Abstract
Montmorillonite-rich local bentonite was modified with four different quaternary alkyl salts: hexadecyltrimethylammonium bromide ((HMA)(Br)), tetra(kis)decylammonium bromide ((TKA)(Br)), tetrabutylammonium tetrafluoroborate ((TBA)(BF4)), and tetrabutylphosphonium tetrafluoroborate ((TBP)(BF4)) to produce organoclays. The organoclays produced were characterized by Fourier transform infrared spectroscopy, thermogravimetric analysis (TGA), and x-ray diffraction (XRD). The XRD results indicate that the d-spacing of the organoclays increased as a consequence of the exchange of Na+ ions in the clay galleries with the cation of the surfactants with long alkyl tails. The d-spacing of the bentonite increased from 1.2 nm to 1.78 nm, 2.56 nm, 1.48 nm, and 1.64 nm after modification with HMA, TKA, TBA, and TBP cations, respectively. TGA analysis of the organoclays showed that the decomposition temperatures of the organoclays were higher than the melt processing temperature of polypropylene (PP), permitting the use of these organoclays in melt processing of PP. Ternary composites of PP/PP-grafted maleic anhydride/organoclays were prepared using a twin screw extruder followed by injection molding for characterization. Transmission electron microscopy analysis of the composites showed intercalated structures as well as microcomposite formations. Mechanical properties of pure PP were improved through ternary composite formation.
Keywords
Introduction
Production of organoclays from montmorillonite (MMT) has attracted much attention owing to wide application areas of these materials. MMT is the most widely used smectite clay mineral in the polymer-layered silicate nanocomposite (PLSN) production 1 –3 due to nanoscale enhancement in certain properties of polymers with the addition of small amounts of filler (2–5 wt%) compared to other conventional fillers. Several studies were carried out in the literature focusing on the effects of organoclays on mechanical, barrier, rheological, and thermal properties of polymer nanocomposites. 4 –10
MMT belongs to 2:1 type (two-dimensional layers with a thickness of 0.96 nm) smectite clay mineral family with one octahedral alumina sheet inserted between the two tetrahedral silica sheets. Stacking of these sheets leads to van der Waals gaps or galleries. As a result of the isomorphous substitution within the layers (e.g. tetrahedral Si4+ replaced by Al3+ or octahedral Al3+ replaced by Mg2+), a charge deficiency is generated and this deficiency is compensated by exchangeable cations (e.g. Na+, Ca2+, and K+). 11,12
Bentonite may contain mostly smectite clay minerals such as MMT, beidellite, nontronite, saponite, and other nonclay impurities such as quartz, feldspar, and clinoptilolite depending on the geological deposit. 13,14 Onal et al. 13 purified Resadiye bentonite from Turkey by a sedimentation method and found that this method was successful in isolating Na-smectite similar to Na+-MMT. Separation of clay and nonclay parts from natural bentonite to produce pure MMT has great importance in the polymer-based nanocomposite production, since the degree of purity of MMT affects the delamination of clay layers in the polymer matrix where nonpurities and nonexchangeable cations apart from Na+ may act as trapping points in the matrix. 15
Prior to nanocomposite production, hydrophilic clay layer surfaces should be made lipophilic in order to be used in mostly hydrophobic polymers such as polypropylene (PP), polyethylene, and polystyrene. With this aim, generally, surfactants with long tails of alkyl ammonium/phosphonium ions are intercalated between the clay galleries via cation exchange reactions in which the cations of MMT (e.g. Na+, Ca+2, and K+) are exchanged with the cations of organic surfactants. 11 This process increases the basal spacing of the clay layers depending on the length of the alkyl tail of the surfactant. Although organophilization reaction is achieved, the organoclay layer surfaces are still incompatible with most polar polymers, therefore a third component should be used as a compatibilizer in the PLSN to achieve a high degree of delamination of organoclay layers. These compatibilizers mainly have reactive polar groups on their backbone, thus they can have interactions and bonding with the organoclay layer surfaces. 