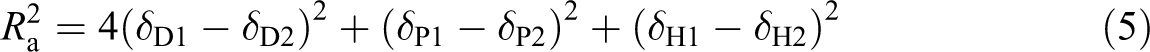Abstract
In this article, Hansen solubility parameters (HSP) have been used to evaluate the dispersion state and interfacial strain transfer ability of purified, nitric acid-functionalized and octadecylamine-functionalized single-walled carbon nanotubes (SWNTs) as the reinforcing materials in a poly(vinylidene fluoride) (PVDF) matrix. Composites were prepared by solution blending of PVDF and SWNTs and injection molding. The observations of the dispersion were obtained by dynamic light scattering and light optical microscopy and compared with the HSP results. The interfacial strain transfer between the polymer matrix and the carbon nanotubes in SWNTs/PVDF composite was studied by measuring the shift of the Raman two-dimensional peak position under tension. It was found that the strain transfer is affected by the degree of homogeneity of the fillers in the composites, the carbon nanotube bundle size and the affinities between two materials.
Introduction
Single-walled carbon nanotubes (SWNTs) are promising materials as polymer reinforcements in composite materials. 1 –3 However, due to the high aspect ratio of SWNTs and their tendency to aggregate into bundles, the dispersion of carbon nanotubes is a major challenge. The mechanical performance of the composite materials depends not only on the properties of the matrix and the reinforcing fillers but also on the surface adhesion between the matrix and the filler. The modification of SWNT affects the adhesion characteristics between the SWNTs and the matrix.
Many experimental studies have been carried out to investigate the dispersion of various modified SWNTs 4 –6 and the SWNTs/matrix interfacial adhesion, 7 –9 in order to improve the mechanical properties of nanocomposites.
The Hansen solubility parameter (HSP) method is a convenient and effective way in predicting compatibility between two materials 10 ; materials with similar HSPs will show good physical affinities in relation with the surfaces, thus improving the dispersion and adhesion. There are plenty of practical applications of HSP, such as aiding in solvent selection in different polymer systems, characterization of polymer blends and additives and predicting the dispersion of solutes in solvents. 11 –14 A few works have been done on SWNT/polymer composites using HSP so far. 13,15
In this work, poly(vinylidene fluoride) (PVDF) is used as the polymeric matrix. PVDF is used in a wide range of applications due to its good corrosion resistance and good mechanical properties. Furthermore, it has piezoelectric properties making it suitable for actuators and sensors. 16 –18 In this respect, adding SWNT to PVDF changes the electrical characteristics and also increases the electromechanical response. 19 –26 In this study, solution blending was used as the manufacturing method in the process of the composites and purified SWNTs; nitric acid (HNO3)- and octadecylamine (ODA)-functionalized SWNTs were used as the fillers. In order to get well dispersion of SWNT in the PVDF matrix, the SWNT have to be stably dispersed in the solvent, the polymer should be dissolved in the solvent and the physical affinity between SWNT and PVDF need to be strong. In this article, we propose using HSP to predict the dispersion and adhesion of SWNT with or without surface modification in a PVDF matrix. If the HSP method is successful in predicting the dispersion and adhesion properties, it might be used as a general method to help combining the polymer with the right modification and solvent and thus reducing work load.
Hansen solubility parameters
In Hansen’s approach to solubility parameters, the total cohesive energy E of a liquid is divided into three separate parts: the nonpolar dispersion interactions E
D, permanent dipole–permanent dipole interactions E
P and the hydrogen bonding interactions E
H, respectively, as shown in equation (1). E is determined experimentally by measuring the energy required to evaporate the liquid, and break all of its cohesive bonds in the evaporation process
Dividing the energy in equation (1) by molar volume V gives the energy densities in equation (2)
