Abstract
Poly(vinylidene fluoride-co-hexafluoropropylene) (PVDF-HFP) nanocomposites with different contents of nanoclays were prepared by melt mixing. The clays used included unmodified clays, a commercially available hydrophilic nanoclay based on bentonite (nanoclay 1) and organically modified clay based on montmorillonite (nanoclay 2). The nanocomposites obtained were analyzed by x-ray diffraction, differential thermal and differential scanning calorimetry analyses and their basic tensile and other characteristics were determined. The distribution of the filler throughout the matrix was found to be good for both the types of nanocomposite materials. Nanoclay 1 induced slight increase in the β-phase and higher deviation of the main diffractions (2θ = 18.6° and 20.1° at 10 mass% filler). The degree of crystallinity of the materials containing nanoclay 2 decreased from 26.4 to 20.5% and from 23.9 to 18.7%, respectively, for the first and second heating. The tensile strength of the materials based on PVDF-HFP with nanoclays 1 and 2 was close to that of the initial copolymer or slightly increased with the addition of nanoclay 2. Young’s modulus was in the range of 215–265 MPa. The heat resistance by Vicat increased by 7°C compared with the initial copolymer.
Keywords
Introduction
Polymer nanocomposites are a new class of materials attracting much attention in the recent decade. It was due to their improved properties compared with those of the initial polymers and classic composite materials. These materials contain ultrafine phase (particle size 1–100 nm) dispersed in the thermoplastic or thermoreactive matrix. The use of small quantities of nanofillers (1–5%) allows imparting properties comparable or better than those achieved using traditional fillers at the concentrations of 15–40%. 1
With regard to the nano-sized particles used so far, clays (and especially lamellar silicates) are of special interest due to their frequent occurrences in nature and low cost. They are composed of several layers packed parallel to each other to form lamellae of thickness of about 1 nm and length in the order of several micrometers. It means that each lamella has high ratio length/thickness that facilitates energy transfer from inorganic-to-organic phase and back. A number of studies on these materials have shown that polymer nanocomposite materials have new, sometimes improved properties that are not exhibited by the individual phases or traditional composite materials. 2 –11 The most effective improvement in the properties was achieved when only 1-nm-thick layers of nanoclays were dispersed within the polymer matrix. Unfortunately, some polymers are not exfoliated between nanoclay sheets. For this reason, improvement in properties was not observed for many nanomaterials. The nanoclay particles would probably glue together in agglomerates or penetrate partially into the polymer to form intercalation structure.
Fluoropolymers such as poly(vinylidene fluoride) (PVDF) and its copolymers with hexafluoropropylene (HFP) are thermoplastics of high thermal stability and are technologically important because of their availability in different crystalline forms. Introducing HFP units to the PVDF backbone generates copolymer that exhibit certain advantages compared with the homopolymer such as enhanced piezoelectricity and improved mechanical behavior. 12,13 Both PVDF and PVDF-HFP fluoropolymers are semicrystalline. The crystalline structure of the PVDF-HFP is similar to that of PVDF. The degree of crystallinity of the PVDF-HFP is sufficiently reduced in comparison with pure PVDF, whereas the flexibility and chemical resistance are enormously enhanced. 14,15
The first data on nano-sized composites based on nanoclays and PVDF were published by Priya and Jog. 16 –18 They used PVDF (Solef 1008) and two different types of nanoclays (Cloisite® 6A and Cloisite 20A) at contents of 1.5, 3.5 and 7%. These polymer composites were obtained from nanoclay and PVDF melt in a ‘Brabender’ mixer. They found that the degree of crystallization of the composites was much higher than that of the initial polymer. The composites obtained showed increased temperatures of melting (by 6–8°C) and crystallization (by 10–12°C) as well as increased degree of crystallization and decreased crystallinity by about 17%.
Liu et al. 19 studied the use of organically modified nanoclays based on montmorillonite (MMT) and PVDF of high molecular weight (M w = 530,000 g/mol). Both solid and gel-like nano-sized composites containing LiClO4 − salt were obtained from polymer solution in dimethylformamide on glass plates. Kim and White studied composite materials on the same basis as a part of an investigation of a series of nanocomposites with various fluorine-containing polymers. 20,21 They used nonmodified nanoclay (Cloisite Na+) or organically modified MMT and the same PVDF (Solef 1008) to obtain nanocomposites by inclusion from melt. The results obtained from the wide angle x-ray diffraction (XRD) showed that the intercalation with PVDF did not occur when unmodified nanoclay was used. The organically modified nanoclay was exfoliated at a content of 3 wt% filler and intercalated at higher nanoclay content.
Giannelis et al. reported a significant increase in the tensile strength of nanocomposites based on PVDF and organic clay (Cloisite 30B). 22 This was considered because it was induced by the organically modified MMT. Both XRD and infrared Fourier spectroscopic studies showed that MMT particles caused the formation of PVDF β-phase. The SEM micrographs showed spherulite morphology both for the initial polymer and that filled with unmodified and organically modified MMT. The presence of large isotropic shperulites is to be expected in the initial PVDF. The spherulite size in the α-phase (∼3 µm) significantly decreased after adding MMT. In PVDF containing 5 wt% organic clay, only β-phase crystallites sized ∼0.6 μm were formed. This suggests that the organic MMT induced phase transition from arranged α-crystallites to nonarranged fibrous crystallites of β-phase. The formation of β-phase facilitates the improvement in the hardness and strength of the PVDF composites. The fibrous β-phase was considered to decrease the plastic flowing of the materials under applied pressure. This might induce more efficient energy dissipation in nanocomposites, thus retarding the formation of cracks.
The same authors found that the Young’s modulus of the composite materials increased from 1.3 to 1.8 GPa, while the elongation increased from 20 to 140%. The strength of the nanocomposite filled with organic MMT was about 700% higher than that of the initial PVDF. 22 Such simultaneous improvement in the hardness and strength of the partially crystalline PVDF filled with organic clays is very important from technological point of view. It was due to the fact that the addition of organic clay to most of the partially crystalline thermoplasts results only in improved hardness but without deterioration of plasticity or strength.
Using twin-screw extruder, Pramoda et al. 23 obtained different nanocomposites containing 0, 1, 2 and 5 wt% nanoclay (Nanocor Inc. I. 34 TCN) by compounding in PVDF (Solef®) melt. The β-phase content in the nanocomposites increased with the nanoclay content but there was always significant amount in α-phase remaining in the polymer. This contradicts the statement of Priya and Jog 16 –18 as well as Giannelis et al. 22 who showed almost full transformation into β-phase by the addition of nanoclay.
Dillon et al. 24 reported the preparation of PVDF–nanoclay composite materials by two methods: from solution and by coprecipitation. The content of nanoclay (organically modified MMT Cloisite 15A and Cloisite 25A) was 1–6 wt%. The structure and morphology of the composites obtained were studied. Regardless of the content and morphology of the nanoclay used, β-form of PVDF was observed in all the nanocomposites. The temperatures of crystallization and melting of the polymer increased with the increase in nanoclay content, most probably due to the formation of β-from of PVDF. The introduction of 1–6 wt% nanoclay decreased PVDF crystallinity from 38 to 34%. The decrease did not depend on the type of organic modifier, degree of filling, or nanoclay morphology.
