Abstract
Novel polyimide/clay nanocomposites were fabricated by incorporating organically modified clay at different loading percentage of 1–5 wt% within the polyimide (PI) matrix. A novel PI was synthesized through a direct polycondensation reaction between novel 5,5′-((4-methoxyphenyl)azanediyl)bis(isobenzofuran-1,3-dione)and 4,4′-(1,4-phenylenebis(oxy))bis(3-(trifluoromethyl)aniline). The organoclay was prepared from Sodium montmorillonite (Na-clay) and protonated form of diamine (modifier) via ion-exchange reaction. The structural characterization of nanocomposites were made using Fourier transform infrared spectroscopy (FT-IR), X-ray diffraction (XRD), scanning electron microscopy (SEM) and transmission electron microscopy (TEM), the mechanical properties were obtained by tension tests. The results of XRD, TEM, and FE-SEM indicated that clay could well disperse in the PI matrix and were intercalated by diamine and PI macromolecules. The thermal stability and glass transition temperature of nanocomposite at lower clay loadings are similar to PI but gradually decrease with increase in clay content. The tensile properties of PI/Clay nanocomposites showed the highest values at the optimum clay content of 3 wt%. The gas permeation of PI/clay nanocomposites was increased with the increase in loading percentage of organoclay, whereas the gas selectivity was maintained at par to that of neat PI film until it diminished at 5 wt% percentage of organoclay causing mineral agglomeration. PI/clay 3 wt% MMMs exhibited 245% and 240% increase in CO2 and O2 gas permeability respectively, while showing minimal variation in the selectivity of O2/N2 and CO2/N2 compared to that of neat PI matrix.
Introduction
The unrelenting efforts to form novel materials to surpass the ‘upper bound’ limitation lead to the development of state-of-theart organic–inorganic hybrids, generically called mixed matrix membranes (MMMs). MMM is comprised of discrete inorganic phase dispersed in a continuous polymer phase which brings forth novel hybrid material with synergistic advantages from each phase: high selectivity of the dispersed particle, desirable mechanical integrity, and economical processability of the polymer matrix.1–3 At the early stage of MMM development, selective adsorbent or molecular sieve inorganic particles such as zeolite became the main choice for the filler material. 4
Polyimides (PIs) are widely used in the microelectronics industry as buffer coatings, passivation layers, α-particle barriers, interlayer dielectrics, wafer-scale packages, etc., because of their mechanical, thermal and electrical properties, and chemical resistance. In order to improve the utility of PIs in electronic industries, many kinds of PI/clay nanocomposites have been studied using different dianhydrides and diamines. Various dianhydrides widely used to prepare PI/clay nanocomposites are pyromellitic dianhydride (PMDA), 3,3′,4,4′-biphenyltetracarboxylic dianhydride (BPDA), benzophenone-3,3′,4,4′- tetracarboxylic dianhydride (BTDA), 2,2-bis [4-(3,4-dicarboxyphenoxy)phenyl]propane dianhydride, oxydiphthalic anhydride (ODPA) and 2,2-bis(3,4-dicarboxyphenyl) hexafluoropropane dianhydride (6FDA).5–10
Clay minerals have been shown to be good candidates for the preparation of nanocomposites because of their easy availability, low cost, layered structures and ability to become organophilic by cation-exchange reaction. Clay minerals such as montmorillonite, saponite and hectorite have mainly been used to form polymer-based nanocomposites. 11 These clayminerals usually contain hydrated inorganic cations, such as Na+, K+, Ca2+, and are hydrophilic. These clay minerals have to be rendered more organophilic to make them compatible with organic polymers. To attain this, the hydrated cations of clay minerals are exchanged with cationic surfactants, such as quaternary alkylammonium cations. 12
On the one hand, the molecular diffusion and shear critically depend on the surface modification of montmorillonite (MMT), such as an optimal structure, size and adsorption content of surfactant. The organic modification of MMT decreases the energy of adhesion between layers, thus facilitating the molecular diffusion and shear.13,14 The increase of the number of alkyl tails or the adsorption content of surfactants will increase the interlayer distance and weaken the attraction between adjacent platelets.4,15 However, it also increases steric hindrance, and the molecular diffusion of polymers becomes more difficult.
The chemical structure and melt viscosity of polymers influence the molecular diffusion and shear. Balazs proposed a structure of polymer that was beneficial to the exfoliation via molecular diffusion.13,16 The polymer should contain fragments attracting to the MMT surface in order to promote the compatibility between the polymer and MMT; meanwhile the polymer should also contain longer fragments that are not attracted to MMT surface in order to exfoliate MMT layers. As we all known, polar polymers have stronger interaction with the surface of MMT than polar polymers.17–19 The favorable interaction makes polymers intercalate into the galleries of OMMTs easily, and shear stress can also be transferred effectively. But the resistance to diffusion of polar polymers in the interlayers also increases with the enhancement of interactions. Therefore the exfoliation cannot be achieved via molecular diffusion in some polar matrixes. 20 In addition, the melt viscosity of polymer is also a vital factor because different melt viscosities will result in different diffusion ability and shear stress. The exfoliation needs a balance between shear stress that requires high viscosity and diffusion process that needs rather low viscosity.21,22
In order to synthesize novel high performance materials with improved processability and solubility with respect to aromatic PI, PI nanocomposites were prepared through a solution intercalation method using different loading percentage organoclays. The effects of nanofiller type on morphology, thermal properties, gas permeation and mechanical of PI were explored in details.
Materials
5-bromo-2-methylisoindoline-1, 3 dione, 4-methoxyaniline and tetrakis(triphenylphosphine) palladium (0) were purchased from Alfa Aesar and used as received. Commercially available N-methyl-2-pyrrolidinone (NMP), m-cresol, N, N-dimethylacetamide (DMAc), N,N-dimethylformamide (DMF), dimethyl sulfoxide (DMSO), tetrahydrofuran (THF) and other reagents were all used as received. Research grade ethane, ethylene, propane and propylene gases (purity499.95%) were purchased from Technical Gas Service. All the materials and solvents that were used were obtained from the suppliers without further purification. The sodium montmorillonite clay (Closite Na+, cation-exchange capacity (CEC) = 92 mmol/100 g, denoted as Na-MMT) was purchased from Southern Clay Products Co. (USA).
Equipments
Fourier transform infrared spectroscopy (FT-IR) analysis was performed on an FT-IR spectrometer (ALPHA, Bruker) in the frequency range of 400–4000 cm-1 at room temperature. Xray diffraction (XRD) patterns were recorded on a diffractometer (D/max-2500, Rigaku), using CuKα radiation at a scanning rate of 5°/min (from 10° to 80°). Proton nuclear magnetic resonance (1H NMR, 500 MHz) spectra were carried out using Bruker Avance 500 instrument (D-755 of the Roger-Gaudry, Germany) at room temperature (RT) in dimethylsulphoxide-d6 (DMSO-d6). Multiplicities of proton resonance were designated as singlet (s), doublet (d), triplet (t), and multiplet (m). The microstructure of PI/mMMT nanocomposites were measured by scanning electron microscope (FE-SEM) (EVO18, Zeiss). In order to maintain the original appearance of PI/m-MMT nanocomposites, the samples were treated by liquid nitrogen to obtain their fracture surface. The spatial dispersion of m-MMT in polymer matrix was analyzed via TEM, transmission electron microscopy, FEI Tecnai F20S. Thermal gravimetric analysis (TGA) was performed on a Setaram TGA 92 Thermobalance, by heating the clay samples with 10°C/min heating rate from ambient temperature to 800°C at nitrogen atmosphere. The gas permeation performance of PI I/m-MMT nanocomposite membranes were carried out by using 99.999% pure CO2 O2and N2 gases at room temperature at a pressure of 2–10 bars. The desired gas was supplied through lumen side of the membranes bundle. For permeation test, 3 fibres are taken as a pile and one end of the pile was sealed using epoxy glue. After 3 h, another coat of epoxy was performed at pile, followed by 24 h of drying time. Subsequently, after 24 h, pile of fibres are fixed inside the module and glued again so that no gas can pass through except through the fibres shell side. The permeation cell was vacuumed prior to gas permeation test. Membrane permeance and selectivity for CO2, O2 and N2 was calculated by using equations (1) and (2), respectively.23,24
Pi/l is the gas permeance in GPU [1GPU = 1 × 10−6 cm3 (STP)/cm2.s.cmHg]. Subscript, i, represents the penetrating gas, i.e., CO2 or O2 and N2. A, is the effective surface area of the membrane, ΔP is the pressure difference across membrane in cmHg and T (K) is the temperature at which permeation was carried out. αij, represents ideal selectivity of gas i to j.
Experimental
Monomer synthesis
The diimide compound 2 obtained from double N-arylation reactions of 4-methoxyaniline with 5-bromo-2-methylisoindoline-1, 3-dione was carried out in the presence of palladium (II) acetate, rac-2,2′-bis(diphenylphosphino) −1,1′-binaphthyl (rac-BINAP), and cesium carbonate in toluene. The resulting diimide compound (1) was then hydrolyzed with aqueous potassium hydroxide, giving the corresponding tetracarboxylic acid, which in turn was converted to the new dianhydride monomer 2 by the chemical cyclodehydration with acetic anhydride (Scheme 1). Elemental analysis, FT-IR, NMR and 13C-NMR spectroscopic techniques were used to identify structures of the dianhydride monomer. The carbonyl groups of diimide compound exhibit two characteristic bands at around 1720 and 1645 cm−1 could be attributed to imide C = O asymmetric and symmetric stretching, respectively.