2 Production of organoclays has attracted many researchers. Tiwari et al. 16 synthesized novel organoclays with three different amines and observed increase in the basal spacing of the clay, and the thermal analysis showed that these organoclays can be used in PLSN. Kim et al. 17 used different alkylimidazolium salts in the modification of MMT and characterized the resulting organoclays by Fourier transform infrared spectroscopy (FTIR), thermogravimetric analysis (TGA), and x-ray diffraction (XRD). These organoclays were used in PP-clay composite production. Awad et al. 11 synthesized a series of alkylimidazolium-modified organoclays and studied the thermal degradation process of the organoclays in detail using TGA and thermal desorption mass spectroscopy. Lee and Lee 18 analyzed the thermal properties of MMT that was modified with a series of alkyl amine and ammonium salts and showed that the thermal stability of organoclay depends on the nature and arrangement of the surfactant between the MMT layers. Patel et al. 19 purified Indian bentonite by sedimentation and then modified the product with seven different quaternary phosphonium (QP) salts followed by characterization with XRD, FTIR, and TGA. The phenyl group-substituted phosphonium salt was found to have highest thermal stability. Calderon et al. 20 and Xie et al. 21 studied the thermal stability of organoclays modified with quaternary ammonium (QA) and QP and observed that phosphonium ions were more thermally stable than the ammonium ions.
The aim of the current study is to investigate the modification of Resadiye bentonite for potential use of it as a reinforcement in polymer–clay nanocomposite (PCN) production. An extensive and systematic evaluation of the surfactants was performed. Some of the surfactants have not been used before. The effects of purity of MMT, cation exchange capacity (CEC) of the clay, surfactant concentration, surfactant structure such as the central atom, and the length of the surfactant tail were investigated. These organoclays were later used in the production of PP-based ternary composites.
Experimental
Materials
PP MH-418 with a reported melt flow index (MFI) of 4–6 g/10 min was obtained from Petkim Petrokimya Holding A.S., Izmir, Turkey. The compatibilizer PP-grafted maleic anhydride (MAPP) with a brand name of Bondyram 1001 was purchased from Polyram, Ram-On, Israel. It has a reported MFI (ASTM D-1238, 190°C/2.16 kg) of 100 g/10 min and a density of 0.9 g/cm3. Raw bentonite (RB) clay was kindly provided by Karakaya Bentonit A.S., Ankara, Turkey, in yellow powder form. The source of the mineral was Resadiye, Turkey. Its chemical analysis determined by Inductively Coupled Plasma Optical Emission Spectrometer is given in Table 1, showing high contents of silicon dioxide and magnesium oxide, thus indicating a high content of MMT in it. 22 Commercially pure Na+-MMT with a trade name of PGW was purchased from Nanocor Inc., (AMCOL International Corporation, Illinois, USA) for comparison purposes. Properties of the clays are given in Table 2. QA and QP salts that were purchased from Sigma-Aldrich (St. Louis County, Missouri, USA) and used in this study are shown in Table 3. Hexadecyltrimethylammonium (HMA), tetra(kis)decylammonium (TKA) and tetrabutylammonium (TBA) are ammonium salts, whereas tetrabutylphosphonium (TBP) is a phosphonium salt that can be used for higher temperature applications in polymer-based nanocomposites. HMA has one long hydrocarbon tail with 16 carbons. TKA and TBA have four hydrocarbon tails containing ten and four carbons, respectively. TBP is the same as TBA except for the central atom. In order to observe the effect of CEC of the purified bentonite on the modification process, the commercial MMT PGW was also used. A commercial organoclay with a trade name of Cloisite® 25A was also purchased from Southern Clay Products Inc. (Rockwood Additives, Austin, Texas, USA) for comparison with the organoclays produced. Abbreviations for organoclays in Table 3 are based on the cation-associated surfactant, for example, the term TKA was used for the organoclay that was modified with tetra(kis)decylammonium bromide ((TKA)(Br−)). Other cations of the current study were used with different MMT sources. HMA was used by Xiao et al. 23 with smectite MMT®organoclay and by Filho et al. 22 with Brazilian bentonite. TBA was used by Arora et al. 24 with Indian bentonite. TBP was used by Hedley et al. 25 with Kunipia-F clay and by Patel et al. 19 with Indian bentonite.
