Abstract
Conducting polythiophene (PTP)/Na-montmorillonite (Na-MMT) composites have been prepared by intercalative polymerization. The synthesized composites are characterized by Fourier transform infrared spectroscopy, x-ray diffraction (XRD), scanning electron microscopy, transmission electron microscopy (TEM), and thermal analysis. XRD and TEM images showed that the PTP was intercalated into the clay layers. The thermogravimetric analysis and the differential thermal analysis revealed the introduction of Na-MMT results in thermal stability of composites with respect to pure PTP. Furthermore, adsorptive properties, the moisture retention, and water uptake values of composites were investigated. Conductivity behavior of composites was also studied.
Introduction
In recent years, much research attention has been paid to the synthesis and characterization of conductive polymer/clay composite. Clays are inorganic layered materials for the synthesis of polymer/clay composite because it is natural, abundant, inexpensive, and has excellent chemical and thermal resistance. Among many layered materials, the Na-montmorillonite (Na-MMT) has received intensive attention.
Introduction of only a small amount of clay minerals, such as Na-MMT, into the polymer structure may provide remarkable physical and chemical properties of the conductive polymers. 1 –4 Clay minerals can participate in intercalative polymerization with conducting polymers like polypyrrole, polyaniline, and polythiophene (PTP) because these polymers show significant improvements in conductivity and environment stability. 5
Among the polymers, PTP and its derivatives as promising conducting polymers have attracted considerable attention in recent years for commercial applications due to high mobility, good stability, easy synthesis, economic efficiency, and better conductivity than other conductive polymers. 6 They have potential applications in many technological applications in microelectronic devices, catalysts, organic field-effect transistors, chemical sensors, and biosensors. 7 However, its poor thermal and mechanical properties especially thermal stability and solubility to an aqueous solution need to be enhanced.
Therefore, the aim of this article is to improve the thermal stability of PTP through the synthesis with layered materials such as clay. During this method, the thermal properties of conductive polymers in composite form are improving with incorporating clay.
A great deal of work has been carried out regarding the conductive polymer/organoclay composites that were available in the literature, but there is little work about preparation, characterization, and adsorptive properties of intercalated PTP/Na-MMT composites using unmodified smectic clay. Therefore, in the present study, new types of composites from PTP and unmodified Na-MMT were prepared by intercalative polymerization using ammonium peroxydisulfate (APS) as oxidant in aqueous medium. In addition, we also report the adsorptive properties, moisture retention, water uptake values, and conductivity behavior of the composites.
Experimental
Materials
In this study, the monomer thiophene (TP) and oxidant APS were purchased from Merck Chemical Company (Germany). TP was distilled under reduced pressure and stored in a refrigerator prior to use. The oxidant, APS, and all other chemicals were used without further treatment. Na-MMT clay purchased from Reşadiye (Tokat/Turkey) was used as the clay material. Its cation exchange capacity value is 1.08 mol kg−1, specific surface area is 43 m2 g−1, and an interlayer spacing of Na-MMT is 1.00 nm.
Synthesis of PTP/Na-MMT composites
Na-rich montmorillonite was obtained, as previously described in the literature, by the methods of dispersion and sedimentation from its aqueous suspension. 8 Synthesis of PTP/Na-MMT composites in aqueous medium occurred as below via intercalative polymerization. 3
The dispersed Na-MMT (1.0 g) in 25 mL distilled water is vigorously stirred at room temperature overnight. Then the TP monomer and the aqueous Na-MMT suspension in a 100-mL Pyrex glass tube were mixed with different mass ratios as given in Table 1. The APS as oxidant solution in water (3.5 × 10−2 mol L−1) was added as 5 mL to the prepared suspension. The samples were polymerized and adjusted to the polymerization temperature (0°C) for 2 h to obtain PTP/Na-MMT composites using an ultrasonic bath (Bandelin Sonorex-RK100H, Germany). Then the obtained deep black colored powder indicated the formation of PTP. The resulting deep black precipitate was isolated from the reaction mixture by filtration and then was washed with deionized water to remove the unreacted monomer. Obtained composites were dried at 30°C under vacuum. The other samples were prepared by the same procedure using different content of TP. Table 1 shows the polymerization conditions, different contents of TP, and sample codes.
Data on polymerizationa and chemical formation in TP and Na-MMT system.
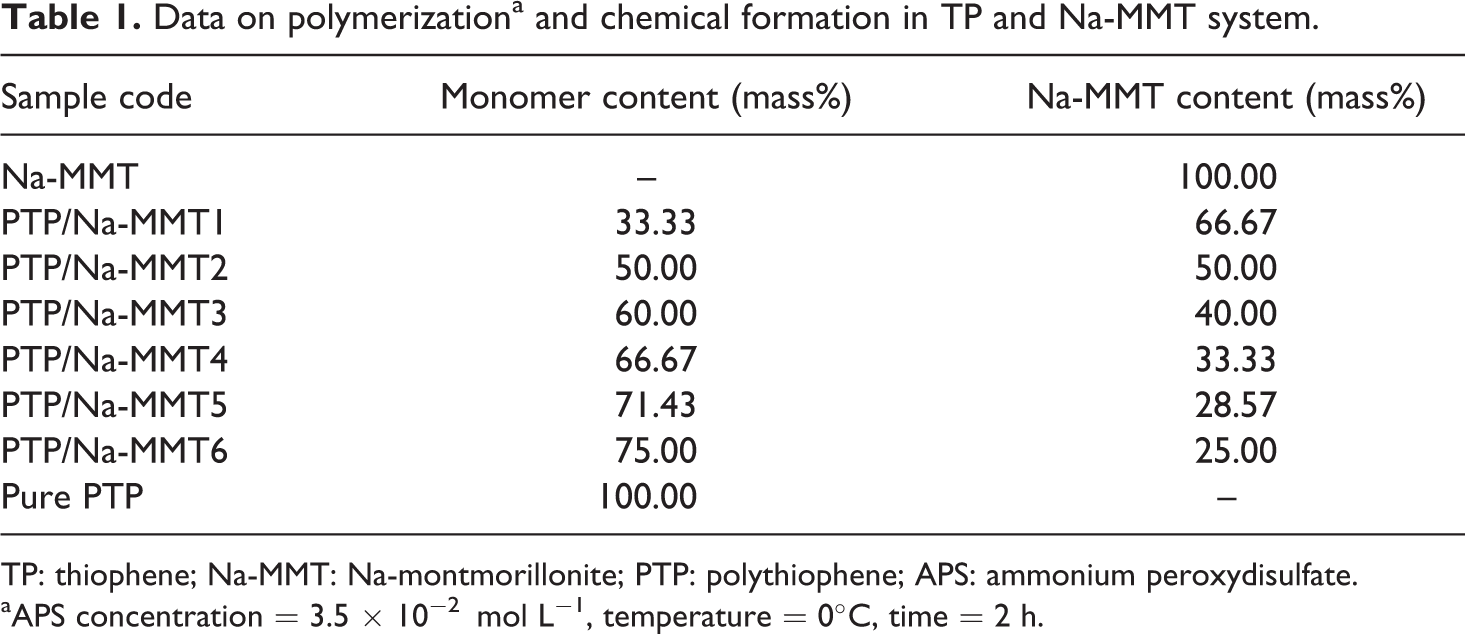
TP: thiophene; Na-MMT: Na-montmorillonite; PTP: polythiophene; APS: ammonium peroxydisulfate.
aAPS concentration = 3.5 × 10−2 mol L−1, temperature = 0°C, time = 2 h.
