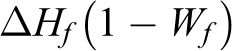Abstract
Polylactic acid (PLA) nanocomposites were prepared using melt blending technique. Two different commercially available nanoclays, Cloisite 93A (C93A) and Cloisite 30B (C30B) at various wt%, have been used to prepare the nanocomposites. The mechanical properties of the nanocomposites have been studied to evaluate the effect of the nanoclay within the matrix polymer. Mechanical tests revealed an increase in the mechanical properties with the incorporation of organically modified nanoclays as compared with PLA matrix. PLA/C30B nanocomposites exhibited optimum tensile modulus at 3 wt% of clay loading. Thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC) studies were also performed to evaluate the thermal behavior of the nanocomposites. Further, viscoelastic behavior in the nanocomposites has been investigated using dynamic mechanical analysis (DMA) to study the damping behavior of the materials. Also the morphological study was carried out through X-ray diffraction analysis, and a better exfoliation or dispersion of C30B clay with PLA matrix was observed. The interaction between the clays and PLA matrix has also been studied using transmission electron microscopy (TEM).
Keywords
Introduction
Over the past decades, polymer/clay nanocomposites have attracted great deal of research interests, both in industry and in academic because of their remarkable improvement in material properties when compared with virgin polymer or conventional micro- and macrocomposites. 1 –7 These improvements include high moduli, increased strength and heat resistance, decreased gas permeability and flame retardency.
Recently, special attention has been paid to the development of nanocomposites based on thermoplastics and organically modified layered silicate organoclays such as montmorillonite (MMT), because of the nanoscale size layer thickness and the intercalation properties. 8 The achievement of exfoliation of these layered silicates in a polymer matrix is essential for preparing true nanocomposites with desired attributes.
There are predominantly two techniques for preparing the nanocomposites through intercalation process that is solution intercalation and melt intercalation, respectively. An effective hybrid formulation was conducted mostly via a solution intercalation. 9 But it takes a longer time for the completion of this technique and solvent removal has been a major impediment. However, melt intercalation has been recognized as a promising approach due to its environmental benign nature. Moreover, it is a more economically viable process. 10 Various commercially available thermoplastics such as styrenic polymers, 11,12 polyolefin, 13,14 nylons 15,16 and so on have been reported to be synthesized using melt blending technique.
Further, with the urgent need for reducing the environmental problems caused by conventional plastic wastes, biodegradable polymers have received much attention in the recent years. Clay materials obtained from renewable resources are also environmentally friendly, naturally abundant, and economical. Hence, the biodegradable polymers blended with organoclay can be a potential candidate in preparing biodegradable nanocomposites with improved properties as compared with the virgin polymers that can enlarge the application field of these materials for various end-use applications.
Recently, inherently biodegradable polyesters polylactic acid (PLA) has received considerable research attention.
17
–19
PLA is a linear aliphatic thermoplastic polyester, produced from renewable resources, with excellent properties comparable to many petroleum-based plastics and is readily biodegradable.
20,21
PLA is produced either by ring-opening polymerization of lactides or by condensation polymerization of the lactic acid monomers, and these monomers are obtained from the fermentation of sugar feedstock, corn, and so on.
22
Commercially available PLA grades are copolymers of poly (
In comparison to other biodegradable aliphatic polyester, such as poly (E-caprolactone) (PCL), poly (butylenes succinate) (PBS), and so on, PLA possesses a relatively high melting temperature. Its environmental friendly characteristics together with superior physical properties can be further modulated with various reinforcements and additives. These features can make PLA an attractive substitute for numerous biodegradable synthetic polymers to penetrate in rich markets. PLA-based nanocomposites have been studied extensively by various authors. Sinha Ray et al. 24 –27 studied various properties of PLA/clay nanocomposites prepared by melt intercalation. The authors have used oligo (E-caprolactone opel) as a compatibilizer for achieving improved performance characteristics. Similarly PLA reinforced with MMT and sepiolite has been investigated exclusively. Pulta et al. 28 also studied some structural and physical properties of PLA/clay nanocomposites and PLA/clay microcomposites prepared using the same melt intercalation technique.
Several authors have recently reported on the preparation and characterization of biodegradable polymer-based nanocomposite. Paul et al. 29 reported the preparation of PLA/MMT nanocomposites by melt intercalation technique using an MMT modified with bis-(2-hydroxyethyl) methyl (hydrogenated tallow alkyl) ammonium cations. Chang et al. 30 reported the preparation of PLA-based nanocomposites with three different kinds of layered silicates via solution intercalation method in N-dimethylacetamide, leading to the formation of intercalated nanocomposites whatever be the clay. In a recent report, Feijoo et al. 31 prepared biodegradable nanocomposites of amorphous PLA and two different types of organically modified montmorillonite obtaining nanocomposites with a morphology of stacked intercalated and partially exfoliated layers. In the present investigation, PLA nanocomposites have been prepared using melt blending technique. A detailed investigation on the effect of nanoclay on mechanical, dynamic mechanical (DMA), thermal and morphological properties of PLA nanocomposites has been reported.
Experimental
Materials
Polylactic acid, PLA 4042D nature work with a density of 1.24 g/cm3, is used. The resin was provided in the form of granules. Two types of organoclay under commercial names of Cloisite 93A (C93A) modified with methyl dehydrogenated tallow ammonium having a cation exchange capacity (CEC) of 90 meq/100 g clay and Cloisite 30B (C30B) modified with methyl bis-2-hydroxyethyl hydrogenated tallow ammonium having a CEC of 90 meq/100 g clay were supplied by Southern Clay Products Inc. (Gonzalez, Texas, USA).
Preparation of nanocomposites
Prior to processing, PLA was dried at 50°C for 6–8 h and the nanoclay was dried at 80°C for 8–10 h, respectively. PLA/organoclay nanocomposites were prepared in a 15-cc conical co-rotating twin screw extruder (DSM Xplore 15, the Netherland) using melt blending technique with barrel temperature of 170, 175 and 180°C with a screw speed of 40 rpm and acceleration rate of 50 rpm/min. Subsequently the melt mixes were transferred to the specimen moulds by the microinjection moulding machine with process parameters of 180°C temperature, 7–7.5 bar injection pressure and a cycle time of 10 s. The specimens were prepared as per the American Society for Testing and Materials (ASTM) D standard.
Mechanical properties
PLA nanocomposite sample specimens of dimension 167 × 12.7 × 3 mm were subjected to tensile test using universal testing machine (UTM, Instron 3386, UK) as per ASTM D-638, at a crosshead speed of 5mm/min and gauge length of 50 mm. Izod impact strength of the specimens of dimensions 63.5 × 12.7 × 3 mm has been measured as per ASTM D-256, and samples were notched with a “v” notch at a depth of 2.54 mm and notch angle of 45°, using Tinius Olsen Model 899 specimen notcher, USA. The impact test was carried out using Tinius Olsen Model IT504 Plastic Impact.
Thermal properties
The glass transition, melting and thermal stability of virgin PLA and PLA nanocomposite samples has been studied using differential scanning calorimetry (DSC; Perkin-Elmer Diamond DSC, USA) and thermogravimetric analyzer (TGA; Perkin-Elmer Pyris-7, TGA, USA) respectively.
Samples of ≤5 mg were heated from 30 to 200°C at a rate of 5°C/min under a nitrogen atmosphere. The sample was kept at 200°C for 1 min. Corresponding glass transition and melting temperature were determined from the temperature at the maximum values of glass transition and melting peaks.
The degree of crystallinity can be determined using the following equation:
where ΔHm = heat of fusion of sample; ΔHf = corresponds to the heat of fusion for 100% crystalline material; Wf = mass fraction of the filler. 32
The crystallinity of PLA in the composites was calculated where the heat of fusion of 100% crystalline PLA ΔHf was set equal to 93.6 (J/g). 33
Similarly, thermal stability of PLA and its nanocomposite samples have been studied using samples of 5–10 mg weight under an air atmosphere at a heating rate of 10°C/min from the room temperature to 500°C. The initial and final degradation temperature and the corresponding percentage weight loss for all the samples were noted.
Heat distortion temperature (HDT)
The HDT tests of PLA and PLA nanocomposites were conducted with the samples of dimension 125 mm × 12.7 mm × 3 mm with a HDT tester (M/s GOTECH HV-200-C3, Taiwan) according to the ASTM D-648 method at a heating rate of 2°C/min and load of 66 psi.