The solubility parameters are defined as the square root of the energy densities. δ
t is the total solubility parameter also known as the Hildebrand solubility parameter,
27,28
and δ
D, δ
P and δ
H are the dispersion, polar and hydrogen bonding solubility parameters, respectively, in equation (3)
Solubility properties can be visualized using a spherical graphical presentation for a solute and solvents. As shown in Figure 1, the HSP coordinates of the solute are at the center of the sphere and the radius of the sphere R
o indicates the maximum difference in the affinity tolerable for solution to take place. The solvents are divided into two groups depending on the interaction with the solute; good solvents are within the sphere and bad ones are placed outside of it. The relative energy difference (RED) number is defined as the distance according to equation (4) Classical method for Hansen solubility parameters (black dot: good solvent; white circle: bad solvent).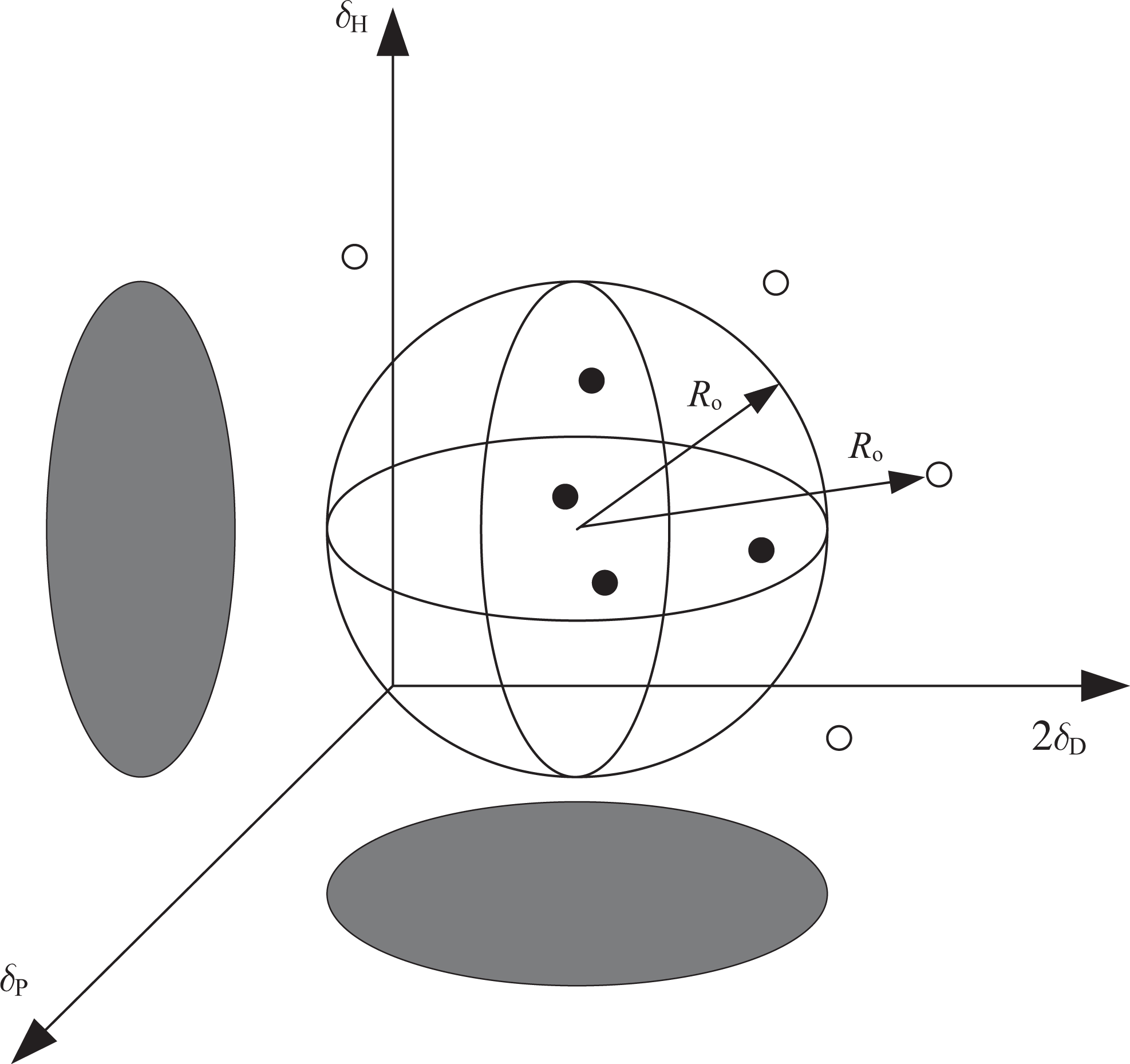
where R
a is the distance between the solvent and the solute and is calculated as equation (5)
The RED values for good solvents are normally below 1.0, while bad solvents will have RED values higher than 1.0.
The calculation procedure of HSP and RED values is presented in a previous publication. 29
Interfacial adhesion
The interfacial adhesion between the PVDF and the SWNTs was evaluated by measuring the Raman second order of the D peak so called 2D peak shift under tension. Raman spectroscopy has proven to be a powerful tool in the carbon nanotube research.
30
It has been used to characterize the nanotube structure, such as to determine the diameter of nanotubes, distinct metallic and semiconducting SWNTs and test the purity of the SWNTs.
31
Furthermore, the Raman bands are found to shift in position and intensities at a mechanical deformation of the composites, which can be used to evaluate the degree of the interfacial adhesion between the matrix and SWNTs. Especially, the position of the second-order Raman 2D band is sensitive to the mechanical deformation of the carbon nanotubes. At a low strain, in the elastic regime, the data can approximately be fitted by a linear relationship. At a larger strain, the Raman response becomes insensitive to strain, due to weakening of the interfacial adhesion as the polymer chains possibly slide at the nanotube interface. An empirical linear relationship exists between the SWNT 2D wave number shift and the applied elastic strain.
7,32,33
If the SWNT 2D wave number difference between zero strain and the applied strain (ε) is defined as the Raman wave number shift
For composites, good reinforcement requires a sufficient strain transfer from polymer matrix to filler. 34 The maximum shear stress is limited by the interfacial adhesion. The mechanisms of strain transfer from a matrix to filler can be summarized as micromechanical interlocking, chemical bonding or weak van der Waals bonding between the carbon nanotubes and the matrix 35 as well as residual compressive stresses on the nanotubes. 36,37
Experimental
Materials and nanotube composites preparation
The nanotube composites were produced by Carbolex Grade A SWNTs and a commercial Sigma-Aldrich 427144-100G PVDF supplied from Sigma-Aldrich Co. (MO, USA).
SWNT purification
The nanotubes were initially purified through annealing in the air at 275°C for 1 h and followed by refluxing in 6 M hydrochloric acid for 6 h to remove the amorphous carbon and the metal catalysts particles. The suspension was washed with deionized water 5–10 times and filtered through 0.45 µm PVDF-membrane filter.
SWNT functionalized by HNO3
Purified SWNTs of 0.5 g were refluxed in 50 ml 5 M HNO3 for 1 h. As shown in Figure 2, the purified SWNTs were cut shorter by the HNO3 reflux and leave the SWNTs open ended, the ends being decorated with oxygenated functionalities such as carboxylic acids. 38,39
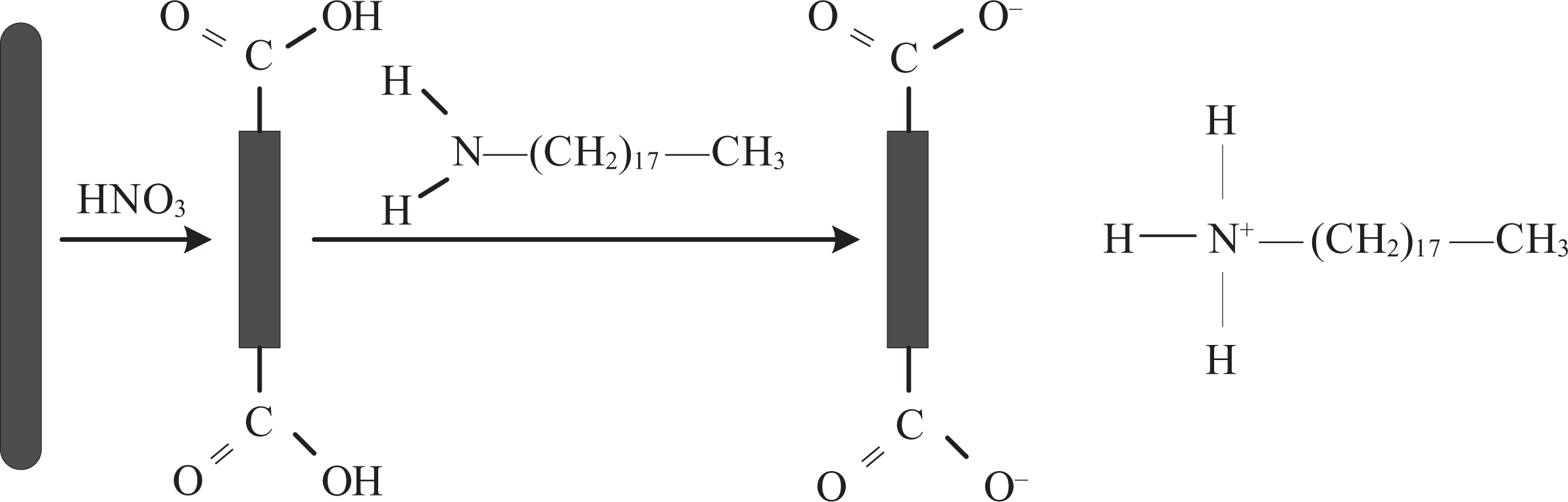
Schematic depiction of SWNTs treated by HNO3 and ODA. SWNT: single-walled carbon nanotube; HNO3: nitric acid; ODA: octadecylamine.
