Abstract
The organically modified montmorillonite (OMMT)-filled polyamide 6 (PA6) nanocomposites were toughened with epoxidized natural rubber (ENR). The PA6, ENR (15–30 wt%) and OMMT (4 parts per hundered (phr)) were melt compounded using counterrotating twin-screw extruder followed by the injection molding. X-ray diffraction (XRD) results indicated that the OMMT platelets in PA6/ENR/OMMT nanocomposites were well dispersed. The Fourier transform infrared (FT-IR) spectra showed graft esterification reaction between PA6 and ENR during processing. It was found that the addition of ENR (up to 20% wt) increased the impact strength and elongation at break of the nanocomposites. Scanning electron microscopy (SEM) images revealed well-dispersed ENR particles. Differential scanning calorimetry (DSC) results showed that the presence of ENR and OMMT had negligible effect on the glass transition of PA6 with a slight decrease in crystallization temperature and crystallinity in PA6/ENR/OMMT nanocomposites.
Introduction
The combination of both mechanical and thermal properties allows polyamide 6 (PA6) to be employed for highly specified end uses and often for metal replacements. 1 Despite its inherent mechanical properties, PA6 is sensitive to crack propagation and becomes brittle at low temperature which limits its applications. 2 Recent achievements in nanocomposites technology have fueled the need for new knowledge and findings in the field of polymer nanocomposites resulting in the development of several polymer nanocomposites such as polyethyelene, 3 polypropylene, 4 polystyrene 5 and natural rubber (NR). 6 The field of PA6 nanocomposites based on layered silicates, such as organically modified montmorillonite (OMMT) has also gained its popularity among scientists and industrialists. 7,8 The nanoscale distribution of such high-aspect ratio fillers in nanocomposites causes huge improvements to the polymer matrix in terms of mechanical, fire retardant, rheological, gas barrier and optical properties, especially at low clay content (<5 wt%) in comparison with conventional microcomposites (>30 wt% of microfiller). 9 –11 Interestingly, the distribution of nanoclay in PA6 is proven to be well dispersed without the introduction of compatibilizing agents due to the hydrogen bonding interactions between amine, carbonyl groups of PA6 and hydroxyl groups of OMMT. 12,13
Regardless of the improvements achieved in the development of PA6/OMMT nanocomposites, the polymer’s notch sensitiveness and brittleness have limited its performance for high-end applications, and the toughness of PA6/OMMT nanocomposites can be improved by the introduction of tough materials with considerable sacrifice in stiffness. 12 On the contrary, effective toughening agents must be functionalized due to the polarity of PA6. Therefore, it is essential to improve the interfacial adhesion by reactive blending between PA6 and rubber phase. Maleated rubber such as maleated styrene–ethylene/butylene–styrene copolymer (mSEBS), 14 –17 maleated ethylene–propylene–diene copolymer (mEPDM), 18,19 maleated ethylene–copropylene (mEPR) 20 –22 and maleated ethylene–octene elastomer (mPOE) 23 –25 has been popular in the recent years as the grafted maleic anyhydride groups react with the amine groups of PA6 yielding an in situ graft copolymers on the interphase of both phases. 26 In theory, the formation of this graft copolymer promotes the compatibility of rubber with PA6 leading to a decreased interfacial tension and good dispersion of rubber particles, and finally improved the mechanical toughness of the composite. 13
Although NR can be used as a toughening agent for many thermoplastics, it may not be compatible with PA6 due to the dissimilarity in polarity. 2 Therefore, the polarity of NR is improved via epoxidization of unsaturated chains in NR to form epoxidized NR (ENR). Previous investigation by Tanrattanakul et al. 2 on toughening PA6 with ENR was successful with the increment in impact strength parallel to ENR content, and another study by Xie and his coworkers 27 revealed that the carboxyl end groups of PA6 can chemically react in situ with epoxy groups of ENR during processing, resulting in the formation of ester groups. Nevertheless, the potential use of ENR as a toughening agent for PA6/OMMT nanocomposite is still unexplored. Recent study by our group 28 revealed interesting findings with the use of ENR to overcome the brittleness of PP/OMMT nanocomposites. The present study aims to develop rubber toughened PA6/OMMT nanocomposites and investigate the effect of various ENR contents on the mechanical and morphological properties of PA6 and PA6/OMMT nanocomposites.
Materials and method
Materials
PA6 was obtained from Toray Plastics Japan (product name Amilan CM1017-K) with melt mass flow rate (MFR) of 35 g/10 min at 230°C and 2.16 kg load and density of 1.14 g/cm3. ENR with 25 mol% epoxidation (grade EPOXYPRENE 25) and specific gravity 0.97, used in this study, was used as received from the Malaysian Rubber Board, Malaysia. OMMT obtained from Nanocor Inc., Arlington Heights Illinois, USA (Nanomer 1.30TC) was organically modified with octadecylamine with mean dry particle size of 16–22 µm.
Nanocomposites preparations
PA6 pellets were dried at 82°C for 24 h prior to compounding. PA6/ENR blend and nanocomposites as stated in Table 1 were compounded by simultaneous addition of all components to Brabender Plasticoder P 2000 counterrotation twin-screw extruder. PA6 and ENR blends were prepared by weight percentage (wt%) while OMMT was added as parts per hundred (phr) of PA6/ENR blends. The barrel temperature profile adopted during compounding for all formulations was 190°C at feed section, increasing to 240°C at the die head. The screw rotation speed was fixed at 60 rpm. The extruded materials were injection molded into standard tensile, flexural and Izod impact specimens using a JSW (Muraron, Japan) Model NIOOB II injection-moulding machine with a barrel temperature range of 200°C–240°C. All test specimens were left under ambient conditions in desiccators for at least 24 h prior to testing.
Designation of samples and their compositions.
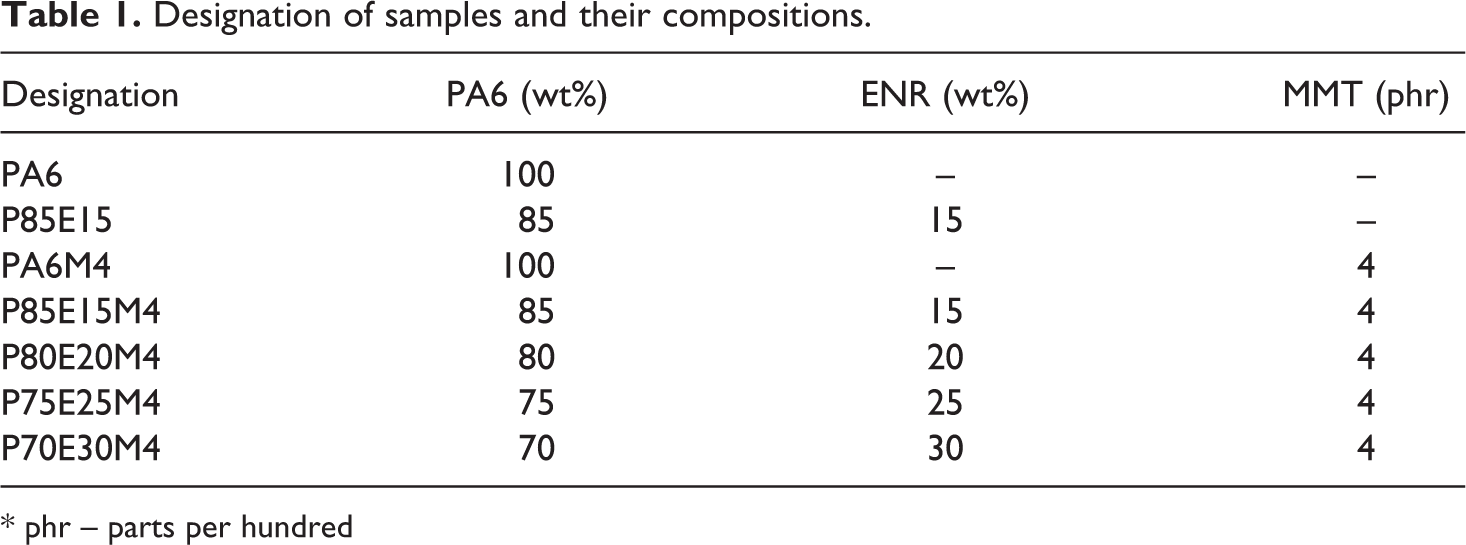
* phr – parts per hundred
Mechanical analysis
Tensile test was carried out according to ASTM D638 using an Instron (Bucks, UK) 5567 under ambient conditions with a crosshead speed of 50 mm/min. Flexural test was done according to ASTM D790 by Instron (Bucks, UK) 5567 under ambient conditions with a crosshead speed of 5 mm/min. Izod impact tests were carried out on notched impact specimens according to ASTM 256, using a Toyoseiki (Tokyo, Japan) impact testing machine under ambient conditions. Five specimens of each formulation were tested and the average values were reported.
