Abstract
Hydrogel of single-walled carbon nanotubes and polyaniline has been used for thermopower engineering applications due to desirable thermal, electrical, and mechanical properties as well as tunable degradability. In this article, we fabricated nanoporous composite scaffolds from hydrogel of single-walled carbon nanotubes and polyaniline polymer using a standard in situ polymerization process. Our solution-based fabrication method prevented single-walled carbon nanotube aggregation which resulted in enhancing thermal, electrical, and mechanical properties with keeping optimum flexibility in the porous composite scaffold. We compared the mechanical, electrical, and thermal properties of nanoporous composites with different single-walled carbon nanotube loadings. The porous composite scaffold with a 25 wt% showed higher electrical conductivity, ultimate tensile strength, and tensile modulus. Lastly, our solution fabrication method prevents aggregation single-walled carbon nanotube and could help to build the thermoelectrical materials for flexible electronic applications.
Introduction
The rapidly increasing interests in flexible electronic devices and the wearable display industry have motivated the advancement of lightweight and inexpensive energy storage devices. 1 –5 The nanocomposites of nanoparticles with conductive polymer matrix represent a unique system in which properties of its components partially or completely mix and can emerge with new material behavior. The traditional nanocomposites are well-studied, the superposition of components of the composite play a very important role in holding the composites. The surface area comes to dominate as the filler shrinks in size and leads to the unique behavior of the porous nanocomposite scaffolds.
Recently, conjugated polymers have gained special attention due to their broad range of industrial applications, 6 –10 which are arising from their excellent optical, electrical, 11 mechanical, and thermal properties. 12 –16 Polyaniline (PANI) is the most attractive polymer among all the conjugated polymers owing to its controllable levels of doping, and it has been using for a broad range of industrial applications, such as sensors, anticorrosion coating, tissue engineering, and reduction of toxicity in water. 17 –23
Carbon nanotubes offer a larger aspect ratio, high surface area per mass, lower density (∼1.3 g ml−1) with exceptional mechanical, thermal, electrical, and optical properties, 24,25 which would make them an ideal additive to impart these multifunctional properties to polymers. The single-walled carbon nanotube (SWCNT) aerogels are the three-dimensional networks, consisting of interpenetrating pores, which are an active network for fabricating nanoporous polymer nanocomposite scaffolds. The SWCNTs with high stiffness along with a larger surface area per volume, larger aspect ratio, and homogeneous dispersion at high weight fraction have a stronger interfacial adhesion with polymers to promote multifunctional properties. 26 –28 The nanotubes aggregate easily when we mix them with polymers due to Vander Waal’s attraction between nanotubes and polymers. As a result, simple and direct dispersion of 1–25 wt% of nanotubes into polymers leads to significantly lower electrical and mechanical properties. 11,29 –37 To avoid bundling and slippage of nanotubes at its surface, there is a solution-based fabrication method, followed by in situ polymerization, which desirably improves the electrical and mechanical properties. 11,30,36 –46
In this work, we fabricated SWCNT/PANI nanoporous composite aerogels by using the method of a hydrogel of SWCNT and followed by in situ polymerization. Camphor sulfonic acid (CSA) is used to increase electron transport in these nanoporous composite scaffolds. We examined the electrical conductivity, thermal analysis, and mechanical properties of CSA-treated SWCNT/PANI nanoporous composite scaffolds. The electrical conductivity was increased from 7 S cm−1 to 60 S cm−1 at 25 wt% SWCNTs. The tensile modulus of porous nanocomposites (volume fraction: 50%) was increased with the addition of SWCNTs and reached 70 MPa at 25 wt% which indicates 200% improvement over the pristine polymer.
Methods and characterization
Materials
SWCNTs-CG-200 purchased from South West Nano Technologies, and sodium dodecylbenzene sulfonate (SDBS), aniline, CSA, and ammonium peroxydisulfate purchased from Sigma-Aldrich.
