Abstract
The thermal degradation behaviors of poly(vinyl alcohol) (PVA) and PVA/zinc oxide (ZnO) composite are investigated using differential thermal analysis (DTA). The degradation activation energy (
Introduction
Inorganic–organic polymer composites have received wide interest because the addition of inorganic particles to polymers can enhance conductivity, mechanical toughness and optical and catalytic activities. 1 Polymer composites have been found successful in many applications, such as organic batteries, microelectronics, nonlinear optics and sensors. Consequently, it is important to understand the effects of the incorporation of particles on the physical and chemical properties of the composites. Among the organic polymers, poly(vinyl alcohol) (PVA) is a possible candidate to be used as biodegradable, nonhazardous and environmentally benign 2 because of its film-forming ability, good chemical stability and good mechanical properties. 3,4
The mechanical ball milling is strain-induced synthesis of various chemical compounds. 5 A multiscale continuum thermodynamic and kinetic theory can explain the key phenomena occurring during strain-induced structural changes under extreme condition. 6,7 With a ball milling method, a decrease in the crystallinity of the PVA is expected. This is because ball milling can partially change the PVA structure into low crystallinity due to the break down of PVA chain by the introduction of high energy.
The thermal behavior of polymers can be improved if the information about the thermal degradation kinetics and degradation mechanisms can be employed to decrease the thermal degradation rate or increase the heat resistance. It has been reported that nanoparticles can enhance thermal degradation of nanocomposites. 8 –10 The aim of this work is to study the effect of zinc oxide (ZnO) as filler on the thermal stability of the PVA. Also, we determine the degradation kinetic parameters of PVA/ZnO composites.
Experimental
The starting material is commercial ZnO nanoparticles (purity 99.99%, Sigma Aldrich, Saint Louis, USA), and the powder of PVA is obtained from S.D. Fine-Chem. Ltd, India) with a molecular weight of 14,000 laboratory reagent (LR). The weights of ZnO nanoparticles are 1, 2 and 5% of weight of PVA powder. The mixed powders are milled in a mechanical ball-milling machine (type KM1 VEB, Leuchtenbau, Germany) for the duration of 2 h in an agate vial. Agate ball is used to avoid contamination. The milling is carried out in an air atmosphere, and after a certain period (maximum 15 min), the milling is interrupted for cooling down the machine and for ensuring that the temperature of the mills does not exceed 50°C. The ball to powder mixture weight ratio is taken as 20:1.
The analysis of the milling products is performed by the differential thermal analysis (DTA) measurements using the Shimadzu thermal analysis apparatus (DTG-60H) Japan. The sample is placed in a quartz basket suspended from the arm of the balance by means of a quartz wire. They are heated under nonisothermal conditions at different heating rates (5–35°C min−1) under air atmosphere. The sample temperature is adjusted using a Heraeus temperature controller (type TRK) involved in the apparatus of the thermal analysis.