Composite characterization
After synthesis, the insertion of PTP into layers was examined using an Inel Equinox 1000 powder diffractomer using CoKα x-rays whose wavelength was 0.178901 nm. The Na-MMT and composites were in the powder form for x-ray diffraction (XRD) analysis.
The chemical structure of samples were taken in KBr pellets and recorded on a Perkin Elmer 100 Model Fourier transform infrared (FTIR) spectrophotometer in the range 450–4000 cm−1.
Thermogravimetric analysis (TGA) and differential thermal analysis (DTA) were taken on a Shimadzu simultaneous DTA-TG apparatus (DTG-60H Model) thermal analyzer under nitrogen atmosphere at a heating rate of 10°C min−1, and α-Al2O3 was used as an inert material. The temperature range studied was from room temperature to 1000°C.
The scanning electron microscopy (SEM) images were investigated by a LEO-435 Model scanning electron microscope (England). All specimens were coated with gold prior to examination.
Transmission electron microscopy (TEM) was operated with a LEO-906 with an accelerating voltage of 80 kV. The samples for TEM were prepared by curing epoxy resin and were microtomed with a Leica ultracut-R. Ultrathin sections were cut with a glass knife and deposited on one layer of carbon 300 mesh copper grids. The contrast between the layered clay and polymer phase was sufficient for imaging, and so observed by TEM without staining.
The adsorption and desorption isotherms for nitrogen were obtained at 77 K using a volumetric adsorption instrument fully constructed of Pyrex glass and connected to high vacuum was used in the experiments. 8
The percentage moisture retention was calculated from the difference between the masses of the conditioned and unconditioned samples. 4,9 The weighed dry samples in the form of pellet were conditioned at 25°C in the medium having 100% humidity for 24 h for the moisture retention measurements.
The mass percentage water uptake of samples was determined by the difference between the weightings of dry and wet samples in the form of pellet. Dry samples were weighed and then kept in distilled water for 2 h at 25°C. After immersion, the wet samples were wiped using filter paper and reweighed immediately. 9,10
The prepared powders were compacted under 10 ton cm−2 for 5 min by a uniaxial press (SPECAC Model, UK) using stainless steel mold. Diameter and thickness of the cylindrical pellets were about 10 mm and 1 mm, respectively.
A Keithley 6517A multimeter (USA) was used for conductivity measurements. DC conductivities (Λ) of the pellets were determined at room temperature using conventional four-probe technique. 3,6
Results and discussion
FTIR spectroscopy
Figure 1(a) to (c) shows the FTIR spectra of Na-MMT, pure PTP, and PTP/Na-MMT6 composite. The characteristic peaks of the Na-MMT at 1040 cm−1 (Si–O), 525 cm−1 (Al–O), 470 cm−1 (Mg–O), 3460 cm−1, and 3630 cm−1 (O–H stretching) and the characteristic peaks of PTP at 794 and 1005 cm−1 (C–H vibrations), 1043, 1450, and 1760 cm−1 (C=C of the TP ring) are observed in Figure 1(a) and (b). The FTIR spectra of the PTP/Na-MMT6 composite reveal the peaks associated with pure PTP and the Na-MMT in Figure 1(c). This spectrum showed the formation of PTP and its incorporation in the prepared PTP/Na-MMT composite. This band may also indicate that the Na-MMT particles were physically covered by PTP macromolecules. These results were consistent with previously published data 6,11 –14 and demonstrated that PTP was contained in this composite.
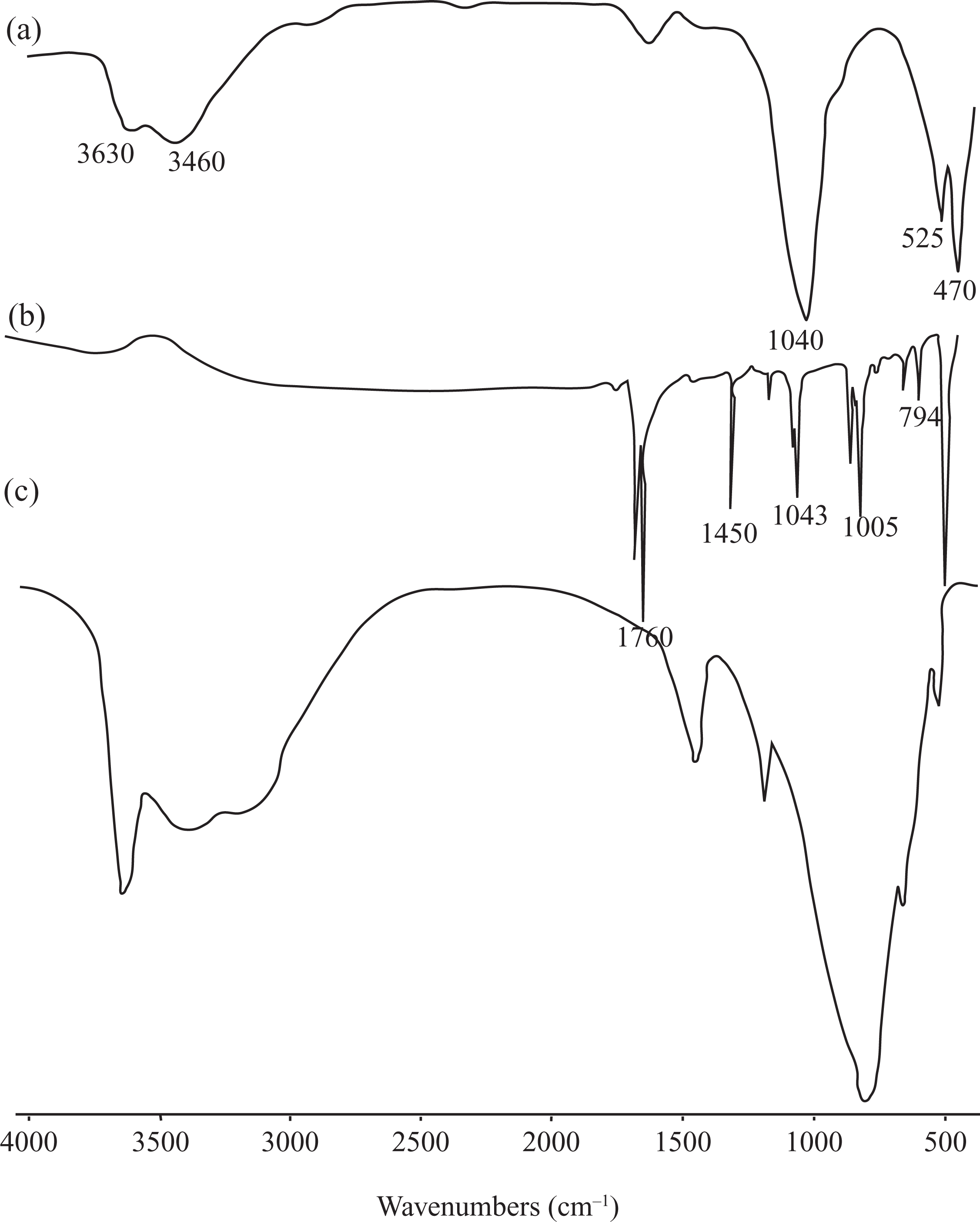
FTIR spectra of (a) Na-MMT, (b) pure PTP, and (c) PTP/Na-MMT6 composite. FTIR: Fourier transform infrared spectroscopy; Na-MMT: Na-montmorillonite; PTP: polythiophene.
