Abstract
In this work, a novel polymer magnetic composite based on thermoplastic acrylate pressure-sensitive adhesive (acrylate PSA) filled with the nickel plating multiwall carbon nanotubes (Ni/MWNTs) were prepared using Ni/MWNTs obtained by electroless plating method. Then their morphology and properties were characterized by transmission electron microscope, scanning electron microscope (SEM), X-ray diffraction (XRD), vibrating sample magnetometer (VSM) and so on. Both SEM and XRD indicate that a large number of nickel particles are homogenously coated on the surface of MWNTs. SEM reveals that the Ni/MWNTs are homogeneously dispersed in thermoplastic acrylate PSA. The VSM show that the saturation magnetization (M s) of the Ni/MWNTs is 92.0 emu/g and the increase in the Ni/MWNTs content could enhance the M s of the thermoplastic acrylate PSA/Ni/MWNTs composites and decrease their coercivity (H c). When the content of Ni/MWNTs is 10.0 wt%, the magnetic and mechanical properties of the thermoplastic acrylate PSA/Ni/MWNTs composites are optimum.
Keywords
Introduction
Recently, electromagnetic (EM) waves have been used in various industrial applications, so that the demand for utilization of microwaves of GHz range has drastically increased, and some electromagnetic interference (EMI) problems are serious and also has baneful influences on human bodies. 1 Electromagnetic wave (EMW)-shielding materials are effective in reducing the EMI and not only are valued by people in its application in military, but simultaneously also have an extremely widespread application prospect in the civil aspect. 2 –4 For EMW-shielding materials, typical metals (like cobalt, iron, nickel and aluminum) have been described in the prior research works, 5 –8 which possess high conductivity and high magnetic properties. But their disadvantages such as high density, easy corrosion and poor processability restrict their high-frequency applications.
To attain better efficiency in EMW shielding, polymer magnetic composites with low density, designable and dimension stability have been suggested and developed. 9 –12 The polymer magnetic composites are usually composed of two parts. One is the polymer that provides favorable compatibility. The other is a magnetic filler, which is often the inorganic magnetic nanoparticle, such as Co, 13 Ni, 14 Fe3O4 15 and Er2O3. 16 Carbon nanotubes (CNTs) are popular fillers because of their exceptional mechanical, thermal and electrical properties. 17 –20 However, excellent magnetic properties cannot be obtained for unmodified CNTs because their maximum magnetization saturation is small. In order to optimize the performance of CNTs as magnetic fillers, it is necessary to modify CNTs by coating and filling with other nanomaterials. Nickel powder is a typical magnetic filler of high conductivity and high-saturation magnetization. But its disadvantages are high density and high cost. 21 Nickel plating multiwall carbon nanotubes (Ni/MWNTs) should be an effective approach to obtain the magnetic filler with high magnetic properties and moderate cost.
For polymer magnetic composites, the polymers that are widely studied and reported are thermosetting resins such as phenolic resin, 22 epoxy resin 23 and silicone resin 24 in recent years. Thermoplastic acrylate pressure-sensitive adhesive (PSA) is permanently tacky and can quickly adhere to solid surfaces because it does not undergo any physical transformation or chemical reaction during the bonding process. Therefore, the advantages of thermoplastic acrylate PSA include the convenience of use, environmental friendly and low cost compared with the other polymers. 25 However, studies of polymer magnetic composites based on thermoplastic acrylate PSA are very limited. In this study, polymer magnetic composites obtained comprised thermoplastic acrylate PSA with Ni/MWNTs prepared by electroless plating method. The morphology and properties of the Ni/MWNTs and the thermoplastic acrylate PSA/Ni/MWNTs composites were characterized by transmission electron microscope (TEM), scanning electron microscope (SEM), X-ray diffraction (XRD), vibrating sample magnetometer (VSM), and so on.
Experimental
Materials
Butyl acrylate (BA), methyl acrylate (MA), acrylic acid (AA) and hydroxyethyl acrylate (HEA) were purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China), and were purified by washing with aqueous sodium hydroxide (NaOH) solution and deionized water, followed by distillation under reduced pressure. Rosin resin was supplied by Sinopharm Chemical Reagent Co., Ltd and was used as a tackifier resin. The initiator 2,2-azodiisobutyronitrile (AIBN) was purchased from Shanghai Pu Shan Chemical Co., Ltd (Shanghai, China), and was purified by recrystallization in methanol. 2-Butanone (MEK), provided by Tianjin Fu Yu Fine Chemical Co., Ltd, was used in all the polymerization procedures.
The MWNTs with a purity of 90% used in the present study, synthesized via catalyst-assisted chemical vapor deposition (CVD) process, were purchased from Chengdu Alpha Nano Technology Co., Ltd (Chengdu, China). NaOH, 98% nitric acid (HNO3), 36% hydrochloric acid (HCl), stannous chloride (SnCl2), palladium dichloride (PdCl2), boric acid (H3BO3), sodium hypophosphite (NaH2PO2·H2O), nickel sulfate (NiSO4·6H2O), 28% ammonia (NH3·H2O) and malic acid (HOOC–CHOH–CH2–COOH) were provided by Sinopharm Chemical Reagent Co., Ltd. Distilled water was used in all the electroless plating procedures.
Sample preparation
The crude MWNTs were mixed with 3 M HNO3 and refluxed for 24 h at 105°C, and were then suspended and refluxed in 5 M HCl for 10 h at 110°C. After acid treatment, the samples were calcined in static air at 500°C with the reaction time of about 1.0 h, and the purified MWNTs were obtained.
