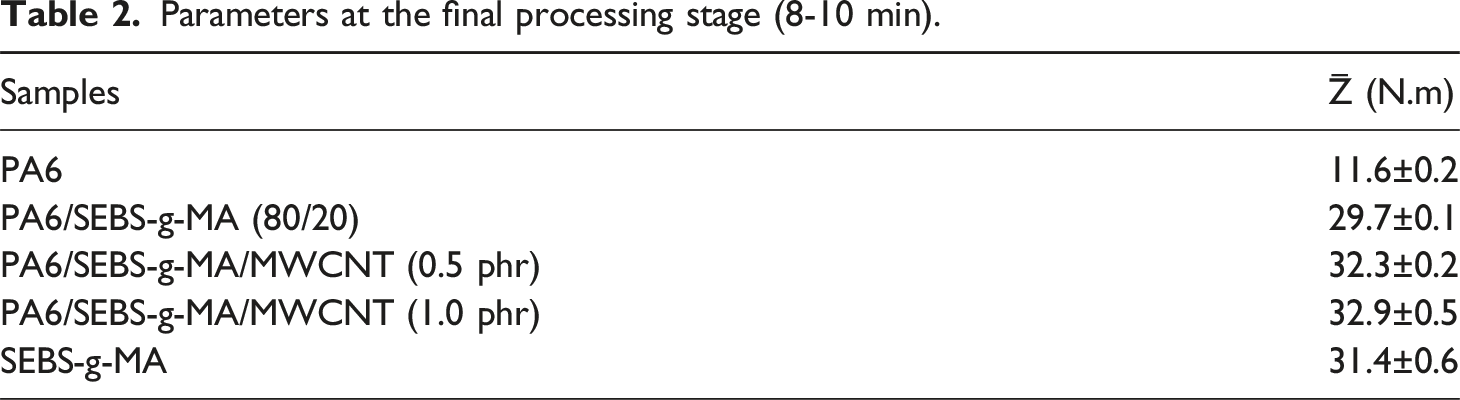
Torque rheometry is a widely used method for evaluating the processability of polymers, polymer blends, and nanocomposites, as it reflects the rotational resistance experienced by the mixing rotors. In general, higher torque values are associated with increased viscosity.35,36 Figure 2(a), and (b) shows the torque profiles over time for neat PA6, SEBS-g-MA, the PA6/SEBS-g-MA (80/20 wt%) blend, and the corresponding nanocomposites containing 0.5 and 1.0 phr of multiwalled carbon nanotubes (MWCNTs). Table 2 summarizes the average torque values (Z̅) measured in the 8–10 minute interval. Curves of the pure polymers, the PA6/SEBS-g-MA blend and the nanocomposites with 0.5 and 1 phr of MWCNT, for: torque versus time (a); magnification between 8 and 10 minutes (b). Parameters at the final processing stage (8-10 min).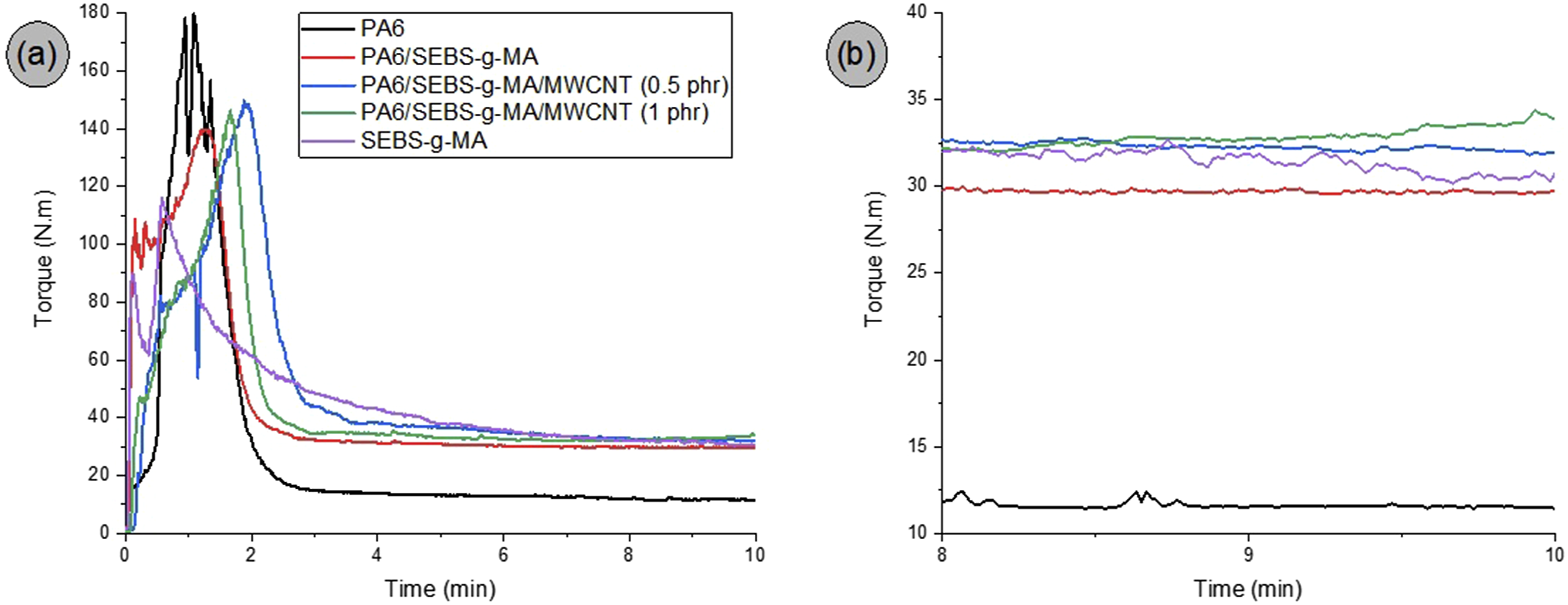
In Figure 2(a), an initial increase in torque values can be observed during the early stages of processing, reaching a maximum peak. This behavior is attributed to the dissipation of mechanical energy caused by friction between solid particles. After approximately 4 minutes, all formulations appear to be fully melted, leading to a stabilization of the torque. Figure 2(b) presents the torque versus time curves in the 8–10 minute interval, during which the melt reaches a steady viscosity, a critical condition for consistent extrusion and injection molding performance.
Neat PA6 exhibited the lowest average torque value (11.6 N·m), while SEBS-g-MA showed a significantly higher torque of 31.4 N·m. The PA6/SEBS-g-MA blend displayed an intermediate torque of approximately 29.7 N·m, falling between the values of the pure components. The addition of just 20% SEBS-g-MA to PA6 resulted in a notable increase in the matrix torque, indicating interactions between the functional groups of PA6 and SEBS-g-MA. These interactions led to an increase in the viscosity of the PA6 matrix, which was reflected in the higher torque values. This enhanced interaction suggests improved compatibility between the PA6 and SEBS-g-MA phases, contributing to better impact strength and elongation at break, as discussed later. Several studies in the literature37–39 report that maleic anhydride groups can react with the terminal amine groups of PA6, facilitating the in situ formation of a copolymer at the interface and thus increasing viscosity.
