Abstract
The electrical and thermal properties of two series of composites, single-filler aluminum (Al) and silver (Ag) and hybrid-filler Al/Ag-filled high-density polyethylene (HDPE) composites, are reported. The composites were prepared by mixing using an internal mixer followed by compression molding. Studies on the electrical properties of single-filler composites showed an insulator-to-conductor transition in HDPE/Ag and HDPE/Al between 45–55 wt% and 55–65 wt% filler loadings, respectively. This is due to the formation of conductive paths that spanned throughout the polymer matrix, as revealed by morphology study. Thermogravimetric analysis results indicated that the degradation temperature of the composites decreased with the incorporation of 15 wt% Al- and Ag-fillers but increased slightly after the addition of 55 wt% of the filler. Investigation on the effect of hybrid composites showed that high-electrical conductivity was achieved, exhibiting percolation phenomenon in HDPE/80Ag20Al and HDPE/70Ag30Al composites where in these composites, 20% and 30% from 55 wt% of Ag were replaced with Al fillers, respectively. Furthermore, thermal stability significantly increased when hybridization was conducted to form the composite.
INTRODUCTION
In recent years, the development of electronic products has undergone considerable growth, and because of this, there has been an increasing demand to satisfy more stringent requirements for some electronic components. For example, a more electrically and thermally conductive material is required for typical electronic assembly parts, lower conductivity values to prevent electrostatic charge in certain applications, and a defined electromagnetic shielding and conductivity range for electronic components, such as enclosures, sensors, and control units [1]. This growing demand has generated significant research interest in electrically conductive polymer composites that exhibit the properties of both metals and polymers, such as ease of processability, low density, and outstanding electrical and thermal characteristics [1–3].
The easiest method of producing conductive polymer composites is to fill in an insulating polymer with highly conductive particles or fillers. Aluminum (Al), silver (Ag), copper, nickel, and graphite are widely used as conductive fillers in polymer matrices to improve the electrical conductivity of polymers [2,4,5]. Among the various conducting particles, Ag is the most commonly used because it exhibits the largest electrical conductivity, high-thermal conductivity, and chemical durability. It also possesses the lowest contact resistance [6–9]. However, Ag is relatively expensive. Recently, a method for incorporating two types of particulate fillers simultaneously into a polymer matrix has been put forward. This method not only offers economic advantages by diluting a more expensive reinforcement or filler with cheaper materials, but it can also be used to achieve synergistic effects and improvements in the mechanical and functional properties of the composite material [10,11]. In this study, Al and Ag are incorporated into high-density polyethylene (HDPE), instead of only pure Ag, as fillers to reduce cost. Al is also very conductive and has a relatively lower cost than Ag.
This study investigates the electrical and thermal properties of conductive HDPE composites filled with two different types of metal fillers: Al and Ag. The effect of filler size and shape on the composite properties is investigated given that both Al and Ag come in different sizes, 83 and 9 µm, and shapes, irregular and flaky. Moreover, the effectiveness of a hybrid sample containing both conductive fillers in terms of its electrical and thermal properties is reported in this study.
EXPERIMENTAL
Materials
Composites Preparation
Formulation for HDPE filled with single filler.