Results and discussion
DTA of PVA and PVA/ZnO composite
Figure 1 shows the DTA traces of unfilled PVA and PVA filled with 5 wt% ZnO nanoparticles at heating rates (
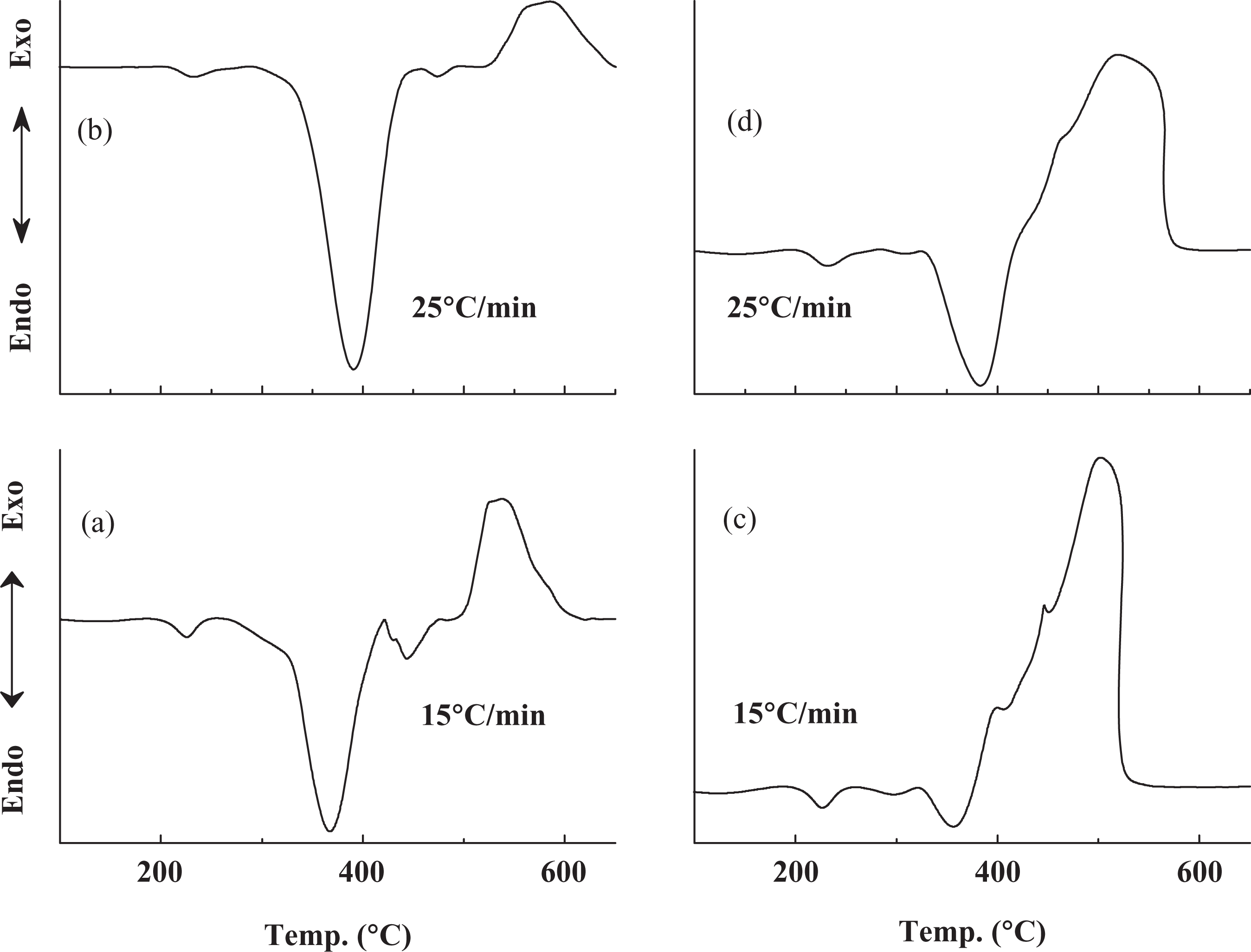
DTA curves for: PVA (a and b) and PVA/ZnO composite (c and d). PVA: poly(vinyl alcohol); DTA: differential thermal analysis; ZnO: zinc oxide.
The large exothermic peak centered at 535°C meets the full degradation of the polymer. A higher heating rate leads to degradation at higher temperature, which results in a dependence of distribution of the size of volatile products on the heating rate. In other words, degradation temperature influences the size of the volatile products. Figure 1(b) shows that the degradation occurs at 580°C as the heating rate becomes 25°C min−1.
In the case of PVA/ZnO composite, the first decomposition stage is indicated by endothermic peak at 357°C and the second stage is represented by two kink-like exothermic peaks at 397 and 447°C (Figure 1(c)). Therefore, the decomposition is accompanied by the release of polymer–ZnO links, which may be responsible for the change in the direction of the heat flow and the new decomposition event in the case of PVA/ZnO composite.