Dynamic mechanical properties
The dynamic mechanical analysis (DMA) was performed with a dynamic mechanical analyzer (DMA Q 800, M/s TA Instruments, USA). The experiments are carried out at a fixed frequency of 1 Hz and at a heating rate of 3°C/min. The tests were conducted in a three-point bending mode at a temperature range of 30 to 120°C using a specimen of dimension 40 mm × 10 mm × 3 mm.
Transmission electron microscopy (TEM)
The nanoscale morphology of PLA nanocomposites has been studied using TEM (JEOL-1200EX, Japan). Samples for TEM imaging were sectioned using Leica ultracut UCT microtome at room temperature, using a diamond knife (M/s Leica, UK). Sections of 50–70 mm thickness were collected from water on 300-mesh carbon-coated copper grids and kept overnight on filter paper for drying. Subsequently, TEM of the sections was carried out using the electron microscope at an accelerating voltage of 100 KV.
Wide angle x-ray diffraction (WAXD)
X-ray diffraction (XRD) analysis was carried out using XRD-7000, M/s Shimadzu, Japan, which have a monochromator and a Cu radiation source of wave length 1.54 Å operating at 40 kV and 30 mA. The basal spacing or d-spacing (d 001) reflection of the clay gallery and the PLA-based nanocomposites was calculated using Bragg’s law equation (i.e. nλ = 2dsinθ) by monitoring the diffraction angle 2θ from 2° to 10° at a scanning rate of 2°/min.
Results and discussion
Mechanical properties
The variation in mechanical properties of PLA and its nanocomposites as a function of clay loading and clay type is enumerated in Table 1. It is evident that the incorporation of organoclay to the tune of 1 to 5 wt% results in increased tensile strength and modulus. The tensile modulus of the nanocomposites increased from 3550 Mpa to 4156 and 3966 Mpa in case of PLA/C30B and PLA/C93A nanocomposites, respectively, with increasing nanoclay loading from 1 to 5 wt%. However, the tensile strength of virgin PLA shows a marginal increase from 38.22 Mpa to 39.8 Mpa in PLA/C30B and 40.7 Mpa in PLA/C93A nanocomposites at 3 wt% of clay loading. Test result depicted in Table 1 reveals that the tensile strength of nanocomposites is lower than that of virgin matrix at a lower clay content of 1 wt%, which subsequently increases with the increase in clay content to 3 wt%. The reduction in tensile strength at 1 wt% of clay loading probably due to the development of two phases of immiscibility between the matrix and clay as well as due to the improper reinforcement of the clay, which induces cracks. The optimum tensile strength observed at 3 wt% of clay loading due to the nanoparticles and better interfacial properties resulted in improved stress transfer at the interface. Further, beyond the percolation limit the additional clay loading in PLA has much less effect due to the additional silicate layers incorporated in the polymer regions that are already affected by other silicate layers. So at 5 wt% of clay loading the tensile strength of PLA nanocomposites started to decrease due to the agglomeration of clay particles, which induces cracks in the nanocomposites. In all the cases, the elongation at break decreased with filler loading as observed in the case of all other filled systems.
Tensile properties of PLA nanocomposites.
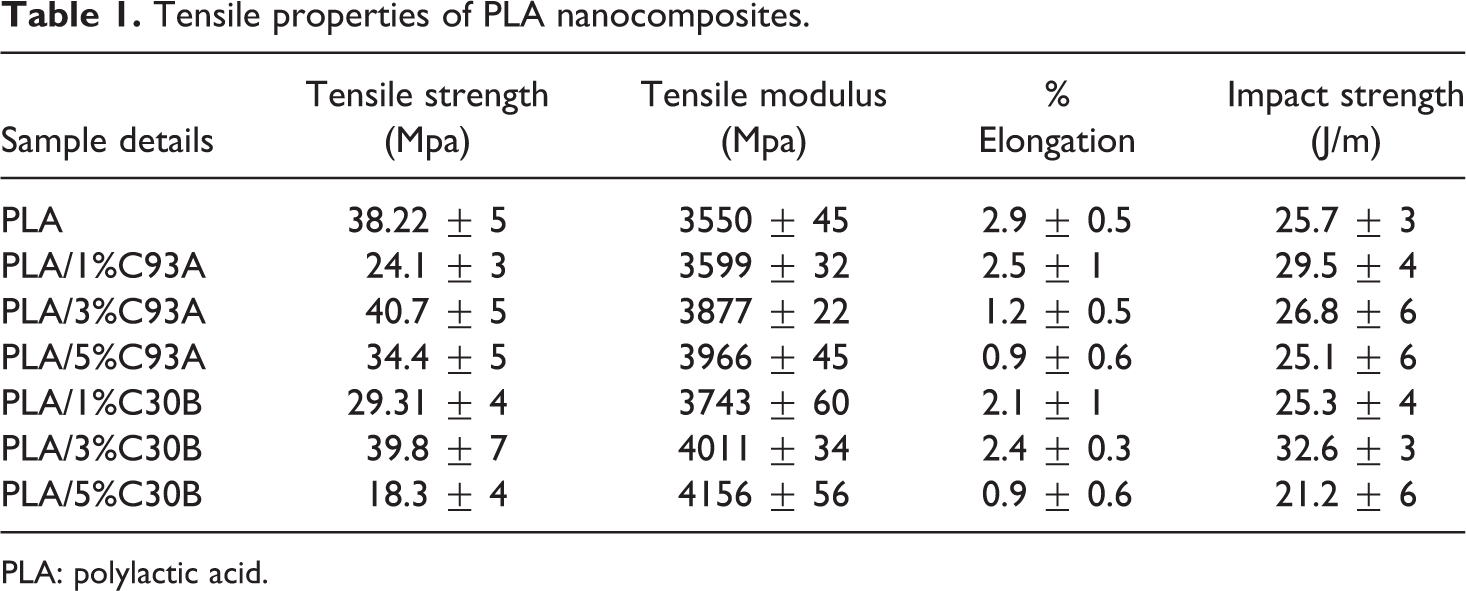
PLA: polylactic acid.
Similarly, the impact strength of the matrix polymer increased with the increase in the clay loading from 1 to 3 wt% in case of PLA/C30B nanocomposites beyond which there was a decrease in the strength. Impact strength increased to 26.84% in case of PLA/C30B nanocomposites at 3 wt% of clay loading. PLA/C93A nanocomposites show enhanced impact properties as compared to the PLA matrix upto a 3 wt% of C93A clay loading. On the contrary, the variation in impact strength observed in case of PLA/C93A follows decreasing trend to the tune of increased clay loading. From the data, it is evident that the nanocomposites prepared using C30B organoclay exhibit optimum mechanical performance vis-à-vis C93A, which confirms strong interaction between PLA matrix and C30B nanoclays. This is due to the interaction of carboxyl groups of PLA and hydroxyl ethyl groups of the intercalant methyl bis-hydroxy ethyl tallow (MT2EtOH) residing at the surface of silicate layers of organically modified clay C30B, enhancing the interfacial interaction that leads to improved capability and dispersion of the clays within the PLA matrix. Conversely, in case of C93A, the presence of two hydrogenated tallow (HT) groups and one methyl group in its intercalant imparts more hydrophobic characteristics of organoclay which is probably responsible for lesser interaction and its less compatibility within the polar PLA matrix. Also the PLA-based nanocomposites prepared using the organoclay having one alkyl tail (inC30B) shows a larger increase in basal spacing than those containing two alkyl tail (in C93A).
The detailed study of tensile properties elaborated through the stress–strain behavior of virgin PLA and the nanocomposites is represented in Figure 1. It is observed that virgin PLA shows a 3% strain. Incorporation of nanoclay induces brittleness within the polymer matrix. PLA/clay nanocomposite system undergoes brittle failure after reaching its ultimate tensile strength at a strain of about 2.5% and 1.5% in case of PLA/C30B and PLA/C93A nanocomposite system, respectively. The presence of nanoclay results in an increase in the ultimate tensile strength of the virgin PLA from 38 MPa to 39.5 MPa and 40.5 MPa for C30B and C93A nanocomposite systems, respectively. It is also observed that with C93A clay loading, the nanocomposite exhibited a very little elongation due to its brittle nature. Further, as observed from the stress–strain curve, the yield strength of the nanocomposites increases with the modulus. This indicates incorporation of clay into PLA to improve both stiffness and strength in an analogous manner.