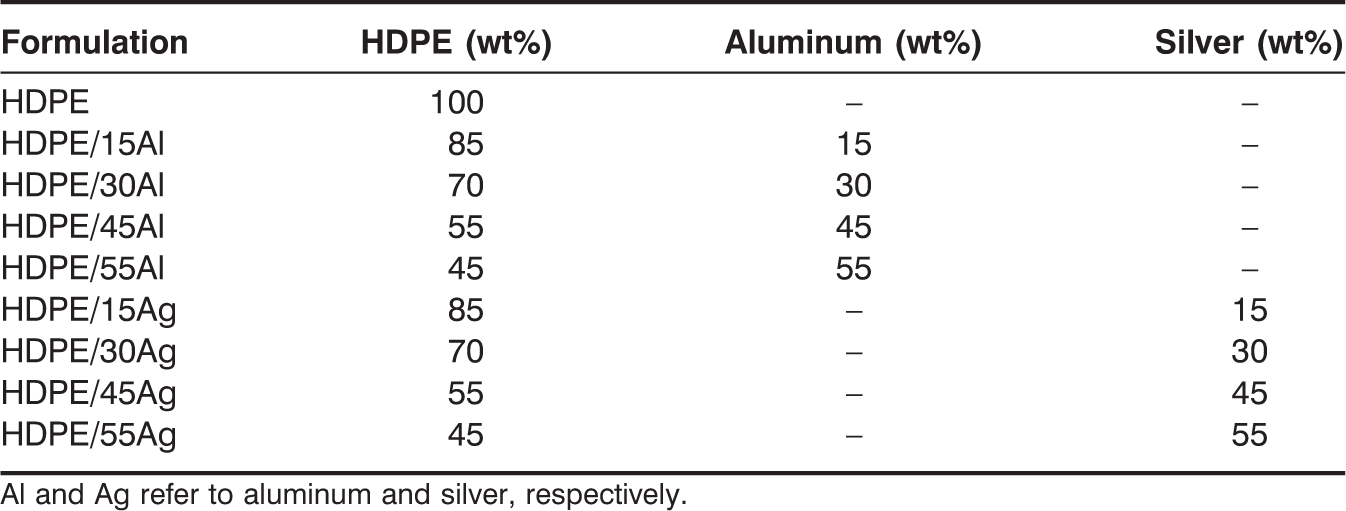
Al and Ag refer to aluminum and silver, respectively.
Formulation for HDPE filled with hybrid filler.
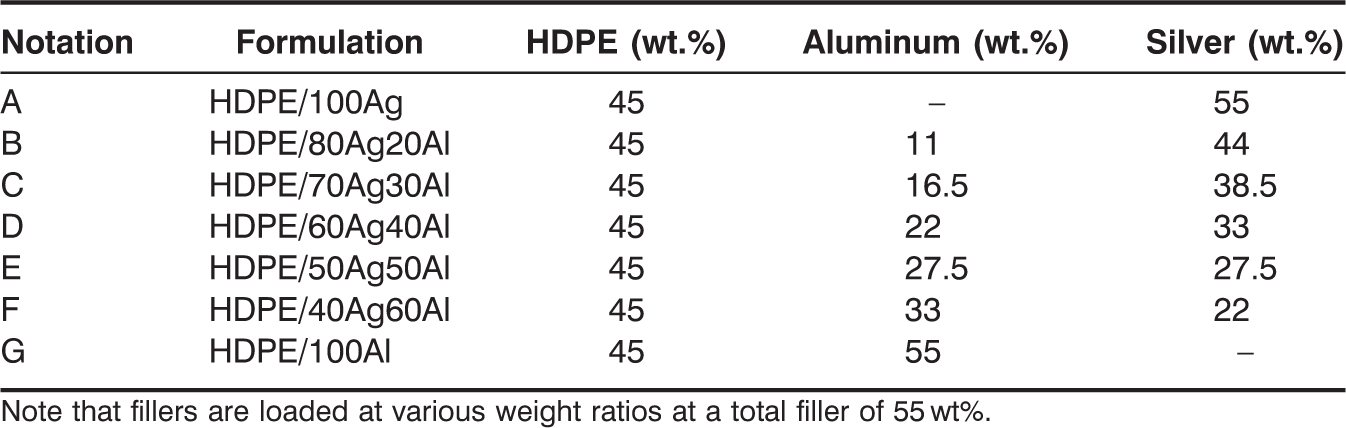
Note that fillers are loaded at various weight ratios at a total filler of 55 wt%.
Characterization and Measurement
CONDUCTIVITY TEST
The electrical conductivity of the composites was determined through the electrical volume resistivity test using an LCR meter. Samples were cut into 3 × 3 cm2 squares and polished using sand paper prior to testing to ensure good electrode-sample interface contact. Ag paint was applied to the opposite faces of the samples to fill the microvoids and provide an intimate electrical interface between the sample and the electrode. The testing instrument was calibrated before performing the measurements. A constant voltage of 1 V was supplied to the samples attached between two copper electrodes; the resistance of the samples was measured after 1 min. Three samples for every percent of filler content were tested, and the average values of the volume resistivity were taken into consideration. Volume resistivity was calculated using Equation (1):
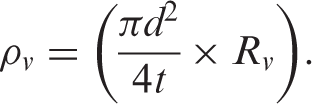
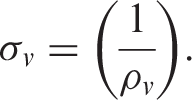
MORPHOLOGY
The fracture surface morphology of selected HDPE composites was analyzed using a scanning electron microscope (SEM; model ZEISS SUPRA 35 VP). The fracture surface of the sample was coated with a gold-palladium layer using a Sputter Coater Polaron SC 515 to avoid electrostatic charging during observation. The dispersion of the filler throughout the composite and the filler agglomeration were investigated using SEM.
