Abstract
The coeffect of β-nucleating agent (β-NA) and different core–shell particles (CSPs) on improving the mechanical properties and thermal stability of polypropylene random copolymer (PPR) composites were investigated. Different CSPs, that is, the regular POE/nano-calcium carbonate (CaCO3) and the novel PE/nano-CaCO3 CSP were prepared through melt blending. The transmission electron microscopic micrograph demonstrated that the core–shell structure was successfully prepared and the CSP dispersed homogeneously in the PPR matrix. β-NA was also introduced into the PPR matrix to control the crystalline structure. The results of differential scanning calorimetry, wide-angle x-ray diffraction, and polarization optical microscopy showed that the relative content of β-form crystal was greatly enhanced in the presence of β-NA. The results of the mechanical tests and thermal gravimetric analysis indicated that the comprehensive mechanical properties and thermal stability were all greatly improved, especially for the sample with the incorporation of both β-NA and PE/nano-CaCO3 CSP.
Keywords
Introduction
Polypropylene random copolymer (PPR) is one of the most widely used thermoplastics for its wide range of advantages, such as low cost, easy processing, low density, easy design, and corrosion resistance. It has been widely applied in film, rigid packing, and pipes. However, it can still hardly meet the application demands in some severe conditions owing to its low impact resistance and notch sensitivity. 1,2 Several modification methods have been used to overcome this problem. However, most of these methods lower the stiffness and strength of the materials. 3 –5 So, a new way for improving the toughness of PPR is of great importance and benefit.
It is well known that elastomer is one of the most effective modifiers for PPR. 6,7 However, the fracture resistance improvement is usually at the cost of lowering the stiffness and heat stability. So, rigid particles are usually added simultaneously to compensate this shortcoming. For PP, nano-calcium carbonate (nano-CaCO3) is one of the most effective fillers. 5,8 The combination of elastomer and rigid particles to achieve the so-called core–shell particles (CSPs) has been proved another toughening method. 9,10 Nevertheless, the toughening efficiency of such CSP is still poor, and the stiffness would also deteriorate. 3,11,12 Thus, it is desirable to achieve high toughening efficiency without lowering its stiffness.
In fact, PPR is semicrystalline and can form several crystal modifications, including α, β, γ and mesomorphic smectic form. 4 So, the fracture resistance of PPR is also controlled by the crystal properties. Lately, β-form PP (β-PP) has been attached great importance owing to its higher toughness and heat stability than the regular α-form crystal, though it occurs more rarely for its relatively lower thermal stability. 13,14 Several methods have been used to form β-PP, among which adding β-nucleating agent (β-NA) is the most effective one. 15 –21 However, the rigidity of the PP would decrease a lot due to the lower stiffness of β form crystal compared to α form. 4,22
Only controlling the crystalline structure or adding CSP is not enough to achieve high toughening efficiency without lowering the stiffness. In this article, to overcome the shortages mentioned above, two kinds of CSPs were prepared, including the regular polyolefin elastomer (POE)/nano-CaCO3 CSP and the novel polyethylene (PE)/nano-CaCO3 CSP. They were incorporated into PPR with β-NA simultaneously to investigate the joint effect. For comparison, PPR with only β-modification (β-PPR) and neat PPR were also prepared. The distribution and morphology of CSP, the crystallization properties, the mechanical and thermal properties improvement of the PPR composites were investigated.
Experiments
Materials
PPR (trade name: C180, melt flow index MFI = 0.3 g/10 min at 230°C and 2.16 kg) was manufactured by Sinopec Yangzi Petrochemical Co. Ltd (China). Rare-earth β-NA (trade name WBG) was purchased from Guangdong Winner Functional Materials Corporation (China). PE (trade name: SP980, MFI = 0.6 g/10 min at 190°C and 2.16 kg) was purchased from LG Chem (Seoul, South Korea). Polyolefin elastomer (POE) with the trade name ENGAGE 8450 (MFI = 3.0 g/10 min at 190°C and 2.16 kg) was bought from Dow Chemical (Midland, Michigan, USA). Nano-CaCO3 (particle diameter (D): 60–80 nm) was purchased from Gofen calcium carbonate Co. Ltd (Jiangxi, China). Maleic anhydride-grafted PP (PP-g-MAH) was manufactured by Shanghai Sunny New Technology Development Co. Ltd. (Shanghai, China). Irganox 1010 and 168 are all commercially available.
Sample preparation
The CSPs were prepared in the following procedure: All the pellets were totally dried in a drying oven at 80°C overnight. PE or POE, nano-CaCO3, and PP-g-MAH were melt blended by a corotating twin-screw extruder (TSE-35A, length/D = 48; Nanjing RuiYa Polymer Processing Equipment Limited Company (Jiangsu, China)) with the rotation speed of 150 r min−1 and the temperature along the barrel was increased from 130 to 190°C. The extrudates were cooled in a water bath and then cut into pellets by a pelletizer. The obtained blend pellets, that is, the CSP, were then dried again at 80°C in the drying oven.
After that, the CSP, PPR, β-NA, and Irganox 1010 and 168 were again mixed in the twin-screw extruder with the extruding temperature of 140–200°C from hopper to die and the rotation speed of 120 r min−1. The blend pellets were obtained after cooled down with water bath and dried in a drying oven at 80°C for 8 h. Then, the dried pellets were injection molded into standard specimens for properties testing, conducted on an injection machine (CJ80M2, Chengde Plastic Machinery Limited Company, China), with the melting temperature from 200 to 230°C, and the mold temperature was 40°C. Table 1 illustrates the composition of the blends.