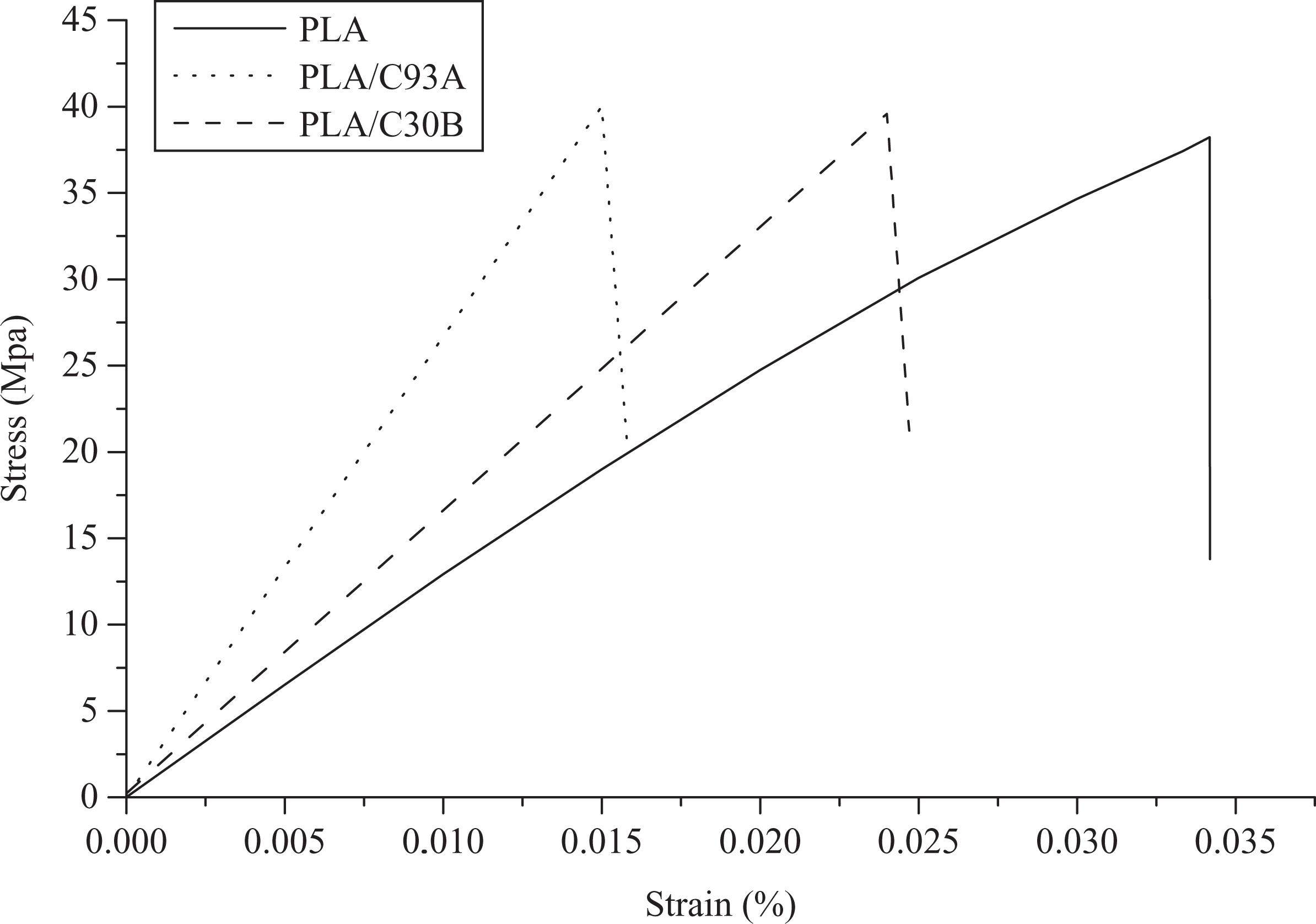
Stress–strain curve of PLA and its nanocomposites. PLA: polylactic acid.
Thermal properties
DSC
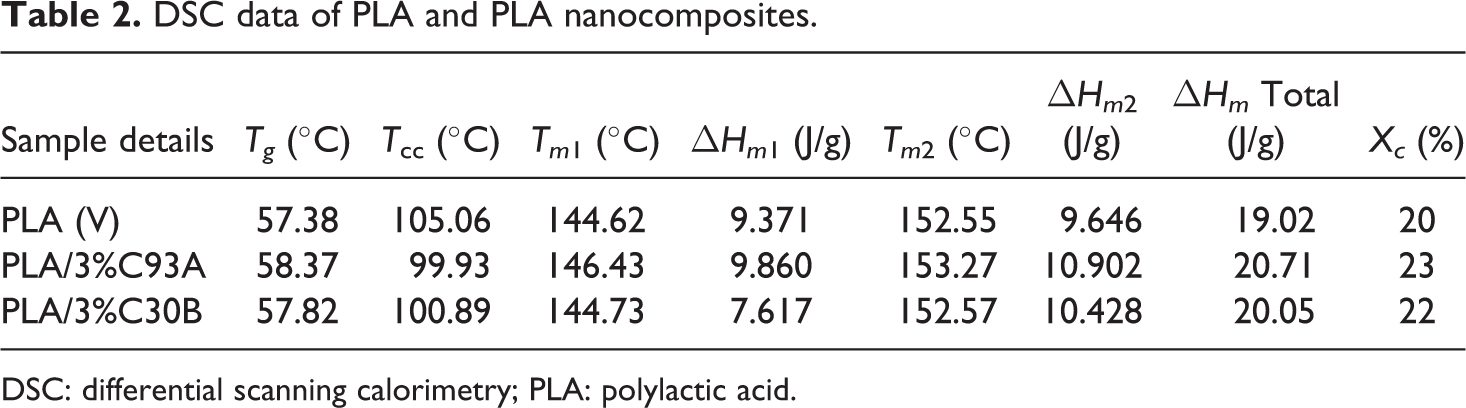
Figures 2 –4 depict the DSC thermograms of virgin PLA and its nanocomposites derived from the second heating scan. Corresponding glass transition (Tg ), melting temperature (Tm ), crystallization temperature (T cc) and enthalpy of fusion is also represented in Table 2. The measurements have been performed immediately after melt quenching scans to erase the previous thermal history in the samples.
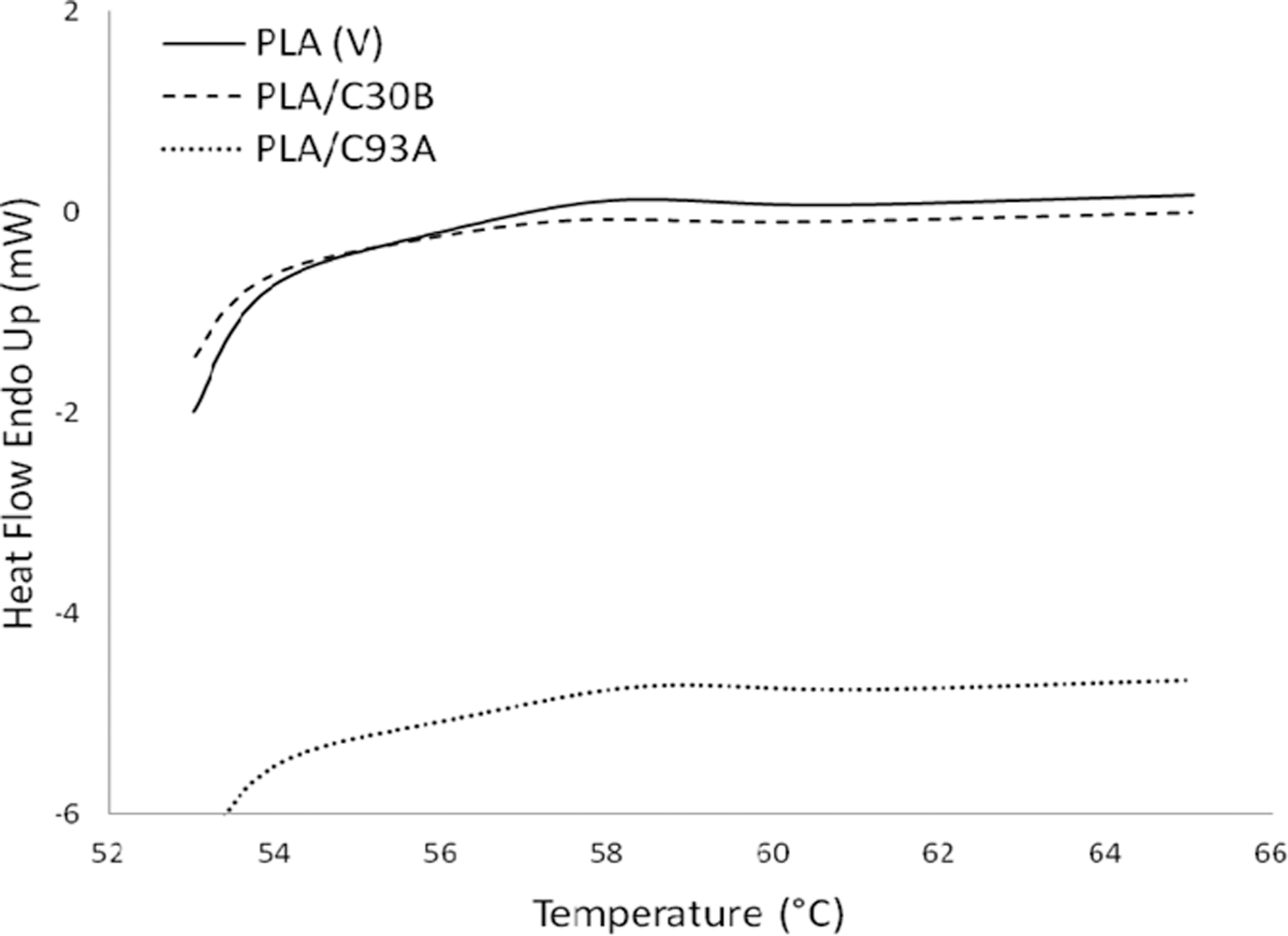
DSC curve for glass transition temperature (Tg ). DSC: differential scanning calorimetry.