THERMOGRAVIMETRIC ANALYSIS
Thermogravimetric analysis (TGA) was carried out using a Perkin-Elmer Pyris 6 TGA analyzer to confirm the filler content and to determine the thermal stability of the composites. Samples of 10 mg were prepared for the test. The samples were heated from 30°C to 600°C at 20°C/min in a nitrogen atmosphere.
RESULTS AND DISCUSSION
Characterization of Single Metal Filler Filled HDPE Composites
Figure 1 shows a graph illustrating the effect of various Al and Ag loadings on the volume electrical conductivity of HDPE/Al and HDPE/Ag composites. As expected, the volume conductivity for both composites increases with the filler content. This is in agreement with previous studies on the effect of particle loading on the electrical conductivity of metal-filled polymer composites [15,16]. The studies showed that the composites were in an insulating region until a critical or threshold concentration of the conductive filler was achieved. In Figure 1, at lower filler loadings (below the percolation threshold), it is shown that the filler particles act like conductive islands in a sea of electrically insulating polymer, and no significant increase in conductivity is observed. As more particles are introduced, the conductive particles become more crowded and are more likely to come in contact with each other. At the percolation threshold, the majority of the filler particles are in contact with at least two of their nearest neighbors, thereby establishing a continuous conductive chain or network throughout the HDPE plastic. An electrical charge can then flow without encountering the high resistance of the polymer. As a result, the conductivity values of the conductive Al- and Ag-filled HDPE composites increase dramatically.
Electrical conductivity of HDPE/Al and HDPE/Ag composites as a function of filler loading.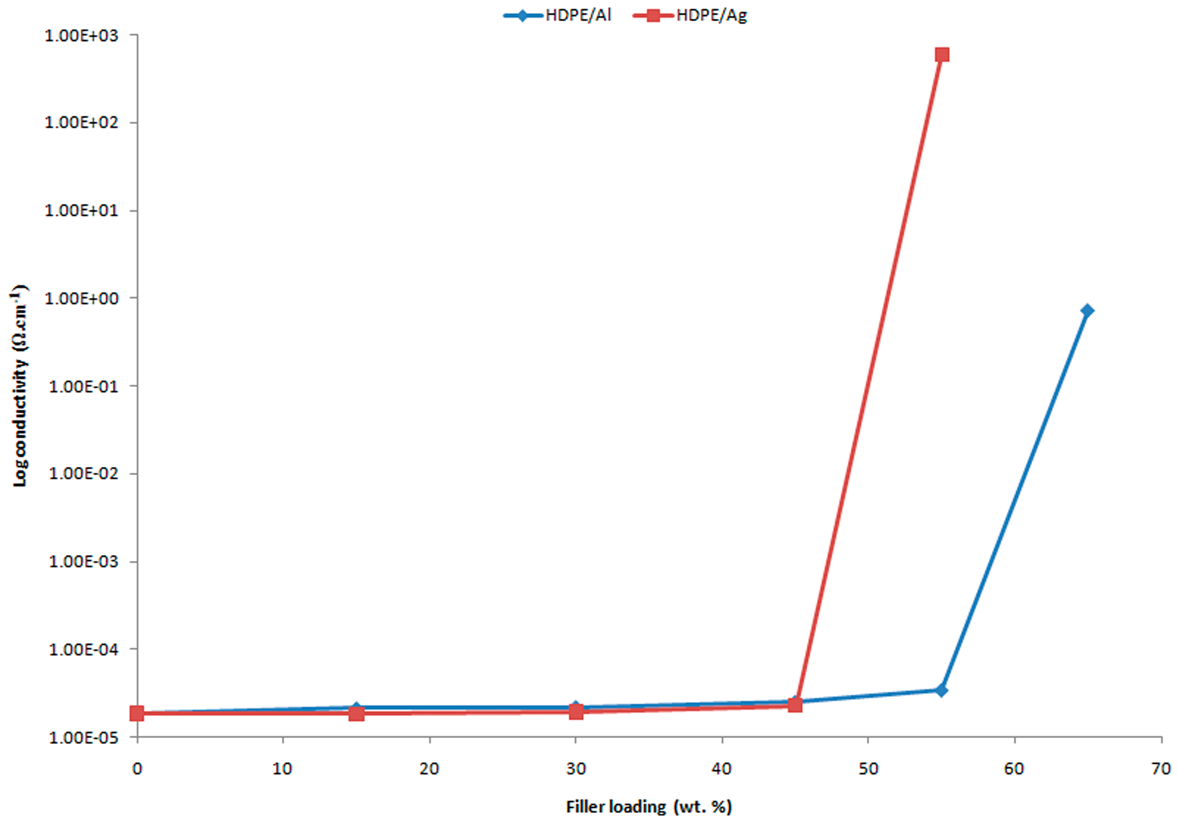
By referring to Figure 1, the percolation threshold is observed to occur between 45 and 55 wt% for the Ag filler loading and between 55 and 65 wt% for the Al filler loading, where the conductivity increases sharply by seven orders and more than four orders of magnitude, respectively. The lower percolation concentration achieved by HDPE/Ag compared with HDPE/Al is due to the flaky shape of the Ag particles, which provides a more stable conducting composite than nondimensional particles (irregular) because of their large surface area per unit volume and provides better metal-to-metal contact. According to Xue [16], a larger aspect ratio can help reduce the minimum metal content