Regarding the PA6/SEBS-g-MA/MWCNT nanocomposites, as shown in Table 2, the stabilized torque values increased to 32.3 N·m and 32.9 N·m for 0.5 and 1 phr of MWCNT, respectively. The presence of carbon nanotubes restricted the mobility of the PA6/SEBS-g-MA polymer chains by acting as physical barriers to flow, resulting in greater shear resistance, which in turn increased both torque and viscosity.40,41
Fourier Transform Infrared Spectroscopy (FTIR)
Figure 3 displays the FTIR spectra of neat PA6, SEBS-g-MA, the PA6/SEBS-g-MA blend, and the corresponding nanocomposites containing 0.5 phr and 1 phr of MWCNT. FTIR for pure PA6, pure SEBS-g-MA, PA6/SEBS-g-MA blend and nanocomposites with MWCNT.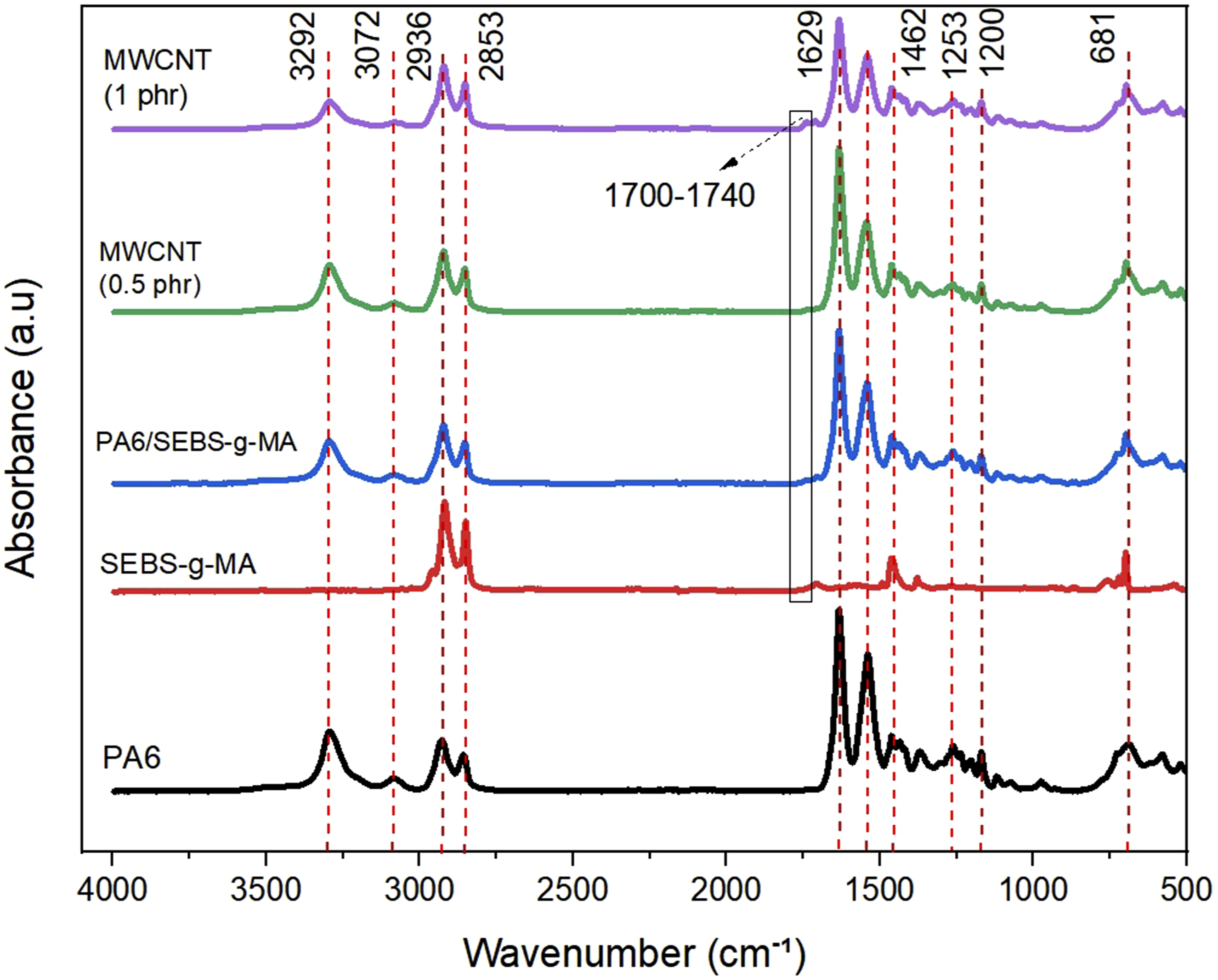
The bands observed at 1535 cm−1 and 1629 cm−1 correspond to the vibrations of the amide groups, specifically the C–N and C = O bonds. Bands at 3292 cm−1 and 3072 cm−1 are attributed to the stretching of N–H groups, while those at 2936 cm−1 and 2853 cm−1 correspond to the stretching of CH2 groups in the amorphous and crystalline regions of PA6, respectively. The bands detected at 1462 cm−1 and 1200 cm−1 are assigned to the vibrations of CH2 groups adjacent to N–H and C=O, indicating the predominant formation of the monoclinic α-phase in the PA6 structure, consistent with observations reported by Alatawi et al., 42 Rwei et al., 43 and Yan et al. 44 For SEBS-g-MA, the main bands are observed between 3050 and 3000 cm−1 (styrene with weak bands), 2950–2850 cm−1 (aliphatic C–H from the ethylene–butylene segment), 1460–1380 cm−1 (CH2/CH3 deformations of the aliphatic chain), and 910–700 cm−1 (out-of-plane vibrations of the aromatic ring). As demonstrated in the literature, 45 FTIR generally shows a band at 1780 cm−1 for the maleic anhydride group. However, the maleic anhydride band around 1780 cm−1 was not detected in SEBS-g-MA, possibly due to the low degree of functionalization. A small band in SEBS-g-MA around 1705 cm−1 suggests the presence of a carboxylic acid group or the C = O stretching of hydrolyzed MA.45,46 Overall, the characteristic PA6 bands were preserved in the spectra of the PA6/SEBS-g-MA blend and the PA6/SEBS-g-MA/MWCNT nanocomposites, indicating that the incorporation of SEBS-g-MA and carbon nanotubes did not induce significant structural changes in the PA6 matrix. However, a subtle band appeared for the PA6/SEBS-g-MA/MWCNT (1 phr) nanocomposite between 1700 and 1740 cm−1, which may suggest the formation of imide groups. Phan et al. 47 reported the presence of a band in this FTIR spectral region, attributing it to the carbonyl absorption of imide groups formed as a result of interactions between amine groups and maleic anhydride. A similar behavior was observed by Barra et al. 48 during the development of reactive blends of PA6/EPDM-MA. They suggested that the band appearing between 1700 and 1730 cm−1 could be associated with the carbonyl absorption of imide groups formed at the interface between the elastomer domains and the PA6 matrix.