In another publication, 25 it was shown that the use of 1–2 wt% nanoclay effectively improved the tribological properties of the initial PVDF. At the same degree of filling, the mechanical properties were also good. The hardness increased and polymer crystallinity decreased with the increase in nanoclay content. The materials containing 5 wt% nanoclay had higher wear resistance than that of the initial PVDF, worse compatibility between nanoclay and PVDF and lower degree of crystallinity.
PVDF nanocomposites containing clay based on MMT were prepared by mixing in melt. 26 The fillers used were nonmodified clay, clay based on ammonium base and two organically modified clays obtained by cation exchange with hexadecylpyridine chloride and octadecyltriphenylphosphonium bromide. The temperatures of melting and crystallization of the initial polymer increased by 10 and 13°C, respectively, with wt% phosphonium-modified clay. The elongation increased ∼200% with the introduction of only 5 wt% ammonium-modified clay. The tensile strength remained the same or slightly increased with the introduction of clay. In most of the cases, the modulus slightly decreased except for the samples containing phosphonium-modified clay or high content of nonmodified clay (5 wt%). The dielectric constant increased with the increased content of filler. Formation of β-form of PVDF was observed in all the nanocomposites with organically modified clays, regardless of their content.
Kelarakis et al. 27 reported for the first time on structure–properties relationships in a series of clay nanocomposites based on PVDF-HFP. The introduction of organically modified nanoclays to PVDF-HFP facilitated the transformation of the polymer crystals from α- to β-form. The degree of transformation depended on the nature of the modifier used on clay surface and the strength of the interaction between the clay and the polymer. The nanocomposite materials based on this copolymer showed increased values of elongation at break compared with the initial copolymer as well as higher dielectric permeability in wide temperature range.
There are few publications in the literature on the preparation and studies of the properties of composite materials based on PVDF-HFP. They are related mostly to their application in membranes, separators for lithium-ion batteries, and so on. 28 –31
A new method of surface modification based on the ‘grafting from’ polymerization process enabled to prepare original silica nanoparticles covered with PDVF chains. 32 This method was developed to radical grafting of tetrafluoroethylene and vinylidene fluoride telomers onto the silica-bearing vinyl groups 33 to synthesize nanohybrids that exhibited high thermostability (>400°C) and good hydrophobility (surface tension from 15 to 44 mN m−1 for silica).
The aim of the present study is to obtain lamellar clay nanocomposites based on PVDF-HFP with hydrophilic or organically modified clay and study their basic properties.
Experimental
Materials
Poly(vinylidene fluoride-co-hexafluoropropylene) referred as PVDF-HFP is a copolymer (15% HFP comonomer) with a melting temperature of 117°C and a melt index of 6.52 g/10 min (220°C, load 98 N) as powder purchased from Solvay, France.
Two commercial clays were used: Nanomer® PGV, which is hydrophilic nanoclay based on bentonite, and Nanomer I.31 PS based on MMT, surface modified with 0.5–5.0 mass% amino-propyltriethoxysilane and 15–35 mass% octadecylamine, denoted as nanoclays 1 and 2, respectively (products of Nanocor® Inc., Arlington Heights, IL, USA).
Sample preparation
The compositions containing 1.5, 3, 6 and 10 mass% nanoclays 1 and 2 were mixed with PVDF-HFP and homogenized as powder by stirring at 50–60°C for 10 min, and then twice in a Brabender at 200°C. They were pressed on a laboratory press PHI (England) between aluminum foils under the following conditions: samples thickness of about 1 mm, temperature of 200°C, melting period at 200°C for 3 min, pressing pressure of 12 MPa and cooling rate of 40°C/min.
Characterization
Melt index
The melt indices of the initial polymer and the nanomaterials based on it were determined by the melt flow index (MFI; grams per 10 minutes) method on an apparatus MFI 3350 Prodemat (France) at a temperature of 200°C and a load of 98 N.
Differential thermal analysis
The analyses were carried out on a derivatograph ‘STA 449F3 Jupiter’ (Netzsch, Germany) in the range of 20–700°C, heating rate of 10°C/min, sample initial mass of 12 mg, in air and corundum crucible.
DSC measurements
The behavior of the samples with a mass of about 4 mg under melting and crystallization in nitrogen atmosphere was analyzed using simultaneous thermal analyzer STA 449F3 Jupiter under the following conditions: first heating from 20 to 240°C at a rate of 10°C/min, isothermal period of 1 min at 240°C (to remove any traces of crystalline structure) followed by cooling to 20°C and second heating to 240°C at the same rate. The degree of crystallinity of the samples was calculated at ΔH 100% = 104.7 J/g for 100% crystalline PVDF-HFP. 34
X-Ray structure analysis
The XRD patterns were taken by x-ray diffractometer with generator Iris-M (Russia) and goniometer URD-6 (2θ; Germany) at an atmospheric pressure, room temperature and Ni-filtered Cu target Kα radiation in the interval 2θ = 4°–50°.
Light microscopy
The light microscope studies were performed on an apparatus AmplivalPol (Germany) at a magnification of ×400.
Tensile properties
The tensile strength (σ), elongation (ε) and the other characteristics of the initial PVDF-HFP and the nanocomposite materials based on it were measured on a dynamometer INSTRON 4203 (England) at a speed of 100 mm/min and at room temperature.
Heat resistance by Vicat
The Vicat softening points were determined under the following conditions: thickness of the pressed samples of 2–4 mm, cross-section of the finished rod of 1 mm2, load of 9.81 N and rate of increase in temperature of 50°C/h (Fritz Heckert, Germany).
Results and discussion
Figure 1 shows micrographs of the nanocomposite materials prepared from PVDF-HFP and the nanoclays used. The filler dispersal in the matrix was observed to be fairly good at 10 mass% content of nanoclay 1 or 2. This was more pronounced for the materials based on nanoclay 2 due to the organophilic character of nanoclay 2 compared with nanoclay 1.

Microphotographs of nanocomposite materials based on PVDF-HFP with 10 mass% nanoclay 1 (a) and nanoclay 2 (b). PVDF-HFP: poly(vinylidene fluoride-co-hexafluoropropylene).
Figures 2 and 3 present the XRD patterns of the initial PVDF-HFP, nanoclays 1 and 2 and the nanocomposite materials based on them. It can be seen that the filler nature has certain effect on the diffraction patterns of the composite materials studied. The materials containing 10 mass% organically modified clay showed higher deviation in the main diffraction reflections at 2θ = 18.6° and 20.1° (Figure 3). In the same figure, notable changes in peak locations and intensities were observed, indicating for changes in the quantity and perfectness of the crystalline phase in the compositions. Taking into account the deviations of the main reflections to smaller angles, it can be suggested that the packing factor of these nanocomposites decreased because of the more stressed state of the segments crystallizing in the interphase regions. This was explained with the better interaction between nanoclay 2 and PVDF-HFP matrices. Nanoclay 2 induced significant increase in the content of copolymer β-form in the nanocomposites obtained, as can be deduced from the XRD patterns of the compositions containing 10 mass% nanoclay 2.