SWNT functionalized by ODA
HNO3-treated SWNTs of 0.23 g was dried at 100°C for 18 h. ODA of 2.01 g was added, and the mixture was held at 125°C for 6 days, during this period, ODA melted and reacted with the nanotubes. Hereafter, 800 ml ethanol was added to dissolve the remaining ODA and the mixture was then ultrasonicated for 30 min and filtered through 0.45 µm PVDF-membrane filter. The filtrate was washed by ethanol in an ultrasonic bath and filtered again two times to make sure that all ODA was removed. ODA was used to directly react with shortened SWNT-COOH to form an octadecylammonium after the modification; a simple acid-based reaction 39 is shown in Figure 2. A proton-transfer has taken place to form an ionic bond between the nanotube and amine-group. Finally, the carbon nanotubes were dried at 100°C for 1–2 days.
Preparation of composites
The as-prepared carbon nanotubes were ultrasonically dispersed in N, N-dimethyl formamide (DMF) for 1.5 h using an 80 W ultrasonic rod generator in order to form a stable suspension, and an ice bath was used to keep the suspension cold during sonication. At the same time, PVDF was also dissolved in DMF by mechanically stirring at 60°C for 2 h. The carbon nanotube/DMF suspension was added to the PVDF solution, the solution was ultrahigh shear mixed for 30 min at the speed of 5000–6000 r/min. Afterward, the solution was poured into large trays to forms a thin layer, which was dried at 60°C for 8 h to completely remove the solvent DMF. Subsequently, dog bone test specimens (ISO 527-2) were injection molded (Thermo HAAKE Minijet II) at 260°C as the melt temperature, the mold was heated to 90°C, holding at 95 MPa for 10 s.
Characterization
In order to evaluate the HSP of the carbon nanotubes, small amount of SWNT powders were added to 17 different kinds of solvents with known HSP, the tested tubes were sealed and mixed for 24 h in an ultrasonic bath to form suspension, and then the suspensions were left for observation. After 48 h, the dispersion stability was determined based on the observation of the sedimentation and the swollen state of SWNTs.
HSP experiment is based on the observation of the interaction or absence of interaction between the studied materials and solvents with known HSP. 10,40 The difference in interaction is used to divide the solvents into two groups, ‘good’ and ‘bad’ solvents. Good solvents interact strongly with the material, while the bad solvents give little interaction. In this case, the degree of sedimentation is used to divide solvents into good or bad. Figure 3 shows the purified SWNTs in (a) DMF and (b) acetone in test tubes after sonication and 48 h stagnation. Purified SWNTs dispersed well in DMF but sedimented in acetone, and this is taken as the criteria to distinguish between the good or bad solvents. The data are processed by a Matlab program based on Hansen’s method 41 to determine the HSP and radius of the sphere R o for the tested SWNTs.

Purified SWNTs in (a) DMF and (b) acetone kept in quiescent state for 48 h after sonication. SWNT: single-walled carbon nanotube; DMF: dimethyl formamide.
The SWNT/DMF suspension (0.1 mg ml−1) after tip sonication was analyzed by Zetasizer Nano ZS from Malvern Instruments using dynamic light scattering (DLS) to characterize the dispersion of SWNTs in DMF. The particles in suspension are constantly moving due to Brownian motion; the Brownian motion speed is related to the particle size, which is defined by the Stokes–Einstein equation. The Zetasizer Nano ZS illuminates the particles with laser and measures the rate of fluctuations in the intensity of the scattered light. The software calculates the particle size according to the ISO standard 13321:1996(E). The average hydrodynamic particle diameter (Z-average) is calculated by the Zetasizer software by the determination of the rate of diffusion. Along with the Z-average, the polydispersity index (PDI) is calculated as an indicator of the particle size distribution, the PDI scale ranges from 0 to 1, with 0 being monodisperse and 1 being polydisperse. The dispersion of carbon nanotubes in PVDF matrix was characterized by light optical microscopy on thin composite sections, which were ground from the bulk dog bone specimens.
The composites were tested in a Raman spectrometer (Renishaw Invia Raman Microscope) using He-Ne laser (632.8 nm) focused through X5 objective lens on the sample, forming a laser spot about 20–40 µm in diameter. The small dog bone specimens were loaded in a custom-made tensile test rig placed on the microscope table. The laser polarization was parallel to the loading direction and an analyzer also parallel to the load direction was used – the so called VV geometry. The specimen was gradually loaded up to different strain levels and Raman spectra were taken at each strain level.
Results and discussion
HSP results
In Table 1, the HSP results are depicted for the purified SWNTs, HNO3-functionalized SWNTs, ODA-functionalized SWNTs and PVDF, which were calculated based on the experimental data given in Tables 2 to 4 and reference data of Bottino et al. 42 In Table 2 to 4, the RED values have been calculated and as can be seen, the calculated RED values do not, in all cases, reflect the experimental observations. This is because it is impossible to create a sphere that includes all the good solvents and exclude all the bad solvents; anyway the program gives the optimal result.
HSP, radius of interaction R o of purified SWNT, 5 M HNO3-functionalized SWNT, ODA-functionalized SWNT and PVDF.