Composition of the blends.a
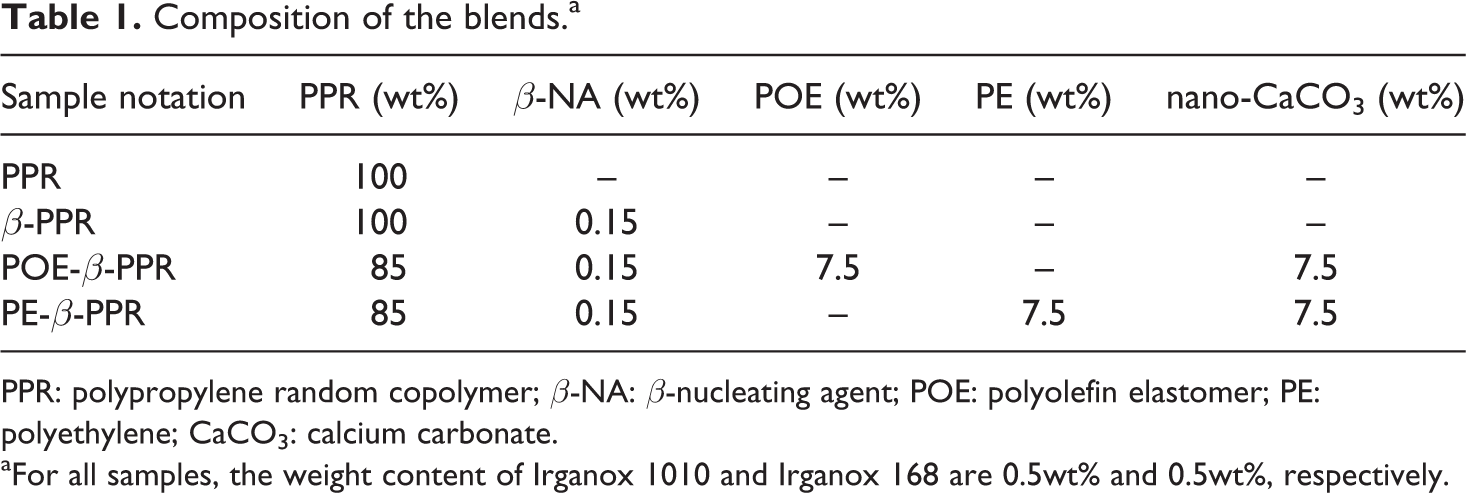
PPR: polypropylene random copolymer; β-NA: β-nucleating agent; POE: polyolefin elastomer; PE: polyethylene; CaCO3: calcium carbonate.
aFor all samples, the weight content of Irganox 1010 and Irganox 168 are 0.5wt% and 0.5wt%, respectively.
Characterizations
Morphological study
Transmission electron microscopy (TEM, JEM-1400, JEOL, Tokyo, Japan) was used to investigate the dispersion and phase morphology of CSP in the PPR matrix. The samples for investigation were cut into ultrathin flakes from the rectangular specimens.
Differential scanning calorimetry
Differential scanning calorimetry (DSC) measurements were performed on the 200PC (NETZSCH, Germany) to study the crystallization behavior and melting characteristics of samples under nitrogen atmosphere. The enthalpy and the temperature had been calibrated with indium at different rates. About 6–8 mg samples were heated from room temperature to 200°C at the rate of 10°C min−1 and held for 5 min to eliminate the mechanical and thermal history. After melting, the samples were cooled to 50°C at 10°C min−1 to study the crystallization behavior and then reheated to 200°C at a heating rate of 10°C min−1 to study their melting behavior and polymorphism. The degree of crystallinity (Xc) of each sample was calculated from the data obtained during the second heating process by

The relative content of the β-phase PP (Φβ) was calculated according to following relationship:

Wide-angle x-ray diffraction
Wide-angle x-ray diffraction (WAXD) was employed to study the crystalline structure of PPR matrix. WAXD patterns of the samples were obtained with a D-max2550 VB-PC x-ray diffractometer (Rigaku Corporation, Japan) at room temperature. The copper Kα irradiation source was operated at 40 kV and 100 mA. The scanning range was 5 to 50° with the rate of 3° min−1 and a step length of 0.02°. The K value representing the β-form content in PPR was determined from x-ray diffractograms according to Turner-Jones et al.
24
:
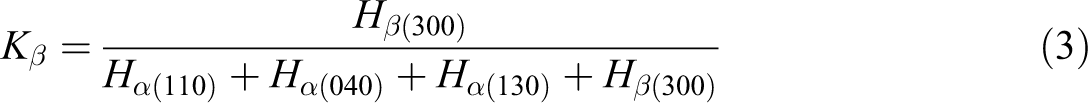
Polarization optical microscopy
A polarization optical microscope (POM, XPL-30TF, China) with a hot stage was used to study the crystallization morphology of the composite. First, the specimen was cut from the sample and hot-compressed into a thin film at 240°C and maintained at 240°C for 5 min to erase the thermal and mechanical prehistory. Second, the sample was rapidly cooled to a certain temperature and maintained at this temperature until the isothermal crystallization was finished.
Mechanical testing
Notched Izod impact strength was measured by an impact tester (JJ-20, Changchun Intelligent Instrument and Equipment Limited Company, China) according to ISO 180-2001. The tensile property of injection-molded specimens were measured using a computerized universal testing machine (New Sansi, Shenzhen, China) at the tensile speed of 50 mm min−1 according to ISO 527-2-1994 standards. All the measurements were carried out at room temperature, and five samples were tested from each composite and the average results were recorded.