DSC curve for cold crystallization temperature (T cc). DSC: differential scanning calorimetry.
DSC curve for melting temperature (Tm ). DSC: differential scanning calorimetry.
DSC data of PLA and PLA nanocomposites.
DSC: differential scanning calorimetry; PLA: polylactic acid.
As evident from Figure 2, the peak in the region at 57.38°C is attributed to the Tg of PLA. No significant changes in Tg was observed in the case of both PLA/C93A and PLA/C30B nanocomposites, and this finding is similar to those reported in reference. 35, 36, 38 Further, the evidence of step-like shape for Tg reveals the physical aging in the samples. 34 The small increase in Tg in case of the nanocomposites can be attributed to the restriction in the mobility of the polymer chains as a consequence of bonding or adsorbing on the clay surface.
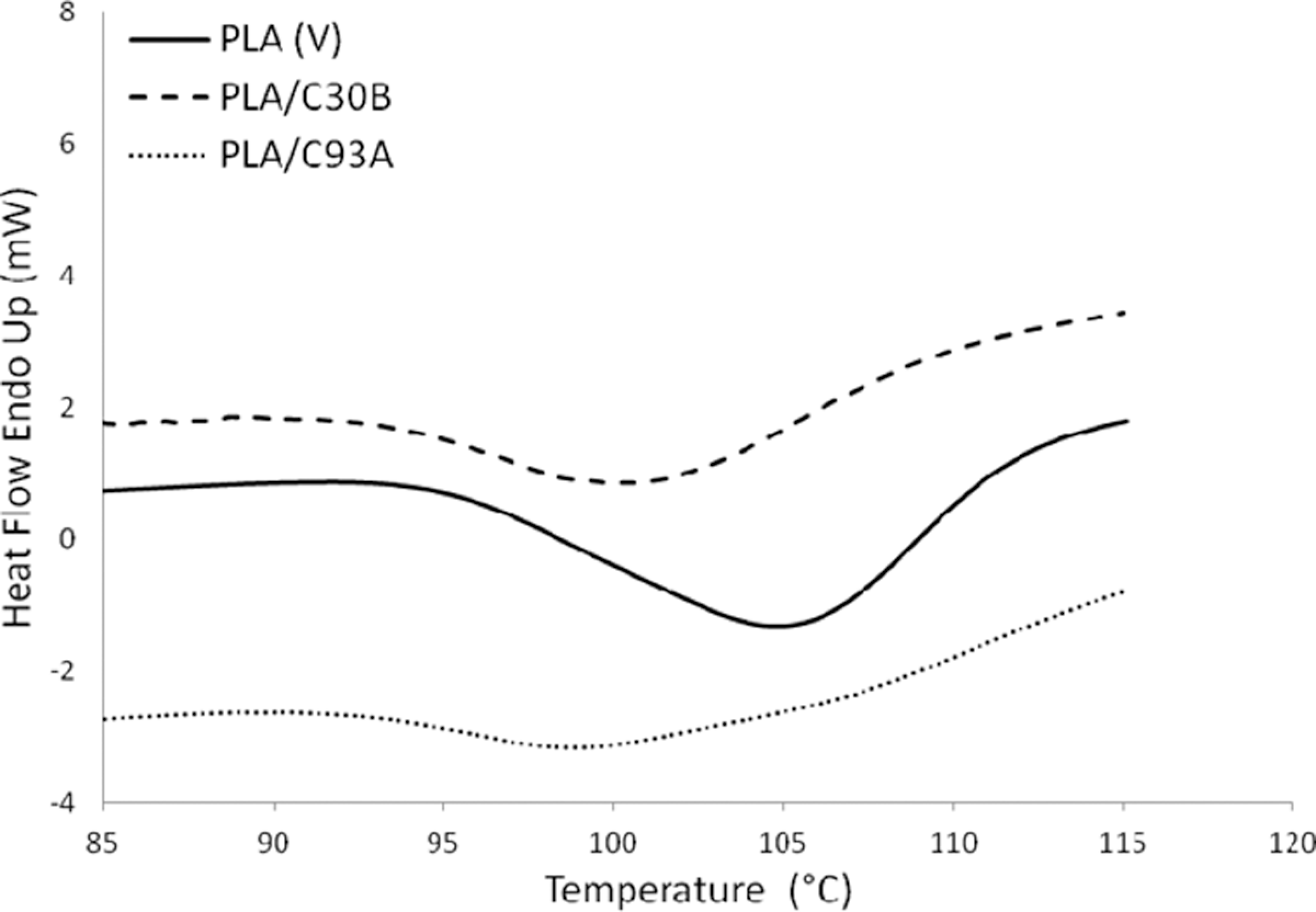
The second transition peak observed in the DSC endotherms at around 105°C represents cold crystallization peak of virgin PLA. As observed in Figure 3, the exotherms reveal reduction in T cc of the matrix polymer at approximately 4 to 5°C with the incorporation of nanoclays. The Tc decreases from 105°C for unfilled PLA to 99.93°C for PLA/C93A and 100.89°C for PLA/C30B nanocomposites, respectively. This decrease in cold crystallization temperature is primarily because of the large surface area of the nanoclays which act as effective nucleating sites, thereby facilitating the crystallization process of PLA. Similar facts have been reported by various authors where the crystallization temperature of PLA has been reduced by 5 to 10°C with the incorporation of nanoclays. Ray et al. 35,36 concluded that nanoclays enhanced the rate of crystallization of PLA. Lee et al. 37 studied the thermal properties of a poly (L-lactic acid) (PLLA) nanocomposite system and observed that the recrystallization temperature (Tc ) decreased by the addition of clay when compared to neat PLA. Nam et al. 38 and Ray et al. 36 have shown that the crystal size of PLLA nanocomposites was significantly smaller than the pure PLLA and the order of spherulites was lower.
