Abstract
Pyridinium- and phosphonium-based filler surface modifications (tetraphenylphosphonium, hexadecylpyridinium and hexadecyltriphenylphosphonium), which are more thermally stable than the conventionally used ammonium modifications, were exchanged on the filler surface. Polypropylene nanocomposites with the modified fillers were prepared and characterized for gas barrier, mechanical, calorimetric and thermal properties. Mixed morphology consisting of single layers and tactoids of different thicknesses was observed in the composites. The developed morphology in the composites was a result of better thermal stability of the system as well as the nature of the surface modification. Though the completely aromatic surface modification was most thermally stable, it did not improve the composite properties significantly owing to poor interfacial intermixing with the polymer. On the other hand, impressive improvements in the gas barrier and mechanical properties were observed for the filler modifications which included long alkyl chains. These improvements were also better than the corresponding ammonium modifications. The incorporation of the filler led to enhanced thermal resistance of the composites.
Keywords
Introduction
Since the earlier reports on polyamide nanocomposites by Toyota researchers two decades back, in which the layered silicate tactoids could be dispersed into individual nanometer thick platelets in the polymer matrix, large number of research studies have focused on various aspects of such nanocomposites. 1–3 Both polar and nonpolar polymer matrices were subsequently employed to generate polymer nanocomposites using different synthesis methods like melt intercalation, solution intercalation, in situ polymerization and so on. 4–10 Owing to the polar nature of the layered silicate clay platelets and residual polarity even after their surface modification with organic modification molecules via cation exchange on the surface, good dispersion of such platelets and significant enhancement of the properties is generally achieved for polar polymers. However, for nonpolar polymer matrices like polyethylene and polypropylene, owing to the mismatch between the surface polarities of the organic and inorganic phases, the incorporation of the layered silicate filler into the polymers leads to only moderate enhancement in the performance, thus requiring more specific surface modifications. 11–13 Thermal stability of the organic ammonium-based surface modifications at the compounding temperature is another concern. As melt compounding is the commonly used method for the generation of the polyolefin nanocomposites, higher compounding and processing temperatures are required. Such higher temperatures may already be at the borderline of the onset of degradation of the conventional ammonium-based surface modifications. 14 For example, the onset of thermal degradation in the case of organic ammonium modifications is generally observed around 180°C, and the processing temperature of polypropylene is generally 200°C or higher, thus, indicating that the high-temperature processes are detrimental to the interface development in the nanocomposites. 14 The thermal degradation is observed to follow the Hoffmann degradation path in which weaker C–N bonds get broken leading to complete change in the structure and properties of the modification. Thus, even a small extent of such bond rupture can lead to significant impact on the morphology and properties of the composites. The smaller molecular weight species generated after the onset of degradation may also react differently with different polymer matrices. Any theoretical study of the degradation aspect is also missing and perfect thermal stability of the system is assumed in such theoretical models.
As an alternative to the conventional ammonium-based filler surface modifications, thermally stable modifications like imidazolium, pyridinium, phosphonium and so on have been reported in some of the studies. 15–24 Owing to their chemical architecture, these modifications are observed to be significantly thermally stable than the ammonium-based modifications for both onset of thermal degradation and peak degradation temperature values. He et al. 23 reported monoalkylimidazolium modification for the layered silicate platelets and subsequent generation of polypropylene nanocomposites using in situ approach. Mittal 24 reported the synthesis of 1-decyl-2-methyl-3-octadecylimidazolium bromide for exchange on the filler surface. The generated nanocomposites had much higher reduction in the oxygen permeation as compared to the composites with filler carrying ammonium-based modification owing to better interface development between the polymer and the filler.
Owing to their thermal stability, modifications with phosphonium head groups were also explored. Xie et al. 25 also reported on the thermal degradation mechanism of phosphonium-modified montmorillonites, which was different from the degradation pattern of ammonium modifications. The initial degradation of the alkyl phosphonium-modified fillers followed reaction pathways of β elimination and nucleophilic displacement at phosphorus. On the other hand, aryl phosphonium-modified fillers had decomposition via either a reductive elimination or radical generation through homologous cleavage of the P-phenyl bond. Ha et al. 17 subsequently reported polypropylene nanocomposites with two phosphonium-based modifications, trihexyltetradecylphosphonium decanoate and trihexyltetradecylphosphonium tetrafluoroborate, and reported better thermal stability for the composites. Though such modifications are of immense importance for the generation of thermally stable, organically modified fillers for the polymers requiring high processing temperatures, such systems have not been yet optimized as evident from the small number of research studies available on composites incorporating these modifications. Even fewer studies are present for the polyolefin matrices. Polyolefins owing to their tremendous commercial usage need to be developed into thermally stable nanocomposites in order to widen the application potential, thus, it is of interest to further explore the polyolefin nanocomposites with such thermally stable, organically modified fillers. The current study reports the generation of polypropylene nanocomposites with layered silicates modified with phosphonium- and pyridinium-based modifications (different chemical architectures from already reported in the literature). Fillers with two different cation exchange capacities were used. The morphology, mechanical, gas barrier and calorimetric properties of the generated polypropylene nanocomposites have been analyzed.
Experimental
Materials
Homopolymer grade polypropylene (H733-07) was procured from Dow (Dow Plastics, Horgen, Switzerland). The polymer had a melt flow index of 7.5 g/10 min (230°C at 2.16 kg load) and a density of 0.9 g cm−3. Purified sodium montmorillonites with trade names of Cloisite Na and Nanofil 757 were purchased from Southern Clay Inc. (Gonzales, Texas, USA). The cation exchange capacity (CEC) value for Nanofil filler was measured to be 680 µeq g−1, whereas a value of 880 µeq g−1 was measured for Cloisite substrate. 26 Tetraphenylphosphonium chloride and hexadecylpyridinium chloride were procured from Sigma (Switzerland), whereas hexadecyltriphenylphosphonium chloride was supplied by Alfa, Switzerland. Ethyl acetate, ethanol and dioxane were used as solvents and were supplied by Fluka, Switzerland. Ultrapure Millipore water (pH = 5.8, G = 1 mS cm−1) was used for the cation exchange reactions.
