Abstract
Rare earth solution (RES) surface modification and acid treatment methods were used to functionalize carbon nanotubes (CNTs). CNT composite thin films were prepared on hydroxylated glass substrates by a self-assembling process from specially formulated solution. Atomic force microscopy, x-ray photoelectron spectrometry, and scanning electron microscopy were used to characterize the thin films. Tribological properties of the CNT thin films sliding against a steel ball were evaluated on a friction and wear tester. The experimental results showed that the friction coefficient of the glass substrate was reduced from 0.85 to 0.1 after the formation of CNT composite self-assembled film on its surface, and the film exhibited good wear resistant property. The superior friction reduction and wear resistance of CNT composite films were attributed to the improvement of load-bearing capacity afforded by the chemical bond between CNTs and the films as well as good adhesion of the films to the substrate.
Keywords
Introduction
Over the last decade, the field of microelectromechanical systems (MEMS) has expanded considerably, catering to a wide variety of applications ranging from industrial, consumer, defense, aerospace, to biomedical applications. 1 Miniaturized devices are traditionally made from silicon (Si) due to the well-established designs and fabrication processes related to the integrated circuit technology. However, Si does not have good tribological properties. 2 In order to improve the tribological properties of Si, researchers worldwide have resorted to a variety of surface modification techniques such as chemical and topographical modifications. Chemical modification includes thin films/coatings such as self-assembled monolayers (SAMs) and diamond-like carbon coatings. 3 Obviously, it is essential to develop super lubrication method for MEMS to advance their practical applications.
Since their discovery in 1991, 4 carbon nanotubes (CNTs) have attracted much interest of researchers because they possess many unique properties, 5 –7 such as high tensile and flexural strength, high elastic modulus, and self-lubrication property. If CNTs are aligned to form nanotube composite films, 8 they can provide both mechanical support and self-lubrication for the films. Also, theoretical calculations indicate that, in analogy to the well-known lubricating properties of van der Waals bonded graphite, the individual cylinders of CNTs might be expected to easily slide or rotate with respect to one another. Such unique properties of CNTs make them potentially useful in the lubrication of the interfaces between moving components in MEMS.
However, it is known that CNTs are not compatible with all solvents because of their chemical inertness, and it is very hard to obtain a suitable modifier to improve their surface activities. These properties lead to poor dispersion capability and weak bonding of CNTs when they are used in composite films as reinforcing elements. Many studies have been conducted to enhance the adhesion between CNT and substrate by surface treatment of CNTs. 9 However, the interfacial adhesion between CNTs and substrate was not strong enough by these methods, which affected the mechanical and tribological properties of the composite films.
Recently, several authors demonstrated that the reinforcement of polymers 10 and nanoparticles can significantly improve mechanical strength and wear resistance. Interestingly, reinforcement by conventional micrometer-sized inorganic particle had a deleterious effect on wear because the particles get detached from the composites during wearing, act as an abrasive, and induce a three-body wear mechanism. In case of nanoparticles, the bonding with the matrix is stronger due to the higher surface area-to-volume ratio. This yields a higher reinforcement effect and better mechanical resistance. Moreover, the particle angularity, and, thus, their abrasivity, 11 is remarkably reduced with decreasing particle size.
RES surface treatment has been successfully applied to improve the adhesion between carbon fiber and their matrixes 12 –14 in composites. In this study, RES was applied in the CNTs surface modification, and CNTs composite thin films were prepared on glass substrates by self-assembly process. The films were characterized and their tribological properties were investigated by means of many advanced analyzing methods. Also, the mechanism of the formation of the self-assembled composite film and the effect of CNTs on tribological behaviors of the films were discussed.
Experimental procedure
Sample preparation
Materials
The purified CNTs (purchased from Nanotech Port Co. Ltd, Shenzhen, China) were produced from carbon monoxide by chemical vapor deposition method. Glass plates were used as substrates for the transfer of SAM film. The glass plates were cut into pieces with a surface size of approximately 10 × 10 mm2 before cleaning and modifying. 3-Mercaptopropyl trimethoxysilane (MPTS) was purchased from Sigma Aldrich (Deisenhofen, Germany) and used as received. All other chemicals used in chemical manipulations were of analytical reagent grade. Deionized water used in this study was purified using Barnstead Nanopure apparatus (Thermo, Shanghai).
Si wafers were treated in freshly prepared Piranha solution (volume ratio 7:3 of 98% sulfuric acid (H2SO4) and 30% hydrogen peroxide (H2O2)) at 90.1°C for 2 h to get a hydroxyl-terminated surface. The resulting modified Si substrates were extensively washed with dichloromethane for three times to remove the unbonded silane-coupling agents by ultrasonic cleaning. The hydrogen-terminated Si wafers were obtained by etching in a saturated aqueous solution of ammonium fluoride (mass fraction 40%) for 10 min.