XRD characteristics
Insertion of the TP into the layer of Na-MMT was also examined by XRD. XRD patterns provide useful information about the d-spacing values (d 001) of Na-MMT and the composites. Distance between the layers, were estimated by following the Bragg’s equation (d = λ/2sinθ). Figure 2(a) to (g) represents the XRD pattern of the Na-MMT and a series of PTP/Na-MMT composites with different monomer content, respectively. It can be seen from Figure 2 that the XRD pattern of Na-MMT is different from that of composites pattern. The XRD pattern of Na-MMT showed a peak at 1 nm (2θ = 10.30°), which was shifted toward lower angles for the composites due to the intercalation of PTP between the Na-MMT layers. It was found gradually increasing with the increase in monomer percentage in the composite. The obtained results are presented in Table 2. As shown in Table 2, the average Na-MMT interlayer distance shifted from 1.00 nm (2θ = 10.30°) to 1.22 nm (2θ = 8.43°), and no further increase in interlayer distance at higher PTP content was observed. These results prove and support that the PTP molecules are intercalated into the interlayer space of clay and expand the interlayer spacing, and an intercalated PTP/Na-MMT composites has been synthesized. The above result is also close to that reported by many other researchers. 14,15
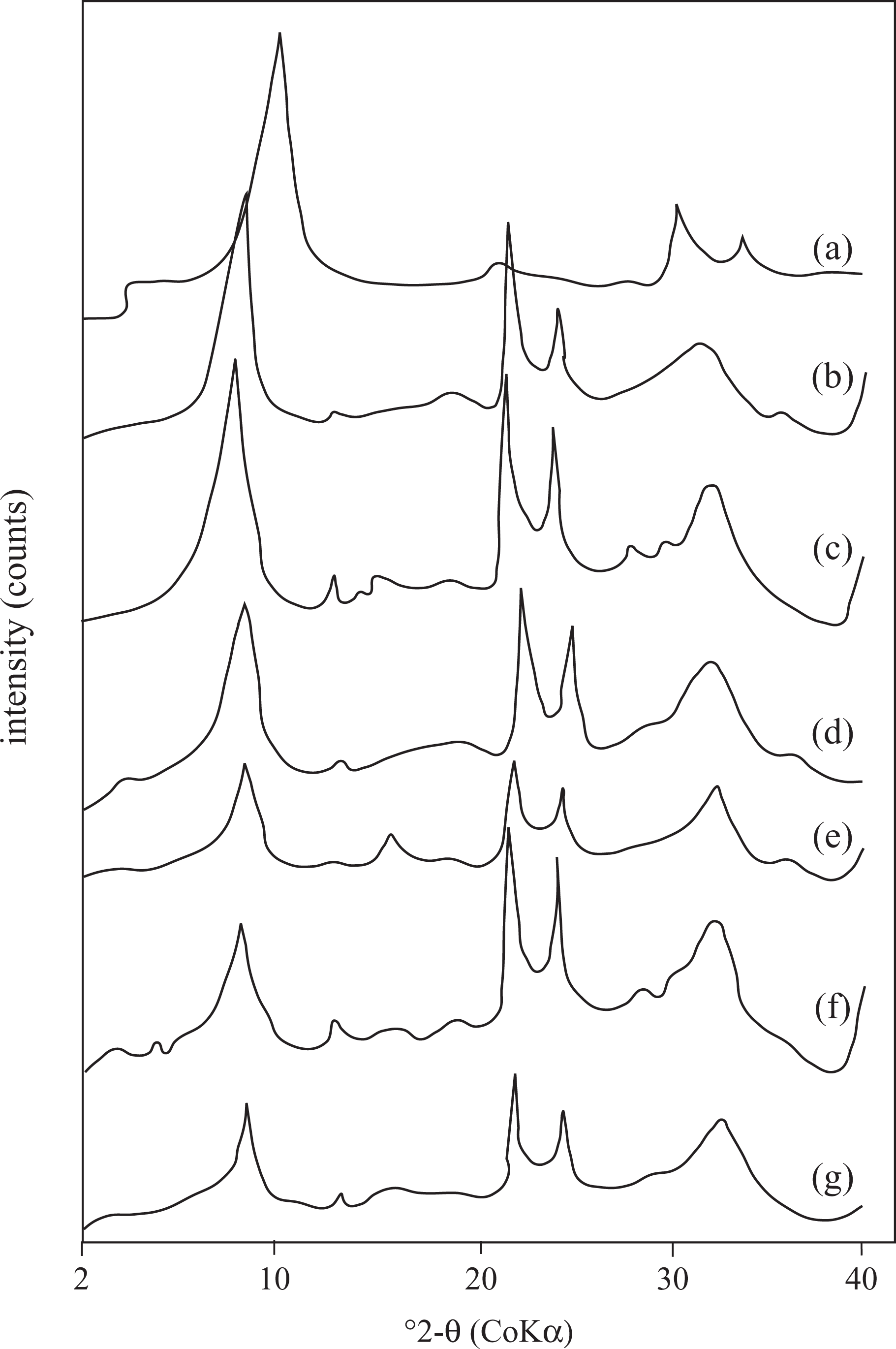
XRD patterns of (a) Na-MMT, (b) PTP/Na-MMT1, (c) PTP/Na-MMT2, (d) PTP/Na-MMT3, (e) PTP/Na-MMT4, (f) PTP/Na-MMT5, and (g) PTP/Na-MMT6 composites. XRD: x-ray diffraction; Na-MMT: Na-montmorillonite; PTP: polythiophene.
XRD data for Na-MMT and PTP/Na-MMT composites.