25
After hydrolyzed and cyclodehydrated to dianhydride monomer, the carbonyl groups shifted to around 1827 and 1717 cm−1. Figures 1 and 2 illustrates the 1H-NMR and 13C-NMR spectra of the dianhydride monomer 2 and these spectra agree well with the proposed molecular structure. Thus, the results of all the spectroscopic and elemental analyses suggest the successful preparation of the target dianhydride monomer 2. Synthesis rout of novel diimide monomer. 1H-NMR spectrum of diimide monomer. 13C-NMR spectrum of diimide monomer.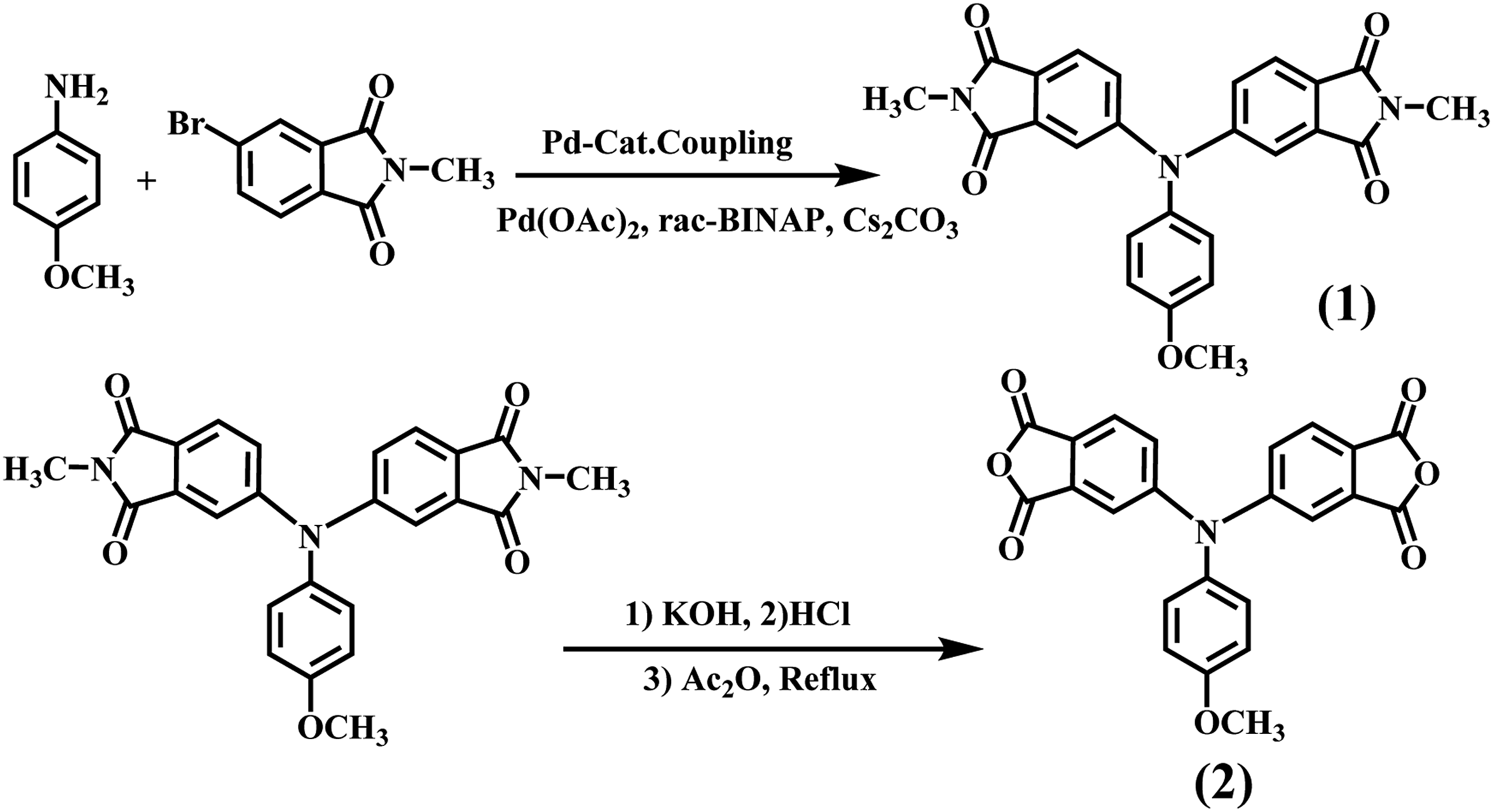
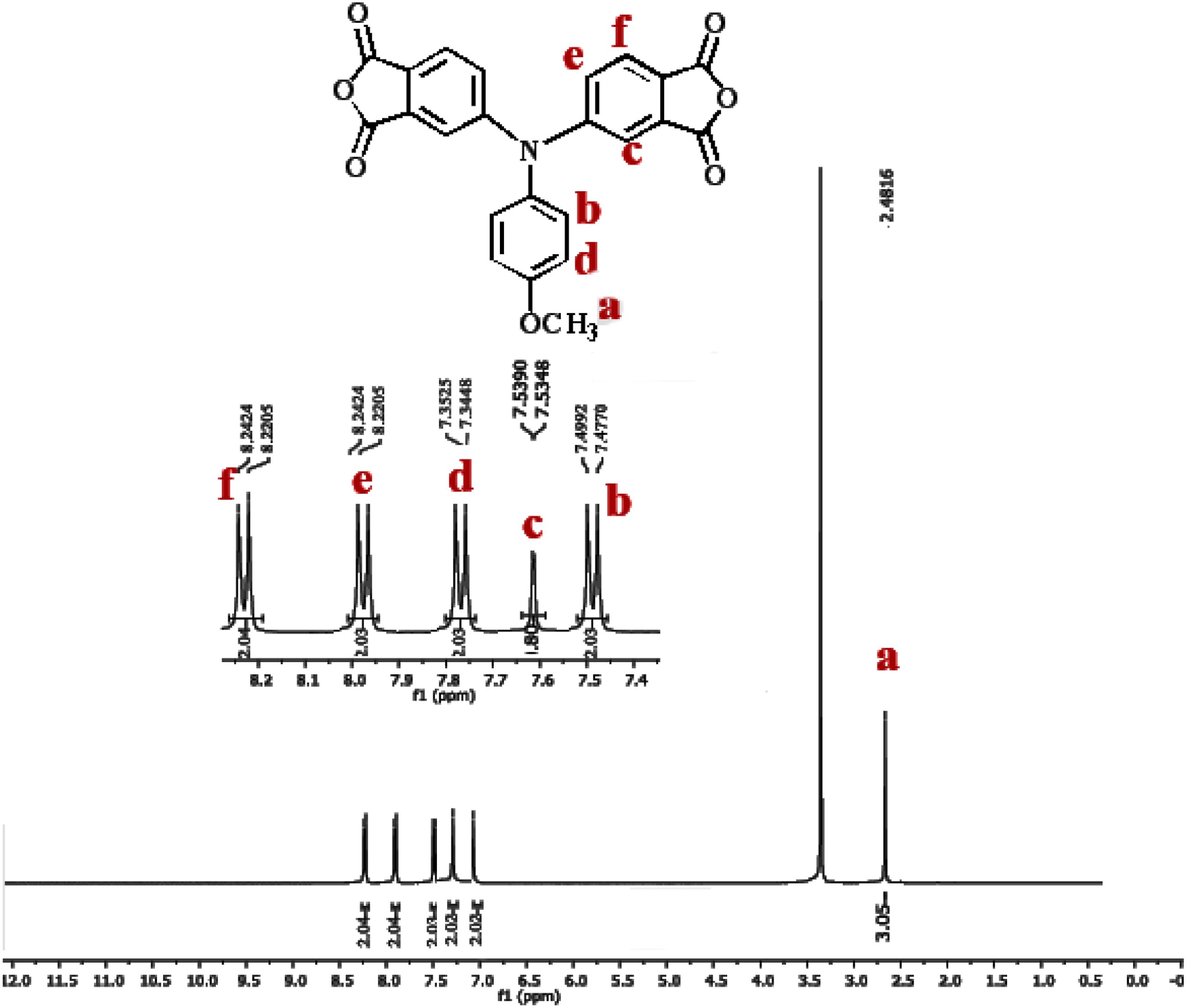
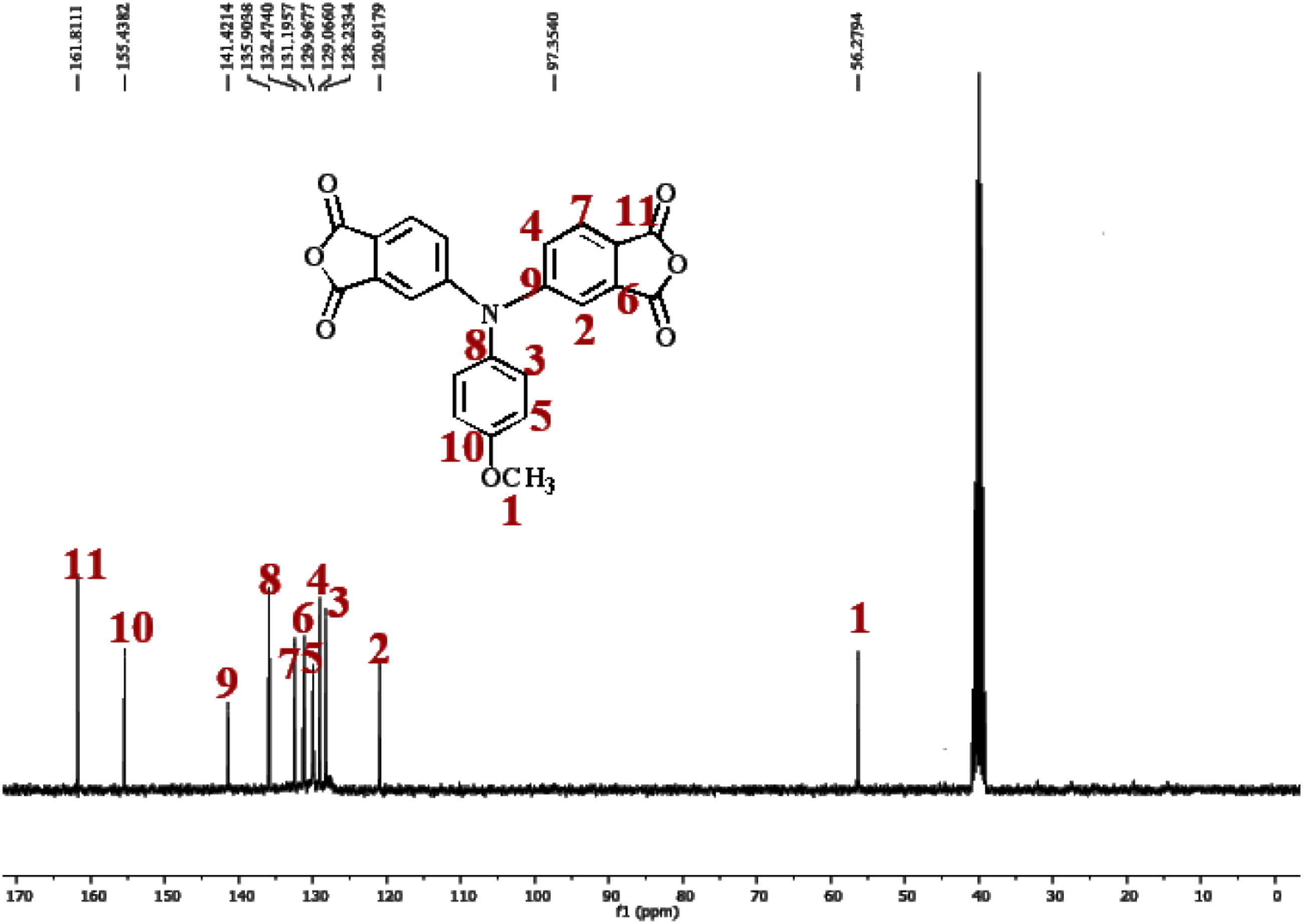
Polymer synthesis and preparation PI with amine end-capped
Novel PI was prepared by the one/pot, high/temperature solution polymerization of new dianhydride monomer with aromatic diamine (4,4′-(1,4-phenylenebis(oxy))bis(3-(trifluoromethyl)aniline) in m Synthesis rout of novel PI and PI with amine end-capped monomer. 1H-NMR spectrum of polymer (PI).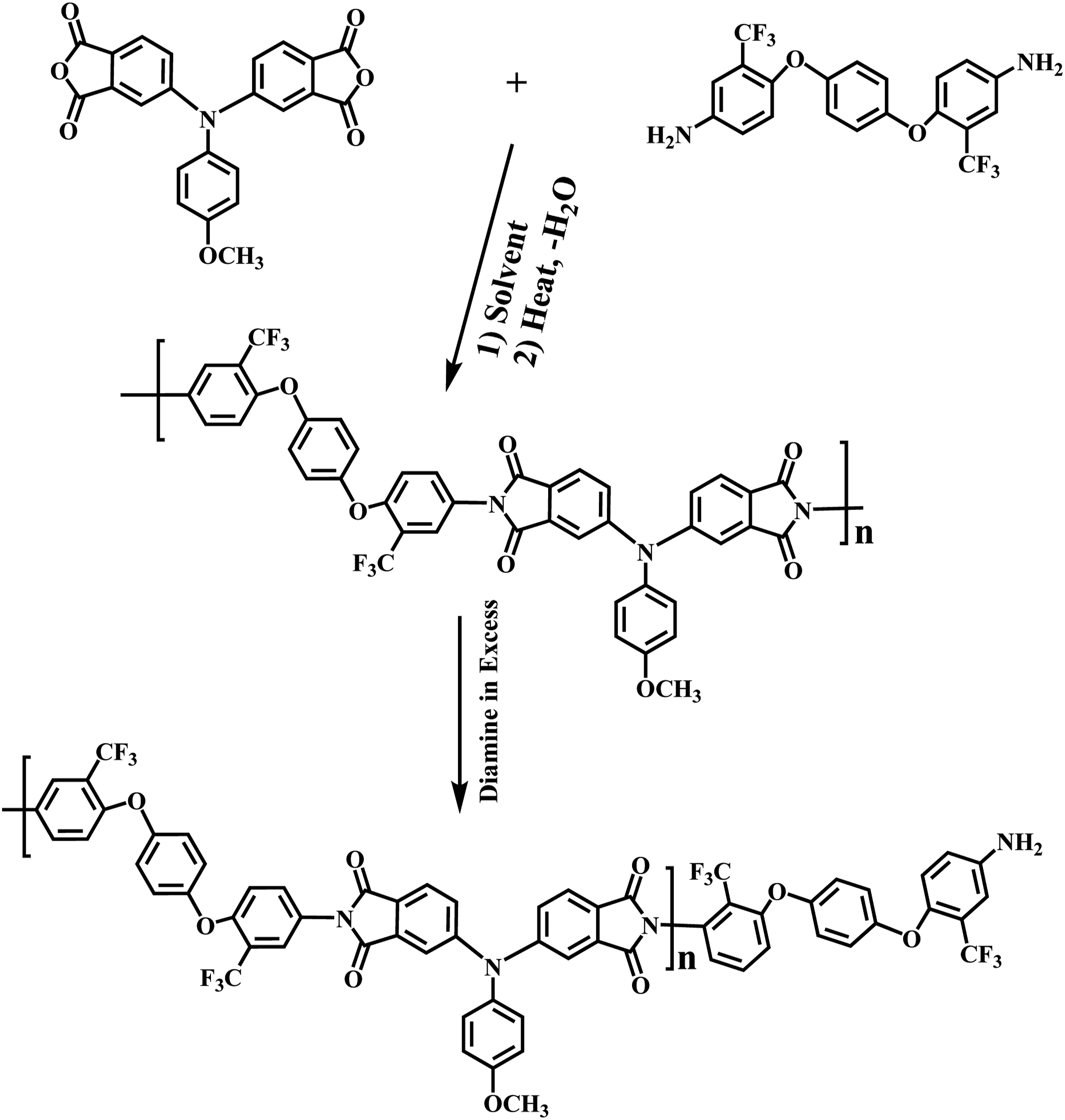
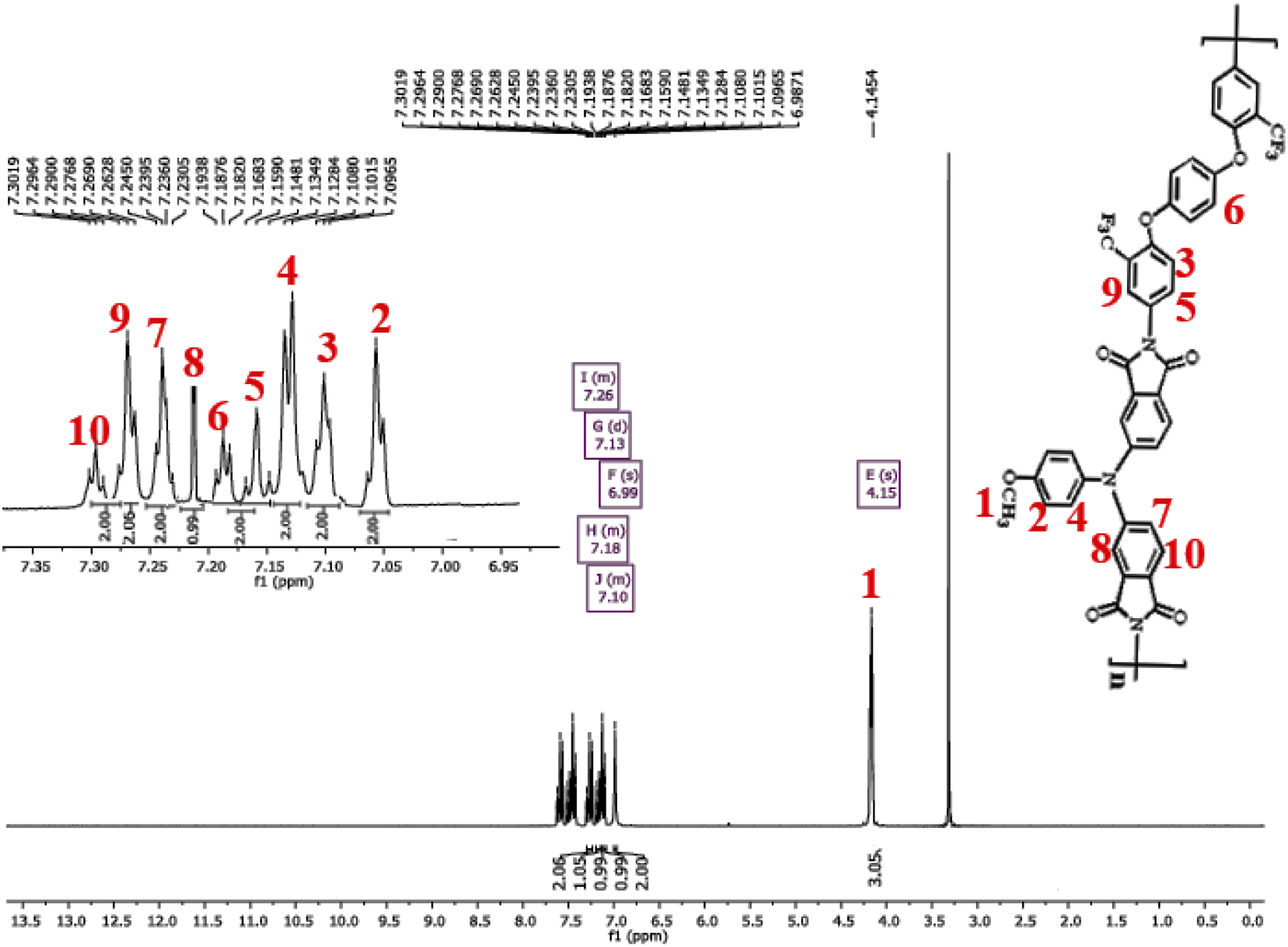
PI was also characterized by elemental analysis techniques, and the results are in good agreement with the calculated ones for the proposed structures. These results in sum confirmed the successful formation of the new PI.
Fabrication of organoclay and PI/Clay nanocomposites
The organoclay was prepared by a cation-exchange method, which is a displacement of the sodium cations of Na-clay with the quaternary ammonium ions of the surfactant. Typically, 10 g of Na-clay was mechanically stirred with 500 mL of deionized water at 25°C for 3 h to swell the clays. Then, 15 mL protonized diamine/HCl solution was added dropwise into the Na-MMT suspension. The mixed suspension was stirred at 65°C overnight to obtain the organoclay layers. The precipitates of organoclays were isolated by suction-filtration using Whatman filter paper placed in a 500 mL beaker again and washed with 300 mL of hot deionized water. This process was repeated four times to ensure the removal of excess ammonium salt of diamine. The final product obtained by filtration was dried under vacuum at 65°C for 6 h.
