Abstract
A novel β-nucleating agent of isotactic polypropylene (iPP), the potassium salt of 1,4,5,6,7,7-hexachlorobicyclo[2.2.1]hept-5-ene-2,3-dicarboxylic acid (CA-19), was found and its effects on the mechanical properties, content of β-crystal, and crystallization behavior of iPP were investigated. The results showed the content of β-crystals of nucleated iPP (
Keywords
Introduction
Isotactic polypropylene (iPP) is a polymorphic material with several crystal modifications including the monoclinic (α), trigonal (β), and orthorhombic (γ) forms. 1 In the past decades, β-nucleated iPP (β-iPP) has attracted extensive attention due to its excellent thermal and mechanical performance. 1 –21 The toughness and the elongation at break of β-iPP are much higher than that of α-iPP, which is very important from the viewpoint of industrial application. However, β-iPP is thermodynamically less stable and is therefore more difficult to obtain under the conditions typically found in industrial processes. Numerous methods have been developed to produce significant quantities of β-phase, such as quenching the melt to a certain temperature range, 5,6 directional crystallization in a thermal gradient field, 7,8 shearing or elongation of the melt during crystallization, 9 vibration-induced crystallization, 10,11 or using β-nucleating agents (β-NAs). 12 –21
Of these methods, the addition of β-NAs has proved to be the most efficient way to obtain iPP particles with high content of β-iPP. Until now, only three classifications of compounds have been mainly used as β-form NAs: the first ones are organic pigments,
12
–14
such as γ-quinacridone (Dye Permanent Red E3B), Indigosol Grey/IBL, Indigosol Golden Yellow IGK, and Cibantine Blue 2B; the second one includes a few aromatic amide compounds,
15
–17
such as
In this article, the effects of the novel β-NA CA-19 on the mechanical properties, content of β crystals, crystallization peak temperature (
Experimental
Materials
The iPP (trade name T30S) used in this study was kindly provided as a powder by Jiujiang Petroleum Chemical Co. (Jiujiang City, China); it had a melt flow index of 2.9 g/10 min (230/2.16 kg),
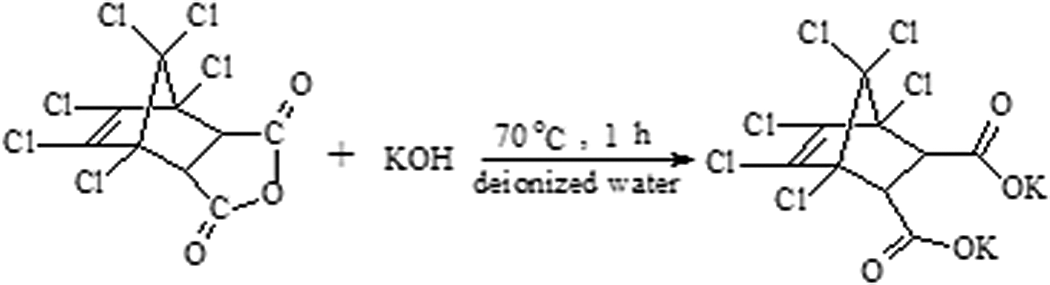
Synthesis reaction of CA-19. CA-19: 1,4,5,6,7,7-hexachlorobicyclo[2.2.1]hept-5-ene-2,3-dicarboxylic acid.

Morphology of the nucleating agent CA-19. CA-19: 1,4,5,6,7,7-hexachlorobicyclo[2.2.1]hept-5-ene-2,3-dicarboxylic acid.
Thermal properties of CA-19 were measured using a Perkin-Elmer Pyris 1 thermogravimetric analyzer (PerkinElmer, Shelton, CT, USA) in flowing nitrogen atmosphere (Figure 3). The result indicated that there was no crystal water in the molecules. According to the thermogravimetric analysis result, CA-19 has excellent thermal stability (the temperature of loss of 5 wt% of CA-19 was 275°C).
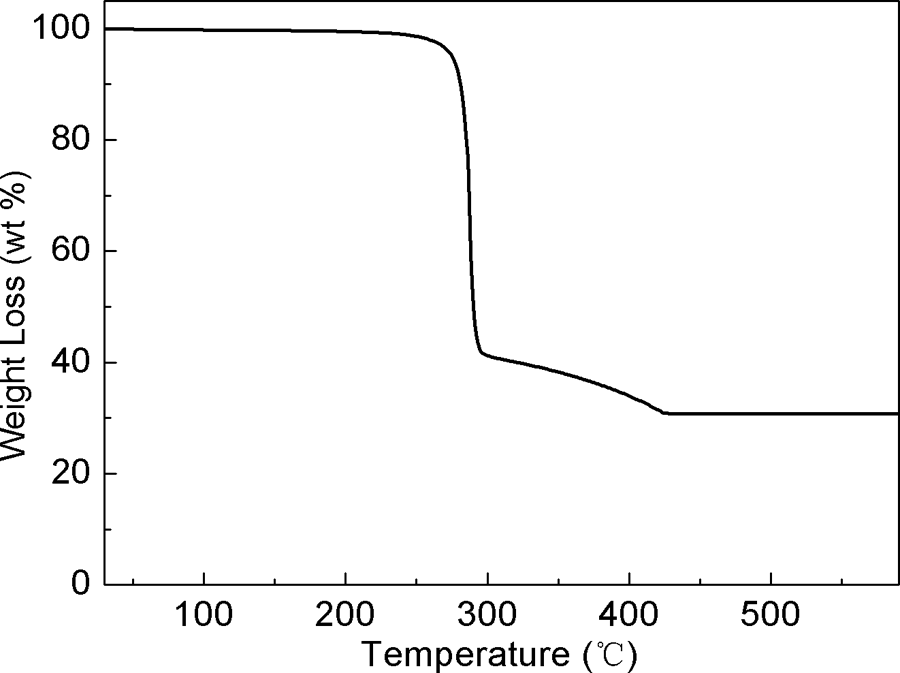
Thermogravimetric analysis profile of CA-19 measured in nitrogen atmosphere. Heating rate: 10°C/min. CA-19: 1,4,5,6,7,7-hexachlorobicyclo[2.2.1]hept-5-ene-2,3-dicarboxylic acid.
Sample preparation
The nucleating agent CA-19 and the iPP powders were dry blended in a high-speed mixer for 5 min. Then, the mixture was extruded by a twin-screw extruder (HT-30, Nanjing Rubber and Plastics Machinery Plant Co., Ltd, Nanjing City, China) through a strand die and pelletized. The pellets were molded into standard test specimens by an injection-molding machine (CJ-80E, Guangdong Zhende Plastics Machinery Plant Co., Ltd, Foshan City, China). The concentrations of the nucleating agent polypropylene were 0.05, 0.1, 0.25, 0.5, 0.75, and 1 wt%, and these samples were denoted as PP0.05, PP0.1, PP0.25, PP0.5, PP0.75, and PP1, respectively. The pure iPP sample was designated as PP0, which was prepared by the same method for comparison.
