Abstract
New Schiff-base polyesters were prepared from a dihydroxy monomer (E)-1-(5-(4-hydroxy-3-chlorobenzylidene)thiocarbamoylaminonaphthyl)-3-(4-hydroxy-3-chlorobenzylidene)thiourea. The processable pyridine or thiophene-based poly(azomethine–ester)s (PAEs) possess high molar mass of 111 × 103–127 × 103 g mol−1. The 10% weight loss (521–527°C) and glass transition temperature (287–291°C) depicted high thermal stability. Polyaniline (PAN) doped with dodecylbenzenesulfonic acid (PAN/DBSA), prepared by in situ doping polymerization, was blended in solution/melt with PAEs. Field emission scanning electron microscopy of melt blended PAEs/PAN/DBSA showed nanolevel homogeneity of microstructure accountable for superior electrical conductivity (1.9–2.8 S cm−1). The miscible nanoblends also exhibited high heat stability (T 10; 500–507°C) and mechanical strength (45.51–48.17 MPa) relative to reported azomethine/PAN-based structures.
Keywords
Introduction
Recently, heat-resistant linear polyester materials have received great eminence owing to their demand in advance applications. 1 However, there are several limitations because of poor physical, mechanical, thermal, and processing characteristics. 2 In this regard, introduction of heteroaromatic rings into the backbone has been explored as an expedient approach to acquire thermally stable polymers.3,4 New types of conducting organic polymers have also secured researcher’s interest due to their excellent electrical properties, superior thermal stability, high surface area, good environmental stability, and corrosion resistance along with suitable strength and fracture toughness.5,6 Incidentally, poly(azomethine)s (polyimines/Schiff-base polymers) with a system of conjugated –C=N– bonds exhibit valuable properties mainly associated with the conjugated backbone and the presence of imine sites.7,8 Thermal stability of some poly(azomethine)s has found to be similar to polyamides and also hold semiconductor properties.6 –8 These polymers are generally difficult to process due to their high melting point and low solubility. The technical applications of polyimines have strongly been restricted by their sensitivity to acidic hydrolysis and thermal exchange reactions between the azomethine groups. Many efforts have been focused on enhancing these characteristics by varying the nature of components or the introduction of flexible or bent substituents into the backbone.9,10
Over the last decade, significant progress has been achieved to improve the processability and mechanical properties of conducting polyanilines (PANs). In PAN, a conjugated double bond structure, composed of the benzenoid ring between the quinoid imine and the benzenoid amine structures, forms the conjugation π-bond system. The conjugated double bonds permit easy electron mobility throughout the molecule due to electrons delocalization. Among the known oxidation states of PAN, only doped emeraldine-based form, being a partially reduced and partially oxidized form, is conductive. The incorporation of PAN into an electrically insulating conventional polymer matrix has received attention due to the possibility of combining the good processability and mechanical performance of the conventional polymers with the electrical properties of PAN. These blends are normally obtained by in situ polymerization of aniline in the presence of an insulating polymer or by blending soluble or fusible PAN with other polymers in solution or melts.11,12 The mixing of polymers has become commercially and technologically more important than the synthesis of homopolymers and copolymers in the last decade. Blending allows creating new materials with specific properties for the desired application at low cost. Since the conducting polyblends are stable and retain the mechanical properties of the host polymer, films, fibers, and coatings can be fabricated by solvent evaporation or by melt processing for use in antistatic applications, for electromagnetic shielding, microwave absorption devices, and/or for transparent conducting films. 13 Blending of PAN with conventional polymers through solution processing has been reported to be efficient when both polymers are soluble in a common solvent. The solubility of PAN in common solvents has been achieved through a counter-ion-induced processability method, where a specific functionalized protonic acid is used as protonating agent.14,15 Doping is the process by which polymers that are insulators are exposed to charge transfer agent (dopant). The properties of PAN not only depend on the oxidation state but also on its protonation state/doping level and also on the nature of dopants. The electrical conductivity of undoped PAN is reported to be in the range of 10−10–10−7 S cm−1. Upon acid doping, it attains higher values in the range of 10−2–100 S cm−1, depending on the dopant. These dopants enhance the solubility of PAN in common organic solvents and enable the formulation of conducting blends in which PAN and other organic polymers are dissolved and blended in a common solvent. For instance, a functionalized acid dodecylbenzenesulfonic acid (DBSA), having long alkyl chains, is capable of increasing the solubility of PAN doped with DBSA (PAN/DBSA) in different solvents, thereby, increasing the compatibility with polymer matrices. 14 PAN/DBSA complex is also used in melt blends, owing to its fairly high thermal stability.16,17
This endeavor initially deals with the fabrication of poly(azomethine–ester)s (PAEs) and later the development of PAE/PAN/DBSA blends using two techniques. The thermal stability and electrical conductivity of new PAEs were found to increase by the incorporation of pyridine, thiophene or azomethine linkages in the backbone. A dihydroxy compound, (E)-1-(5-(4-hydroxy-3-chlorobenzylidene)thiocarbamoylaminonaphthyl)-3-(4-hydroxy-3-chloro-benzylidene)thiourea (HCBTCANT), has been designed and exploited to develop polymers with high molar mass and improved physical properties. Second, the present study is focused on some novel PAE/PAN/DBSA blends prepared by the dispersion of PAN in PAEs matrix using two methods (1) solution blending and (2) melt blending. The effects of the varying PAN content and the blending technique employed on morphology, thermal stability, mechanical strength, and conductivity of the blends were investigated. Miscible nanoblend morphology was observed upon the melt blending of PAEs with PAN, depicting their fine compatibility. Thermal and mechanical stability in addition to electrical conductivity of the melt-blended-miscible materials were found to be superior relative to those obtained using other technique.
Experimental
Materials
Aniline (analytical grade, Merck, Nottingham, UK) was distilled twice under vacuum and stored under nitrogen (N2) in a refrigerator. Ammonium peroxydisulfate (APS; 99%), N-methylpyrrolidone (NMP; 99%), dimethyl sulfoxide (DMSO; 99%), and chloroform (99%) were also procured from Merck. DBSA (technical grade), 2,6-pyridinedicarbonyl chloride (PDC; 97%), thiophene-2,5-dicarbonyl dichloride (TDC; 97%), 1,5-diaminonaphthalene (DAN; 97%), 3-chloro-4-hydroxybenzaldehyde (98%), N,N-dimethylacetamide (DMAc; 99%), xylene (99%), and toluene (99.8%) were obtained from Aldrich (St. Louis, Missouri, USA). Ammonium thiocyanate (98%), dimethylformamide (DMF; 99%), and tetrahydrofuran (THF; 99.5%) were obtained from Fluka (Biocen GmbH, Duesseldorf, Germany).
Measurements
Perkin-Elmer (Washington, USA) 2400 CHN elemental analyzer was used for the elemental analysis. Infrared spectra were recorded using Fourier transform infrared (FTIR) spectrometer, Model no. FTSW 300 MX, manufactured by Bio-Rad (California, USA) (4 cm−1 resolution). Nuclear magnetic resonance (NMR) spectra were scanned at room temperature using Bruker (Tokyo, Japan) Spectrometer (300.13 MHz for proton ( 1 H) NMR and at 75.47 MHz for carbon ( 13 C) NMR) in deuterated DMSO (DMSO-d 6). Inherent viscosity (η inh) was measured in THF (30°C) with an Ubbelohde viscometer for 0.5 g dL−1 polymer solution. The weight-average molecular weight (M w) was calculated using gel permeation chromatography (GPC) with THF as an eluent and refractive index detector. Thermal stability of the polymers was verified using Mettler Toledo (California, USA) thermogravimetric analysis (TGA)/SDTA 851e thermogravimetric analyzer with 1–5 mg of the sample in aluminum oxide crucible at a heating rate of 10°C min−1. Differential scanning calorimetry (DSC) was performed using Mettler Toledo DSC 822e differential scanning calorimeter taking 5–10 mg of samples in aluminum pans and heated at a rate of 10°C min−1. X-Ray diffractometer (3040/60 X’pert PRO, California, USA) with nickel-filtered copper Kα radiation (40 kV, 30 mA) was used to obtain x-ray diffractogram of polymers at room temperature. Field emission scanning electron microscope (FESEM) of freeze-fractured samples was performed using JSM5910 (JEOL, Japan). Electrical conductivities of thin films were measured using a Keithley (California, USA) 614 electrometer and the four-probe method. Stress–strain response of the blend samples were obtained on Universal Testing Machine INSTRON (Norwood, United States) 4206 according to the ASTM 638 method. A crosshead speed of 100 mm min−1 was used during the test.
Synthesis of TCANT
DAN (0.2 mol), concentrated hydrochloric acid (HCl; 16 mL), ammonium thiocyanate (0.2 mol), and deaerated water (120 mL) were mixed and heated in a porcelain dish for 2 h on a steam bath. The mixture was allowed to cool to room temperature. Afterward, the mixture was gradually evaporated to dryness for 6–7 h. The product obtained was boiled with charcoal in ethanol, filtered, and cooled. 1-(5-Thiocarbamoylaminonaphthyl)thiourea (TCANT) was recrystallized from ethanol and dried under vacuum at 90°C for 36 h.18,19 Elemental analysis for C12H12N4S2 (1-(5-thiocarbamoylaminonaphthyl)thiourea): calculated = %C 52.15, %H 4.38, %N 20.27; found = %C 51.07, %H 4.19, %N 19.41. FTIR (potassium bromide; KBr): 3319 cm−1, 3276 cm−1 (N–H stretch), 3036 cm−1(aromatic C–H stretch), 2934 cm−1(aliphatic C–H stretch), 1598 cm−1 (N–H bend), 1411 cm−1 (C–N stretch), 1153 cm−1 (C=S stretch). 1H NMR (300.13 MHz, DMSO-d 6, δ ppm): 4.14 (s, 4H, Ha), 9.26 (s, 2H, Hb), 6.45 (d, 2H, Hc), 7.01 (t, 2H, Hd), 7.22 (d, 2H, He). 13C NMR (75.47 MHz, DMSO-d 6, δ ppm): thiocarbonyl C=S 177.2 (C1), 141.4 (C2), 107.3 (C3), 125.1 (C4), 110.2 (C5).
