Abstract
The melt flow behaviours of low-density polyethylene (LDPE)/palm kernel shell (PKS) composites were studied. Acrylic acid (AA) was used as a chemical modifier for PKS. The effect of filler loading and the presence of AA in melt flow behaviour of composites were determined. The melt flow index of the composites decreased with the increase in the filler loading. The apparent viscosity of the composites was found to exhibit linear relationship with reciprocal of the temperature. The study on the thermal properties showed that higher filler loading tend to reduce the onset temperature as the PKS possessed lower degradation temperature compared to the LDPE. The presence of the filler in LDPE polymeric matrix improved the thermal stability of the composites. The addition of AA provided better interfacial bonding between the LDPE matrix and the PKS filler, where higher onset temperature and lower weight loss were observed for LDPE/PKS composites with AA. The activation energy of the LDPE/PKS composites was increased with increasing filler loading. At similar filler loading, the addition of AA increased the activation energy of the LDPE/PKS composites.
Introduction
Nowadays, natural filler–reinforced polymers draw more attention of the researchers compared to the synthetic filler–reinforced polymers. The natural filler–reinforced polymers are of light weight, nontoxic, of low cost and able to decrease environmental pollution. There are many research done based on the natural fillers in polymeric composites. The commonly used natural fillers are wood fibres and flour, kenaf fibres, henequen fibres, pineapple-leaf fibres and corn starch. 1 –6 Malaysia, as the main oil palm production country, produces large quantity of palm kernel shell (PKS). PKS is hard stony endocarp that surround the kernel. It is produced annually in large quantity and usually burnt as waste material. Thus, it has high potential to be used as natural filler in polymeric composites due to its high availability and low cost.
The properties of the polymer such as low-density polyethylene (LDPE) will be altered as the filler is incorporated into the system. The polymers possess hydrophobic nature which is incompatible with the hydrophilic nature of the natural fillers. The natural fillers contain hydroxyl groups from lignin and cellulose. These hydroxyl groups can be modified in order to improve the interfacial bonding between the filler and the matrix. 7 Several investigations on acrylic acid (AA) as the coupling agent have been reported by several workers. 8 –11
The studies on the melt flow properties of the filled polymer system are important in order to establish better processing conditions and maximize the product performance. Rheological measurements conducted in various steady-state and dynamic environments are used widely for determining the sensitivity of a material during processing. 12 Generally, the presence of filler in thermoplastic and elastomers increases the melt viscosity and may result in unusual rheological effects. The rheological studies of polymeric composites have been carried out extensively by these workers. 12 –21
In this study, the effect of temperature and filler loading on the melt flow behaviour of untreated and treated LDPE/PKS composites with AA was investigated. The thermal properties of the LDPE, PKS, untreated and treated LDPE/PKS composites with AA at different filler loadings were also determined using thermogravimetric analysis (TGA).
Experimental
Materials
The LDPE was supplied by Titan Chemical, Johor, Malaysia, with the density of 0.922 g/cm3 and melt flow index (MFI) of 0.33 g/10 min. The PKS used as fillers was obtained from Malpom Oil Palm Processing, Nibong Tebal, Malaysia. The PKS was dried at 80°C for 24 h to evaporate the moisture content. Then the cleaned PKS was crushed into powder form. The average particle size of 75 μm was measured by Malvern particle size analyzer. The AA with a molecular weight (Mw = 72.06) and density of 1.051 g/ml was supplied by Fluka (Penang, Malaysia). Ethanol was used as a solvent to dissolve AA in filler treatment. Ethanol (96%) was supplied by Fisher Scientific (M) Sdn. Bhd., Shah Alam.
Filler treatment
The AA is delivered in liquid form. PKS was mixed with 3% (v/v) of AA in ethanol. Then the solution was added into the flask by constant stirring for 1 h. The treated filler was dried at 80°C for 24 h for complete evaporation of ethanol.
Composites preparation
Composites were prepared using a Z-Blade mixer at a temperature of 180°C and rotor speed of 50 r/min. The LDPE were loaded into the mixing chamber for 5 min. After 5 min, the filler was added and mixed continuously for 10 min. The total mixing time of composites was 15 min. Finally, the composites were removed from the mixing chamber and pressed into thin sheets by compression moulding. Compression moulding involved preheating at 180°C for 9 min followed by compressing for 6 min and subsequent cooling under pressure for 4 min. The formulation of the treated and untreated LDPE/PKS composites with AA is shown in Table 1.
Formulation of untreated and treated LDPE/PKS composites with AA.

AA: acrylic acid; LDPE: low-density polyethylene; PKS: palm kernel shell.
Melt flow index test
MFI test was carried out according to American Society for Testing and Materials (ASTM) 1238. The die length and diameter are 8.000 mm and 2.095 mm, respectively. The entry angle was 180°. After a residence time of 6 min, the melt was extruded through the die. The testing temperature used were 180°C, 190°C, 200°C and 210°C with the load of 5 kg.
A relationship between shear stress (τ
w
) and apparent shear rate (γ
a
) at the wall can be written as follows:




The Arrhenius equation is shown as below:

Thermogravimetric analysis
TGA was carried out using a Perkin Elmer Pyris Diamond TG-DTA. The samples, weighting about 15–25 mg, were subjected to heating rate of 20°C/min at temperature range of 30–650°C under nitrogen flow of 50 ml/min.
Results and discussion
Melt flow behaviour
Figure 1 shows the effect of temperature on the MFI of the LDPE and the LDPE/PKS composites of different filler loadings. The MFI of the LDPE and LDPE/PKS composites exhibited linear relationship with temperature, which increased with temperature that is from 180°C to 210°C. The increase in MFI values at higher temperature is due to the accelerated molecular motion, availability of greater free volume and also decreasing entanglement density and weaker intermolecular interaction. At the same temperature, the MFI of the LDPE composites is higher than the LDPE/PKS composites, which indicates its lower viscosity compared to the composites. The incorporation of the filler into the polymeric system decreases the MFI of the composites. The presence of filler will disturb the normal flow of the composites melt and hinder the mobility of chain segment of the flow. At higher filler loading, the flow of the composites was disturbed and resisted. As a result, the composites with high filler loading possess lower MFI compared to that at lower filler loading. The treatment of PKS with AA had changed the flowability of the composites. Figure 2 shows the MFI of the composites which was influenced by the presence of AA. At a similar filler loading, the treated LDPE/PKS composites exhibited higher MFI compared to the untreated composites. It is indicated that the presence of AA decreased the viscosity of the composites and enhanced their flowability. The AA improves the compatibility between hydrophilic filler and hydrophobic matrix. The interfacial bonding between the matrix and filler was enhanced and showed better flowability.