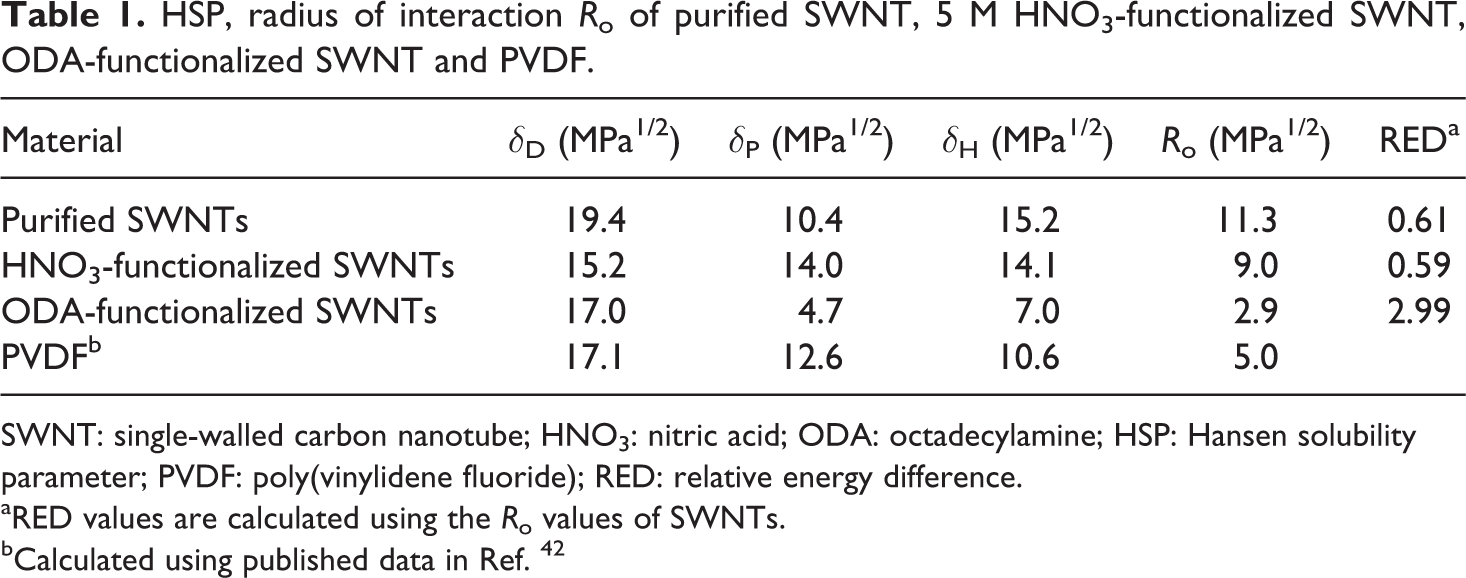
SWNT: single-walled carbon nanotube; HNO3: nitric acid; ODA: octadecylamine; HSP: Hansen solubility parameter; PVDF: poly(vinylidene fluoride); RED: relative energy difference.
aRED values are calculated using the R o values of SWNTs.
bCalculated using published data in Ref. 42
Dispersion of purified SWNTs in various solvents.
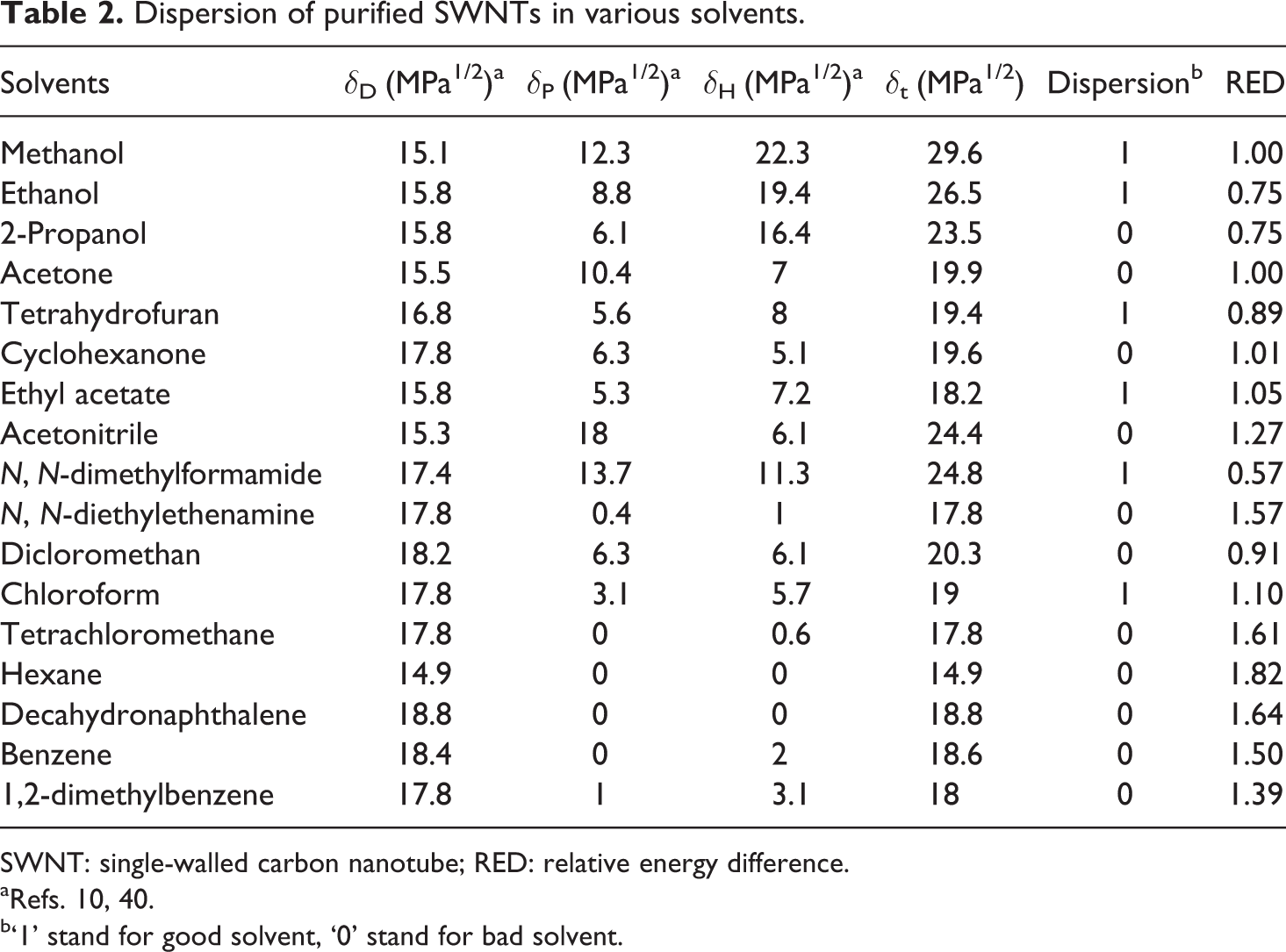
SWNT: single-walled carbon nanotube; RED: relative energy difference.
aRefs. 10, 40.
b‘1’ stand for good solvent, ‘0’ stand for bad solvent.