Synthesis of HCBTCANT
TCANT (0.02 mol), 3-chloro-4-hydroxybenzaldehyde (0.04 mol), and absolute alcohol (250 mL) were agitated in a Dean–Stark apparatus for 30 min at room temperature. The above mixture was further refluxed for 2.5 h after the removal of azeotropic mixture at 100°C. The reaction contents were cooled to collect the solid and washed with dilute HCl and hot water. Finally, the product obtained by recrystallization (ethyl alcohol) was dried at 80°C for 36 h to give HCBTCANT with 95.1% yield. Elemental analysis for C26H18Cl2N4O2S2 (((E)-1-(5-(4-hydroxy-3-chlorobenzylidene)thiocarbamoylaminonaphthyl)-3-(4-hydroxy-3-chloro-benzylidene)thiourea): calculated = % C 56.42, % H 3.28, % N 10.12; found = % C 54.89, % H 3.11, % N 9.80. FTIR (KBr; Figure 1): 3439 cm−1 (–OH), 3297 and 1599 cm−1 (secondary amine N–H stretch and bend), 3075 cm−1(aromatic C–H stretch), 2928, 2859 (aliphatic C–H stretch), 1612 cm−1(–C=N– stretch), 1124 cm−1 (C=S stretch). 1H NMR (300.13 MHz, DMSO-d 6, δ ppm) given in Figure 2: 5.03 (s, 2H, Ha), 6.71 (d, 2H, Hb), 7.35 (d, 2H, Hc), 7.49 (s, 2H, Hd), 8.02 (s, 2H, He), 8.83 (s, 2H, Hf), 6.53 (d, 2H, Hg), 7.14 (t, 2H, Hh), 6.98 (d, 2H, Hi). 13C NMR (75.47 MHz, DMSO-d 6, δ ppm) is shown in Figure 3: 155.8 (C1), 117.4 (C2), 128.7 (C3), 127.8 (C4), 130.7 (C5), 124.3 (C6), 163.3 (C7), thiocarbonyl C=S 184.1 (C8), 141.2 (C9), 109.1 (C10), 125.3 (C11), 111.8 (C12).
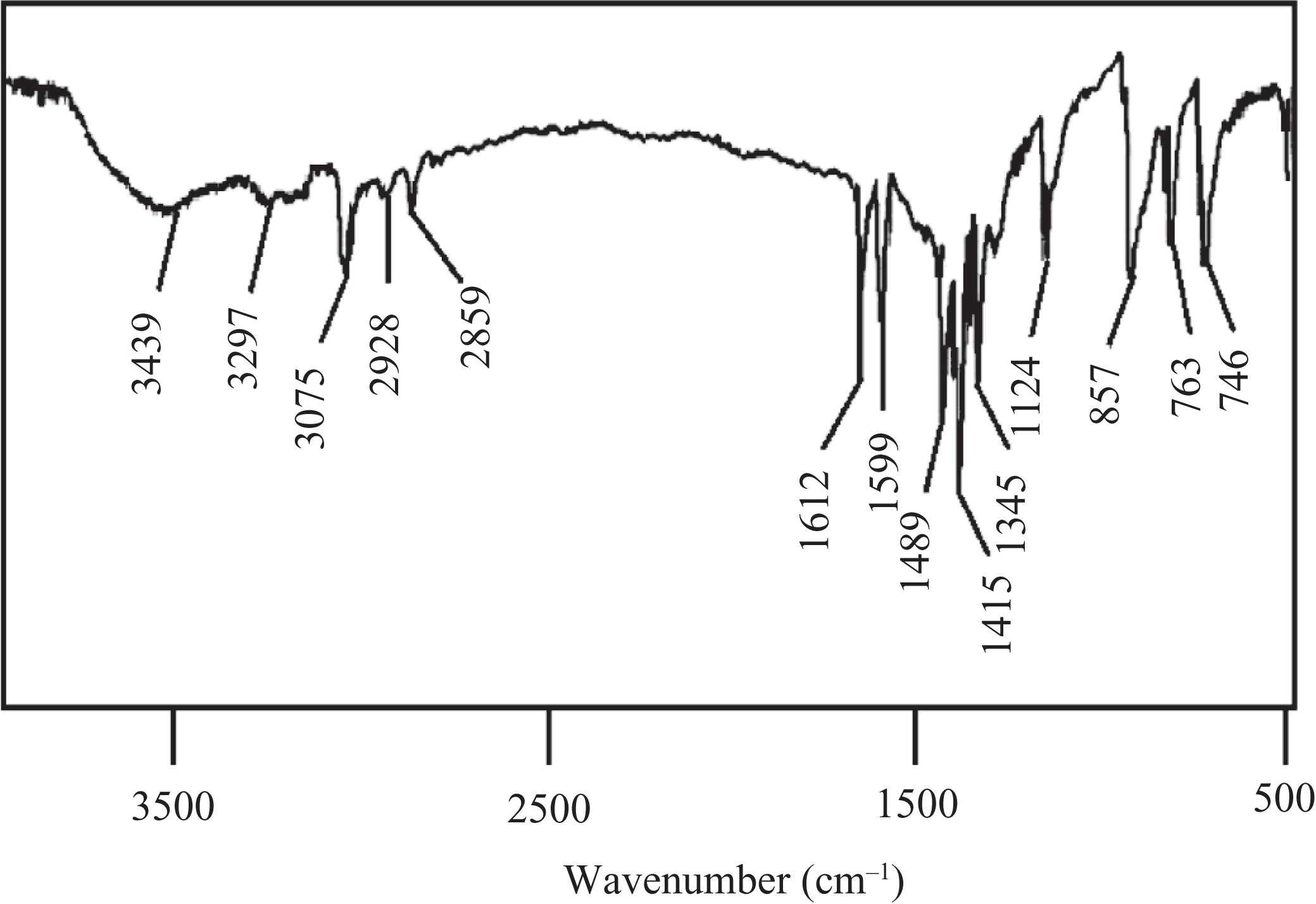
FTIR spectrum of HCBTCANT. FTIR: Fourier transform infrared; HCBTCANT: (E)-1-(5-(4-hydroxy-3-chlorobenzylidene)thiocarbamoylaminonaphthyl)-3-(4-hydroxy-3-chloro-benzylidene)thiourea.
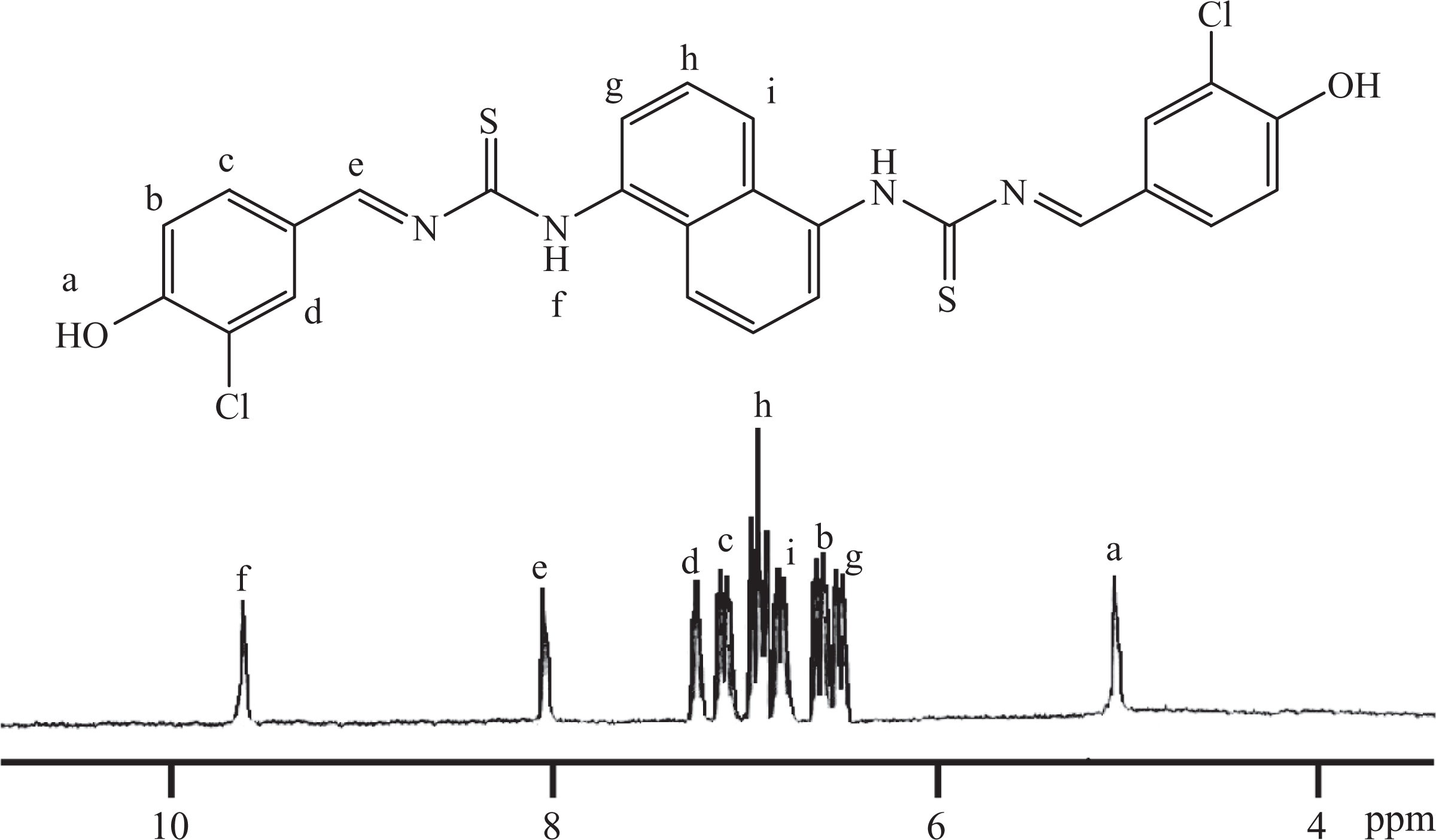
1H NMR spectrum of HCBTCANT. 1H NMR: proton nuclear magnetic resonance; HCBTCANT: (E)-1-(5-(4-hydroxy-3-chlorobenzylidene)thiocarbamoylaminonaphthyl)-3-(4-hydroxy-3-chloro-benzylidene)thiourea.