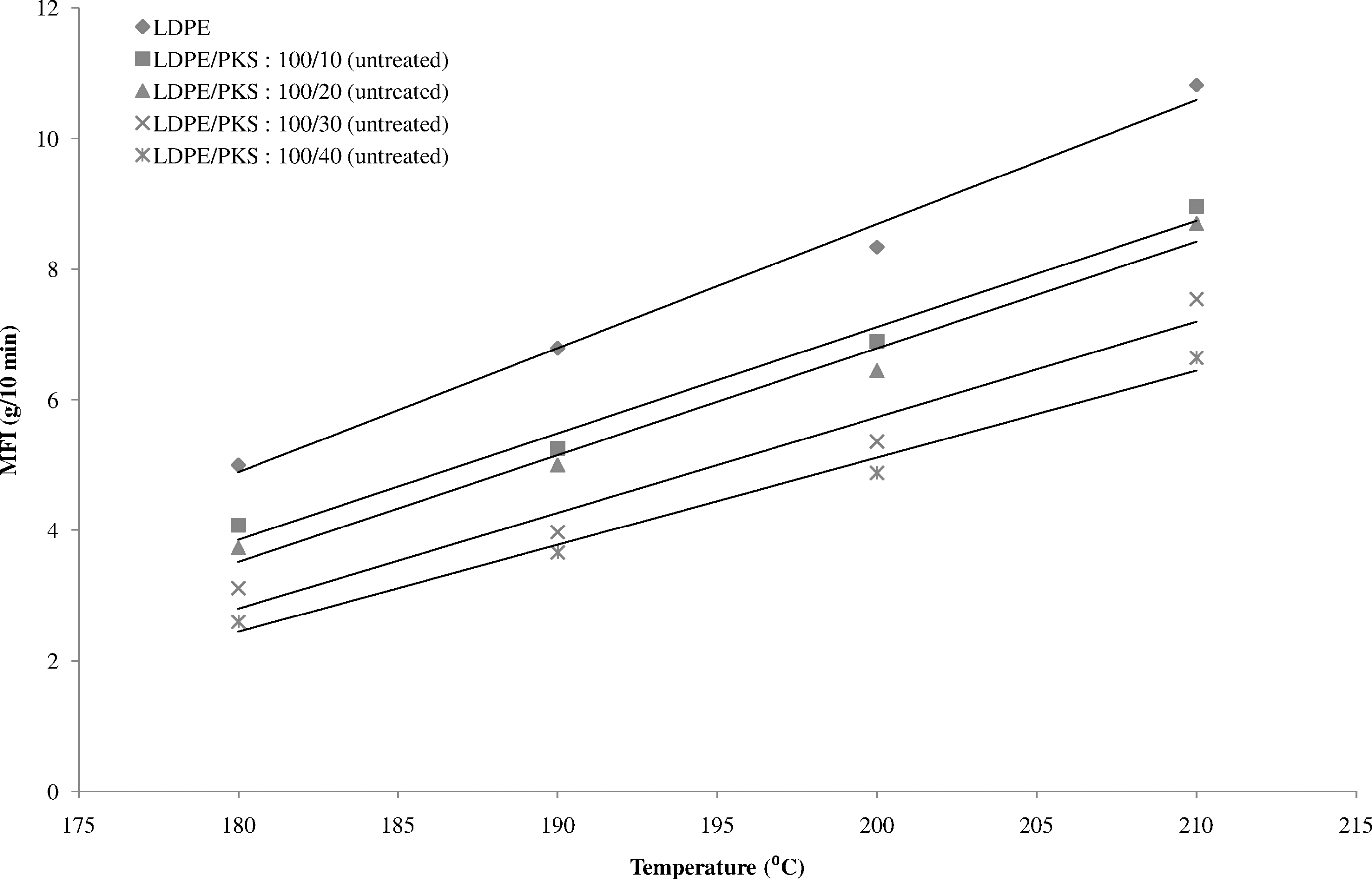
The melt flow index (MFI) of untreated LDPE/PKS composites at different temperatures. LDPE: low-density polyethylene; PKS: palm kernel shell.
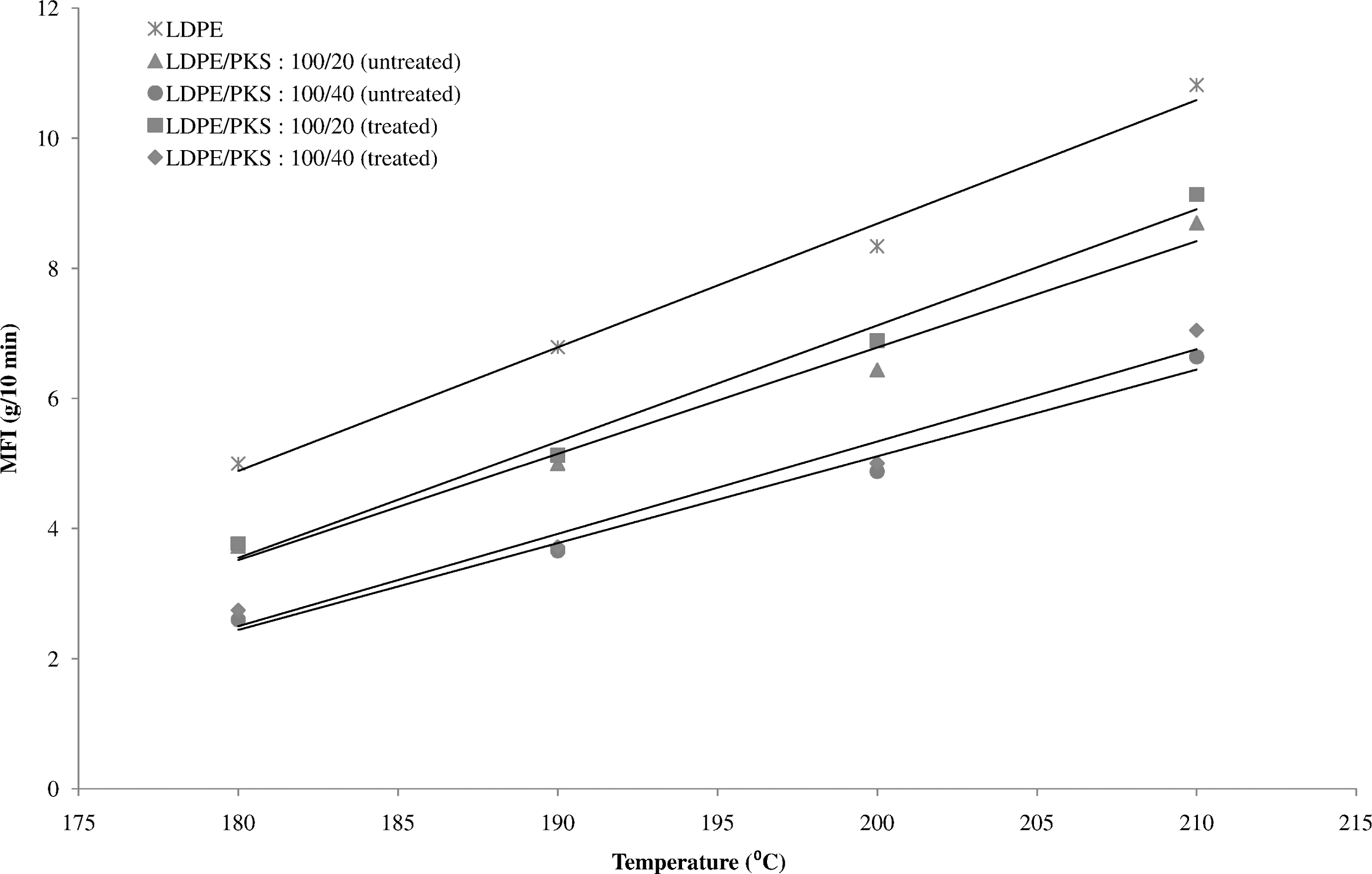
The melt flow index (MFI) of untreated and treated LDPE/PKS composites with AA. AA: acrylic acid; LDPE: low-density polyethylene; PKS: palm kernel shell.