Chemical analysis of bentonite.
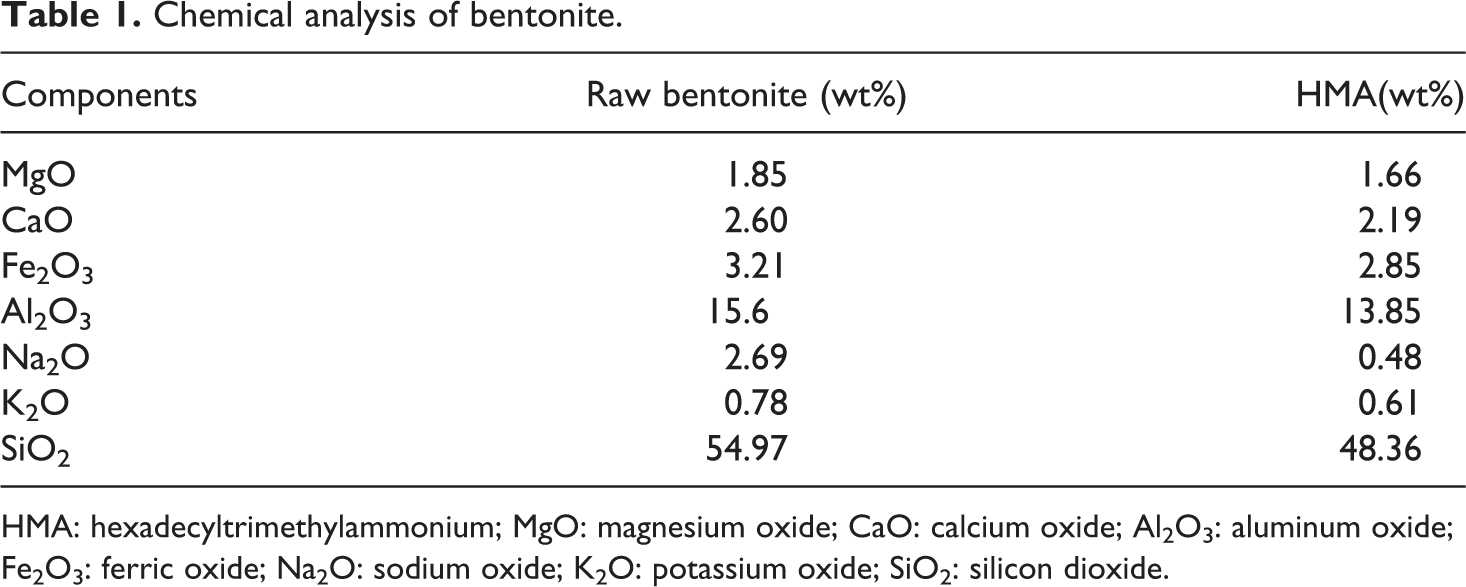
HMA: hexadecyltrimethylammonium; MgO: magnesium oxide; CaO: calcium oxide; Al2O3: aluminum oxide; Fe2O3: ferric oxide; Na2O: sodium oxide; K2O: potassium oxide; SiO2: silicon dioxide.
Properties of clays.

aDetermined by the methylene blue method.
bGiven by the producer.
Structure of the salts used for bentonite modification and abbreviations of organoclays.