Morphological analysis
X-ray diffraction (XRD) analysis was carried out on a Siemens (Berlin, Germany) D5000 X-ray diffractometer. The diffraction patterns were recorded with a step size of 0.02° from 2θ = 2.0 to 10.0°.The interlayer distances (d-spacing) of the OMMT in the nanocomposites were derived from the peak positions (d001 reflections) in the XRD scans, according to the Bragg equation (1). 
where n is an integer, θp is the diffraction angle giving the primary diffraction peak, and λ is the X-ray wavelength. In these experiments, λ = 0.154 nm (Cu Kα) and n = 1 were used.
Philips ZL 40 Scanning electron microscopy (SEM) was used to observe the morphology of the PA6/ENR blend and nanocomposites. All the SEM samples were impact fractured perpendicular to the injection-moulding direction. The samples were etched in toluene at 50°C for 3 h to extract the ENR phase and dried in oven at 40°C for 24 h. Samples were coated with a thin layer of platinum prior to examination under the electron beam. An operating voltage of 25 kV and magnification of 3000× was used. The particle size of ENR particles was calculated from the diameter of the particles which were approximated to spheres. More than 150 particles were evaluated and these were taken as the mean diameter and hence mean size of the particles.
Chemical structure analysis
Fourier-transform infrared (FTIR) spectra were recorded by a Perkin Elmer FTIR spectrophotometer with a resolution of 2 cm−1 for scans more than the wave number range of 3600–800 cm−1.
Thermal analysis
The melting and crystallization behavior of the blend and nanocomposites were studied under nitrogen by differential scanning calorimetry (DSC; Perkin–Elmer DSC-6), using 8–10 mg sample sealed into aluminum pans. The temperature was raised from 30°C to 300°C at a heating rate of 10°C/min; and after a period of 1 min, it was swept back to 10°C/min. The fusion enthalpy ΔHm
(PA6) was measured and the degree of crystallinity Xc
(PA6) was calculated from the following equation:
26
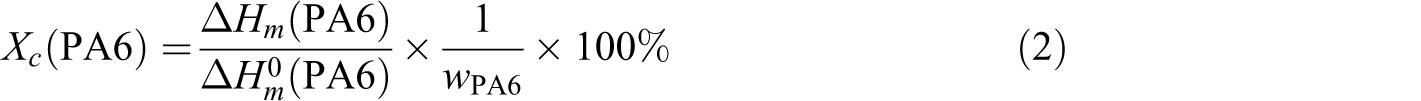
where
Results and discussion
Mechanical analysis
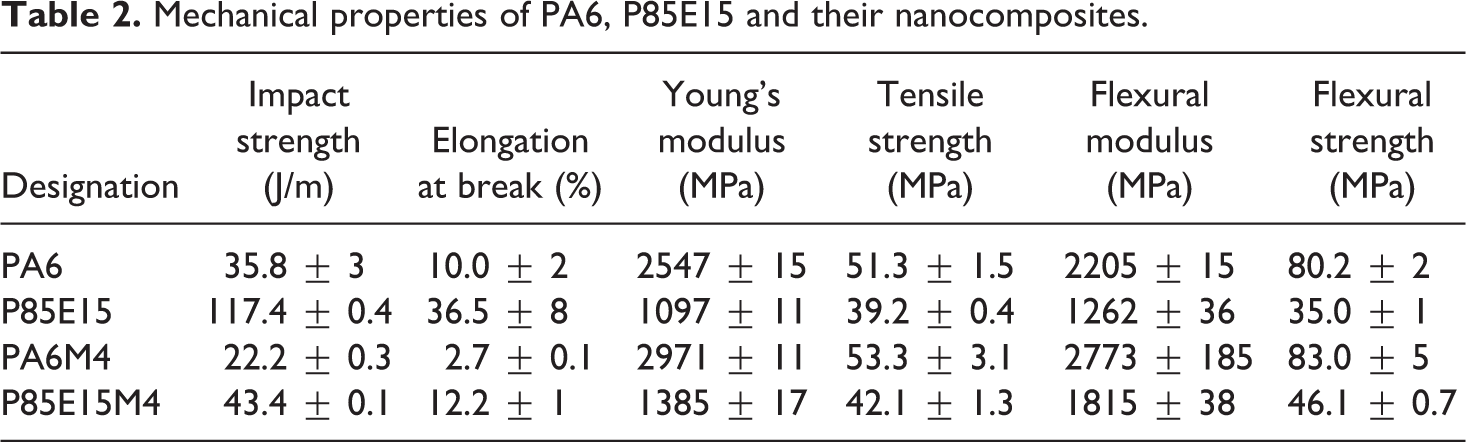
The mechanical properties of PA6, PA6/ENR and their nanocomposites are tabulated in Table 2. It can be observed that the impact strength and elongation at break values of PA6 decreased with the addition of 4 parts per hundred (phr) OMMT in PA6/OMMT nanocomposites. This shows that the presence of OMMT as stress concentrators in PA6 matrix led to embrittlement as discussed by Alexander and Dubois. 9 Therefore, the subsequent steps of our work have been directed toward improving the toughness of PA6/OMMT nanocomposites with ENR as an impact modifier. As seen from Table 2, the incorporation of 4 phr OMMT to PA6/ENR blend also led to a reduction in elongation at break (73%) due to the restraints on mobility of the PA6 chains caused by OMMT in PA6. Similar observation was noted by Kusmono et al., 26 where the elongation at break value decreased from 11.9% to 3.5%, with addition of 4 phr OMMT in PA6/PP and PA6/PP/mSEBS nanocomposites, respectively.
Mechanical properties of PA6, P85E15 and their nanocomposites.
From Figure 1, it can be observed that the impact strength values increased to maximum at 20 wt% (cal. 31%) and thereafter, it started to decrease showing the potential use of ENR to improve the toughness of PA6/OMMT. Previous studies also showed improvement in impact strength of PA6 nanocomposites with blending of rubbery phase such as mSEBS, 15 mEPDM 19 and mEPR. 20 The relationship between elongation at break and ENR content is also shown in Figure 1. The elongation at break of PA6 nanocomposite increased around 640% with 20 wt% blending of ENR. Similar trend was reported by Nishitani and coworkers 16 who observed an increment in elongation values with the blending of mSEBS (rubbery phase) into PA6/OMMT nanocomposites. As highlighted by Horiuchi et al., 29 good interfacial adhesion between PA6 and ENR is essential to achieve high elongation at break values in PA6/ENR/OMMT nanocomposites. Therefore, it can be proposed that the observed improvement in elongation at break and impact strength in the present study might be due to the good interfacial adhesion between PA6 and ENR in PA6/ENR nanocomposites.