Mechanical properties
The mechanical properties were measured according to ASTM test methods, such as D-638 for the tensile strength and D-790 for the flexural modulus, using a universal testing machine (MTS Systems Co., Ltd, Foshan City, China). The Izod impact strength was tested on the basis of D-256, using an impact tester (MTS Systems Co., Ltd, Shenzhen City, China.). The reported values of the mechanical properties were averaged from five independent measurements.
Differential scanning calorimetry
Differential scanning calorimetry (DSC; Diamond, Perkin-Elmer, Shelton, CT, USA) was carried out to study the crystallization peak temperature and analyze the nonisothermal crystallization kinetics. Temperature was calibrated before the measurements using indium as a standard medium. The slices, 3–5 mg, were cut from the injection-molded samples.
Samples were heated to 200°C at a rate of 10°C/min under a nitrogen atmosphere and held for 5 min to eliminate their thermal and mechanical history before cooling at the specified cooling rate (2.5, 5, 10, 20, and 40°C/min). The exothermal curves of heat flow as a function of temperature were recorded to analyze the nonisothermal crystallization process.
Polarized optical microscopy
The morphology studies of pure iPP and nucleated iPP were performed with the aid of an Olympus BX51 (Olympus, Tokyo, Japan) polarized optical microscope with a DP70 digital camera, and it attached THMS600 hot stage. The extruded samples were placed between two microscope slides, melted, and pressed at 200°C for 5 min to erase any trace of crystals, and then rapidly cooled to a predetermined crystallization temperature. The samples were kept isothermally until the crystallization process was completed (for pure iPP, the isothermal crystallization time was 60 min; while for nucleated iPP, it was 30 min), while photographs were taken.
Wide-angle x-ray diffraction
The extruded samples were placed between two cover glasses on a hot stage (Figure 4: Hot stage 1) at 200°C. The melted specimen was then quickly placed onto another hot stage (Figure 4: Hot stage 2) set to the desired temperature in the range 90–140°C. The samples were held isothermally until the crystallization process was completed. Then the crystal structure of the samples was investigated by wide-angle x-ray diffraction (WAXD). The WAXD patterns were recorded in transmission with a Rigaku D/max-2550VB/PC apparatus (Japan). Using Cu Kα (λ = 1.54 Å) radiation, the spectra were recorded in the 2 The preparation process of the samples for WAXD. WAXD: wide-angle x-ray diffraction.
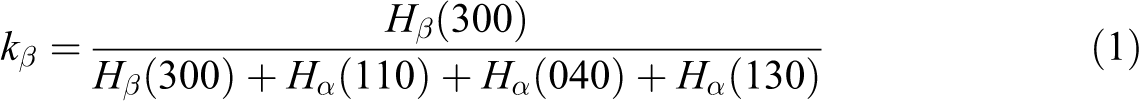
where
Theory of nonisothermal crystallization
The Avrami equation
26,27
widely used to describe polymer isothermal crystallization of polymers is given by
where
The Avrami equation has been extended by Ozawa
28
to develop a simple method to study nonisothermal experiments. The general form of the Ozawa theory can be written as follows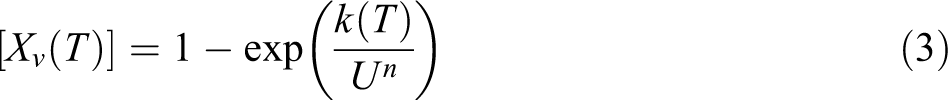
where
Caze et al.
23
put forward a method to modify the Ozawa equation. They assumed an exponential increase in
On the basis of their findings on the crystallization behavior of poly(ethylene terephthalate) and polypropylene (PP), Kim et al. proposed
29

where 
Combination of equations (3)
to (5) yields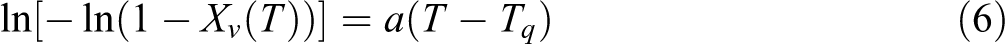
Hence, a linear plot of ln[−ln(1 − 
As such, the parameter
Results and discussion
Mechanical properties
From the viewpoint of industrial application, it is necessary to investigate the effects of a nucleating agent on the mechanical properties. The mechanical properties of iPP are known to be strongly affected by the amount of a nucleating agent. Therefore, the effects of the novel nucleating agent CA-19 on mechanical properties of iPP were investigated as a function of concentration of CA-19. The results are shown in Figure 5.
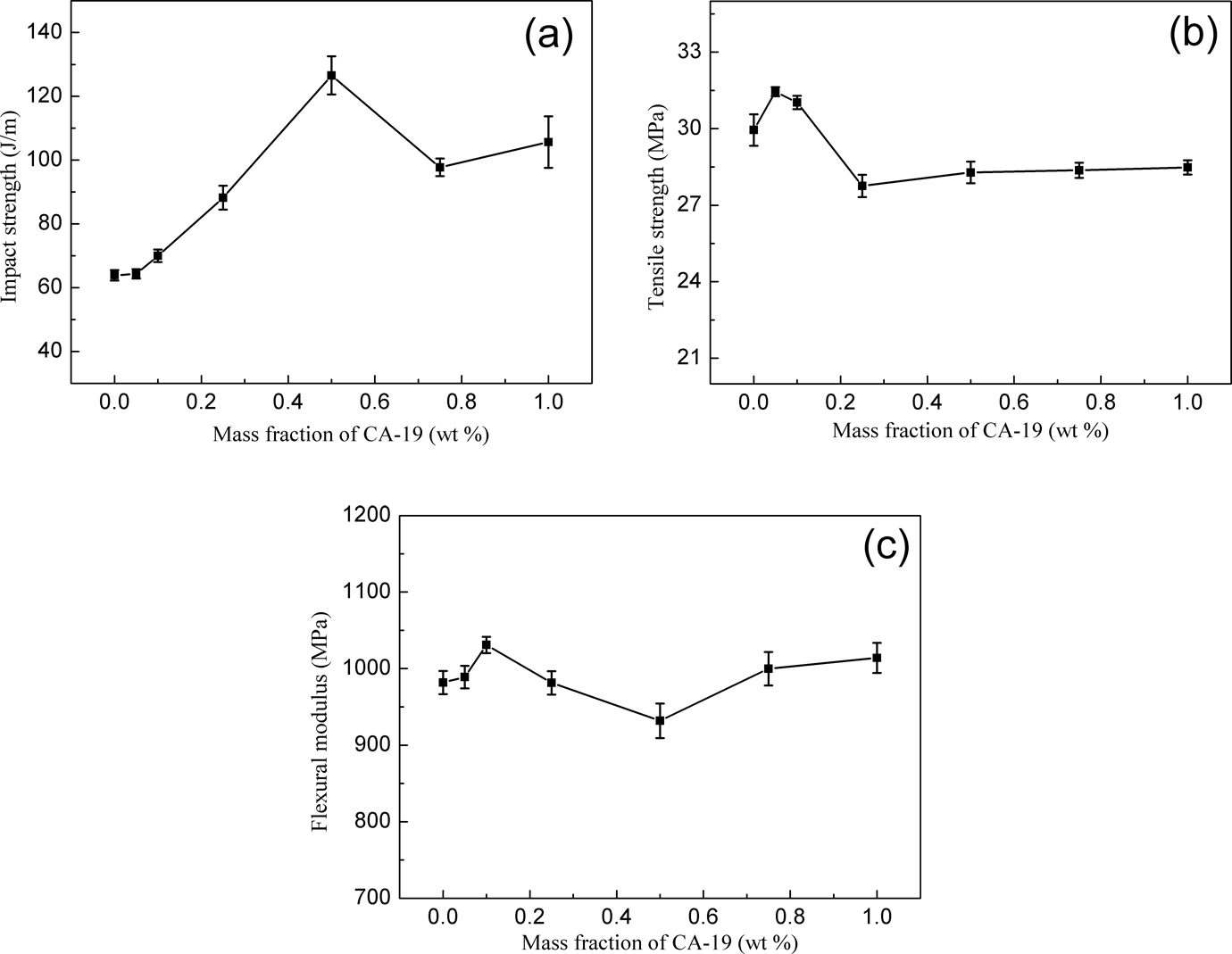
The mechanical properties of iPP nucleated with different content of CA-19, (a) impact strength; (b) tensile strength; and (c) flexural modulus. iPP: isotactic polypropylene. CA-19: 1,4,5,6,7,7-hexachlorobicyclo[2.2.1]hept-5-ene-2,3-dicarboxylic acid.