CNT modification
Two surface modification methods were used in the CNT treatment: acid modification and RES modification (the reaction procedure is shown in Figure 1). The typical acid treatment process was as follows: 200 mg of purified CNT was suspended in 400 ml of a 3:1 mixture of concentrated H2SO4(90%)/nitric acid (HNO3; 70%) and sonicated in a water bath for 22 h at 50°C. The resultant acid-treated CNTs were collected on a polytetrafluoroethylene filter with the pore size of 500 nm (H050A047A, Advantec Co. Ltd, Ehime, Japan) and then washed for four times with water and methanol, respectively. The RES modification process was as follows: the mass ratios of CNTs to RES (1:2) were uniformly immersed into RES for 10 min and then were dried for 4 h at 120°C. Rare earth compound lanthanum(III) chloride (LaCl3) purchased from Yuelong New Materials Co. Ltd (Shanghai, China) was used as the main component of RES applied in surface modification, and the concentration of LaCl3 in RES was 0.3 wt%. LaCl3, ethylenediaminetetraacetic acid (EDTA), ammonium chloride, and hydrogen nitrate were dissolved in ethanol to prepare the RES. The final pH of the solution was 5. EDTA, ammonium chloride, and hydrogen nitrate were commercially obtained without further purification.
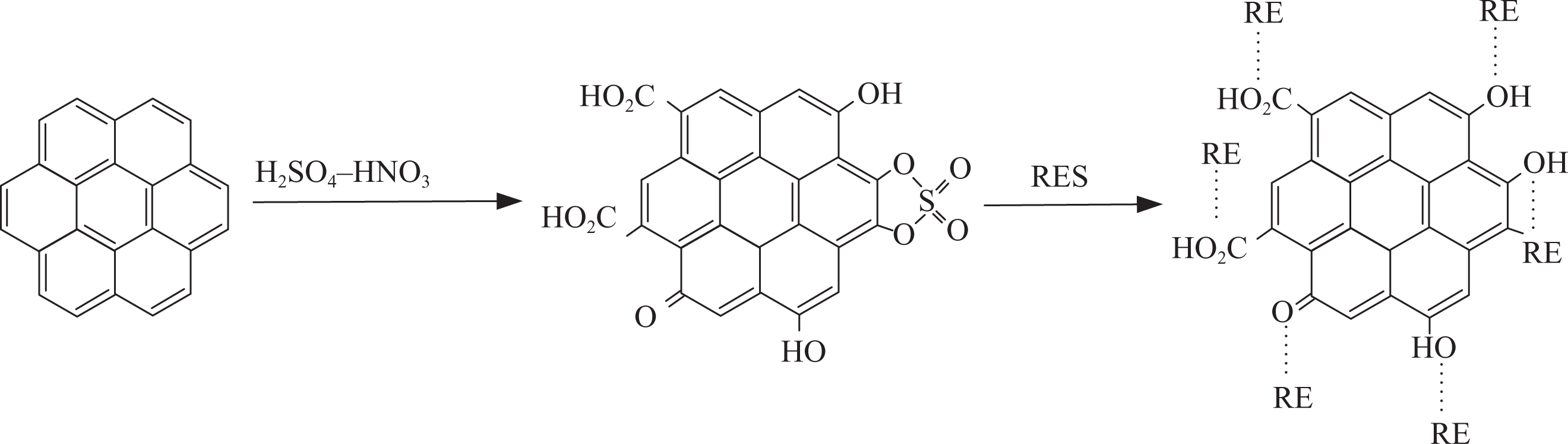
The schematic diagram of the functionalization of CNTscarbon nanotubes.
Preparation process of CNT composite films
First, preparation of MPTS self-assembled film was performed using the following procedure: with proper prehydration, the hydroxide glass substrates were capable of bonding covalently to the hydrolyzed silane molecules via condensation reaction of silanol groups. Glass substrates were immersed in Piranha solution (H2SO4:H2O2 = 7/3(v/v)) at 90°C for 30 min to produce hydroxy radicals on the surfaces. Then, the substrates were carefully rinsed with deionized water and dried. After that the hydroxide substrates were dipped into dehydrated benzene solution containing 0.5 mM of MPTS solution with polyethylene for 12 h, to allow the formation of self-assembled MPTS SAM. Finally, the substrates were cleaned ultrasonically with chloroform, acetone, and deionized water in turn to remove other physisorbed ions or molecules and dried for 1 h at 120°C and then cooled in a desiccator. The oxidization of SH groups to the desired SO3H groups was carried out by dipping the substrates into a solution of 30% HNO3 at 80°C for 1 h, followed by washing with distilled water and dried in nitrogen, and sulfonated MPTS-SAM film was obtained.