Figure 3 shows the effect of temperature on the apparent viscosity of untreated LDPE/PKS composites at different loadings of PKS. It can be seen that the apparent viscosity had increased with increasing 1/T and filler loading. The addition of filler into polymeric matrix decreased the flow properties of composites to some extent because the friction between the matrix melt and the filler was increased. As a result the transition or mobility of the molecules was hindered leading to an increase in the melt viscosity or decrease in MFI. Figure 4 shows the effect of AA on the viscosity of LDPE/PKS composites at different temperatures. The linear relationship was obtained between ln apparent viscosity of the composites and reciprocal of absolute temperature (1/T). This indicates that the dependence of the shear viscosity of LDPE/PKS composites on temperature obeys the Arrhenius equations as shown in equation (5). At similar filler loading, the LDPE/PKS composites with AA showed lower apparent viscosity compared to the composites without AA. The presence of AA enhanced the filler dispersion and interfacial bonding between the polymer matrix and filler. The filler can be bonded better with the matrix and flowed easily. As a result, the viscosity of the composites reduced.
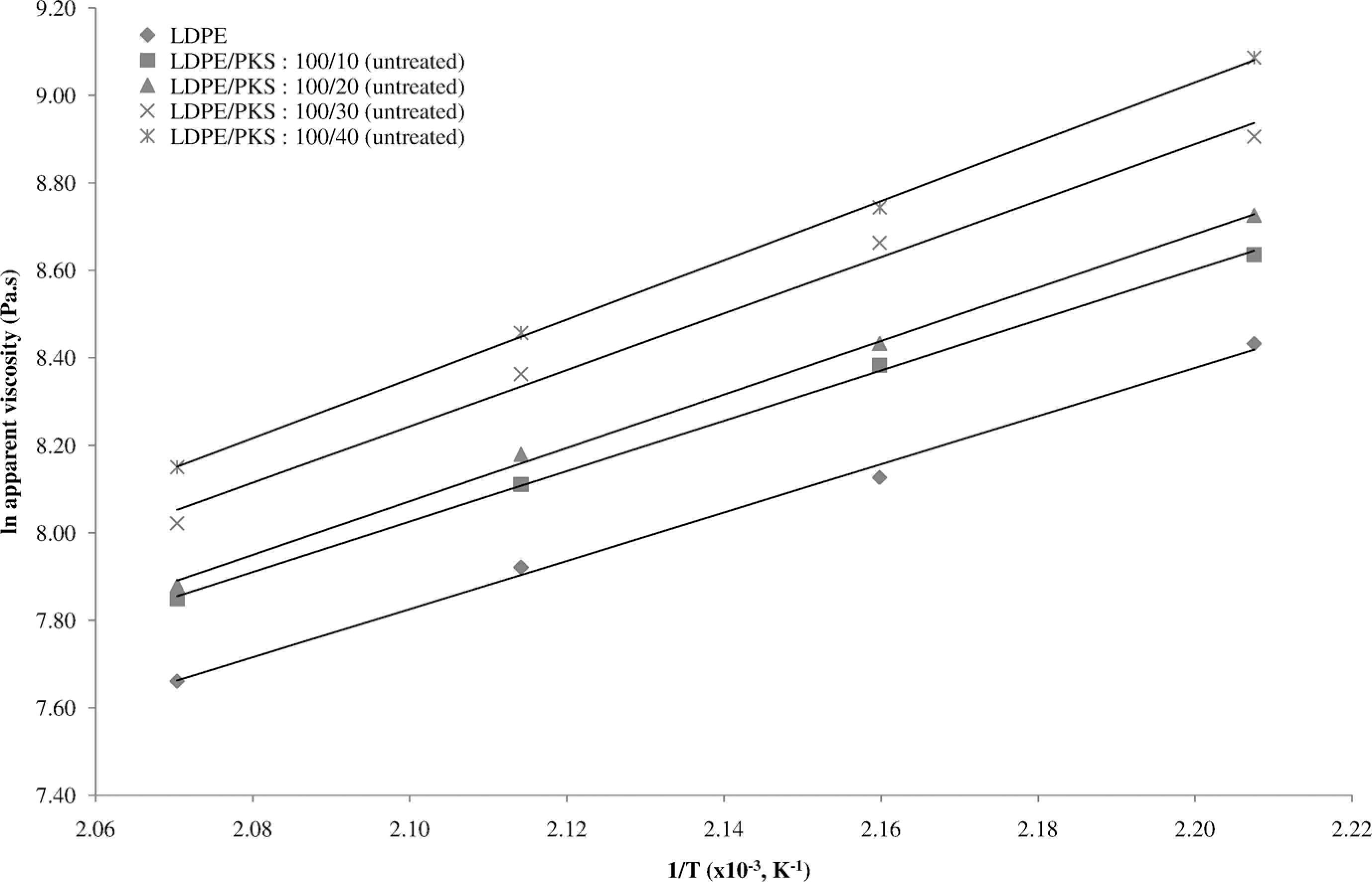
The apparent viscosity versus temperature of untreated LDPE/PKS composites at different temperatures. LDPE: low-density polyethylene; PKS: palm kernel shell.