Figure 5(a) shows that the impact strength of nucleated iPP increased rapidly with increasing CA-19 when it was less than 0.5 wt%, then decreased until the CA-19 contents reached 0.75 wt%, and then basically remained constant with the further increase in CA-19. When the content of CA-19 was 0.5 wt%, the impact strength of the nucleated iPP improved about two times compared with that of pure iPP, indicating that CA-19 could significantly improve the toughness of iPP. In Figure 5(b), the tensile strength of nucleated iPP slightly increased and then decreased rapidly until the concentration of CA-19 reached about 0.25 wt%, and then remained essentially constant with further increase in concentration. In Figure 5(c), flexural modulus of nucleated iPP slightly increased and then decreased until the concentration of CA-19 reached about 0.5 wt%; it then increased with a further increase in concentration. Thus, 0.5 wt% seems to be the best concentration for CA-19 in terms of mechanical properties.
Effect of the nucleating agent on the content of β-crystals of iPP
DSC and WAXD are usually used to characterize the content of β-crystals of iPP. However, the β-crystal content of iPP cannot be accurately calculated from DSC melting curves. One reason is that the exact determination of the β-content is difficult because the melting peaks of the α- and β-modification overlap each other. Another reason is the existence of βα-recrystallization of β-crystals during melting process. Therefore, WAXD is more accurate than DSC to characterize the content of β-crystals of iPP, which is what we used for the quantification of β-phase content in the isothermally crystallized iPP samples. The β-crystal contents of iPP are known to be strongly affected by the crystallization temperature. 21 Therefore, the effects of the isothermal crystallization temperature on the content of β-crystals of iPP nucleated with 0.5 wt% CA-19 were investigated by WAXD. The WAXD scans are shown in Figure 6.
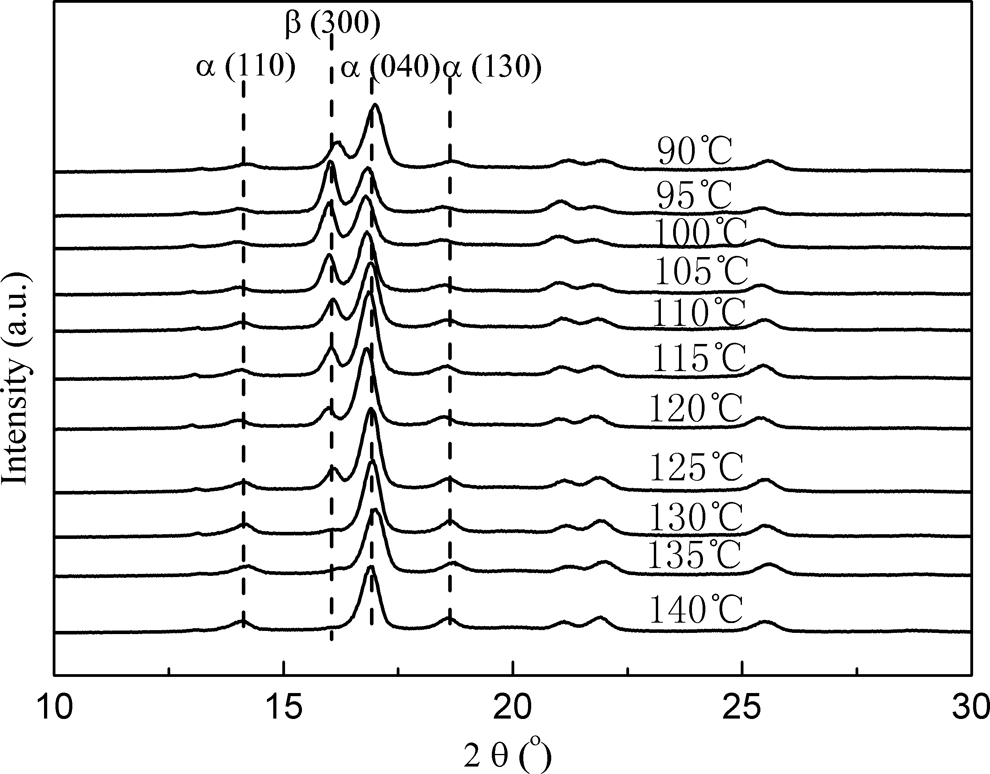
WAXD patterns of sample PP0.5 with different isothermal crystallization temperatures under quiescent conditions. WAXD: wide-angle x-ray diffraction; PP: polypropylene.