Dispersion of HNO3-functionalized SWNTs in various solvents.
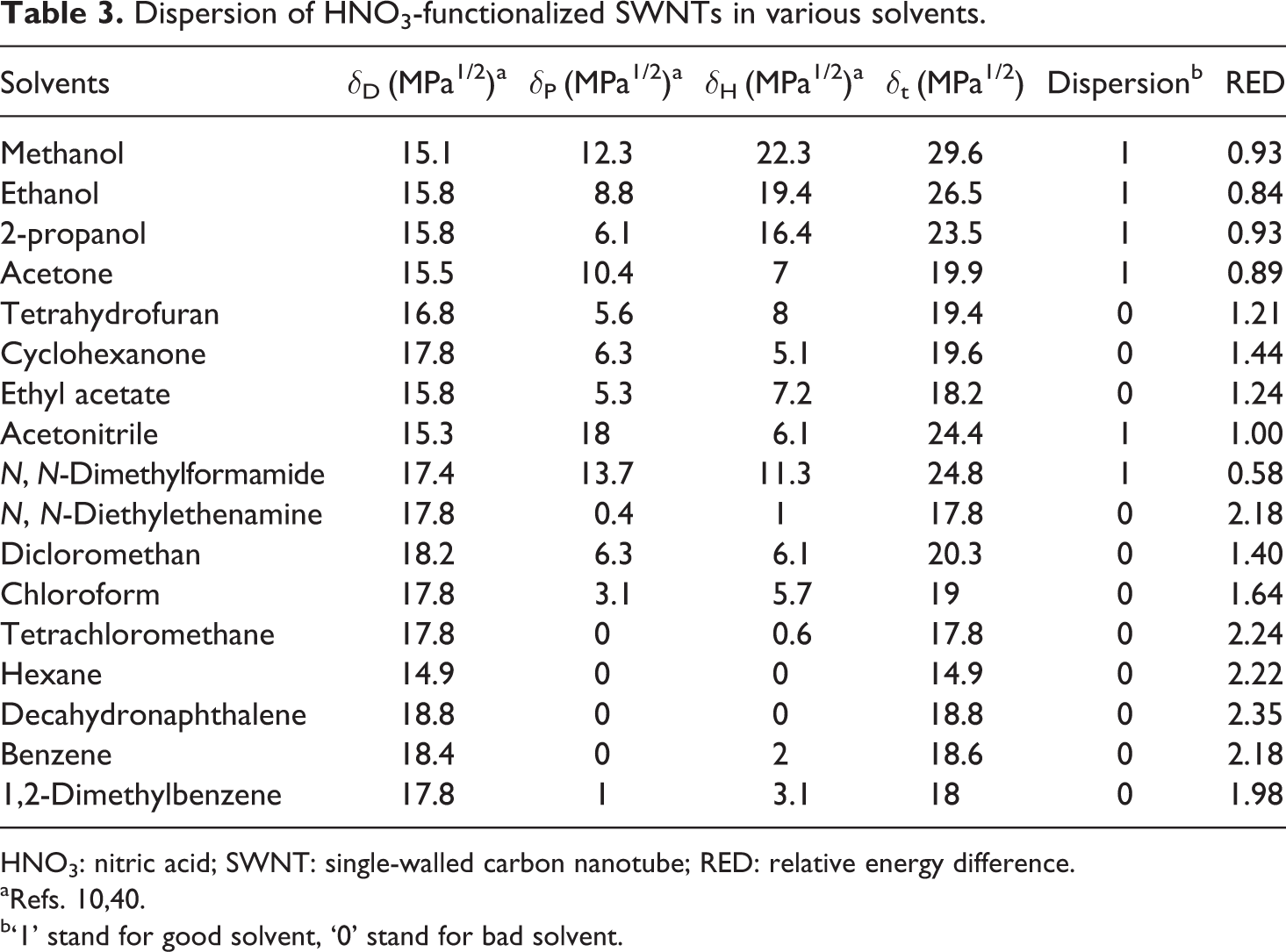
HNO3: nitric acid; SWNT: single-walled carbon nanotube; RED: relative energy difference.
aRefs. 10,40.
b‘1’ stand for good solvent, ‘0’ stand for bad solvent.
Dispersion of ODA-functionalized SWNTs in various solvents.