After the purification procedure, the Ni/MWNTs were prepared using the following procedure. A weighed amount (1 g) of purified MWNTs were oxidized in 100 ml of NaOH (2 mol/L) at 40°C for 2 h. After rinsing with distilled water, the oxidized MWNTs were sensitized in 50 ml solution consisting of SnCl2 (0.04 mol/L) and HCl (0.5 mol/L) at room temperature for 1 h. Afterward, the sensitized MWNTs were activated in 50 ml activator solution containing PdCl2 (0.002 mol/L), HCl (0.5 mol/L) and H3BO3 (0.3 mol/L) at room temperature for 1 h. The treated MWNTs were rinsed with distilled water until their pH was about 7 and then dried completely at 110°C. Subsequently, the activated MWNTs were immersed in 150 ml solution consisting of NiSO4·6H2O (0.1 mol/L), C4O5H6 (0.2 mol/L) and NH3·H2O (0.5 mol/L), with the addition of appropriate drops of 50 ml NaH2PO2·H2O (0.2 mol/L) solution and stirred for 1 h by maintaining the temperature of the reaction at 80°C. The nickel plating process was illustrated in Figure 1.
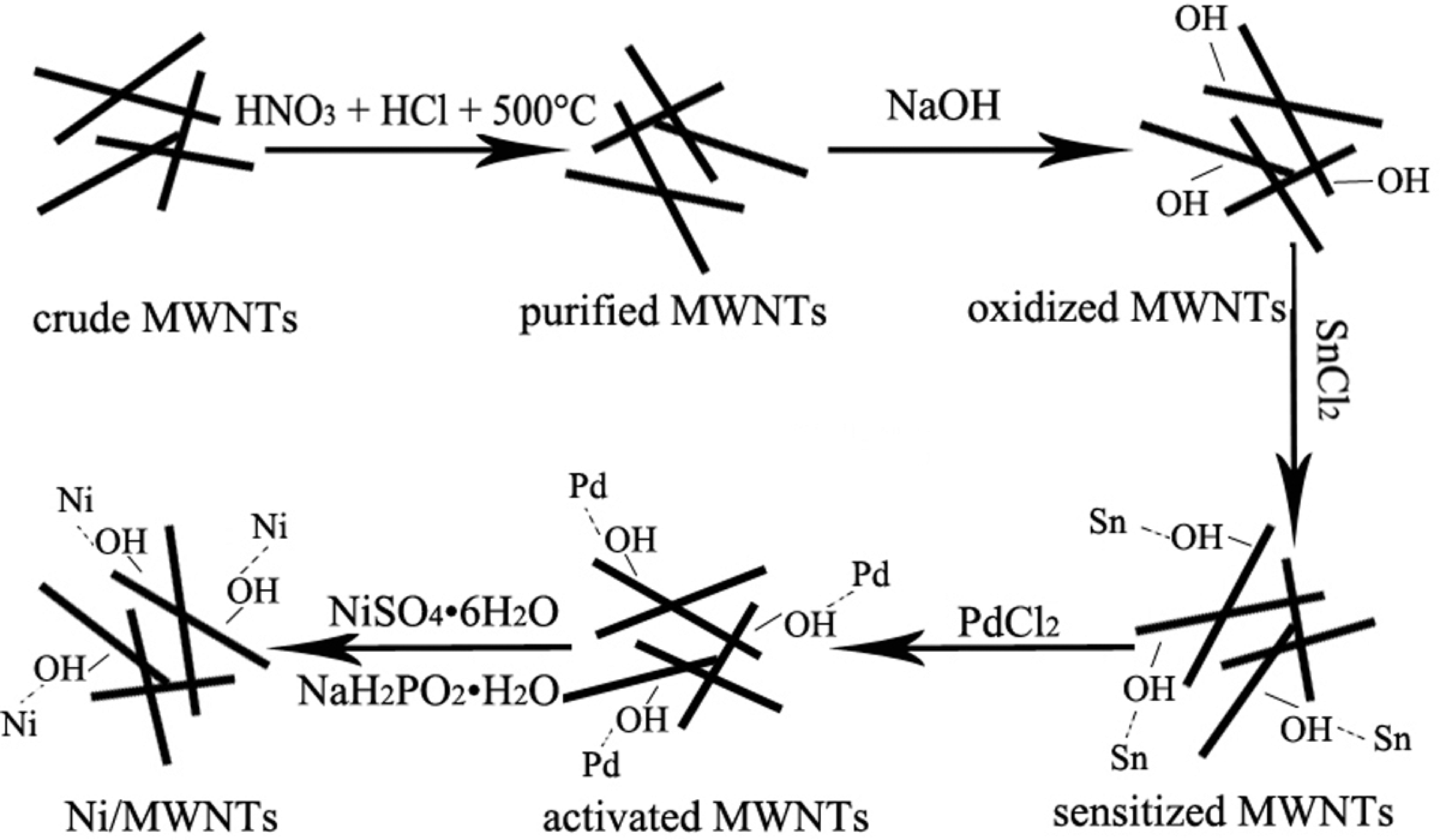
Schematic illustration of the nickel plating process.
A typical procedure for solution polymerization of thermoplastic acrylate PSA is described as follows. First, 50.0 g MEK was placed in a 250-ml, separable four-necked flask equipped with an efficient stirrer, and a reflux condenser in a nitrogen atmosphere. The MEK was stirred at 300 rad/min and was heated to 78°C. Then, a mixture of 55.8 g BA, 33.5 g MA, 8.0 g AA, 2.7 g HEA and 1.5 g AIBN was added to the flask. The reaction mixture was stirred at 300 rad/min and at 80°C for 2.5 h. Next, 1.5 g of AIBN was added to the flask and the temperature of the reaction was maintained at 80°C for 3.5 h. Finally, 10.0 g rosin resin was added to the mixture after the mixture was cooled to approximately 25°C and the thermoplastic acrylate PSA was obtained.