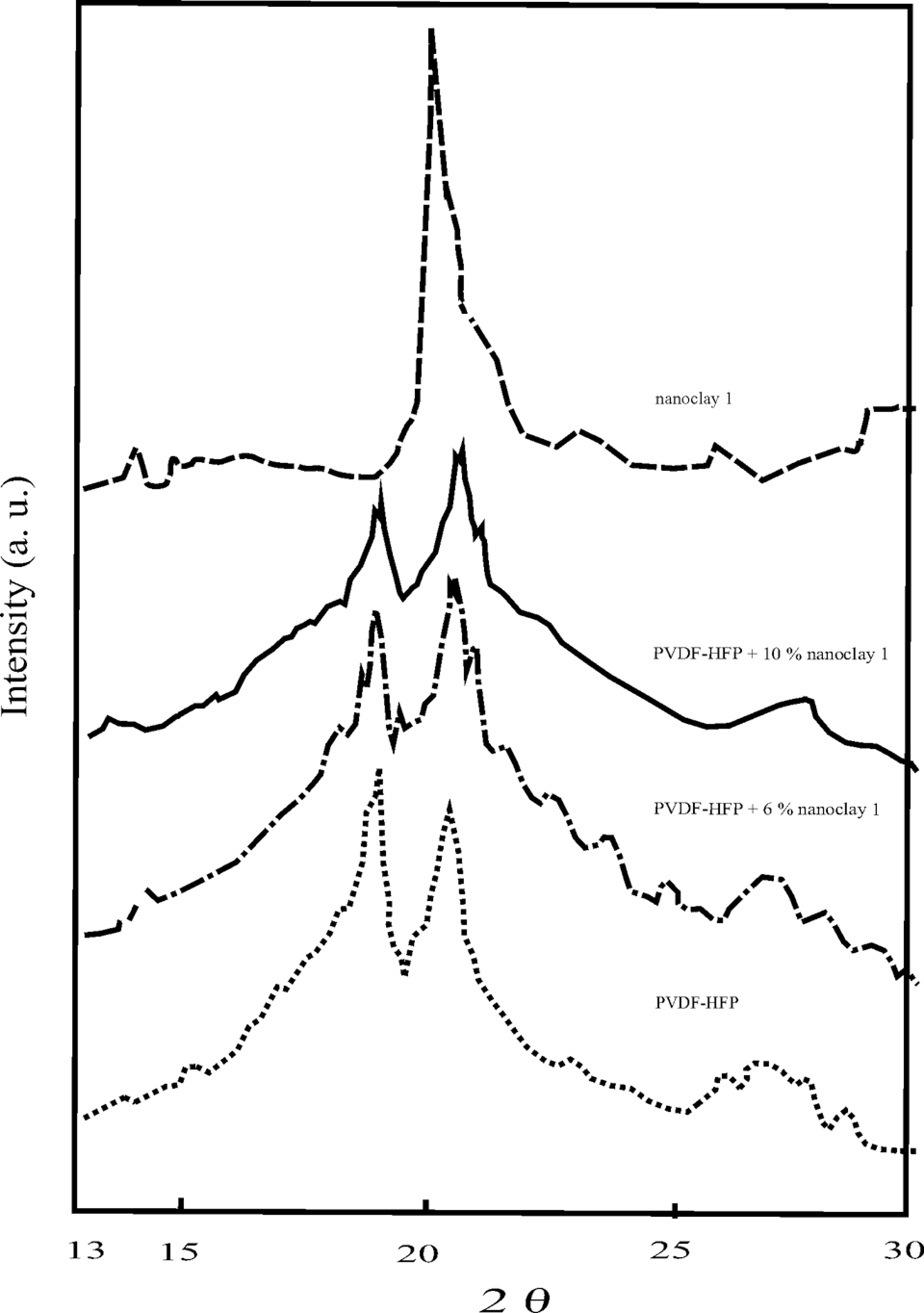
X-Ray diffraction patterns of the initial PVDF-HFP, nanoclay 1 and the nanocomposite materials. PVDF-HFP: poly(vinylidene fluoride-co-hexafluoropropylene).
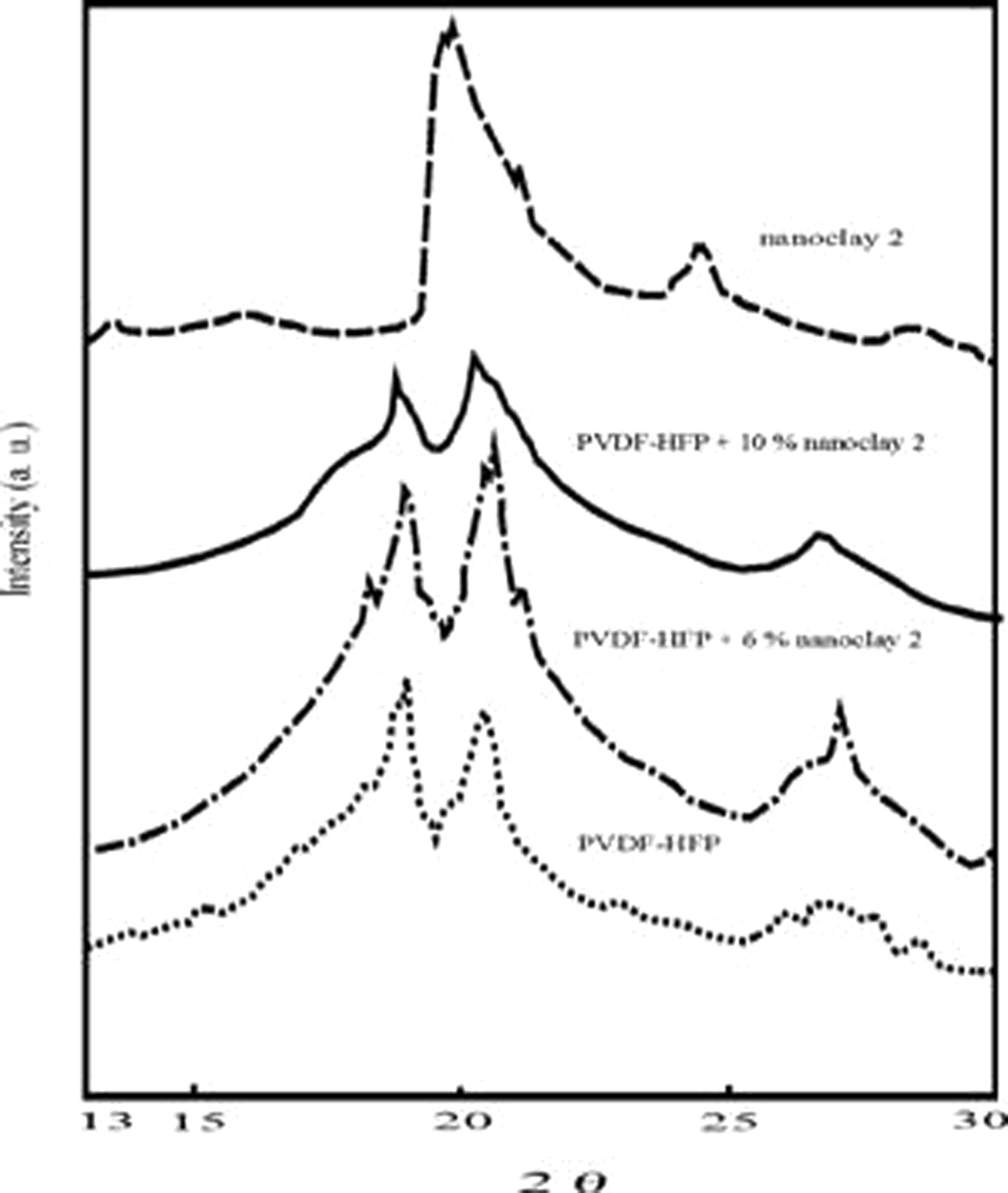
X-Ray diffraction patterns of the initial PVDF-HFP, nanoclay 2 and the nanocomposite materials. PVDF-HFP: poly(vinylidene fluoride-co-hexafluoropropylene).