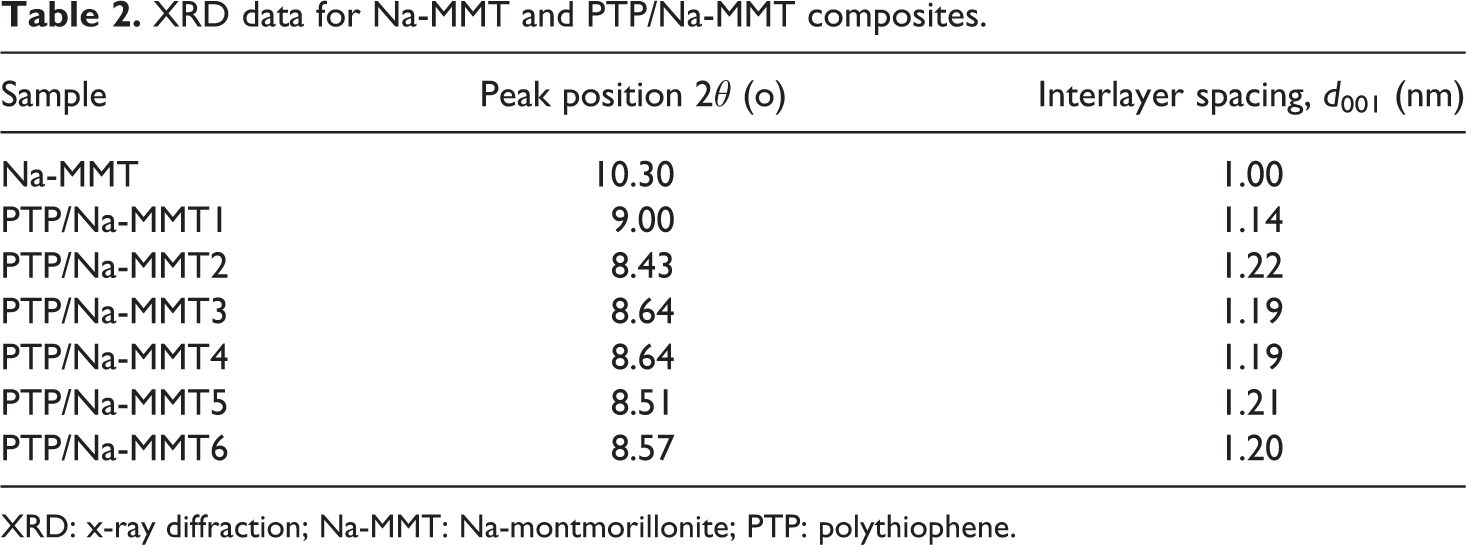
XRD: x-ray diffraction; Na-MMT: Na-montmorillonite; PTP: polythiophene.
Scanning electron microscopy
Scanning electron micrographs revealed some interesting morphological differences between the Na-MMT and PTP/Na-MMT6 composite. Figure 3(a) shows overlapping of tight and smooth plates of the clay. 14 After polymerization, the composite shows significant changes in the morphology (Figure 3(b)). It can be seen that PTP polymerization occurred within the Na-MMT layers. Composite exhibits a spongy and porous structure. Clay layers dispersed homogeneously in the polymer matrix and the interlayer spacing of Na-MMT is expanded. This morphological pattern was also observed by other researchers. 6,16

SEM of (a) Na-MMT and (b) PTP/Na-MMT6 composite at a magnification of × 5000. SEM: scanning electron micrographs; Na-MMT: Na-montmorillonite; PTP: polythiophene.
Transmission electron microscopy
TEM was used in order to confirm the morphology of the PTP in the clay layers. The dark lines represent the cross section of the MMT layers. Some agglomeration of clay platelets was observed. The composite image shows that the clay layers embedded in the polymer matrix evidence the intercalative polymerization. TEM micrograph of PTP/Na-MMT2 composite had layered structure and homogenously dispersed nanoscale, which is consistent with XRD d-spacing data. This feature was similar to the observations of other researchers. 16,17 The average particle sizes as evaluated from Figure 4 were varying from 100 to 190 nm.

TEM image of PTP/Na-MMT2 composite. TEM: transmission electron microscopy; Na-MMT: Na-montmorillonite; PTP: polythiophene.
Thermal properties
TGA and DTA results of the Na-MMT, pure PTP, PTP/Na-MMT3, and PTP/Na-MMT4 composite are shown in Figures 5 and 6 (a) to (d). The TGA curve of Na-MMT indicates that there are two stages of decomposition (Figure 5(a)). The first one is small due to the loss of absorbed water when the temperature is lower than 125°C and mass loss is ∼6.13%. When the temperature is higher than 800°C, the water resulting from the structural –OH groups of Na-MMT begins to be removed. The total mass loss is only 20.82%, up to 1000°C. As could be expected, the Na-MMT shows a high thermal stability. 14
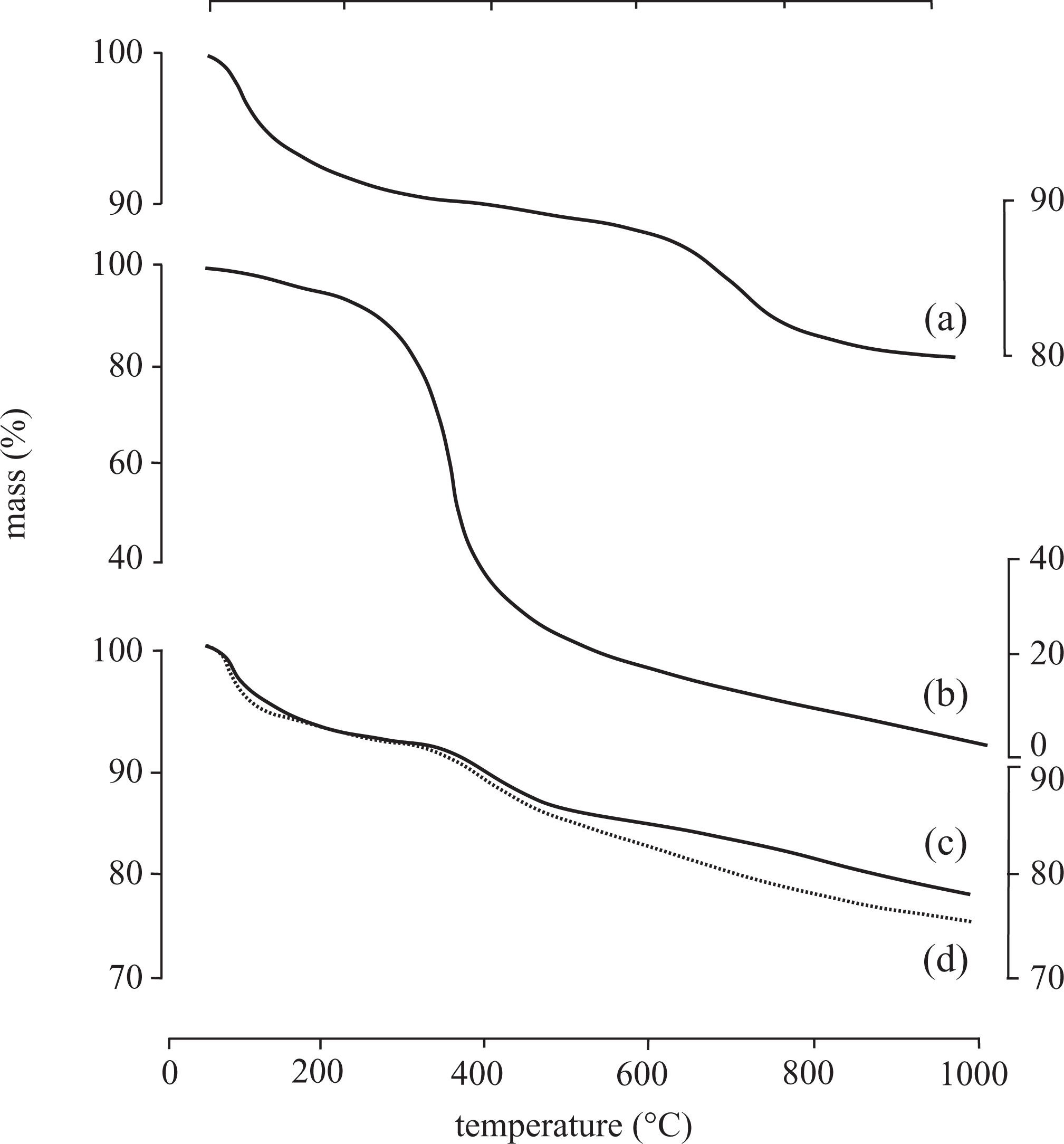
TGA curves of (a) Na-MMT, (b) pure PTP, (c) PTP/Na-MMT3, and (d) PTP/Na-MMT4 composite obtained in nitrogen atmosphere at a heating rate of 10°C min−1. TGA: thermogravimetric analysis; Na-MMT: Na-montmorillonite; PTP: polythiophene.