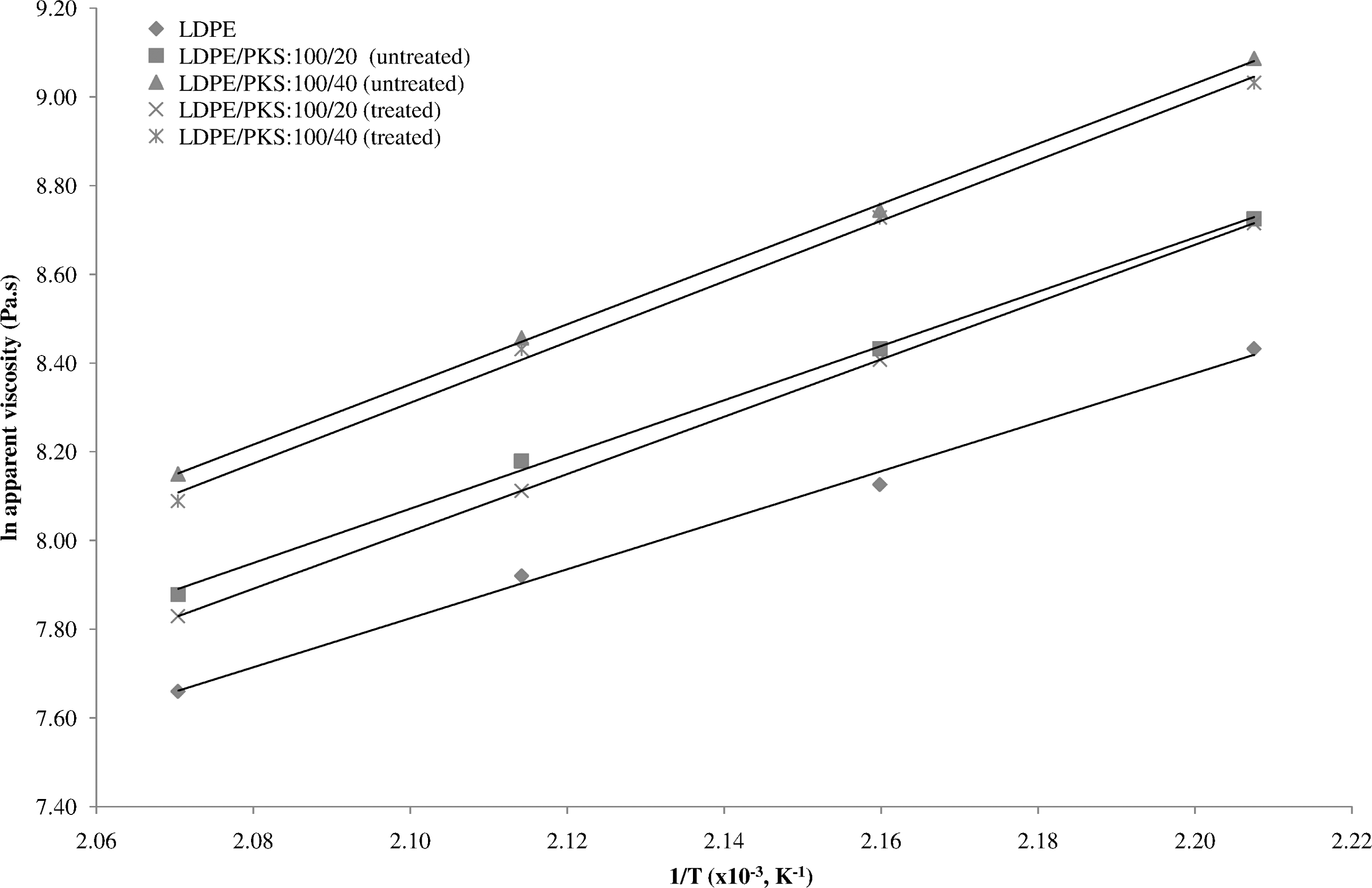
The apparent viscosity versus temperature of untreated and treated LDPE/PKS with acrylic acid (AA) at different temperatures. LDPE: low-density polyethylene; PKS: palm kernel shell.
Table 2 shows the values of A and E a of LDPE/PKS composites with different filler loadings. It can be seen that all values of linear correlation coefficients (R 0) are more than 0.991, which indicate that ln apparent viscosity and 1/T have a strong positive linear correlation. The activation energy, E a, of a material provides important information on the sensitivity of material towards changes in temperature. The higher the activation energy, the more temperature sensitive is the material. The results shown in Table 2 indicate that the addition of PKS into LDPE matrix increased the activation energy of composites. This is due to the different thermal properties of PKS and LDPE matrix. Table 3 shows the constant related to melt viscosity (A) and the activation energy (E a) of the untreated and treated of LDPE/PKS composites. At similar filler loading, the addition of AA into LDPE/PKS composites increased the activation energy of the composites. This is due to the better interfacial bonding between the LDPE matrix and PKS with addition of AA.
Values of A, E a and R 0 of untreated LDPE/PKS composites at different filler loadings.

A: constant related to melt viscosity; E a: activation energy of viscous flow; LDPE: low-density polyethylene; PKS: palm kernel shell; R: universal gas constant.
Values of A, E a and R 0 of untreated and treated LDPE/PKS composites with AA.
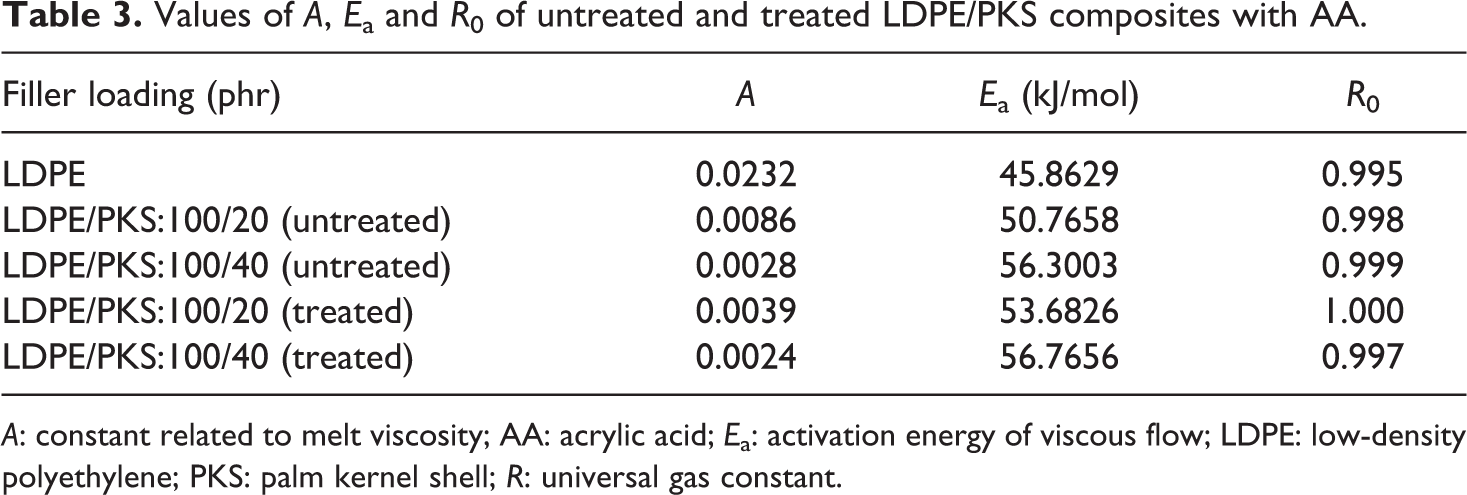
A: constant related to melt viscosity; AA: acrylic acid; E a: activation energy of viscous flow; LDPE: low-density polyethylene; PKS: palm kernel shell; R: universal gas constant.