Cation exchange
To exchange the sodium ions on the surface of the montmorillonites with phosphonium and pyridinium cations, 8 g of montmorillonite was dispersed in 500 ml of water by stirring at 70°C for 2 h followed by the addition of 200 ml of ethanol. The dispersion was sonicated (ultrasonic horn at 70% amplitude) for 10 min followed by shear mixing for another 10 min (Ultra-Turax T50, IKA, Staufen, Germany). To this dispersion, the required amount of phosphonium or pyridinium salt (100–130% equivalent of the CEC of the montmorillonite) in 100 ml of ethanol was added dropwise under continuous stirring. Thus, the final concentration of montmorillonite in the dispersion was 1 wt%. The reaction mixture was allowed to stir overnight at 70°C. This was followed by filtration of the organically modified filler and washing with hot water–ethanol and subsequently with hot ethanol. In order to completely wash the excess or unexchanged surface modification molecules, the modified filler was again suspended in a solvent in which the optimum swelling of the filler was achieved. The filler was similarly sonicated (5 min, 70% amplitude), stirred overnight at 70°C, filtered, washed and dried at 70°C under reduced pressure. It was necessary to ensure that the modified filler is free from any excess or unexchanged surface modification molecules as such an excess is known to adversely affect the composite properties due to their low thermal stability. 14,27,28 Such modified filler purity was confirmed by high-resolution thermogravimetric analysis (TGA). In the event of indication of the presence of excess surface modification molecules by high-resolution TGA, the washing procedure was repeated till satisfactory filler purity was achieved. Finally the filler was freeze-dried using dioxane as solvent and sieved through 60 μm mesh.
Nanocomposite preparation
Nanocomposites were prepared with 3 vol% of inorganic filler fraction and the required amounts of modified filler and polymer for this purpose were calculated as reported earlier. 10,29 The compounding of filler and polymer was achieved in a twin-rotor batch compounder (Plasticorder W 50 EH, Brabender, Duisburg, Germany) using a compounding temperature of 170°C. The polymer pellets were molten at 170°C at 40 r/min, filler was then gradually added within 10 min at 50 r/min and the mixture was kneaded for further 10 min at 60 r/min. Pure polypropylene was also similarly compounded in order to expose it to same shear and heat history as the polymer in composites. The compound was then quickly taken from the compounder in order to avoid thermal degradation in air and thick plates of 1.5 mm thickness were compression molded in a brass frame between two thin aluminum plates under vacuum. 30 Thin films (approx 100 μm thickness) of the pure polymer and the composites were also similarly pressed by compression molding. 10
Characterization of nanocomposites
For the mechanical testing of the polymer and composites, dumbbell-shaped samples (type 5B) were stamped out from the compression-molded plaques. A stamping press (H. W. Wallace, Croydon, Surrey, England) with a die conforming to the standard of ISO 527-2 was used for this purpose. Universal testing machine Zwick Z020 with testXpert 9.01 software (Zwick, Ulm, Germany) coupled with a Video-Extensometer V4.19.02 (Messphysik, Furstenfeld, Austria) was used for the tensile testing of the dumbbell-shaped samples at room temperature. Different extension speeds were used during the single test: 0.1 mm min−1 for the measurement of elastic modulus and 6 mm min−1 for the measurement of other tensile properties. Elastic modulus was determined in the range of 0.05–0.25% strain and an average of 5 measurements was reported.
Oxygen permeation studies through the polymer and nanocomposite films were carried out on OX-TRAN 2/20 ML (Mocon, Minneapolis, Minnesota, USA) modules at 23°C and 0% relative humidity. The exact thickness of the films was required to normalize the gas permeation rates, which was measured by a process reported earlier. 29
As mentioned earlier, high-resolution (Hi-Res) TGA of the modified fillers was performed in order to confirm the purity of the filler surface. In such a device Q500 Thermogravimetric Analyzer (TA Instruments, New Castle, Delaware, USA), the heating rate is coupled to mass loss so that the sample temperature is not raised until the mass loss at a particular temperature is completed. Similar analysis was also carried out for selected composites. Measurements were performed in air atmosphere in the temperature range 50–900°C at a heating rate of 20°C min−1. Dynamic TGA was also performed to compare the thermal stability of modified fillers. The sample was first equilibrated at 140°C. Then, the sample was heated to 180°C and kept isothermally at this temperature for 90 min. Heat capacity values of the polymer were measured in Differential scanning calorimetry (DSC) 7 (Perkin Elmer, Norwark, Connecticut, USA) using a heating rate of 10°C min−1. 31 A temperature range of 100–180°C was used.
Wide-angle Scintag XDS 2000 diffractometer (Scintag Inc., Cupertino, California, USA) using CuKα radiation (λ = 0.15406 nm) in reflection mode was used to collect X-ray diffraction patterns for the filler in the powder form as well as in composites. The morphology of the composites was evaluated by bright field transmission electron microscope (Zeiss EM 912 Omega). Thin nanocomposite films were etched with oxygen plasma for 5 min and embedded in an epoxy matrix followed by sectioning of 70–90 nm thick sections. These were subsequently supported on 100 mesh grids sputter coated with a 3 nm thick carbon layer.
Results and discussion
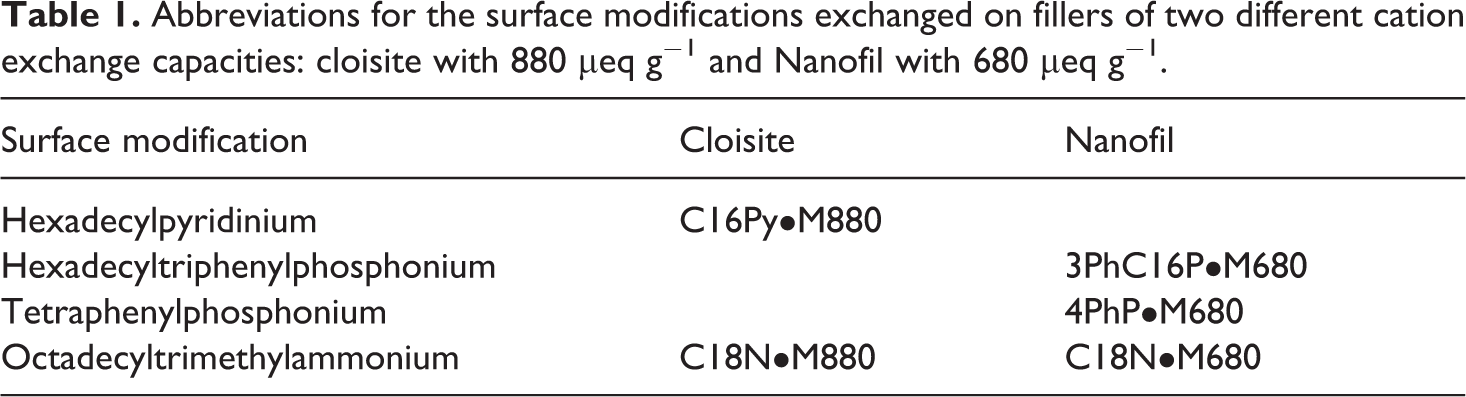
The importance of thermal stability of the polymer as well as organic filler modification at the compounding temperature for sufficient period of time is vital for the development of nanocomposite morphology in which the interface between the filler and the polymer is optimally defined. As the thermal degradation of the ammonium-based surface modification is always of concern especially for polymers requiring higher compounding temperatures, more thermally stable pyridinium- and phosphonium-based surface modifications were exchanged on the filler surface. The chemical architectures of these modifications were also varied in order to study their interactions with polymer. Tetraphenylphosphonium modification was aromatic in nature and had no long alkyl chain generally present in the organic modifications which leads to higher basal plane spacing in the modified filler. Hexadecyltriphenylphosphonium had both aromatic and aliphatic components, whereas hexadecylpyridinium comprised only of long hexadecyl chain in the molecule. The long hexadecyl chains in these surface modifications help in significantly separating the clay platelets thus reducing the attractive forces among them and their similar chemical nature as polypropylene was also expected to help in better intermixing at the interface. Figure 1 shows the chemical structures of these phosphonium and pyridinium modifications. Table 1 also shows the abbreviations for the surface modifications exchanged on the filler surface. Fillers with two different cation exchange capacities (680 and 880 μeq g−1) were used. Comparisons with earlier reported composites containing filler treated with ammonium-based modification octadecyltrimethylammonium 10,30 were also performed. Compounding temperature was maintained at 170°C in order to avoid excessive degradation of polymer or surface modification. The compounding time was also optimized to be sufficient to ensure a good mixing but not long to degrade the polymer. Inorganic filler fraction of 3 vol% was maintained in all the composites so as to compare the composite properties.