The chemically modified CNTs (2 mg) were thoroughly dispersed in 20 ml of N,N-dimethylformamide with sonication for 2 h at room temperature. Then, the substrates deposited with sulfonated MPTS-SAM film were immersed in the resulting mixed solution at 90°C for 12 h, to allow the assembly of CNT composite films. Finally, the substrates were cleaned ultrasonically with deionized water to remove other physisorbed ions or molecules and dried for 2 h at 120°C and then cooled in a desiccator.
Experimental apparatus and measurements
Microscopic study was conducted on NanoScope IIIa atomic force microscopy (AFM; Digital Instruments Inc., Tonawanda, New York, USA) in contact mode. All measurements were performed in the air atmosphere at 23°C and 37% relative humidity (RH). X-Ray photoelectron spectrometry (XPS) analysis was conducted on PHI-5702 XPS/Auger electron spectroscopy system (Shenzhen Sansi, Shenzhen). During the XPS analysis, a 250-W aluminum Kα line (hν = 148616 eV) was used, the pass energy was 29.350 eV, and C1s with the binding energy of 284.6 eV was used as the reference. Contact angles of water were measured by sessile drop method using OCA-20 measurement apparatus (DataPhysics Instruments GmbH, Filderstadt, Germany) under ambient laboratory conditions (24°C and 40% RH). A deionized water droplet (approximately 20 µl) was applied to the substrate surface, allowed to settle for several seconds, and then photographed on a slow-scan charge-coupled device camera. Measurements were made at a minimum of five different spots on each sample. The results stated below were averages of all these measurements. The worn surfaces of the films were investigated using a scanning electron microscope (SEM; model CSM950, OPTON Co. Ltd, Munchen, Germany).
Tribological tests were carried out on the wafer before and after hard baking at 150°C for 2 h. The friction and wear tests were carried out on a UMT-2MT reciprocating friction tester, the main part of the apparatus is shown schematically in Figure 2. The prepared films on the glass substrates were rubbed by a GCr15 steel ball with a diameter of 3 mm in an ambient condition (RH: 40–44%). The composition of GCr15 bearing steel ball comprises of carbon: 0.95–1.05 wt%, Si: 0.15–0.35 wt%, manganese: 0.20–0.40 wt%, chromium: 1.30–1.65 wt%, and the remaining amount by iron, with a bulk hardness of HRC65 ± 5 was used as the counterpart. The steel ball was cleaned with acetone on supersonic bath before each test. The sliding distance for each pass is 5 mm. The coefficient of friction and the number of sliding passes were recorded automatically. In the whole sliding process, the friction coefficient remains stable with little fluctuation for a long period and rises to a higher stable value at last dozen of passes. The corresponding sliding pass numbers are recorded as the wear life of the film. Three replicate tests were carried out for each specimen. The average friction coefficients and wear lives of the three replicate tests are cited in this article. The relative error for the replicate tests was no more than 5%.
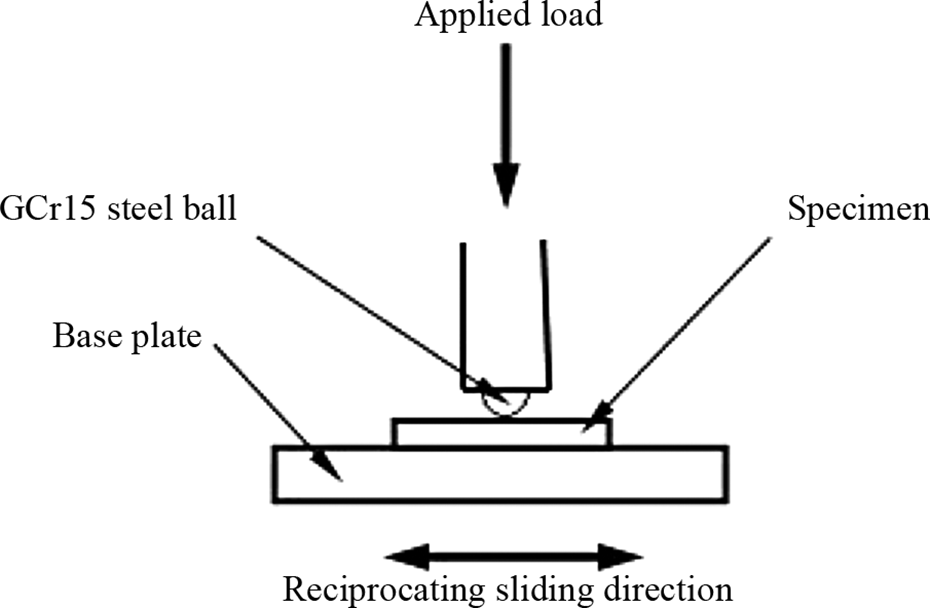
The schematic diagram of the friction pair.