In Figure 6, (110) at 2
The effect of the isothermal crystallization temperature on the
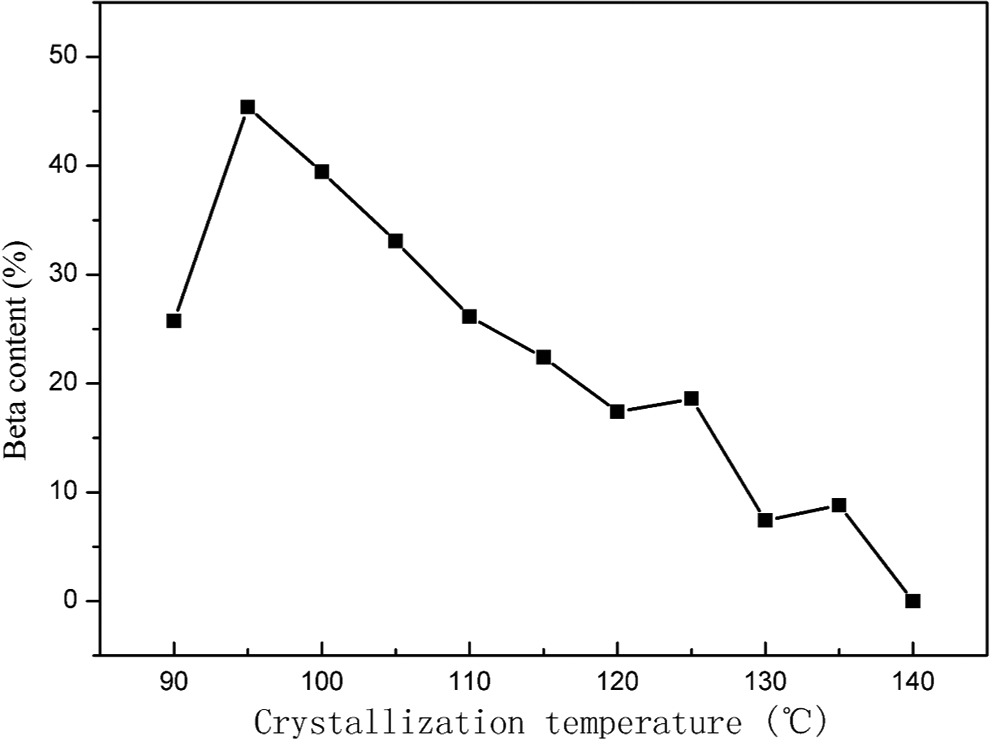
It is well known that the nucleating efficiency of β-NAs depends on not only crystallization temperature but also their concentration.
21,31
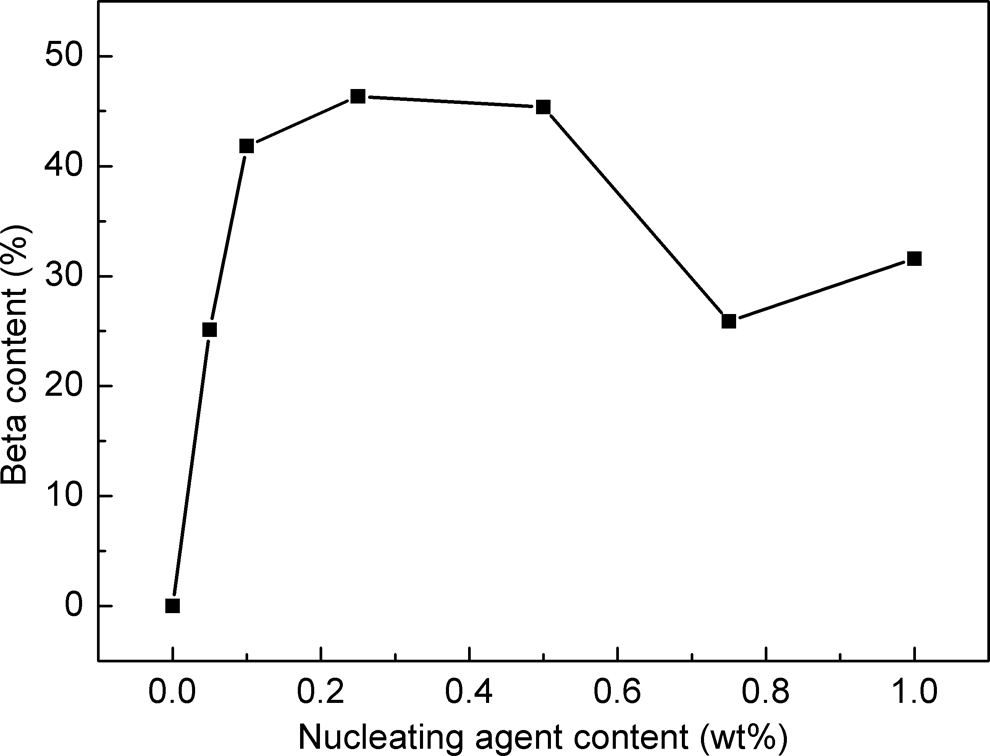
Therefore, the nucleating efficiency of CA-19 with different concentrations was also investigated. The WAXD patterns of nucleated iPP with different concentrations of CA-19 crystallized at 95°C under static conditions are shown in Figure 8. The
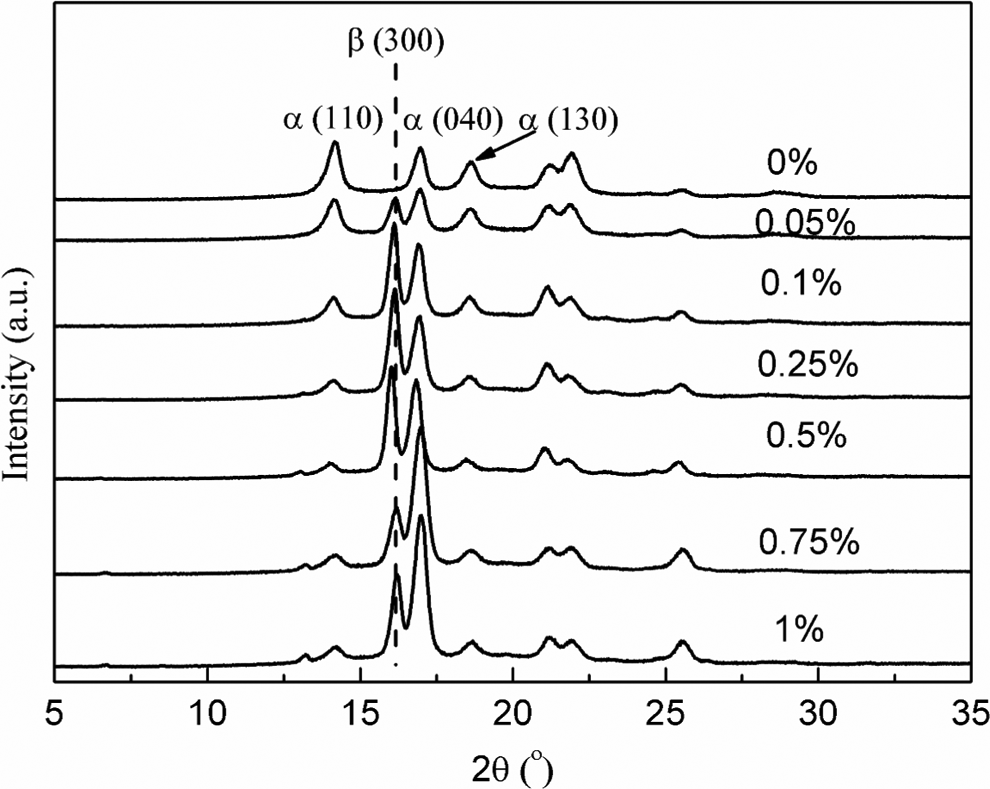
WAXD patterns of sample PP0 (pure iPP) and nucleated iPP with different CA-19 contents crystallized at 95°C under quiescent conditions. WAXD: wide-angle x-ray diffraction; iPP: isotactic polypropylene; PP: polypropylene; CA-19: 1,4,5,6,7,7-hexachlorobicyclo[2.2.1]hept-5-ene-2,3-dicarboxylic acid.