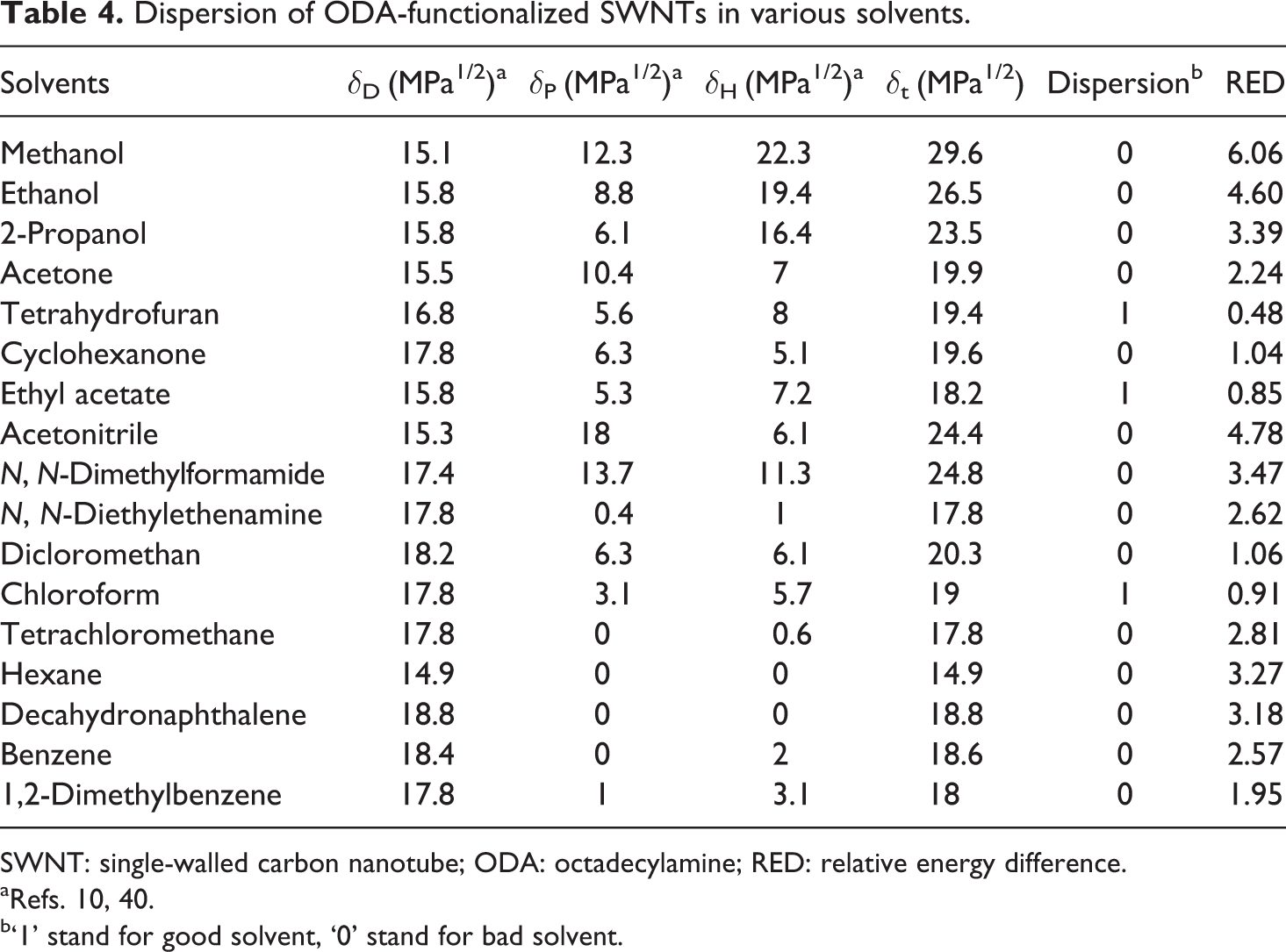
SWNT: single-walled carbon nanotube; ODA: octadecylamine; RED: relative energy difference.
aRefs. 10, 40.
b‘1’ stand for good solvent, ‘0’ stand for bad solvent.
DMF was chosen as the solvent during the processing, which is a good solvent for PVDF. 42 Purified SWNTs and HNO3-functionalized SWNTs are also dispersed well in DMF in contrast to ODA-functionalized SWNTs, which are not well dispersed in DMF. In Table 1, the HSPs of PVDF are calculated as 17.1, 12.6 and 10.6 MPa1/2, respectively.
Here, we consider PVDF as a solvent for the SWNTs and the RED values are calculated using the R o values of SWNTs. As can be seen in Table1, the RED values of purified SWNTs and HNO3-functionalized SWNTs in PVDF are 0.61 and 0.59, respectively. This indicates good physical affinities. However, the ODA-functionalized SWNTs possess higher RED value (2.99) and should exhibit poorer solubility in PVDF. Figure 4 shows the HSP spheres of the carbon nanotubes and the center of the solvents including PVDF. The location and the radius of the SWNT sphere change as the surface of SWNT is modified. The size of the ODA-functionalized SWNT sphere became very small indicating difficulties in interaction with most solvents and polymers. The position was also shifted toward reagents with less polar and hydrogen bond interactions. This seems naturally because of the nonpolar and hydrophobic nature of the attached amine with long aliphatic hydrocarbon chains sticking out from the nanotube. The ODA-functionalized SWNTs might be more compatible with polymers like polypropylene or polyethylene, that possess less polar and hydrogen bonding nature.
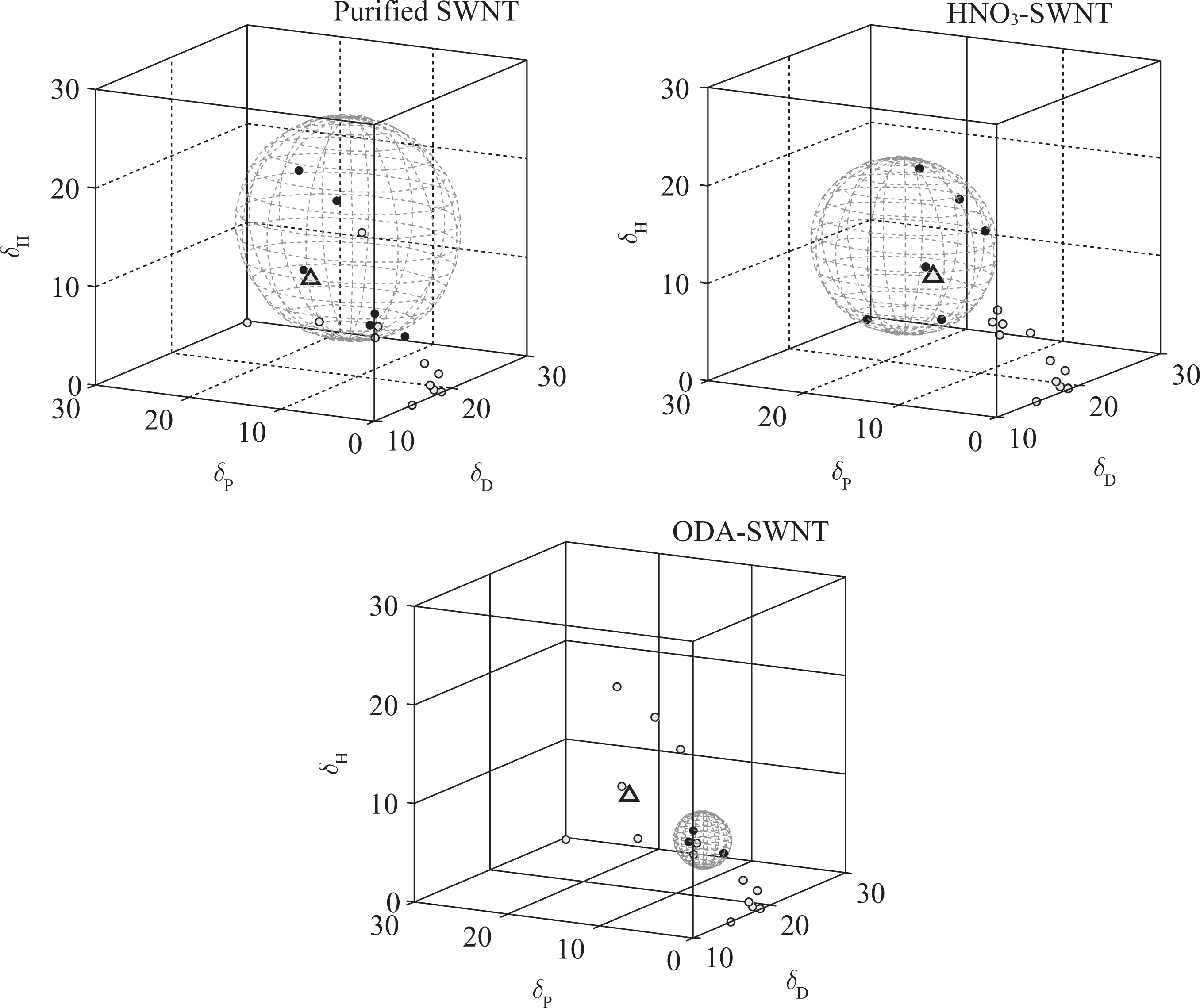
HSP spheres of various SWNTs with solvents (black solid dots: good solvent; gray dots: bad solvent; gray triangle: PVDF). SWNT: single-walled carbon nanotube; HSP: Hansen solubility parameter; PVDF: poly(vinylidene fluoride).