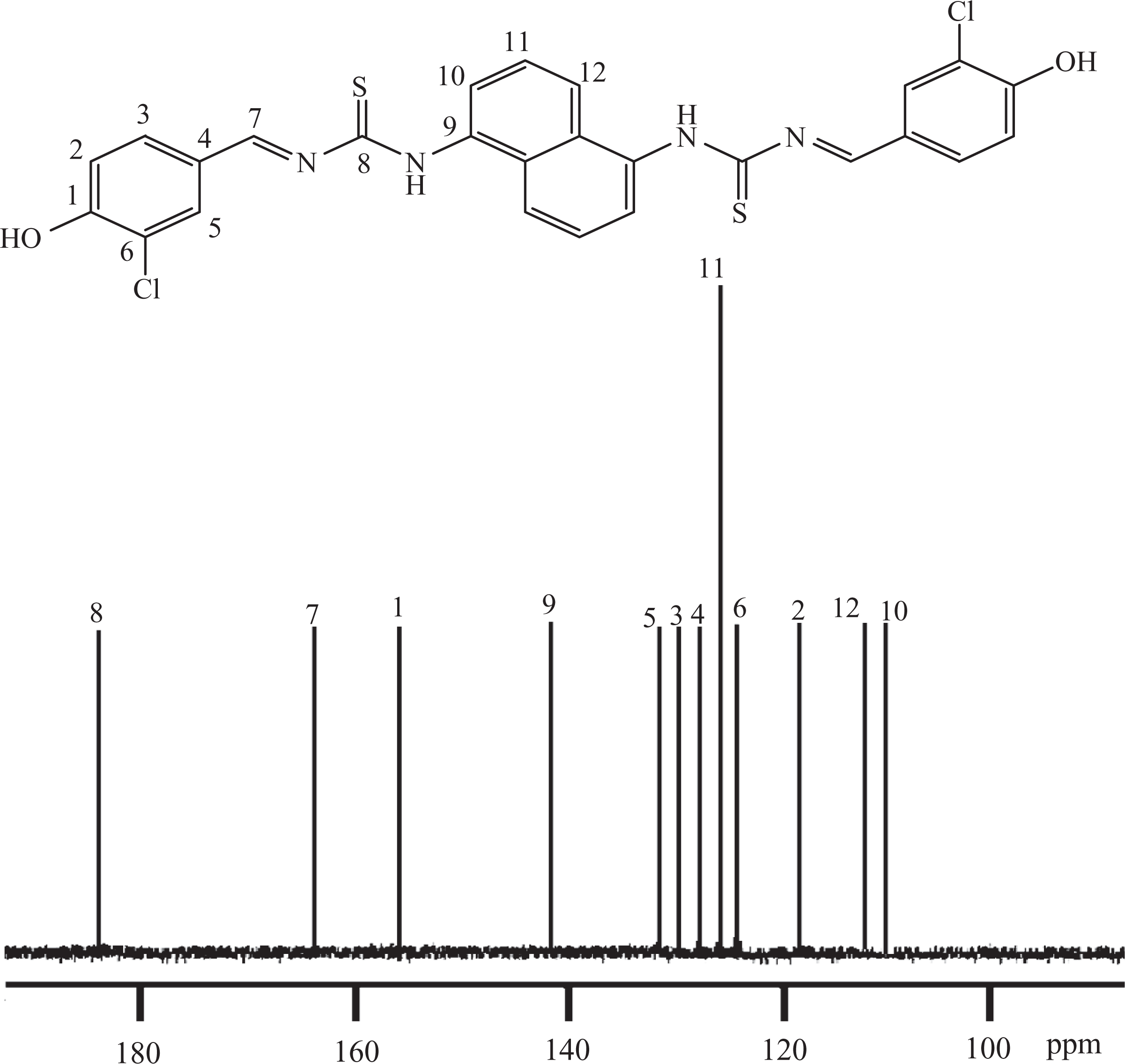
13C NMR spectrum of HCBTCANT.13C NMR: carbon nuclear magnetic resonance; HCBTCANT: (E)-1-(5-(4-hydroxy-3-chlorobenzylidene)thiocarbamoylaminonaphthyl)-3-(4-hydroxy-3-chloro-benzylidene)thiourea.
Synthesis of PAEs
Into a 250-mL round-bottomed flask, HCBTCANT monomer (0.01 mol) and DMF (50 mL) were charged and heated at 100°C with stirring under N2 atmosphere. Then, 0.01 mol diacid chloride (PDC or TDC) dissolved in DMF (20 mL) was added to the reaction mixture and heated further at 150–160°C for 20 h. 20 The mixture was filtered and the solution was slowly poured into methanol. The resultant precipitate was filtered, washed with methanol, and dried at 100°C under vacuum. The solid residue was dissolved in hot chloroform, precipitated in methanol, and dried again. Yield of the obtained polymer ranged between 93 and 95%. Spectral data of PAE 1 corroborated the structure of the polymer synthesized (Figure 4): 3338 cm−1(N–H stretch), 1599 cm−1 (N–H bend), 3057 cm−1 (aromatic C–H stretch), 2913, 2869 cm−1 (aliphatic C–H stretch), 1736 cm−1 (ester C=O stretch), 1612 cm−1(–C=N– stretch), 1124 cm−1 (C=S stretch). 1H NMR (300.13 MHz, DMSO-d 6; Figure 5): PAE 1 displayed doublets nearly at 8.34 and 8.73 ppm and triplets at 8.61 ppm owing to pyridine unit protons. The naphthalene ring displayed chemical shifts around 6.92 (triplet), 7.23 (doublet), and 7.38 (doublet) ppm. A singlet at 8.01 ppm was attributed to the azomethine proton. Furthermore, the aromatic ring protons appeared about 7.61 and 7.79 ppm as doublet, while singlet was found at 7.89 ppm.
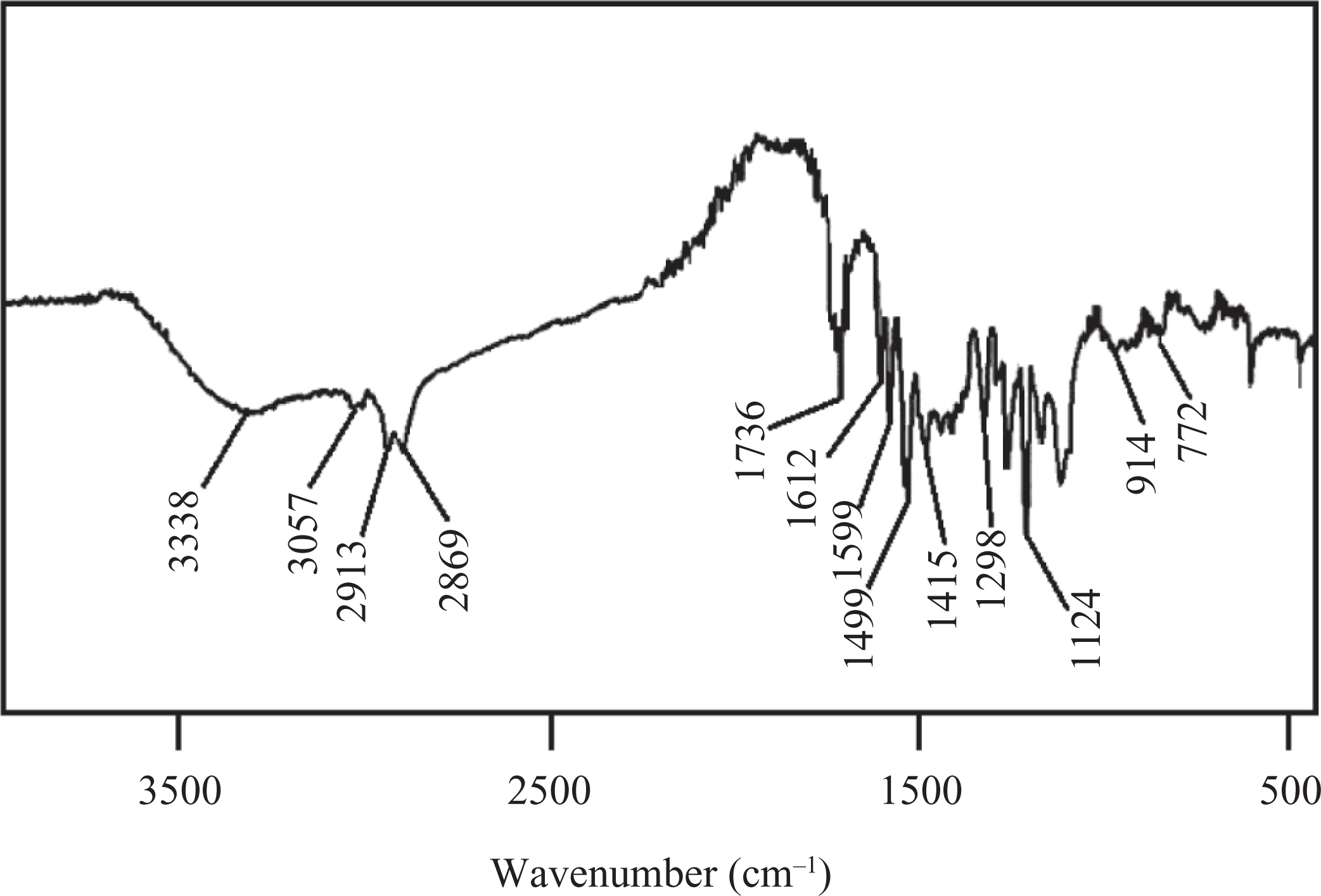
FTIR spectrum of PAE 1. FTIR: Fourier transform infrared; PAE: poly(azomethine–ester).
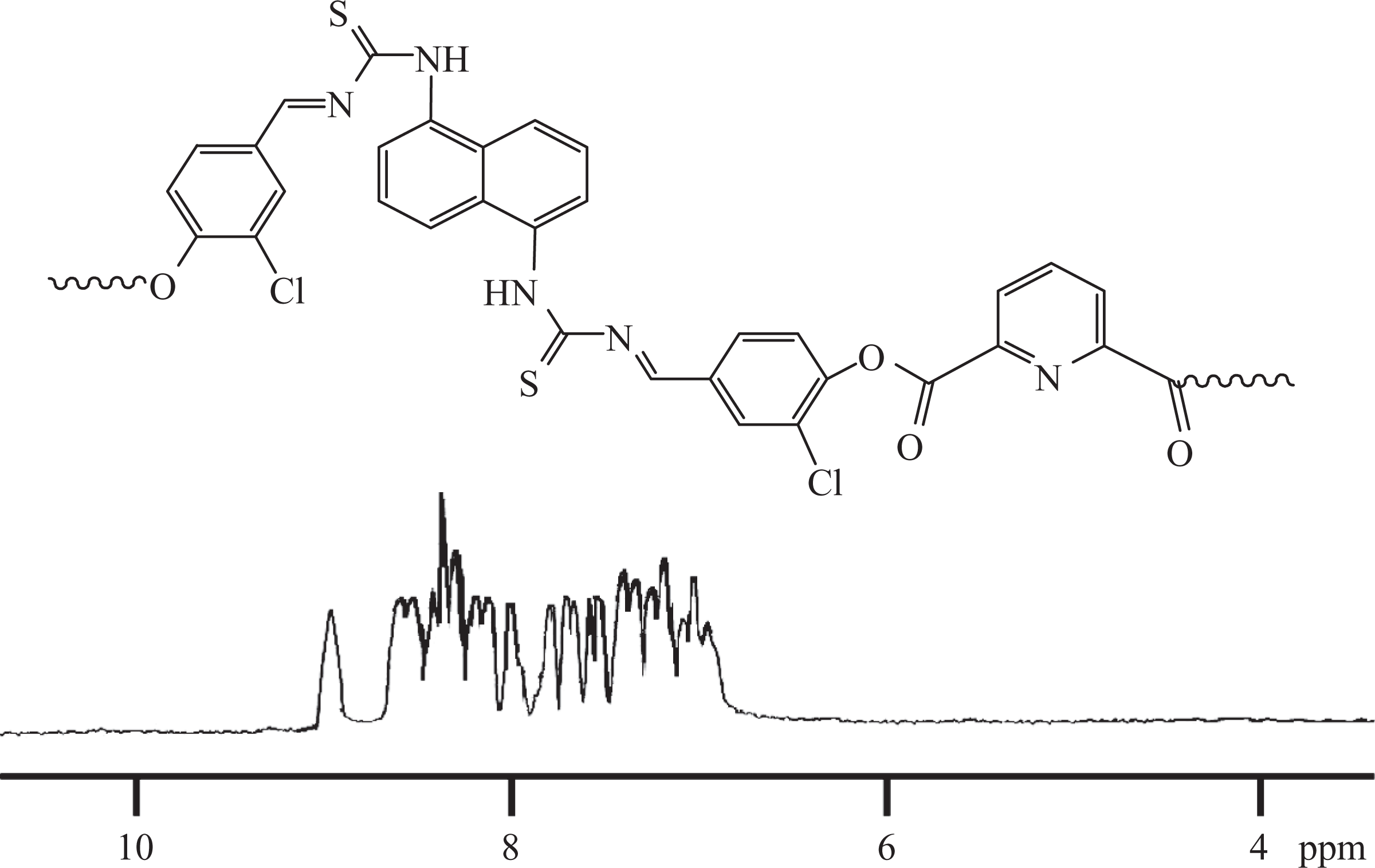
1H NMR spectrum of PAE 1. 1H NMR: proton nuclear magnetic resonance; PAE: poly(azomethine–ester).