Preparation of SWCNT hydrogel
SWCNT aerogels were prepared according to the literature 25 and the SWCNT concentration with SDBS was 30 mg ml−1. Then the gel was transformed into different molds to remove SDBS completely, the hydrogel was transferred into 20 ml of nitric acid (1 M) aqueous solution at room temperature for 2 h. To remove acid completely, the hydrogel was rinsed with deionized (DI) water.
SWCNT/PANI aerogel composites
To synthesize SWCNT/PANI composite aerogels, the SWCNT hydrogel was soaked into the 25 ml CSA/An aqueous solution [(CSA) = (An) = 0.05 M] for 1 h. The infiltrated SWCNT hydrogel was carefully transferred into the 20 ml of CSA/APS solution [(CSA) = 0.05 M and (An)/(APS) = 2:1] at 3–4°C for 3 h. Once the polymerization process (green color) was completed, the SWCNT/PANI composite was washed by DI water several times, followed by drying at 60°C for 3 h to obtain the SWCNT/PANI nanoporous composites. The weight of SWCNT and PANI in the composite was determined as the difference of weight of SWCNT and the final weight of the composites.
Morphology
The SWCNTs within these scaffolds are individually dispersed and randomly oriented, as captured in high-resolution scanning and transmission electron microscopy images of their cross sections.
Mechanical characterization
The tensile stress (σ) was measured as a function of tensile strain (?) at the rate of 0.2 mm s−1 at room temperature with a50-N load cell using an Instron 5940 series tabletop testing system (TA Instruments).
Thermal analysis
The differential scanning calorimetry (DSC) measurements were carried out with a Q20 DSC (TA Instruments) at a heating rate of 3°C min−1. Pristine polymer and nanoporous composites with 12 wt% SWCNT were also tested over a temperature range of −20°C to 300°C. Thermogravimetric analysis (TGA) was carried out under atmospheric air over a temperature range of 25–800°C using a Q50 TGA (TA Instruments). The specimens were heated at a rate of 5°C min−1.
Raman spectroscopy
The Raman spectra were collected using an inverted Raman confocal microscope (in Via Raman microscope, Renishaw) with a 20 × (0.40 NA) objective (Leica Microsystems) and 785 nm (1.58 eV) laser line. For spectra collection, the laser power was set to 10 mW to avoid heat-induced damage to the samples. The laser spot size was 1–2 µm with an exposure time of 3 s. Raman spectrum intensity was then normalized by its G-band intensity.
Electrical conductivity measurements
For electrical conductivity measurements of the nanoporous composite, copper foil leads were connected or attached to the short ends of the rectangular nanocomposites with silver paste, and resistance was measured using a two-probe contact method.
Results and discussion
The fabrication of SWCNT/PANI nanoporous composite aerogel is shown in Figure 1(a) . The fabrication method included four steps, that is, (1.) the preparation of SWCNT gels followed by transferring gel into rectangular molds, (2.) aniline monomers infiltration, and (3.) in situ polymerization of PANI polymer within the SWCNT gel network followed by infiltration of CSA doping, followed by (4.) drying composite at 60°C. The morphology of SWCNT aerogel shows a highly nanoporous structure (Figure 1(b) and 1(c)). The SWCNT aerogel sample was in-filtered with PANI by using in situ polymerization (Figure 1(d)).

(a) The schematic of fabrication steps of SWCNT/PANI nanoporous composite. (b) The TEM image of pristine SWCNT aerogel. (c) The SEM images of SWCNT aerogel. (d) The SEM images of SWCNT/PANI composite. SWCNT: single-walled carbon nanotube; PANI: polyaniline; TEM: transmission electron microscopy; SEM: scanning electron microscopy.