Dispersion state
Table 5 shows the DLS data for the three types of SWNTs in DMF solvent; the mean size of purified SWNTs, HNO3-treated SWNTs and ODA-modified SWNTs dispersed in DMF were 191.8, 175.8 and 328.5 nm, respectively. By comparing the particle size, the HNO3-treated SWNTs exhibited the smallest Z-average particle diameter and the most uniform distribution with very low PDI of 0.345. On contrast, the ODA-modified SWNTs dispersed in DMF had the largest Z-average diameter of 328.5 nm and the largest PDI at 0.495, which indicate the existence of large agglomerates and bundles.
Results for DLS measurement for various SWNTs in DMF solvent.
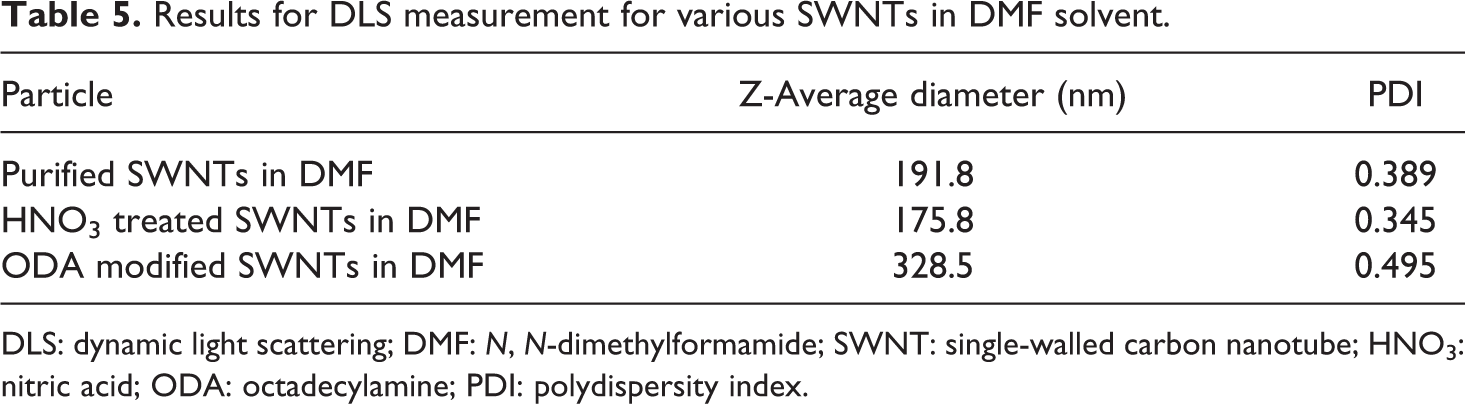
DLS: dynamic light scattering; DMF: N, N-dimethylformamide; SWNT: single-walled carbon nanotube; HNO3: nitric acid; ODA: octadecylamine; PDI: polydispersity index.
There is a reasonable agreement between the Z-average particle diameter and the RED values of SWNTs in DMF. This is especially true for the ODA-modified SWNTs. However, HNO3-treated SWNTs exhibits smaller particle size in DMF than purified SWNTs; this could be due to the debundling and cutting during the functionalization. 38
Figure 5 exhibits the optical micrographs of SWNTs/PVDF composite films containing 1 wt% purified SWNTs, HNO3-functionalized SWNTs and ODA-functionalized SWNTs, respectively. There are no obvious agglomerations of the nanotubes in Figure 5(a) and (b) with purified SWNTs and the HNO3-functionalized SWNTs/PVDF composites, shown in Figure 5(c) and (d), and the dispersions seem quite homogeneous. Numerous black spots, which attribute to nanotube agglomerates, appear on the film with ODA-functionalized SWNTs, shown in Figure 5(e) and (f). The observations of dispersion agree very well with the predictions obtained by HSP method.

The light optical micrographs of SWNTs/PVDF nanocomposite films at different magnification containing: (a) and (b) 1 wt% purified SWNTs; (c) and (d) 1 wt% HNO3-functionalized SWNTs; (e) and (f) 1 wt% ODA-functionalized SWNTs. SWNT: single-walled carbon nanotube; HNO3: nitric acid; ODA: octadecylamine; PVDF: poly(vinylidene fluoride).
Strain transfer
HNO3 treatment of SWNTs is known to cause debundling, 38 and this debundling is also evidenced by the SEM images published previously. 43 The debundling is expected to improve the strain transfer between the matrix and the nanotubes.