The thermoplastic acrylate PSA/Ni/MWNTs composites were prepared by mixing the thermoplastic acrylate PSA and the Ni/MWNTs at room temperature for 60 min in an efficient stirrer that was operating at the stirring rate of 300 rad/min. To obtain homogeneous dispersion and to prevent the Ni/MWNTs settling in thermoplastic acrylate PSA, the thermoplastic acrylate PSA/Ni/MWNTs composites were placed in a ultrasonic bath for 2 h.
Characterization and property
The TEM (H-800 model, Hitachi Co., Ltd, Japan) was used to analyze the internal morphology of the crude MWNTs and the purified MWNTs.
SEM (JSM-6360LV model, JEOL Ltd, Japan) and energy-dispersive spectroscopy images of the purified MWNTs, the Ni/MWNTs and the thermoplastic acrylate PSA/Ni/MWNTs composites were collected by JSM-6360LV.
The XRD was performed using a XRD-7000 instrument (X’ Pert MPD PRO type, Panalytical Co., Ltd, Holland) at a scan rate of 0.02°. The X-ray pattern was collected for 2θ from 10° to 80°.
Magnetic properties of the Ni/MWNTs and the thermoplastic acrylate PSA/Ni/MWNTs composites were examined at room temperature using the VSM (Lakeshore 7307, The Nanjing University Instrument plant, China).
The 180° peel strength and the shear strength of the thermoplastic acrylate PSA/Ni/MWNTs composites were measured with computer-controlled electronic tensile testing machine (CMT-8502 model, Shenzhen Sansi Co., Ltd, China). The 1Cr13 stainless steel was coated with the PSA film to achieve a thickness of 50 μm at room temperature (23 ± 2°C) and 65 ± 5% relative humidity.
Results and discussion
Structures of the crude MWNTs, the purified MWNTs and the Ni/MWNTs
Various synthesis methods have been developed for the production of CNTs. However, the raw materials prepared by these methods contain impurities. Purification of CNTs is important for applications because of their excellent intrinsic properties. To verify the validity of purification treatment for MWNTs, we performed TEM imaging of MWNTs both before and after purification treatment. Results from this analysis are shown in Figure 2. Figure 2(a) is a typical TEM image of the crude MWNTs. From Figure 2(a), we can see that the MWNTs are black hollow tubes with a diameter ranging from 40–60 nm and some dark spots and dark lumps adsorbed onto the tips and sidewalls of MWNTs. The dark spots represent metal catalyst nanoparticles and the dark lumps are the amorphous carbon. The impurities in MWNTs contain metal catalyst nanoparticles and amorphous carbon. Figure 2(b) shows the TEM image of the purified MWNTs. As shown in Figure 2(b), almost all metal catalyst nanoparticles and the amorphous carbon are removed and the bundles of MWNTs have clean tube walls. The results indicate that the purification treatment is effective.

Transmission electron microscope (TEM) images of the crude multiwall carbon nanotubes (MWNTs) (a) and the purified MWNTs (b).
The Ni/MWNTs are prepared by electroless plating which is on the basis of transferring metal obtained from solution to substrate surface by reducing the metal ion using chemical agent. The SEM images of the MWNTs and the Ni/MWNTs are shown in Figure 3. Figure 3(a) shows that the purified samples have a large number of CNTs with a typical sausage-like structures. Almost no amorphous carbon and metal catalyst nanoparticles can be observed. It is difficult to measure the accurate length of the MWNTs from the SEM observation because of their twisting morphology, but the length can be estimated to be more than several tens of micrometers. The diameter of the MWNTs is about 50 nm. Figure 3(b) is the SEM image of the Ni/MWNTs. Figure 3(b) shows that in addition to the MWNTs bundles, a large number of white spots are homogenously coated on the surfaces of the MWNTs and the white spots indicate the nickel particles that show oblate form. We also observed that the size of the Ni/MWNTs is estimated to be around 150 nm in diameter and calculated the thickness of the coating layer to be about 50 nm.

Scanning electron microscope (SEM) images of the purified multiwall carbon nanotubes (MWNTs) (a) and the Ni/MWNTs (b).
In order to analyze the content of Ni and P on the surface of MWNTs, we performed energy-dispersive (EDS) spectrum of Ni/MWNTs. Figure 4 shows the EDS result of the Ni/MWNTs. From the EDS spectrum, it can be seen that the major components of the Ni/MWNTs are Ni and C. The elements P and O are also the components of the Ni/MWNTs. The elementary analysis for the Ni/MWNTs shows that the content of Ni, C, P and O in the Ni/MWNTs is 70.52, 19.14, 4.24 and 6.10 wt%, respectively. The content of Ni on the surface of the MWNTs is very high, which is an indication that the Ni/MWNTs have been successfully prepared.