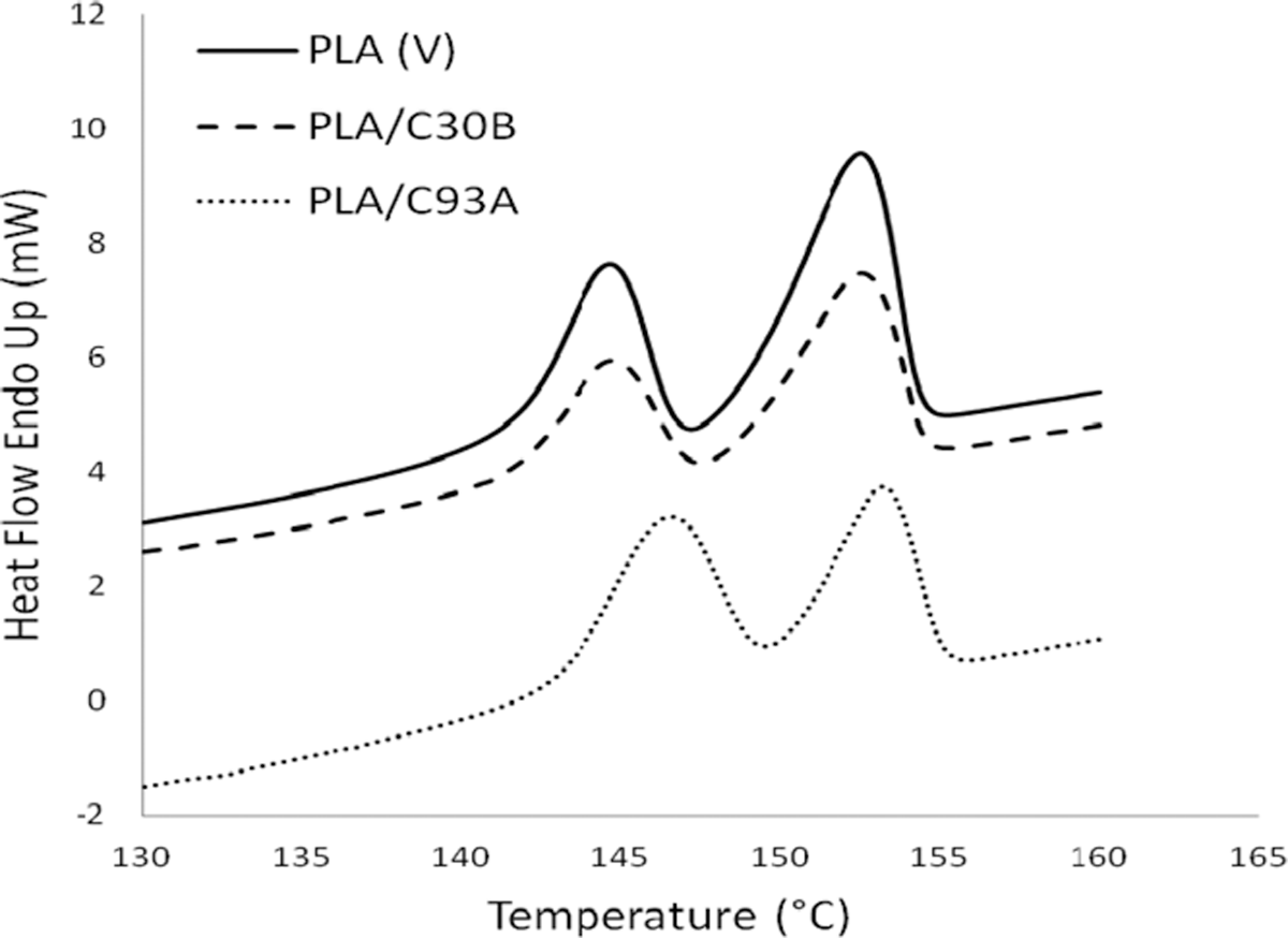
The third transition in the DSC thermograms indicated the melting temperature (Tm ) of virgin PLA and its nanocomposites. As evident from the Figure 4, both the virgin matrixes and the nanocomposites depict double melting peaks. On cooling, the crystallization of PLA is negligible, whereas it crystallizes on heating (cold crystallization) giving a crystalline phase that melts with a two-peak endotherm. The double melting behavior in PLA can be linked to the formulation of different crystal structures 39 –41 —the α-form (pseudo- orthorhombic, pseudo- hexagonal or orthorhombic), 41 melting at higher temperature, and the β-form (orthorhombic or trigonal 41 ) that melts in correspondence of the endotherm at lower temperature. The virgin PLA showed the distinct peaks at around 149 and 152°C. The shoulder or low temperature peak is probably formed on the melting endotherm of the original crystallites 42 ; whereas the imperfect crystals that melt at this temperature recrystallize to form crystals at a higher temperature. A similar characteristic of double melting endotherm was also displayed in case of both the nanocomposites systems. Also there was no appreciable change in the Tm of PLA with the incorporation of organically modified nanoclays.
The degree of crystallinity of PLA and its nanocomposites is represented in Table 2. It is evident that Xc increased marginally with the incorporation of nanoclays. It is believed that C93A could act as a nucleating agent to increase the crystallinity of PLA. Furthermore, in case of PLA/C30B nanocomposites, the OH group present in the surfactant of C30B nanoclay forms H-bond at the interface which restricts the crystallization process and crystallite formation of PLA that subsequently reduces the Xc as compared with PLA/C93A nanocomposites.
TGA
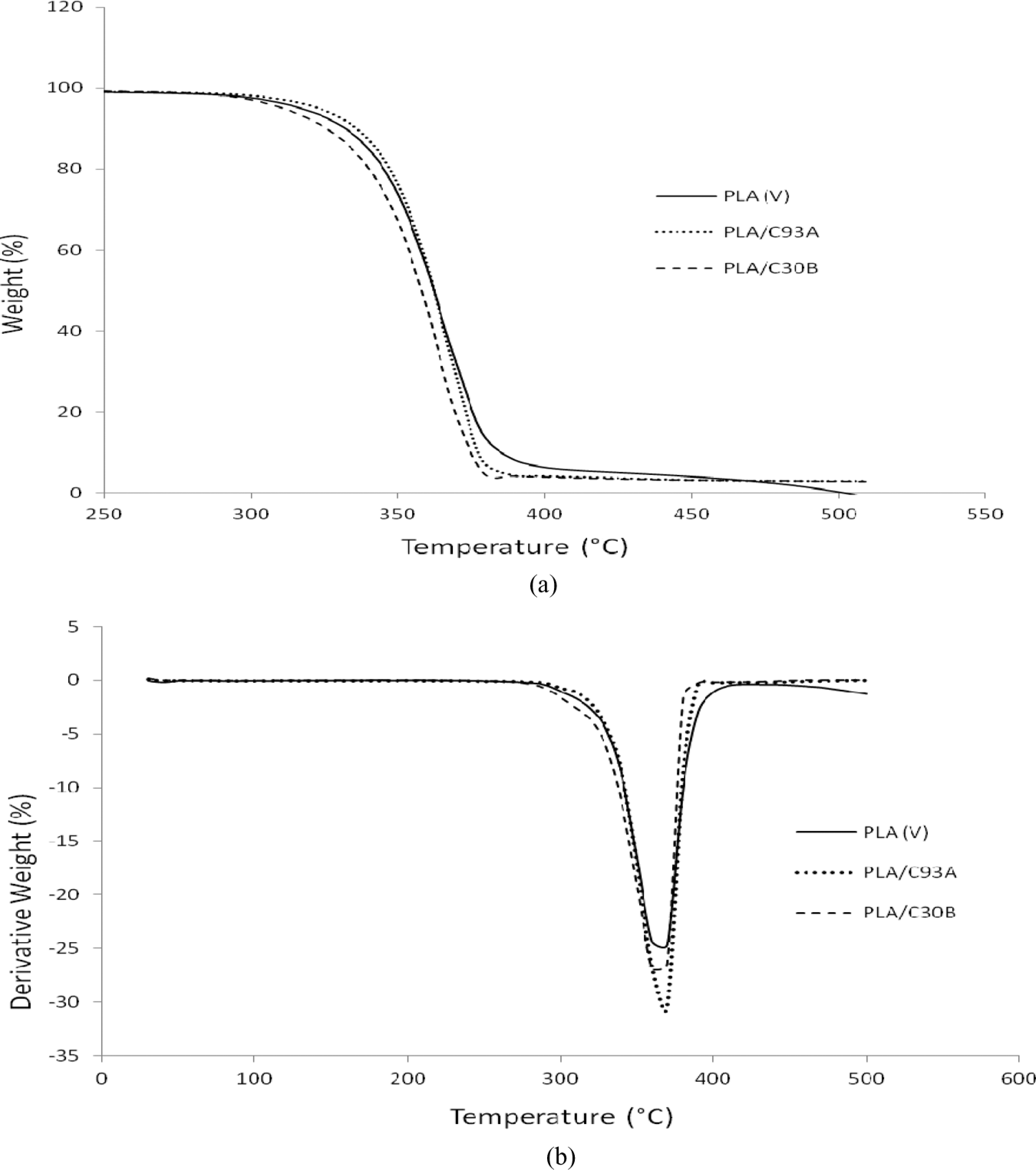
The TGA thermograms of virgin PLA and its nanocomposites are depicted in Figure 5 and Table 3. It is evident that the virgin PLA exhibits an initial degradation temperature of 339.26°C and final degradation temperature of 382.51°C. The incorporation of clay content into the polymer matrix was observed to enhance the thermal stability by acting as a superior insulator and mass transport barrier to the volatile products generated during decomposition. Table 3 shows the Ti , T 50, Tf of the virgin PLA and the nanocomposites. Ti refers to the initial decomposition temperature, while T 50 refers to the decomposition temperature at 50% mass loss and Tf is the final decomposition temperature. It is evident from Table 3 that the Tf of PLA, PLA/C93A and PLA/C30B nanocomposites is 382.51, 379.80 and 377.08°C, respectively. This reveals that the addition of nanoclays decreased the final decomposition temperature of matrix polymer. This behavior is probably because the organically modified clays (C93A and C30B) act as deformation accelerators at high temperature.