In contrast to the materials with modified clay, those with nonmodified ones (Figure 2) showed deviations in the opposite direction (2θ = 18.6° and 20.1°) at the same filler concentration and 10 mass%. This was probably due to the densification of the segments in the interphase regions.
It can be seen from the differential thermal analyses of the samples containing 3 and 10 mass% filler that these samples containing nanoclay 1 had higher thermal stability. Their initial rate of destruction and 10% weight loss were 344 and 323°C and 410 and 380°C for the samples with 3 mass% nanoclays 1 and 2, respectively (Table 1). These values for the nanocomposite materials with 10 mass% nanoclays 1 and 2 were 360 and 340°C and 410 and 370°C, respectively. The highest destruction rate of the composites based on PVDF-HFP and nanoclay 1 was also higher than those based on nanoclay 2.
Thermal properties of nanocomposite materials based on PVDF-HFP with nanoclays 1 and 2.
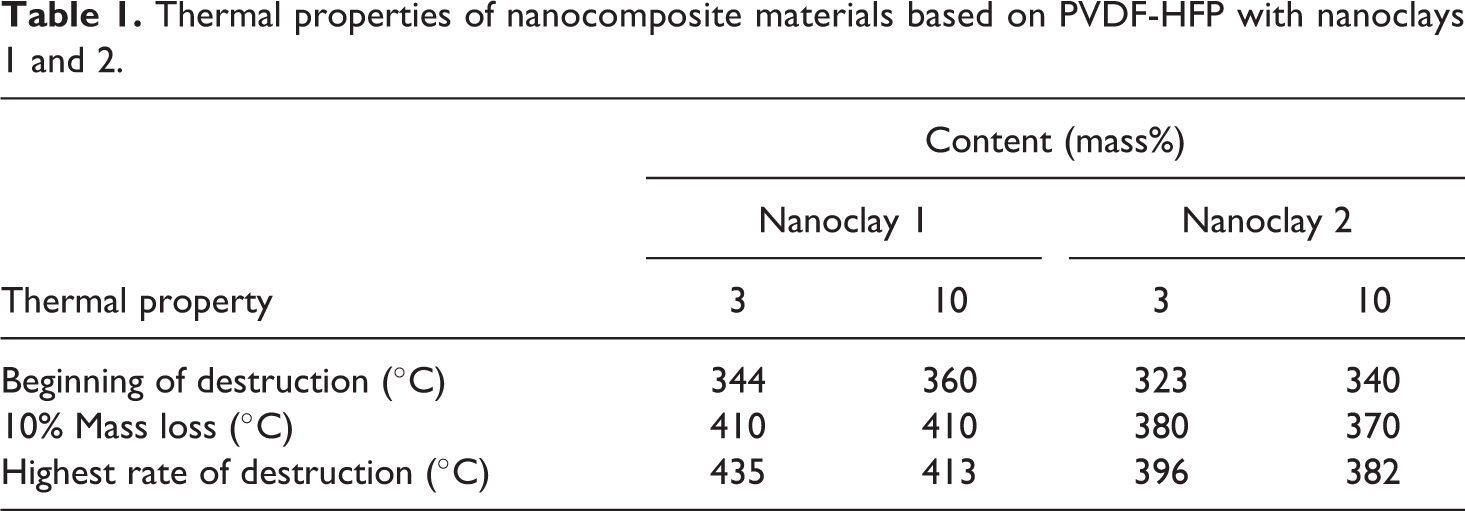
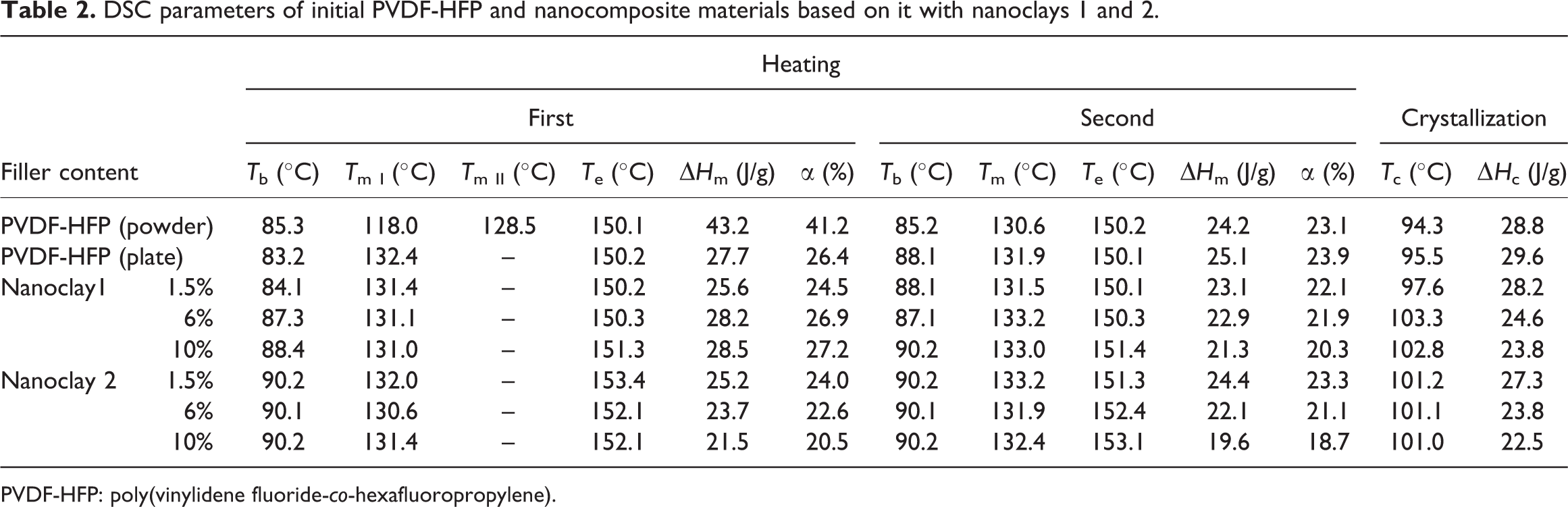
Table 2 presents the differential scanning calorimeter (DSC) parameters for the initial PVDF-HFP and the nanocomposites based on it with the two types of nanoclays used. The results obtained show that the introduction of nanoclay 1 or 2 into PVDF-HFP did not change the temperatures of beginning (T b) and end (T e) of melting for both the heating processes and both types of samples; these temperatures are about 90 and 151–153°C, respectively. It can be seen from the same table for the different series of samples that the melting temperature (T m) during the two heating processes remained the same (131–133°C).
DSC parameters of initial PVDF-HFP and nanocomposite materials based on it with nanoclays 1 and 2.
PVDF-HFP: poly(vinylidene fluoride-co-hexafluoropropylene).