Thermal properties
Figure 5 shows the comparison of the TGA of PKS, LDPE and untreated LDPE/PKS composites of different filler loadings. In Figure 5, it can be seen that the weight loss of PKS started at 40°C. This is due to the evaporation of volatile and moisture content. The degradation started to take place more rapidly at 204°C. The derivative thermogravimetric (DTG) curves of PKS, LDPE and untreated LDPE/PKS composites in Figure 6 show two decomposition peaks for the PKS, which indicated its two-step degradation. The first and second peak temperatures were 282.3°C and 344.8°C, respectively, which showed the degradation of cellulose and lignin. According to Nicholas, 22 cellulose decomposed at 280°C and ended at 300–350°C and lignin decomposed at 300–350°C and ended at 400–450°C. The weight loss of LDPE occurred in a one-step degradation process from 400°C to 500°C, as shown in Figures 5 and 6. The weight loss in LDPE started at 280.9°C and continued very slowly before reaching the temperature of 400°C. At higher temperature, that is, above 400°C, the degradation process took place rapidly due to the further breakdown of the LDPE into gaseous products. The thermal degradation of the LDPE/PKS composites was a combination phenomenon of thermal degradation of PKS and LDPE. Figure 6 shows the three-step degradation exhibited by LDPE/PKS composites. The first two steps weight loss at 230--300°C were due to the degradation of PKS, while the following weight loss was caused by the degradation of LDPE at 470--500°C. The addition of the PKS into LDPE decreased the onset temperature of the composites due to the lower decomposition temperature of PKS.
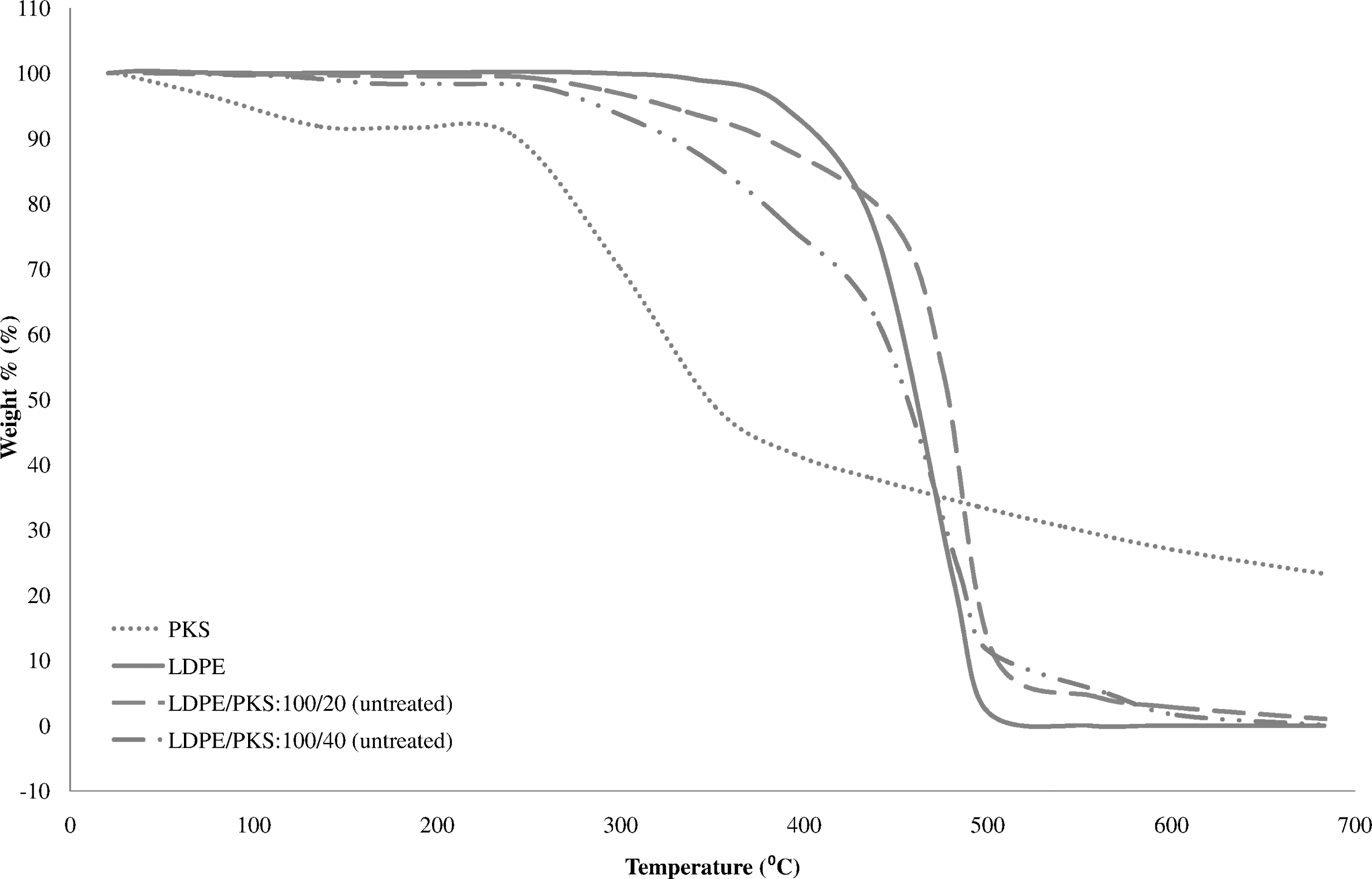
Comparison of thermogravimetric analysis of PKS, LDPE and untreated LDPE/PKS composites of different filler loadings. LDPE: low-density polyethylene; PKS: palm kernel shell.