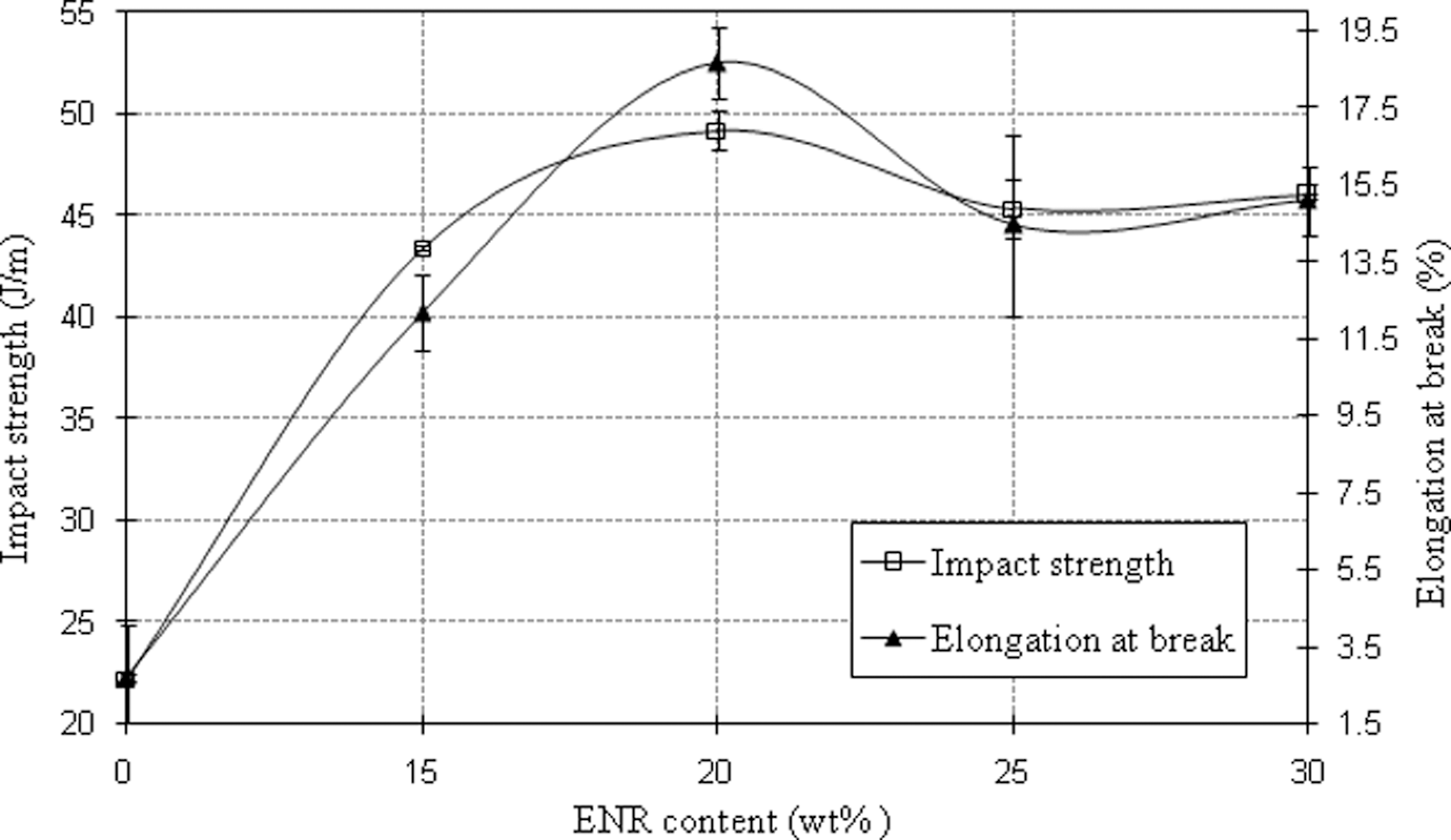
Impact strength and elongation at break of PA6/OMMT nanocomposites with various ENR content. PA6: polyamide 6; OMMT: organically modified montmorillonite; ENR: epoxidized natural rubber.
The enhanced interfacial adhesion attributed to the possible grafting reaction between PA6 and ENR is illustrated in Figure 2. The graft esterification reaction between epoxy groups of ENR and carboxylic end groups of PA6 has resulted in the formation of ester bonds between PA6 and ENR. The resulted structure offers many polar sites for hydrogen bonding interactions between octadecylamine (an intercalant group in organically modified OMMT) and PA6. The OMMT used in this study contained about 30 wt% octadecylamine intercalant. 13 ENR epoxy groups (both reacted and unreacted) also may have possible hydrogen bonding interactions with OMMT platelets as shown in Figure 2. The grafting reaction between PA6 and epoxy groups of ENR was investigated in detail by Xie et al. 27
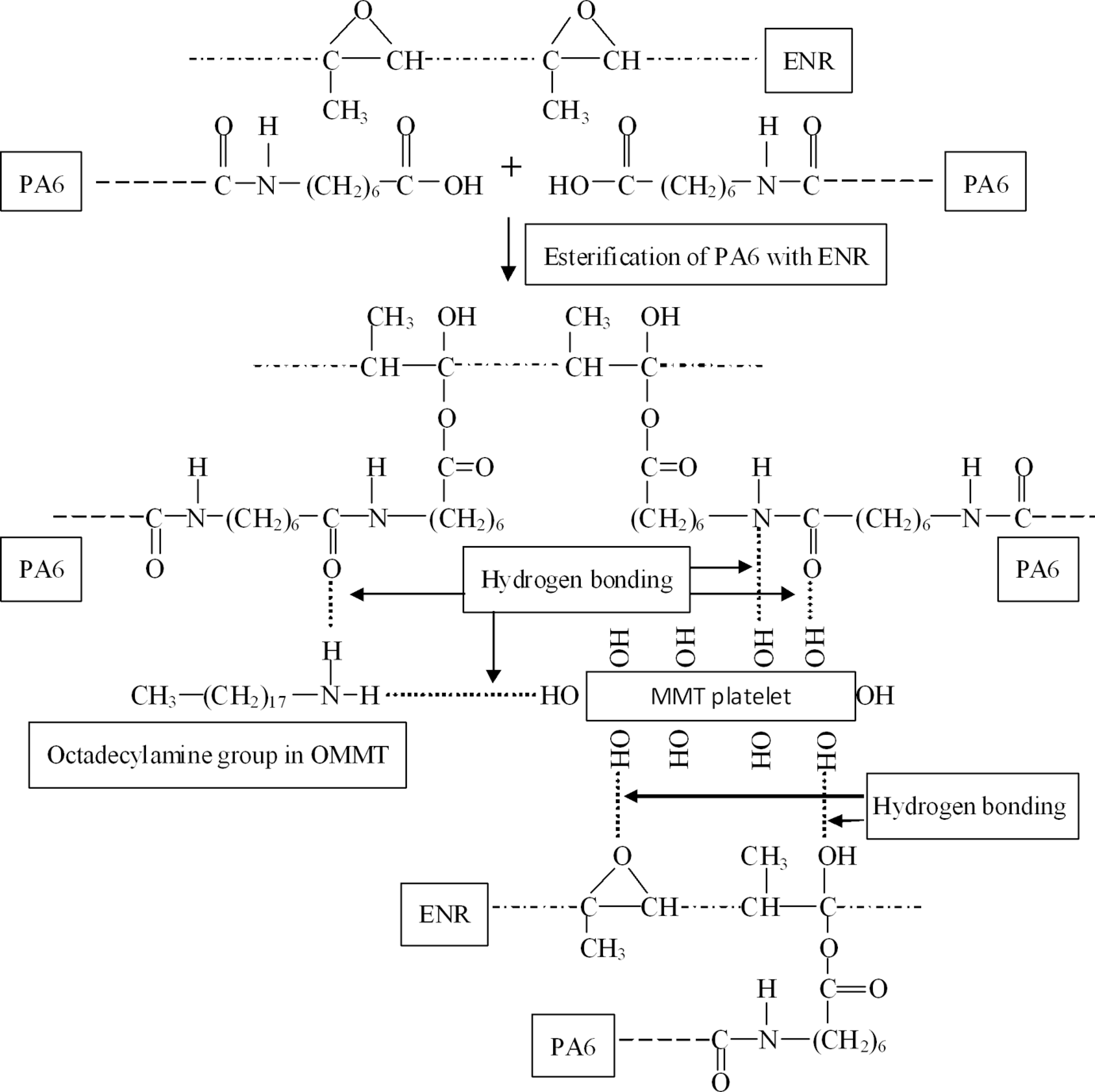
Schematic representation of the esterification reaction and hydrogen bonding in PA6/ENR/OMMT nanocomposite. PA6: polyamide 6; OMMT: organically modified montmorillonite; ENR: epoxidized natural rubber.