Results and discussion
XPS analysis of the CNT surfaces
Figure 3 shows typical wide-scan photoelectron spectra of (a) untreated CNT and (b) RES-treated CNT. Apart from strong C-signals (C1s photoelectron), the spectrum of the untreated CNT (Figure 3(a)) shows relatively weak O-signals (O1s), which is due to the near surface oxygen-containing functional groups of the nanotubes. The effect of RES treatment can be distinctly seen in Figure 3(b) where the spectrum shows strong oxygen lines and a weak graphite line.
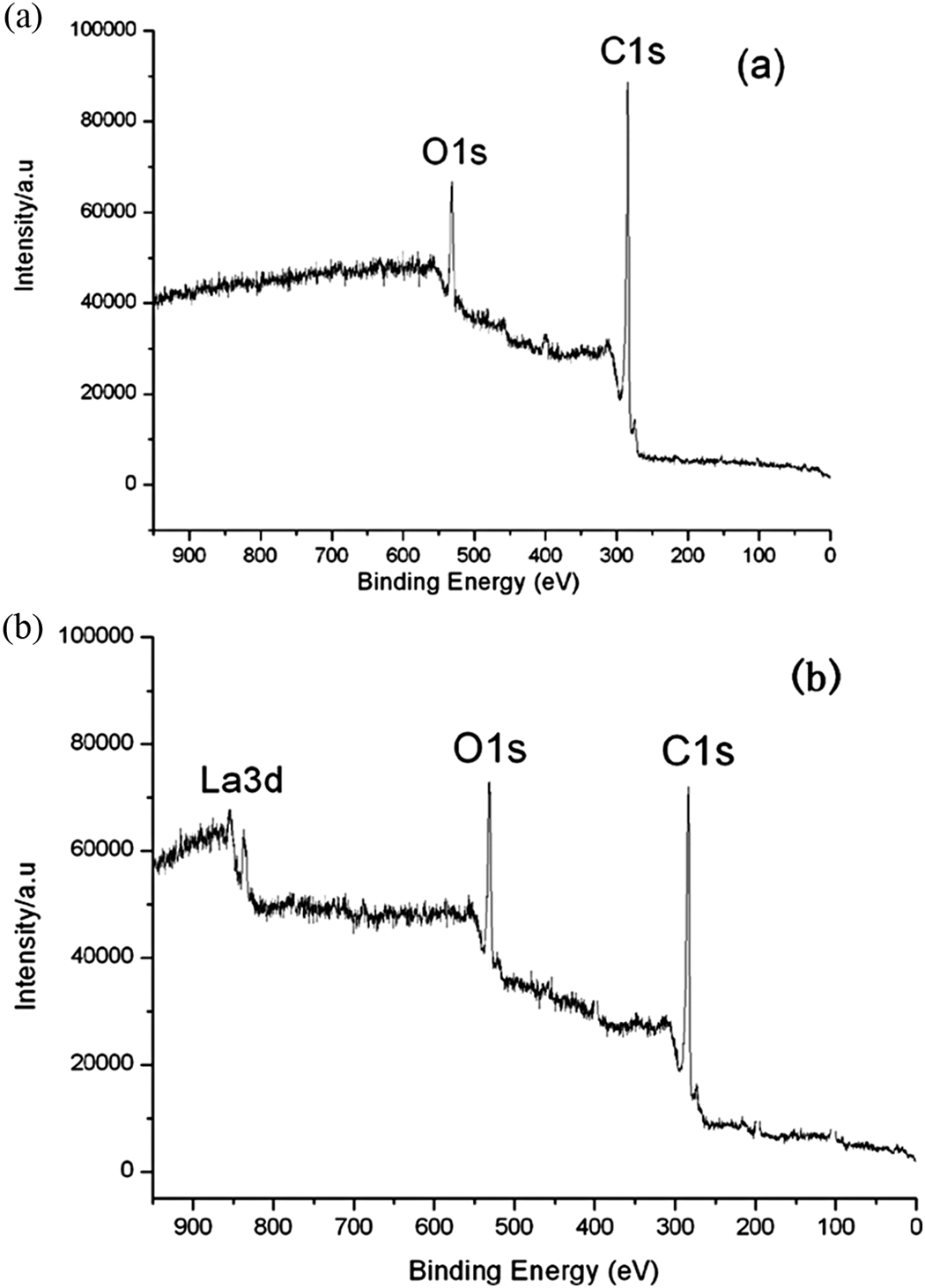
Wide-scan photoelectron spectra of carbon nanotubes.