Scanning electron microscopy
The impact fracture surfaces of the samples got after Izod impact tests were investigated with a JSM-6360LV scanning electron microscopy (SEM, JEOL, Japan). The excitation energy used was 10 keV. The observations were focused on the difference of the crack initiation zone, crack propagation zone, and the later stage of the crack propagation. To achieve good electric conductivity, all samples were coated with gold–palladium by a sputter coating instrument.
Thermogravimetric analysis
The heat stability of the samples was studied by thermogravimetric analysis (TGA, STA449 F3 Jupiter, NETZSCH, Germany). Each sample weighing about 10 mg was heated from room temperature to 800°C at the rate of 10°C min−1 under nitrogen atmosphere.
Results and discussion
Phase morphology and distribution of CSPs
The morphology and dispersion of CSPs in the PPR composites were investigated by TEM. Figure 1(a) and (b) showed that these two kinds of CSPs dispersed homogeneously in the PPR matrix and showed no obvious agglomeration. In Figure 1(c) and (d), a typical core–shell structure can be observed (as shown by the dotted line circle). Figure 1(e) and (f) demonstrate the morphology of CSP at a higher magnification. Figure 1(c) and (e) show the morphology of the POE/nano-CaCO3 CSP at different magnification. But only small amounts of CSP can be observed, and the thickness of shell layer is really thin as can be seen in Figure 1(e). For PE-β-PPR shown in Figure 1(d) and (f), the spheres become bigger, implying thicker shell phase. The diameters of the nano-CaCO3 core are in the range of 70–80 nm, while the thickness of the PE shell is about 20–30 nm according to Figure 1(f). The mechanical properties also proved that the CSP contributes a lot to the good comprehensive properties of the modified PPR. The difference between the POE/nano-CaCO3 and PE/nano-CaCO3 CSP is caused by their different MFI. The MFI of POE (3.0 g/10 min) is much higher than that of PE (0.6 g/10 min), leading to the higher melt strength of PE than POE at the same processing temperature. So, the POE phase will disperse into the PPR matrix more easily instead of covering the nano-CaCO3 than PE.