required to reach a certain conductivity value. However, with respect to the morphology of the composite samples (Figure 2), no obvious metal-to-metal contact is observed. The Ag flakes are not well dispersed in the HDPE matrix. Instead, they form large particle agglomerates in the system. From the SEM micrograph in Figure 2(a), which represents the HDPE composite filled with 45 wt% of Ag, the agglomerates are observed to be distant from each other. Through this, the possibility of electron transfer is avoided. Thus, at 45 wt% of Ag loading, the composite is still in the insulating region. After the filler is added up to 55 wt% (Figure 2(b)), the particle agglomerates seem to form close to each other, allowing electron transfer to occur. This explains the high-conductivity value achieved by HDPE/55Ag. The same phenomenon can also be observed in Al-filled HDPE composites, as shown in Figure 2(c) and (d).
Fracture surface of HDPE filled with (a) 45 wt% Ag, (b) 55 wt% Ag, (c) 55 wt% Al, (d) 65 wt% Al (100 × magnification).
On the other hand, Ag exhibits a higher electrical conductivity than Al (Table 1). The conductivity of Ag and Al is 0.59 × 106 and 4 × 105 Ω/cm, respectively. This may be a reason for the lower percolation concentration as well as the higher electrical conductivity achieved by HDPE/Ag compared with HDPE/Al. However, other factors, such as the distribution of filler, void content, and oxide layer, may also influence the electrical conductivity of the composites.
Figure 3 depicts the TGA results showing the degradation temperature of the composite samples together with their percent of weight change as a function of filler content. The unfilled HDPE shows the highest weight loss with no residue. Adding the fillers, the thermal degradation of the composites shifts toward lower temperatures, which is more pronounced for both Al-and Ag-filled HDPE at a low filler content (15 wt%). A probable reason for the observed behavior may be due to the high-heat capacity and thermal conductivity of Al and Ag, enabling these particles to reach a higher temperature more quickly than the surrounding matrix [2]. The heat capacities of Al and Ag are 24.200 and 25.350 J/mol/K, respectively, and their thermal conductivities are 204 and 418 W/m/K, respectively. This causes the HDPE chains to start degrading at a lower temperature. When the Al and Ag content is increased up to 55 wt%, the degradation temperature at 5% weight loss is increased to 443°C for HDPE/Al and 445°C for HDPE/Ag, which is still lower than that for the unfilled HDPE.