Figure 6 shows the strain induced 2D Raman shifts in SWNT/PVDF composites under tension. The slope in the elastic regime for purified SWNTs in PVDF is −30.4 cm−1 per strain, whereas it is −78.6 cm−1 per strain for HNO3-functionalized SWNTs and −47.7 cm−1 per strain for ODA-functionalized SWNTs in PVDF composites.
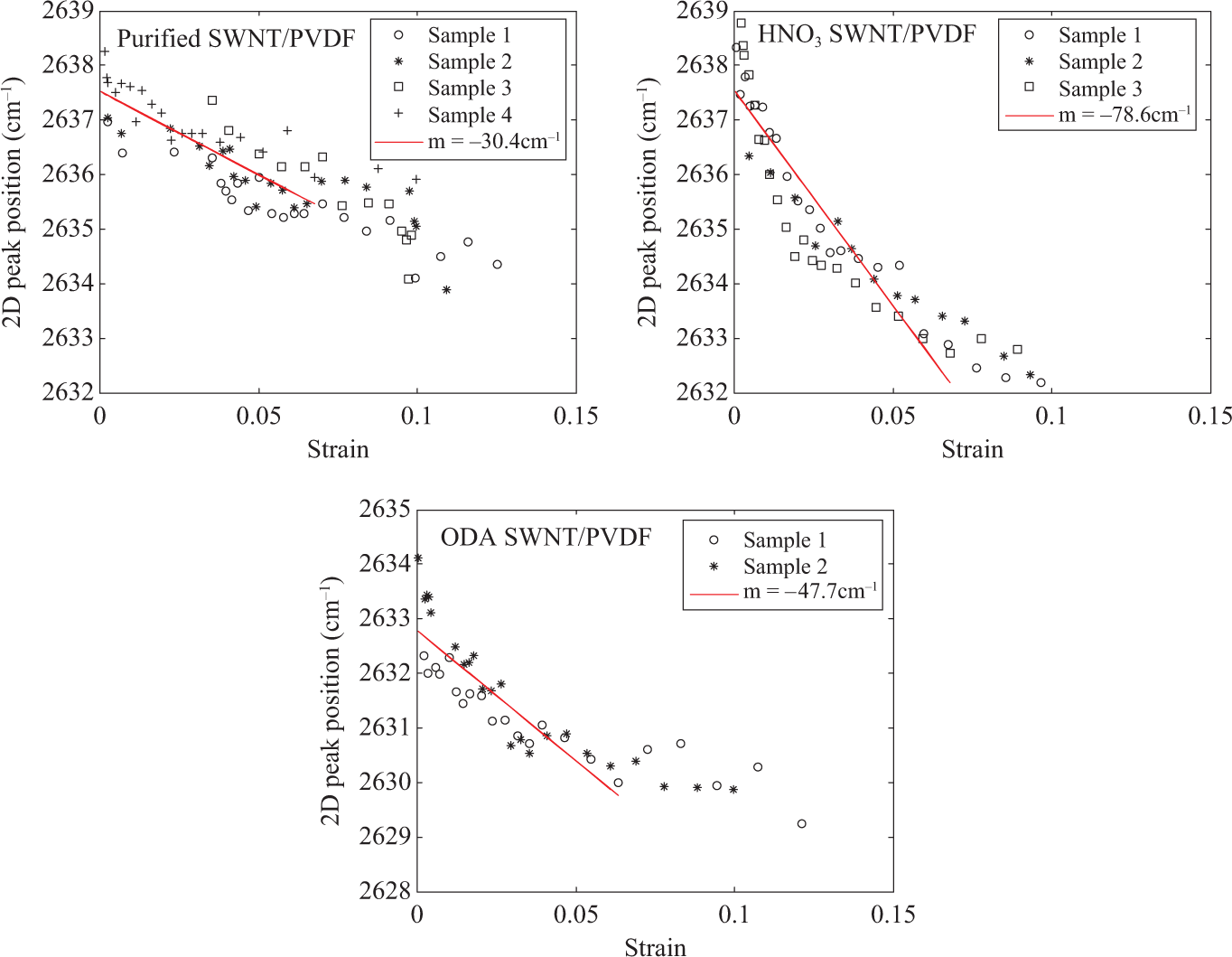
The strain response of the 2D peak position of PVDF composites filled with various nanotubes. PVDF: poly(vinylidene fluoride); 2D: two-dimensional.
Looking at the maximum shift of 2D peak position, it is largest (∼6 cm−1) for the HNO3-functionalized SWNT, whereas it is 3.5 and 3 cm−1 for the purified and ODA-functionalized SWNT, respectively. Only for the ODA-functionalized SWNT, a steady-state is observed, which is an indication of poorer interface.
The variation of the Raman shift between purified SWNTs/PVDF and functionalized SWNTs/PVDF could be explained by different bundle sizes and also by the inferior compatibility as indicated by HSP. The small Raman shift of the purified SWNTs/PVDF is due to slipping of the individual SWNTs within the large bundles and the average strain of the nanotubes is thus decreased. The HNO3 functionalization debundles the nanotube bundles 38 and forms carboxyl groups at free ends of SWNTs and defect sites at the surface. Although isolated SWNTs are not achieved, the slippage between the individual carbon nanotubes is minimized. The carboxyl groups could form hydrogen bonds with the PVDF chains, which increase the interfacial adhesion and help the stress-transfer from PVDF to the SWNT. The ODA-functionalized SWNTs show less strain transfer in the PVDF composites perhaps mainly due to the inferior dispersion state of the carbon nanotubes. On the other hand, it could also be related to the poor physical affinity between the ODA-functionalized nanotube surface and the PVDF matrix, which the HSPs demonstrates.
Conclusions
HSP could be used to help selecting solvent for composites processing, and predicting the dispersion state and interfacial properties of SWNTs in PVDF polymer matrix. Purified and HNO3-functionalized SWNTs dispersed well in DMF and PVDF, while ODA-functionalized SWNTs dispersed poorly, which were in agreement with the compatibility predicted by HSP.
The stronger interfacial adhesion is related to both absence of agglomerates and debundling of the carbon nanotubes. The SWNTs functionalized by HNO3 in PVDF show improved strain transfer compared with the purified SWNTs, this could be explained by the debundling of the SWNTs caused by the HNO3 treatment, while the ODA-functionalized SWNTs exhibits less strain transfer, which also agree well with the HSP prediction.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.