TEM micrograph of POE-β-PPR and PE-β-PPR. (a) The dispersion of the POE/nano-CaCO3 CSP; (b) the dispersion of the PE/nano-CaCO3 CSP; (c) the morphology of the POE/nano-CaCO3 CSP; (d) the morphology of the PE/nano-CaCO3 CSP; (e) the morphology of the POE/nano-CaCO3 CSP at higher magnification; and (f) the morphology of the PE/nano-CaCO3 CSP at higher magnification. TEM: transmission electron microscopy; POE: polyolefin elastomer; PPR: polypropylene random copolymer; PE: polyethylene; CaCO3: calcium carbonate; CSP: core–shell particle.
Crystallization and melting behaviors
DSC cooling and melting curves of different samples are shown in Figure 2 and the corresponding data are listed in Table 2. As shown in Figure 2(a), the pristine PPR exhibits the crystallization temperature (Tc) of 110.1°C. In the presence of β-NA, the Tc of β-NA is increased to 123.9°C, much higher than that of the pure PP, showing great nucleation effect of β-NA. For the composites with both CSP and β-NA, they show the similar Tc value compared with β-PPR. It has already been proved that nano-CaCO3 had strong effect on the PP crystallization, 5,25,26 while the test results here does not show this tendency. However, this further proved the formation of CSP. In other words, the nucleation effect of nano-CaCO3 becomes inconspicuous most likely due to that almost nano-CaCO3 particles are encapsulated by POE or PE phase and their heterogeneous nucleating effect for PPR crystallization are prevented. So, the overall results well showed great nucleation effect of β-NA and the formation of CSP.
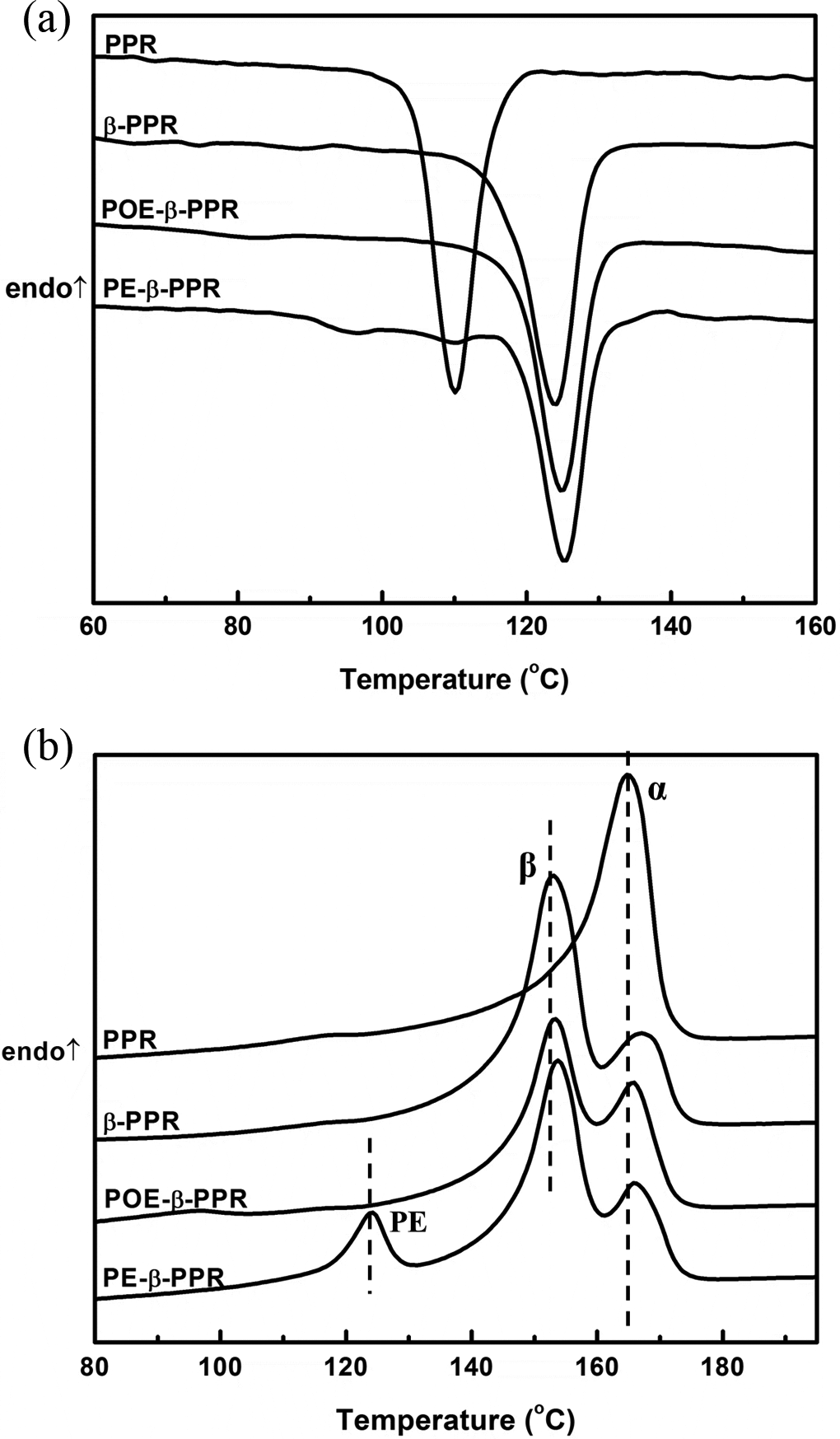
DSC cooling (a) and heating (b) curves of different samples. DSC: differential scanning calorimetry.
Crystallization and melting characteristics of PPR composites.
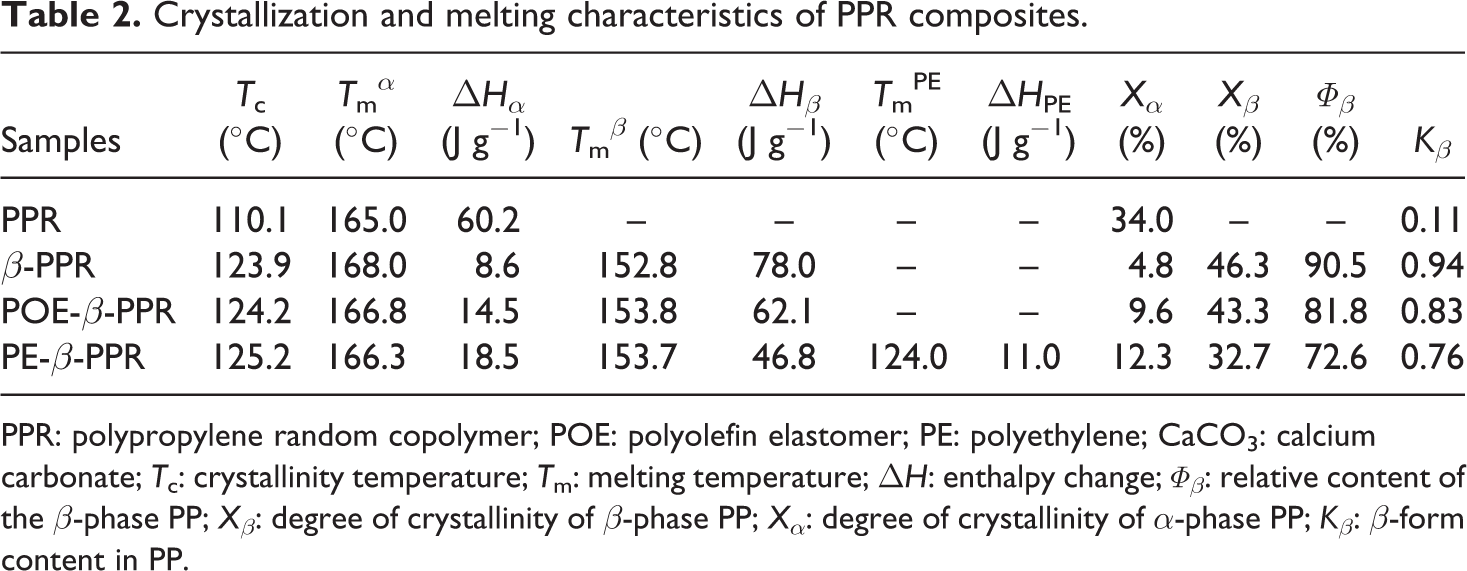
PPR: polypropylene random copolymer; POE: polyolefin elastomer; PE: polyethylene; CaCO3: calcium carbonate; Tc: crystallinity temperature; Tm: melting temperature; ΔH: enthalpy change; Φβ: relative content of the β-phase PP; Xβ: degree of crystallinity of β-phase PP; Xα: degree of crystallinity of α-phase PP; Kβ: β-form content in PP.