The PI/Clay nanocomposite membranes were prepared by mixing suitable amounts of the PI and clays in a flask for a particular concentration. The reaction mixture was agitated to high speed stirring at 80°C for 4 h and at 60°C for 20 h for uniform dispersion of organoclay platelets in the PI matrix. Various compositions (1, 3 and 5%) of organoclays were prepared by mixing various amounts of clays to the PI solution (Scheme 3). Thin PI/Clay films of uniform thickness were obtained by pouring the hybrid solutions into Petri dishes, followed by solvent evaporation at high temperature. These films were further dried at 85°C under reduced pressure to a constant mass. Fabrication of organoclay and PI/Clay nanocomposites.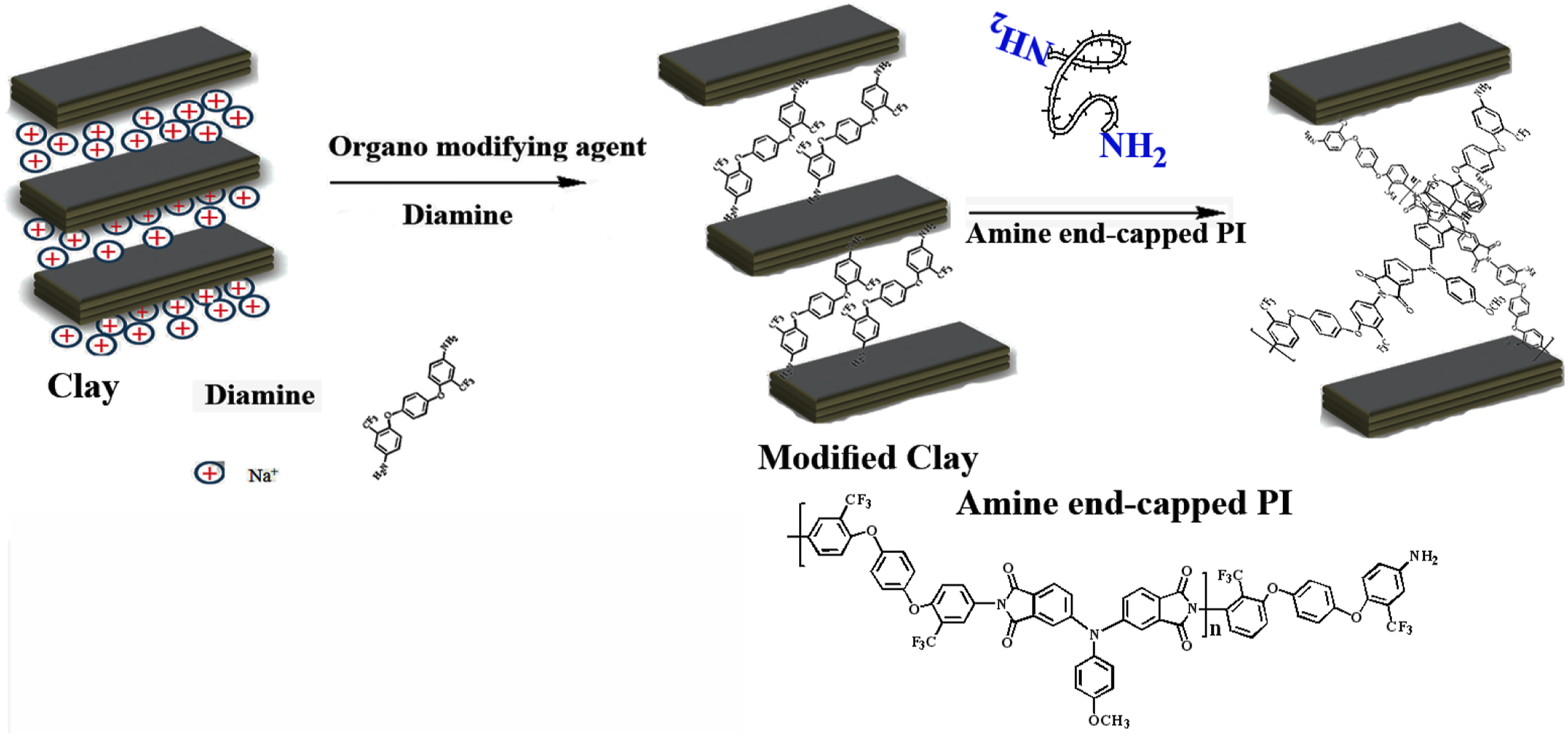
Results and discussion
FT-IR spectra of nanocomposite membranes
Figure 4 shows the FT-IR spectra of the (a) Na-Clay, (b) organoclay, (c) pure PI and (d) PI/Clay 1% materials. FT-IR spectra of Na-Clay and organoclay are shown in Figure 4(a) and (b). The FT-IR spectrum of Na-Clay showed the characteristic absorption bands of the Si–O, Al–O and Mg–O moieties at 1039, 513 and 462 cm−1, respectively. The incorporation of organic groups in organoclay was confirmed by the presence of peaks at 3050 cm−1 (ν C = C–H), 2850 cm−1 (νNH2+) and 1650 cm−1 (δC = C aromatic). In the FT-IR spectrum of PAA, the characteristic absorption bands of amides and carboxyl groups appeared at 3335 and 1665 cm−1, respectively, whereas they disappeared in the spectrum of the imide.
27
The appearance of new peaks at 1782 cm−1 (asymmetric C = O stretching), 1372 cm−1 (νC–N) and 1050 and 742 cm−1 (ring deformation) indicated the complete conversion of PAA to PI. In the nanocomposite sample, the presence of a peak at 1050 cm−1 corresponding to the Si–O stretch indicated the incorporation of organoclay in the PI matrix (Figure 4(d)). FT-IR spectra of the (a) Na-Clay, (b) organoclay, (c) pure PI and (d) PI/Clay 1% materials.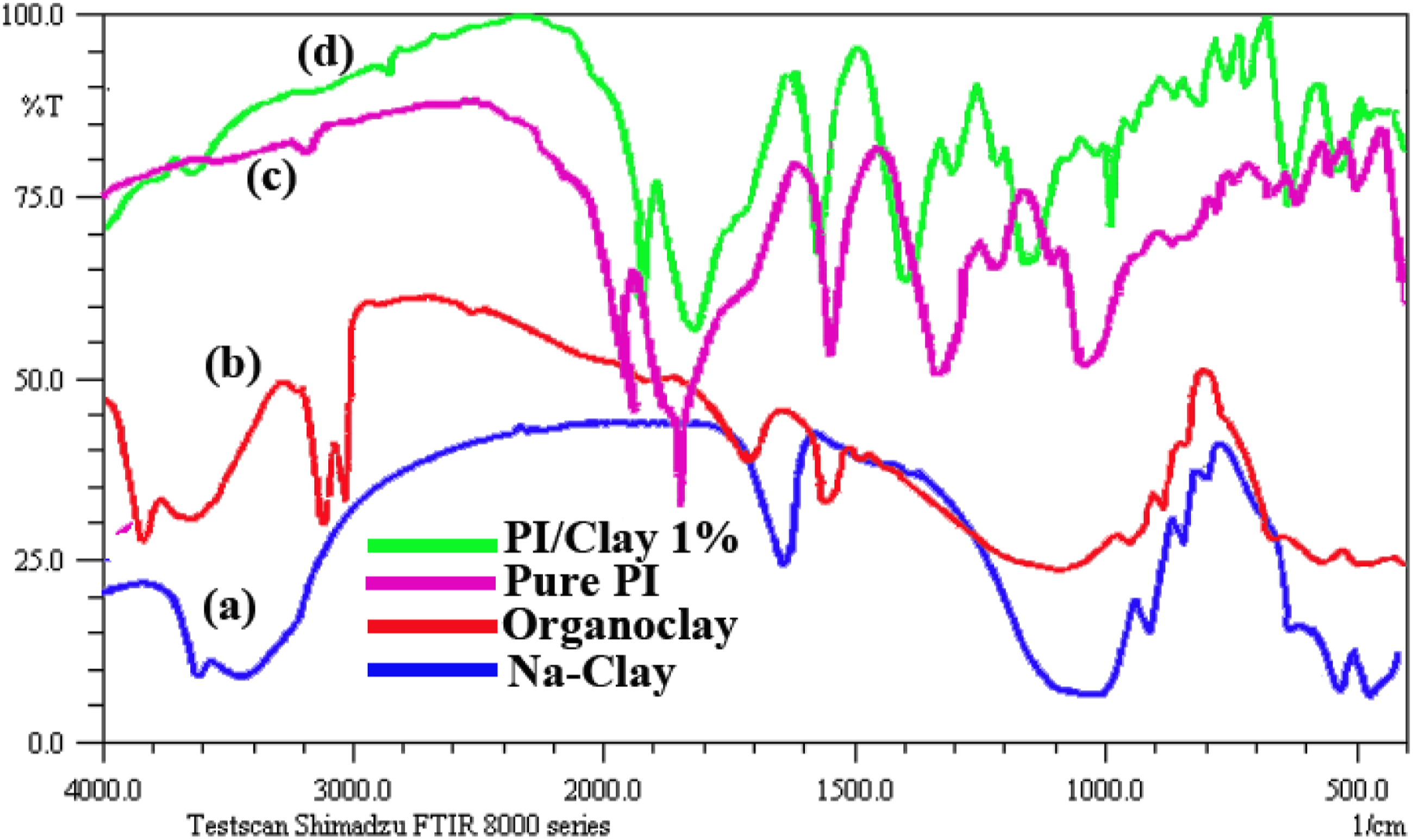
XRD patterns of nanocomposite membranes
Figures 5(a)–(f) shows the XRD patterns of clay samples and polymer nanocomposite samples, respectively. For Na-Clay, the diffraction pattern shows a distinct peak at 2θ = 4.68° corresponding to 18.22 A°, which is assigned to the 001 lattice spacing of montmorillonite. After modification with dodecyltrimethylammonium XRD patterns of the (a) Na-Clay, (b) organoclay, (c) pure PI and (d) PI/Clay 1%, 3% and 5%materials.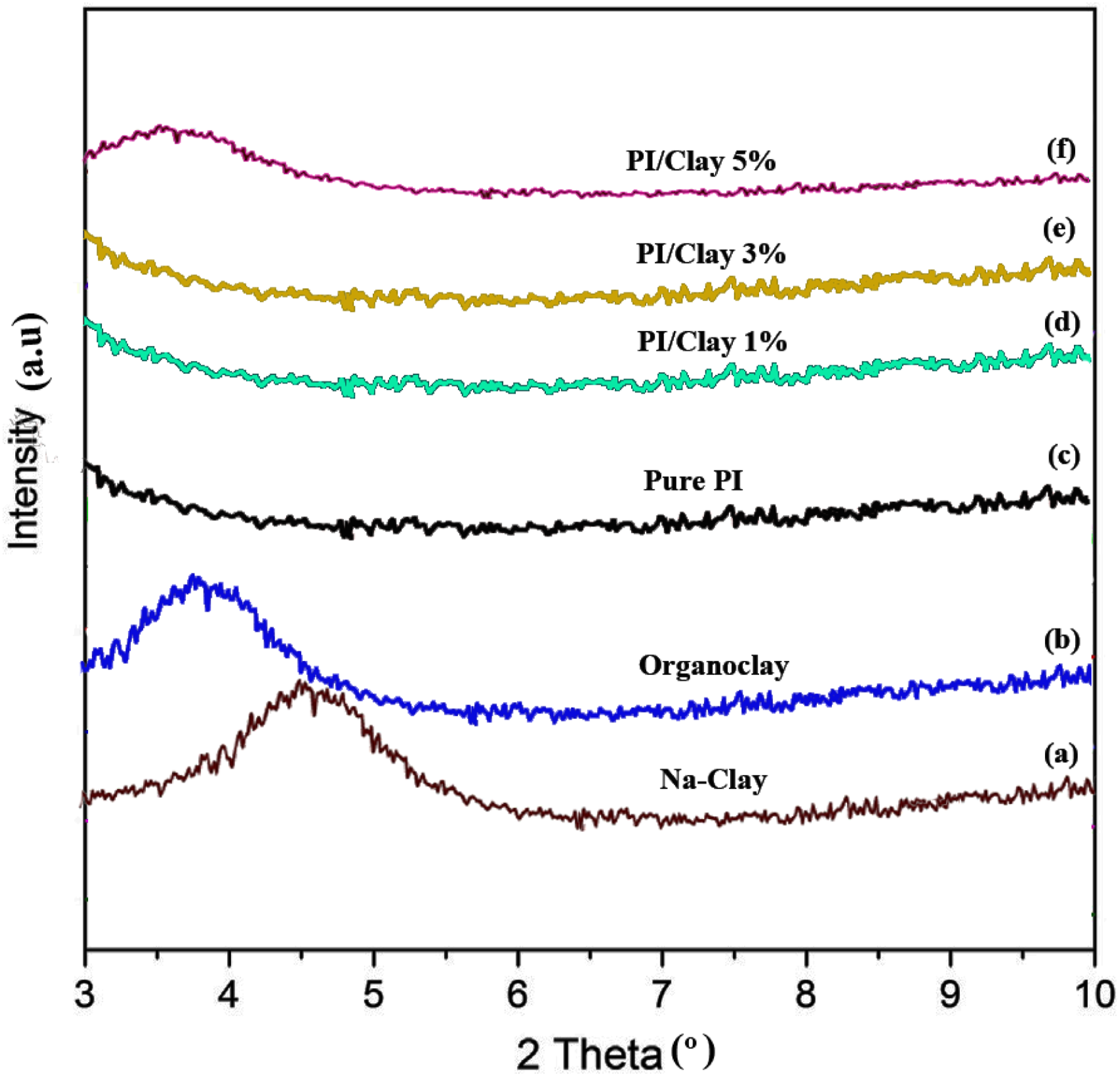
diamine, 2θ decreased to 3.83°, i.e. the d spacing increased to 21.25°A for organoclay. The greater spacing in the organoclay, when compared to Na-Clay, would help the silicate layers to disperse easily.