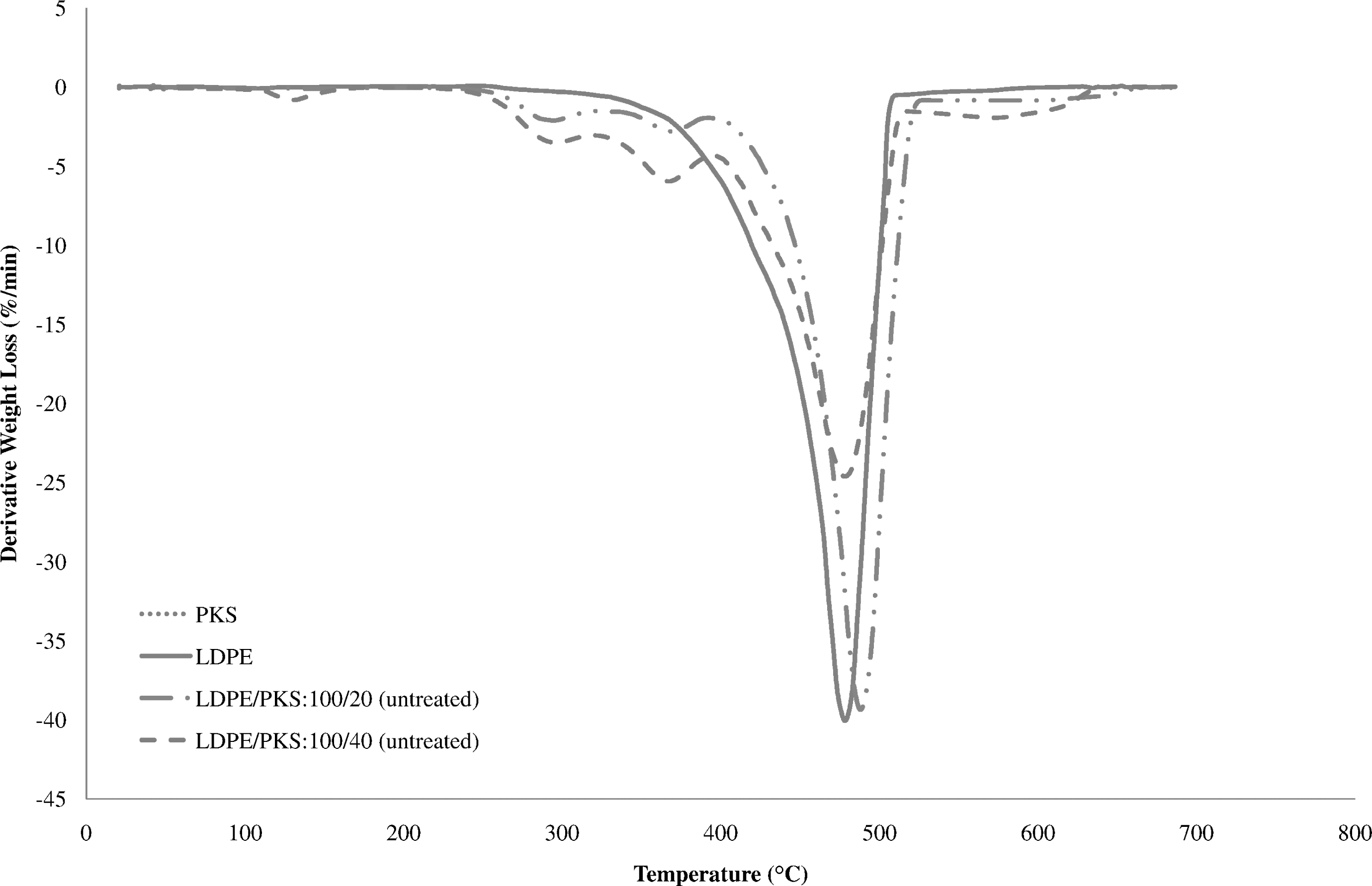
Comparison of derivative thermogravimetry (DTG) curves of PKS, LDPE and untreated LDPE/PKS composites of different filler loading. LDPE: low-density polyethylene; PKS: palm kernel shell.
Figure 7 shows the effect of AA on the TGA of the LDPE/PKS composites at 40 php PKS. The DTG curves of untreated and treated composites are shown in Figure 8. The addition of AA into the composites with 40 php filler loading exhibited higher onset temperature at 218.8°C compared to that of untreated composites at 217.5°C. Table 4 summarizes the derivative data (DTG) for the PKS, LDPE, untreated and treated LDPE/PKS composites with AA. Table 5 shows the total percentage weight loss of PKS, LDPE and LDPE/PKS composites. The total weight loss of LDPE/PKS composites decreased with increasing PKS loading. The addition of PKS improved the thermal stability of the composites. At a similar filler loading, the addition of AA improved the thermal stability of the LDPE/PKS composites with lower total weight loss compared to the untreated composites. The better thermal stability of composites with AA was attributed to the better interfacial bonding between the LDPE matrix and the PKS with addition of AA.
Themogravimetric analysis of PKS, LDPE, untreated and treated LDPE/PKS composites with AA.
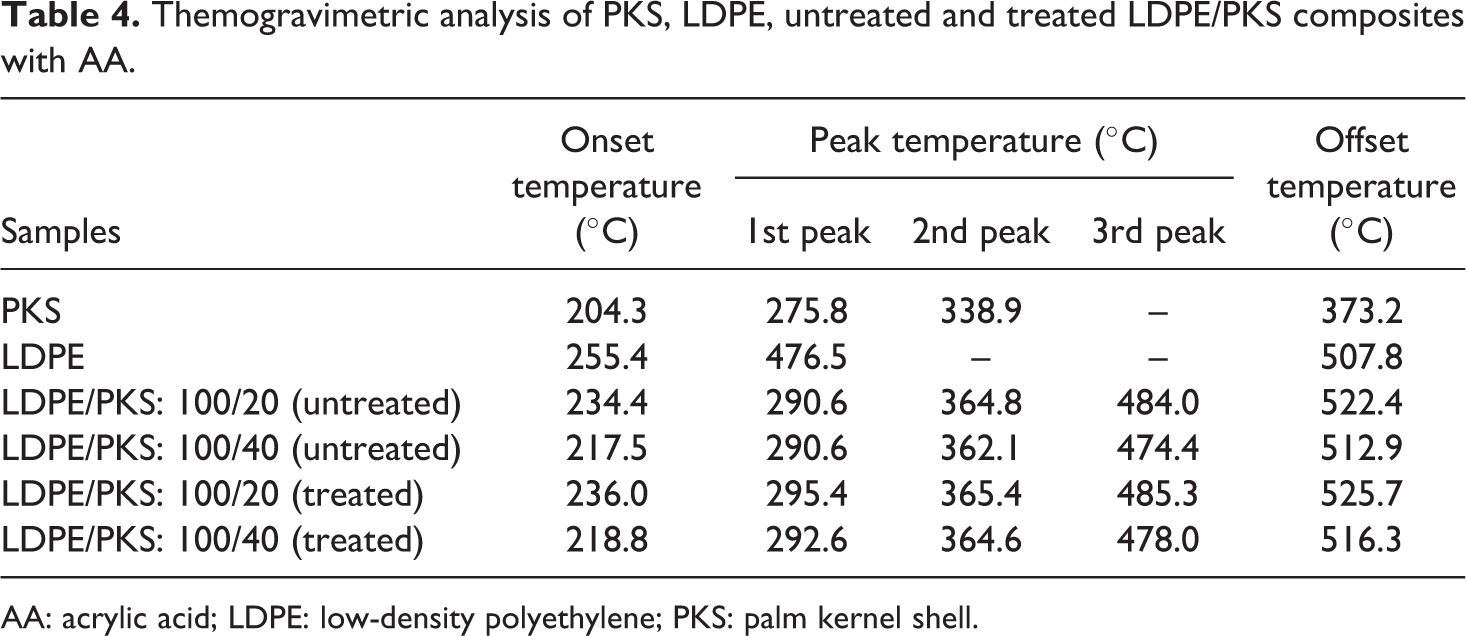
AA: acrylic acid; LDPE: low-density polyethylene; PKS: palm kernel shell.