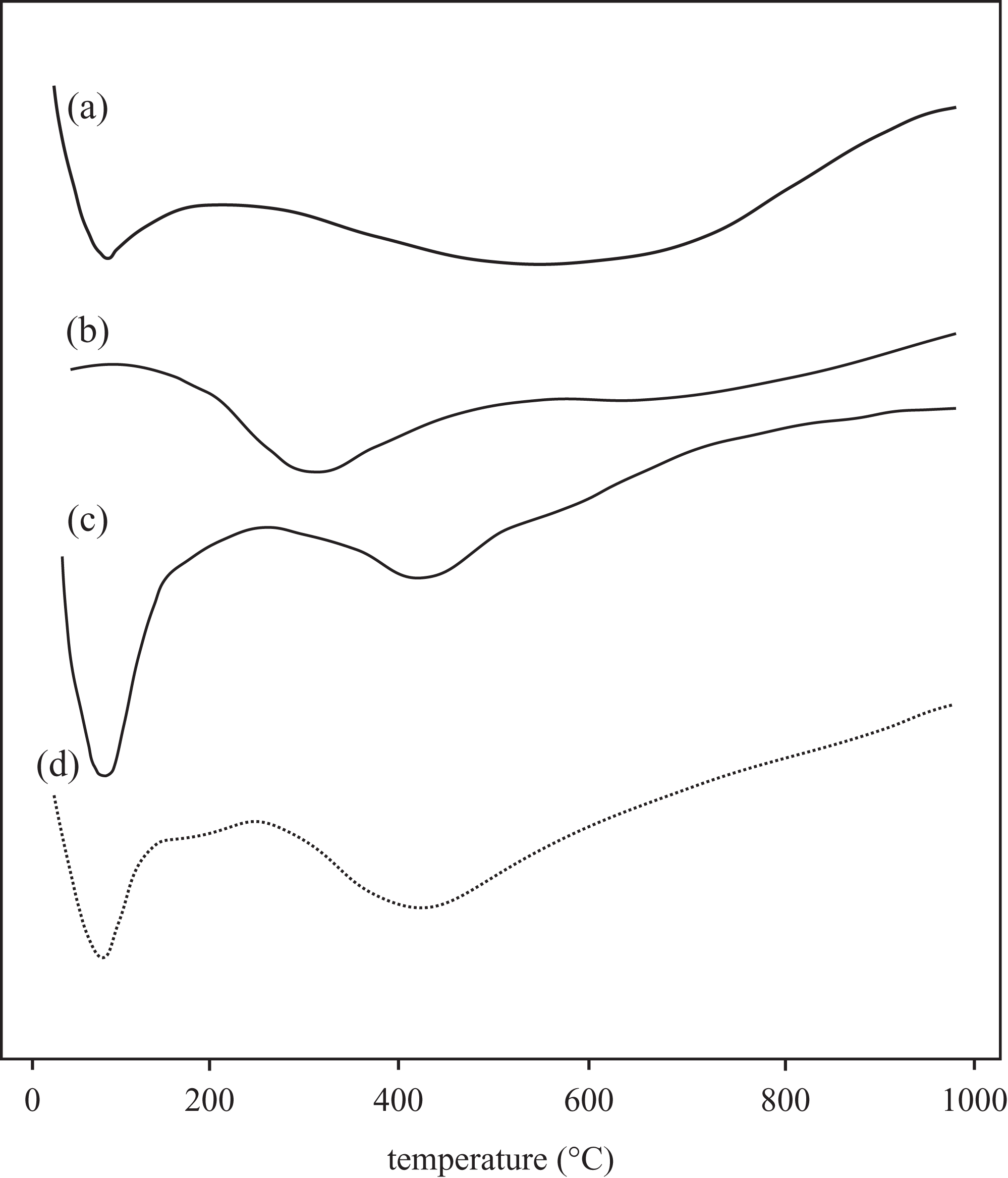
DTA curves of (a) Na-MMT, (b) pure PTP, (c) PTP/Na-MMT3, and (d) PTP/Na-MMT4 composite obtained in nitrogen atmosphere at heating rate of 10°C min−1. Na-MMT: Na-montmorillonite; PTP: polythiophene.
As shown in Figure 5(b), a small mass loss (∼6.27%) just below 100°C is attributed to the loss of absorbed water of pure PTP. Pure PTP begins to decompose at approximately 250°C, while at high temperature range (after 300°C), the decomposition rate increases, and finally the second mass loss is nearly 100.00% at 1000°C, which was attributed to thermal decomposition of the PTP chains. This stage is the thermal decomposition of polymer. Consequently, pure PTP should exhibit lower thermal stability relative to Na-MMT.