In nanocomposite samples, no distinct peak was observed at lower clay concentrations (up to 3%), whereas for 5% clay concentrations, a distinct peak at about 2θ = 3.98° (d = 14.4 A°) was observed. Usually, there are two types of nanocomposites depending upon the dispersion of clay particles. The first type is an intercalated polymer–clay nanocomposite, which consists of well-ordered multilayers of polymer and silicate layers a few nanometers thick and these structures would give rise to distinct peaks in XRD patterns as in the case of 5% nanocomposite sample. In these nanocomposite samples, the extent of polymer penetration was not sufficient to delaminate the ordered structures. The second type is an exfoliated polymer–clay nanocomposite, in which is there is a loss of ordered structures due to the extensive penetration of polymer into the silicate layers. Such types would not produce distinct peaks in the XRD patterns. As there was no coherent XRD signal at lower clay concentration (up to 3%), one can say that these nanocomposite samples were exfoliated or delaminated. 28
Morphology of nanocomposite membranes
Figures 6(a) and (b) presents the TEM images of PI/Clay 3% and PI/Clay 5% nanocomposites. In these images, part of clay nanoplatelets denoted as the dark lump have been intercalated and some individual nanoplatelets disperse homogeneously in PI matrix with a slice size lower than 0.5 μm. A minor extent of clay reveals a number of agglomerates in PI/Clay 3%; however, the dispersion of individual silicate layers in the polymer matrix for PI/Clay 5% is less homogeneous than PI/Clay 3%. In Figure 6(c), the magnified sample edge of PI/Clay 5% is given to present the morphology of the nanocomposites more clearly. From Figure 6(c), we confirm that the PI/Clay nanocomposites are prepared successfully and the nanoplatelets are embedding in the PI matrix as we think. It also can be seen the distribution of the clays in the PI matrix which depends on the amount of the clay which is consist with the result of XRD.
29
Even so, both PI/Clay 1% and PI/Clay 5% are considered as a partial intercalated system. The TEM micrographs of (a) PI/Clay 3%, (b) PI/Clay 5% and (c) The sample edge of PI/Clay 5%.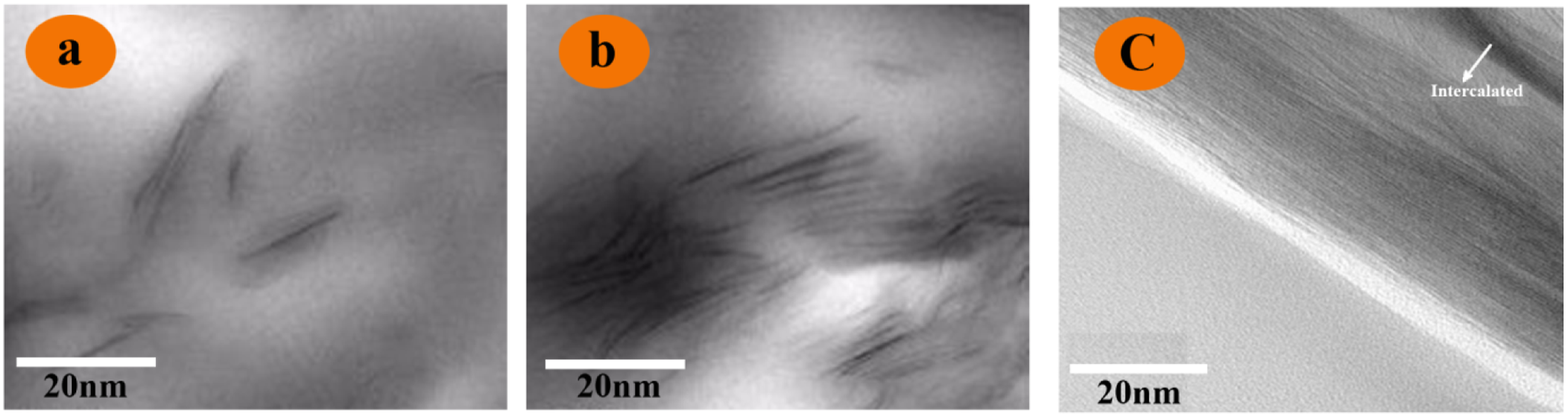
The FE-SEM micrograph of pure clay is shown in Figure 7(a), while neat PI and the nanocomposites FE-SEM images are displayed in Figure 7(b)–(d). The image of clay reveals an apparent layered structure and the original stack size of lamellar structure is close to 3–5 μm. From the cross-section images of the PI/Clay composites, it can be seen that the surface morphology changes according to the clay loading. For brevity, we just list some representative micrographs. Neat PI shows a smooth surface, and the evenly dispersed white dots in the PI matrix are residual Zinc catalysts which have been verified by EDAX (Energy-dispersive X-ray spectroscopy). Moreover, PI/Clay 3% reveals a homogenous dispersion of clay in the polymer matrix, though the surface becomes a little rough. The image of PI/Clay 5% displays a rougher surface and presents some microscale fillers and gaps due to the agglomeration of individual silicate layers. Both the micrographs and XRD results indicate that the organic modified clays (organoclay) can homogeneously disperse in the polymer matrix at a small amount; however, agglomeration may form with higher loadings of clay added. The FE-SEM micrographs of (a) Pure Clay, (b) Pure PI (c) PI/Clay 3% (d) PI/Clay 5%.