X-ray diffraction (XRD)
Figure 4 shows the X-ray diffraction (XRD) patterns of neat PA6, the PA6/SEBS-g-MA blend, and the nanocomposites containing 0.5 and 1.0 phr of carbon nanotubes. X-ray diffractograms of pure PA6, PA6/SEBS-g-MA (80/20) blend and nanocomposites, with 2θ scanning from 10 to 50°.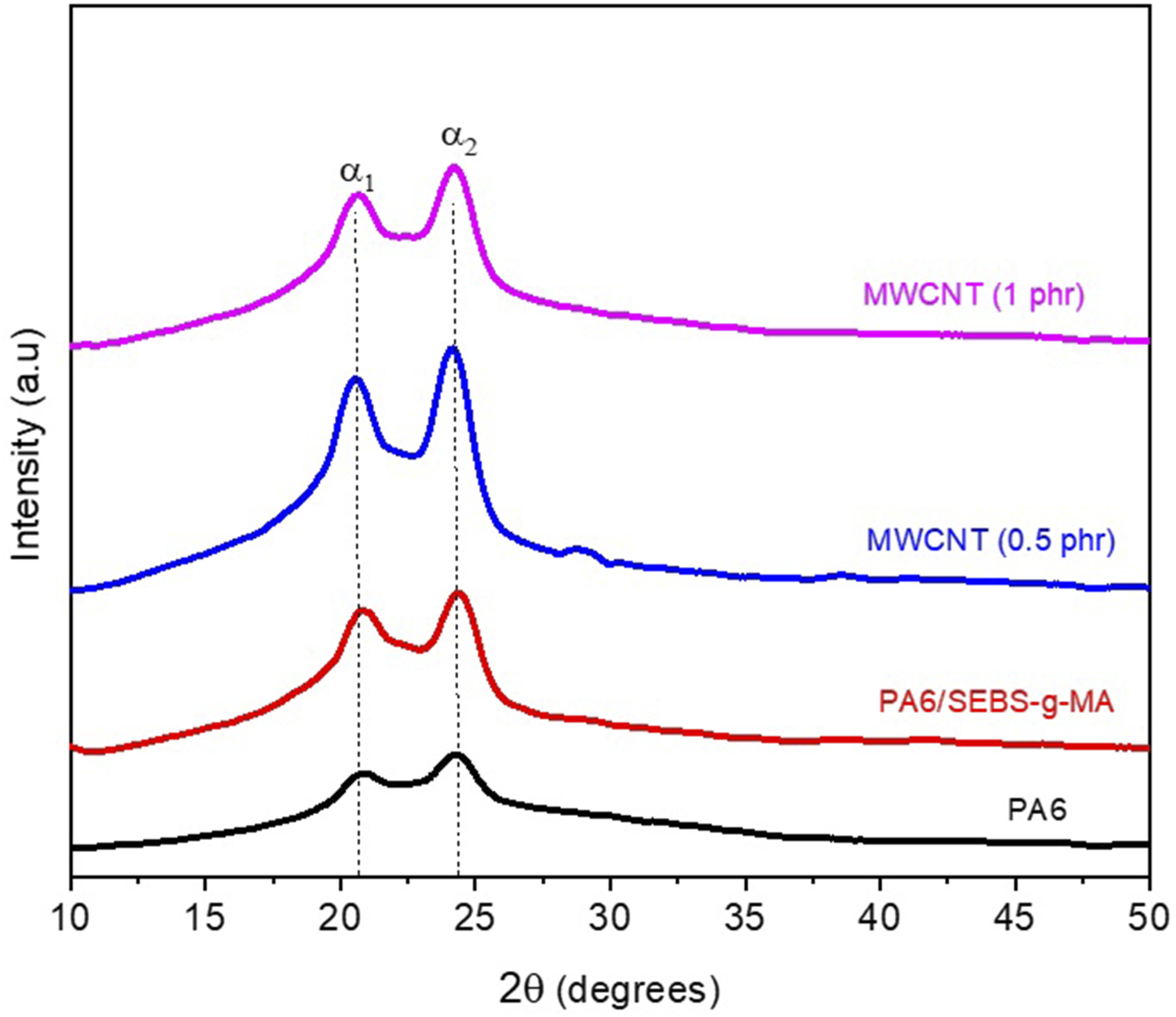
In Figure 4, two prominent peaks at approximately 20° and 24° are observed for all compositions, consistent with the findings reported by Medeiros et al. 49 These peaks correspond to the crystalline α1 and α2 phases of polyamide 6, associated with the diffraction planes (100) and (002)/(202), respectively. This confirms that the α-phase is the predominant crystalline structure in pure PA6, the PA6/SEBS-g-MA blend, and the nanocomposites, corroborating the trend observed in the FTIR analysis.
Differential Scanning Calorimetry
Figure 5(a), (b) shows the DSC curves corresponding to the crystalline melting temperature (Tm) and crystallization temperature (Tc) for neat PA6, the PA6/SEBS-g-MA blend, and the PA6/SEBS-g-MA/MWCNT nanocomposites containing 0.5 phr and 1 phr of MWCNT. The corresponding thermal properties are summarized in Table 3. Neat PA6 exhibited a Tc of approximately 190.6°C. The incorporation of 20 wt% SEBS-g-MA into the PA6 matrix shifted Tc to a slightly lower value, 188.2°C. Because SEBS-g-MA is an essentially amorphous elastomer, its presence may have restricted the mobility of PA6 chains, hindering the molecular organization required for crystallization. In contrast, the addition of 0.5 phr and 1 phr of MWCNT to the PA6/SEBS-g-MA blend resulted in an increase in Tc, indicating an acceleration of the crystallization process at higher temperatures. In this context, the carbon nanotubes acted as nucleating agents, promoting the onset of crystallization of the blend at higher temperatures compared with the system without nanofillers. DSC curves obtained for PA6, PA6/SEBS-g-MA, and the nanocomposites during: (a) heating; (b) cooling. Thermal properties of PA6, PA6/SEBS-g-MA blend and nanocomposites. ΔH = crystalline melting enthalpy; Xc = degree of crystallinity. *Xc = 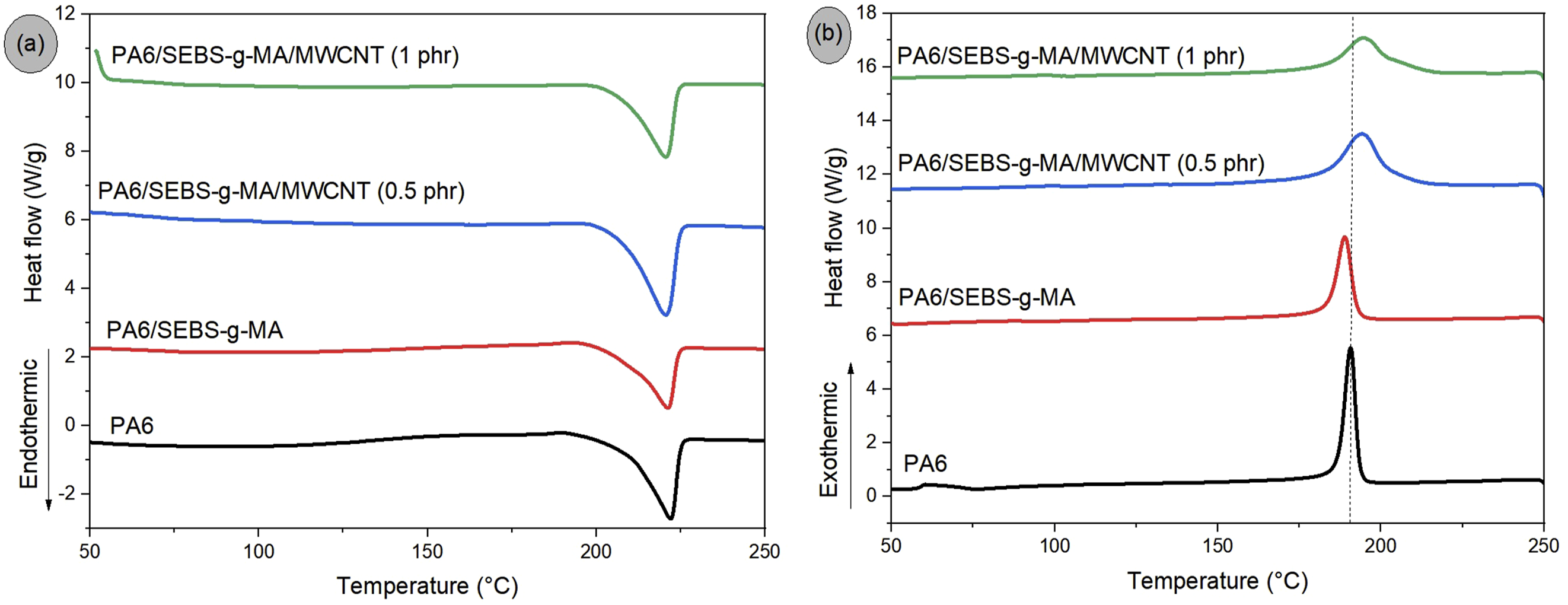