The ability of PA6 and ENR to undergo reactions resulted in better dispersion of ENR in PA6 matrix as observed in the SEM images (morphological analysis). It is known that polarity also promotes interfacial adhesion between ENR and PA6, making them compatible. 2 The polarity of ENR may pose an advantage for ENR as a toughener in PA6 compared to other synthetic rubbers such as EPDM, 30 SEBS 31 and POE, 32 which requires compatibilizers to promote surface adhesion between rubber and PA6 phases. The decrease observed in impact strength and elongation at break of PA6/OMMT nanocomposites at 25 wt% and 30 wt% ENR content in Figure 1, however, may be due to the possible agglomeration of ENR at higher content (refer to SEM analysis). Similar observation was reported by previous researchers 15,18 with various types of rubbers to toughen PA6/OMMT nanocomposites.
Figure 3 shows that the Young’s modulus and tensile strength of PA6/OMMT decreased with increasing ENR content. Previous studies also observed a similar trend with blending of impact modifiers such as mSEBS, 14 mEPDM, 18 mEPR 20 and mPOE 23,25 into PA6/OMMT nanocomposites. The decrease in modulus and strength is due to the elastomeric nature of ENR which has low stiffness and strength. 2 However, the reduction in Young’s modulus and tensile strength is inconsequential with the presence of OMMT. The high aspect ratio of OMMT platelets has enabled it to perform as a reinforcing agent. 33 –37
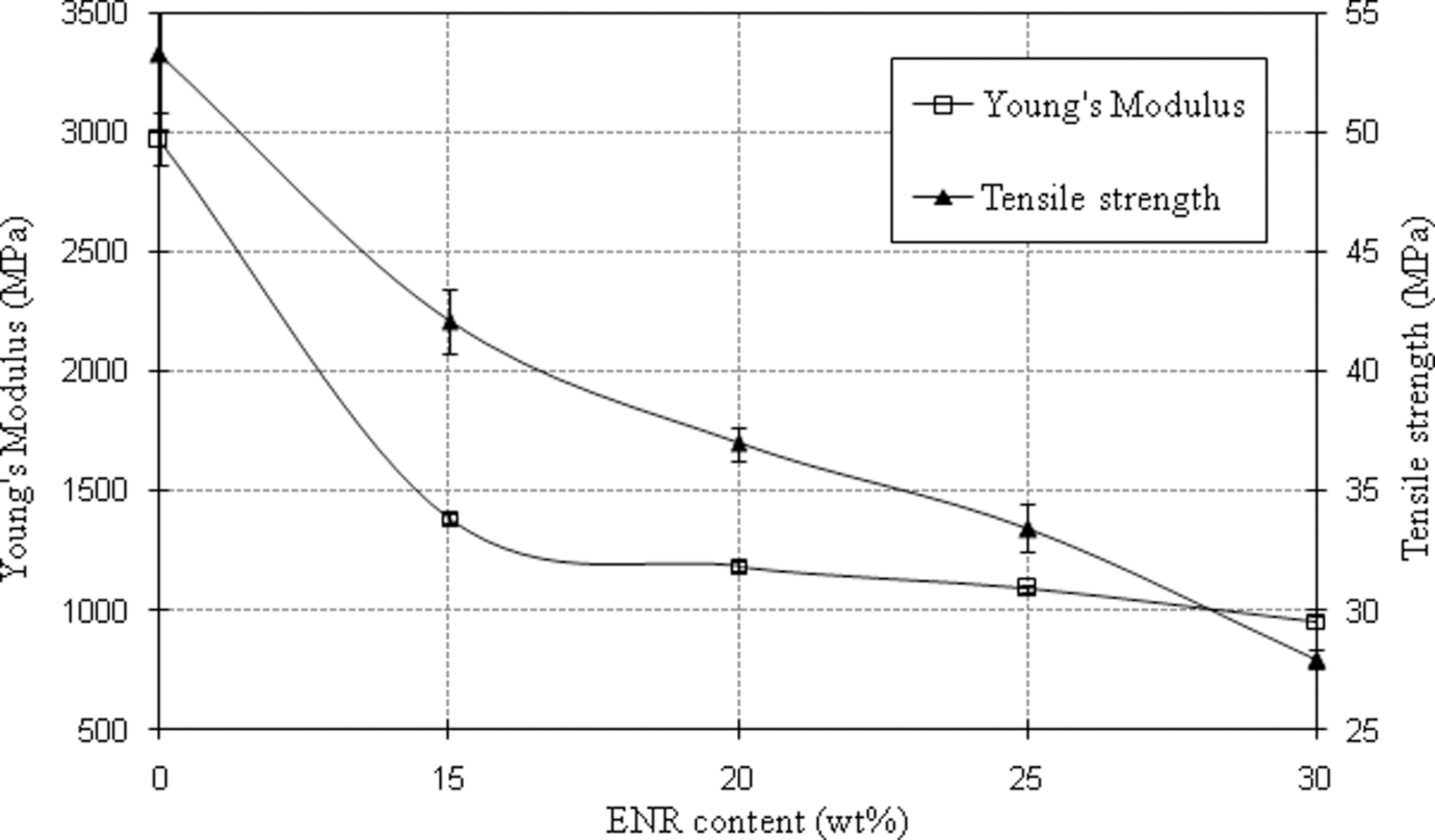
Young’s modulus and tensile strength of PA6/OMMT nanocomposites with various ENR content. PA6: polyamide 6; OMMT: organically modified montmorillonite; ENR: epoxidized natural rubber.
The flexural modulus and strength of PA6/OMMT nanocomposites also decreased gradually with increasing ENR content as shown in Figure 4. In addition, the presence of OMMT as a reinforcing agent prevented further decrement in the stiffness of PA6 nanocomposites compared to PA6 with 15 wt% of ENR as shown in Table 2. Table 2 also shows that the flexural modulus and strength of PA6 improved with addition of 4 phr OMMT, indicating that OMMT platelets restricted the movement of PA6 chains during deformation. Shishan et al. 38 also reported that modulus of PA6 nanocomposites increased with increasing OMMT content up to 6 wt% of OMMT. Moreover, the possibility of hydrogen bonding interactions between ENR and OMMT as described earlier (Figure 2) facilitated the dispersion of OMMT.