Crystallization behavior
In order to check the effect of the nucleating agent on the crystallization behavior, the DSC nonisothermal crystallization curves (the cooling rate is 10°C/min) of iPP with different CA-19 contents are plotted in Figure 10.
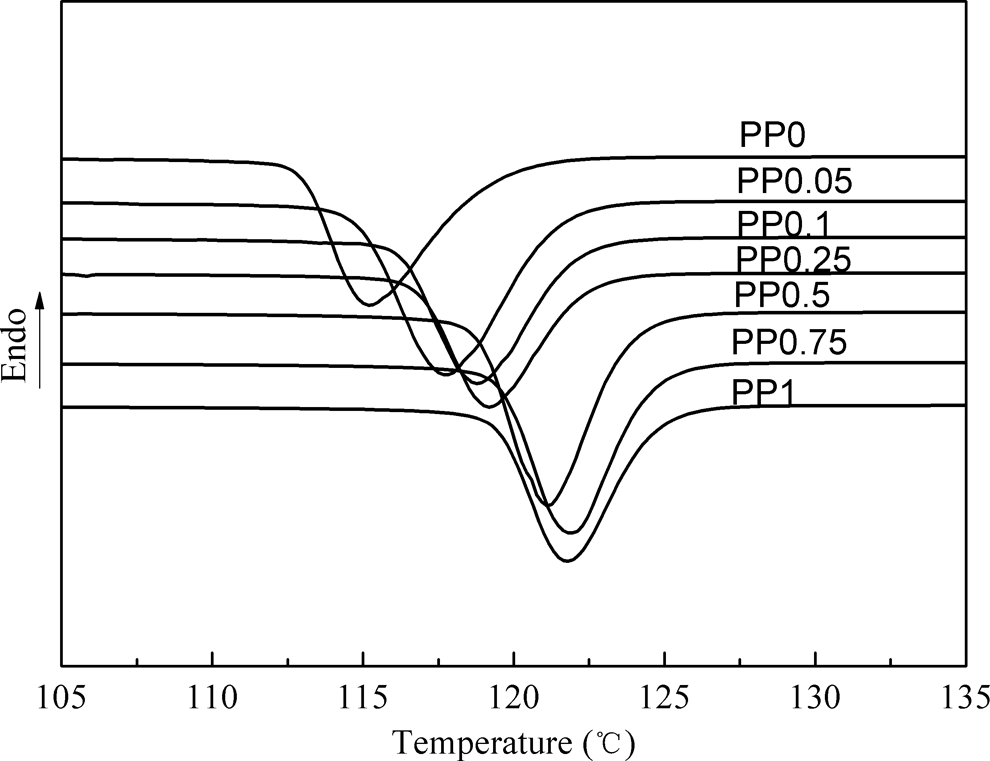
Differential scanning calorimetry crystallization curves of iPP with the different content of CA-19. iPP: isotactic polypropylene; CA-19: 1,4,5,6,7,7-hexachlorobicyclo[2.2.1]hept-5-ene-2,3-dicarboxylic acid.
For the pure iPP (PP0),
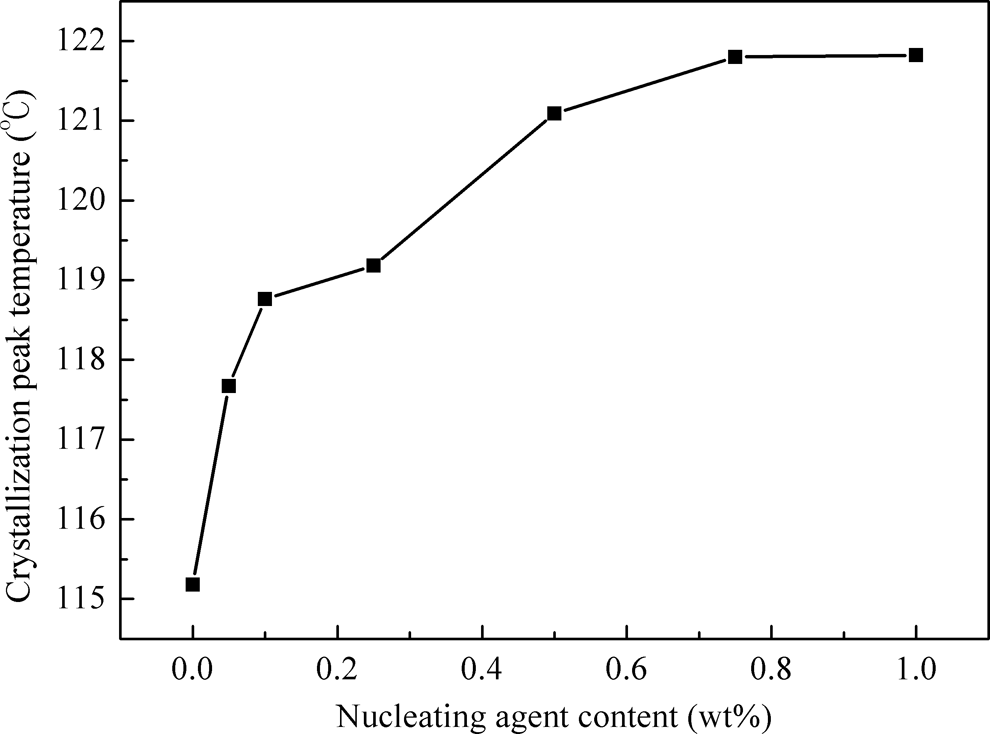
The effect of the content of CA-19 on crystallization peak temperature of iPP. iPP: isotactic polypropylene; CA-19: 1,4,5,6,7,7-hexachlorobicyclo[2.2.1]hept-5-ene-2,3-dicarboxylic acid.