TGA and derivative TGA curve of PLA and PLA nanocomposites. PLA: polylactic acid; TGA: thermogravimetric analysis.
The Ti , T 50 and Tf temperatures of PLA nanocomposite system.
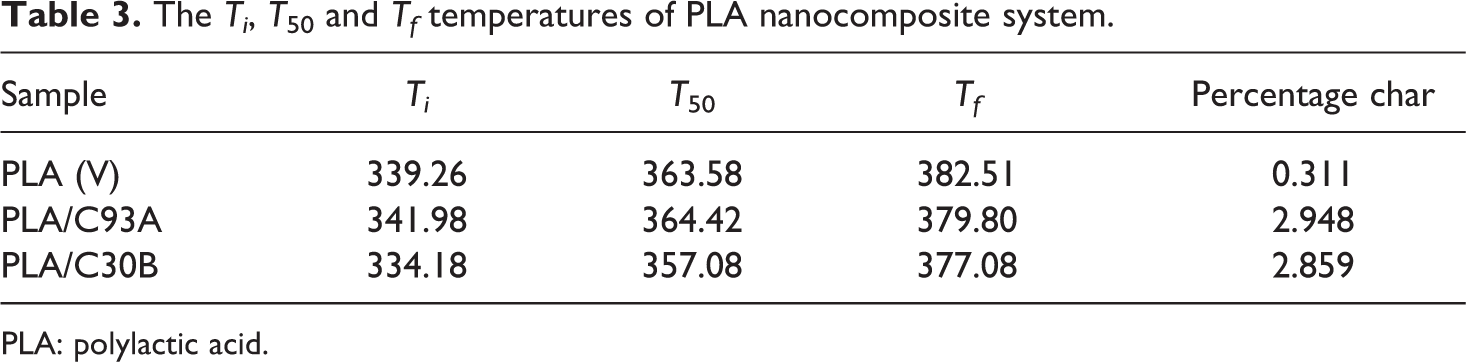
PLA: polylactic acid.
Clay acts as a heat barrier, enhancing the thermal stability in the layered silicate nanocomposites and assisting in the formation of char after thermal decomposition. PLA/C30B nanocomposites depicted lesser final degradation temperature as compared with PLA/C93A and virgin PLA, which is probably due to early decomposition of methyl tallow bis-2-hydroxyethyl intercalant containing OH groups that subsequently accelerate PLA decomposition. On the contrary, PLA/C93A having methyl dehydrogenated tallow intercalant with more hydrophobic characteristics shows marginally higher Ti of PLA matrix, which shows that the layered silicate act as efficient heat barrier on the surface of PLA matrix. In all the nanocomposites, the percentage char was more than the virgin polymer, and this indicated improved flame retardency in the matrix polymer in the presence of nanoclay. Various authors also reported the improved thermal stability for PLA-based nanocomposites prepared with various kinds of organo modified lactic species (OMLS). 43 –47
HDT
The incorporation of nanoclay into the PLA promotes a higher HDT as compared with virgin PLA. Sinha Ray et al. 48 observed higher HDT of PLA in the nanocomposites at different load conditions. In the present study, virgin PLA exhibited a HDT value of 58°C which increased to 62.2 and 60.2°C in case of PLA/C93A and PLA/C30B nanocomposites, respectively. This indicates strong interaction between PLA and C93A in the PLA/C93A nanocomposites. As evident from the melting thermograms discussed in the earlier sections, incorporation of nanoclays does not have any appreciable effect in the Tm of PLA matrix. Hence the improvement in HDT originates from the better mechanical stability of nanocomposites due to the reinforcing effect of the dispersed nanoparticles. Also intercalated clay layers within the matrix contribute to increased crystallinity, thereby increasing the HDT. This increase in HDT of neat PLA after nanocomposite preparation is a very important property improvement not only from the industrial point of view but also in the molecular level control of some properties on the silicate layers, that is, crystallization through interfacial interaction between PLA molecules and SiO4 tetrahedral layers.
DMA
Figures 6 and 7 represent the storage modulus (E’) and tan δ of PLA and its nanocomposites as a function of temperature. As evident from the figures, three different regions could be identified; the glassy state (below 45°C), the glass transition (approximately 50–80°C) and the rubbery plateau (approximately between 80 and 100°C). Virgin PLA exhibited a glass transition of 73°C obtained from the tan δ curve. Through this transition the neat PLA softened as shown by the reduction of E’ by more than 1 order of magnitude. Finally, in the rubbery plateau the matrix polymer became extremely soft from 90 to 100°C.
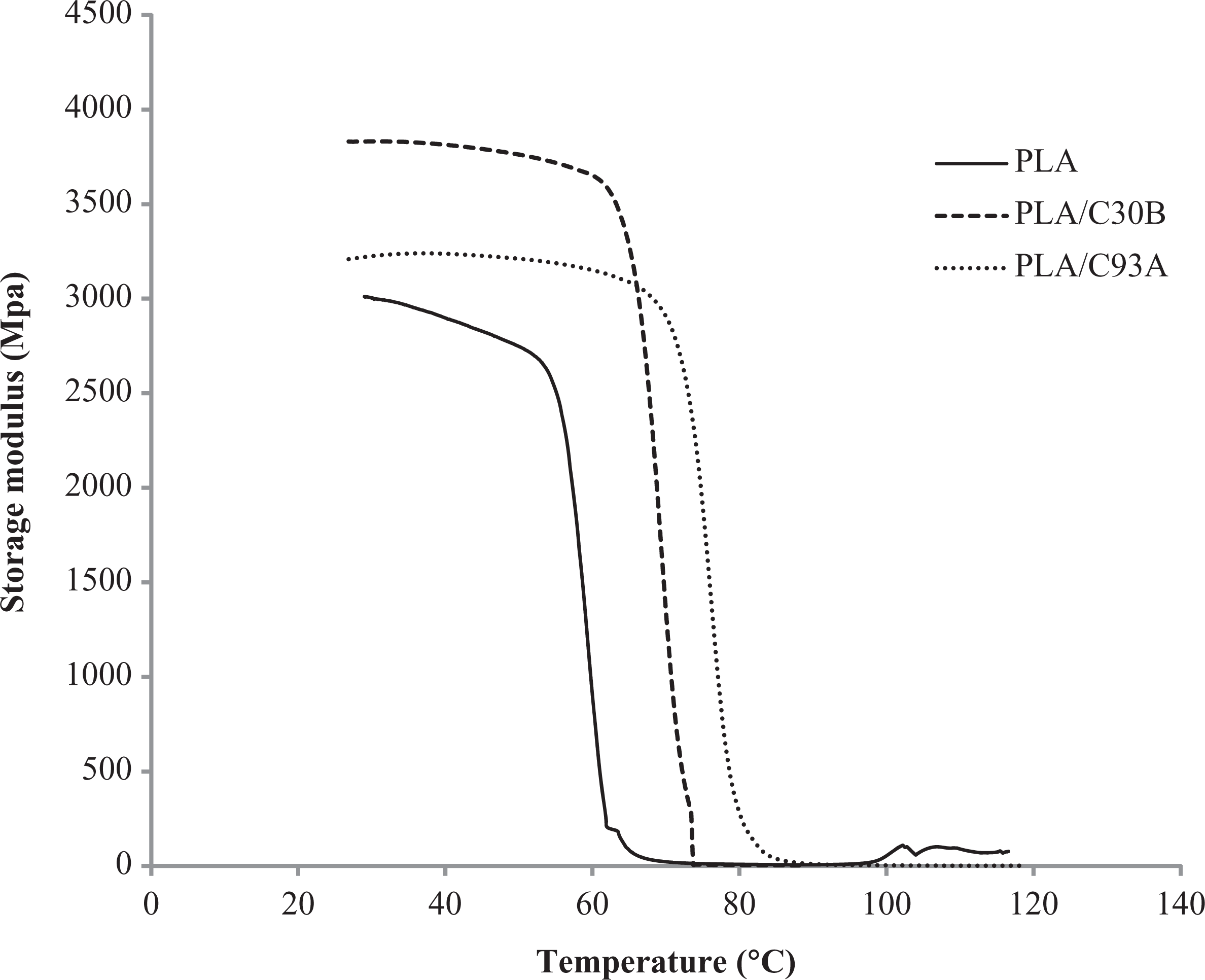
Storage modulus o PLA nanocomposites. PLA: polylactic acid.