Chemical structures of (a) tetraphenylphosphonium, (b) hexadecyltriphenylphosphonium and (c) hexadecylpyridinium modifications.
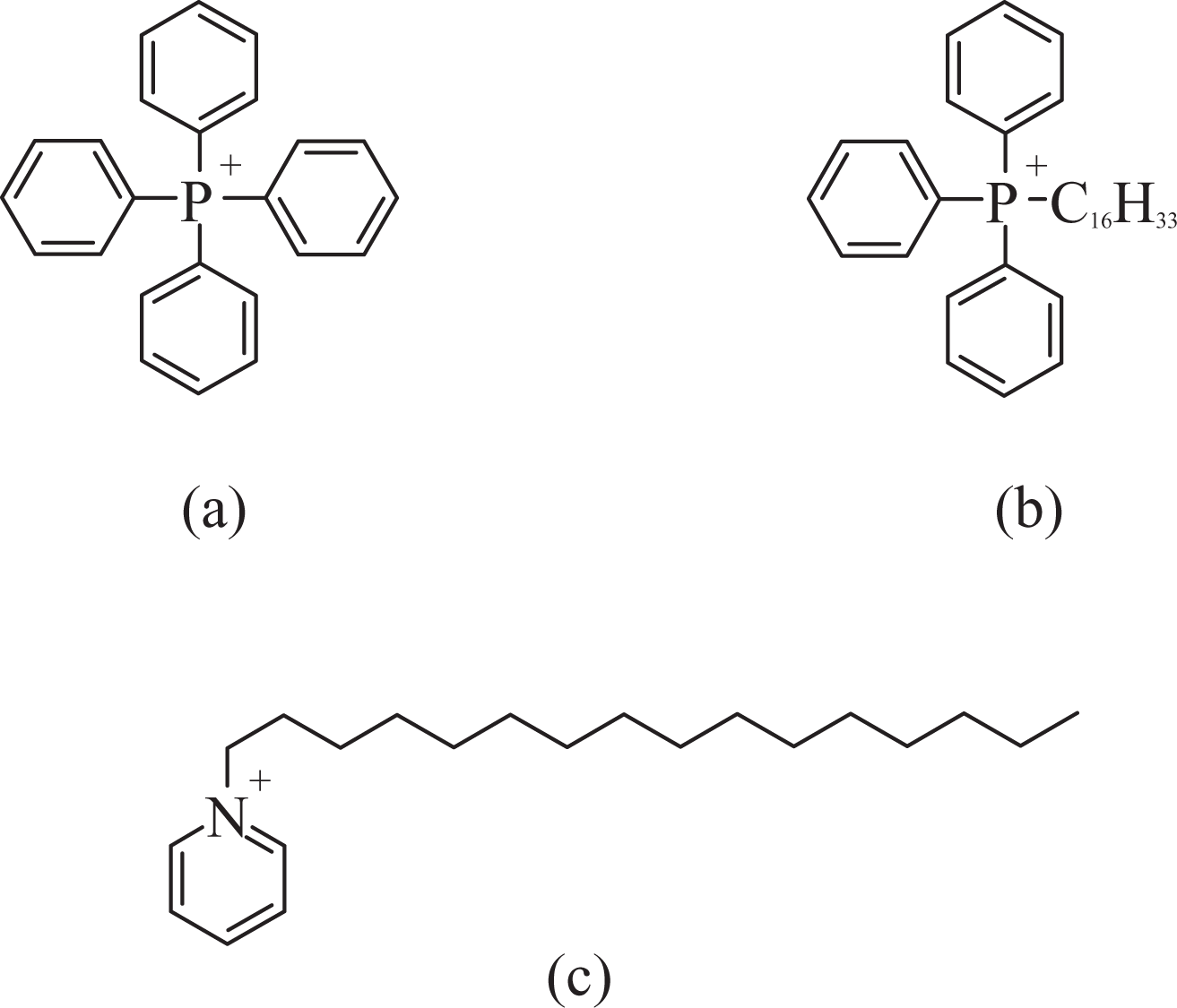
Figure 2(a) compares the thermal performance of pyridinium-modified filler (C16Py•M880) with octadecyltrimethylammonium-modified filler (C18N•M880). The delayed onset of degradation in the case of C16Py•M880 was clearly observed, indicating better thermal stability. The peak degradation temperature was also observed to be more than 45°C higher in the case of C16Py•M880 filler (263°C for C18N•M880 in comparison with 301°C for C16Py•M880). Figure 2(b) also compares the thermal responses of C18N•M680 with phosphonium-modified fillers 3PhC16P•M680 and 4PhP•M680. Similar to the case of pyridinium-modified filler, the phosphonium-modified fillers too had much better thermal stability than the ammonium-modified filler as indicated by higher onset of degradation temperatures. 4PhP•M680 had especially very high thermal stability as no degradation was observed to occur even at 400°C. Thus, the pyridinium and phosphonium modifications were more suitable for the high-temperature compounding than the corresponding ammonium modifications especially for polymers like polypropylene.