TGA Analysis of PI/Clay nanocomposite membranes
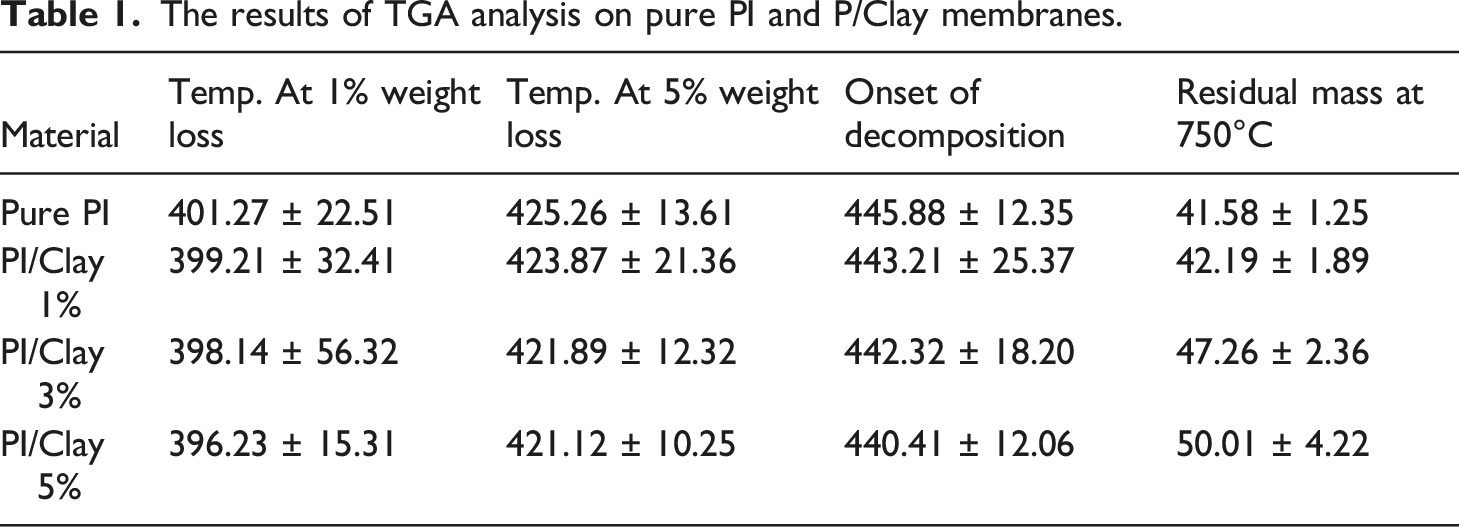
In order to investigate the thermal stability of the PI/Clay nanocomposites, thermogravimetric analysis (TGA) was performed. The TGA was performed under nitrogen atmospheres. The TGA weight loss curves and first derivative TGA curves for reference PI and the nanocomposite samples under nitrogen atmosphere (pyrolytic environment) are presented in Figure 8. In nitrogen atmosphere the pure PI and the PI/Clay nanocomposites exhibited only one step decomposition. The main degradation process during TGA analysis under nitrogen atmosphere is due to the combined degradation processes—polymer chain degradation through end group initiated mechanism and the thermal degradation of the products formed during polymer chain degradation. Under nitrogen environment, the thermal decomposition of polymer is leading to thermally stable cross-lined carbonaceous species which are not undergoing any further decomposition due to the presence of inert nitrogen atmosphere and hence the large amounts of reside at the end of TGA analysis. The results of TGA analysis such as temperature at 1% weight loss, temperature at 5% weight loss, onset of decomposition, and the amount of residue at end of TGA analysis (at 750°C) are summarized in the Table 1. It is clear from the results that the temperatures at 1% and 5% weight loss as well as the onset of decomposition is lower for the nanocomposites compared to pure PI. This observation could be due to the degradation of diamine modifier which is present in Na-Clay. The diamine present in the Na-Clay is known to undergo Hoffman degradation around 190°C.30,31 The amount of residue left over on TGA analysis (at 750°C) under nitrogen atmosphere for PI/Clay nanocomposites is higher than that of the pure PI. (a) TGA weight loss curves and (b) first derivative TGA curves for PI/clay nanocomposites during TGA analysis under nitrogen atmosphere. The results of TGA analysis on pure PI and P/Clay membranes.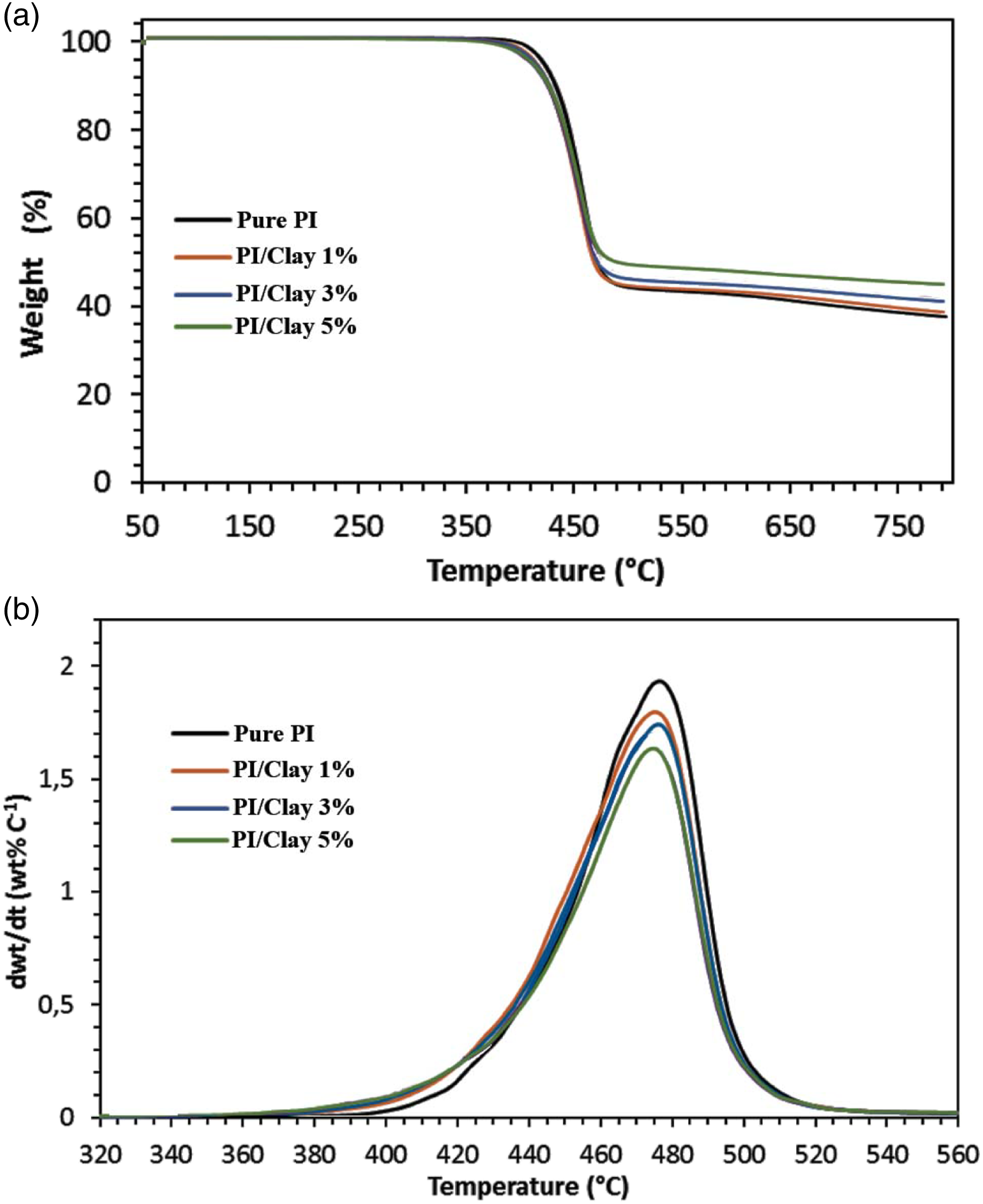
DSC Analysis of PI/Clay nanocomposite membranes
Results of DSC analysis of PI and PI/Clay nanocomposites Tg, Tm1, Tm2 and Tc.
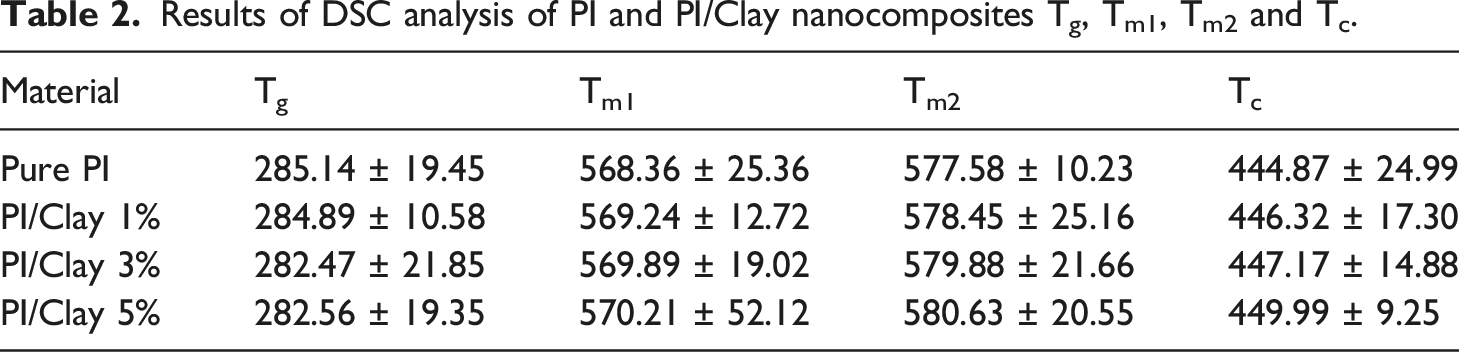
A double melting peak appeared for both pure PI and the PI/Clay nanocomposites. This behavior could be due to the presence of a dual lamella thickness distribution which is formed during crystallization of PI. The nanocomposite samples also presented double melting peaks, indicating that added clay has no influence on the crystal morphology in the PI/Clay nanocomposites. As seen from the thermograms, the position of the first melting (Tm1) is unaltered and the second melting peak temperature (Tm2) is slightly affected by presence of clay filler. The low temperature first melting peak is observed at about 568–570°C while the high temperature main peak position varies between 577–580°C depending on the clay content. In addition, with increase in clay loading, the first melting peak shoulder is less pronounced, being obvious for the nanocomposite containing 5 wt% of clay. This is probably because clay at higher loadings starts to influence crystallization behaviour and morphology. The DSC crystallization endotherms of pure PI and PI/Clay nanocomposites are presented in Table 2 and Figure 9. A slight shift in the crystallization peak position towards the higher temperature is observed for the nanocomposites containing clay. This means that the nanocomposite samples crystallize at higher temperature compared to the pure PI, as also seen from the corresponding Tc values in Table 2. The shift in crystallization peak towards higher temperature is due to the heterogenous nucleating effect of dispersed clay mineral in PI matrix. DSC thermographs of pure PI and PI/Clay nanocomposites.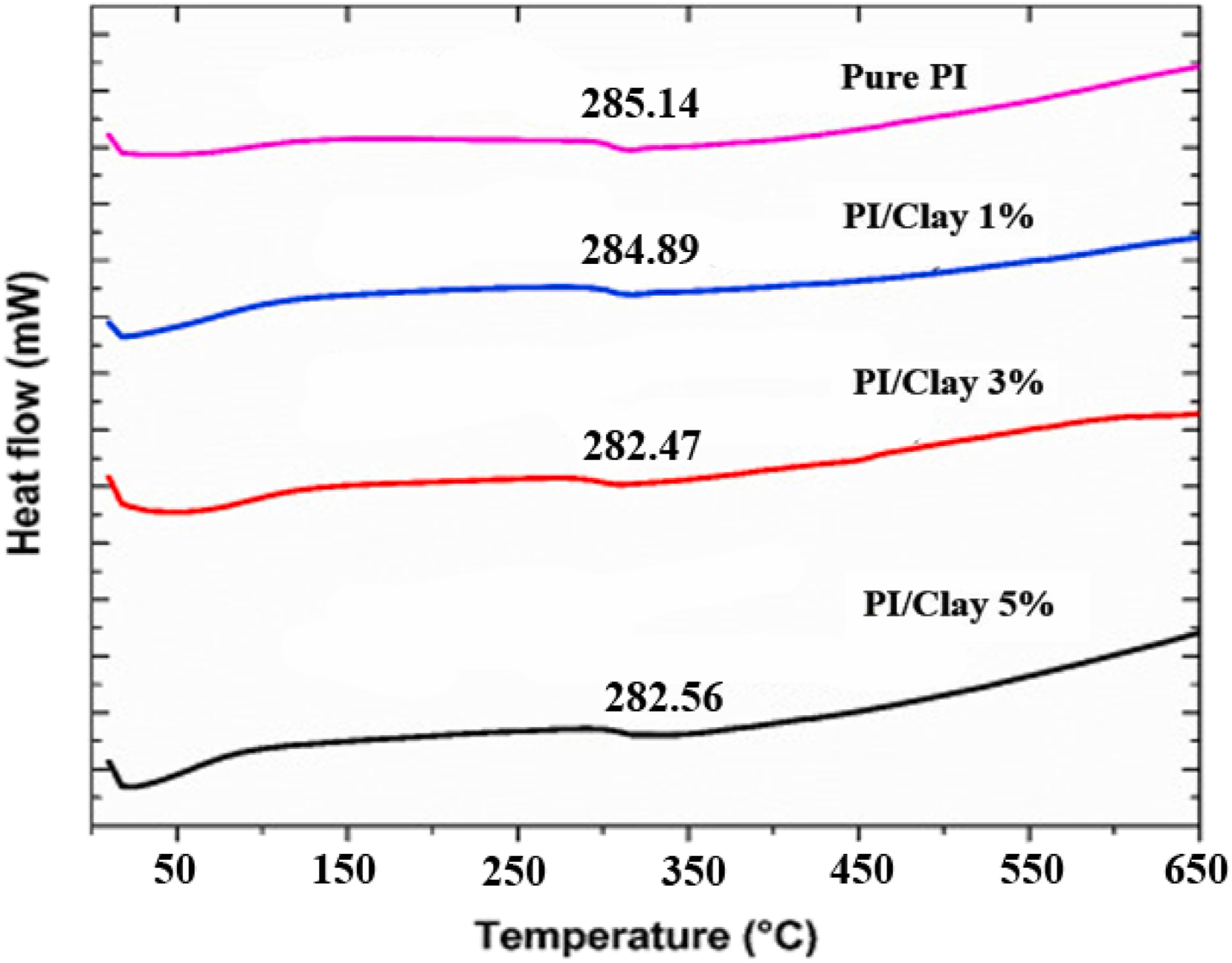