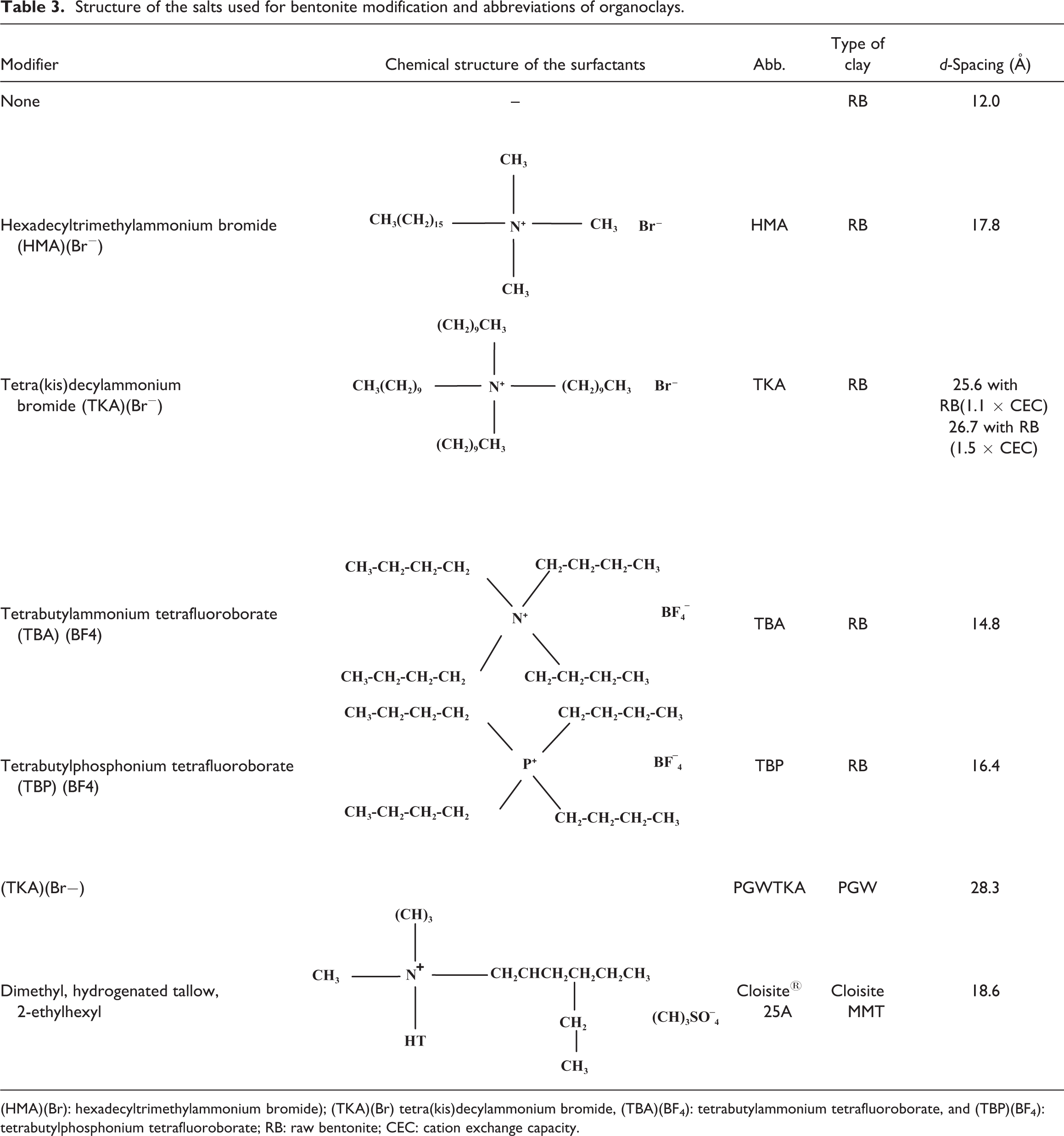
(HMA)(Br): hexadecyltrimethylammonium bromide); (TKA)(Br) tetra(kis)decylammonium bromide, (TBA)(BF4): tetrabutylammonium tetrafluoroborate, and (TBP)(BF4): tetrabutylphosphonium tetrafluoroborate; RB: raw bentonite; CEC: cation exchange capacity.