Degradation kinetics
The degradation process of the PVA takes place in two steps.
12
In the first step, the H2O is eliminated and the residual acetate groups generate water, nonconjugated polyenes and acetic acid. The second degradation step is dominated by chain-scission reactions and cyclization reactions, and continual elimination of residual acetate groups is also found in this step. The overall degradation activation energy (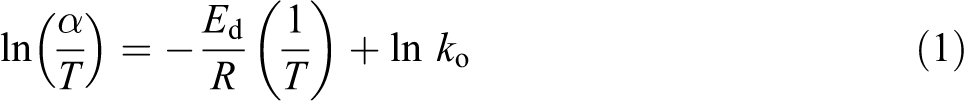
where
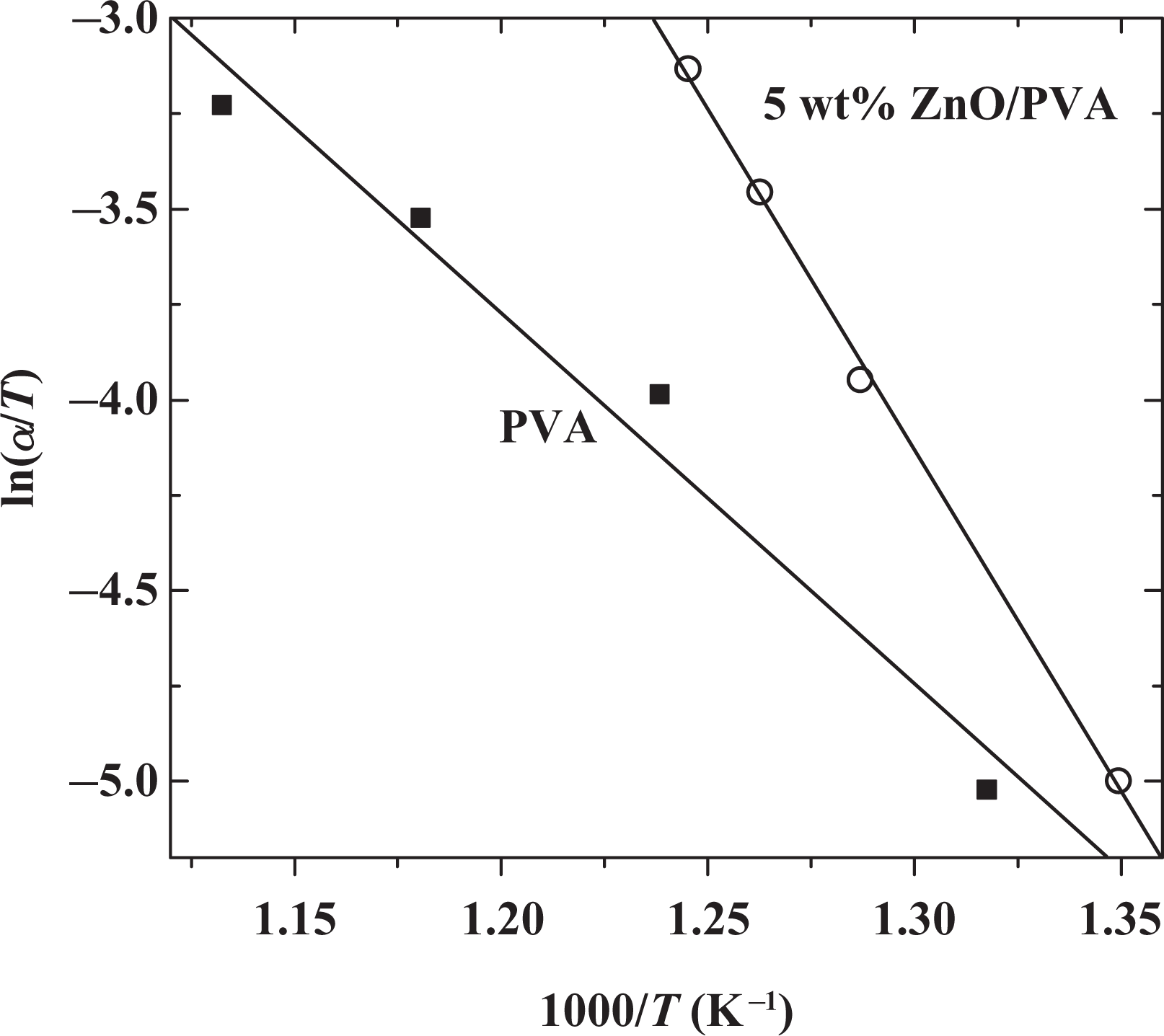
Plots of ln(
The frequency factor
The interpretation of DTA data is provided by the formal theory of transformation kinetics as the degradation rate constant (
The constant
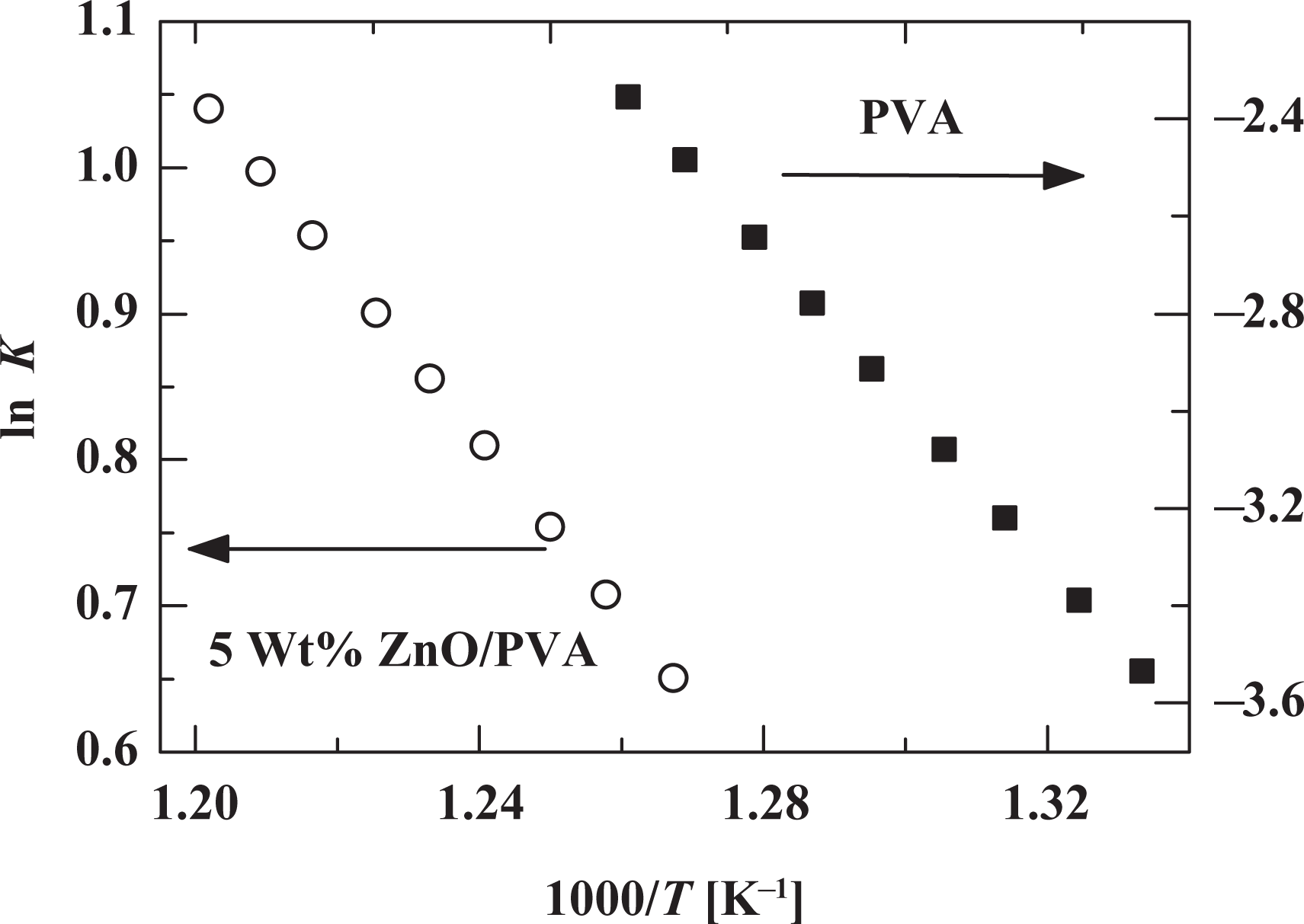
Temperature dependence of the degradation rate constant for PVA and ZnO/PVA composite at heating rate of 15°C min−1. PVA: poly(vinyl alcohol); ZnO: zinc oxide.