Mechanical properties of PI/Clay nanocomposite membranes
The tensile properties of pure PI and PI/Clay nanocomposites with various clay contents are reported in Figure 10. Compared to pure PI, the tensile strength, elongation at break and modulus of PI/Clay nanocomposites dramatically increased with increasing clay content up to 3 wt%. This is due to the exfoliated structure of silicate layer providing the optimum reinforcement. However, the tensile properties decreased when a high content (5 wt%) of clay platelets was added. This suggests that the clay partially remains the intercalated and stacked structure, causing the weakly reinforcing effect, the non-idealized nanoclay dispersion, and the presence of microvoids as shown in FE-SEM micrographs. Tensile strength, Elongation at break and Modulus of pure PI and PI/Clay nanocomposites with various inorganic nanoclay contents.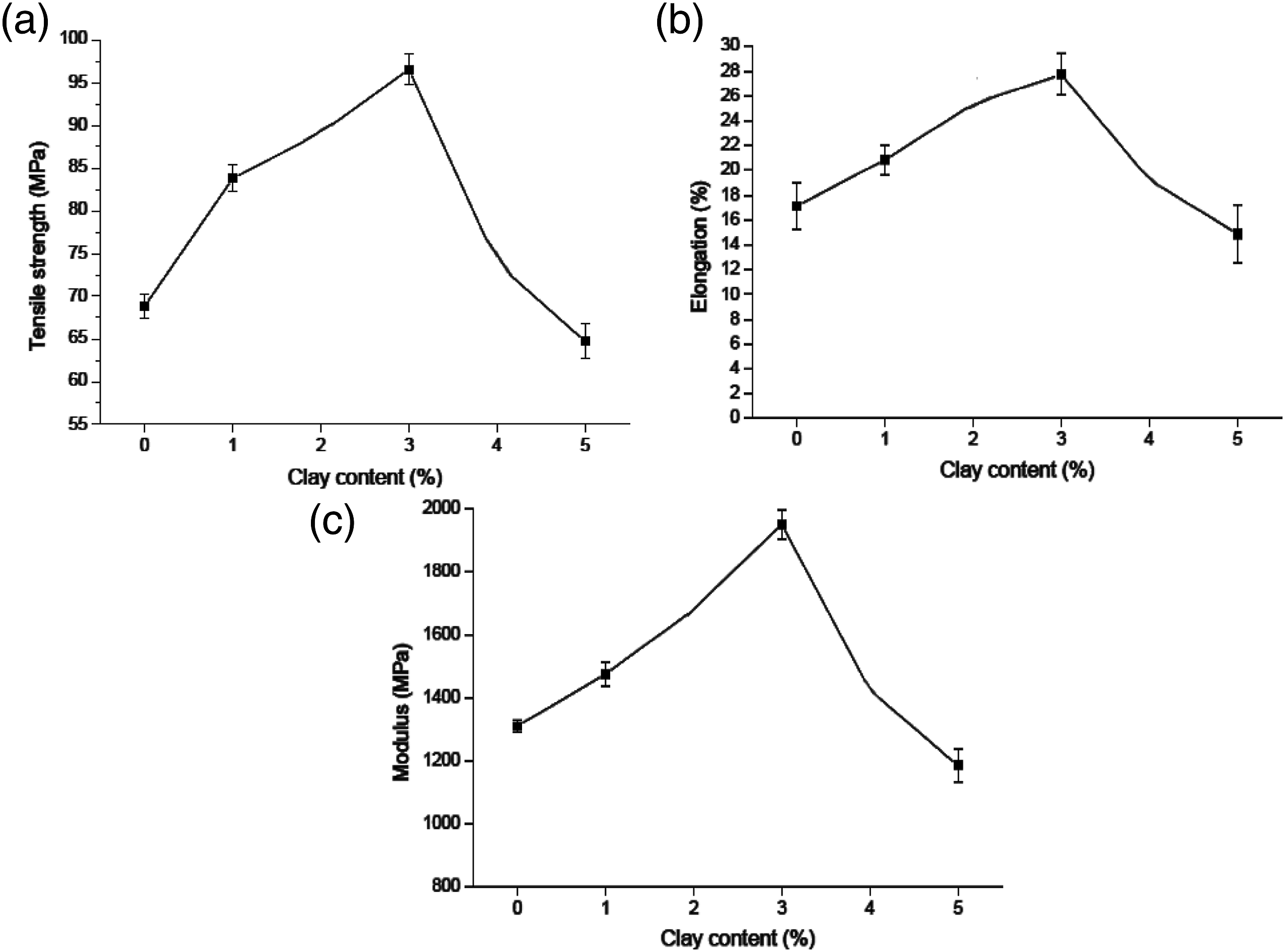
Tensile strength of the PI/Clay nanocomposites was highly dependent upon the clay platelets concentrations and was improved with increased clay concentration. This is due to the high strength of the clay platelet. Initially, the strength was improved by 12% for only 1 wt% clay incorporation. At higher concentrations, the rate of modulus increase became less, e.g., there is only 15% increase of the strength for 3 wt% clay by sonication, which can be attributed to the presence of unexfoliated aggregates in the structure under higher clay concentrations. The strength was also related to the method of clay dispersion; nanocomposites by sonication had slightly higher strength than those by shear mixing. Better dispersions by sonication increased the interfacial contact area between the clay particles and PI matrix, thus providing a better coupling and a more efficient restriction of epoxy chain mobility under load. 32
Tensile modulus was also related to the clay dispersion. Sonication dispersed PI/Clay nanocomposites lead to improved tensile modulus, and an increase of 25% in tensile modulus was observed for only 1 wt% clay platelets content. The nanocomposite modulus at 5 wt% clay platelets concentration lead to decrease and it is still near that of neat PI. Overall, exfoliated structures lead to improved modulus and strength, while the intercalated structures lead to decreased tensile strength and modulus. With sonication, exfoliation and intercalation structures probably coexisted at higher clay concentration with intercalated flocs as defects thus resulting in reduced mechanical strength. The interfacial interaction between clay and matrix is thus reduced by poorer dispersion at high clay concentration and crack fracture could initiate in clay galleries. Decreased tensile and modulus strength has also been attributed to the presence of aggregation of clay particles which is due to the inefficient dispersion or clay concentrations above the percolation threshold. Aggregation not only reduces the interfacial interactions between clay and polymer but also generate macrocracks. Large aggregates and intercalated structures co-existed in shear mixed composites, so there was even less interfacial interaction between the clay and the PI matrix compared to the sonication dispersed composites, as will be observed in FE-SEM and TEM images. 33