Synthesis of PAN/DBSA
The emulsion polymerization of aniline was carried out according to a reported procedure.21,22 In a typical reaction, DBSA (0.075 mol) and aniline (0.051 mol) were dissolved in 250 mL of toluene with continuous stirring at 0°C. An aqueous solution of APS (0.021 mol) in water (20 mL) was then slowly added keeping the mixture at 0°C. The mixture was allowed to stir for further 6 h to attain the complete polymerization. The reaction mixture was poured into methanol and the precipitate was filtered, washed with methanol, and dried under vacuum. PAN/DBSA (a dark green powder), prepared by this method, may display a conductivity up to 1.2 S cm−1.
PAEs/PAN/DBSA blends
Solution blending
Solutions of PAN/DBSA and PAEs in chloroform were prepared separately. The PAE solution was added to the solution of conducting polymer at required proportions. The mixed solutions were ultrasonicated (30 W and 50 Hz) for 5 h. Thin films (approximately 160 µm thickness) were casted by pouring the blend solutions on glass petri dishes, and solvent was evaporated at 40°C for 24 h.
Melt blending
Melt blending of the components was carried out by melt mixing in a Brabender plastograph, using a previously described method. 23 Flat plaques, 2.3 mm thick, for the conductivity measurement, were prepared by compression molding.
Results and discussion
Development of PAEs and their miscible blends
Synthesis of new polyesters with azomethine–thiourea–naphthalene–chloro–pyridine/thiophene backbone architecture is an uncharted research zone. In general, azomethine, pyridine, and thiophene groups, when incorporated into the backbone, impart heat stability to the polymer with lowering its solubility. Conversely, chloro unit provides lower energy of internal rotation to the polymer chains. A new generation of conducting and thermally stable polymers was designed exhibiting cumulative effect of the above-mentioned groups. The synthetic route to new dihydroxy monomer HCBTCANT and its polycondensation with PDC/TDC is given in Figure 6. High-performance polyesters, having good η inh (1.59–1.73 dL g−1) and M w (111–127 × 103 gmol-1), were obtained in high yield. Thermal stability data showed that the structural modification of polyesters improved the heat stability and glass transition without affecting their solvent miscibility. In the second phase of this original research toil, PAN/DBSA was prepared using in situ doping polymerization and employed for the blend manufacture. The blending of PAEs with PAN/DBSA (via solution/melt technique) unlocked a remarkable route to electrically conducting miscible nanoblends owing to the physical interaction between two matrices (Figure 7). The blends also exhibit fine thermal and mechanical profile.
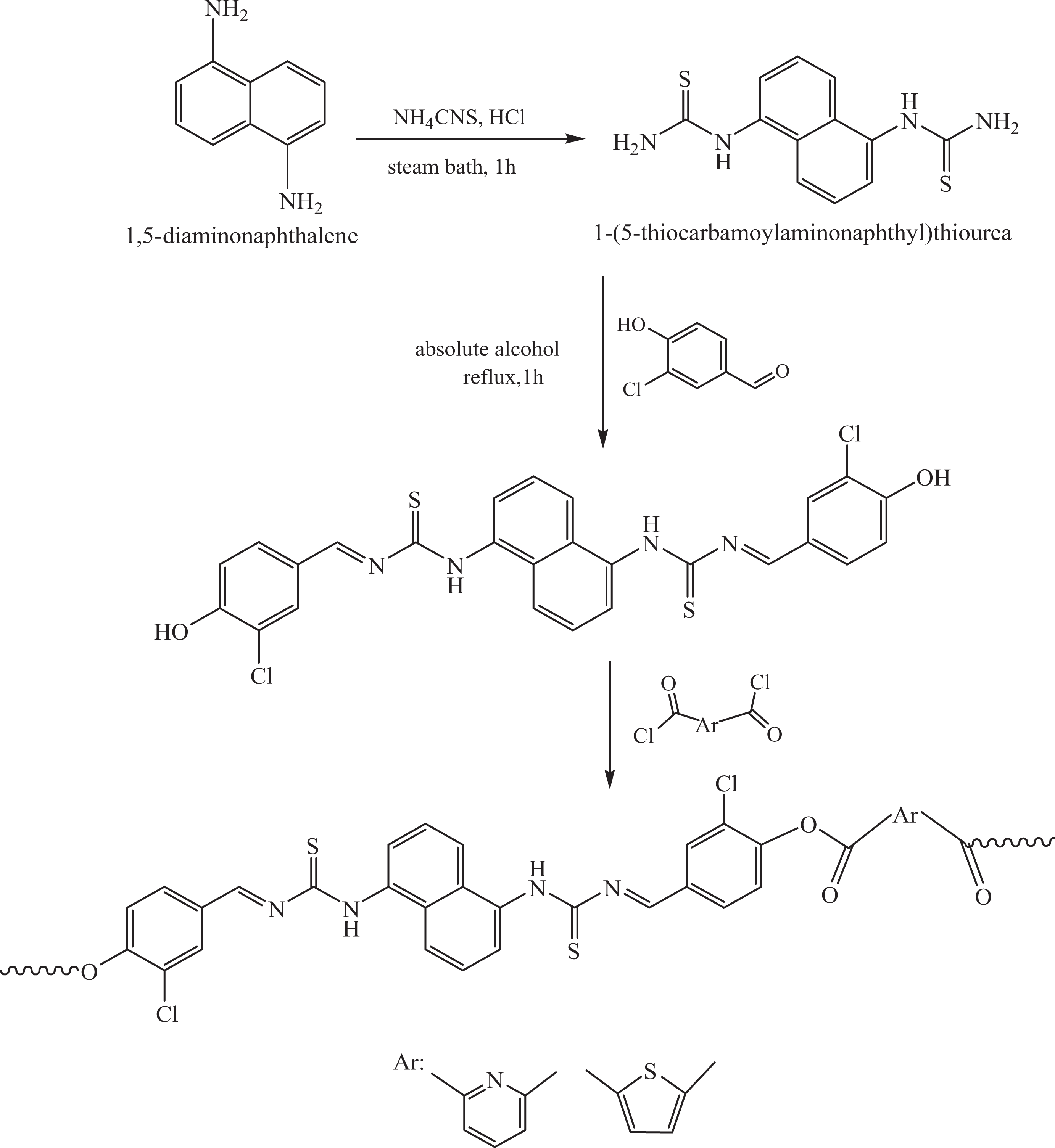
Schematic representation of the synthesis of monomer and poly(azomethine–ester)s.
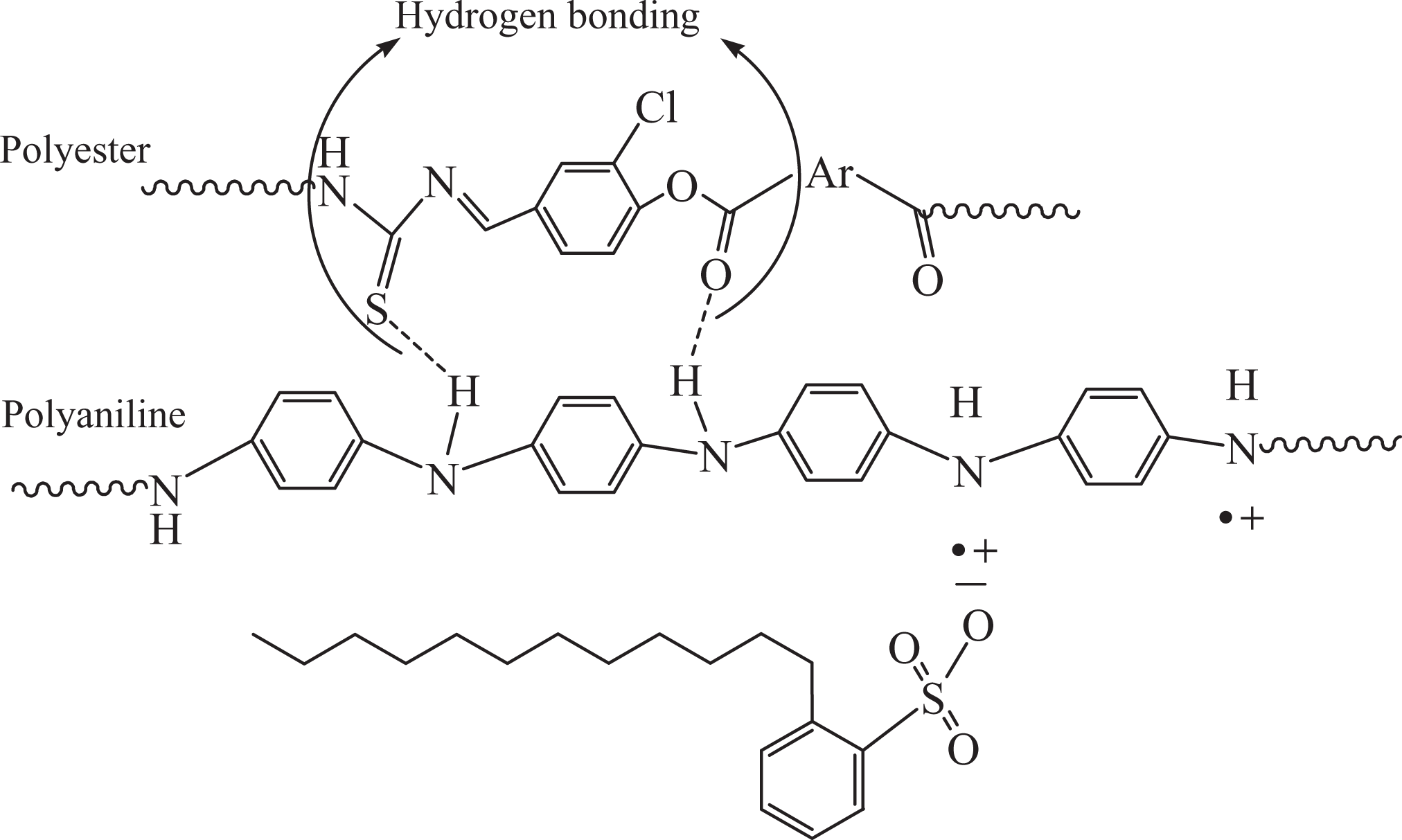
Schematic representation of the hydrogen bonding between poly(azomethine–ester) and PAN/DBSA components in the blend. PAN: polyanilines; DBSA: dodecylbenzenesulfonic acid.