All the DSC thermograms of pure pressed PVDF-HFP and the materials containing nanoclay 1 or 2 showed one peak for their melting. The only exception is the initial powdery copolymer by the first heating, where two peaks were observed at 118.0 and 128.5°C (Figure 4). The double melting peak was presumably due to the melt recrystallization and imperfect crystallites present in pure PVDF-HFP. 35 It is interesting to note that the commercial homopolymer Kynar® only shows one peak for melting, which suggests that the double peaks might result from different ‘head–head’ or ‘tail–tail’ bonding in the different PVDF samples. These bondings stipulate different configuration micro-inhomogeneities of the chains, which affect their packing into crystalline phase and their thermodynamic behavior. 24 The DSC thermograms of clay nanocomposites based on PVDF-HFP had also one melting peak. Most probably, it was due to the nanoclays playing the role of crystallization centers for the polymer to promote smaller and more homogeneous lamellae. They all melt simultaneously during the heating.
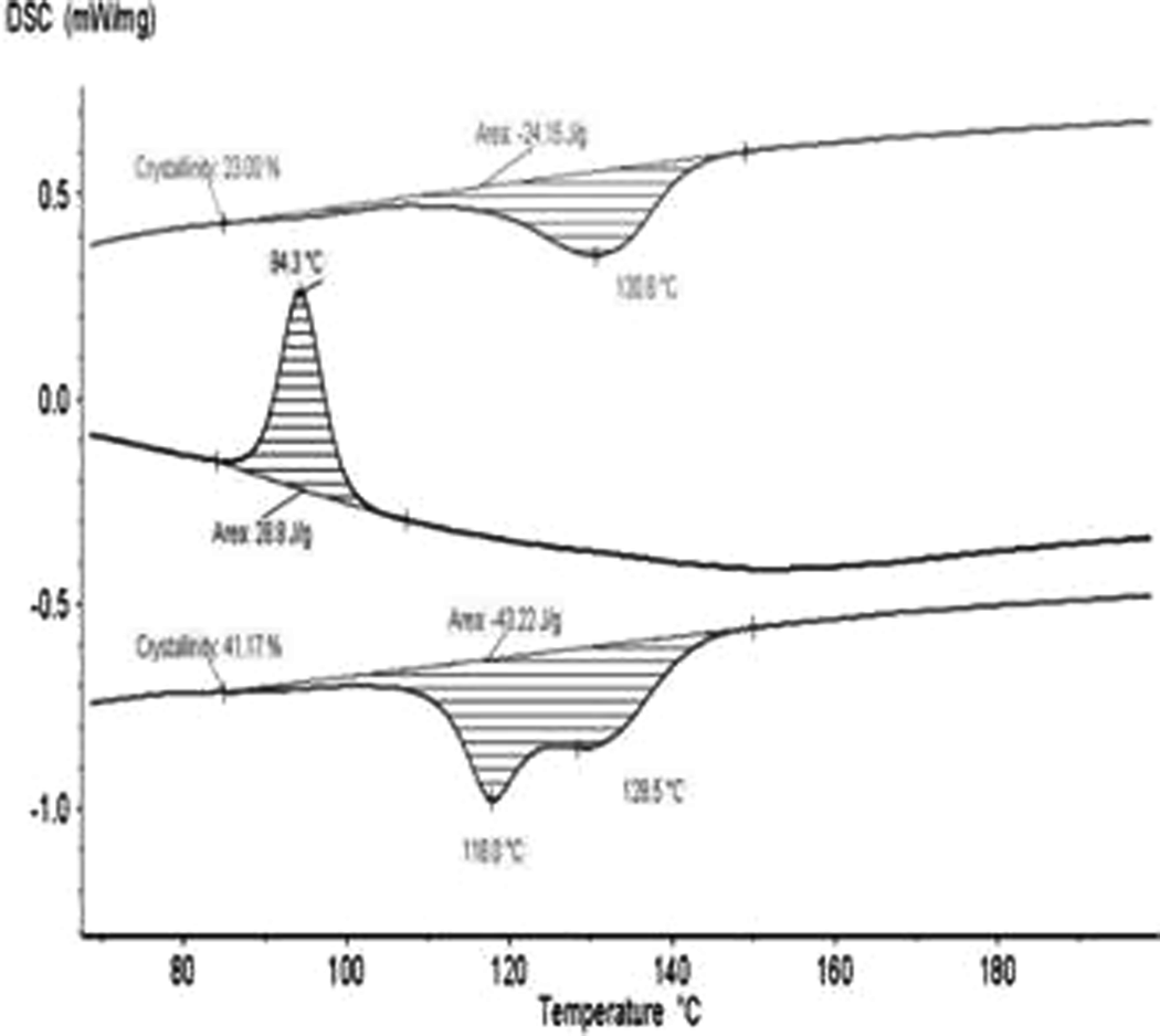
DSC thermogram of the initial powdery PVDF-HFP under heating and crystallization. DSC: differential scanning calorimeter; PVDF-HFP: poly(vinylidene fluoride-co-hexafluoropropylene).
The temperature of crystallization (T c) of the initial pressed copolymer slightly increased from 95.5 to 102.8 and 101.0°C, for the materials containing 10 mass% nanoclay 1 or 2, respectively (Table 2). Besides, no notable differences were observed for the T c and T m of both the types of nanocomposite materials. These temperatures remained unchanged with the increase in the content of nanoclay 1 or 2 within the interval studied (1.5–10 mass%). All this is consistent with the XRD data, which indicates that the addition of nanoclay promoted crystallization in β-polymorphic modification, which has higher T c and T m.
Similarly, Table 2 shows in detail the degree of crystallinity (α) of the initial PVDF-HFP and the series of samples based on it with the two types of nanoclays. For the pressed copolymer, it was found to be ∼26%; while for the composite materials with nanoclay 1, α was in the intervals from 26.4 to 27.2% and from 23.9 to 20.3% for the first and second heating, respectively. The degree of crystallinity of the initial powdery copolymer was considerably higher, that is, 41.2%. The values of the degree of crystallinity were smaller than those of nonmodified PVDF. 24 This is due to the content of HFP in the copolymer and the bonding head–head and tail–tail in to copolymer used for the present study.
It can be seen from the data presented in the table that the introduction of the two types of clays into PVDF-HFP resulted in decreased values in the enthalpies of crystallization and melting (ΔH c and ΔH m).
The MFI of the nanocomposite materials obtained (Figure 5) was found to decrease with the increase in the filler content (C, mass%) from 6.52 g/10 min for the pure copolymer to 3.13 and 4.83 g/10 min for the composites with nanoclays 1 and 2, respectively.