For the composites, the mass loss of the PTP/Na-MMT3 composite started at 350°C, and exhibited a mass loss of 23.18% at 1000°C. But, the TP content further increased from 60.00 to 66.67 mass% for the PTP/Na-MMT4 composite has a lower decomposition temperature (340°C) and exhibited a mass loss of 25.30% at 1000°C because the clay content is low in this sample. The mass loss rate of pure PTP is seen higher than those of PTP/Na-MMT3 and PTP/Na-MMT4 composites. Incorporation of PTP with Na-MMT would be expected to enhance the thermal stability of PTP/Na-MMT3 and PTP/Na-MMT4 composites relative to that of PTP. So, the PTP/Na-MMT3 and the PTP/Na-MMT4 composites have a higher decomposition temperature (350 and 340°C), which are 50 and 40°C more than that of pure PTP. This enhances that the thermal stability of PTP/Na-MMT3 and PTP/Na-MMT4 composites are due to Na-MMT with layered silicate structure acts as barrier to heat flow. It is also noted that only small amount of Na-MMT is effective in improving the residual mass% and lowering thermal degradation rate for composite. These views are consistent with the observed trend. 6,14
The DTA curves of Na-MMT, pure PTP, PTP/Na-MMT3, and PTP/Na-MMT4 composite were also shown in Figure 6(a) to (d). DTA results are in accordance with TGA results. In the DTA curve of the Na-MMT, the endothermic peaks resulting from the dehydration (25–125°C) and the dehydroxylation (400–800°C) are observed. Pure PTP shows the endothermic peak between 150 and 450°C, which is associated with the decomposing of structure. Figure 6(c) and (d) indicates those two endothermic peaks resulting from the dehydration of composites and the decomposition of PTP in the composite, respectively. The thermal decomposition of PTP/Na-MMT3 and PTP/Na-MMT4 shifted the higher temperature than pure PTP, which implies that the composite system has the enhanced thermal stability due to the intercalation of PTP between Na-MMT clay layers.
Adsorptive properties
The specific surface areas were determined from nitrogen adsorption isotherms of Na-MMT and PTP/Na-MMT1 composites in Figure 7. Here, p is the adsorption equilibrium pressure, p° is the vapor pressure of liquid nitrogen, and p/p° ≡ x is the relative equilibrium pressure. The adsorption capacity (n; mol g−1) is defined as molar amount of nitrogen adsorbed per 1 g of sample.
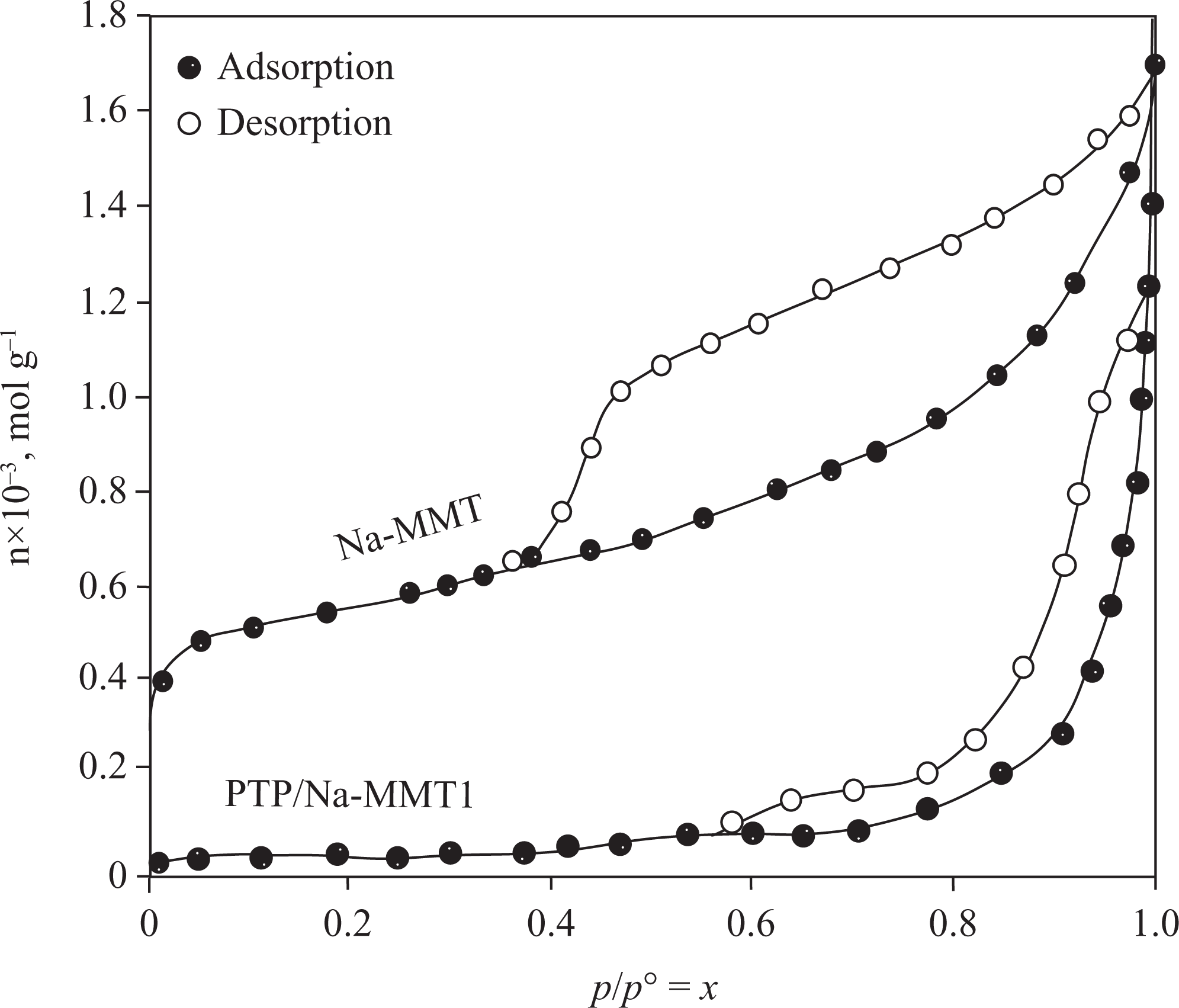
The adsorption and desorption isotherms of the nitrogen on the Na-MMT and PTP/Na-MMT1 composite. Na-MMT: Na-montmorillonite; PTP: polythiophene.
As could be expected, the Na-MMT showed the higher adsorption capacity than PTP/Na-MMT1 composite. The Brunauer–Emmett–Teller (BET) equation was applied to determine the specific surface areas (A) for Na-MMT and PTP/Na-MMT1 composites using N2 adsorption data as shown in Figure 7, and the specific micro-mesopore volumes (V) were calculated from the desorption data in Figure 7. 18,19 To see whether the adsorption data satisfied the BET equation or not, the graph in Figure 8 was plotted. The straight lines seen in these graphs verify that the adsorption data satisfy the BET equation.