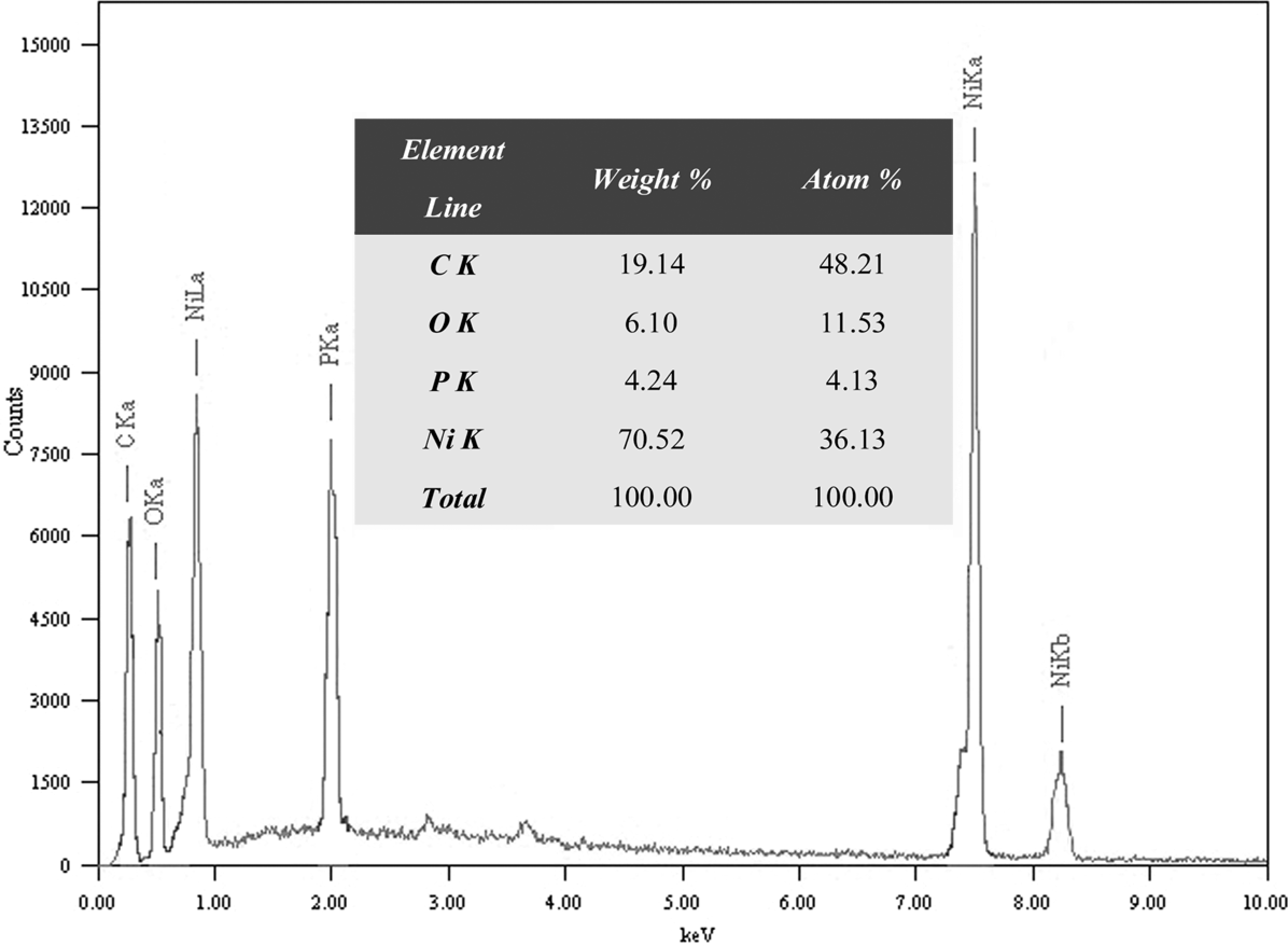
Energy-dispersive (EDS) spectrum of the Ni/MWNTs. MWNTs: multiwall carbon nanotubes.
The XRD patterns of the crude MWNTs, the purified MWNTs and the Ni/MWNTs are shown in Figure 5. In the diffraction pattern (Figure 5(a)) of the crude MWNTs, a sharp and intense peak at 2θ = 26.4° and a weak peak are observed, which correspond to (0 0 2) and (1 1 0) planes of typical graphitized MWNTs, respectively. Figure 5(a) also shows 2 weak peaks at 2θ = 44.5° and 76.4°, and they are (1 1 1) and (220) planes of Ni crystals, which indicate that the metal catalyst residuals of the crude MWNTs obtained by CVD method are Ni nanoparticles. After the purification process (Figure 5(b)), the (1 1 1) and (2 2 0) planes disappeared, demonstrating elimination of the metal catalyst residuals. The fact that the (0 0 2) and (1 1 0) planes in Figure 5(b) are stronger than that shown in Figure 5(a) reveals the amorphous carbon coating the MWNTs surface is consumed by oxidation reaction. As shown in Figure 5(c), 2 strong peaks at 2θ = 44.5° and 76.4° are clearly observed, which can be well indexed to the (1 1 1) and (2 2 0) planes of cubic fcc-type Ni crystals (JCPDS 65-2865). And the intensity of the typical diffraction peaks of MWNTs in Figure 5(c) has been greatly weakened because the surface of purified MWNTs is covered by a large number of Ni particles.