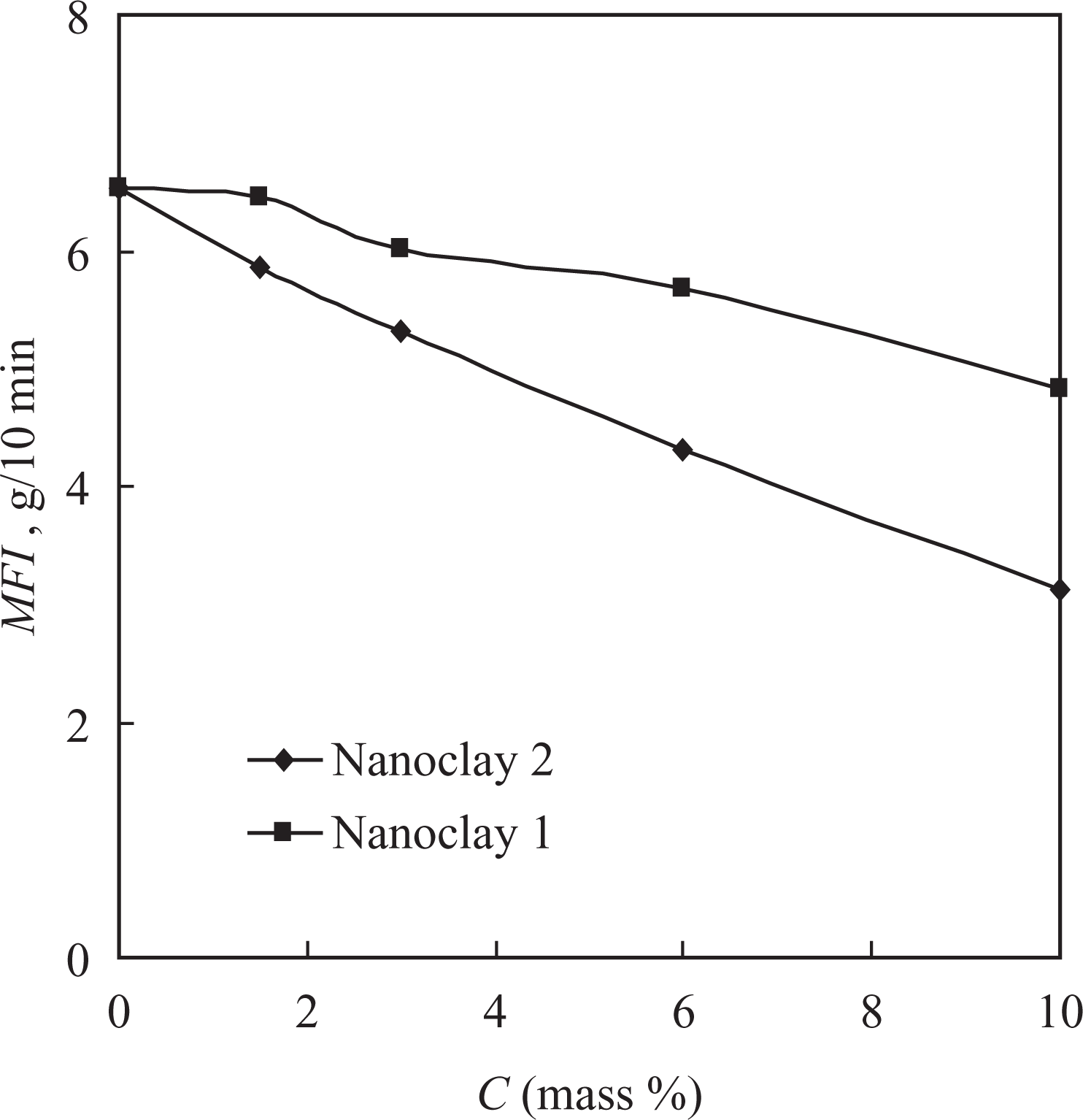
Dependence of the MFI of the nanocomposite materials based on PVDF-HFP on the filler content. MFI: melt flow index; PVDF-HFP: poly(vinylidene fluoride-co-hexafluoropropylene).
The tensile properties of the nanocomposites obtained were also studied. The tensile strength (σ) for the materials based on PVDF-HFP with nanoclay 2 increased from 29.8 to 31.1 MPa at 1.5 mass% filler content and then decreased to 13 MPa at 10 mass% (Figure 6). For the materials with nanoclay 1, the tensile strength decreased at 3 mass% to 20.6 MPa and remained almost the same (about 20 MPa) with the increase in the filler content to 10 mass%.
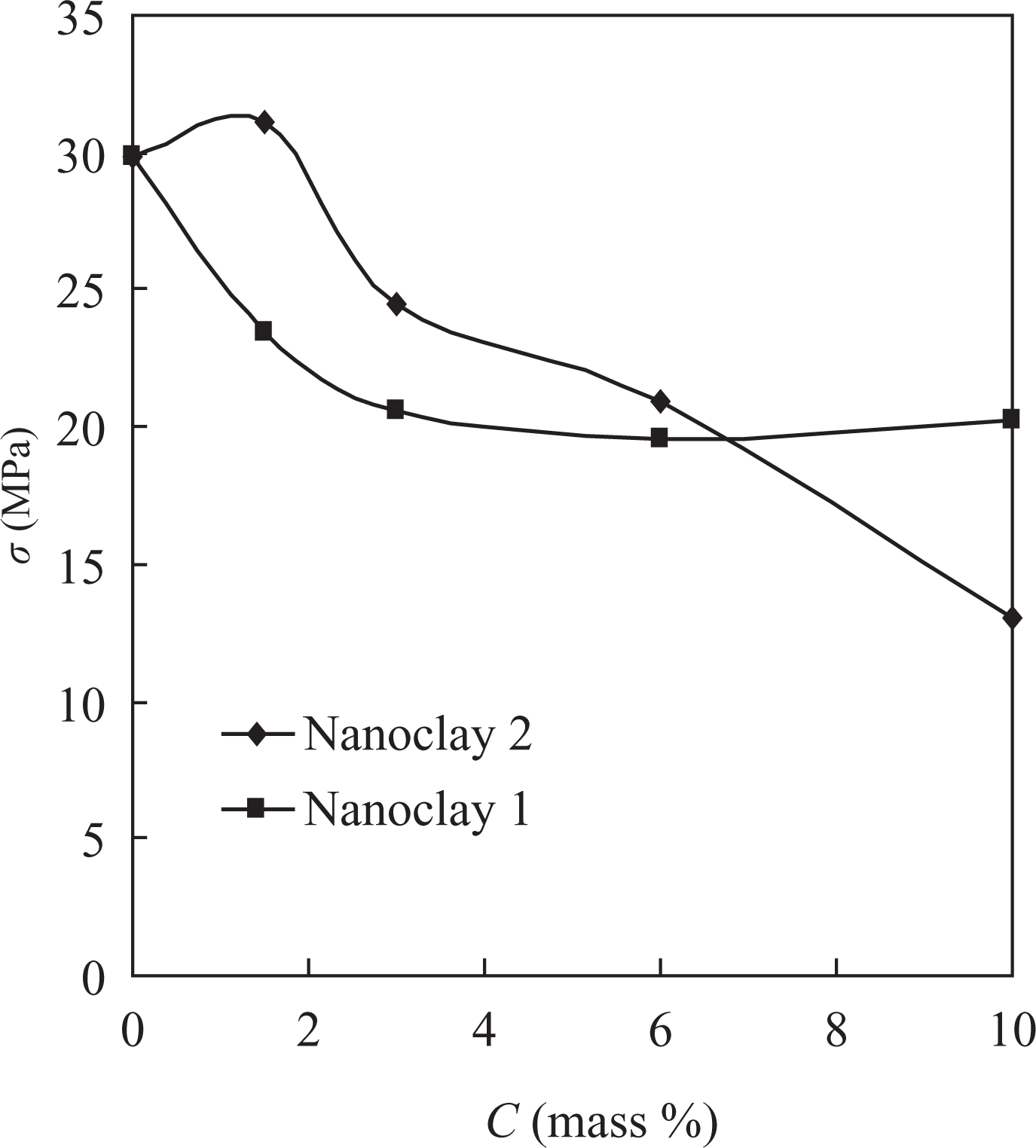
Dependence of the tensile strength (σ) of the nanocomposite materials based on PVDF-HFP on the filler content. PVDF-HFP: poly(vinylidene fluoride-co-hexafluoropropylene).