Thermogravimetric analysis (TGA) thermograms of (a) C18N•M880 (I) and C16Py•M880 (II); (b) C18N•M680 (I), 3PhC16P•M680 (II) and 4PhP•M680 (III).
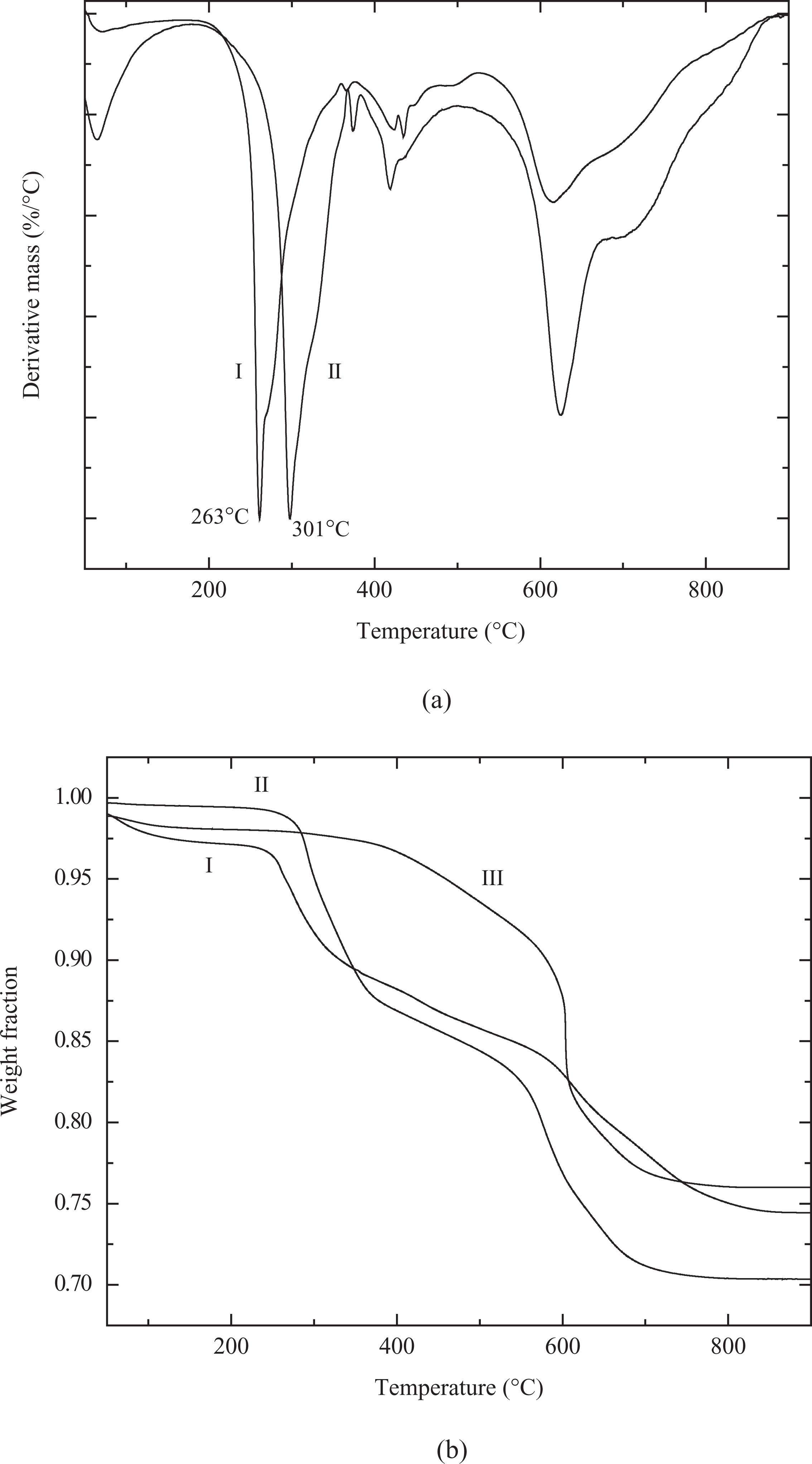
In order to ascertain whether the higher thermal stability of the pyridinium and phosphonium modifications is also retained during the complete cycle of compounding, dynamic TGA was also performed for 3PhC16P•M680 and compared with earlier reported dioctadecyldimethylammonium-modified filler 24 as shown in Figure 3. The analysis was carried out at 180°C under isothermal conditions for 90 min. The ammonium-modified filler was observed to have higher magnitude of weight loss than the phosphonium-modified filler indicating faster thermal degradation. Though the difference in weight loss between the two fillers is not very significant, as the degradation in the case of ammonium modification occurs by the breaking of weaker C–N bonds, therefore, even a small amount of such bond rupture can be detrimental to interfacial interaction between the filler and the polymer. Thus, the TGA studies confirmed the superior thermal performance of phosphonium- and pyridinium-modified fillers over the ammonium-modified fillers.
Dynamic thermogravimetric analysis (TGA) thermograms of 3PhC16P•M680 (I) and 2C18N•M880 (II).
Table 2 lists the basal plane spacing values of the pyridinium- and phosphonium-modified fillers as well as of the 3 vol% composites. Figure 4 also shows the X-ray diffractograms of the filler powders and the fillers in composite films. Absence of any diffraction peak attributed to the pristine filler in the modified filler diffractograms indicated that the exchange of ions on the surface with the organic modifications was satisfactorily achieved. C16Py•M880 was observed to have a basal plane spacing of 1.75 nm which was similar to the C18N•M880 filler value of 1.84 nm. Though 3PhC16P•M680 has also only a single long alkyl chain as C16Py•M880, in this case, much higher basal plane spacing of 2.12 nm was observed. This is owing to the higher cross-sectional area of the modification molecules which helps them to occupy more space per cation on the filler surface, leading to more vertical posturing of the alkyl chains which results in higher basal plane spacing. Though 4PhP modification had no long alkyl chain in the chemical structure, the basal plane spacing was still higher than C16Py•M880 owing to the similar effect of higher area of cross-section of the molecules. The basal plane spacing of the filler in the composites was almost same as modified fillers except 3PhC16P•M680 where some polymer intercalation in the filler interlayers was evident. However, such response from the X-ray diffraction studies of unaffected basal plane spacing values in the composites is expected and earlier reported. 10,30 Owing to the absence of any polar interaction between the filler and apolar polymer chains and owing to the absence of any externally added amphiphilic compatibilizer, complete delamination of filler in nanometer platelets cannot be expected. However, owing to the optimum surface modification and subjected shear in the compounder, a decrease in tactoid thickness occurs and such a phenomenon can be observed in transmission electron microscopy. Even reduction in peak intensity in X-ray diffractograms is sometimes considered as an indication of the presence of intercalated or exfoliated morphology, but the intensity is also influenced by sample preparation as well as concentration and orientation of the filler in the composites, thus, requiring other characterization method. Figure 5 shows the transmission electron micrographs of the polypropylene composite with 3PhC16P•M680 filler. Tactoids of varying thicknesses were present in the micrographs. Presence of some single platelets also confirmed partial exfoliation of the filler even in the absence of conventionally added low-molecular-weight compatibilizers. Apart from the presence of mixed morphology of single platelets and tactoids of varying thickness, the platelets were also occasionally observed to be bent and folded and lacked any alignment. Other composites also exhibited similar mixed morphology, however, the tactoid thicknesses were higher in the case of 4PhP•M680 than C16Py•M880, indicating less filler delamination in the case of 4PhP•M680. This can be the result of the presence of four phenyl groups in the surface modification which may not intermix uniformly with the polypropylene chains at the interface. On the other hand, the presence of alkyl chains in the chemical structures of C16Py•M880 and 3PhC16P•M680 would have helped to entangle well with the polypropylene leading to their delamination by shear.