Organosolubility
Qualitative solubility was determined by dissolving 15 mg of the polymer in 1 mL of the solvent, stirring for 24 h at room temperature and heating up to the solvent’s boiling temperature for the samples that remained insoluble at room temperature. The solubility properties of PAEs were studied in various solvents. As shown in Table 1, PAEs were soluble in all polar aprotic solvents (DMSO, DMAc, and DMF) at room temperature except NMP. However, the samples were soluble in NMP and THF on heating. Less polar toluene also could not dissolve the polymers even on heating. Generally, aromatic polyazomethine, polythiophene, or heteroaromatic polymers have a very low solubility in organic solvents because of their rigid backbone structure. In this regard, the introduction of flexible substituents in the main chain offers positive effect on the solubility of these polymers. In chlorinated solvents such as chloroform and tetrachloroethane, PAE 1 and 2 were only partially soluble at room temperature. This can be attributed to the presence of pyridine and thiophene moieties along the backbone in addition to the development of hydrogen bonding between –OCO–/C=S with NH groups, responsible for the backbone rigidity. Both the polymers were easily soluble in chloroform on heating. Similar effect was observed when PAEs were dissolved in xylene. The overall good solubility of new polymers bearing azomethine and heteroaromatic structure can be explained by the presence of flexible (chloro) or bulky (naphthalene) linkages in the main chain, which created a distance between the macromolecular chains preventing close packing, as a result the diffusion of solvent is facilitated. The solubility was also found to be superior relative to the previous azomethine or thiophene-based polymers.24,25
Solubility behavior of PAEs.a
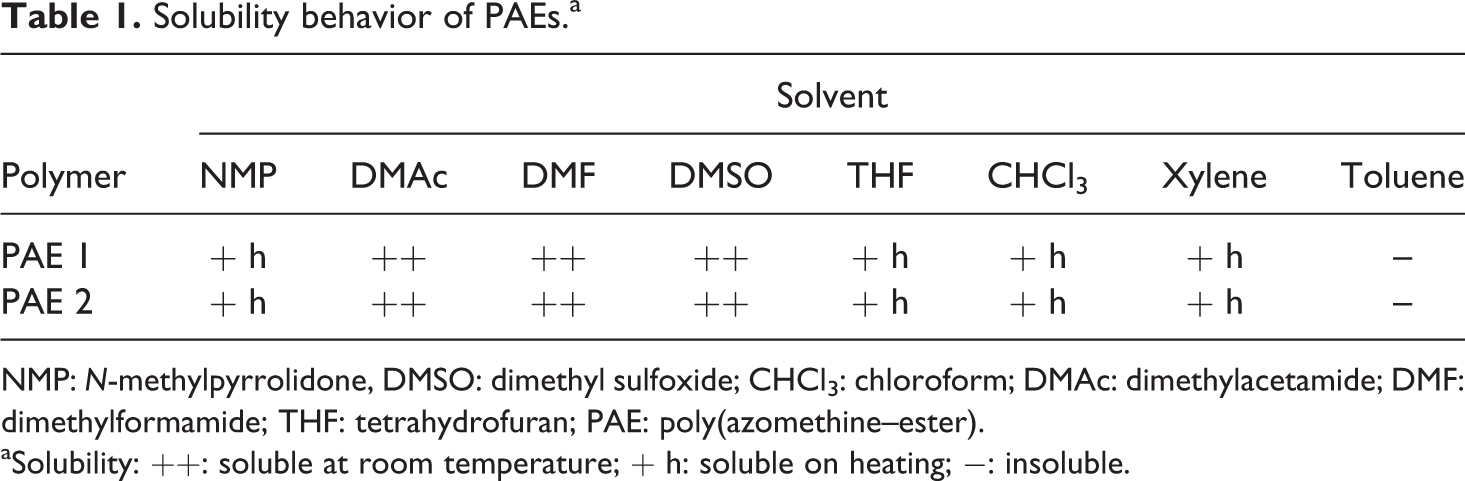
NMP: N-methylpyrrolidone, DMSO: dimethyl sulfoxide; CHCl3: chloroform; DMAc: dimethylacetamide; DMF: dimethylformamide; THF: tetrahydrofuran; PAE: poly(azomethine–ester).
aSolubility: ++: soluble at room temperature; + h: soluble on heating; −: insoluble.
Molecular weight
Molecular weight of polymers was studied using η inh and GPC measurements (Table 2). Inherent viscosity (η inh) of a polymer is generally considered as a measure of its molecular weight. η inh of PAEs measured in THF (30°C) was in the range 1.59–1.73 dL g−1. A relationship between the polymer structure and η inh can be clearly observed in this study. The data obtained showed higher viscosity of PAE 1 (1.73 dL g−1) and was found to decrease for the polymer derived from HCBTCANT and TDC (1.59 dL g−1). Higher η inh of PAE 1 with pyridine-based structure, –HCBTCANT–PDC–, was indicative of the higher chain stacking efficiency. This may be accredited to more rigid and symmetrically aligned polymer chains of PAE 1 bearing six-membered heteroaromatic architecture. GPC results revealed that HCBTCANT monomer had fine polymerization potential to yield sufficiently high-molecular-weight polymers with the diacid chlorides. Similar to the above examination, polycondensation of new dihydroxy compound with PDC gave higher molecular weight (M w; 127 × 103 gmol−1) compared with PAE 2 (M w; 111 × 103 gmol−1). Briefly, new PAEs exhibited much higher molar mass relative to the previously synthesized azomethine/thiophene-derived polyesters.24,25
η inh, M w, polydispersity, and percentage yield of PAEs.
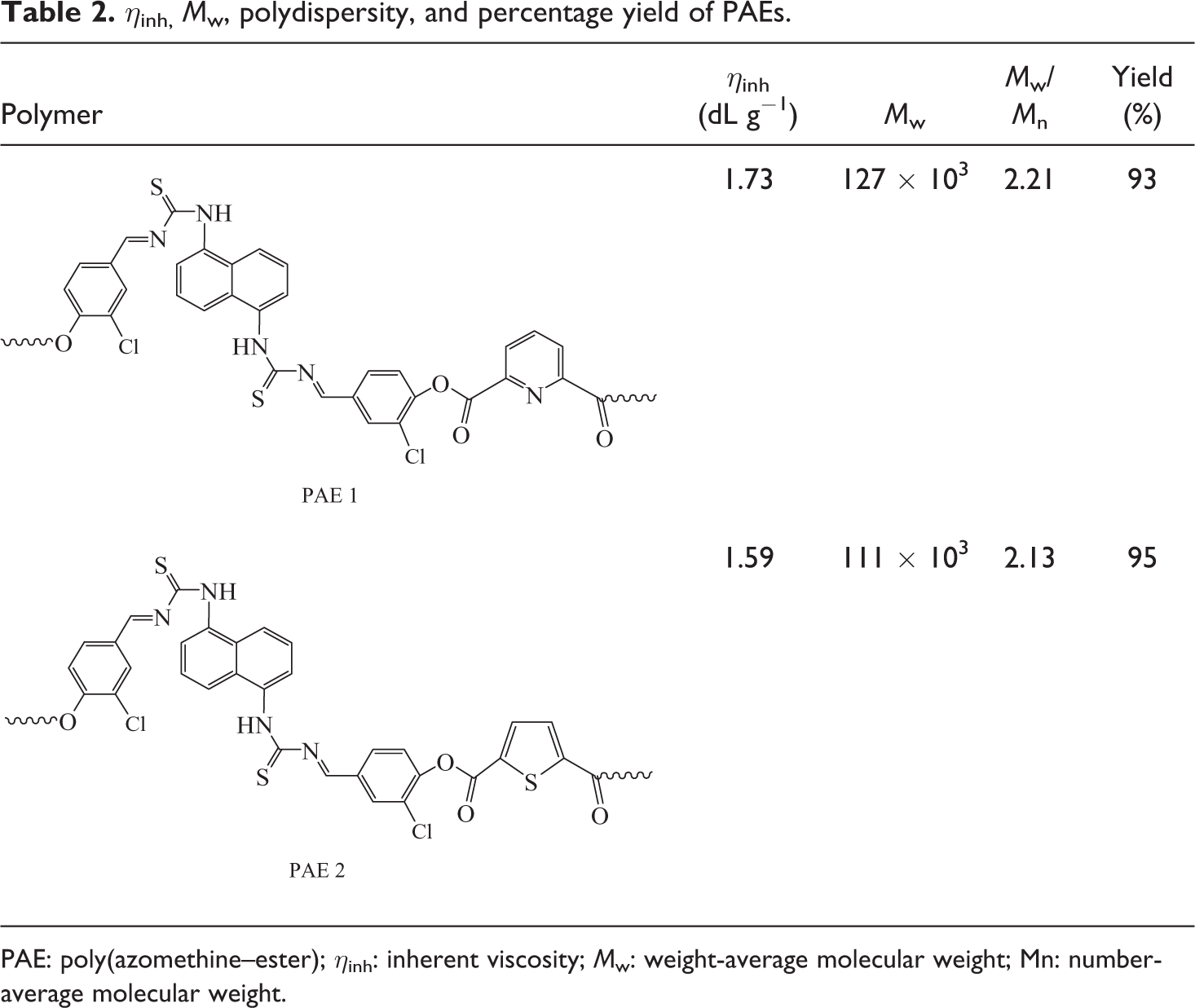
PAE: poly(azomethine–ester); η inh: inherent viscosity; M w: weight-average molecular weight; Mn: number-average molecular weight.