The mechanical characteristics were carried out on the composite scaffold with different SWCNT loadings at 50% volume fraction composite scaffolds using tensile stress (σ) versus tensile strain (ε) measurements (Figure 2). The composite scaffold samples used for all mechanical measurements were rectangular. We did not use the dog bone-shaped samples because their width to length ratio is larger and stress is fairly uniformly applied along with these samples. The tensile modulus is the calculated slope of the linear region of stress versus strain at lower strain limits. The mechanical properties of composite scaffold depend on nanostructure parameters such as pore size, SWCNT concentration, the spatial distribution of SWCNT, and polymer. Firstly, all samples are CSA treated followed by drying at 60°C and the composite scaffolds were maintained with 50% volume fraction in a well-controlled manner. The pristine PANI sample was found to be fracture at a higher strain (8–10%) compared to the nanoporous composite scaffold at 25 wt% SWCNT loading (strain 2.8%). The pristine polymer was showed linear deformation at 0.3% strain with 35 MPa and then followed by a gradual deformation. The polymer sample showed up to 2.8% stretchability which indicates the specimen break (Figure 3(a)). The ultimate tensile strength (UTS) for nanoporous composite scaffold at 25 wt% SWCNT loading was approximately 0.8 MPa (400%) times that of the pristine PANI polymer (Figure 3(b)). The tensile modulus of the nanoporous composite scaffold at 25 wt% SWCNT loading was approximately increased 70 MPa (200%) times improvement over the pristine PANI polymer (Figure 3(c)). The composite scaffold at 25 wt% SWCNT loading showed higher tensile modulus, UTS, and higher stuffiness compared to the pristine PANI polymer due to the stronger interfacial interaction between polymer and nanotubes. It could also be speculated that the reduction of pore size at higher SWCNT loading leads to more friction, stronger interfacial interactions between SWCNT and PANI polymer which helps to increase the tensile modulus and UTS.
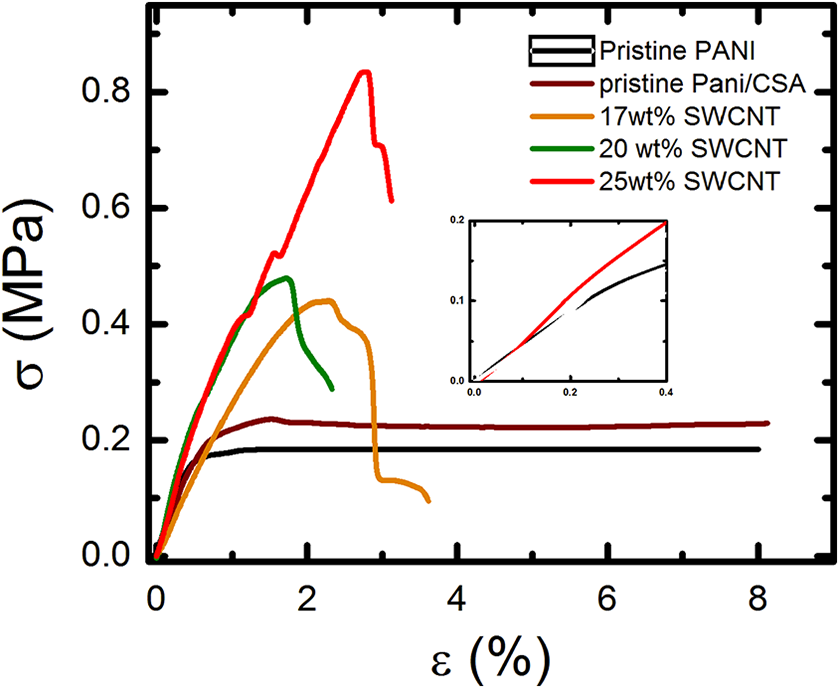
Stress–strain diagrams for pristine PANI and their nanoporous composites. PANI: polyaniline.

Mechanical characteristic of composites. (a) The tensile strain as the function of SWCNT concentrations. (b) UTS as a function of SWCNT concentrations. (c) Elastic modulus as the function of SWCNT concentrations. SWCNT: single-walled carbon nanotube; UTS: ultimate tensile strength.