The melting behavior of pristine PPR is quite different from the samples after modification. As can be seen from Figure 2(b), the melting curve of pristine PPR exhibited only one single melting peak at 165°C, which corresponding to the melting of α-form crystal. However, with the appearance of β-NA and CSP, the samples showed multiple melting peaks. For β-PPR and POE-β-PPR, they exhibit double endothermic peaks, corresponding to the fusion of β-phase PP and α-phase PP from low to high temperature. And, there are three peaks in the melting curve of PE-β-PPR. The melting peak at about 167°C is attributed to the melting of α-PP, while the 154°C melting peak is caused by the β-PP melting, and the melting peak at around 124°C is believed to be the melting of PE crystals. The absolutely dominant β-crystal melting peak indicated that the PPR composites after modification crystallized mostly in β-form in the presence of β-NA. The Φβ of different samples were calculated by equation (2), and the results are shown in Table 2. In fact, lots of studies have proved that nano-CaCO3 can induce the β-form PP formation, which is also thought to be one of the toughening mechanisms. 27 However, in our study, one can find that β-PPR owns the highest Φβ, and the incorporation of nano-CaCO3 does not increase the Φβ. This further indicates that the nano-CaCO3 particles were covered by POE or PE, and the CSPs had been formed.
The crystalline structures of different samples were further detected by WAXD, and the results are shown in Figure 3. In these spectra, the (110) plane at 2θ = 14.2°, (040) plane at 17.0°, and (130) plane at 18.8° are the main reflection peaks of α-crystal, while the (300) plane at 16.2° is the characteristic peak of β-crystal. The value of the β-form content in PP (Kβ) calculated by the Turner-Jones formula (equation (3)) is also listed in Table 2. Agreeing well with the DSC measurement, pristine PPR formed almost pure α-crystal, while the samples exhibited mainly β-form PP after modification. An intensive diffraction peak at 2θ = 16.2° can be detected, which ascribed to the diffraction of (300) plane of β-crystal PP, indicating the presence of a large amount of β-crystal in the sample.
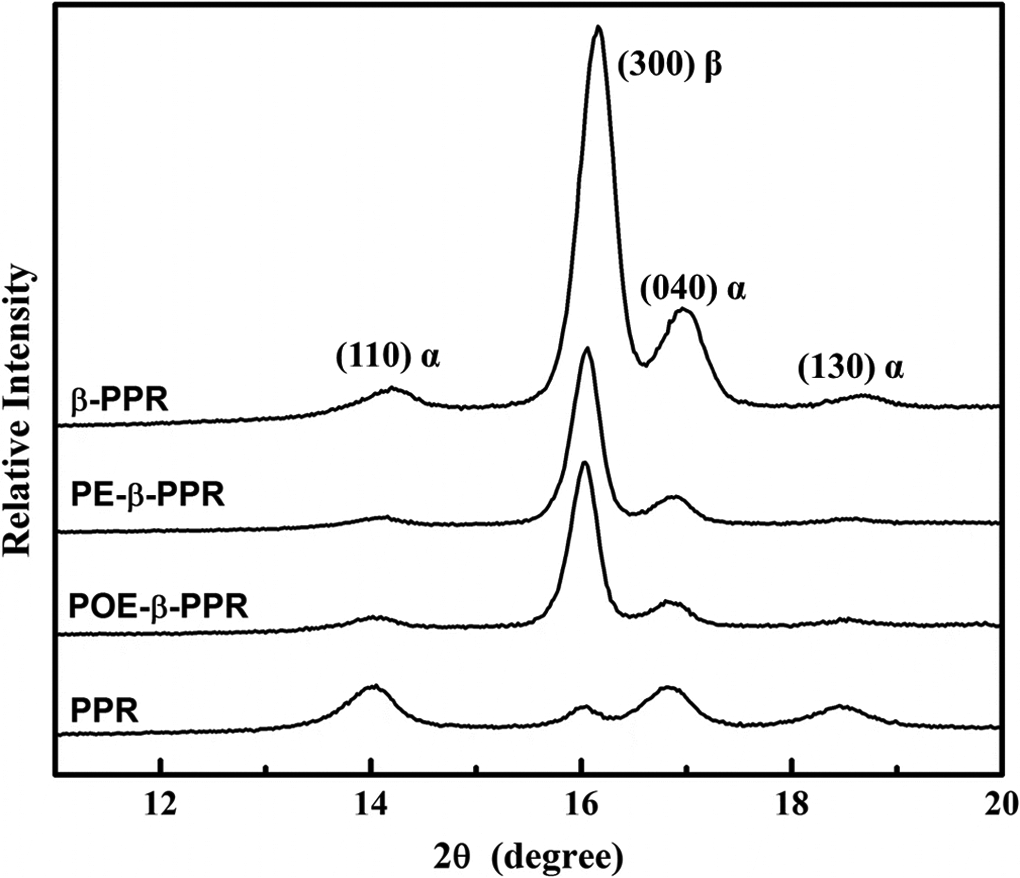
The WAXD spectra of PPR, POE-β-PPR, PE-β-PPR and β-PPR. WAXD: wide-angle x-ray diffraction; PPR: polypropylene random copolymer; POE: polyolefin elastomer; PE: polyethylene.
Figure 4 exhibits the isothermal crystallization morphologies of different samples. Obviously, the pristine PPR (Figure 3(a)) crystallizes in the form of large spherulites with clear boundaries, and the average spherulites diameter is about 100 μm. In the presence of β-NA (Figure 3(b) to (d)), the β-form crystal with acicular form and high birefringence appears and is in the dominant position. The size of the crystals becomes much smaller and the quantity increases dramatically. It is even very difficult to differentiate the spherulites one by one. It can be concluded that the crystalline structure of PPR matrix was successfully controlled by the addition of β-NA.

POM images show the isothermal crystallization morphologies of different samples. (a) PPR, (b) β-PPR, (c) POE-β-PPR, (d) PE-β-PPR. POM: polarization optical microscope; PPR: polypropylene random copolymer; POE: polyolefin elastomer; PE: polyethylene.