TGA curves of unfilled HDPE and HDPE filled with 15 and 55 wt% Al and Ag.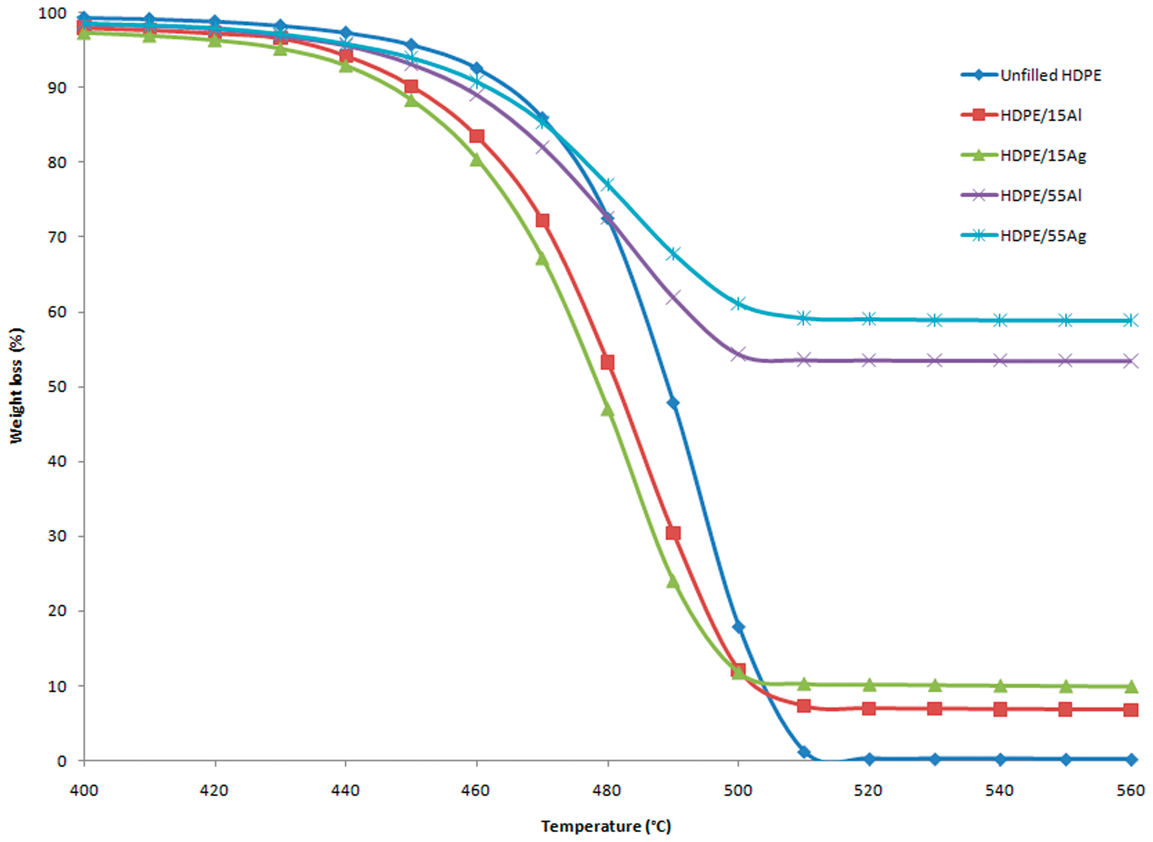
Characterization of Hybrid Metal-filled HDPE Composites
The goal of fabricating hybrid Al- and Ag-filled HDPE composites is to replace Ag with Al. Ag is more expensive than Al. Based on the electrical conductivity values given in the previous section, the percolation threshold of HDPE/Al and HDPE/Ag was observed at 65 wt% and 55 wt% of the filler, respectively. The total filler content between Al and Ag for the hybridization is kept constant at 55°wt%. This is because at this filler loading, the electrical conductivity of HDPE/Ag composites shows an optimum value, whereas for the HDPE/Al composites, the conductivity is still in an insulating region. With hybridization, the conductivity of HDPE/Al may be improved, and the amount of Ag content in HDPE/Ag may be reduced without sacrificing the conductivity. In the following sections, HDPE/100Al and HDPE/100Ag are referred to as HDPE/55Al and HDPE/55Ag, respectively, as investigated in the study on single fillers.