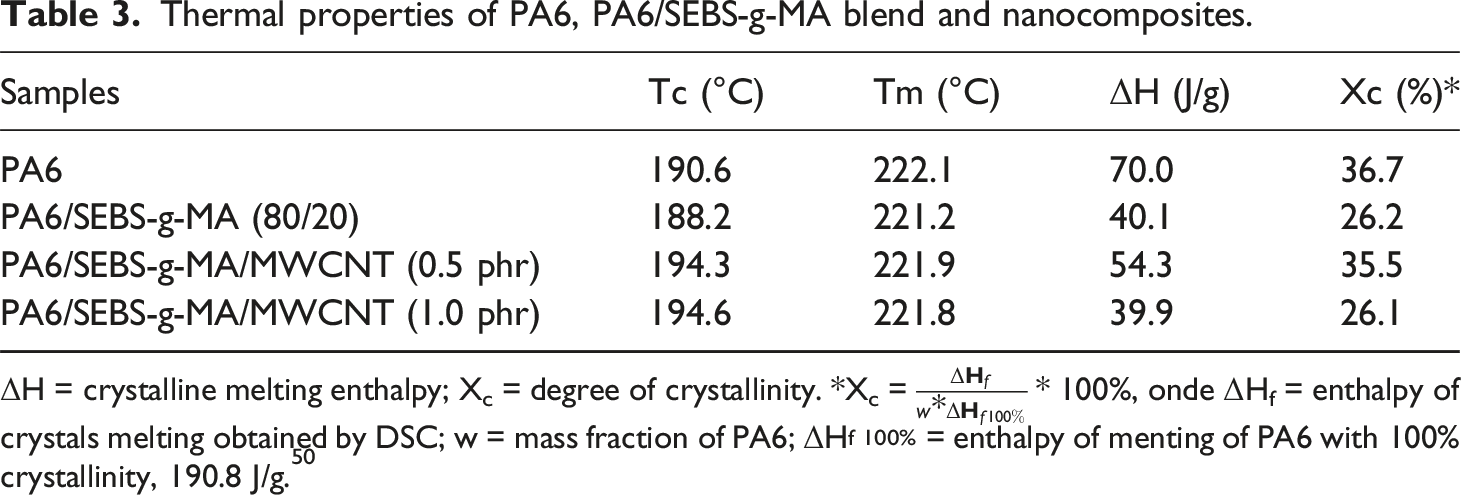
The crystalline melting temperature (Tm) of PA6 was approximately 222.1°C, indicating the predominance of the α crystalline form, which is the most thermally stable, 51 in agreement with the XRD results. The literature 52 reports that PA6 may develop two crystalline phases, α and γ, depending on the processing conditions or the influence of additives. The PA6/SEBS-g-MA blend and the PA6/SEBS-g-MA/MWCNT nanocomposites maintained a Tm close to 222°C, a value similar to that of neat PA6. This behavior suggests that the dominant crystalline structure of PA6 remained unchanged in the samples, preserving the α phase. Consequently, the lamellae that melted exhibited comparable thermal stability, which is reflected in the consistent Tm values. Regarding the melting enthalpy, it was observed that it varied proportionally to the degree of crystallinity of each sample, which is expected since it corresponds to the energy required to melt the crystalline fraction.
The crystallinity degree (Xc) of PA6 was 36.7%, confirming its semicrystalline nature. The addition of 20% SEBS-g-MA to PA6 reduced this value to 26.2%, indicating inhibition of both nucleation and crystal growth processes. As observed in the torque rheometry analysis, the PA6/SEBS-g-MA blend exhibited a significant increase in viscosity compared with neat PA6. This increase in viscosity may hinder the proper packing of PA6 chains, leading to a reduction in crystallinity, as shown in Table 3. The incorporation of 0.5 phr MWCNT into the PA6/SEBS-g-MA blend promoted a recovery of the crystallinity degree to 35.5%, a value similar to that of neat PA6. Although viscosity increased in the PA6/SEBS-g-MA/MWCNT (0.5 phr) nanocomposite, this specific amount of MWCNT favored a nucleating effect, resulting in the formation of a larger quantity of crystals. On the other hand, increasing the concentration to 1 phr MWCNT led to a decrease in crystallinity to 26.1%. At concentrations above 0.5 phr MWCNT, crystal formation in the PA6/SEBS-g-MA blend was inhibited, possibly due to a reduction in the effective surface area and, consequently, in the nucleating capability of the nanotubes.