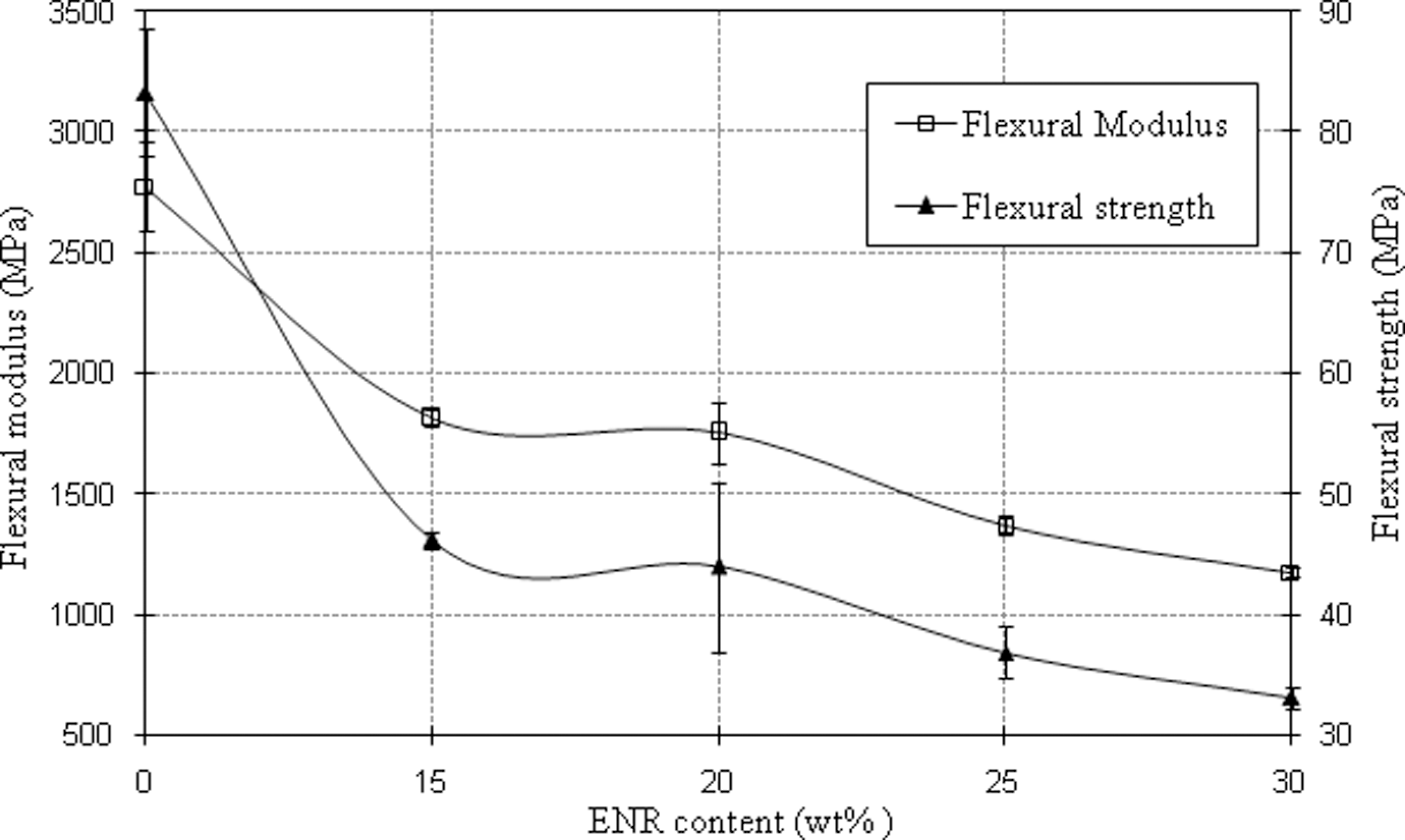
Flexural modulus and strength of PA6/OMMT nanocomposites with various ENR content. PA6: polyamide 6; OMMT: organically modified montmorillonite; ENR: epoxidized natural rubber.
Balanced mechanical properties
One of the most important aspects of materials development in thermoplastics engineering is to achieve a good combination of properties and processability at a moderate cost. As far as mechanical properties are concerned, the main target is to strike a balance of stiffness and toughness. Figure 5 shows the effect of ENR on stiffness and toughness of PA6/OMMT nanocomposites. It is clear that the addition of ENR improved the toughness of PA6/OMMT nanocomposites overcoming its brittleness. However, the blending of the ENR also led to a substantial reduction in stiffness of PA6/OMMT nanocomposites. It can be observed that PA6/OMMT nanocomposites with 20 wt% ENR has a balanced impact strength and flexural modulus values compared to other formulations.
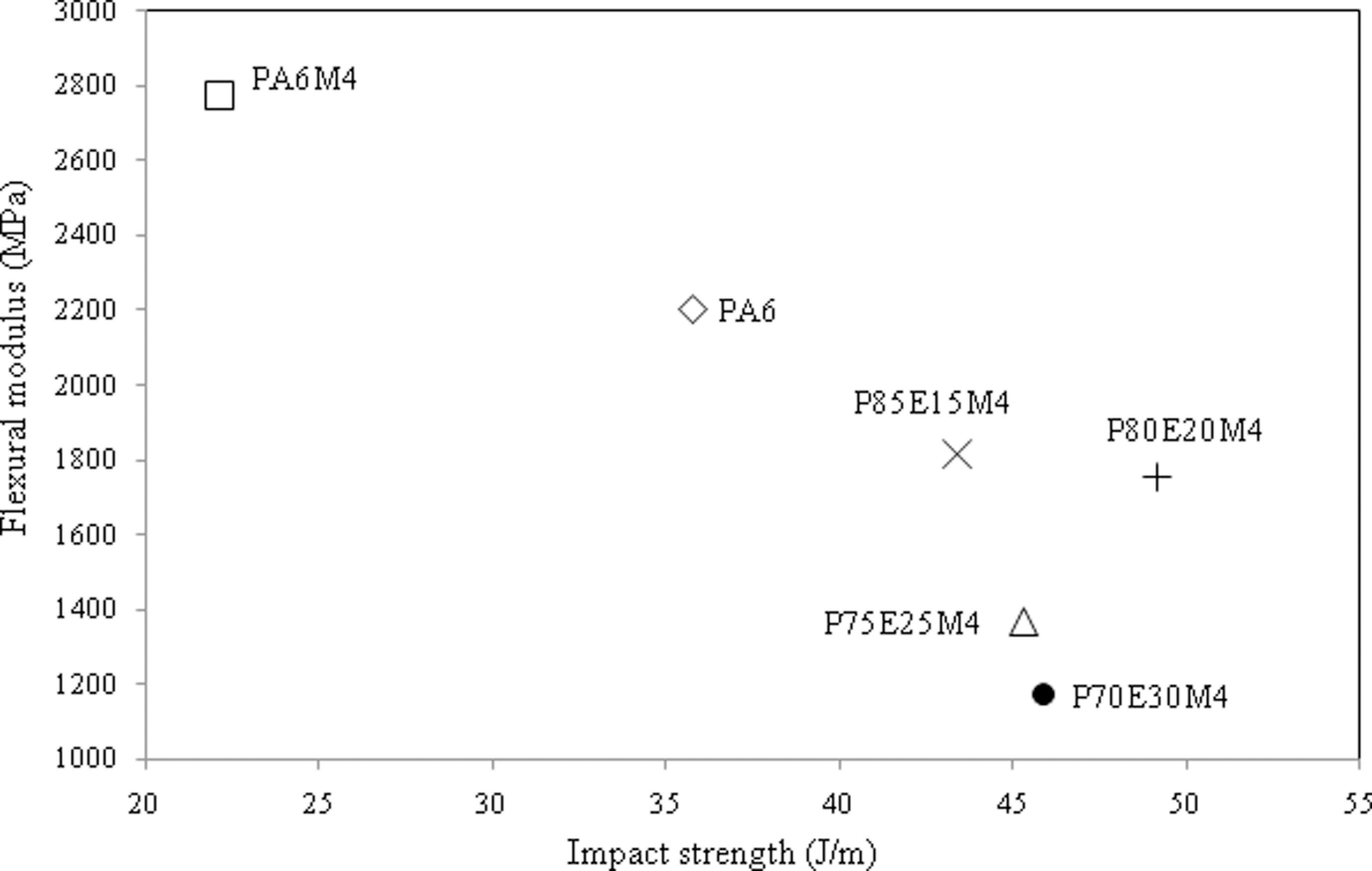
Determination of balanced properties based on impact strength and flexural modulus of PA6/OMMT and PA6/ENR/OMMT nanocomposites. PA6: polyamide 6; OMMT: organically modified montmorillonite; ENR: epoxidized natural rubber.