β-NA affects not only the crystal form but also the morphology of iPP. Polarized optical microscopy (POM) was applied to study the effect of CA-19 on the morphology of iPP. Polarized optical micrographs of pure iPP and nucleated iPP crystallized at 140°C are shown in Figure 12. The spherulite diameter of pure iPP was more than 50 μm, and the incorporation of CA-19 decreased the spherulite sizes. Because of the existence of plenty of nuclei in the iPP/CA-19, the spherulites grow so fast that they rapidly impinge with each other before they grow bigger. Therefore, the spherulite size of nucleated iPP was much smaller than that of virgin iPP. Furthermore, the spherulite size gradually decreased until the concentration of nucleating agent reached 0.75 wt% and then decreased slightly with a further increase in CA-19. This is in good agreement with the results of the crystallization peak temperatures.

Micrographs for pure iPP sample crystallized at 140°C for 60 min and nucleated iPP sample crystallized at 140°C for 30 min: (a) pure iPP, (b) 0.05 wt%, (c) 0.10 wt%, (d) 0.25 wt%, (e) 0.50 wt%, (f) 0.75 wt%, and (g) 1.0 wt%. iPP: isotactic polypropylene; PP: polypropylene.
Nonisothermal crystallization kinetics
The crystallization process of semicrystalline polymers can have a dramatic impact on their mechanical properties and hence its understanding is important for final applications. Practical processes usually proceed under nonisothermal crystallization conditions. In order to search for the optimum conditions in the industrial process and obtain products with better properties, it is necessary to have quantitative evaluations of the nonisothermal crystallization process. The 0.5 wt% content of CA-19 was chosen considering the fact that nucleated iPP with that content showed better comprehensive mechanical properties. The nonisothermal crystallization of iPP and nucleated iPP was carried out by DSC with cooling rates from 2.5°C/min to 40°C/min. The thermograms of neat iPP and nucleated iPP are plotted in Figure 13. It is evident that the crystallization temperature was affected by the cooling rate: the higher the cooling rate, the lower the crystallization peak temperature. Furthermore, it can be recognized that, at the same cooling rates,
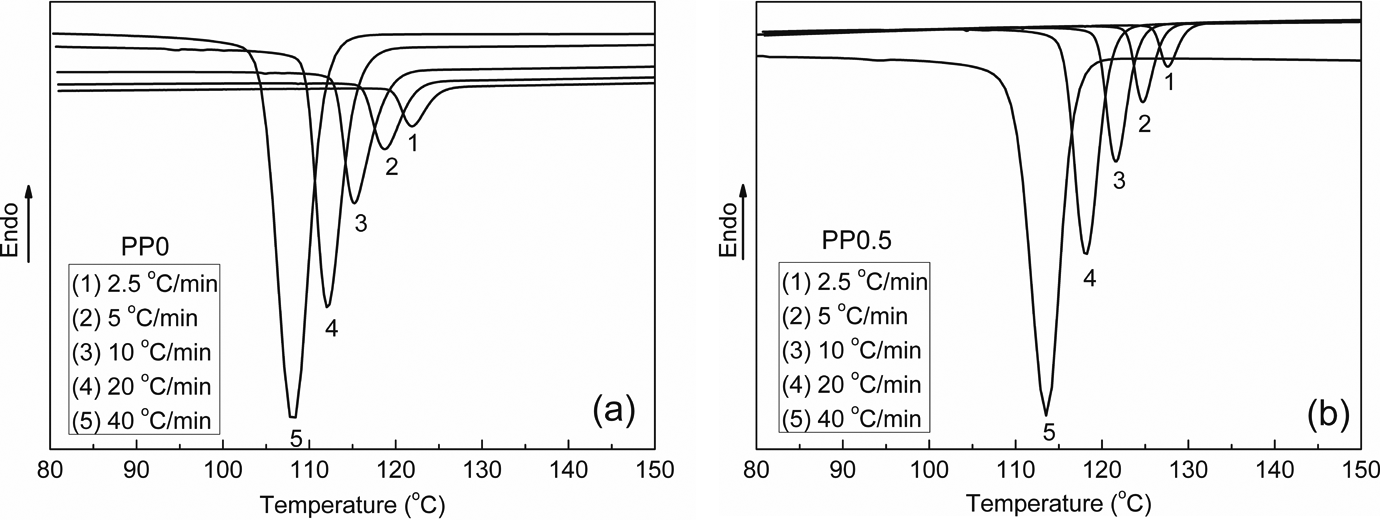
Differential scanning calorimetry cooling curves of (a) PP0 and (b) PP0.5. PP: polypropylene.
By means of integrating the partial areas under the DSC endotherms, the values of the crystalline weight fraction
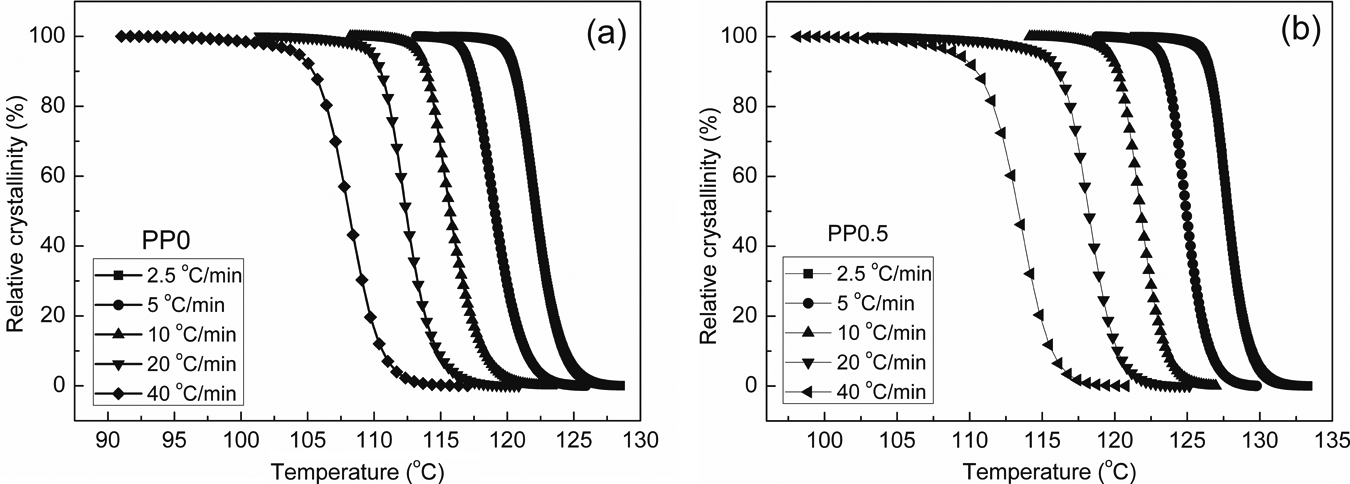
Relative crystallinity of (a) PP0 and (b) PP0.5. PP: polypropylene.