The electrical conductivity values of hybrid composites are presented in Figure 4. Notation of samples (A, B, C, etc.) is based on the formulation in Table 3. From Figure 4, it can be seen that hybridization produces a synergistic effect on the electrical conductivity of the composites. The percolation phenomenon is observed in the hybrid system where based on the electrical conductivity value before percolation threshold (Figure 1), the value increases about five and seven orders of magnitude for HDPE/80Ag20Al and HDPE/70Ag30Al, respectively. The reason for this is perhaps due to the creation of conductive paths, even at reduced Ag content, as a consequence of Al particles favoring the formation of conductive pathways in both systems as observed in Figure 5(a) and (b), respectively. The Al particles are more likely to act as anchors for individual Ag flakes, thereby facilitating electron transfer among the flakes. The positive hybrid effect shown by the positive deviation from the additivity rule (represented by the line) is observed in 70Ag30Al/HDPE composite system. However, the negative hybrid effect shown by HDPE/80Ag20Al is unexpected. This can be explained by the presence of voids that might govern the conductivity values of the composites. The introduction of voids in the composites structure can be caused from two reasons; one is the agglomeration of Ag and Al particles, which results in the existence of an unoccupied space between them that breaks the conductive chain. The other reason is poor filler–matrix adhesion. It is noted that in these composite systems, no bonding occurs at the interface between the filler particles and the matrix. Due to the weak adhesion at the filler–matrix interface, there might be the presence of voids or gap that result in conduction resistant due to the reduction of contact area between metal particles, hence decreasing the conductivity.
Electrical conductivity hybrid-filler filled HDPE composites. Fracture surface of HDPE filled with hybrid Al and Ag particles at various weight ratios: (a) 80Ag20Al, (b) 70Ag30Al, (c) 40Ag60Al at total filler content of 55 wt% observed under 100 × magnification (note that white particle refer to Ag and gray particles refer to Al).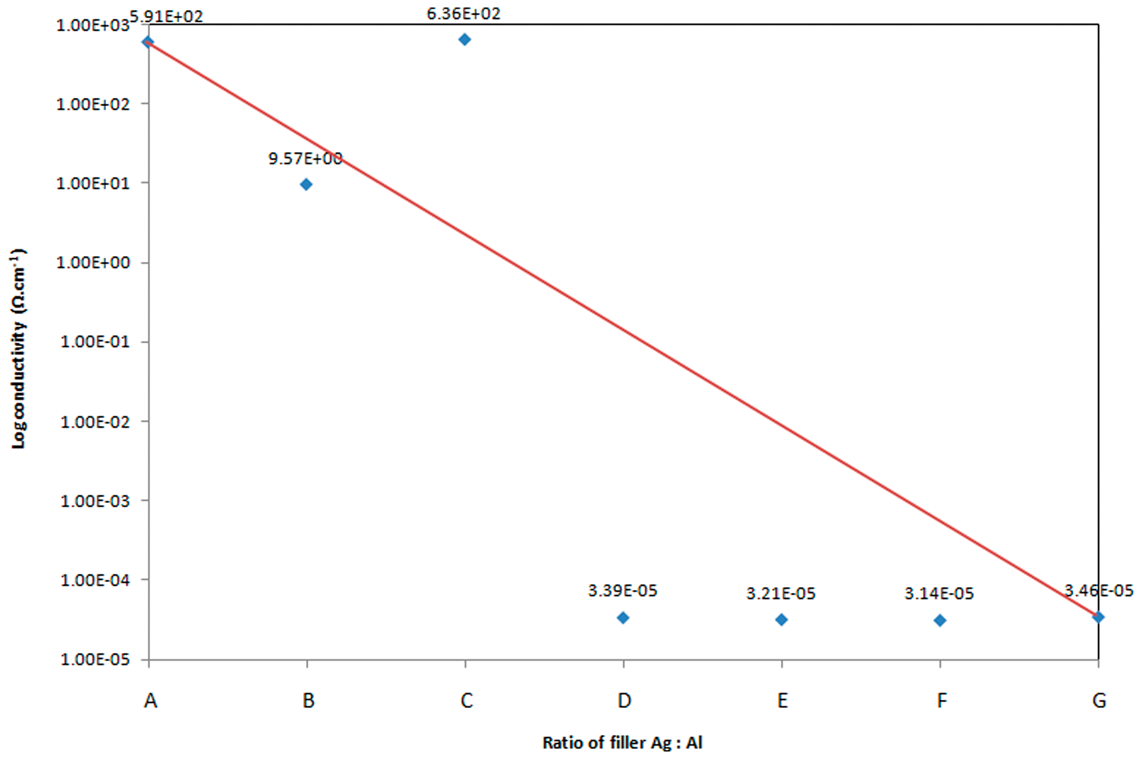

When the Ag content is further decreased, the conductivity drops suddenly, and no significant changes in the conductivity values are found with the replacement of Al, as shown by HDPE/60Ag40Al, HDPE/50Ag50Al, and HDPE/40Ag60Al in Figure 4. Although the conductive paths seem to be created in the morphology of HDPE/40Ag60Al (Figure 5(c)), the conductivity is still in the insulating region. This suggests that, with a high reduction in the Ag content, the composites become insulators. The high content of Al filler inside the HDPE/Ag/Al composite does not help in achieving high-electrical conductivity. Based on the results shown in Figure 1, 55 wt% of Al is still insufficient for an insulator-to-conductor transition to occur. The poor electrical properties obtained at high Al loadings might be due to the oxidation of Al particles. Al filler is known as a reactive metal in which the Al oxide layer can be formed on its surface when exposed to air. The insulating oxide layer shell around Al surface resists the electron transfer between metal particles so that decreasing the conductivity value of the composites [9,17].