The difference in binding energies for La3d is very small and deconvolution of the peaks is necessary to analyze XPS data (Figure 4). The dominant peak at 853.0 eV was assigned to La3d in LaCl3. The 852.3 eV peak shows that the rare earth compounds are capable of coordinating and forming ionic combination when reacting with organic functional groups (such as hydroxyl and carboxyl groups on the CNT) during the process of CNT treatment. 15
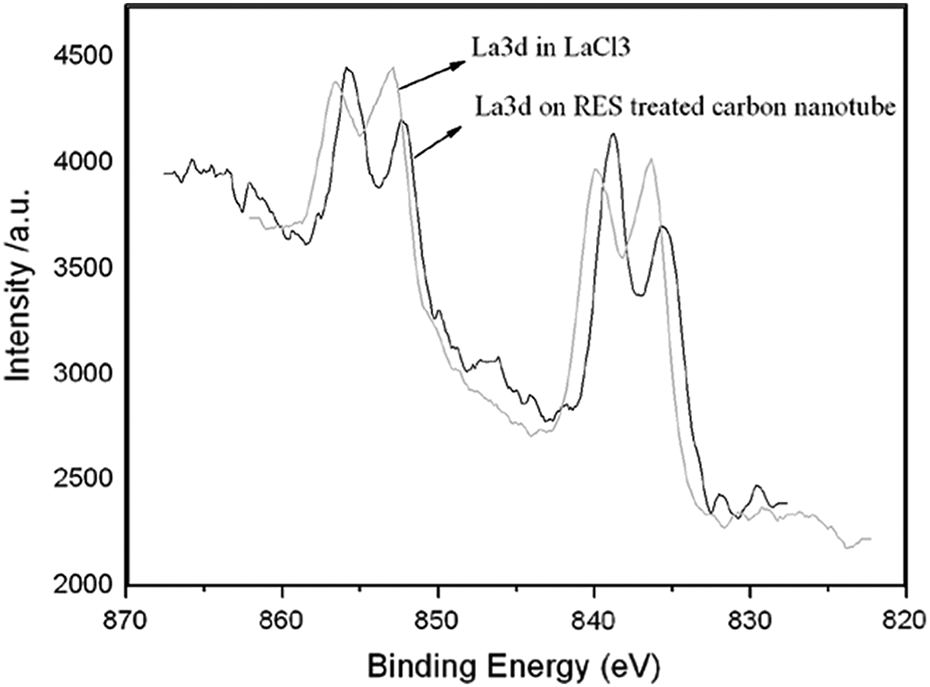
XPS spectra of La3d and carbon nanotube. XPS: x-ray photoelectron spectroscopy; La: lanthanum; CNT: carbon nanotube.
Characterization of MPTS-SAM and CNT composite thin film on sulfonated MPTS-SAM
Figure 5 shows AFM images of hydroxide substrates, MPTS-SAM and oxidized MPTS-SAM, respectively. It is seen that the mean roughness (R a) of the hydroxide substrates is 0.159 nm, of MPTS-SAM is 0.527 nm, and that of oxidized MPTS-SAM is 0.2 nm. From the AFM images and the value of R a, it implies that some molecules have been absorbed onto the substrate. From the SEM image of CNT composite thin films (as shown in Figure 6), distribution of CNTs is dispersive, and single CNT can be observed clearly, which is shown that CNTs are deposited onto the substrate.

AFM images of (a) hydroxide substrate, (b) MPTS- SAM, and (c) sulfonated MPTS-SAM. AFM: atomic force microscopy; MPTS: 3-mercaptopropyl trimethoxysilane; SAM: self-assembled monolayer.
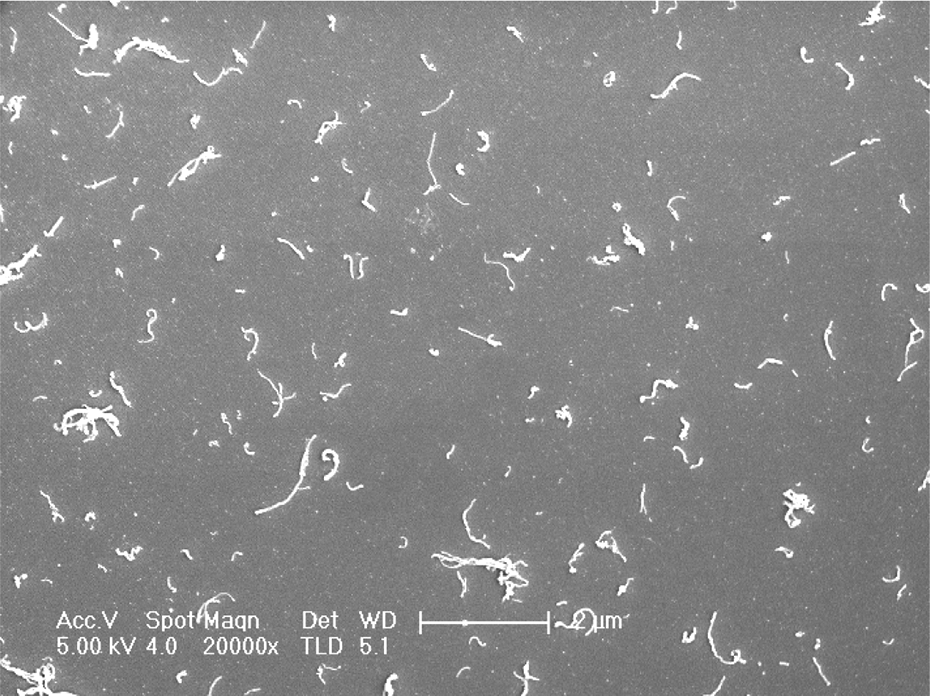
SEM images of CNT composite thin film. SEM: scanning electron microscopy; CNT: carbon nanotube.