The crystallization half-times,
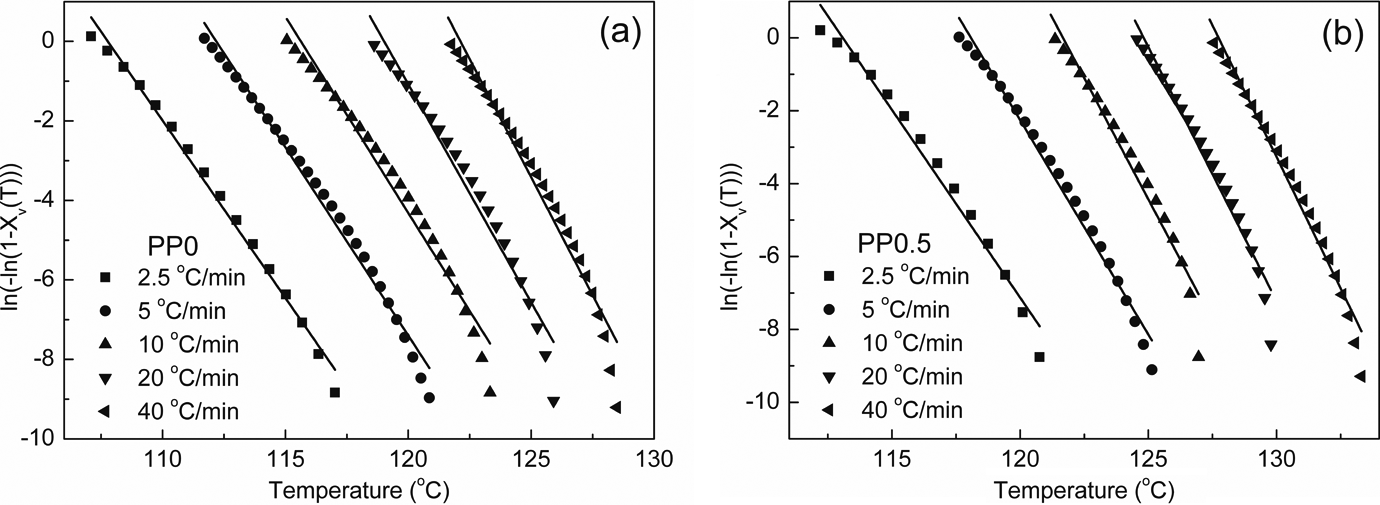
Plots of
Nonisothermal crystallization kinetics parameters for PP0 and PP0.5.
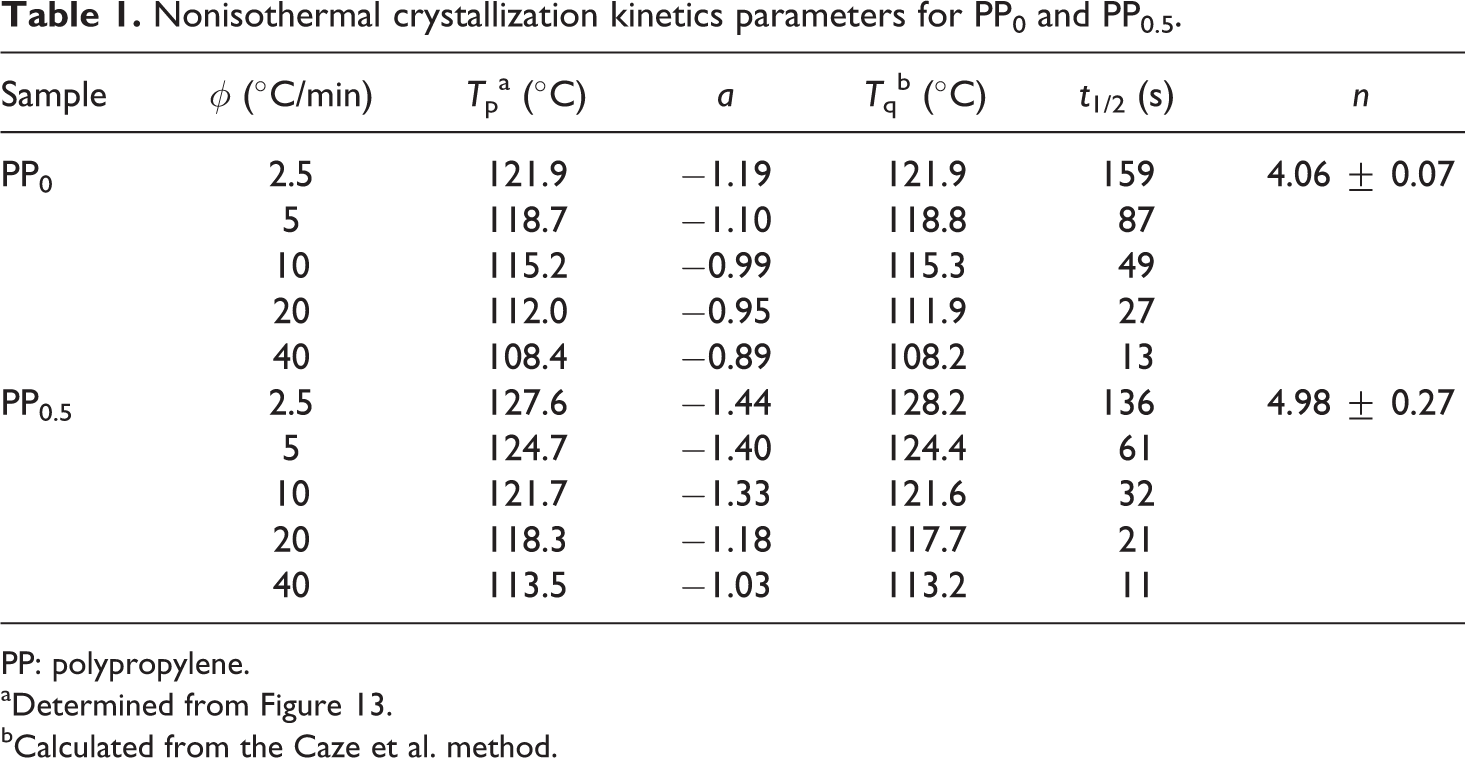
PP: polypropylene.
aDetermined from Figure 13.
bCalculated from the Caze et al. method.