Raman Spectroscopy
Figure 6 displays the Raman spectra obtained for pure PA6, the PA6/SEBS-g-MA blend, and the nanocomposites as a function of the MWCNT concentration. Raman spectra of pure PA6, PA6/SEBS-g-MA blend and nanocomposites.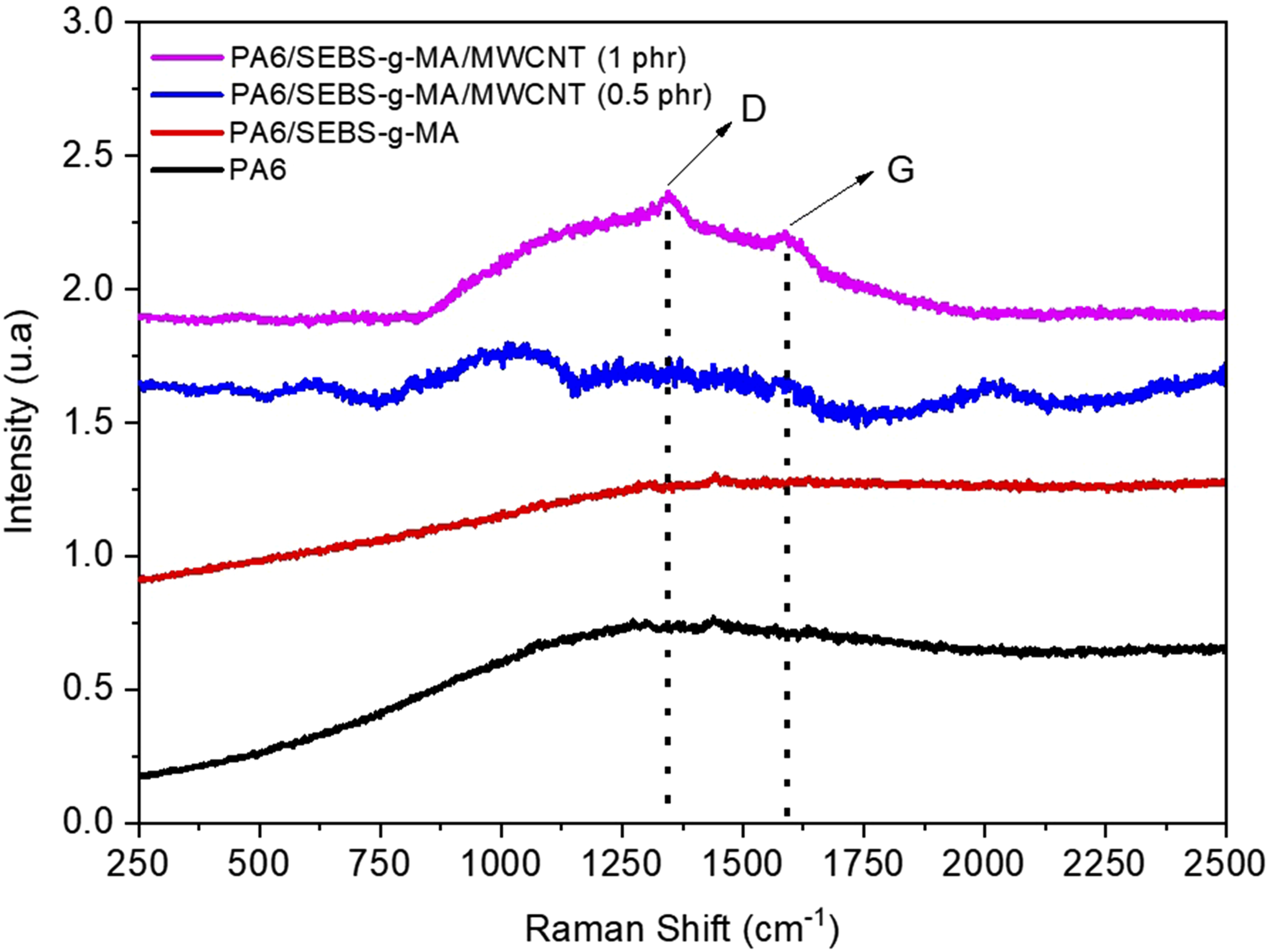
The application of a 532 nm laser was unable to excite characteristic bands for pure PA6 and the PA6/SEBS-g-MA blend. A similar behavior was observed for the PA6/SEBS-g-MA/MWCNT nanocomposite containing 0.5 phr, suggesting that the low concentration of carbon nanotubes was effectively encapsulated in the polymer matrix. However, with the addition of 1 phr MWCNT to the PA6/SEBS-g-MA blend, two distinct bands appeared, as shown in Figure 5. The band at 1343 cm−1 corresponds to the D band, which is associated with structural defects, while the band at 1589 cm−1 corresponds to the G band, related to the longitudinal vibration of carbon atoms in the graphene plane. These findings align with those reported by Cha et al. 53 and Nunes et al. 54 The detected presence of carbon nanotubes well-distributed in the PA6/SEBS-g-MA matrix indicates their accommodation in the polymer chains, which contributes to the increased viscosity observed in the torque rheometry analysis.
Scanning Electron Microscopy (SEM)
Figure 7(a)-(d) displays SEM micrographs of the fracture surfaces for (a) neat PA6, (b) the PA6/SEBS-g-MA (80/20) blend, and the nanocomposites (c) PA6/SEBS-g-MA/MWCNT (0.5 phr) and (d) PA6/SEBS-g-MA/MWCNT (1.0 phr). In Figure 7(a), the fracture surface of neat PA6 appears predominantly smooth, indicating limited plastic deformation. However, the presence of some surface roughness suggests pseudo-ductile fracture behavior, characterized by localized plastic deformation zones coexisting with regions of brittle fracture. For the PA6/SEBS-g-MA blend, shown in Figure 7(b), a heterogeneous phase morphology is observed, with PA6 acting as the matrix phase and SEBS-g-MA forming dispersed, spherical domains. Some SEBS-g-MA particles exhibit weak interfacial adhesion, and evidence of cavitation (void formation) can be seen in the fracture surface. From a fracture mechanics perspective, the blend clearly exhibits a ductile fracture mechanism, characterized by a rough surface texture. This indicates improved resistance to crack propagation and a higher capacity for energy dissipation, which correlates with the enhanced impact strength discussed later. Photomicrographs obtained by SEM of the fracture surface, for: (a) pure PA6; (b) PA6/SEBS-g-MA; (c) PA6/SEBS-g-MA/MWCNT (0.5 phr); (d) PA6/SEBS-g-MA/MWCNT (1.0 phr).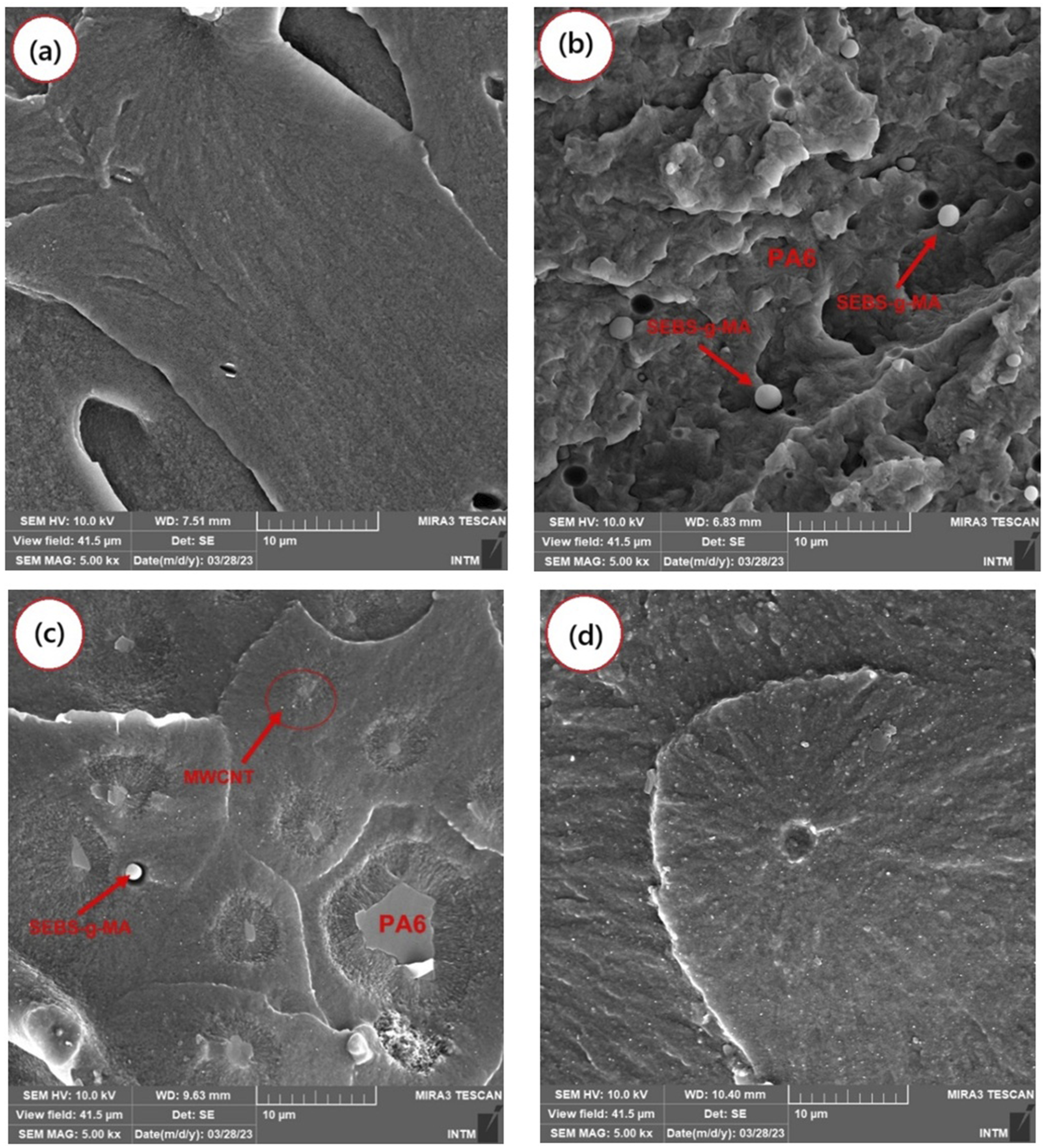