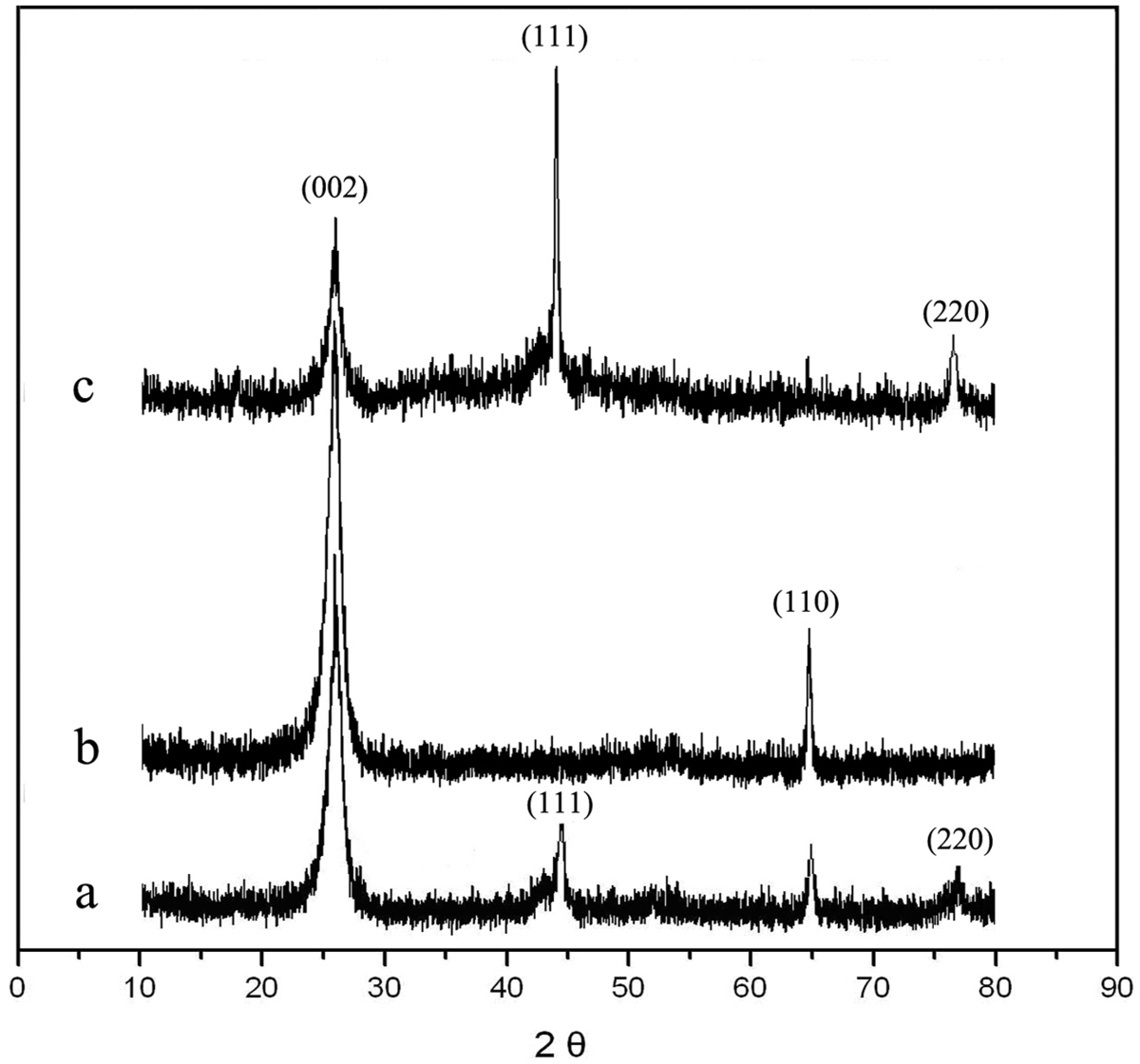
X-ray diffraction (XRD) patterns of the crude multiwall carbon nanotubes (MWNTs) (a), the purified MWNTs (b) and the Ni/MWNTs (c).
Magnetic properties of the purified MWNTs and the Ni/MWNTs
The magnetic properties of the purified MWNTs and the Ni/MWNTs are measured at room temperature on a VSM. Diamagnetic characteristic was observed for the purified MWNTs, with very weak magnetization toward applied magnetic field, and the M s is only 0.026 emu/g (inset in Figure 6). The magnetization–coercivity curve of the Ni/MWNTs in Figure 6 indicates the M s of the Ni/MWNTs is 92.0 emu/g, which is three orders of magnitude higher than that of the MWNTs. The M s of the Ni/MWNTs is higher than that of other fillers. 26 –28 It can be predicted that higher M s can be obtained if the MWNTs are fully coated with well-dispersed Ni particles.
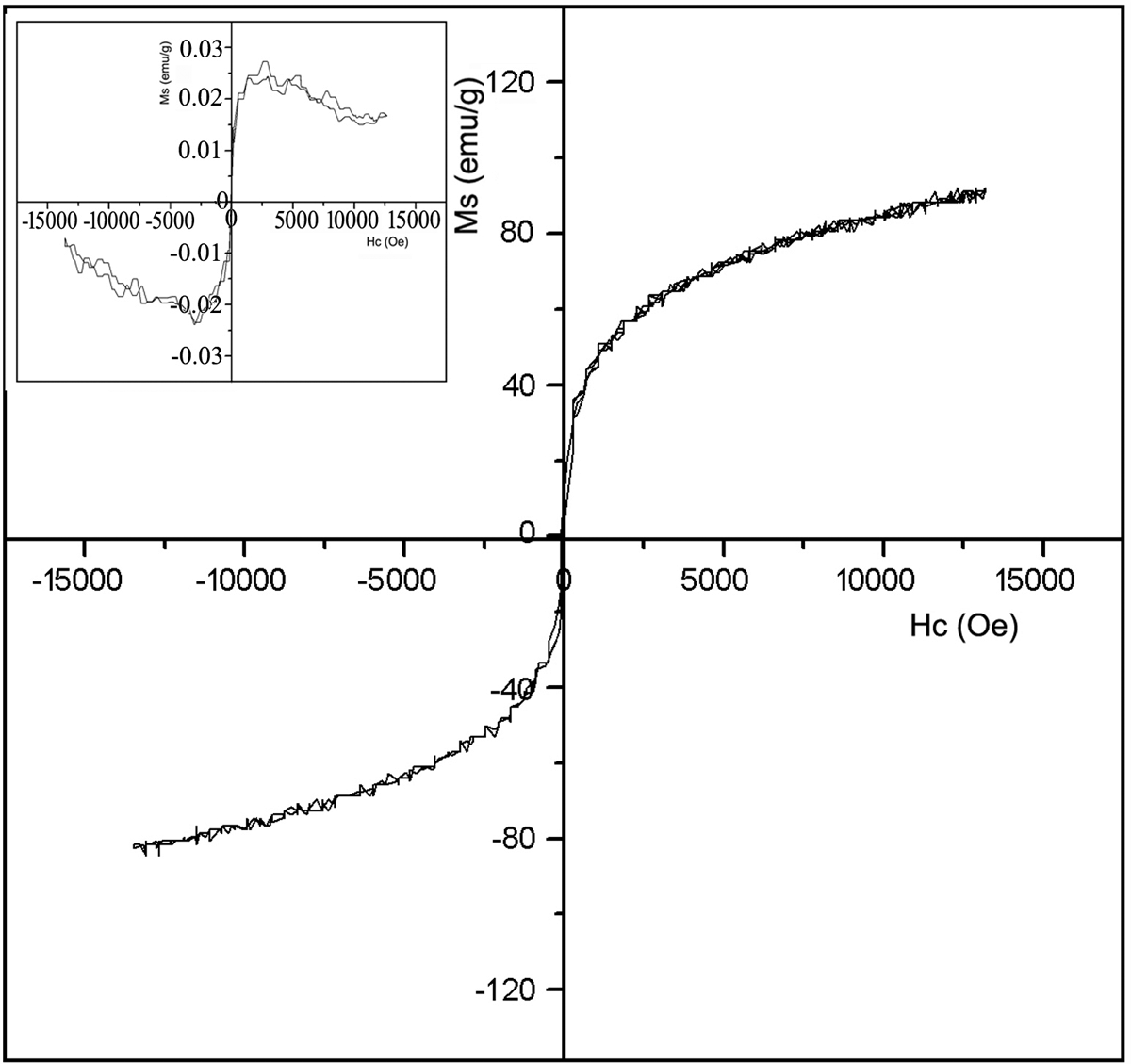
Magnetization–coercivity curves of the purified multiwall carbon nanotubes (MWNTs; inset) and the Ni/MWNTs.
Structure of the thermoplastic acrylate PSA/Ni/MWNT composites
The microstructural characteristics of pure thermoplastic acrylate PSA and the thermoplastic acrylate PSA/Ni/MWNTs composites with 10.0 wt% Ni/MWNTs loading are examined using SEM and the results are displayed in Figure 7. The SEM characterizations of thermoplastic acrylate PSA and the thermoplastic acrylate PSA/Ni/MWNTs composites are done using brittle fractured composites prepared using liquid nitrogen. As shown in Figure 7(a), the cross-section of the pure thermoplastic acrylate PSA is smooth. The SEM image (Figure 7(b)) of the brittle fractured thermoplastic acrylate PSA/Ni/MWNTs composites clearly reveals the distribution of Ni/MWNTs in the matrix. It was observed that the Ni/MWNTs displayed high dispersion ability in the acrylate matrix. Dispersion of the Ni/MWNTs in the acrylate matrix has been identified through the bright dots and the lines in gray areas.