Percentage weight loss of PKS, LDPE, untreated and treated LDPE/PKS composites with AA.
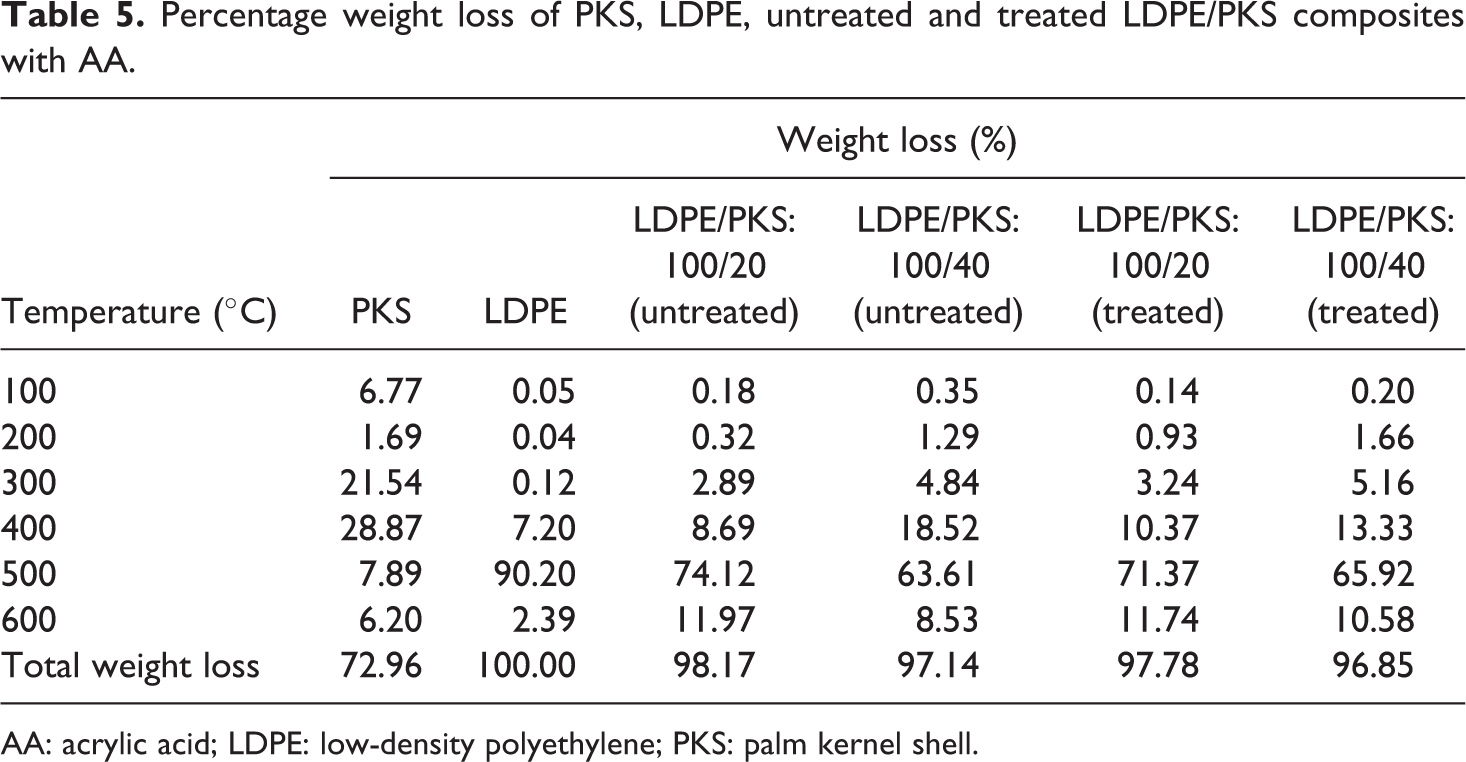
AA: acrylic acid; LDPE: low-density polyethylene; PKS: palm kernel shell.
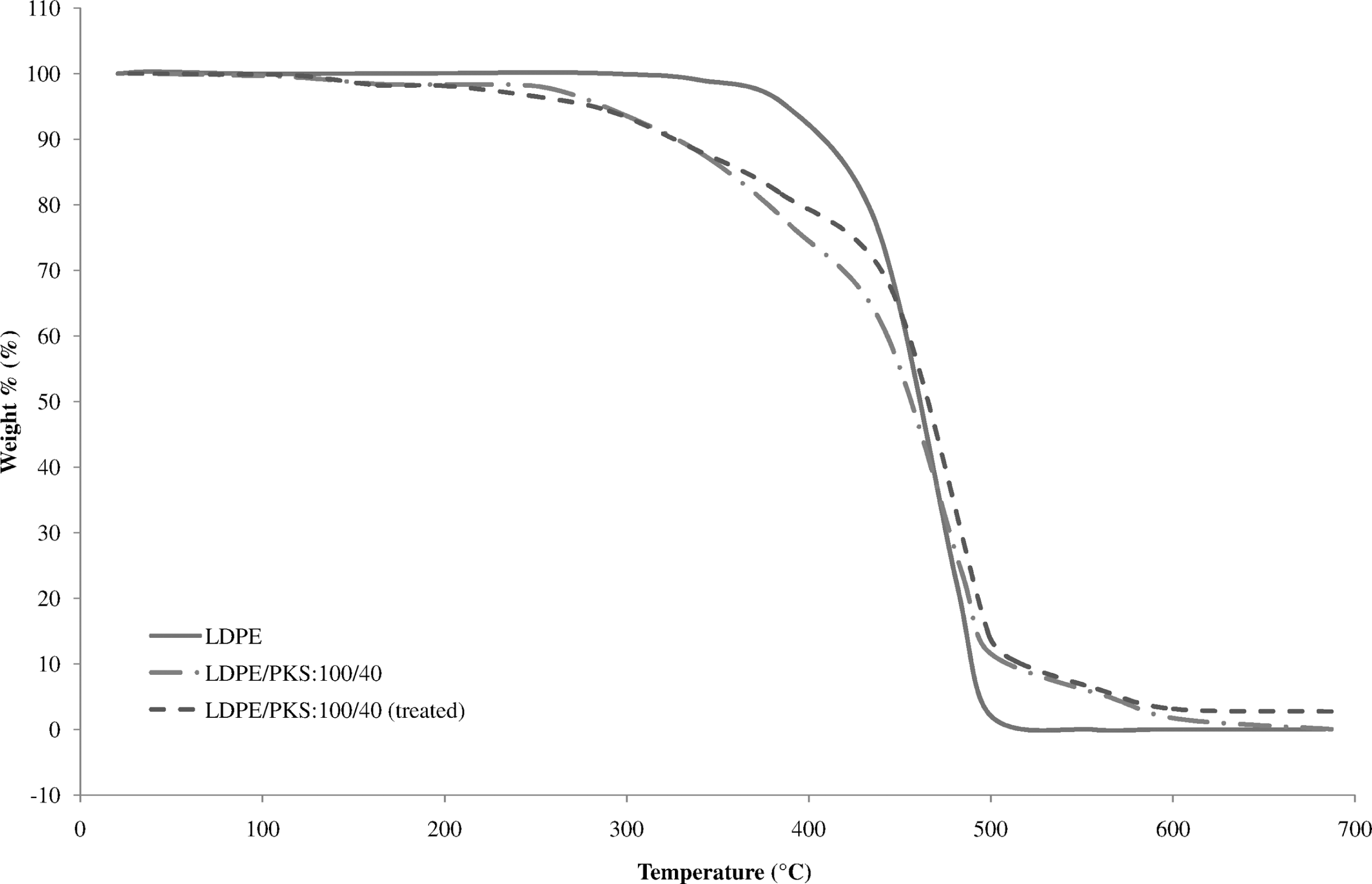
Comparison of thermogravimetric analysis of LDPE, untreated and treated LDPE/PKS composites with acrylic acid (AA). LDPE: low-density polyethylene; PKS: palm kernel shell.
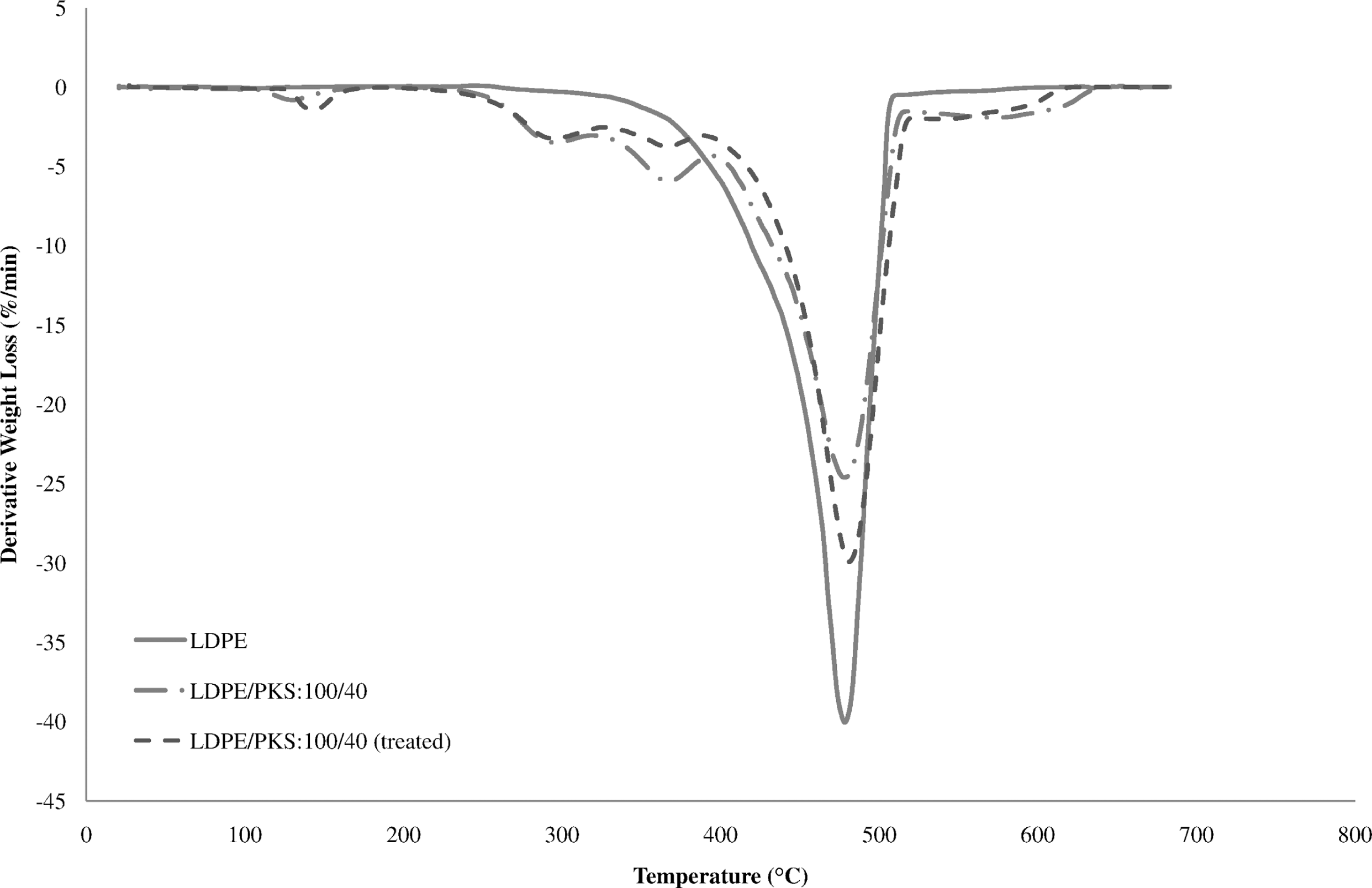
Comparisons of derivative thermogravimetry (DTG) curves of LDPE, untreated and treated LDPE/PKS composites with acrylic acid (AA). LDPE: low-density polyethylene; PKS: palm kernel shell.
Conclusions
The MFI values of the untreated and treated LDPE/PKS composites with AA were decreased with the increase in filler loading. The treated LDPE/PKS composites exhibited lower viscosity compared to untreated LDPE/PKS composites. The apparent viscosity and the activation energy of the composite melt increased with PKS loading. The addition of AA had enhanced the flowability of the composite melt and increased the activation energy of the composites. The onset temperatures of the LDPE/PKS composites were lower compared to the neat LDPE due to the presence of the PKS with lower degradation temperature. The higher PKS loading exhibited better thermal stability of LDPE/PKS composites. The addition of AA has improved the thermal stability of the composites.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