Chemical structure analysis
FT-IR spectroscopy
Figure 6 shows the FT-IR spectra of PA6, P85E15, P85E15M4 and ENR. The bands at ∼3300 cm-1 (N-H stretching), ∼2921 cm-1 (C-H stretching), ∼1640 cm-1 (Amide I, C=O stretching), ∼1540 cm−1 (Amide II, N-H bending + C-N stretching) are the characteristic bands of PA6. 26,39 The appearance of ∼2350 cm−1 with a significant intensity is found to be related to the thermal decomposition of PA6 during processing. 40 Nevertheless, the effect of OMMT and ENR on the thermal decomposition of PA6 was not explored in this study and will be worth a future investigation.
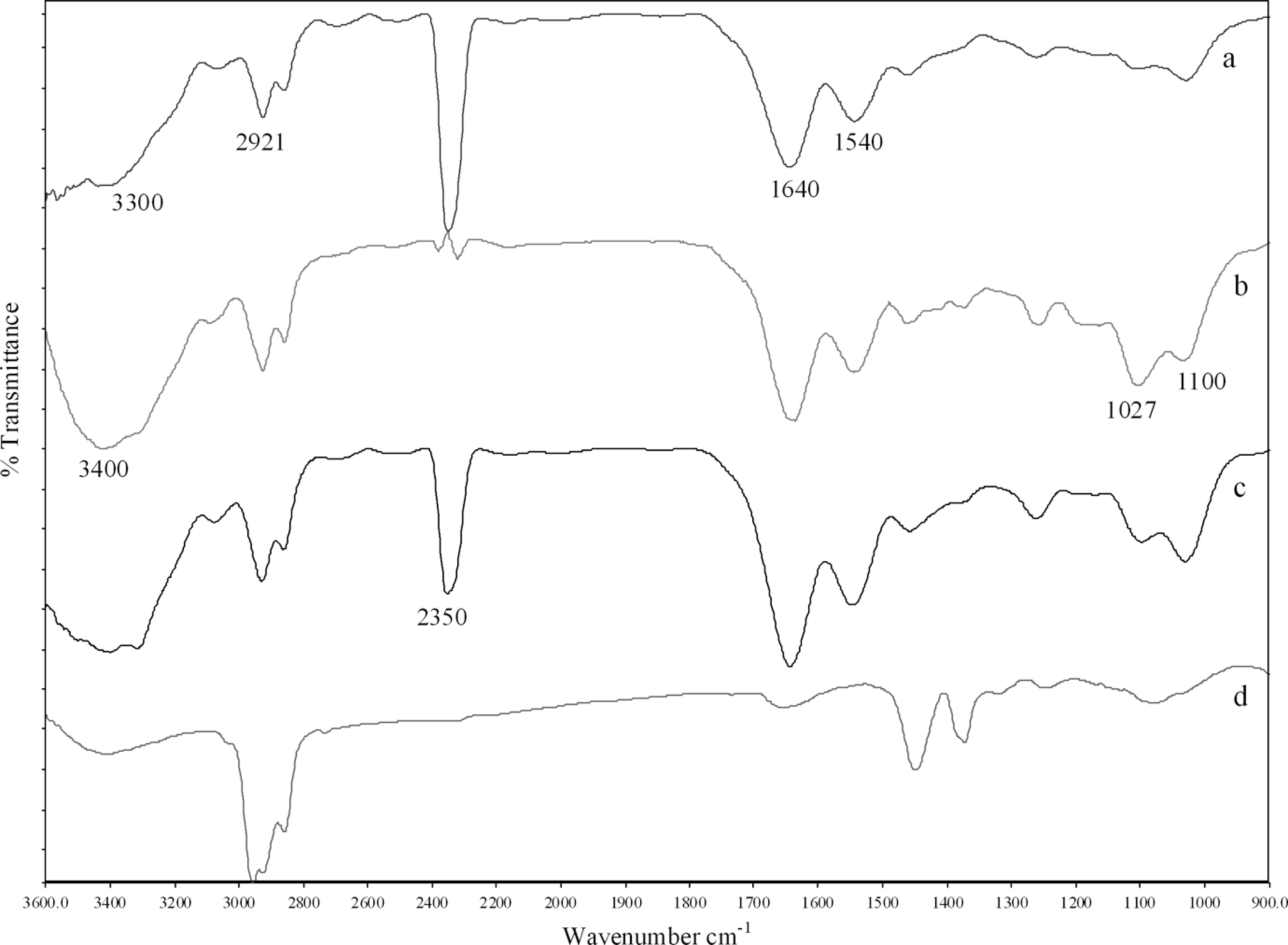
FT-IR spectra of (a) PA6 (b) P85E15 (c) P85E15M4 (d) ENR. PA6: polyamide 6; FT-IR: Fourier transform infrared; ENR: epoxidized natural rubber.
The significant difference in the infrared spectra with blending of ENR is the appearance of C-O stretching (ester) band at ∼1100 cm−1 which indicates the presence of more C-O ester bonds. This provides evidence of graft esterification reaction between PA6 and ENR in PA6/ENR and PA6/ENR/OMMT nanocomposites as proposed by scheme in Figure 2. Similar observation was noted by Xie et al. 27 The appearance of strong absorbance at 3400 cm−1 for PA6/ENR and PA6/ENR/OMMT (Figure 6 (b), (c)) resulted from the formations of new O-H groups in the system. Moreover, the increasing intensity of C-O (alcohol) at ∼1027 cm−1 also explains the formation of more O-H (alcohol) functional groups due to graft esterification reaction. The slight decrease in N-H (amine) band between P85E15 and P85E15M4 also indicates the existence of hydrogen bonding between PA6, ENR and OMMT. Therefore, it can be inferred that the carboxyl groups at the end of PA6 macromolecules could react in situ with the epoxy groups of ENR and open the epoxy rings to form the PA6-g-ENR (as proposed in Figure 2) during processing. The formation of ester bonds improves interfacial adhesion of ENR with PA6 matrix.