Mechanical properties
The fracture resistance of the different samples was detected by notched Izod impact measurement, and the results are shown in Figure 5. For pure PPR, it exhibits the impact strength of 47.8 kJ m−2. It increased to 65.8 kJ m−2 with the presence of β-NA. Surprisingly, the impact strength is dramatically increased with the coeffect of β-NA and the CSP, indicating an excellent toughening effect. It soared to 79.8 and 72.8 kJ m−2 for POE-β-PPR and PE-β-PPR, respectively, much higher than that of pure PPR and β-PPR. Two main reasons lead to the dramatic improvement in toughness. First, lots of β-form crystal appears with the incorporation of β-NA. The β-form PP without cross-hatching allows the initiation and propagation of the plastic deformation and then enhances the energy dissipating process, which leads to the higher toughness than α-PP. Second, the CSP acted as the stress concentrator under the load of stress and promoted the local plastic deformation of the surrounded PPR matrix. When the samples were impacted, the nano-CaCO3 particles that embedded in the PE or POE shell led to the cavitations formed on the interface and brought about the deformation of the CSP. Then, the deformation of the sandbag could dissipate impact energy, thus the impact property would be greatly improved. On the other hand, the impact strength of POE-β-PPR is higher than PE-β-PPR owing to the high intrinsic impact strength of POE.
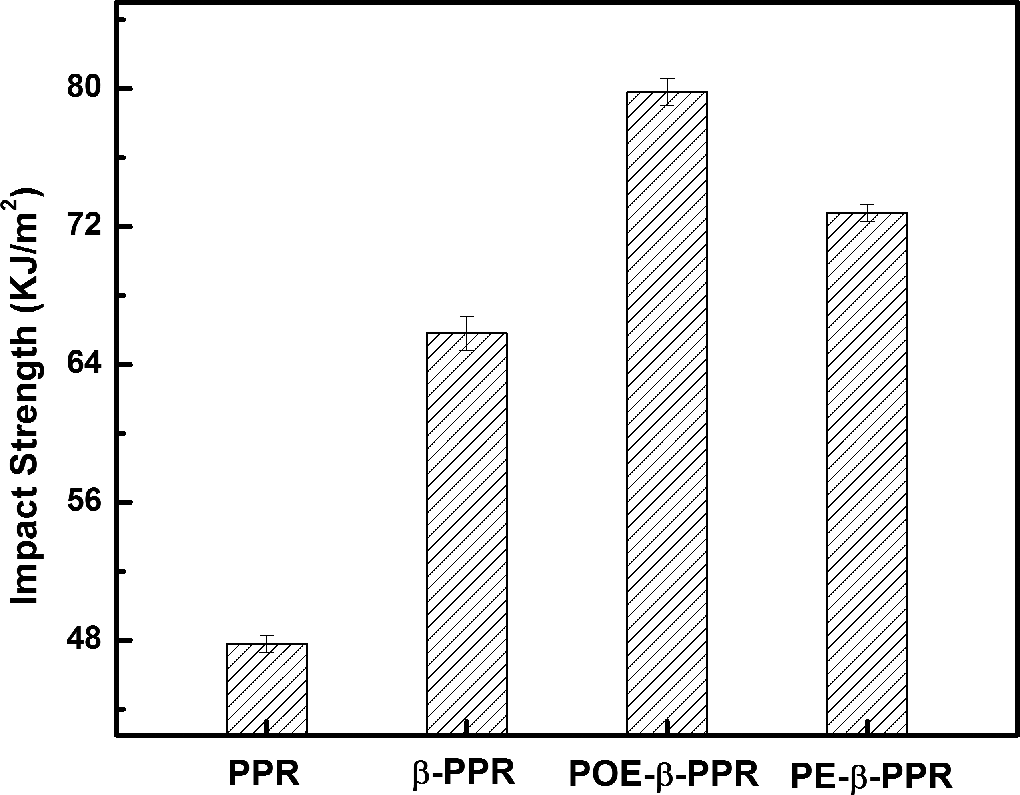
Izod impact strength of different samples.
The impact-fractured surface morphology study is favorable to understand the toughening improvement mechanism of the modified samples. So, the typical impact-fractured surface morphologies are studied by SEM. The overall impact-fractured surfaces are shown in Figure 6(a) to (d). And, they were further subclassified as crack initiation zone (Figure 6(a1) to (d1)), the early stage of crack propagation zone (Figure 6(a2) to (d2)), and the later stage of the crack propagation zone (Figure 6(a3) to (d3)). For pristine PPR, a relatively smooth and flat surface without considerable plastic deformation can be observed on the whole fracture surface. In other words, both the crack initiation and propagation occur at a very rapid rate in pure PP, which stands for the typical morphology of brittle fracture mode. For β-PPR, the fracture surface becomes rather coarse and uneven. But the area of the plastic deformation is still small and concentrated in the region that not far away from the crack roots. Furthermore, a parabolic ridge can be clearly seen in both Figure 6(b) and its enlarged part image, which relates to the change in the stress state. However, with the coeffect of β-NA and the CSP, the morphologies of the whole fractured surface have totally changed. First of all, the whole fractured surfaces of POE-β-PPR and PE-β-PPR become rather coarse compared with the other two specimens. Second, one could also notice that these two samples cannot be fractured completely due to the higher fracture resistance. The nonfractured part of the specimen during the impact process is shown by the rectangular dash lines in Figure 6(c) and (d). Besides, POE-β-PPR shows larger nonfractured parts compared with PE-β-PPR, suggesting the higher fracture resistance of the former sample. These results agree well with the test results of the impact strength. For these two samples, the crack initiation zone are relatively smooth, while large area of plastic deformation and a large amount of striations can be seen in the stage of crack propagation, indicating huge energy dissipation during the impact fracture. So, the overall analysis indicates that the coeffect of β-NA and the CSP induces the brittle–ductile transition of PPR.