Gas permeability studies of PI/Clay nanocomposite membranes
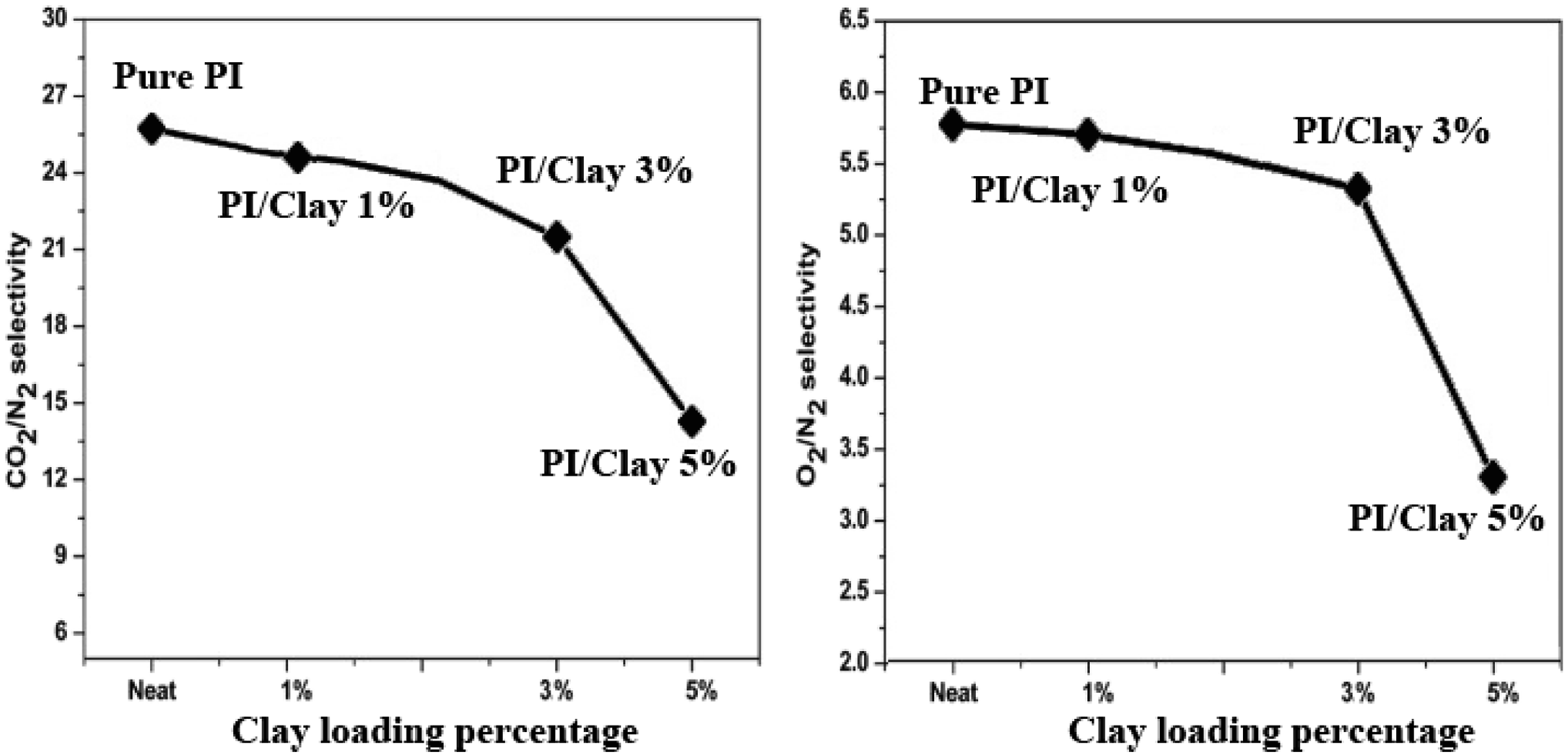
Gas permeation of neat PI and PI/Clay nanocomposites were assessed in terms of gas permeability and selectivity for gas pairs (O2/N2 and CO2/N2). The permeance and the selectivity data were depicted in Figure 11 and Figure 12, respectively. The permeation of CO2, N2, and O2 gases was increased with the increase in the loading percentage of clay. Besides, the selectivity of O2/N2 and CO2/N2 gas pairs were decreased with an increase in clay loading percentage. The relative increase in permeance for CO2 from pure PI to PI/Clay nanocomposite (3 wt%), was about 245% from 17.98 to 61.25 GPU, while the CO2/N2 selectivity was decreased by 17.21% from 26.01 to 21.05. The corresponding increase in permeance for O2 was 240% from 4.91 to 16.89 GPU with a drop of 7% in O2/N2 selectivity from 5.76 to 5.21. Permeance of pure PI and PI/Clay nanocomposites. Selectivity of pure PI and PI/Clay nanocomposites for CO2/N2, and O2/N2 gas separation.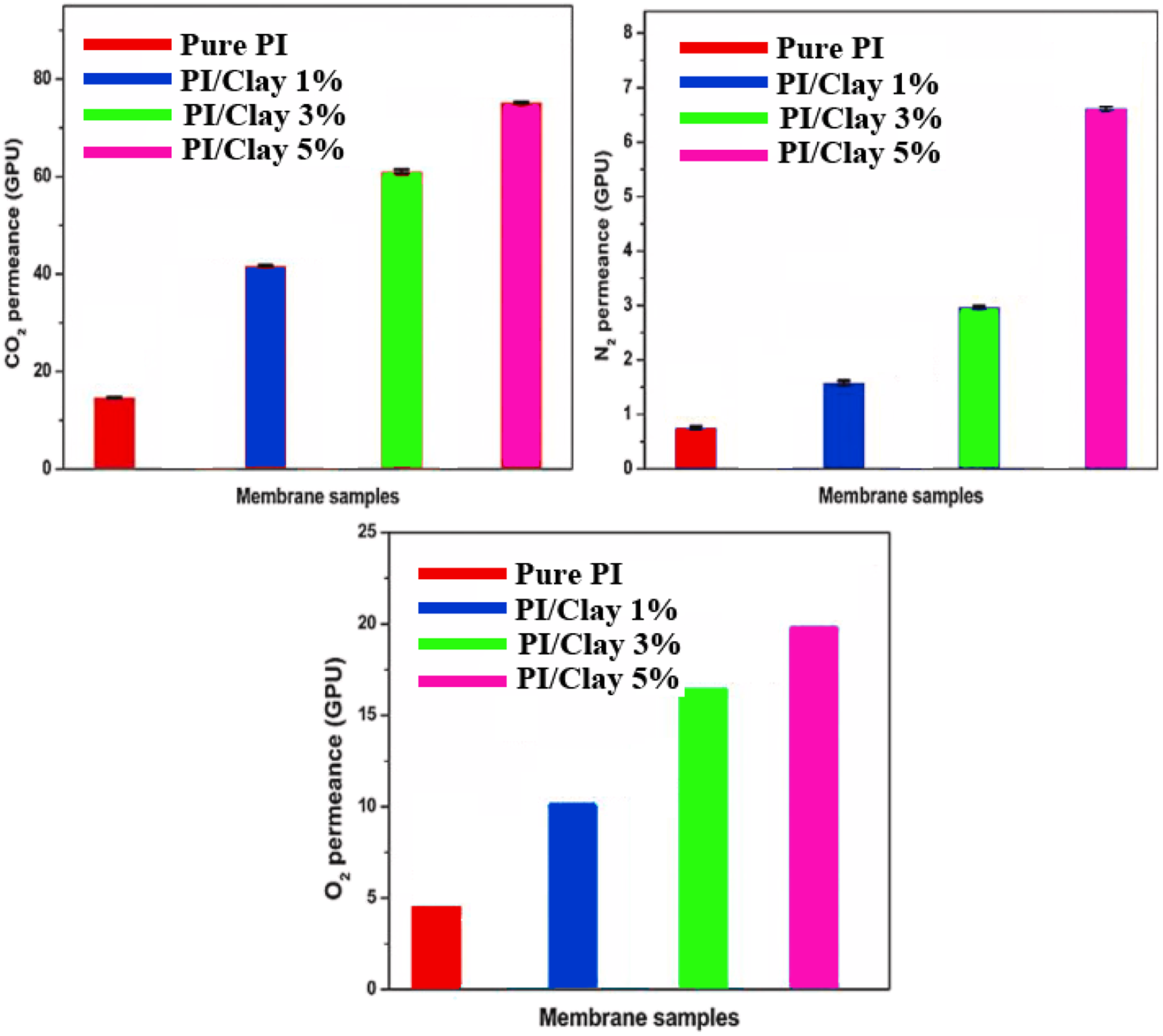
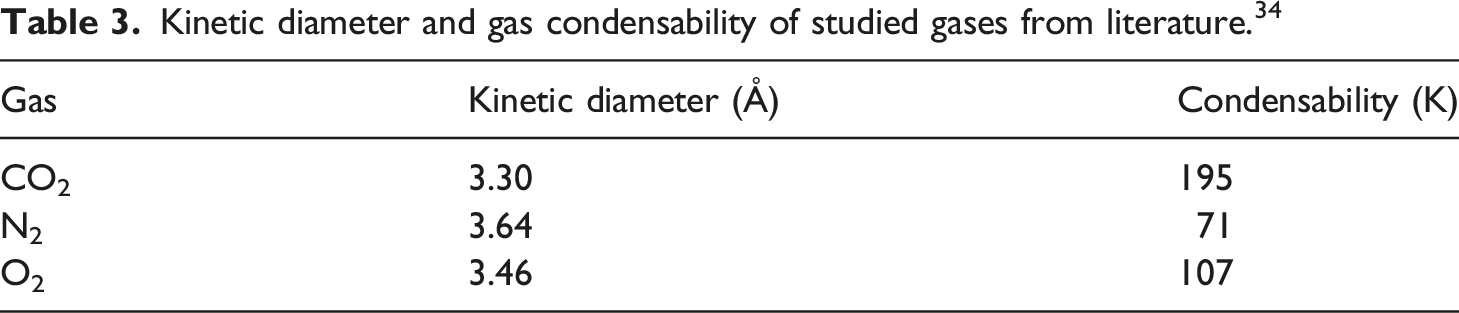
Kinetic diameter and gas condensability of studied gases from literature. 34
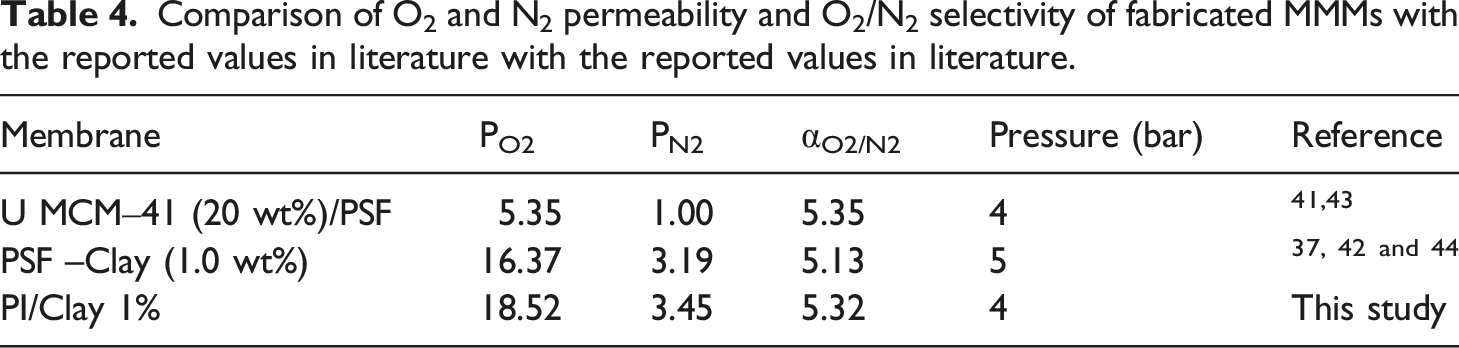
Comparison of fabricated nanocomposites with literature
Comparison of O2 and N2 permeability and O2/N2 selectivity of fabricated MMMs with the reported values in literature with the reported values in literature.
Conclusion
Polyimide containing bis three flours groups designated was synthesized. Then PI/Clay nanocomposites with different concentration of clay platelets were fabricated. The gas permeation, thermal, mechanical and morphological properties were investigated in the nanocomposites. The results of XRD, TEM, and FE-SEM indicated that clay could well disperse in the PI matrix and were intercalated by diamine and PI macromolecules. The nanocomposites a slightly higher crystallisation temperature compared to pure PI. The increased crystallization temperature of nanocomposites is due to a heterogenous nucleating effect of clay. The thermal stability and glass transition temperature of nanocomposite at lower clay loadings are similar to PI but gradually decrease with increase in clay content. The tensile properties of PI/Clay nanocomposites showed the highest values at the optimum clay content of 3 wt%. The experimental observations indicate a significant increase in membrane permeability at a low loading of clay with an insignificant drop in permselectivity. The most efficient membrane was found to be at 3 wt % loading of clay. Organoclays are a relatively novel filler medium to be used in nanocomposites, and exhibit favorable compatible properties for gas separation. The study shows that there is a scope and potential application for the implementation of montmorillonite silicates for gas separation.
Footnotes
Acknowledgements
H.Ahmadizadegan. acknowledges financial support from Iran Nanotechnology Initiative Council (INIC).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Correction (July 2023):
This article has been updated with branch name in the affiliations since its original publication.