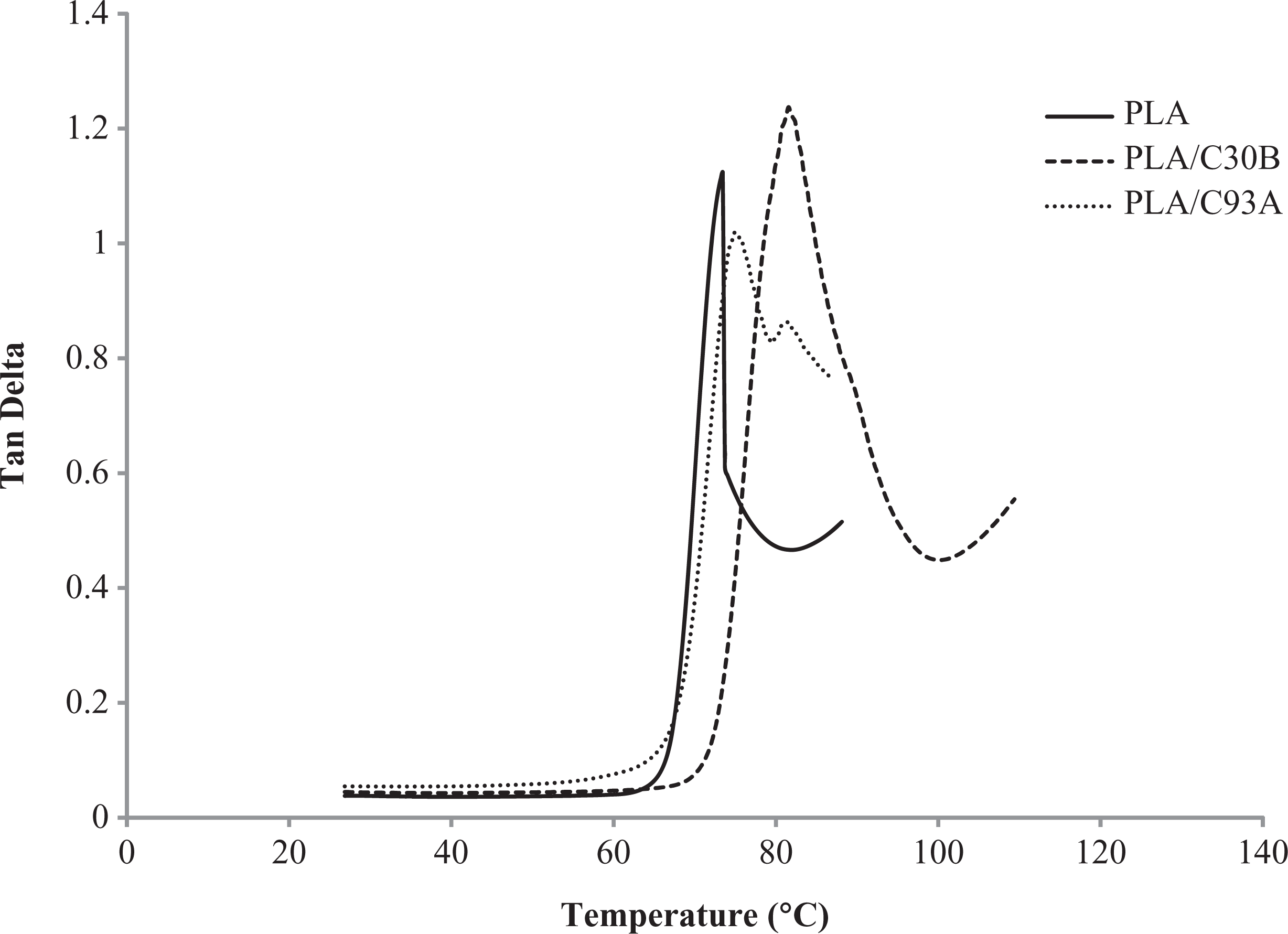
Tan delta o PLA nanocomposites. PLA: polylactic acid.
Further, it is observed that, the presence of organoclays had a strong influence on the viscoelastic response of the resulting nanocomposites in the whole temperature region. In the glassy region, the storage modulus E’ (Figure 6) increased significantly with the incorporation of organically modified nanoclays, which indicates the reinforcing effect imparted by C93A and C30B. Interestingly, nanocomposites prepared using C30B nanoclay exhibited a higher magnitude of E’ than that of PLA/C93A nanocomposites and virgin PLA. This behavior indicates strong hydrogen bond network between the OH groups of methyl tallow bis-hydroxyethyl intercalant of C30B and the C=O group of PLA matrix.
Near the glass transition region of PLA, there was a sudden drop in the matrix modulus. However, with the addition of C30B and C93A, the drop in modulus is compensated with the presence of fillers, due to the segmental immobilization of matrix chains, leading to an increase in E’ and decrease in tan δ intensity (Figure 7). Further, it is also noticed that the Tg of PLA increased from 73.44 to 75.11°C and 81.53°C in case of PLA/C93A and PLA/C30B nanocomposites, respectively. This phenomenon is attributed to improved compatibility of the nanoclay with bulk PLA matrix.
In the rubbery region, an important reinforcing effect was observed due to the presence of the nanoclays. As evident from the tan δ curves, a maximum relaxation was beyond 100°C. The virgin PLA did not reveal any relaxation maximum; however, the PLA/C93A nanocomposite exhibited a peak around 110–115°C. The maximum relaxation beyond 100°C is related to the α process according to mechanical convention which reflects an increase in mechanical loss due to cold crystallization. Further, the intensity of the peak also increases with the incorporation of organoclays.
Morphological properties
WAXD
The XRD patterns of the organoclays (C30B and C93A) and PLA nanocomposites are shown in Figure 8(a) and (b), respectively. The d 001 spacing was calculated from peak positions using Bragg’s law. The primary silicate reflection at 2θ = 4.8° (d spacing ≈1.76 nm) and 3.6° (d spacing ≈2.32 nm) was recorded for the C30B and C93A nanoclay as shown in Figure 8 (a) and (b), respectively. The d spacing is higher in both the case of organoclays in comparison to the MMT (i.e. d spacing ≈1.1 nm) because of the insertion of organic modifier.