Thermal analysis
Thermal stability of the PAEs was studied using TGA and DSC. TGA of PAEs was estimated under N2 atmosphere at a heating rate of 10°C min−1. The weight loss data of PAE 1, PAE 2, and the blends are presented in Table 3 and Figure 8. The thermograms of naphthalene-based PAEs derived from PDC/TDC showed single-stage decomposition up to 650°C and the major weight loss occurred in the range 500–620°C. Effect of the pyridine or thiophene moieties introduced on the heat stability of new polymers was obvious from the results. Initial decomposition (T 0) of PAE 1 prepared from HCBTCANT and pyridine-based diacid chloride was 518°C, 10% gravimetric loss (T 10) of 527°C, and the decomposition extends up to 597°C. Whereas, the thiophene-based PAE 2 revealed slightly lower stability relative to PAE 1, that is, T 0 of 510°C and T 10 up to 521°C. This small difference in the weight loss of PAE 1 and 2 may be attributed to the difference in the backbone tructure. Thiophene, being a five-membered ring, is thermogravimetrically less stable than six-membered aromatic ring and may dissociate easily. Novel polyesters exhibited char yield around 53–55% at 600°C signifying good thermal stability. Moreover, the data suggested the higher stability of PAE 1 and 2 relative to the blend system prepared. Consequently, TGA traces indicated the lower values of thermal properties (T 0; 475–501°C) and char yield (37–45%) for the blends. With the addition of PAN/DBSA into PAE matrix, thermal stability is further reduced. The thiophene-based PAE 2/PAN/DBSA blend depicted lower values in thermal properties as compared to pyridine-based PAE 1/PAN/DBSA system. The structural effects of PAEs and blend systems on the heat constancy were also studied using glass transition temperature (T g) (Figure 9). PAE 1 and 2 showed higher T g of 291 and 287°C, respectively. However, no softening or melting transition was detected in the thermograms. Generally, the incorporation of rigid unit along a polymer backbone inhibits the free rotation of the macromolecular chains and leads to enhanced T g. The presence of stable heteroaromatic structure (pyridine in PAE 1), thus, enhanced the polymer chain rigidity and restricted the segmental mobility. The chain rigidity was also attributed to the intermolecular hydrogen bonding of thio carbonyl or ester carbonyl with N–H, thus enhancing the glass transition. However, the T g values for new blends were found to be slightly reduced on account of enhanced molecular mobility when compared with pure PAE 1 and 2. Therefore, new PAEs bearing azomethine–thiourea–naphthalene–pyridine/thiophene design revealed much higher thermal stability relative to several reported polyesters with these linkages.24 –26
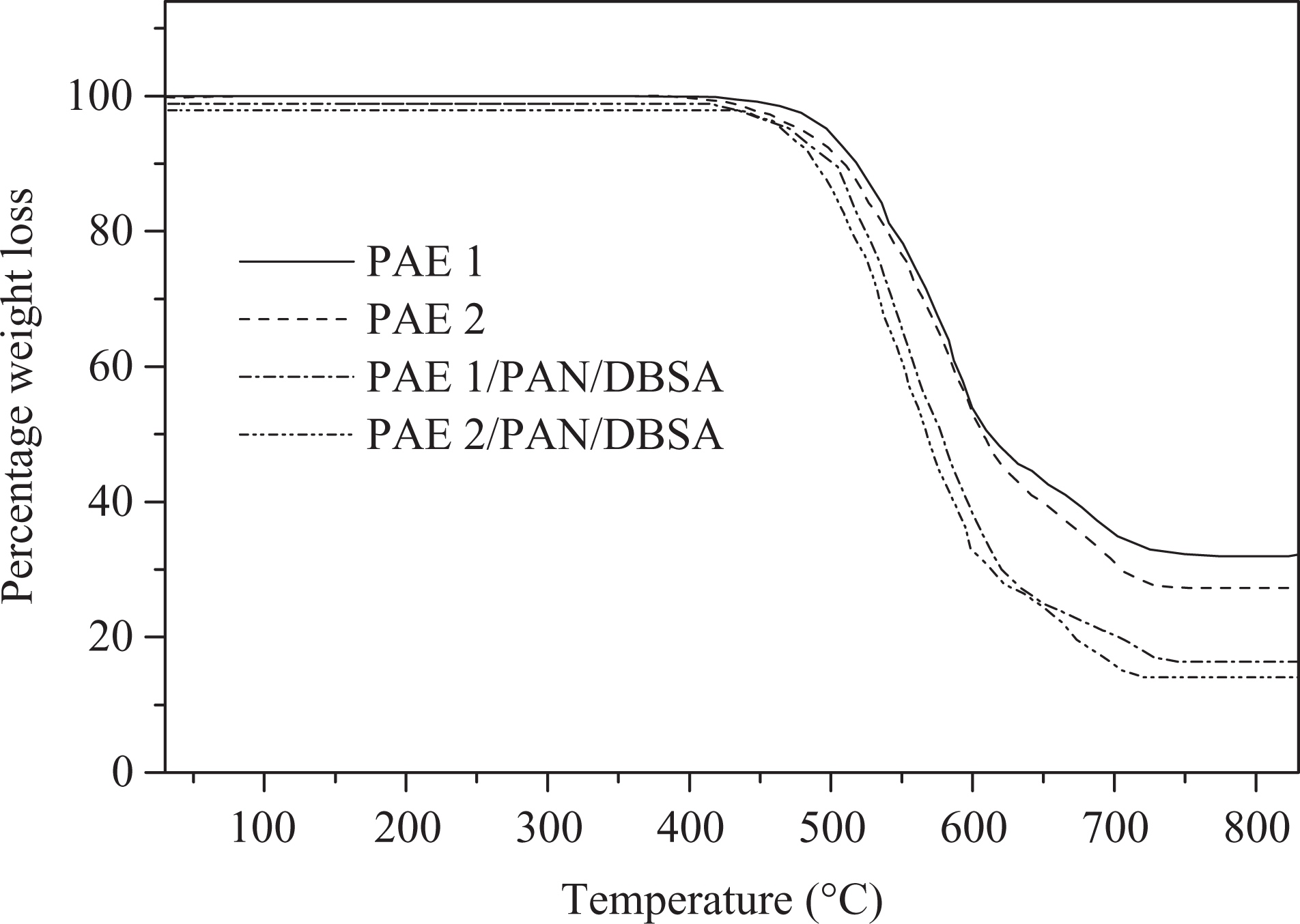
TGA curves of PAEs and blends at a heating rate of 10°C min−1 under nitrogen atmosphere. TGA: thermogravimetric analysis; PAE: poly(azomethine–ester).
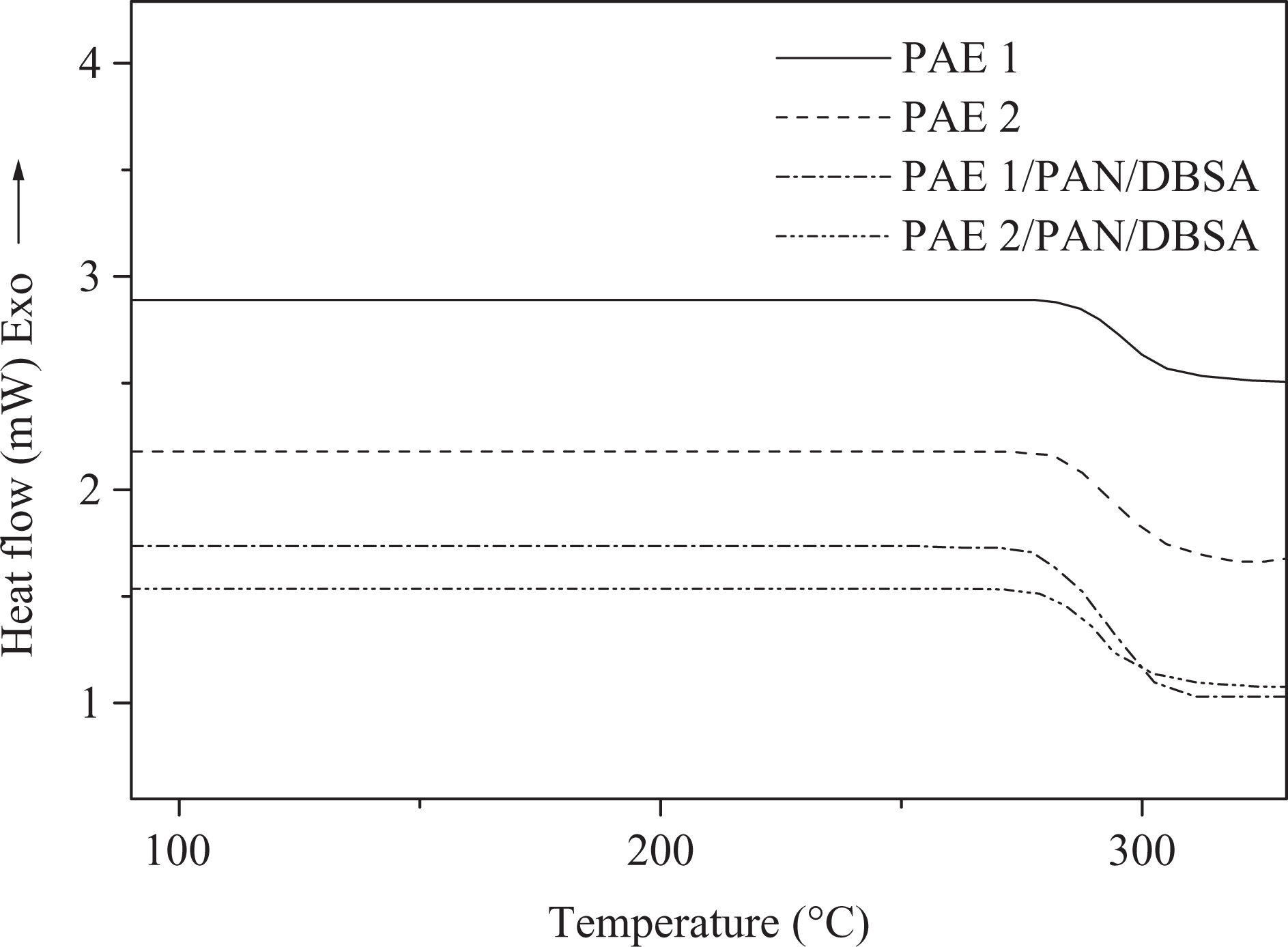
DSC thermograms of PAEs and blends at heating rate of 10°C min−1 under nitrogen atmosphere. DSC: differential scanning calorimetry; PAE: poly(azomethine–ester).
Thermal analyses data of PAEs and PAE/PAN/DBSA blends.