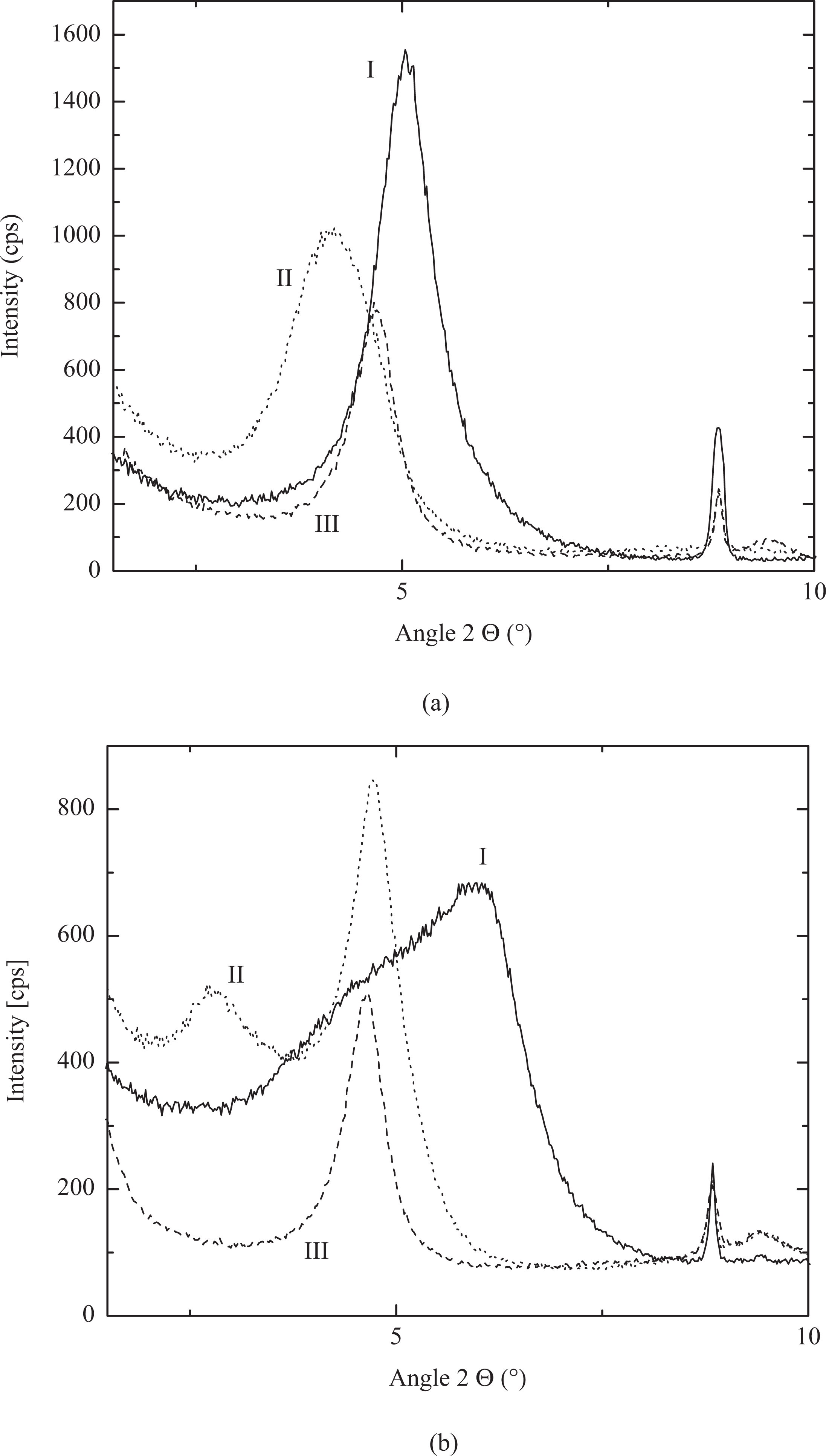
X-ray diffractograms of (a) C16Py•M880 (I), 3PhC16P•M680 (II) and 4PhP•M680 (III) fillers and (b) their 3 vol% polypropylene nanocomposites.

Transmission electron micrographs of 3 vol% composite with 3PhC16P • M680 filler. The arrows indicate either single clay layers or tactoids of varying thicknesses.
Basal plane spacing of organically modified fillers and their polypropylene nanocomposites.