SEM images show the impact-fractured surfaces of different samples at lower magnification. (a) PPR, (b) β-PPR, (c) POE-β-PPR, and (d) PE-β-PPR. The rectangular zone shown by dash lines represents the nonfractured part of the specimen during the impact process. (a1), (a2), and (a3) show enlarged view of subregion marked in (a); (b1), (b2), and (b3) show enlarged view of subregion marked in (b); (c1), (c2), and (c3) show enlarged view of subregion marked in (c); and (d1), (d2), and (d3) show enlarged view of subregion marked in (d). SEM: scanning electron microscopy; PPR: polypropylene random copolymer; POE: polyolefin elastomer; PE: polyethylene.
The tensile properties of neat PPR and different modification samples are also investigated, and the corresponding results are shown in Figure 7. The change of tensile properties depends greatly on the crystal and material internal morphology changing. As is known to all, the yield strength and stiffness of β-form PP is lower than that of α-crystal, while it also has better toughness and heat resistance stability owning to the specific structure of β-crystal. So, the yield strength (Figure 7(a)) decreased with the increasing Φβ or Kβ. However, for the tensile strength and elongation at break shown in Figure 7(b) and (c), their changing trend does not agree with the Φβ or Kβ change. This is attributed to the strain hardening phenomenon. That is to say, owing to the large amounts of β-crystal PP in the matrix, crystallization or orientation in the direction of tensile could happen when the material stretched greatly, thus results in the strength improvement in the tensile direction. So, the tensile strength and elongation at break all increased dramatically. But this is not suitable for the behavior of POE-β-PPR. It did not show the strain-hardening phenomenon, so the tensile strength is relatively lower than PPR, while the elongation also improved due to the presence of β-crystal. As the Φβ or Kβ is also high for POE-β-PPR, this phenomenon must be attributed to the different internal structure. As mentioned above, PE-β-PPR and POE-β-PPR show different CSP morphologies. So, the CSP structure must also play an important role in tensile properties improvement. For Young’s modulus, as shown in Figure 7(d), it has both decreased for β-PPR and POE-β-PPR, owning to the low stiffness of β-form crystal and POE. But surprisingly, the Young’s modulus for PE-β-PPR becomes even a little higher than pristine PPR. This is probably caused by the presence of both the rigid organic (PE) and inorganic (nano-CaCO3) particles, which compensate the deterioration of tensile modulus.
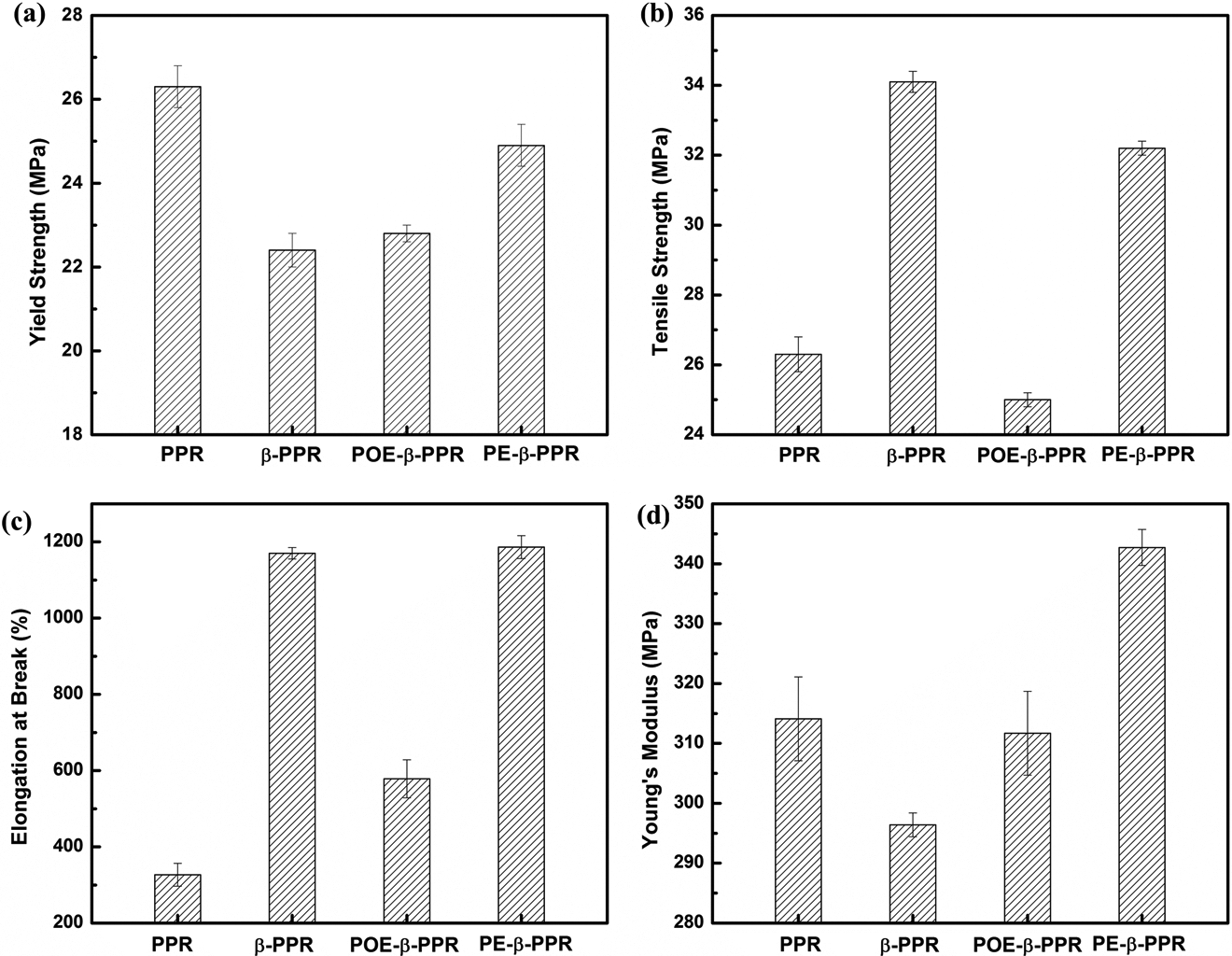
The tensile properties of different samples: (a) yield strength, (b) tensile strength, (c) elongation at break, and (d) Young’s modulus.