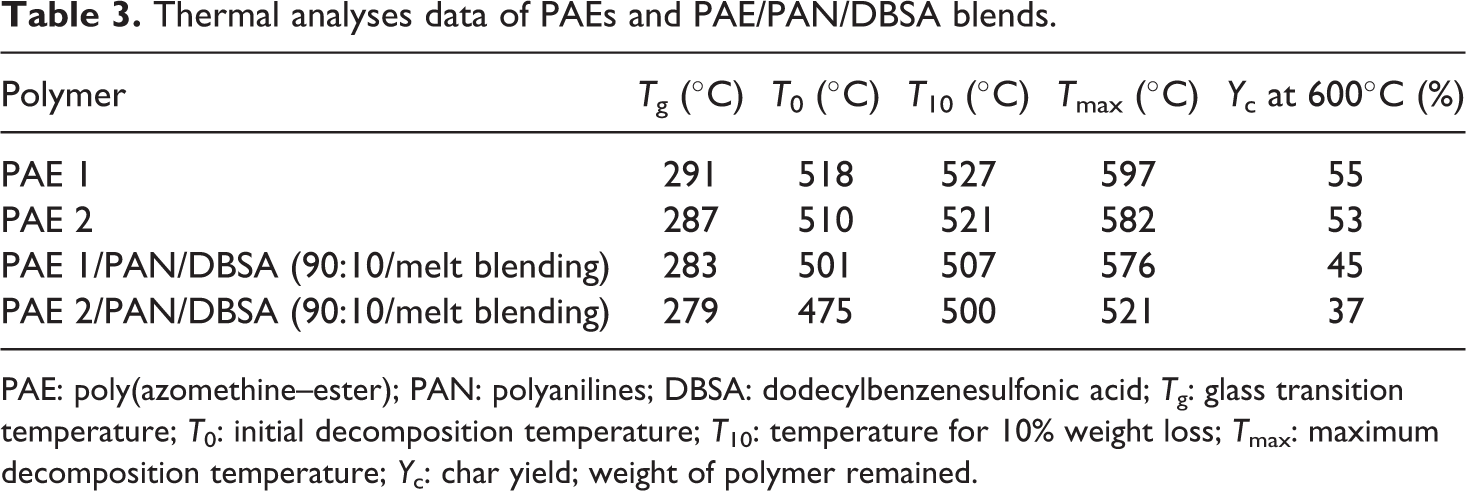
PAE: poly(azomethine–ester); PAN: polyanilines; DBSA: dodecylbenzenesulfonic acid; T g: glass transition temperature; T 0: initial decomposition temperature; T 10: temperature for 10% weight loss; T max: maximum decomposition temperature; Y c: char yield; weight of polymer remained.
XRD analysis
Wide angle x-ray diffraction pattern of PAE 1 is shown in Figure 10. The pyridine-basedPAE exhibited a broad peak indicating wholly amorphous domains. The heteroaromatic rigid-rod aromatic polymers were expected to exhibit crystalline pattern due to better packing of macromolecular backbone. Although, the broad peak with low intensity indicated that the presence of chloro or naphthalene moieties tend to destroy the possibility of reaching a crystalline order. The amorphous nature of PAE 1 was also in agreement with the DSC data as no endotherm was observed.
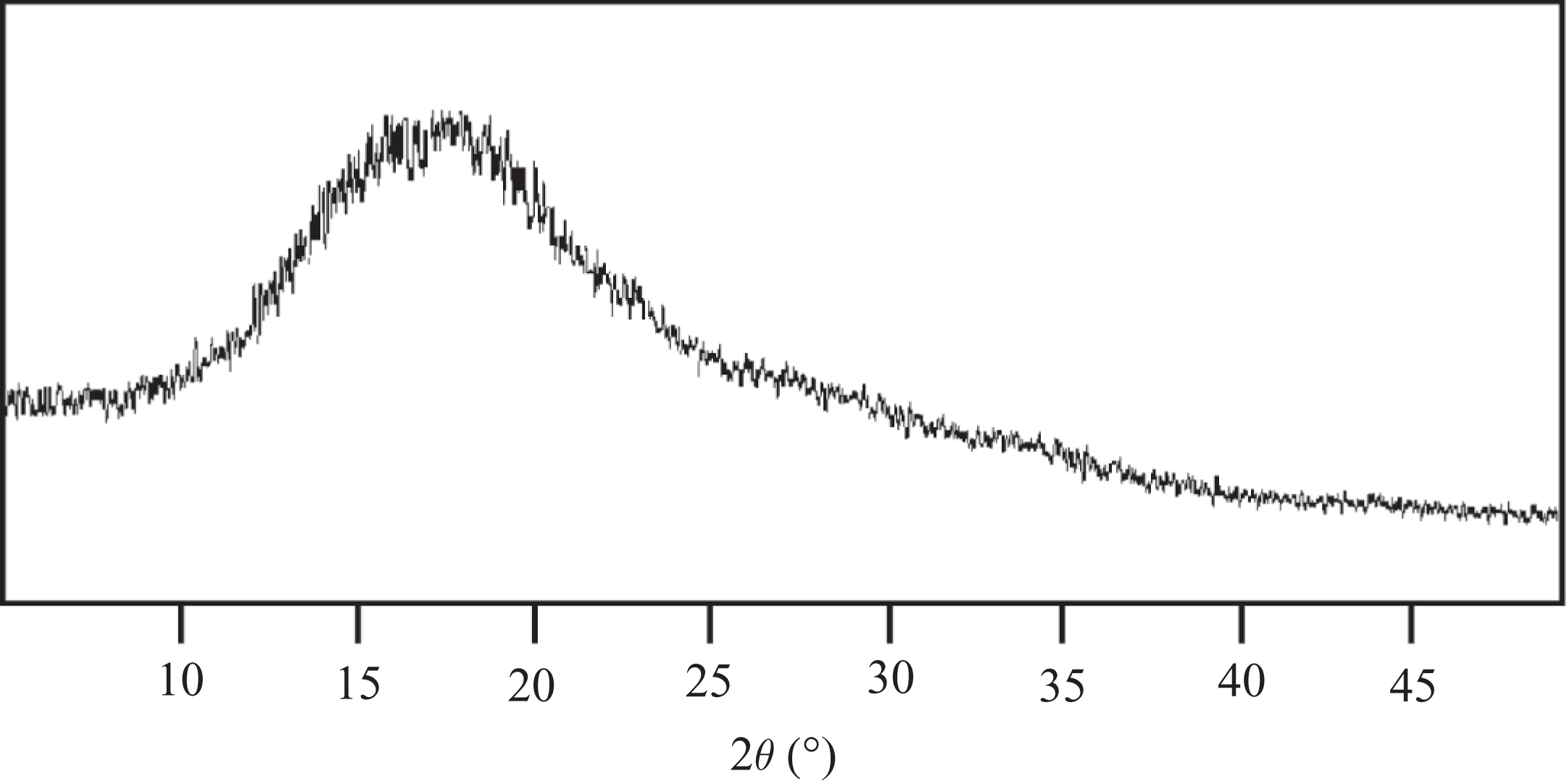
X-Ray diffraction pattern of PAE 1. PAE: poly(azomethine–ester).
Conductivity studies
Pure PAE 1 and 2 showed good electrical conductivity around 9.9 × 10−1 and 8.2 × 10−1 S cm−1, respectively (Table 4). Here, the pyridine moieties were found to significantly increase the conductivity of PAE 1 and its blends. The conductivity of PAE 1/PAN/DBSA with 10% of PAN/DBSA obtained via melt blending was found to be 1.9 S cm−1. The conductivity reached the highest value 2.8 S cm−1 at the 60% PAN/DBSA loading. When comparing the conductivity of blends obtained by two methods, PAEs/PAN/DBSA prepared by melt blending was sufficiently conducting. This may be attributed to better interaction between the blend components (Figure 7) instigating improved combine effect of azomethine/aniline/pyridine or thiophene moieties. The higher conductivity of PAEs/PAN/DBSA obtained by melt blending suggested the success of the method employed. Conductivity of new PAE/PAN/DBSA blends (by solution or melt method) was found to be superior when compared with that of reported PAEs/PAN blends and PAEs doped with silver. 27
Conductivity measurement of PAEs and PAE/PAN/DBSA blends.
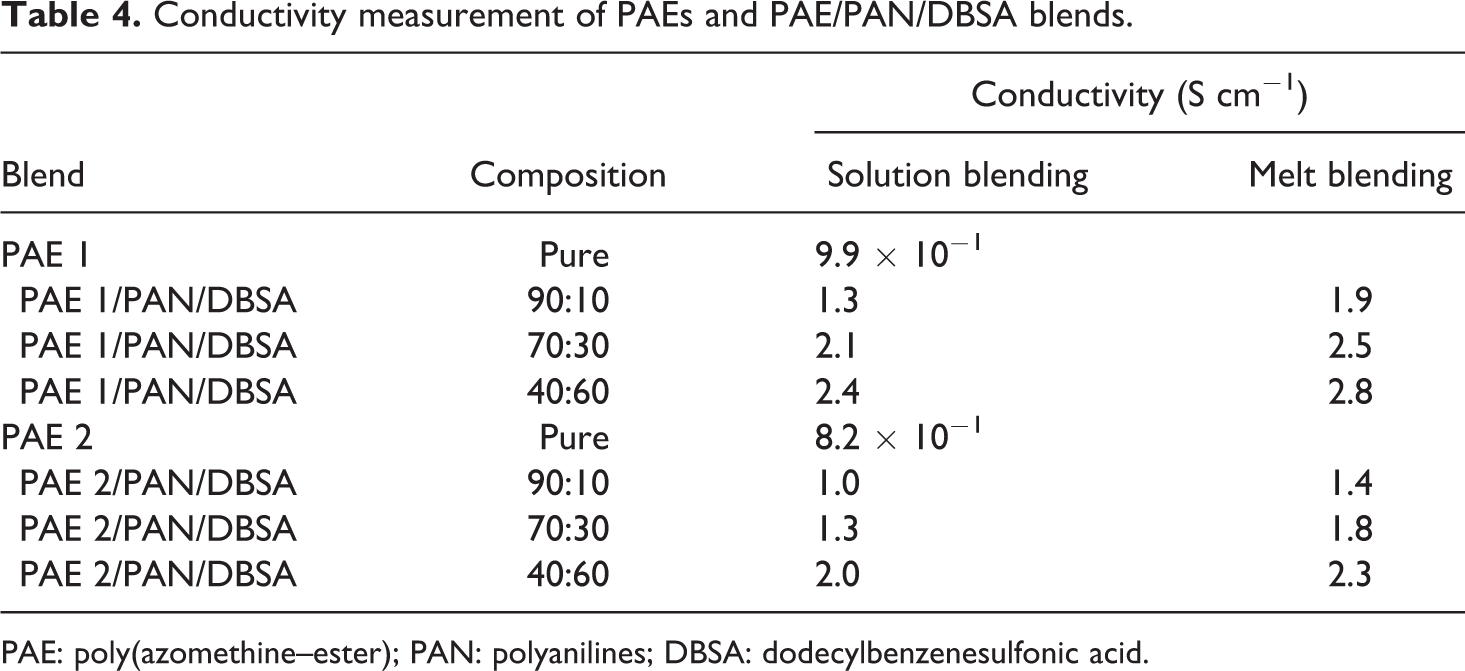
PAE: poly(azomethine–ester); PAN: polyanilines; DBSA: dodecylbenzenesulfonic acid.
Tensile strength of PAE/PAN/DBSA blends
Mechanical behavior of a blend is usually determined by the contribution of each component, the blend morphology and the interfacial cohesion. Table 5 lists the tensile data of new blends with 10% PAN/DBSA. Maximum stress of PAE 1/PAN/DBSA blend was 48.17 MPa, while PAE 2/PAN/DBSA had bit lower value 45.51 MPa (Figure 11). Higher value of the elongation at break was also observed for PAE 1-based blend (1.14) relative to that of PAE 2 (1.10). This can be explained on the basis of increased interfacial interactions between the two components in pyridine-based PAEs/PAN blend. At this juncture, the melt blending was again found to be an efficient method to improve the tensile properties of blends. The lower mechanical strength of solution blended system (maximum stress 25.80–27.16 MPa) suggested that the melt blending technique was quite successful to obtain high-strength materials.