A distinctly different surface morphology was observed with the incorporation of carbon nanotubes into the PA6/SEBS-g-MA system, as shown in Figure 7(c) and (d). Notably, the ductile fracture mechanism became less pronounced in the PA6/SEBS-g-MA/MWCNT nanocomposites compared to the base blend. However, the toughening effect was still evident when compared to neat PA6, as indicated by the rough surface. Both nanocomposites containing 0.5 and 1.0 phr of MWCNT exhibit heterogeneous phase morphologies. In the PA6/SEBS-g-MA/MWCNT (0.5 phr) composition, spherical domains corresponding to the dispersed SEBS-g-MA phase in the PA6 matrix can still be clearly observed. However, with the addition of 1.0 phr MWCNT, the nanocomposite exhibits a more refined morphology. This observation suggests that at this concentration, MWCNTs contribute to morphological stabilization. A similar behavior has been reported in the literature, 55 where carbon nanotubes are described as reducing the coalescence of the dispersed phase through steric hindrance.
Figure 8 shows SEM micrographs of the fracture surfaces at 10,000x magnification for the PA6/SEBS-g-MA/MWCNT nanocomposites containing 0.5 phr and 1.0 phr of carbon nanotubes. In the nanocomposite with 0.5 phr MWCNT (Figure 8(a)), carbon nanotubes are visible, dispersed in the PA6 matrix, with some regions exhibiting fibrillar structures (indicated by arrows). This suggests that the material retains a toughened behavior at room temperature. The addition of 1.0 phr MWCNT (Figure 8(b)) results in a more heterogeneous fracture surface, attributed to the higher concentration of nanotubes, an observation that aligns with the findings from Raman spectroscopy. Photomicrographs of the fracture surface in the region with 10,000x magnification, for the nanocomposites: (a) PA6/SEBS-g-MA/MWCNT (0.5 phr) and (b) PA6/SEBS-g-MA/MWCNT (1.0 phr).
Mechanical Properties
In Figure 9(a), although PA6 is classified as an engineering polymer, its notched impact strength was relatively low, measuring 75.6 J/m. The incorporation of 20 wt% SEBS-g-MA into the PA6 matrix significantly enhanced toughness, resulting in a 262.7% increase in impact strength compared to neat PA6. This indicates that SEBS-g-MA effectively improved energy dissipation in the matrix, reducing the likelihood of premature failure under impact conditions. When 0.5 phr of MWCNTs was added to the PA6/SEBS-g-MA blend, a reduction in impact strength was observed, dropping to 182.3 J/m compared to the unfilled blend. Nevertheless, the material retained a ductile fracture behavior at room temperature and still exhibited a 141.1% improvement over neat PA6. A change in behavior was noted with the addition of 1.0 phr MWCNT, as the impact strength of the PA6/SEBS-g-MA/MWCNT nanocomposite recovered, indicating a positive effect at this higher nanotube concentration. This behavior suggests the existence of a critical MWCNT concentration that optimizes impact strength by enhancing the material’s ability to hinder crack propagation. As observed in the SEM analysis, the PA6/SEBS-g-MA/MWCNT (1 phr) nanocomposite exhibited a more uniform distribution of MWCNTs, which likely served as effective sites for crack deflection or energy dissipation during impact testing. A similar trend was reported by Wang et al.