Now
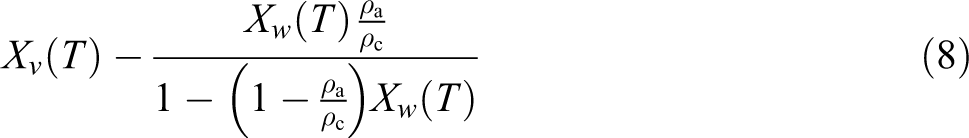
where
Straight lines can be obtained from plots of
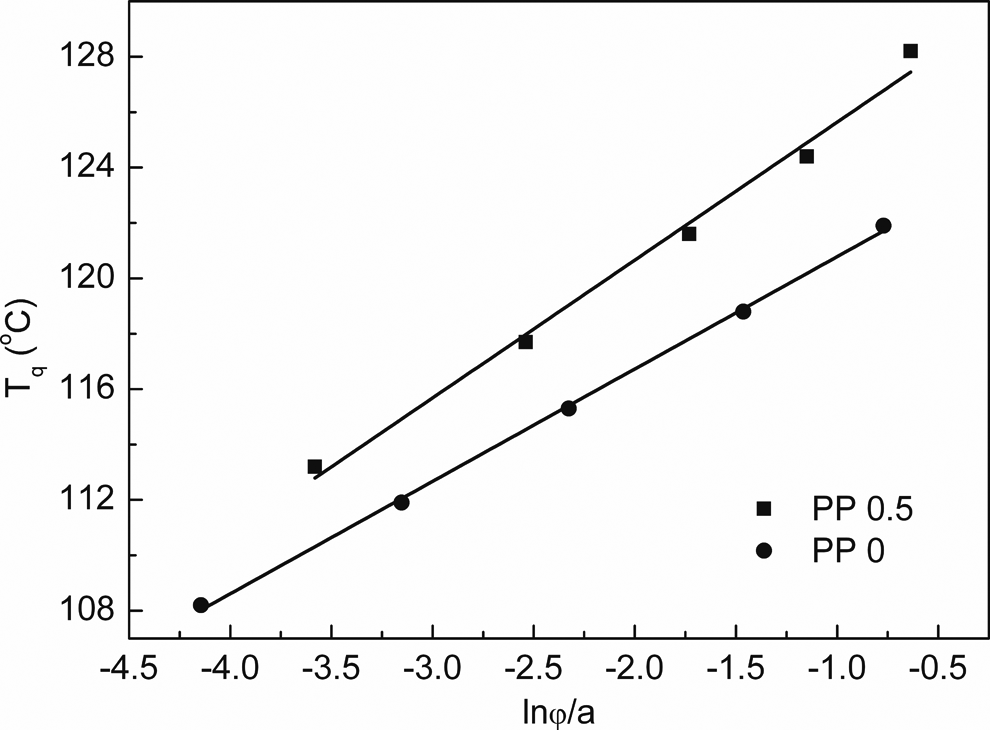
Plots of
Crystallization activation energy
Considering the influence of the cooling rates on the nonisothermal crystallization process, Kissinger proposed that the activation energy could be determined by calculating the variation in the crystallization peak with the cooling rate. 23
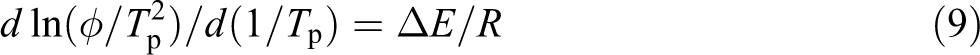
where
As shown in Figure 17, the Δ
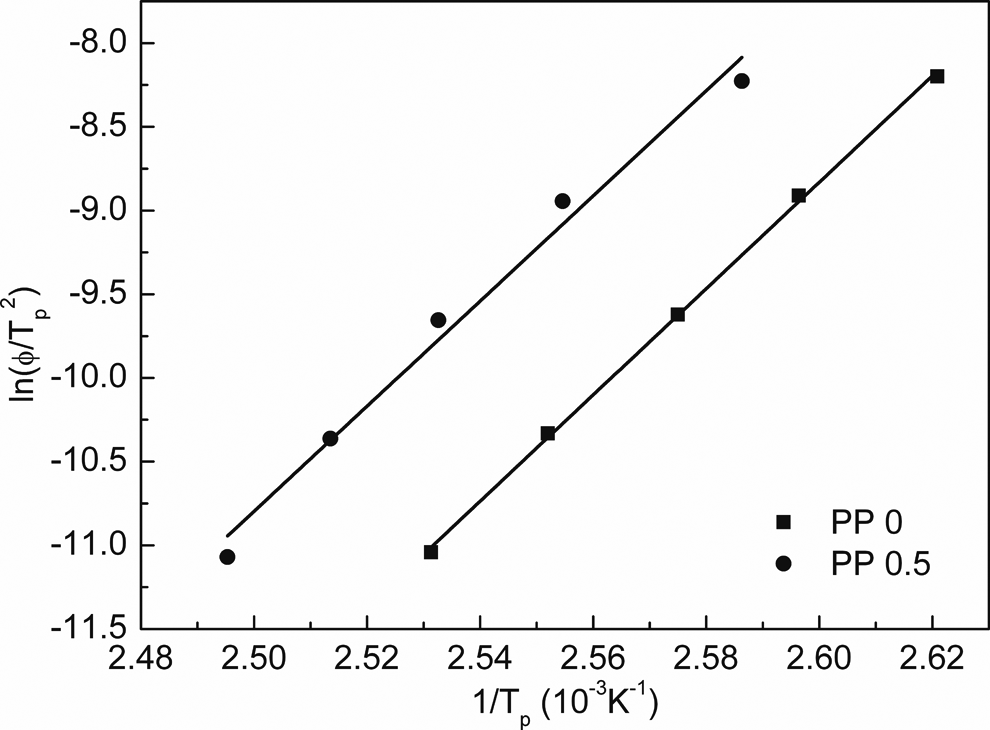
Kissinger plot for calculating the nonisothermal crystallization activation energies for PP0 and PP0.5. PP: polypropylene.
Conclusions
In this work, the effects of the novel β-NA CA-19 on the mechanical properties, content of β-crystals, crystallization behavior, and nonisothermal crystallization kinetics of iPP were investigated. The main conclusions can be summarized as follows: With the increase in CA-19 concentration, the impact strength of iPP first increased up to 0.5 wt% and then decreased. At the concentration of 0.5 wt%, the impact strength of nucleated iPP was about two times higher than that of pure iPP. Meanwhile, the tensile strength and flexural modulus slightly decreased. The results show that CA-19 could significantly improve the toughness of iPP. The The crystallization behavior studied by DSC and POM showed that the nucleating agent CA-19 can increase the crystallization peak temperature of iPP and gradually decrease the spherulite sizes with increase in the content of nucleating agent. The Caze et al. method was used to study the nonisothermal crystallization kinetics of nucleated iPP. The results indicate that the type of nucleation and growth geometry of iPP changed in the presence of nucleating agent CA-19. The crystallization activation energy was determined by the Kissinger method, which showed that the addition of CA-19 slightly decreased the crystallization activation energy of iPP.
Footnotes
Funding
This work was financially supported by National Natural Science Foundation of China (Grant No. 21106042), Research Fund for the Doctoral Program of Higher Education of China (Grant No. 20110074120012), and “the Fundamental Research Funds for the Central Universities.”