Scanning electron microscope (SEM) images of pure acrylate resin (a) and the thermoplastic acrylate PSA/Ni/MWNTs composites (b). MWNTs: multiwall carbon nanotubes; PSA: pressure-sensitive adhesive.
Magnetic properties of the thermoplastic acrylate PSA/Ni/MWNTs composites
Figure 8 shows the magnetization–coercivity curves of the thermoplastic acrylate PSA/Ni/MWNTs composites with 10.0, 20.0 and 30.0 wt% Ni/MWNT. The values of the M s and the H c of all samples, derived from the magnetization–coercivity curves, are given in Table 1. It can be seen that the M s of the thermoplastic acrylate PSA/Ni/MWNTs composites with 10.0, 20.0 and 30.0 wt% Ni/MWNTs is 11.1, 15.8 and 25.7 emu/g, respectively. The results indicate that the M s of the composites increases with an increasing content of the Ni/MWNTs, attributed mainly to the increasing relative content of Ni/MWNTs per unit mass. While the H c of the thermoplastic acrylate PSA/Ni/MWNTs composites decreases with the increase in the content of Ni/MWNTs. The H c of composites with 10.0, 20.0 and 30.0 wt% Ni/MWNTs is 35.3, 29.5 and 9.8 Oe, respectively. According to the ferromagnetic resonance theory, 29 H c is related to its anisotropy that includes magnetic anisotropy, stress anisotropy and shape anisotropy. The thermoplastic acrylate PSA/Ni/MWNTs composites’ shape and stress anisotropy field can be negligible. So the increase in the content of Ni/MWNTs could enhance the exchange coupling that weaken disordered magnetic anisotropy, thus reducing the H c.