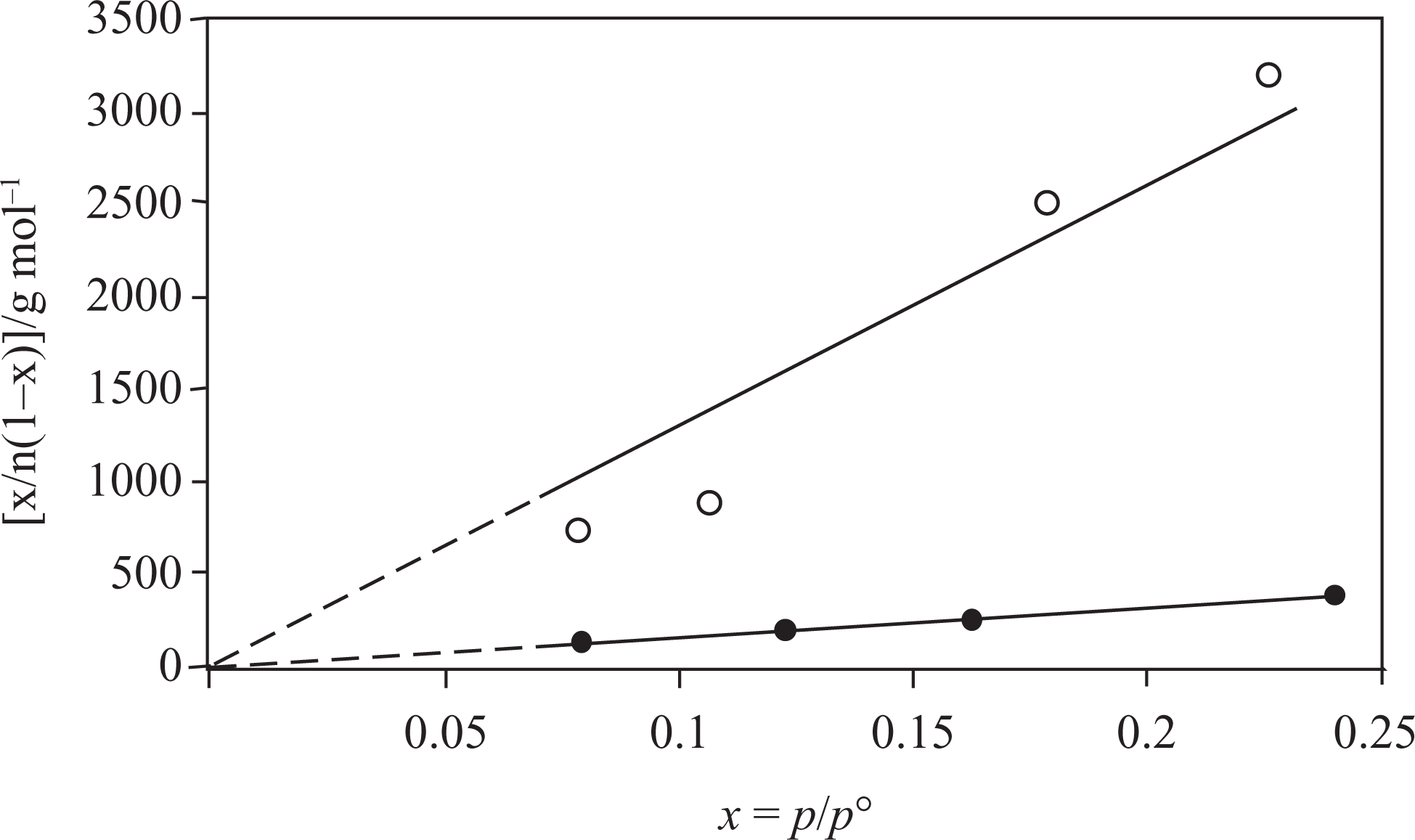
The Brunauer–Emmett–Teller (BET) straight line of the Na-MMT (•) and PTP/Na-MMT1 (^) composite. Na-MMT: Na-montmorillonite; PTP: polythiophene.
The specific surface area and micro-mesopore volume of Na-MMT were 43 m2 g−1 and 0.07 cm3 g−1, respectively. One can see that these values dramatically went down to 7.47 m2 g−1 and 0.04 cm3 g−1 for PTP/Na-MMT1, respectively. This could be explained in terms of covering or filling of the pores by PTP. 20,21
Moisture retention and water uptake measurements
It was observed that from Table 3, moisture retention gradually decreased, while water uptake values of the composites remarkably decreased with the increase in TP content.
The moisture retention and water uptake values of Na-MMT, pure PTP, and PTP/Na-MMT composites.a
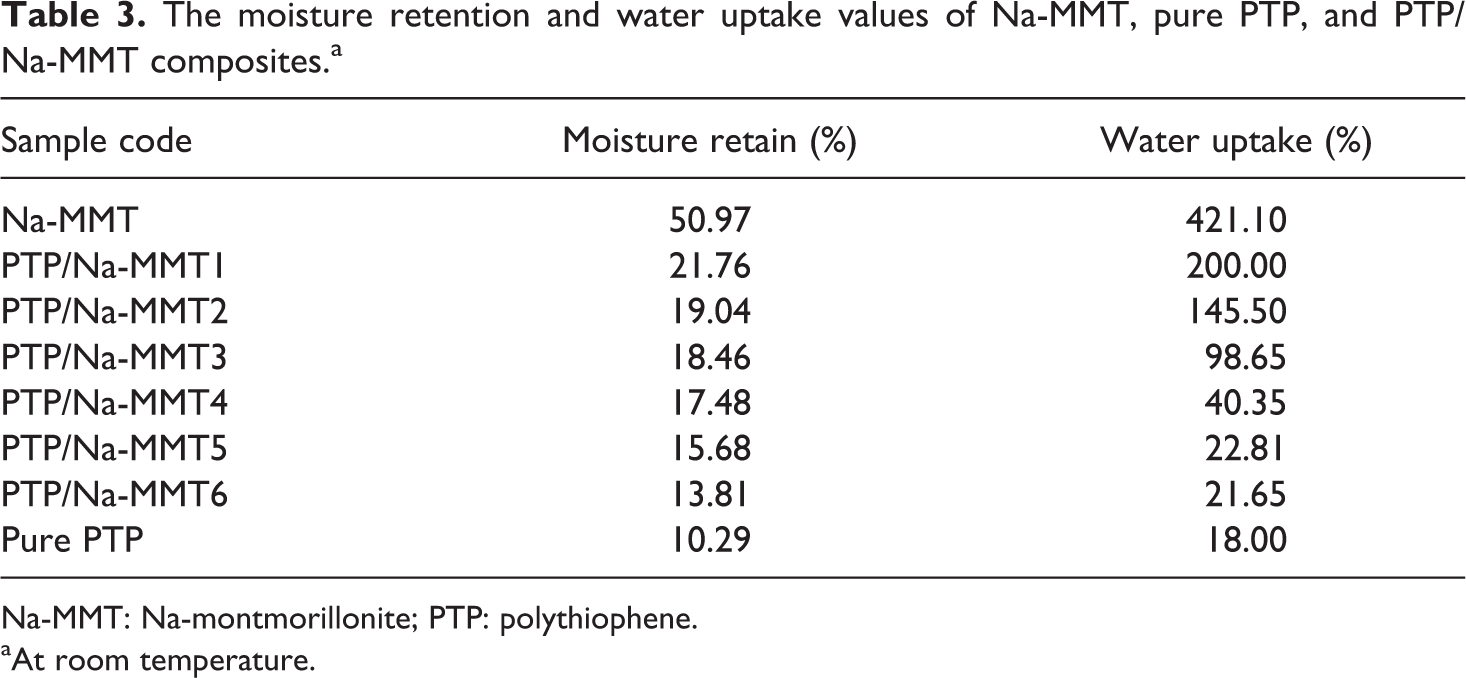
Na-MMT: Na-montmorillonite; PTP: polythiophene.
aAt room temperature.