56
in PLA/EVA nanocomposites containing MWCNTs, highlighting the potential of carbon nanotubes to improve impact performance through microstructural reinforcement. Mechanical behavior of PA6, PA6/SEBS-g-MA blend and nanocomposites, for: (a) impact strength; (b) elastic modulus; (c) tensile strength; (d) elongation at break; (e) stress versus strain curves.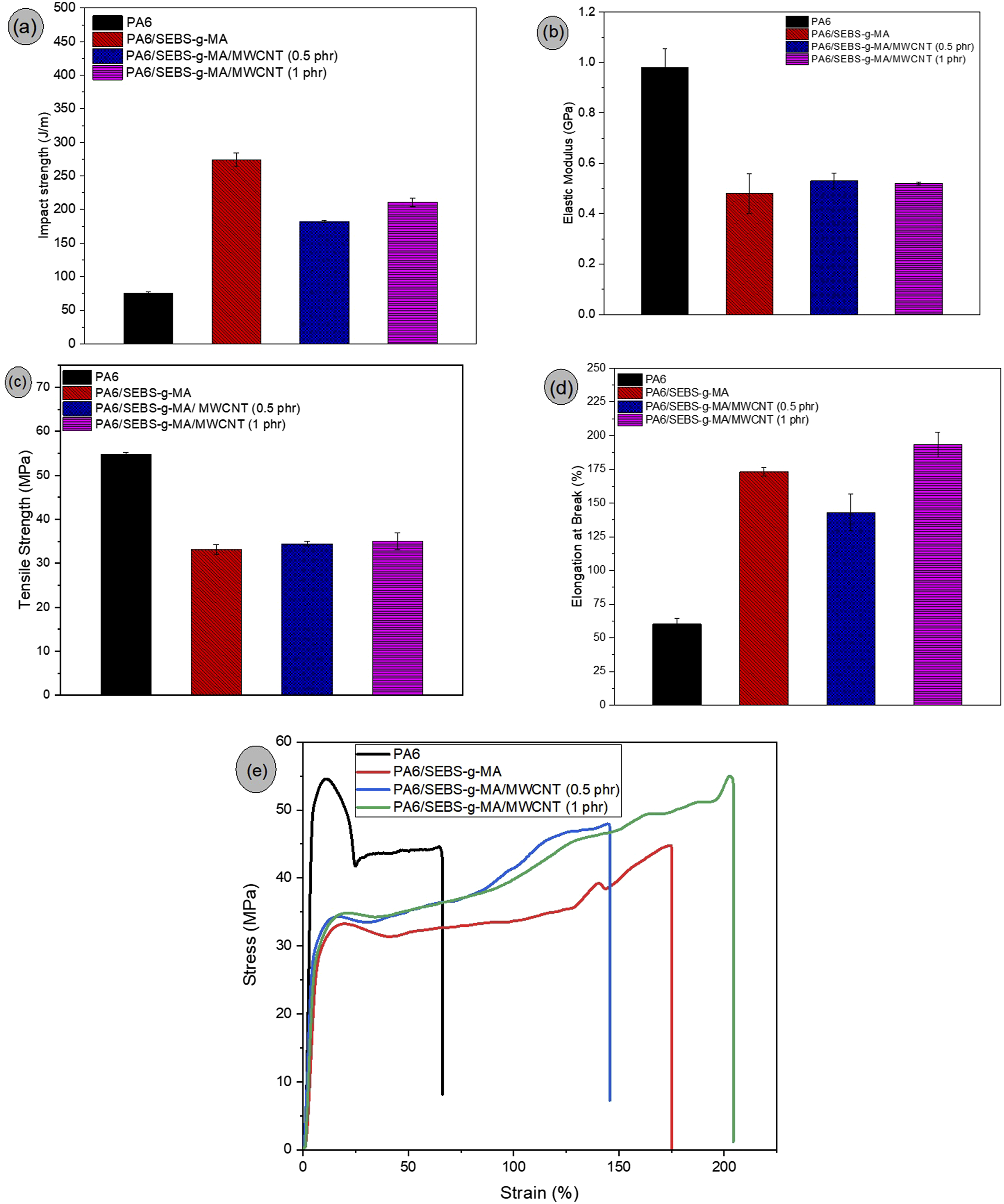
In Figure 9(b), PA6 exhibited the highest elastic modulus (0.98 GPa), indicating the greatest stiffness among the samples analyzed. The addition of SEBS-g-MA led to a reduction in stiffness, which is attributed to the increased flexibility introduced by the elastomeric phase. The incorporation of 0.5 and 1.0 phr of MWCNTs into the PA6/SEBS-g-MA blend resulted in a slight increase in the elastic modulus, suggesting that the carbon nanotubes were accommodated within the polymer matrix, as evidenced by Raman spectroscopy, thereby restricting molecular mobility. However, considering the experimental error margin of the PA6/SEBS-g-MA system, the elastic modulus values of the nanocomposites can be regarded as comparable. Regarding tensile strength, as shown in Figure 9(c), neat PA6 required a high tensile load to undergo deformation, reflecting its inherent strength. The addition of SEBS-g-MA to PA6 led to a noticeable decrease in tensile strength, indicating reduced resistance to elastic deformation. This reduction is attributed to the elastomeric nature of SEBS-g-MA, which contributes flexibility but compromises tensile performance. The formulation containing 0.5 phr of MWCNTs showed a slight 4% increase in tensile strength compared with the PA6/SEBS-g-MA blend, indicating a mild reinforcing effect. The addition of 1.0 phr of MWCNTs resulted in a 7% recovery in tensile strength compared with the base system. The improved performance can be attributed to the uniform distribution of MWCNTs within the PA6/SEBS-g-MA blend, as observed in the SEM analysis, which enabled a more effective reinforcing effect and better stress transfer.
Figure 9(d) illustrates the ductility behavior of the materials, with neat PA6 showing the lowest elongation at break, approximately 60.1%. When PA6 was modified with SEBS-g-MA, a significant increase in elongation was observed, reaching 173.1%. This enhancement in ductility is consistent with the improvements noted in impact strength and suggests a synergistic interaction between the components of the blend that promotes plastic deformation mechanisms. Since elongation at break is measured in the plastic deformation region, the results imply that the PA6/SEBS-g-MA blend exhibits compatibility, contributing to its improved ductile behavior. The incorporation of 0.5 phr of MWCNTs led to a slight decrease in elongation to 143% compared to the unfilled blend, but the value remained well above that of neat PA6. The most notable performance was observed for the PA6/SEBS-g-MA/MWCNT (1 phr) nanocomposite, which exceeded 190% elongation at break. These findings indicate the formation of toughened nanocomposites at room temperature with both 0.5 and 1.0 phr MWCNT concentrations. Figure 9(e) shows the stress–strain curves for neat PA6, the PA6/SEBS-g-MA blend, and the nanocomposites. Neat PA6 exhibited the typical behavior of a tough and ductile material, with plastic deformation after yielding. The degree of ductility increased significantly for the PA6/SEBS-g-MA blend, indicating that the elastomeric fraction interacted with the PA6 matrix, leading to greater deformation. The nanocomposites containing 0.5 phr and 1 phr of MWCNTs in PA6/SEBS-g-MA maintained ductile behavior at room temperature, confirming the trend observed in the impact strength results.