The dependence of the elongation (ε) of the nanocomposites studied was similar to that observed for the strength (Figure 7). With nanoclay 2, the elongation first increased, and at filler concentrations higher than 1.5 mass%, it gradually decreased; whereas for the materials with nanoclay 1, it slightly decreased and remained almost the same at the concentrations higher than 3 mass%.
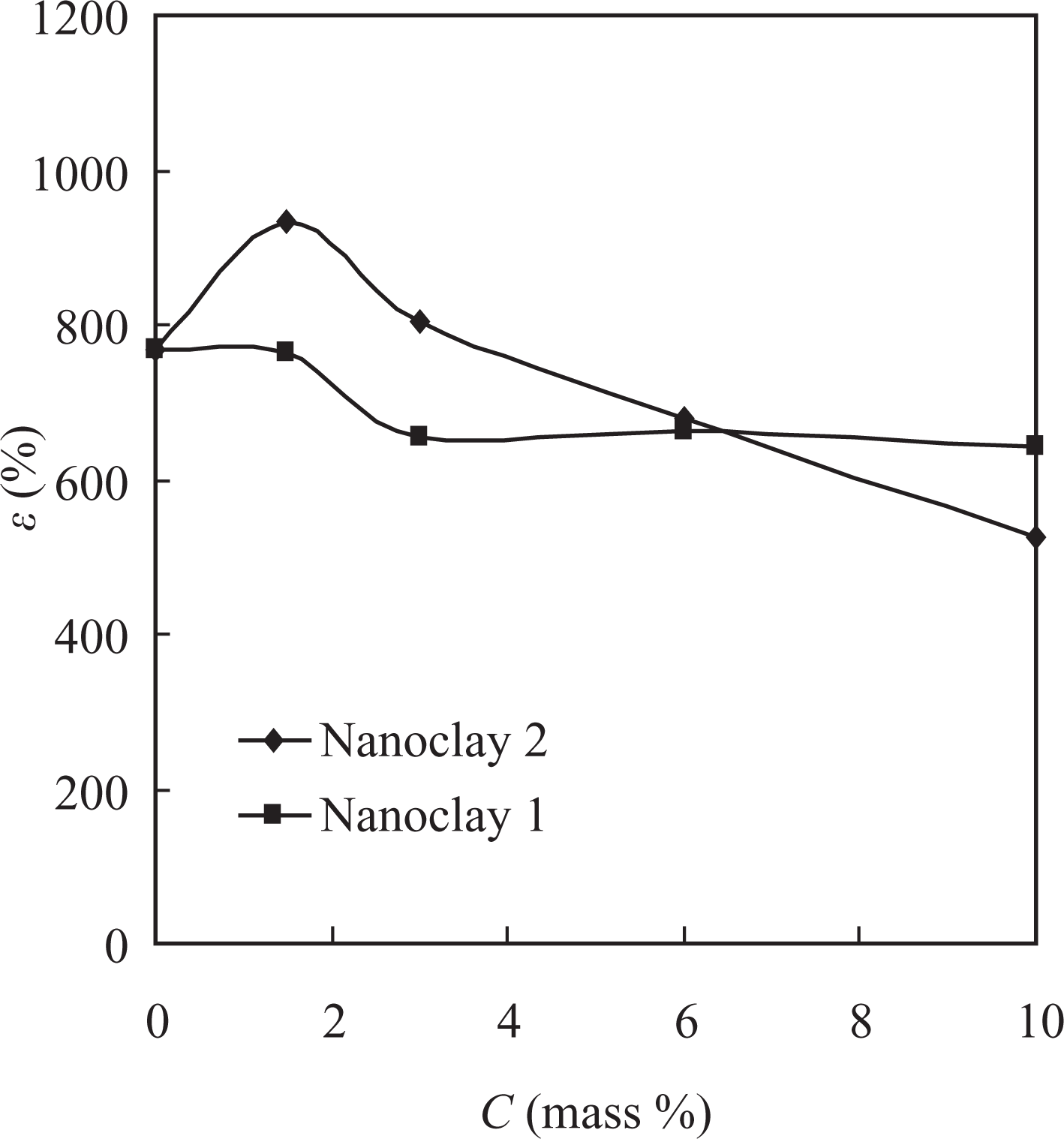
Dependence of the elongation (ε) of the nanocomposite materials based on PVDF-HFP on the filler content. PVDF-HFP: poly(vinylidene fluoride-co-hexafluoropropylene).
The Young’s modulus (Y) and the energy at break (E b) of the nanocomposites obtained were studied (Figures 8 and 9). Obviously, the modulus of the composite materials was almost independent of the content of nanoclays 1 and 2 to remain within the interval 215–265 MPa, while the energy at break decreased to reach 7.5 and 6.0 J for the compositions with 10 mass% contents of nanoclays 1 and 2, respectively.
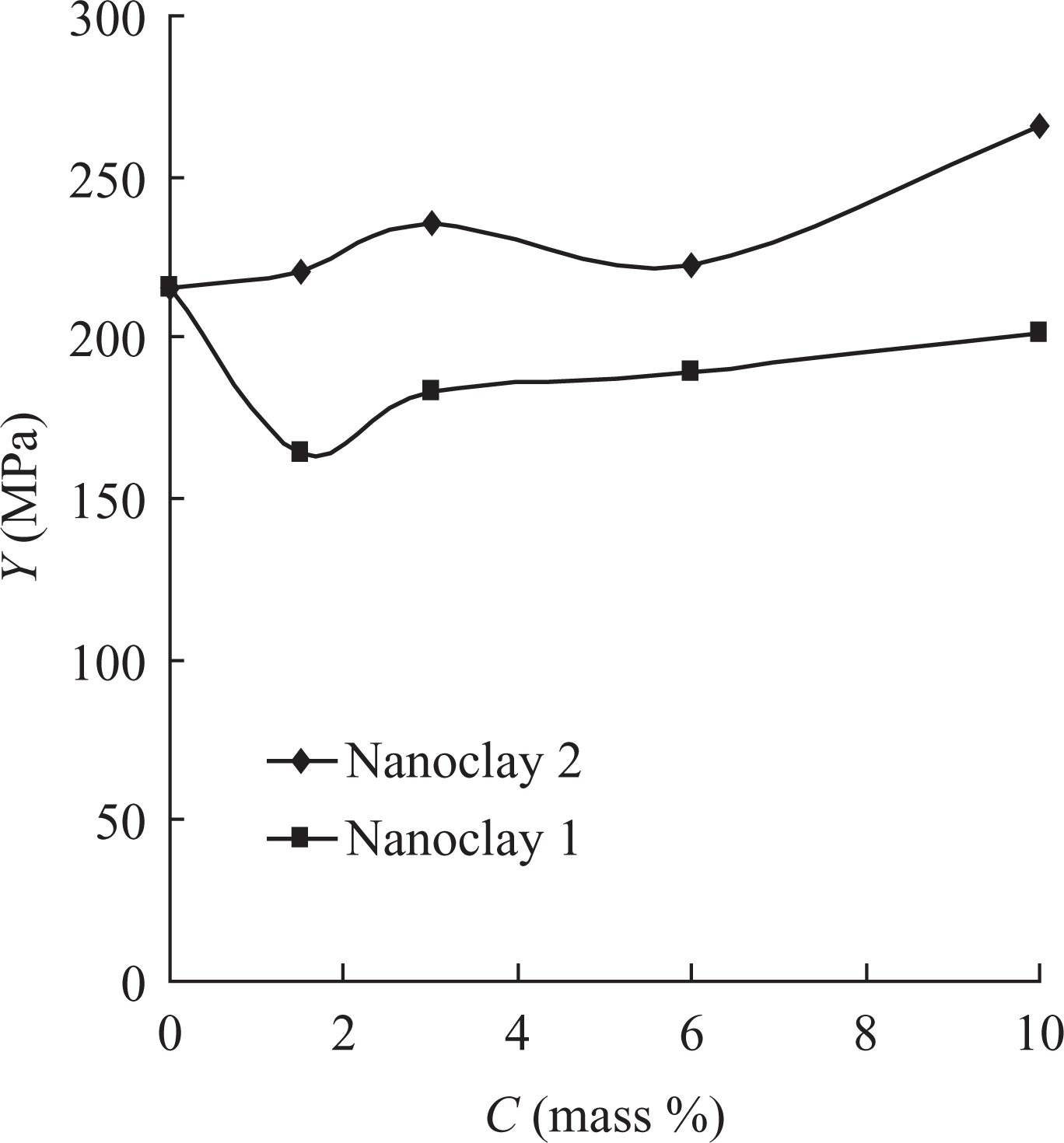
Dependence of Young’s modulus (Y) of nanocomposite materials based on PVDF-HFP on the filler content. PVDF-HFP: poly(vinylidene fluoride-co-hexafluoropropylene).