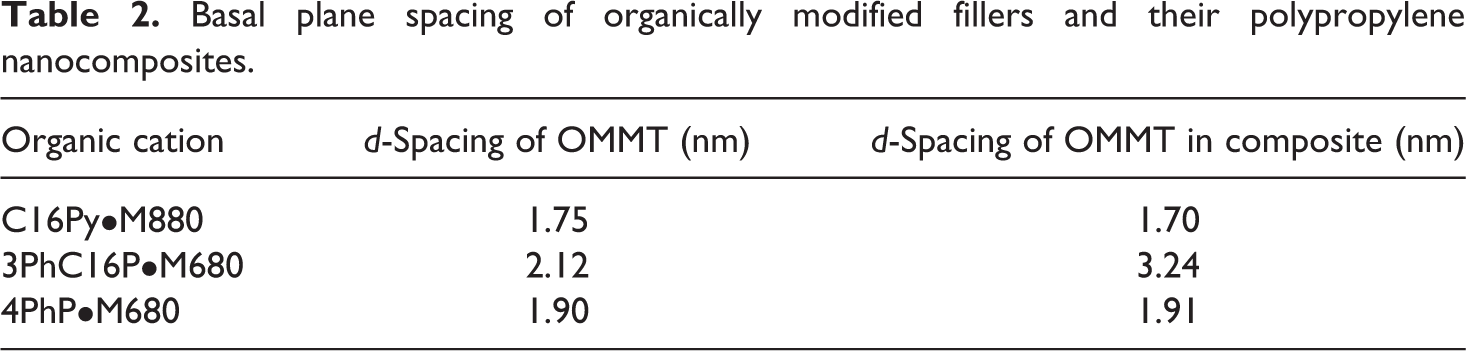
Table 3 shows the oxygen permeation coefficients of the composites with three different modified fillers. The oxygen permeation coefficient through pure polypropylene films was observed to be 89 cm3 μm/m2 day mmHg, which was decreased to varying extents in the nanocomposites. 4PhP•M680 composite showed a decrease of 14% in the oxygen permeation as the permeation coefficient through the composites was measured to be 76.7 cm3 μm/m2 day mmHg. For composites containing 3PhC16P•M680- and C16Py•M880-modified fillers, oxygen permeation coefficients were 70.8 and 69.7 cm3 μm/m2 day mmHg, respectively, indicating permeation reduction of 21.5% and 22% as compared to the pure polypropylene. The ammonium-based composites (C18N•M680 and C18N•M880) were observed to have permeation reduction of 17% as compared to neat polymer, thus confirming better gas barrier properties of 3PhC16P•M680 and C16Py•M880 nanocomposites. 10 The low gas barrier performance of the 4PhP•M680 composite could also be related to the findings from morphology evaluation. The tactoids were thicker in this composite (thus lower aspect ratio), which resulted in lower extent of hindrance to the diffusion of oxygen molecules through the polymer. On the other hand, the aspect ratio of the filler in 3PhC16P•M680 and C16Py•M880 composites was higher owing to lower tactoid thickness leading to better gas barrier performance. Thus, the better thermal stability of the filler modification in combination with the interfacial interactions resulted in the development of better gas permeation resistance.
Oxygen permeability coefficients (23°C, 0% RH) of the 3 vol% nanocomposites.

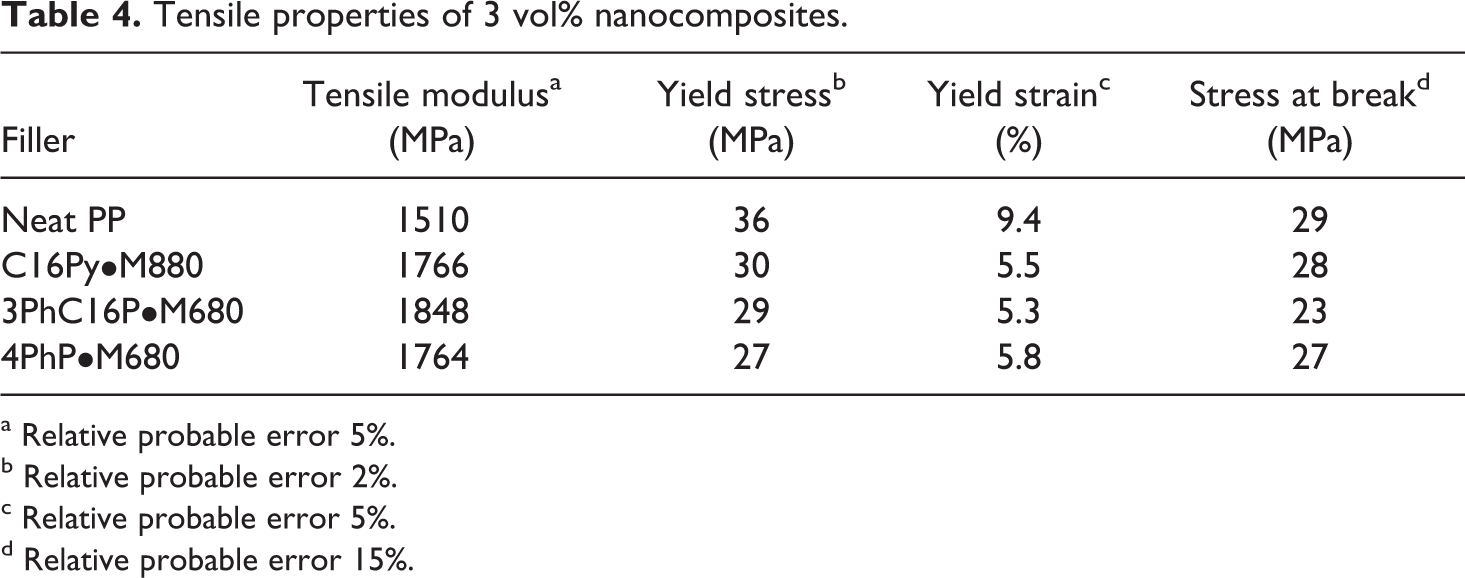
Table 4 describes the tensile properties of the 3 vol% polypropylene nanocomposites with the modified fillers. In all the composites, the tensile modulus was enhanced owing to stress transfer to the inorganic filler particles. An increase of 17% in the tensile modulus was observed for 4PhP•M680 composites as compared to pure polymer. For C16Py•M880- and 3PhP•M680-containing composites, tensile modulus values of 1844 and 1848 MPa were observed, indicating an increase of 22% in the modulus as compared to pure polypropylene. These enhancements were consistent with the barrier performances as well as morphology analysis. These findings also underline the fact that only the exfoliated filler platelets are responsible for the significant enhancement of polymer properties in the nanocomposites as the intercalated platelets or thick platelets do not increase the aspect ratio of the filler. The increase in tensile modulus in the case of 3PhP•M680-containing composite was much better than the corresponding C18N•M680 composite, further confirming the impact of better thermal stability of the filler on composite properties. Other tensile properties were decreased as compared to pure polymer owing to the presence of incomplete exfoliation (presence of thicker tactoids) and strain hardening of the polymer chains confined in the filler interlayers (increased brittleness). The decrease was more pronounced in the case of 4PhP•M680 composites where a reduction of 25% in the yield stress as compared to pure polymer was observed. The yield strain was also the lowest in this case, indicating more brittleness as compared to other composites owing to the presence of thicker filler particles and absence of any interfacial interactions.
Tensile properties of 3 vol% nanocomposites.
a Relative probable error 5%.
b Relative probable error 2%.
c Relative probable error 5%.
d Relative probable error 15%.