In the high resolution scan of XPS spectra of the surface (Figure 7(a)), the S2p peak located at 163 eV is the characteristic of sulfur in –SH group. 16 For the sample surface obtained by oxidizing surface of MPTS film in situ with 30% HNO3, the appearance of an S2p binding energy peak at 168.4 eV is the characteristic of sulfur in –SO3H groups (Figure 7(b)). This indicates that the oxidizing procedure has effect on the terminal –SH groups. Figure 7(c) shows the XPS spectra of La3d on the surface of sulfonated MPTS-SAM coated with RES-modified CNT. The La3d core level band of surface also decomposes into four peaks at binding energies of 835, 838.9, 851.79, and 855.2 eV, but the characteristic of La3+ is 834.8 eV. 17 It can be concluded that La element could react with oxygen element of –SO3H group on the surface of the substrate, which indicates that CNT composite film has been successfully obtained in our work.
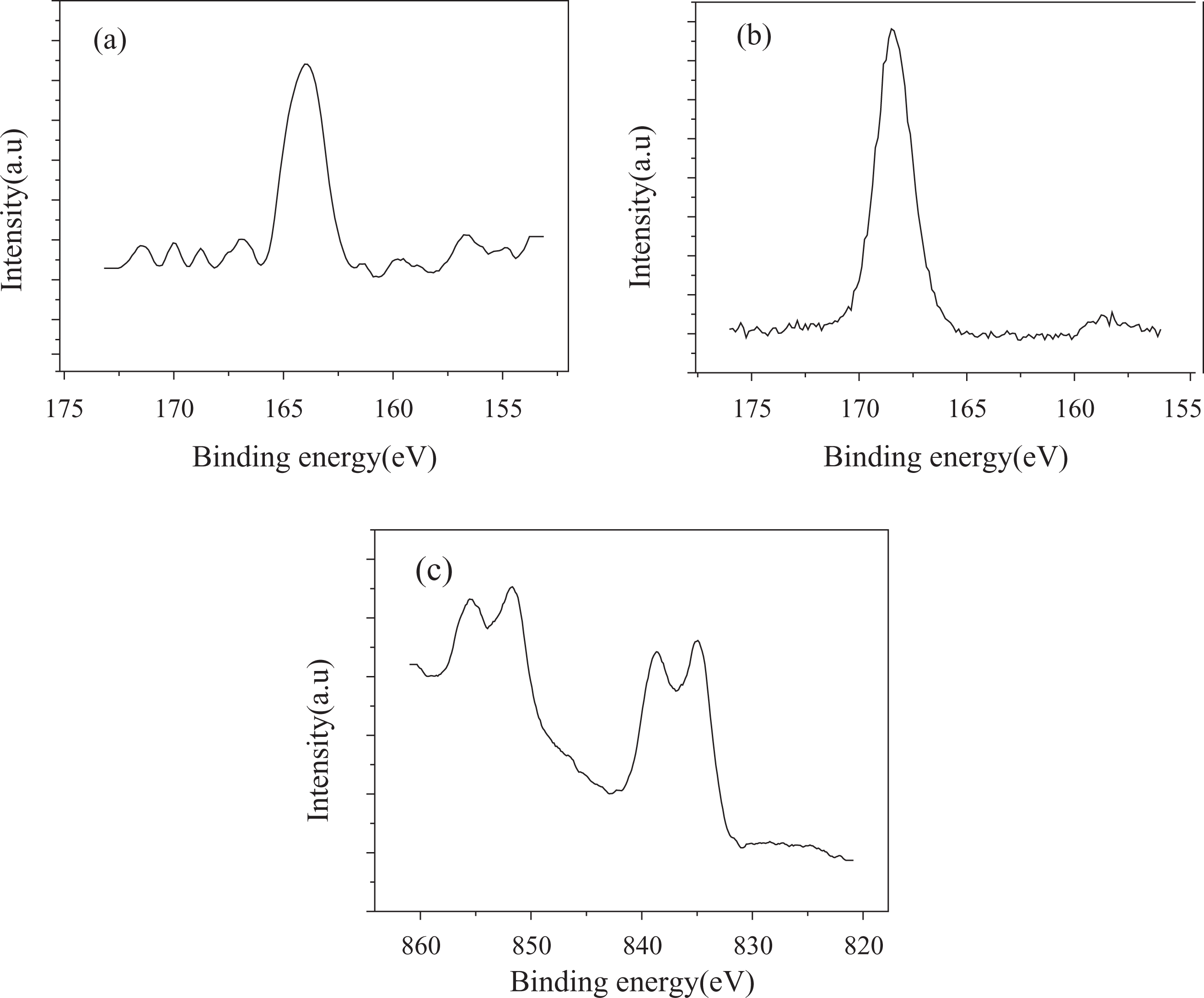
XPS spectra of (a) S2p of MPTS-SAM coated on Si substrate, (b) S2p of oxidized MPTS-SAM coated on Si substrate, and (c) La3d in CNT composite thin film. XPS: x-ray photoelectron spectroscopy; Si: silicon; La: lanthanum; CNT: carbon nanotube; MPTS: 3-mercaptopropyl trimethoxysilane; SAM: self-assembled monolayer.