The decreases in moisture retention and water uptake can be attributed to the hydrophobic character of the PTP and the hydrophilicity of Na-MMT has been changed to an organophilic nature. The decrease in water uptake means the material has more stable properties, which is interesting for practical applications. 22 The decrease in water uptake can be described by the fact that the percentage of clay in the composites is being limited, which reflects that the quantity of the polymer introduced in the layers reaches a limit and is enough to achieve maximum opening of the interlayers of clay 23,24 and the formation of a cross-linked structure to a certain extent, which prevents the insertion of water molecules into the structure. Finally, water resistance of these composites can be greatly improved.
Conductivity properties
The electrical conductivity measurements were conducted at room temperature. The probes were placed on the surface of the pellet of the samples. Figure 9 showed variation in the electrical conductivity of PTP/Na-MMT composites, including different contents of TP. The conductivity of all the composites increased with increasing amount of conducting PTP in the composite. 3,14,25 It was seen that the conductivity increased up to 71.43 mass% of TP in composite 2,6,21 and then percolated to a value of 8.8 × 10− 6 S cm−1 by 75.00 mass% TP.
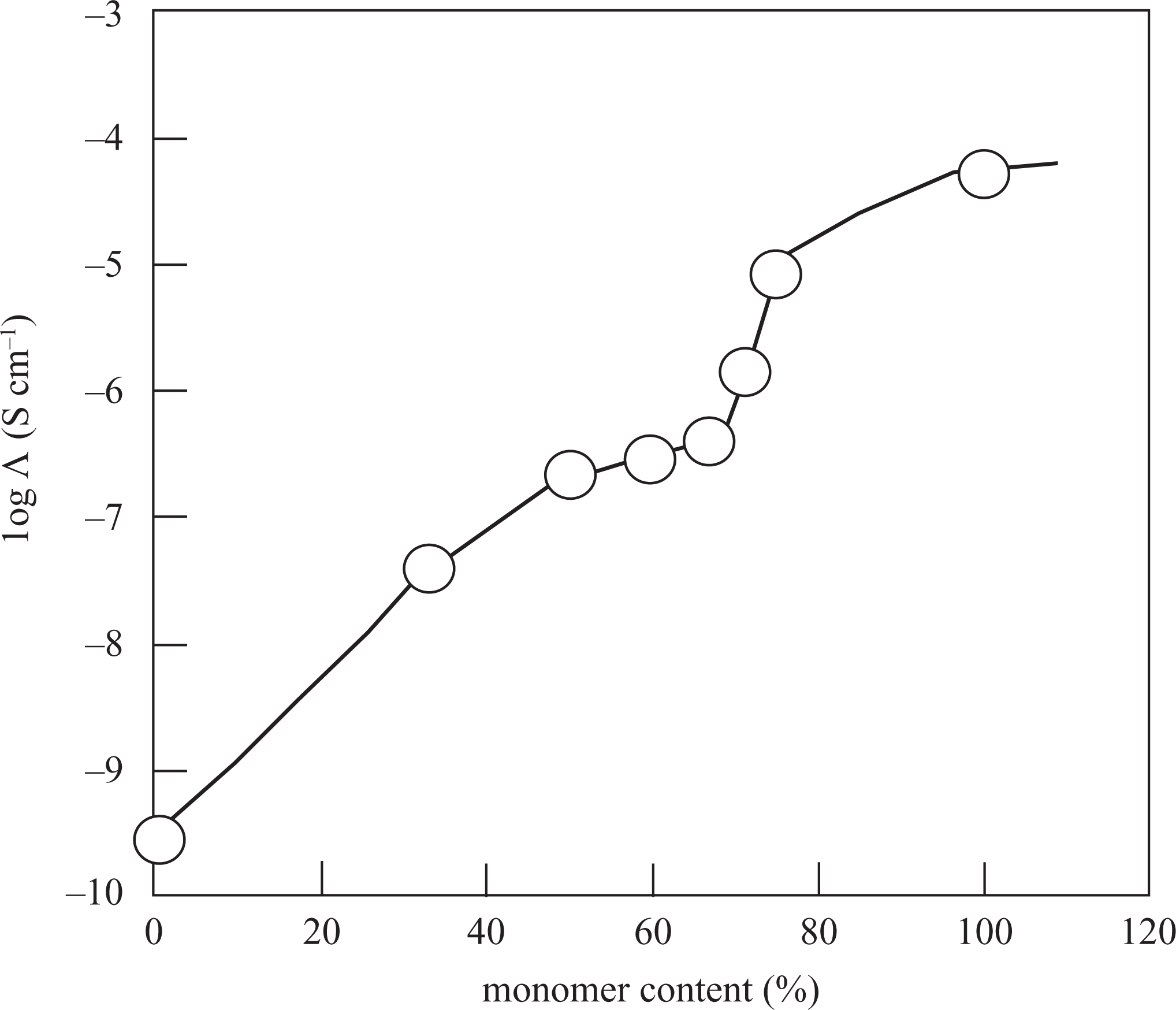
The conductivitiy values of Na-MMT, pure PTP, and PTP/Na-MMT composites including different percentages of TP. Na-MMT: Na-montmorillonite; PTP: polythiophene; TP: thiophene.
Conclusions
Using intercalative polymerization system, PTP/Na-MMT composites was successfully synthesized in an aqueous medium. FTIR, XRD, SEM, TEM, TGA, and DTA techniques were used to characterize PTP/Na-MMT composites. From the TGA and DTA results, we observed that the Na-MMT clay induced the thermal stability. Intercalated composites showed improved thermal stability when compared with pure PTP and Na-MMT clay induced thermal stability. Apparently, the overall thermal stability trends was found to be Na-MMT > PTP/Na-MMT3 > PTP/Na-MMT4 > pure PTP. From the XRD patterns and TEM micrograph, we observed the intercalation of PTP between the Na-MMT clay layers in nanoscale. XRD results also demonstrated the intercalation of conducting PTP between the interlayers of Na-MMT and supported the formation PTP/Na-MMT composites. Incorporation of PTP in the clay structure was determined by FTIR spectroscopy. Adsorption, moisture retention, and water uptake values of composites obviously decreased when compared with that of Na-MMT. A significant decrease in these parameters with increasing monomer content was observed. We also measured the room temperature electrical conductivity of PTP/Na-MMT composites. The conductivities of PTP/Na-MMT composites increased with the increase in TP loading in the composite. Among the composites, PTP/Na-MMT6 composite (containing 75.00 mass% TP) has the highest electrical conductivity.
Footnotes
Funding
This work was supported by the Turkish Scientific and Research Institute for funding a project (no. 108T241) and to Ankara University Research Fund for their financial support (no. 20040705087) to this work.