Morphological analysis
XRD
XRD patterns of OMMT, PA6, PA6/ENR, PA6/OMMT and PA6/ENR/OMMT nanocomposites are shown in Figure 7. The (d001) diffraction in neat OMMT was registered at 2θ = 3.5°, which corresponded to a d-spacing of 2.5 nm (d-spacing and diffraction angle θ is related through Bragg’s relation in Equation 1). The d-spacing of the samples are tabulated in Table 3. As shown in Figure 7(a), the XRD patterns of PA6/OMMT nanocomposites did not show any characteristic peak of OMMT in the range of 2θ = 2–10°, which shows possible intercalation of OMMT platelets in PA6 matrix. Similar observations were reported by various studies 15,18,19,23 indicating that the absence of the corresponding characteristic peak of OMMT platelets at the range of 2θ = 1–3° evidences good dispersion. It is believed that the dispersion of OMMT in PA6 is due to the possible hydrogen bonding interactions between carbonyl groups of PA6 with hydroxyl and amine groups (in intercalant of OMMT) of OMMT as shown in Figure 2. Previous study by Chow et al. 12 also showed that good dispersion of OMMT is achieved in PA6/OMMT nanocomposites due to possible hydrogen bonding interactions. Naderi et al. 41 also reported similar observation.
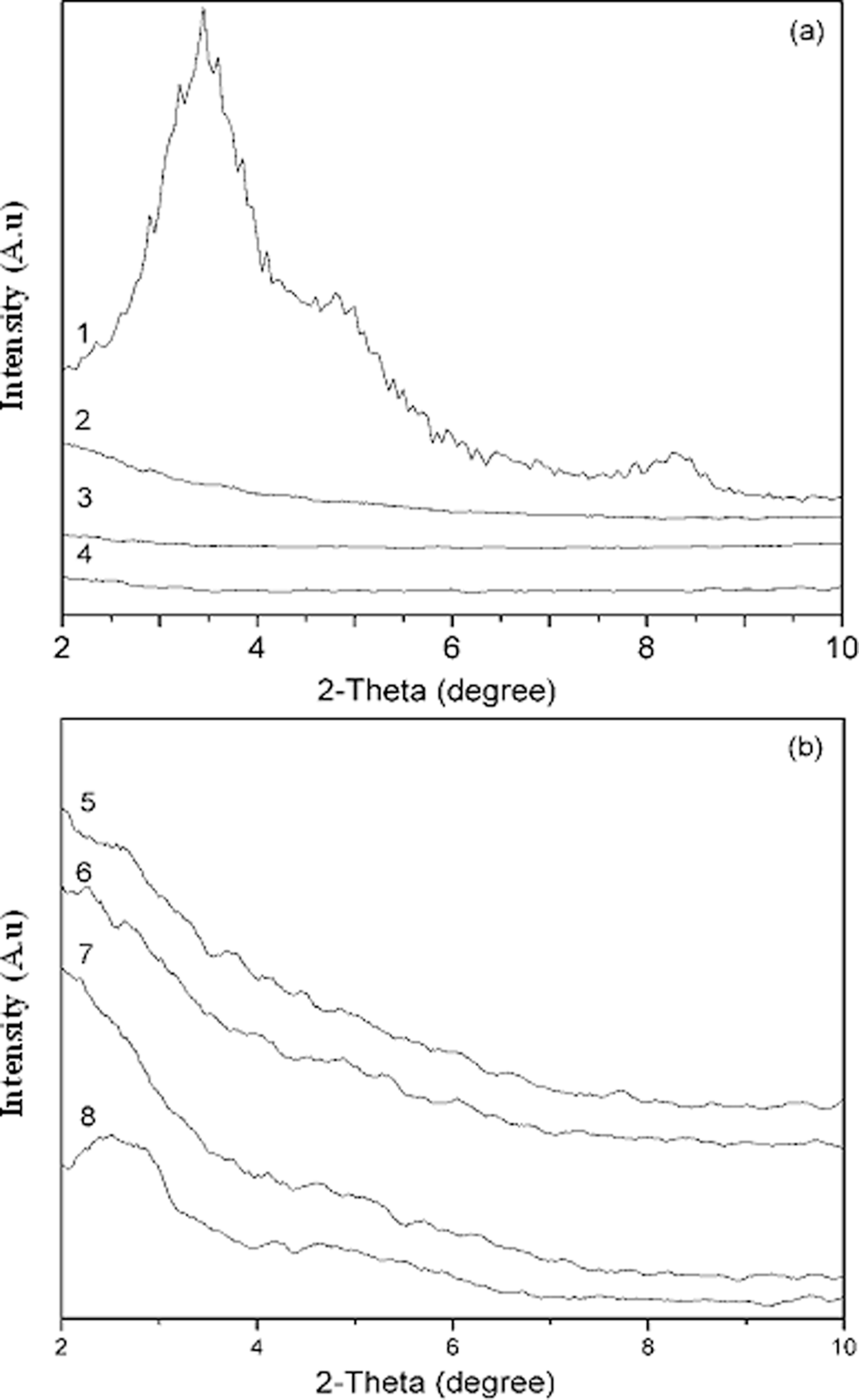
XRD patterns of (a) Pure components and blends: (1) OMMT (2) P85E15 (3) PA6M4 (4) PA6 and (b) PA6/ENR/OMMT nanocomposites: (5) P85E15M4 (6) P80E20M4 (7) P75E25M4 (8) P70E30M4. PA6: polyamide 6; OMMT: organically modified montmorillonite; ENR: epoxidized natural rubber; XRD: X-ray diffraction.
The 2θ angle and d-spacing of MMT for PA6/OMMT and PA6/ENR/OMMT nanocomposites.
Figure 7(b) showed the effect of increasing ENR content on the dispersion of OMMT in PA6/ENR/OMMT nanocomposites. The absence of (d001) peak in the range of 2θ = 1–3° clearly indicates that the OMMT platelets are well dispersed even in the presence of 15–25 wt% of ENR. However, a broad peak at 2θ = 2.5° (d-spacing = 3.5 nm) appeared at XRD diffractograms of PA6/ENR/OMMT nanocompsotites with 30 wt% ENR content is attributed to the agglomeration of OMMT platelets as previously reported by Tjong. 35
SEM
Figures 8 and 9 show the SEM micrographs of PA6, PA6/ENR and its nanocomposites at various ENR contents and the average particle size are shown in Figure 10. From Figure 8(c), it can be observed that the PA6/ENR exhibited a rough fracture surface with two phase morphology where the holes and knobs show the etched ENR phase. Figure 8 (c), (d) demonstrates good distribution of ENR in PA6/ENR blend and PA6/ENR/OMMT nanocomposites. From Figure 10, it can be seen that the average particle size of ENR (with the same ENR content) in PA6 has slightly increased with the addition of 4 phr OMMT.

SEM images of (a) PA6 (b) PA6/OMMT (c) P85E15 (d) P85E15M4. PA6: polyamide 6; OMMT: organically modified montmorillonite; ENR: epoxidized natural rubber; SEM: scanning electron microscopy.

SEM images of PA6/ENR/OMMT nanocomposites with various ENR contents; (a) 15 wt% (b) 20 wt% (c) 25 wt% and (d) 30 wt%. PA6: polyamide 6; OMMT: organically modified montmorillonite; ENR: epoxidized natural rubber; SEM: scanning electron microscopy.