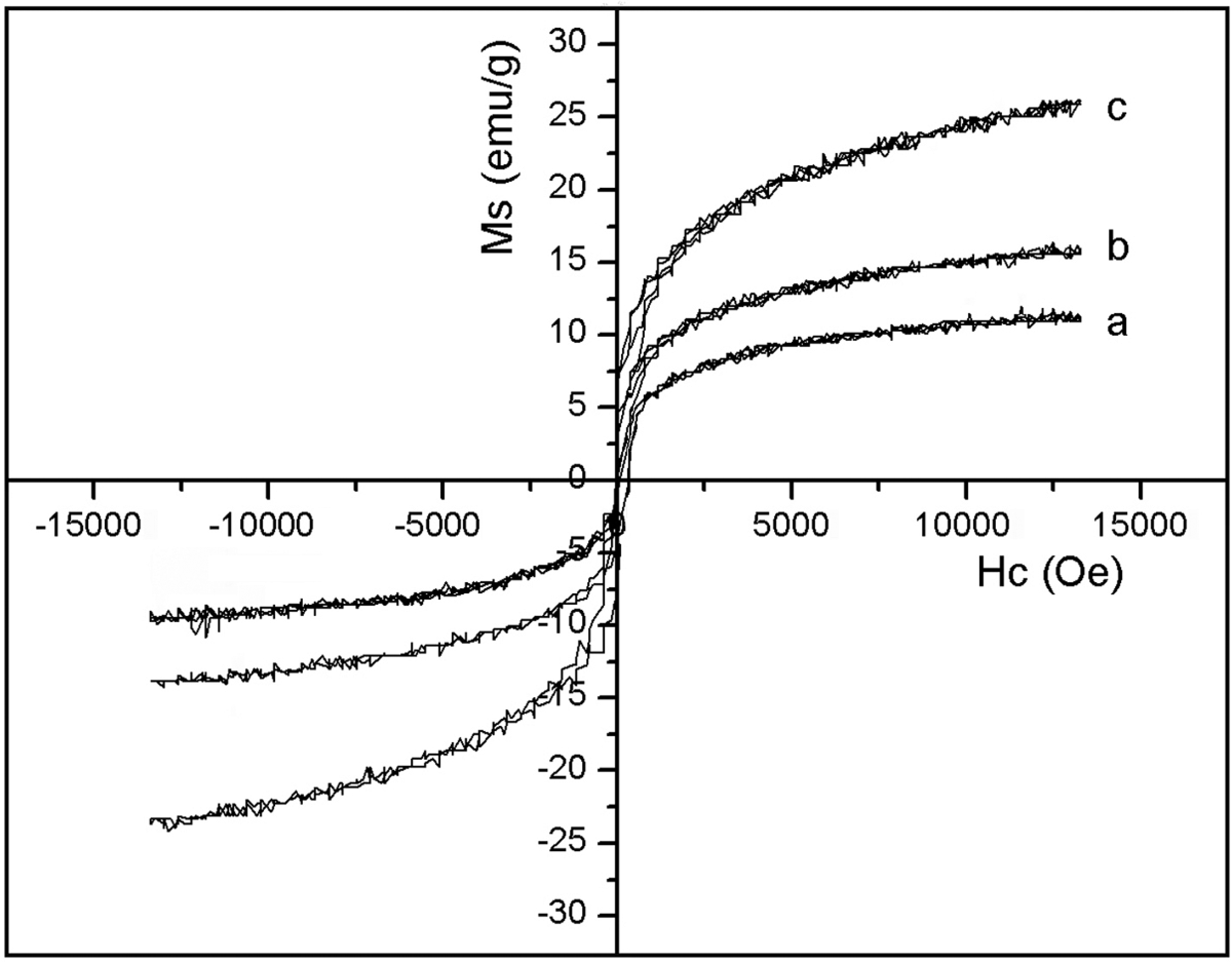
Magnetization–coercivity curves of the thermoplastic acrylate PSA/Ni/MWNTs composites under various contents of the Ni/MWNTs (a: with 10.0 wt% Ni/MWNT, b: with 20.0 wt% Ni/MWNT, c: with 30.0 wt% Ni/MWNT). MWNTs: multiwall carbon nanotubes; PSA: pressure-sensitive adhesive.
The magnetic properties of the thermoplastic acrylate PSA/Ni/MWNTs composites under various contents of the Ni/MWNTs.

MWNTs: multiwall carbon nanotubes; PSA: pressure-sensitive adhesive.
Mechanical properties of the thermoplastic acrylate PSA/Ni/MWNTs composites
The basic mechanical properties included the 180° peel strength and the shear strength of the thermoplastic acrylate PSA/Ni/MWNTs composites, which are measured with computer-controlled electronic tensile testing machine at 25°C at a peel rate of 300 mm/min. The dependencies of the 180° peel strength and shear strength of the thermoplastic acrylate PSA/Ni/MWNTs composites on the content of the Ni/MWNTs are shown in Figure 9. Figure 9(a) shows that the 180° peel strength decreases from 1.171 to 0.194 KN/m when the Ni/MWNTs content increases from 0 to 30.0 wt%. The 180° peel strength is mainly related to the adhesion strength between the adhesive and the surface of the testing panel. The increment in the Ni/MWNTs content could reduce the adhesion strength between the adhesive and the testing panel, thus decreasing the 180° peel strength. As shown in Figure 9(b), the shear strength of thermoplastic acrylate PSA/Ni/MWNTs composites with 0, 10.0, 20.0 and 30.0 wt% Ni/MWNTs is 0.662, 0.732, 0.567 and 0.321 MPa, respectively. The shear strength is related to the cohesive strength of the adhesive. With the increase in the content of Ni/MWNTs, there is an improvement in the rigidity of the polymer chains which in turn increases the cohesive strength. However, when the content of the Ni/MWNTs is high, the fillers are not dispersed well in the composites, resulting in a decrease in their shear strength. Therefore, the shear strength increases first, then gradually decreases. Consequently, considering the 180° peel strength and shear strength at the same time, the mechanical properties