The contact angles of hydroxide substrates, oxidized MPTS-SAM, and MPTS-SAM are 4°, 31°, and 70°, respectively, as shown in Figure 8. When the terminal –SH groups of SAM were oxidized into sulfonate groups (–SO3H) by dipping in a solution of 30% HNO3, the contact angle of samples decreased from 70° to 31°. 18 It indicates that those films were successfully obtained in our work, which is necessary for the formation of CNTs composite film.
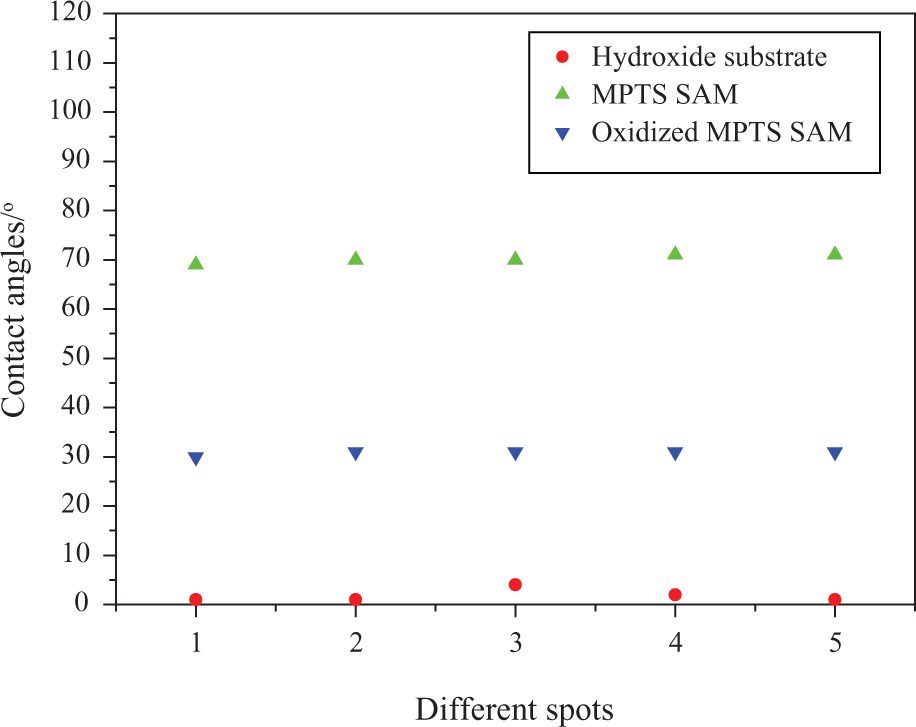
Contact angle of hydroxide substrate and different self-assembled monolayers.
Friction and wear
Figure 9 shows the variation of friction coefficient as a function of sliding pass for those films. It can be seen that the initial friction coefficient of CNT composite thin films is about 0.1 and the wear life of the films exceeds 500 sliding passes, which is obviously longer than that of some organic films. 19 The composite coatings exhibit a running-in period of approximately 250s during which the coefficient of friction (COF) value decreases steadily before attaining a steady state. During the running-in period, these ridges are progressively worn away until a sufficient amount of wear debris covers the surface leading to stable lower COF values.