It was earlier reported that the incorporation of thermally stable imidazolium-modified clay in polypropylene did not have any effect on the crystallization of the polymer in the processing conditions used. 24 It was also of importance to study the similar behavior for the pyridinium and phosphonium fillers employed in this study in order to correctly relate the effects on the nanocomposite properties to the filler exfoliation and interfacial interactions between the filler and the polymer. Table 5 shows the calorimetric behavior of the polypropylene and its composites with different fillers. No significant changes were observed in the onset and peak melting and crystallization temperatures in the pure polymer as well as composites. Figure 6 also shows the DSC thermograms for the crystalline melting and crystallization transitions of the polymer compared with the polymer composites. The thermograms of the different composites were observed to be indistinguishable and also largely overlapped with the pure polymer. Degree of polymer crystallinity calculated by comparison of the enthalpy of melting of polymer in the nanocomposite (corrected due to the presence of filler) and theoretical enthalpy of purely crystalline polymer 32,33 also did not vary in the nanocomposites. It was thus confirmed that the changes in the polymer properties by the incorporation of filler were a result of nanocomposite morphology evolution and not due to changes in the polymer crystallinity.
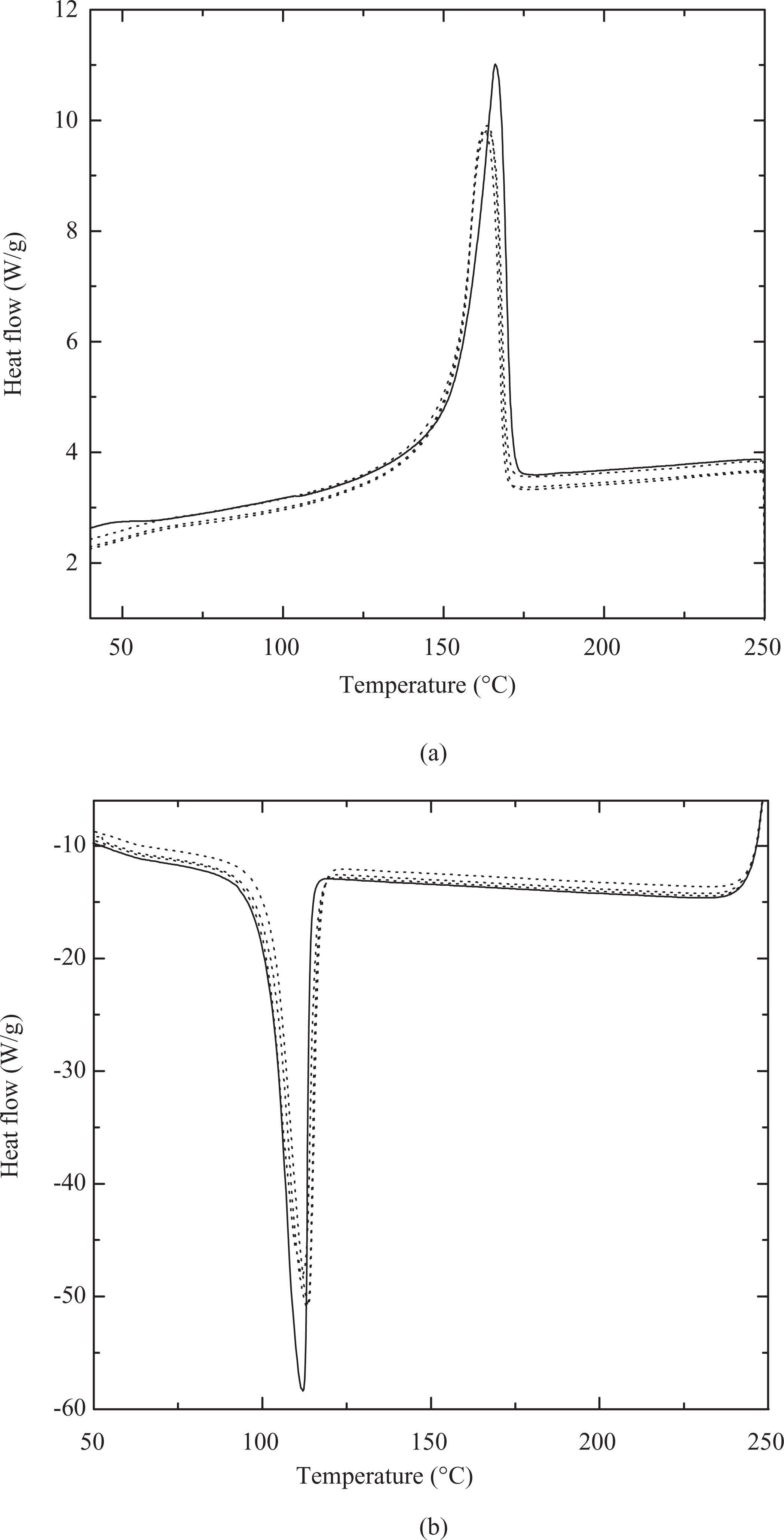
Differential scanning calorimetry (DSC) thermograms of (a) crystalline melting and (b) crystallization transitions of pure polymer (solid line) and 3 vol% composites (dotted lines).
Calorimetric behavior of polypropylene and its 3 vol% nanocomposites.
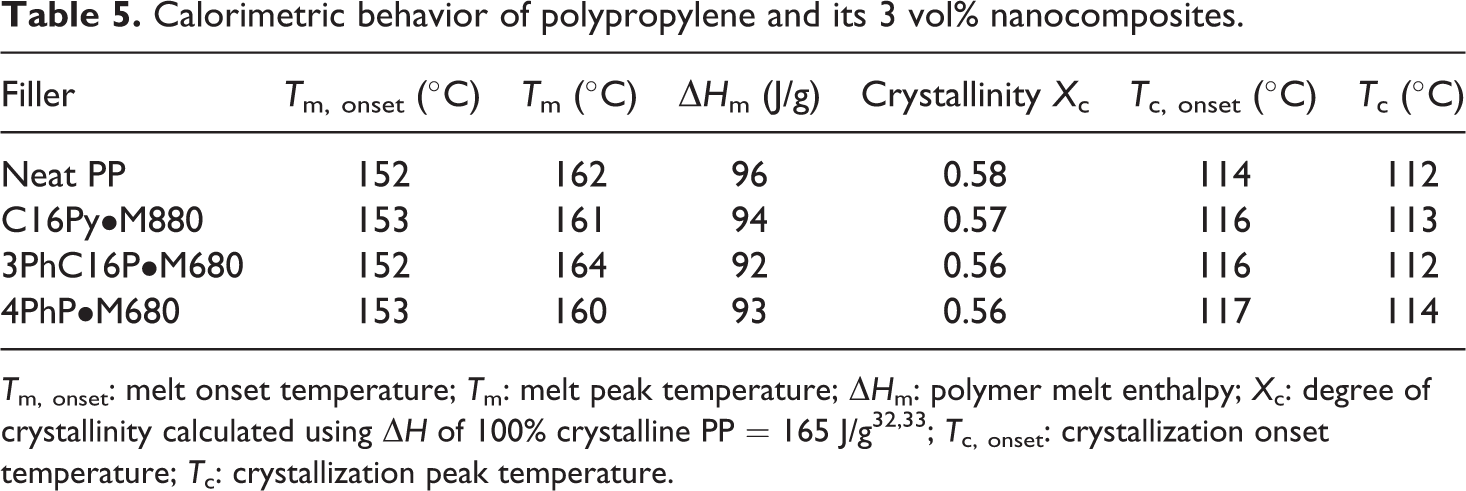
T m, onset: melt onset temperature; T m: melt peak temperature; ΔH m: polymer melt enthalpy; X c: degree of crystallinity calculated using ΔH of 100% crystalline PP = 165 J/g32,33; T c, onset: crystallization onset temperature; T c: crystallization peak temperature.