The order of degradation reaction can be determined by modifying the model suggested by Avrami to analyze the crystallization kinetics.
14
In this model, the crystallized fraction (
where Shape analysis of degradation peak for PVA matrix at heating rate of 15°C min−1. PVA: poly(vinyl alcohol).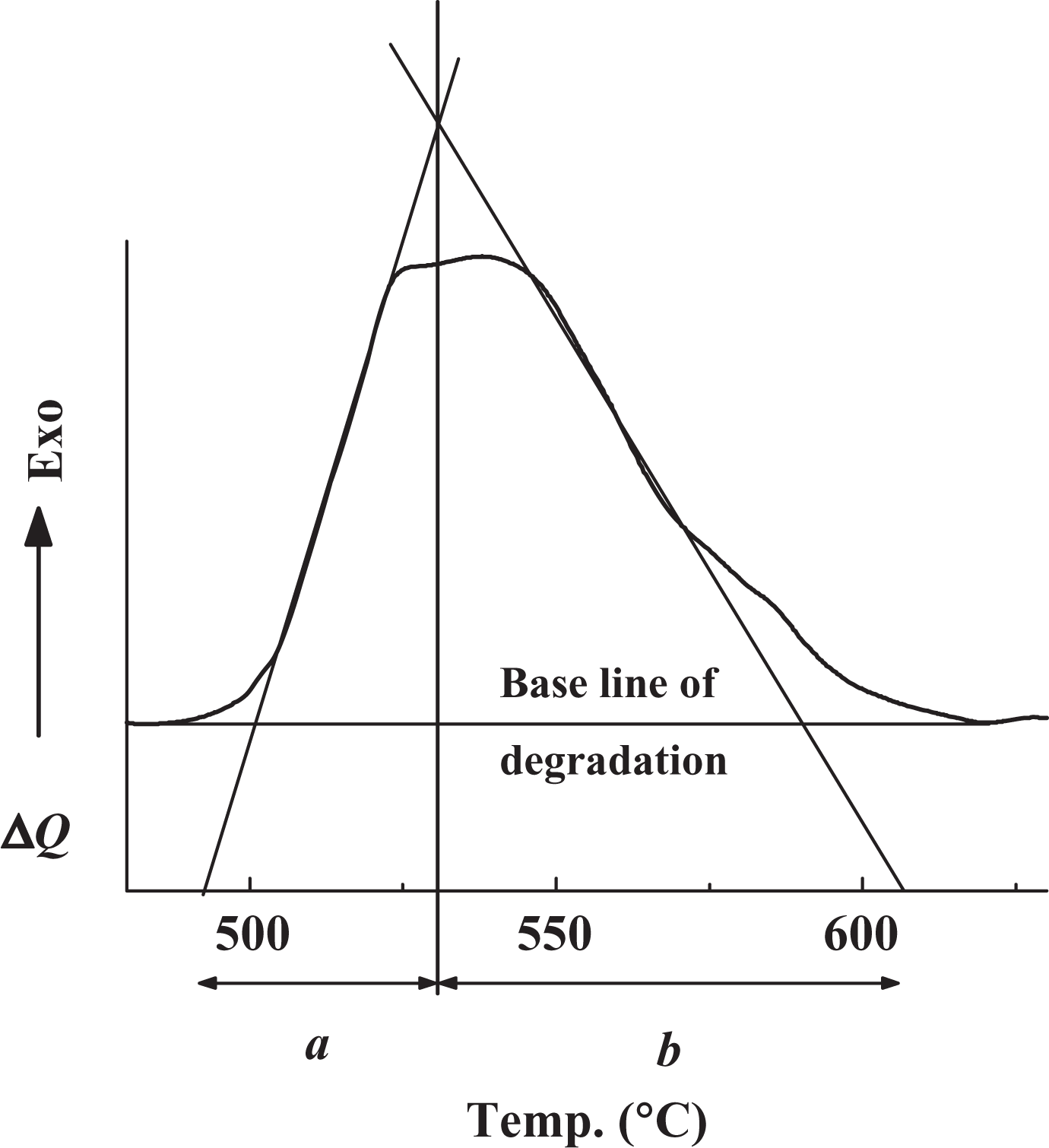

where
The Avrami exponent (
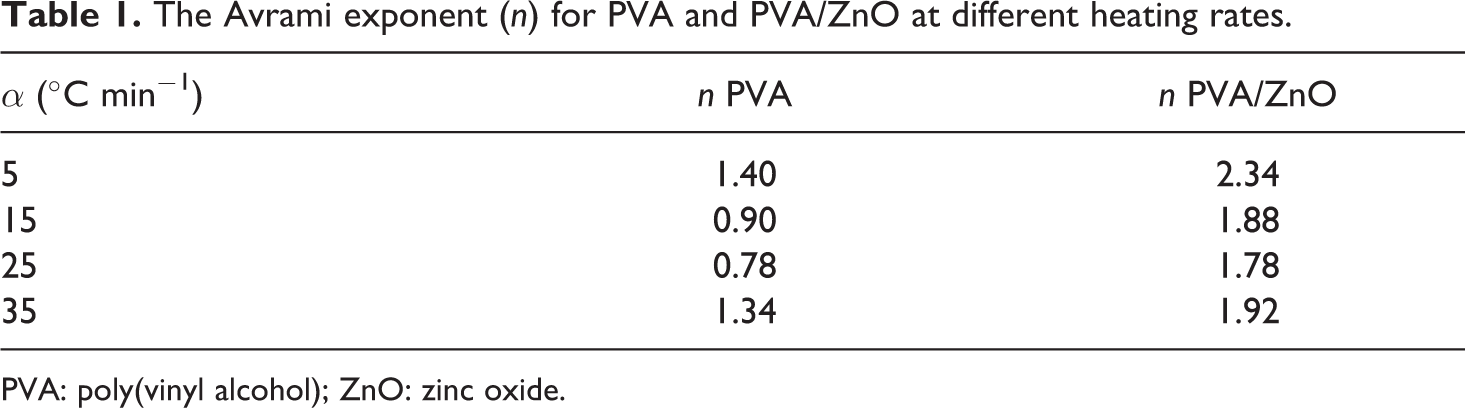
PVA: poly(vinyl alcohol); ZnO: zinc oxide.
Comparison of the experimental results with those for the theoretical models can provide information on the reaction mechanisms for unfilled and filled PVA. The obtained exponent
Conclusion
The DTA measurements reveal that PVA/ZnO composite is more thermally stable than PVA. This is evidenced from the lower value of the degradation activation energy of PVA in comparison with that of PVA/ZnO complex. The behavior of the degradation rate confirms the priority of the degradation of PVA matrix relative to the PVA/ZnO composite. The degradation mechanism of this polymer and its composite follows the Avrami–Erofeev mechanism.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