The origin of mechanical enhancement of these composite scaffolds might also originate from an improved structure, which might be tracked using thermal analysis using DSC. For pristine the PANI sample, the DSC curve on heating showed three transitions at around 93°C, 212°C, and 255°C (Figure 4). According to the literature, 2 the glass transition is observed from our studies at about 93°C. The other two peaks indicate the melting of the pristine polymer. The melting of pristine PANI appears to proceed in a multistep process with a first-order heat flow peak at 212°C, followed by another sharp heat flow peak at 255°C. The melting peak temperature of the nanoporous composite scaffold is increased by 33°C at 25 wt% SWCNTs. The crystallization history has a significant impact on the melting behavior of pristine PANI with the addition of SWCNTs. Whereas the pristine PANI polymer shows a double melting behavior, but the melting of nanoporous composites progressively evolves towards the single melting behavior.
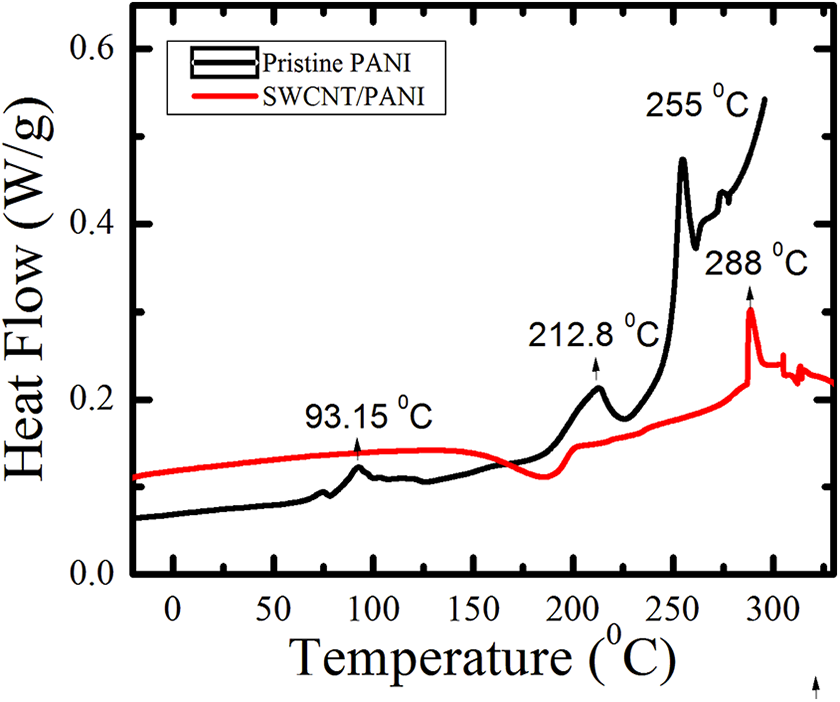
DSC measurements of PANI and nanoporous composites at 25 wt% SWCNT (volume fraction 50% samples). DSC: differential scanning calorimetry; SWCNT: single-walled carbon nanotube; PANI: polyaniline.
The multistep melting features are assumed as a result of polymorphism process and the successive melting of crystal growth with a distinct degree of perfection. From polymorphism process, SWCNTs can induce stronger crystallinity, and no peak is observed on cooling. This indicates that the sample does not have enough time to form a crystal or to recrystalline.
We also examined the stability of the composite scaffold using the TGA as shown in Figure 5. The weight loss of polymer in the composite scaffold is reduced in two different stages; the first-stage degradation process is from room temperature to 200°C. In this stage, degradation is attributed to the release of the moisture entangled within the PANI polymer. The second-stage degradation process is from 270°C to 550°C. The rapid weight loss is due to the breakdown of the backbone of the polymer. It is also showed that the degradation temperature of the composite scaffold is shifted to a higher temperature at 25wt%SWCNT loading (Figure 5). In the composite sample, weight loss is observed in a single stage from 300°C to 550°C. As the degradation temperature increases intensively, the polymer did not degrade even at 800°C in the presence of SWCNTs network which attributes the polymer anchored around the SWCNT network. The improved structure of composite scaffolds might be the reason for increased the thermal stability of composite scaffolds. It requires more energy for nanotube-coated polymer to degrade due to well-defined crystalline structure formation.