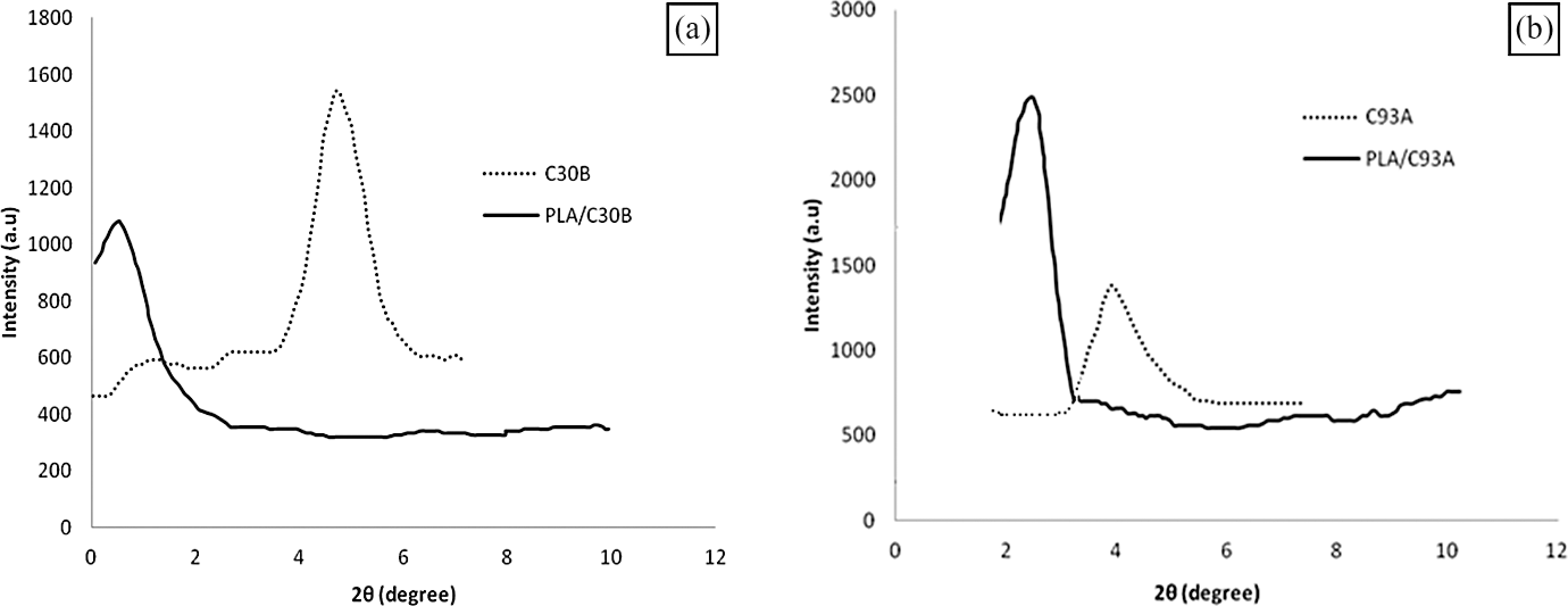
XRD diffractograms of (a) C30B and PLA/C30B and (b) C93A and PLA/C93A respectively. XRD: X-ray diffraction; PLA: polylactic acid.
In case of PLA/C30B nanocomposites, a small peak was observed at the low-angle range which confirms the intercalation of silicate layers of C30B in PLA matrix after the melt mixing. The lack of intergallery clay diffraction is due to the disorderly and random distribution of the clay platelets within the PLA matrix. In contrast to PLA/C30B, PLA/C93A nanocomposites show a high diffraction peak after melt mixing with PLA at high intensity level. There is a shifting in the diffraction peak to lower angle at 2θ = 3.8° (d spacing ≈2.32 nm) to 2θ = 2.9° (d spacing ≈3.04 nm) in case of C93A and PLA/C93A, respectively. This indicates that the layer spacing of C93A was slightly increased, but its ordered structure was not disrupted after melt mixing. Another small peak is also observed in case of PLA/C93A due to the second registry of MMT clay. 49,50
From the above WAXD analysis, we can conclude that PLA/C30B shows the better characteristics with respect to the compatibility and intercalation of the clays with the PLA matrix after melt mixing. PLA/C93A was not exfoliated or significantly intercalated after mixing with PLA. The same observations were also attributed to PCL/C30B and PCL/C93A hybrids. 51 This behavior is primarily due to the hydrophilic nature of C30B clay, and the formation of hydrogen bond between the carbonyl group in the main chain of PLA molecules and the hydroxyl group in the organic modifier of C30B. On the contrary, the C93A nanoclay has no polar interaction because of its nonpolar organic modifier.
TEM
The internal structure of the PLA nanocomposites in the nanometer scale can be obtained by TEM micrographs. Figure 9 (a) and (b) shows the TEM photographs of the PLA/C30B and PLA/C93A, respectively. The dark entities indicate the cross-section of intercalated or stacked clay layers. The stacked silicate layers are due to clustering or agglomeration. The bright fields in the TEM micrograph represent the matrix. The nanocomposites show both intercalated and exfoliated regions. C30B shows better exfoliation or dispersion of clay as compared to C93A with the PLA matrix. Further, few regions showing flocculated as well as intercalated clay galleries were noticed in case of PLA/C93A nanocomposites, thus revealing less compatibility of the clay with the PLA matrix.
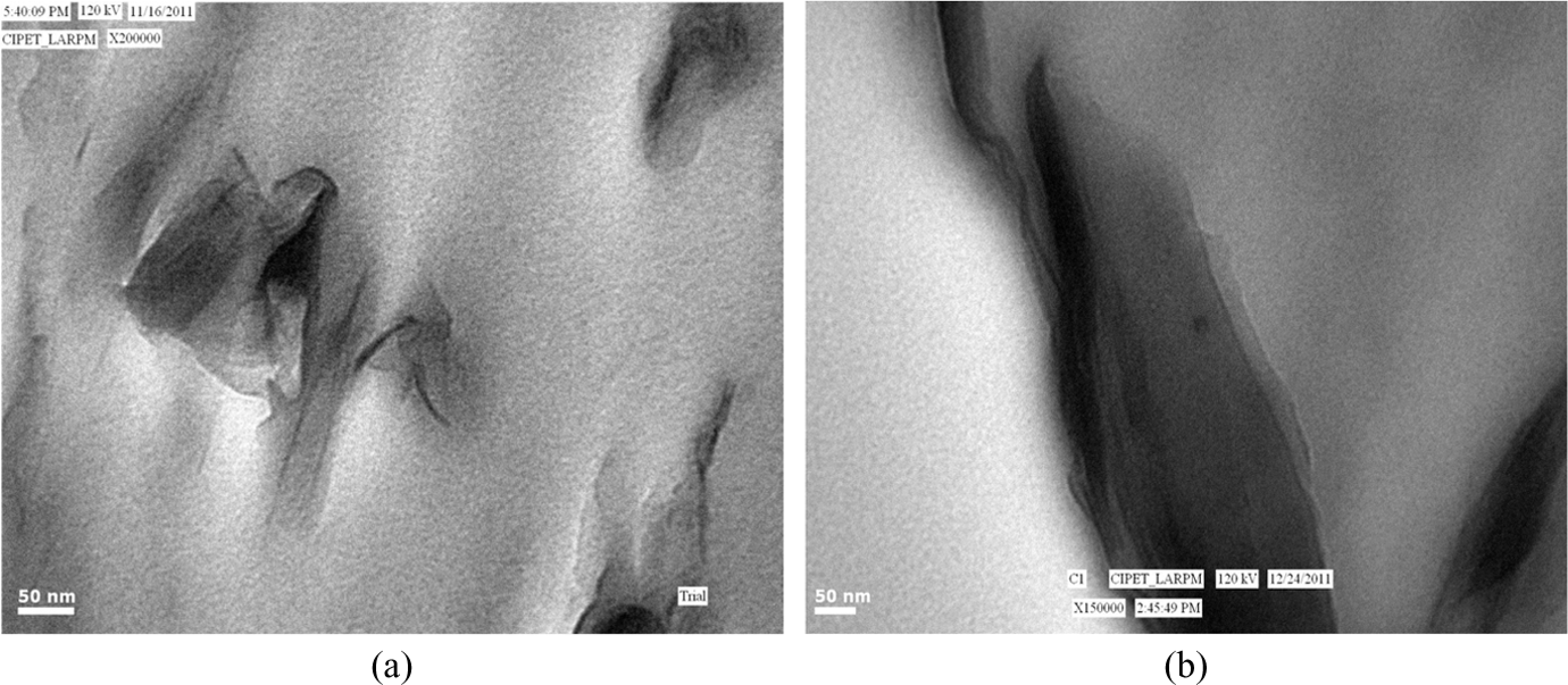
TEM micrographs of (a) PLA/C30B and (b) PLA/C93A. TEM: transmission electron microscope; PLA: polylactic acid.
Conclusion
Incorporation of 3 wt% of cloisite 30B increases the mechanical properties of PLA matrix. Storage modulus versus temperature plots showed an increase in the magnitude of the peaks with the addition of nanoclays. Cloisite 30B has been well dispersed within the matrix PLA, which is clearly evident by the X-ray diffraction analysis. Increase in thermal stability as a result of good interaction between the intercalated/exfoliated C30B nanoclay layers has been observed in TGA. Hence, PLA/C30B clay nanocomposites can be used for various high-performance applications.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.