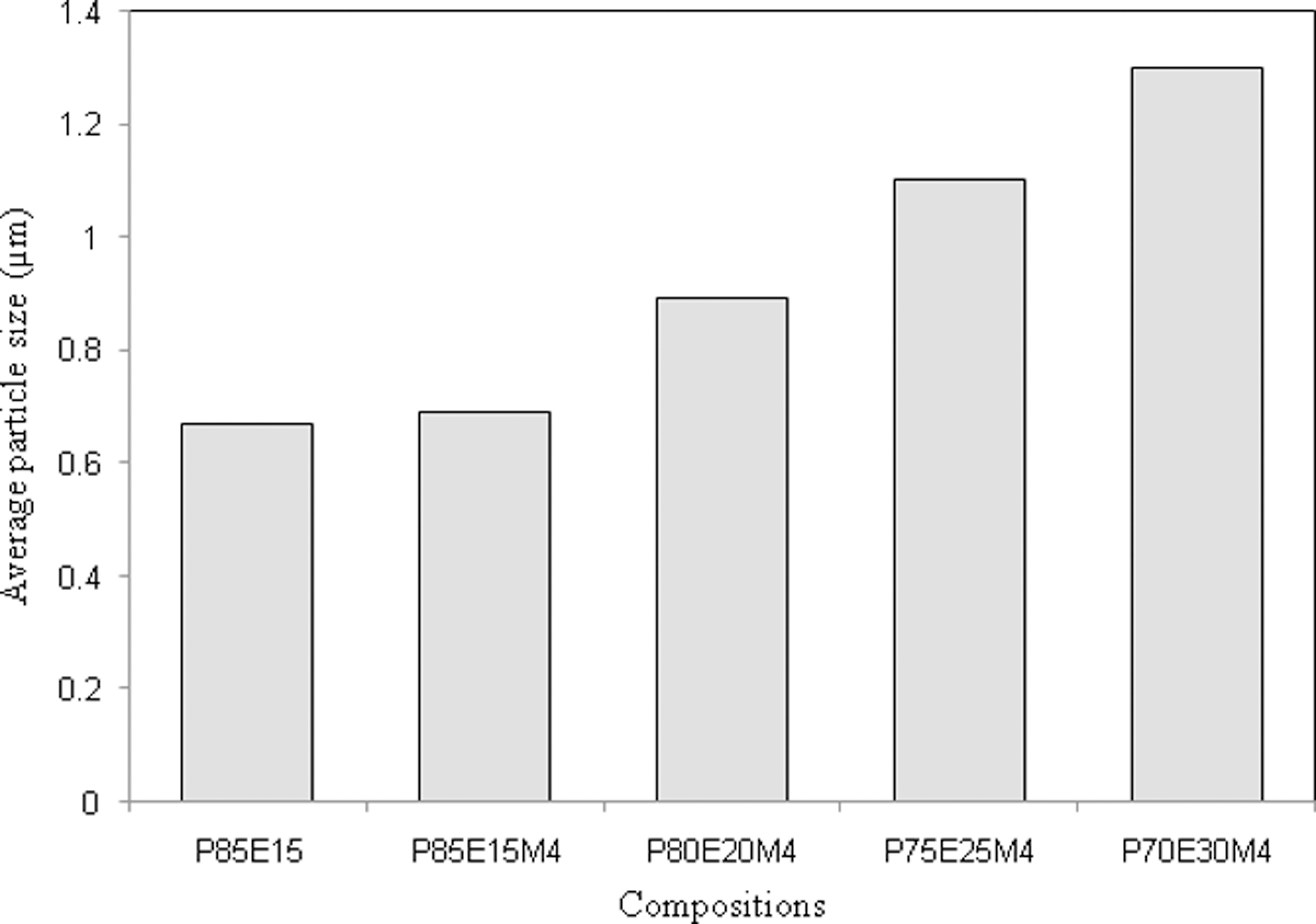
The average particle size of ENR in PA6/ENR and PA6/ENR/OMMT nanocomposites. PA6: polyamide 6; OMMT: organically modified montmorillonite; ENR: epoxidized natural rubber.
The SEM images of PA6/ENR/OMMT nanocomposites with various ENR content (15–30 wt%) are shown in Figure 9. The average particle size of ENR increased with increasing ENR content as observed in Figure 10. Previous study by Gonzalez et al. 15 also observed an increase in rubber’s particle size by blending up to 40 wt% of mSEBS with PA6/OMMT nanocomposites. The drop in impact strength and elongation at break values at 25 and 30 wt% of ENR content (Figure 1) can also be explained with the increase of average particle size of ENR (Figures 9 and 10).
Thermal analysis
DSC
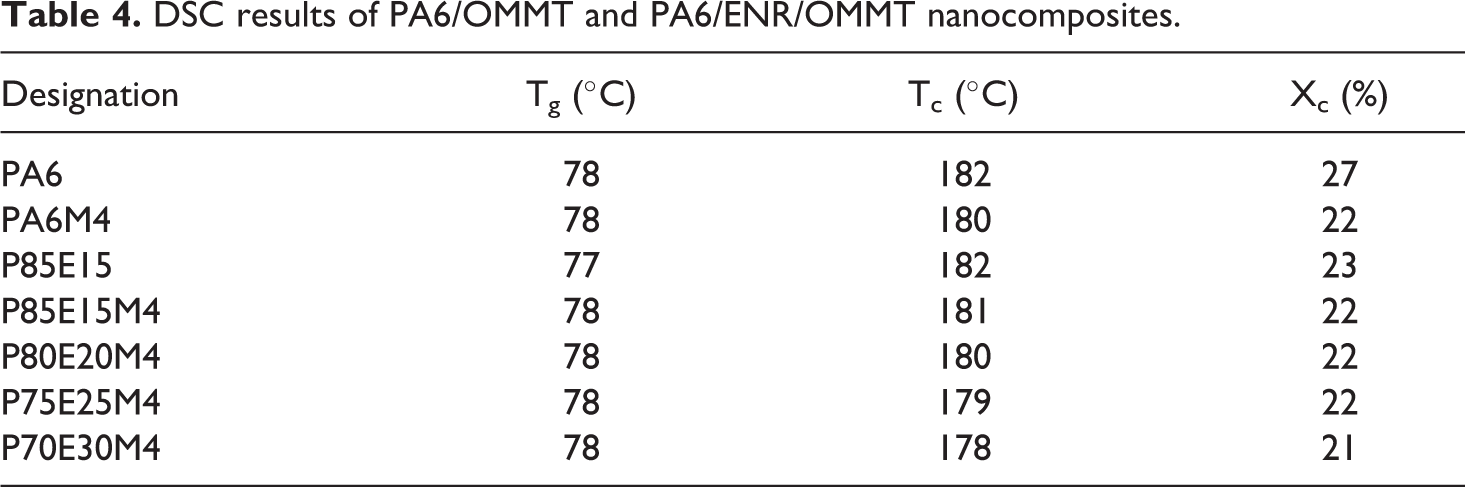
The glass transition temperature (Tg), crystallization temperature (Tc) and percentage of crystallinity (Xc) obtained from DSC analysis for PA6, PA6/ENR and their nanocomposites are shown in Table 4. The Tg of PA6 remained unaltered, while Tc and Xc values reduced slightly with addition of OMMT and ENR. It is believed that the graft esterification between PA6 and ENR at PA6-ENR interface, along with hydrogen bonding interactions inhibited the crystal formation. The increased interactions restrained the mobility of PA6 chains during the crystallization process resulting in the formation of fewer crystalline regions. Previous work by Ashabi et al. 42 also reported similar observation. Tseng et al. 43 showed that the presence of such matrix–rubber interphase interactions decreased the degree of crystallinity of PA6. The study by Tjong et al. 44 on PP/mSEBS/OMMT nanocomposites also found that the incorporation of mSEBS into PP and PP/OMMT nanocomposites reduced the degree of crystallinity of the matrix. However, it should also be noted that the sizes of individual crystal domains in polymer blends are limited in contrast with polymers in bulk which may have contributed to the overall drop in crystallinity.
DSC results of PA6/OMMT and PA6/ENR/OMMT nanocomposites.
Conclusion
The enhancement in impact strength and elongation at break with blending of ENR (<20 wt%) shows that the PA6/OMMT nanocomposites were successfully toughened with ENR. The stiffness and strength were decreased with blending of ENR and the incorporation of OMMT created a balance between mechanical properties of the ENR (20 wt%) toughened PA6/OMMT nanocomposites. The possible use of ENR as impact modifier for PA6/OMMT nanocomposites was explored.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