Figure 10 presents a proposed mechanism for the PA6/SEBS-g-MA blend and the PA6/SEBS-g-MA/MWCNT nanocomposites containing 0.5 phr and 1 phr of MWCNT. Suggested toughening mechanism for the PA6/SEBS-g-MA blend and the nanocomposites.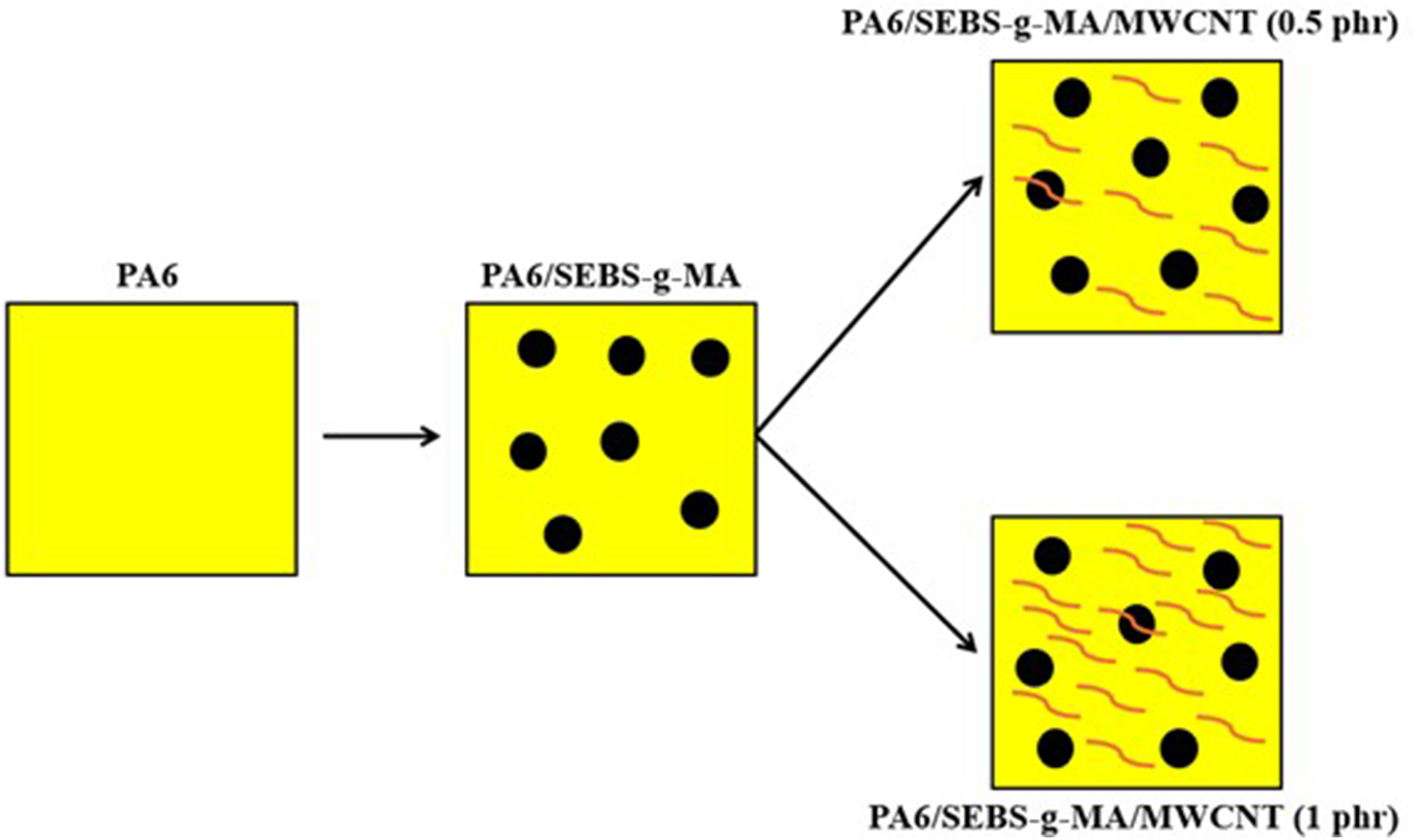
PA6 has a homogeneous morphology with no dispersed phase to aid in the toughening process. As observed in the mechanical tests, PA6 achieved 75.6 J/m for impact strength and 60.1% in elongation at break, suggesting a good level of deformation before fracture. Although it is an engineering polymer, PA6 exhibits a pseudo-ductile behavior, as reported in the literature. 57 I. e, PA6 is ductile when not notched but fractures in a brittle manner when notched, due to its low resistance to crack propagation. 58 Considering this, 20% SEBS-g-MA was added to improve the performance of PA6. It can be observed in Figure 10, for the PA6/SEBS-g-MA system, the formation of a biphasic structure with spherical SEBS-g-MA particles dispersed in the PA6 matrix, which is consistent with the morphology presented in Figure 7(b). When PA6 is subjected to mechanical stress, the SEBS-g-MA phase contributes to inducing toughening mechanisms, such as shear banding and microfibrillation, thus enhancing the energy dissipation level. 59 Consequently, there were significant gains in impact strength and elongation at break, as previously observed in the mechanical tests. When MWCNT is added to PA6/SEBS-g-MA, a hybrid morphology is formed with SEBS-g-MA and MWCNT dispersed in PA6. This contributes to energy dissipation in SEBS-g-MA and crack deflection in the carbon nanotubes, especially for the nanocomposite with a higher MWCNT content (1 phr). Since the PA6/SEBS-g-MA/MWCNT (1 phr) nanocomposite exhibited the best performance in ductility, there was likely a synergy for greater crack deflection and energy dissipation in the PA6 matrix, leading to enhanced toughening performance.
Heat Deflection Temperature (HDT)
Thermomechanical strength by HDT of PA6, polymer blend and nanocomposites.

The HDT of pure PA6 was measured in the 53.7°C, consistent with values reported in the literature.51,57 The PA6/SEBS-g-MA blend showed a slight decrease in HDT to 52.5°C, likely due to the increased flexibility imparted by the SEBS-g-MA. Additionally, the HDT values for the PA6/SEBS-g-MA/MWCNT nanocomposites remained largely unchanged, closely matching those observed for the PA6/SEBS-g-MA blend.
Conclusion
This study demonstrated that incorporating small amounts of carbon nanotubes (MWCNTs) into PA6/SEBS-g-MA (80/20) blends is an effective strategy to achieve a balanced set of mechanical properties. Torque rheometry revealed increased viscosity and improved compatibility between the PA6 and SEBS-g-MA phases, while FTIR, DSC and XRD analyses confirmed the predominance of the α-crystalline phase of PA6. Raman spectroscopy detected the presence of MWCNTs at 1 phr, which was supported by SEM observations showing good nanotube distribution. From a mechanical perspective, the addition of SEBS-g-MA led to a significant increase in the toughness and ductility of PA6, with improvements exceeding 262% in impact strength and 187% in elongation at break. The incorporation of 0.5 phr MWCNT maintained notable gains in impact strength and ductility compared to neat PA6, although with a slight decrease relative to the base blend. The formulation containing 1 phr MWCNT demonstrated superior results in both impact strength and elongation at break, as well as recovery of tensile strength, indicating a synergistic effect between SEBS-g-MA and the nanotubes in the PA6. Overall, these results highlight the technological potential of PA6/SEBS-g-MA/MWCNT nanocomposites, particularly at a concentration of 1 phr, as materials that are impact strength at room temperature, flexible, and melt-processable.
It is important to highlight that the functionalization of carbon nanotubes can play a decisive role in improving their distribution and dispersion, as in the PA6/SEBS-g-MA blend. Functional groups added to the surface of carbon nanotubes tend to increase their chemical affinity with polar phases, promoting stronger interfacial interactions, greater efficiency in stress-transfer processes, and reduced agglomeration. Thus, future studies involving functionalized MWCNTs may deepen the understanding of the reinforcement mechanisms in the PA6/SEBS-g-MA blend and reveal additional gains in mechanical performance, morphological stability, and structural uniformity of the nanocomposites.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: There are no competing or potentially conflicting interests among the authors related to the research, authorship, or publication of this manuscript. Furthermore, responsibility for the content of the article is equally shared by all authors.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support provided by the Foundation for Science and Technology Support of the State of Pernambuco (FACEPE), under grant number APQ-JP-0933-3.03/21. We also extend our gratitude to the Federal University of Pernambuco (UFPE) and the Federal University of Campina Grande (UFCG) for their collaboration and for providing the necessary infrastructure to carry out this research. Additionally, we recognize the National Council for Scientific and Technological Development (CNPq) for funding through research scholarships awarded to Carlos Bruno Barreto Luna (grant number 350025/2023-1) and Edcleide Maria Araújo (grant numbers 312014/2020 and 408779/2022-5).