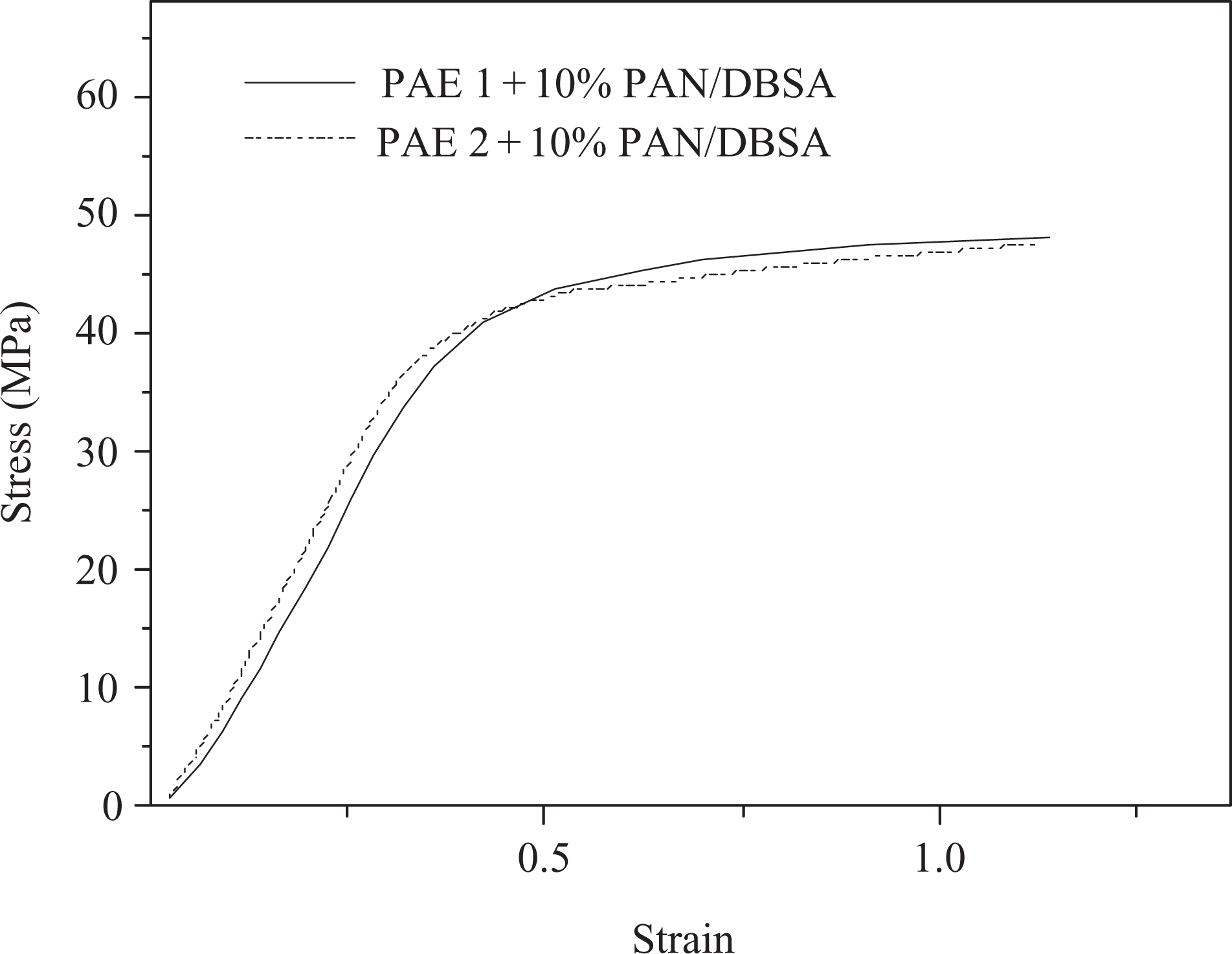
Stress–strain curves of PAEs/PAN/DBSA blends. PAE: poly(azomethine–ester); PAN: polyanilines; DBSA: dodecylbenzenesulfonic acid.
Mechanical properties of PAE/PAN/DBSA blends.
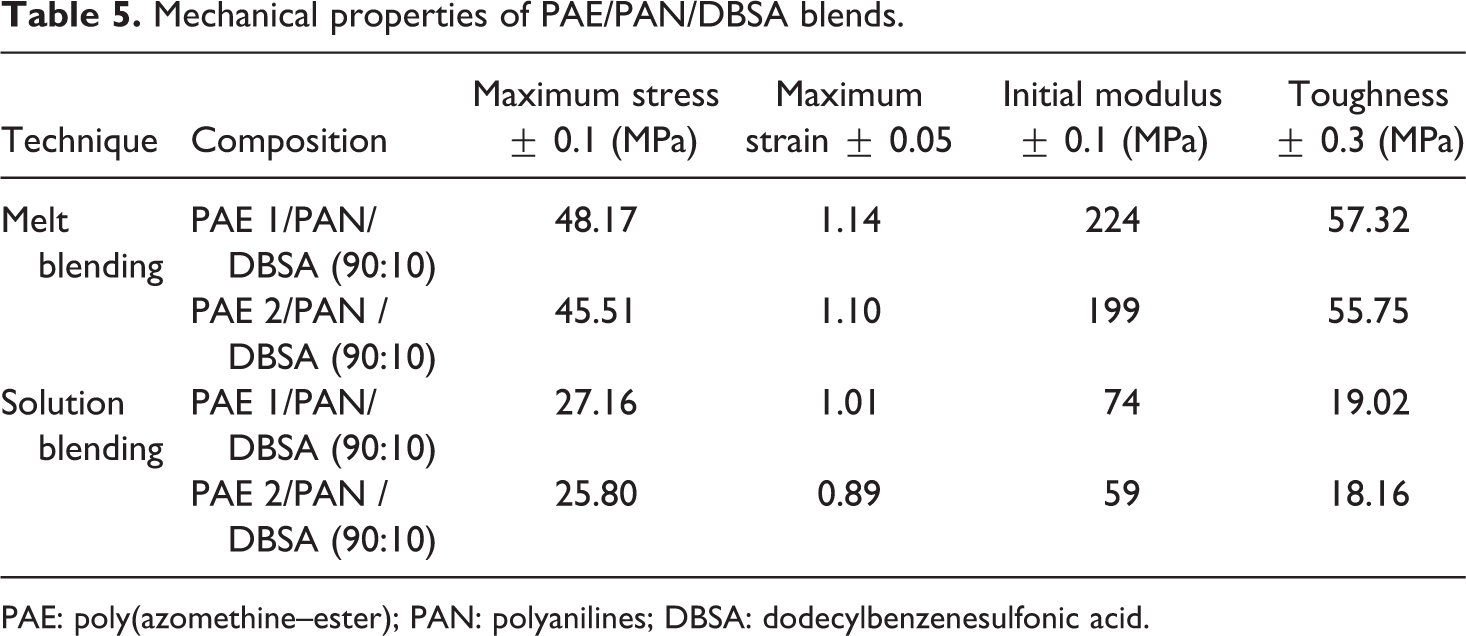
PAE: poly(azomethine–ester); PAN: polyanilines; DBSA: dodecylbenzenesulfonic acid.
Morphological features
The effect of the PAN/DBSA addition on the morphology of PAE 1/PAN/DBSA and PAE 2/PAN/DBSA blends was investigated using FESEM. Morphological comparison demonstrated that melt blending was a fine method to synthesize nanoblends with high electrical conductivity. Figure 12 (a) to (d) presents the morphology of 90/10 wt% of PAE and PAN/DBSA blends. The microstructures of solution blended PAE 1/PAN/DBSA (Figure 12(a)) revealed some aggregate formation at 1 µm, but no significant phase separation was observed. The conducting pathways were also not evident in the solution processed blends (1.3 S cm−1). With the 10% addition of conducting polymer in PAE 2, the aggregates seemed to be coalesced, as shown in Figure 12(c), having relatively low conductivity 1.0 S cm−1. The surface morphology of PAE 1/PAN/DBSA blend obtained by melt blending was significantly changed due to the homogeneous mixing of the two polymers (Figure 12(b)). Nanoblend morphology was, thus, experiential owing to the hydrogen bonding between the two components, leading to better compatibility between PAE and PAN chains. PAE 1/PAN/DBSA (90:10) blend presented a good homogeneous morphology formed by conducting pathways within dispersed PAN particles (1.9 S cm−1). Figure 12(d) also showed the fine dispersion of PAN in PAE 2 matrix presenting nanoscale compatibility in the material prepared by melt blending, thus, improved the conductivity up to 1.4 S cm−1. This type of morphology well explained the high conductivity achieved by PAE/PAN/DBSA blends.

FESEM images of (a) PAE 1/PAN/DBSA (90:10) by solution mixing; (b) PAE 1/PAN/DBSA (90:10) by melt blending; (c) PAE 2/PAN/DBSA (90:10) by solution mixing; (d) PAE 2/PAN/DBSA (90:10) by melt blending. FESEM: field emission scanning electron microscope; PAE: poly(azomethine–ester); PAN: polyanilines; DBSA: dodecylbenzenesulfonic acid.
Conclusions
The research toil confronts the fabrication of novel PAEs using a dihydroxy monomer, HCBTCANT, and various diacid chlorides. Influence of azomethine, pyridine, thiophene, and chloro moieties, introduced in the main chain, on the properties of newly synthesized polyesters were investigated. The foremost objective was to enhance the thermal properties, T g, molar mass, and conductivity while maintaining the processability of polymeric material. Later, thermally and mechanically stable PAE/PAN/DBSA blends with ample electrical conductivity were obtained employing solution and melt blending techniques. The heteroaromatic polymer obtained by pyridine-based diacid chloride exhibited higher thermal properties and glass transition relative to other PAEs and the nanoblend systems. Significantly different morphology and conductivity were detected with the two methods. Nanoblend morphology with conducting pathways was examined in the miscible melt-blended systems. The type of technique employed and the extent of dispersion of conducting polymer inside the polyester matrix strongly affected the electrical properties of the final system. The results demonstrated that conventional melt blending procedure was particularly beneficial for condensation-type polymers. In this study, we have stated few preliminary results on electrical, thermal, and mechanical properties of this copolymer and its blends, which demonstrated promise for their utilization in optoelectronic devices. Future studies may entail attractive consequences regarding their application in single layer organic devices and more complex organic light emitting diodes. 26
Footnotes
Funding
For financial support in the research work, Ayesha Kausar expresses her thanks to the Higher Education Commission of Pakistan.