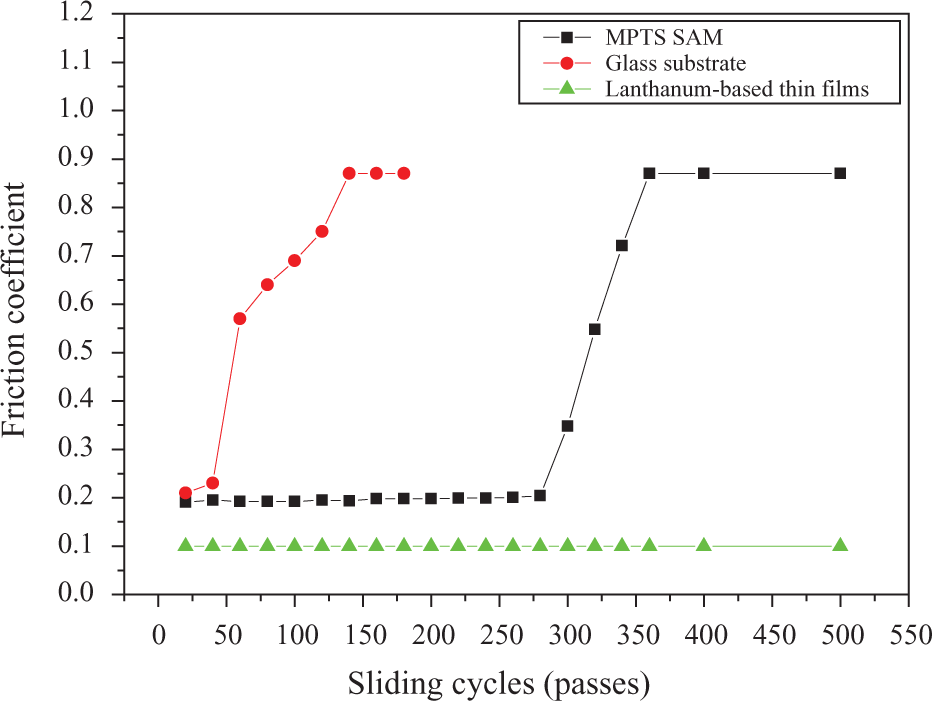
Wear dependence of friction coefficients.
The COF of glass substrate starts from a relative low value of 0.22 and rapidly rises to 0.8, the normal COF between glass and GCr15 steel, after about 120 sliding passes. It is deduced that some contamination is physisorbed on the glass substrate and reduces the COF of the substrate at the beginning. With the increase in sliding cycle, the contamination is worn off quickly, which accordingly leads to an increase in the COF value. It is shown in Figure 9 that MPTS-SAM can obviously reduce the COF of the glass substrate; however, it has poor wear resistance because of its bad load-carrying capability. In contrast, CNT composite film has the lowest COF value of 0.11 and the highest wear life exceeding 5000 passes, which indicates that the friction and wear properties of the film are largely improved. The reason for this lies probably in the stronger interfacial bonding established between the matrix and steel, which results in stronger adhesion and friction.
Rare earth elements have the unique chemical activity, which depends on their special electron structure (– – – 4f 0–14). The rare earth compounds are capable of coordinating and forming ionic combination when reacting with functional groups (such as hydroxyl (C–OH), carbonyl (C=O), and carboxyl (COOH) groups) on the glass substrates or the surfaces of CNTs. 15 With the chemical combination between rare earths and functional groups, CNTs can strongly bond with the substrate, which helps to improve the load-carrying ability and wear life of the composite films.
SEM wear tracks of SAMs are shown in Figure 10. Comparing Figure 10(a) with Figure 10(b), it is seen that the width of wear track of CNT composite films is much narrower, and the films display well-defined tracks without any evidence of complete failure of the SAMs. The higher magnification of Figure 10(b) shows that many CNTs uniformly distribute on the wear track. Relating Figure 10 with Figure 9, it is proved that the CNT composite films are strongly bonded with the substrates in friction.

SEM images of (a) MPTS, (b) CNT composite film at a low magnification, and (c) CNT composite film at a high magnification sliding against steel ball after 500 sliding passes. SEM: scanning electron microscopy; CNT: carbon nanotube; MPTS: 3-mercaptopropyl trimethoxysilane.
Conclusions
CNT composite films can be obtained using self-assembly technique through the chemical combination of rare earths with functional groups.
A very low COF of about 0.1 and a very long wear life of more than 5000 passes were obtained, which made CNT composite films a good candidate for tribological applications.
The uniform distribution of CNTs in composite films improves the stiffness and load-carrying ability as well as the wear resistance of the films.
In friction process, the CNTs that ground off from the composite films might act as microbearings between the two mating surfaces, resulting in the decrease of COF value.
Footnotes
Acknowledgement
The authors thank the Instrumental Analysis Center, Shanghai Jiao Tong University, China, for providing support for the project.
Funding
This work was financially supported by the National Natural Science Foundation of China (No. 50775139).