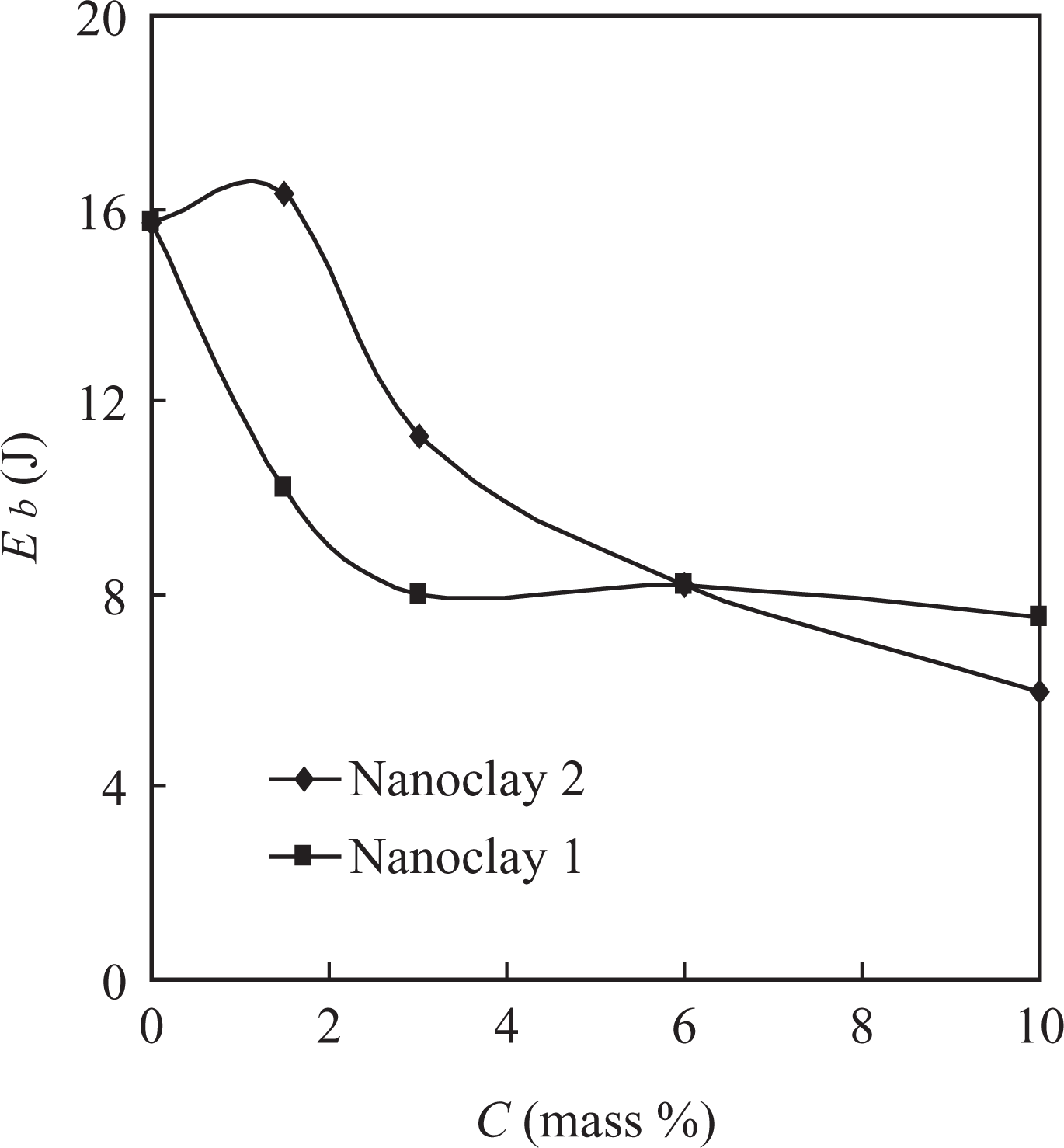
Dependence of the energy at break (E b) of the nanocomposite materials based on PVDF-HFP on the filler content. PVDF-HFP: poly(vinylidene fluoride-co-hexafluoropropylene).
For both types of nanocomposite materials with nanoclays 1 and 2, the heat resistance by Vicat increased by 7°C compared with the initial copolymer, and it was measured to be 129°C at 10 mass% contents of these fillers.
The analysis of the tensile properties of the materials based on PVDF-HFP with nanoclays showed that the results obtained with nanoclay 2 were better. This is due to the higher organophilicity of this filler (surface modified with 0.5–5.0 mass% aminopropyltriethoxysilane and 15–35 mass% octadecylamine) compared with that of nanoclay 1.
Conclusions
PVDF-HFP nanocomposites with contents from 1.5 to 10 mass% nanoclay 1 (unmodified clay based bentonite) and nanoclay 2 (organically modified clay based on MMT) were prepared. The filler dispersion within the matrix was found to be good for both the types of nanocomposite materials. The materials containing 10 mass% organically modified clay showed significant increase in the β-phase and higher deviation of the main diffraction reflections, 2θ = 18.6° and 20.1°. The degree of crystallinity of the materials containing nanoclay 2 decreased from 26.4 to 20.5% and from 23.9 to 18.7% for the first and second heating, respectively. The tensile strength of the materials based on PVDF-HFP with nanoclay 1 increased from 29.8 to 31.1 MPa at 1.5 mass% filler content; while for the materials with nanoclay 1, the tensile strength was observed to decrease to 20.6 MPa at 3 mass% and then remained almost constant. The Young’s modulus was almost unrelated to the content of nanoclay 1 or 2 with the values in the range of 215–265 MPa. For both the types of nanocomposite materials with nanoclays 1 and 2, the heat resistance by Vicat increased by 7°C compared with that of the initial copolymer.
The tensile characteristics of the materials based on PVDF-HFP with nanoclays showed that better results were obtained with nanoclay 2.
Footnotes
Funding
The work was supported by the Bulgarian Ministry of Education and Sciences (National Science Fund).