Thermal stability
The TGA curves of different samples are shown in Figure 8. It shows that the thermal degradation temperature of pristine PPR is about 360°C, and it increased dramatically after modification. For β-PPR, the thermal degradation temperature rises up to about 378°C, owing to the higher thermal stability of β-crystal PP. With the coeffect of β-crystal and CSP, the thermal degradation temperature for POE-β-PPR and PE-β-PPR all increased dramatically, to about 385°C, much higher than that of pure PPR and β-PPR. Also, thanks to the high thermal stability of the nanoinorganic fillers, the weight loss rate of the samples becomes slower, meaning the higher heat stability. For example, when 90% weight loss is selected as a point of comparison, the corresponding temperature is 343.8, 370.1, 382.8, and 381.5°C for PPR, β-PPR, POE-β-PPR, and PE-β-PPR, respectively. Herein, the thermal stability of PPR after modification has been greatly improved with the coeffect of β-crystal and CSP.
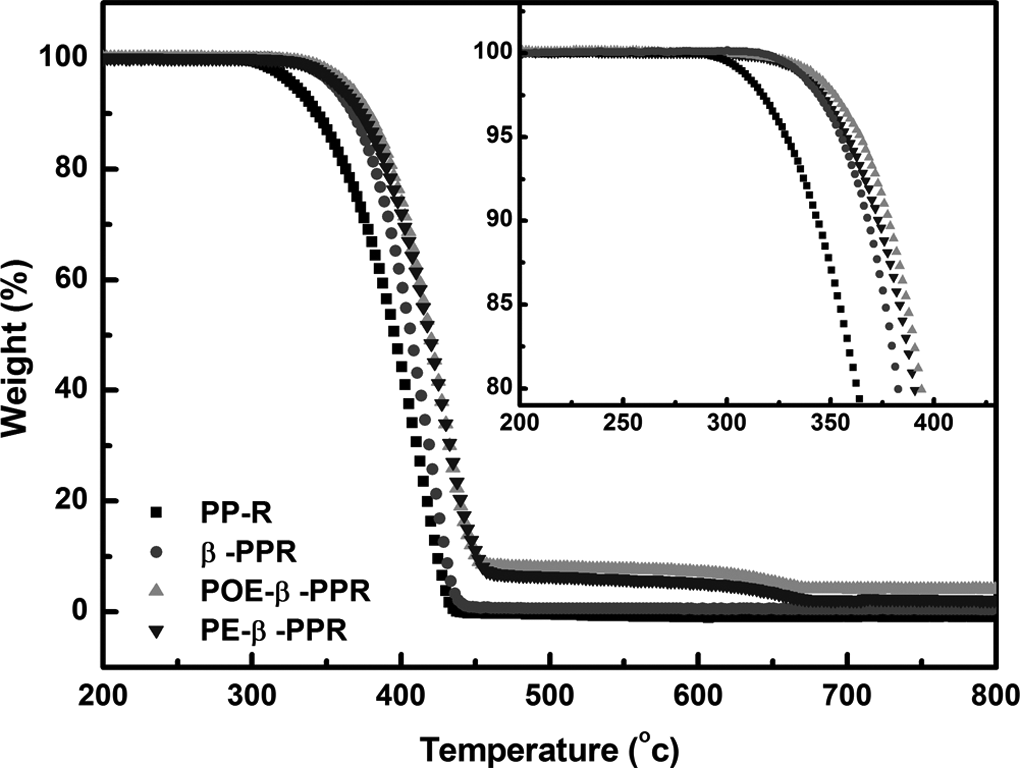
Thermogravimetric analysis curves of different samples.
Conclusion
In this work, the mechanical properties and thermal stability of PPR were greatly improved with the coeffect of β-NA and the core–shell structure. The CSP dispersed uniformly in PPR matrix, and the core–shell structure was easily distinguishable in TEM images. POE-β-PPR and PE-β-PPR showed different core–shell structure, owing to their different melt flow index. This also leads to the different tensile properties. The relative content of the β-crystal was dramatically increased with the addition of β-NA, which is beneficial to the toughness and thermal stability improvement. With the coeffect of β-NA and CSPs, the toughness increased, while the tensile strength and Young’s modulus decreased for POE-β-PPR. On the other hand, the PE-β-PPR achieved excellent toughness and tensile strength without lowering its stiffness. The thermal degradation temperatures of these two samples were all raised by about 25°C and their weight loss rate became much slower. This work provides a novel method for PPR toughening and thermal stability improvement.
Footnotes
Acknowledgements
The authors sincerely acknowledge the National Key Technology R&D Program of China (2012BAD32B01) and the Outstanding Technology Foregoer Plan of Shanghai (12XD1420400) for their support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