Thermal properties of unfilled HDPE, single- and hybrid-fillers filled HDPE composites.
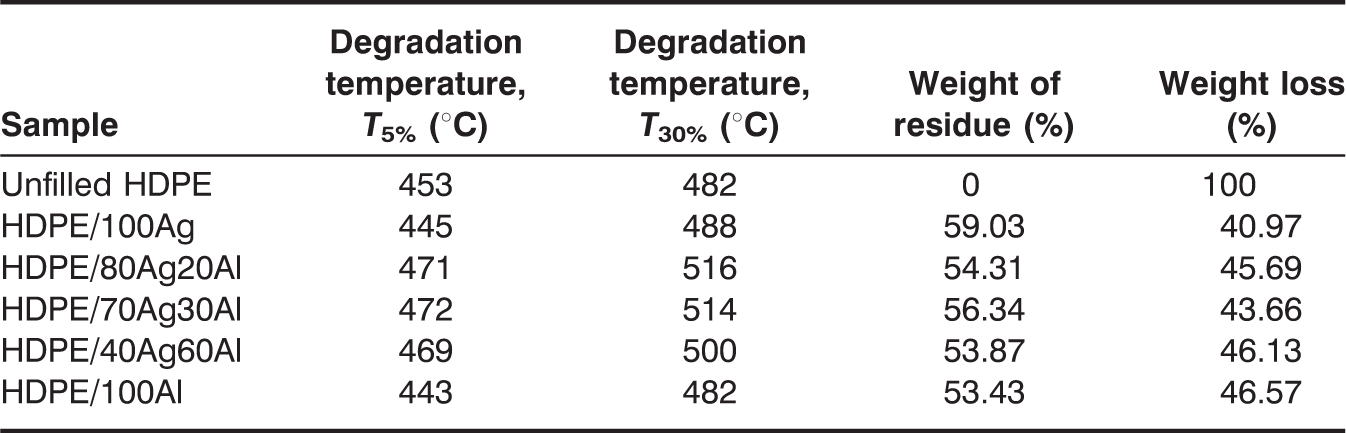
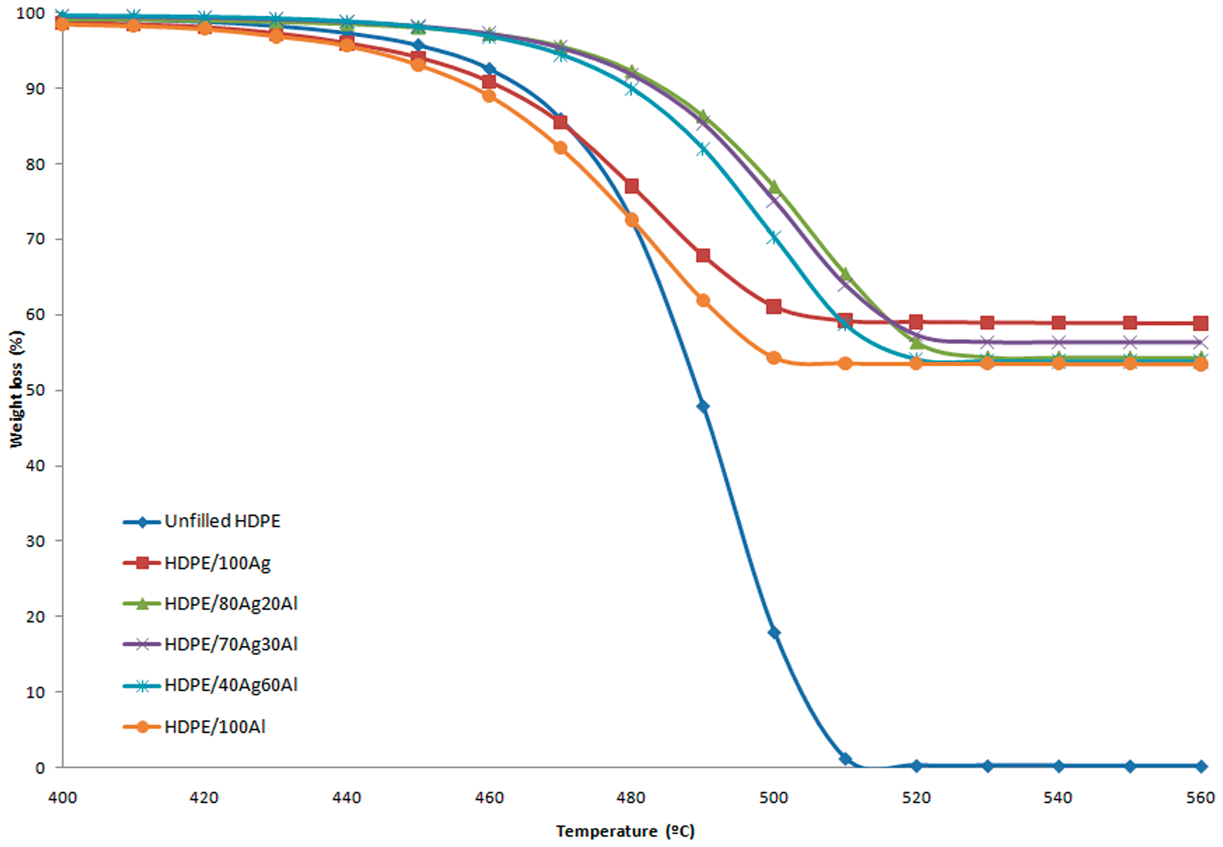
TGA curves of unfilled HDPE, single- and hybrid-fillers filled HDPE.
CONCLUSIONS
The electrical properties of HDPE composites depend greatly on the types and amount of fillers used. HDPE/Ag exhibits lower level of filler loading to achieve percolation threshold compared to HDPE/Al. This is due to the high aspect ratio of the flaky Ag particles, which promotes metal-to-metal contact at lower filler loading. The higher electrical conductivity value achieved by HDPE/Ag can be due to the high conductivity of pure Ag. The addition of 15 wt% of Ag and Al into HDPE decreases the degradation temperature of the HDPE composites, which may be due to the high heat capacity and thermal conductivity of Al and Ag that causes the HDPE chains to start degrading at a lower temperature. However, after the addition of 55 wt% of the filler, the thermal stability of the composites slightly increases. Furthermore, in the hybrid composite system, it is observed that the percolation threshold is achieved with the conductivity value increases about five and seven orders of magnitude for HDPE/80Ag20Al and HDPE/70Ag30Al, respectively. The presence of Al favors the formation of conductive pathways in the hybrid composite. However, with further increase in the Al content of the hybrid system, the conductivity values drop, and the composites become insulators. A study on the thermal properties of the hybrid composites found that the thermal stability of the composites increases significantly. This may be due to the close packing of the hybrid fillers that help in increasing the thermal degradation temperatures of the hybrids.
Footnotes
ACKNOWLEDGMENTS
The authors are very grateful to the Universiti Sains Malaysia and the Ministry of Science, Technology and Innovation (MOSTI) for awarding them with a Research University grant (814055) that made this study possible. The authors gratefully acknowledge the scientific contributions of Professor Lin Ye from the University of Sydney, Australia.