Figure 7 also shows the synergistic improvements in the thermal stability of the nanocomposite. The TGA thermograms of C16Py•M880-modified filler have been plotted with thermograms of pure polymer as well as 3 vol% polymer composite. The polymer was observed to degrade sharply around 260°C, whereas the composite degradation was initiated only after 300°C, owing to the incorporation of filler.
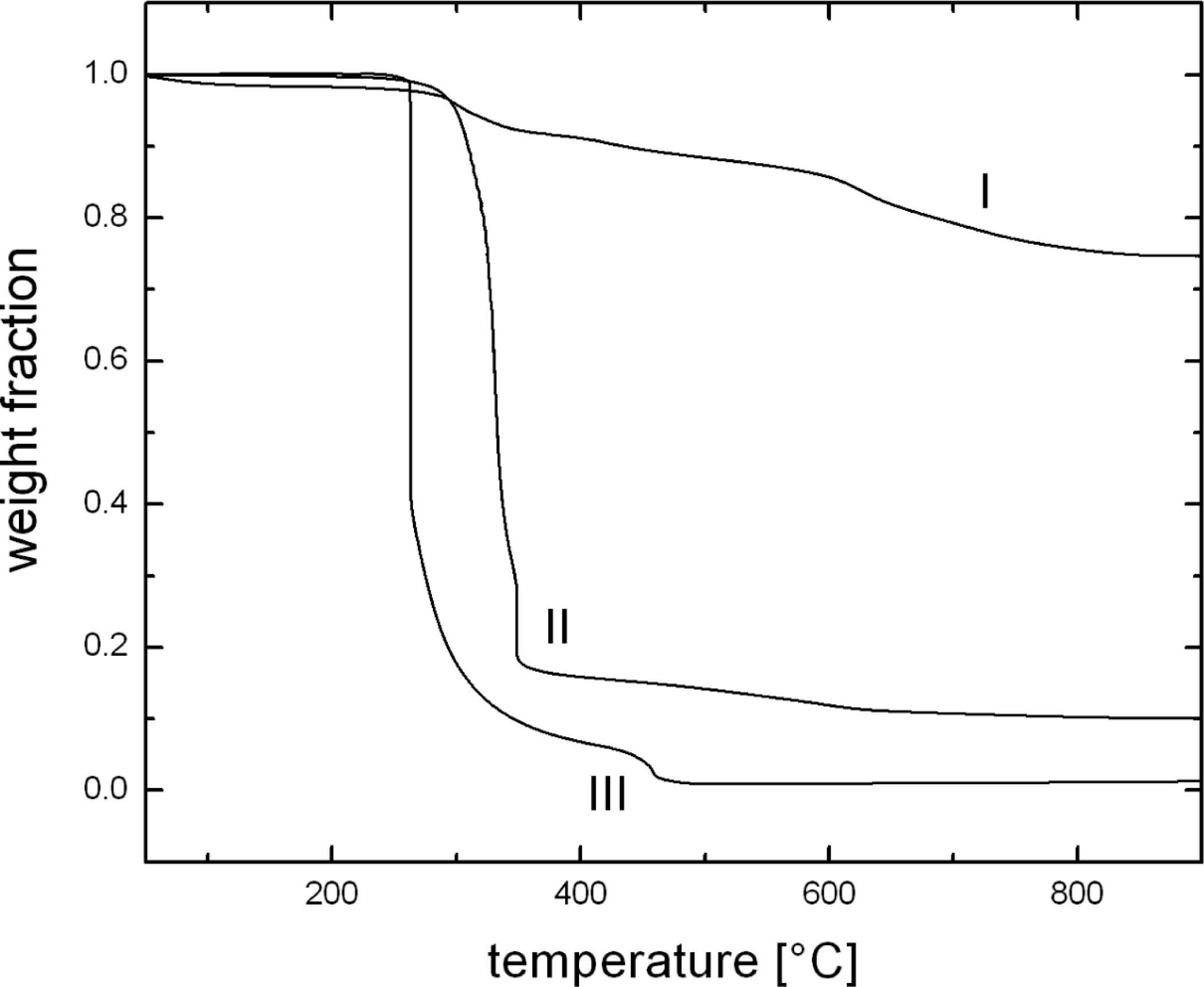
Thermogravimetric analysis (TGA) thermograms of C16Py • M880-modified filler (I), pure polypropylene (III) and 3 vol% polypropylene composite with C16Py • M880 (II).
Conclusions
The better thermal stability of pyridinium and phosphonium surface modifications ionically bound on the filler surface was confirmed through isothermal and nonisothermal TGA. The thermal performance of the polymer was synergistically enhanced after incorporation of modified filler. The morphology generation in the polypropylene nanocomposites was affected by thermal stability of the filler as well as interactions between the filler surface modification and the polymer chains. The most thermally stable modification 4PhP•M680 did not result in the best performance, owing to poor intermixing between the phenyl-based modification and alkyl polymer chains. On the other hand, alkyl chain carrying modifications like C16Py•M880 and 3PhC16P•M680 led to the generation of partially exfoliated nanocomposites. The partial delamination corresponded with significant decrease in oxygen permeation and increase in tensile modulus. Absence of complete exfoliation led to the reduction in other tensile properties and the decrease was the maximum in 4PhP•M680-based composite. The fillers did not affect the crystallization behavior of the polymer.
Footnotes
Acknowledgement
The experimental work was carried out at Institute of Polymers, ETH Zurich, Switzerland.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.