TGA of pristine PANI and nanoporous composites in air atmosphere at 25 wt% SWCNT (volume fraction 50%). TGA: thermogravimetric analysis; SWCNT: single-walled carbon nanotube; PANI: polyaniline.
The Raman spectrum of nanoporous composite scaffolds with different loading of SWCNTs is shown in Figure 6. For comparison, the top panel shows pristine PANI and CSA-treated PANI, and the bottom panel shows pristine SWCNT aerogel and nanoporous composites. The peaks around 150 cm−1 are related to the radial breathing modes (RBM), arising from the coherent vibration of carbon atom in the radial direction. The RBM modes mainly depend on SWCNT diameter. The peak at about 1289 cm−1, named as D-band, is arising in the presence of disorder from the defects located along the sidewalls of carbon nanotubes. 47 The peak at around 1582 cm−1, called G-band, is attributed to the elongation of the carbon–carbon bonds along the longitudinal directional of the carbon nanotubes. The peak at around 2565 cm−1, called G’-band, is known as a second-order harmonic of the D-band. 48 All the characterization peaks associated with the composite scaffolds have shifted 9 cm−1 to a higher wave number. This change is attributed to structural change in the composite scaffold sample which might be due to pristine PANI polymer anchored around the SWCNT network and improve the structure of the composite scaffold. The Raman spectrum is observed as a red-shift for nanoporous composite scaffold compared to the pristine polymer which might be due to the improved structural changes in the porous composite scaffold. Hence, the nanoporous composite scaffold shows higher electrical, thermal, and mechanical properties.

(a) Raman spectrum for pristine PANI and CSA-treated PANI. (b) Raman spectrum for pristine SWCNT and PANI composites. SWCNT: single-walled carbon nanotube; PANI: polyaniline; CSA: camphor sulfonic acid.
The electrical conductivity of CSA-doped nanoporous composites dried at 60°C as the function SWCNTs is shown in Figure 7. After the CSA-doping, the PANI composites dried at 60°C and the electrical conductivity is improved from 7°S cm−1 to 60°S cm−1 (Figure 7(a)). This increase in electrical conductivity might be due to higher crystalline structure formation after CSA doping. We also fabricated the samples with varying the ratio of SWCNT to PANI at two different volume fractions. The electrical conductivity is increased to 1:4 at 30% volume fraction sample (Figure 7(b)). We have chosen 50% volume fraction nanoporous composite scaffolds to characterize the electrical conductivity. The electrical conductivity of nanoporous composites scaffold at 50% volume fraction showed higher electrical conductivity due to improved structure formation. The electrical conductivity of porous composite scaffold increased at 25 wt% SWCNT and it might be due to less contact resistance between CNT-CNT and CNT-polymer. The conducting polymer PANI chains are anchored or wrapped around the junctions of CNT-CNT and reduce the contact resistance between CNT-CNT.

(a) Electrical conductivity of CSA-doped PANI composites is a function of SWCNTs at room temperature. (b) Electrical conductivity of PANI composites versus fraction of SWCNT to PANI. SWCNT: single-walled carbon nanotube; PANI: polyaniline; CSA: camphor sulfonic acid.
Conclusions
We used SWCNT hydrogel solution-based fabrication and in situ polymerization methods to fabricate composite scaffolds of individually dispersed nanotubes with PANI polymer coating to prevent SWCNTs aggregation. We found that the porous composite scaffolds were thermally stable and improved electrical and mechanical properties. The tensile modulus of the composite scaffold at 50% (volume fraction) was increased by 200% over the pristine PANI polymer at 25 wt% SWCNT loading. The electrical conductivity of the composite scaffold was increased by 800% compared to the pristine PANI polymer. The porous composite scaffolds were produced by using our fabrication method, have superior mechanical, electrical, and thermal properties, and might be used in thermoelectric material industry applications.
Footnotes
Acknowledgment
The authors would like to acknowledge CCE and Osmania University, Hyderabad, Telangana.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