of the thermoplastic acrylate PSA/Ni/MWNTs composites are optimum (180° peel strength of about 0.911 MPa and shear strength of about 0.732 MPa) when the content of the Ni/MWNTs is 10.0 wt%, which is excellent and can satisfy the requirements of use as viscoelastic materials in comparison with some other researcher’s results about PSA. 30,31
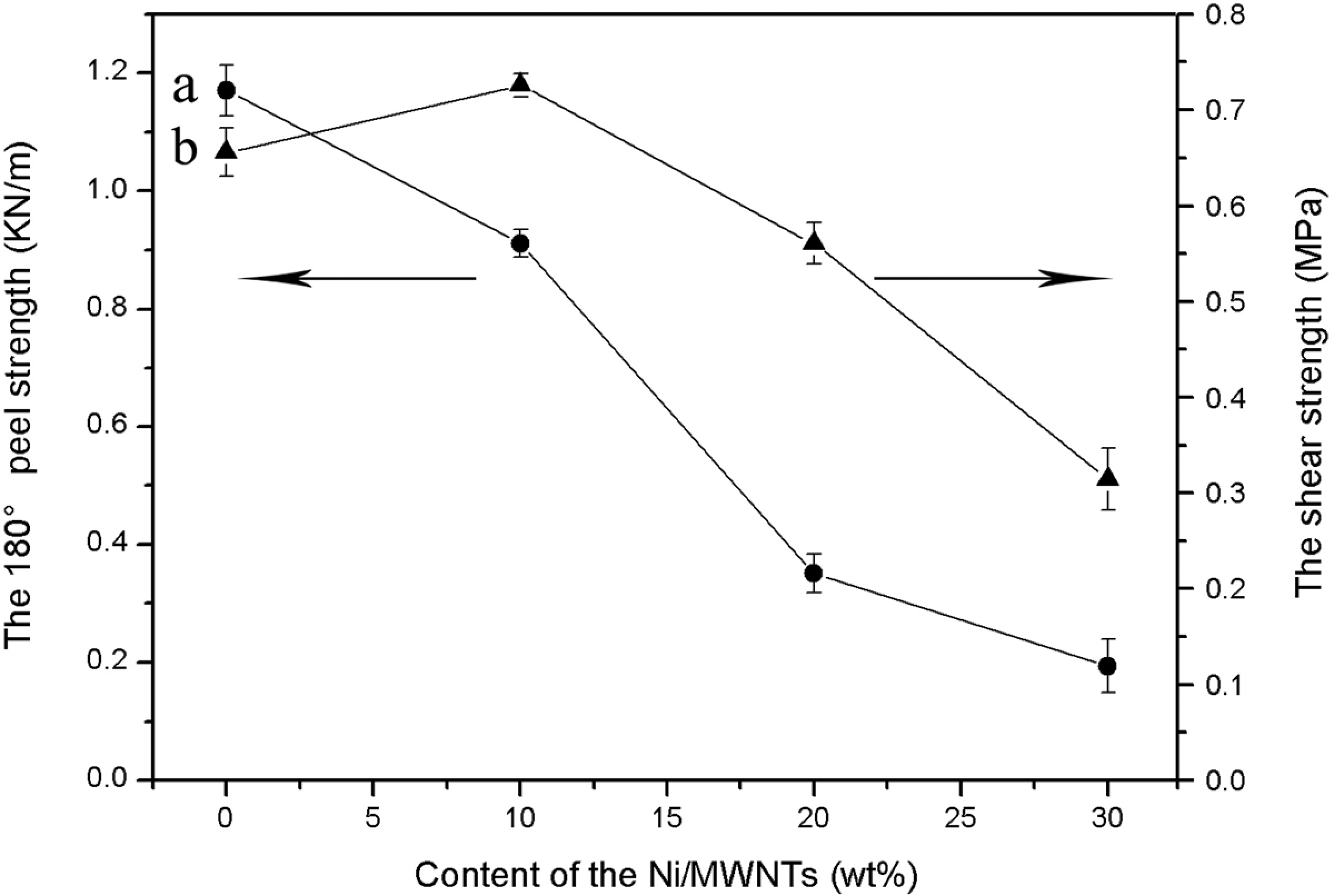
The 180° peel strength (a) and the shear strength (b) of the thermoplastic acrylate PSA/Ni/MWNTs composites under various contents of the Ni/MWNTs. MWNTs: multiwall carbon nanotubes; PSA: pressure-sensitive adhesive.
Conclusions
In this study, the Ni/MWNTs were successfully prepared via electroless plating method. The thermoplastic acrylate PSA/Ni/MWNTs composites were obtained by mixing the Ni/MWNTs and thermoplastic acrylate PSA at room temperature. TEM images, SEM results and XRD results indicate that impurities in the MWNTs were removed after the purification treatment and a large number of nickel particles were homogenously coated on the surfaces of the purified MWNTs. The elementary analysis for the Ni/MWNTs shows that the content of Ni and P in the Ni/MWNTs is 70.52 and 4.24 wt%, respectively. According to the VSM test, the M s of the Ni/MWNTs is 92.0 emu/g and the M s of the thermoplastic acrylate PSA/Ni/MWNTs composites increases while their H c decreases with an increasing content of the Ni/MWNTs. SEM images reveal that the Ni/MWNTs are homogeneously dispersed in thermoplastic acrylate PSA. The increment in the Ni/MWNTs content could decrease 180° peel strength and enhance the shear strength when the content of the Ni/MWNTs is low. When the content of the Ni/MWNTs is 10.0 wt%, the properties of the thermoplastic acrylate PSA/Ni/MWNTs composites are optimum (M s of about 11.1 emu/g, 180° peel strength of about 0.911 MPa and shear strength of about 0.732 MPa).
Footnotes
Acknowledgements
The authors would like to thank the Shaanxi Materials Analysis and Research Center for carrying out the XRD, TEM and SEM characterization. The authors also thank their colleagues in their laboratory for their support.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