Preparation of the organoclays
CEC of Resadiye bentonite clay was determined by the methylene blue procedure according to ASTM/C837. The CEC value of the RB was found to be 67.5 mEq/100 g of clay. In the modification of MMT, 5 wt% of RB was dispersed in water–ethanol (4:1, v/v) mixture and stirred with a stirrer for 4 h to obtain a well-dispersed mixture of the clay. The mixture was heated to 80°C in a jacketed heater. 1.1 × CEC of the salt was dissolved and stirred in a water–ethanol (4:1 v/v) mixture and poured into the bentonite dispersion. Another batch was prepared with 1.5 × CEC with the TKA cation with bentonite to observe the effect of high concentration of the modifier on modification, and this organoclay is designated as TKA50. The sample with PGW (commercial pure MMT) was also modified with TKA at 1.1 × CEC to compare it with the samples modified with bentonite. This sample is named as PGWTKA because it was made of PGW clay and TKA cation. The clay–organic salt mixture was stirred for 24 h at 70–80°C. Then, the solution was filtered through a Buchner funnel filtration system, and the retentate was washed with 4–5 l of hot water–ethanol mixture (1:1, v/v) to have a bromide ion-free paste. The absence of bromide ion was tested with silver nitrate (AgNO3). Washing was stopped when a white precipitate was not observed when a few drops of 0.1 N AgNO3 were added to the filtrate. The paste was smeared in a large glass plate and air dried overnight. Then, it was dried in a vacuum oven for 72 h at 80°C. The dried clay was ground and stored in a desiccator.
Characterization of the organoclays
In order to determine the basal spacings of the clays, the x-ray data of clays and composites were obtained by a 100 kV Philips twin tube X-ray diffractometer (Almelo, The Netherlands) (PW/1050) using CuKα radiation that generated a voltage of 40 kV and a current of 40 mA (λ = 1.54 Å). TGA analysis was performed from room temperature up to 1000°C, under a nitrogen atmosphere, using a Shimadzu DTG-60H thermal analyzer (Columbia, MD, USA) with a scanning rate of 15°C/min. FTIR analyses of pure and modified bentonites were conducted with an IR Prestige-21 Shimadzu equipment operating in the range of 400–4000 cm−1. Clay specimens were prepared by mixing a small amount of clay with potassium bromide (KBr) followed by cold pressing to form discs.
Preparation of the polymer nanocomposites
In the production of ternary composites, a corotating twin screw extruder (Thermoprism TSE 16 TC with L/D = 24, screw diameter = 16 mm) (Staffordshire, UK) was used. In the production of ternary composites, a corotating twin screw extruder (Thermoprism TSE 16 TC with L/D=24, screw diameter=16 mm, barrel length=384 mm) was used. A mixture of organoclay (2 wt%), MAPP (2 wt%), and PP was fed to the extruder through the hopper. These samples are abbreviated as PP/Organoclay2/MAPP2. In addition to these concentrations, nanocomposites with TKA and HMA organoclays were prepared with a clay content of (1 wt%), MAPP (3 wt%), and PP. These samples are abbreviated as PP/Organoclay1/MAPP3.
The temperature profile was 210°C throughout the extruder and the screw speed was 150 r/min and 80 r/min in the first and second melt processing steps, respectively. The extrudates obtained were passed through a water bath and then pelletized. The samples pelletized were dried in a vacuum oven for 4 h at 100°C prior to the second extrusion process. The pellets were dried in a vacuum oven for 12 h at 100°C before injection molding. The samples for mechanical testing were prepared using a laboratory scale DSM Xplore (Geleen, The Netherlands) (micro 10 cc) injection molding machine. Injection pressure was 15 bars, the melt and mold temperatures were 220°C and 30°C, respectively. The specimens were molded according to the standards of ISO 527-5A for tensile tests. Tensile properties were measured with Lloyd LR 30 (High Wycombe, UK) K Universal Testing machine using dog bone-type molded samples at a strain rate of 0.5 min−1.
Results and discussion
XRD of the organoclays
Organoclays have reflection angles shifted to the left in comparison to the pristine bentonites (Figure 1), indicating increases in the basal spacing of the unmodified clay according to Bragg’s law: X-ray results of the modified clays with RB, TKA, HMA, TBA, and TBP. HMA: hexadecyltrimethylammonium; TKA: tetra(kis)decylammonium; TBP: tetrabutylphosphonium; RB: raw bentonite; TBA: tetrabutylammonium.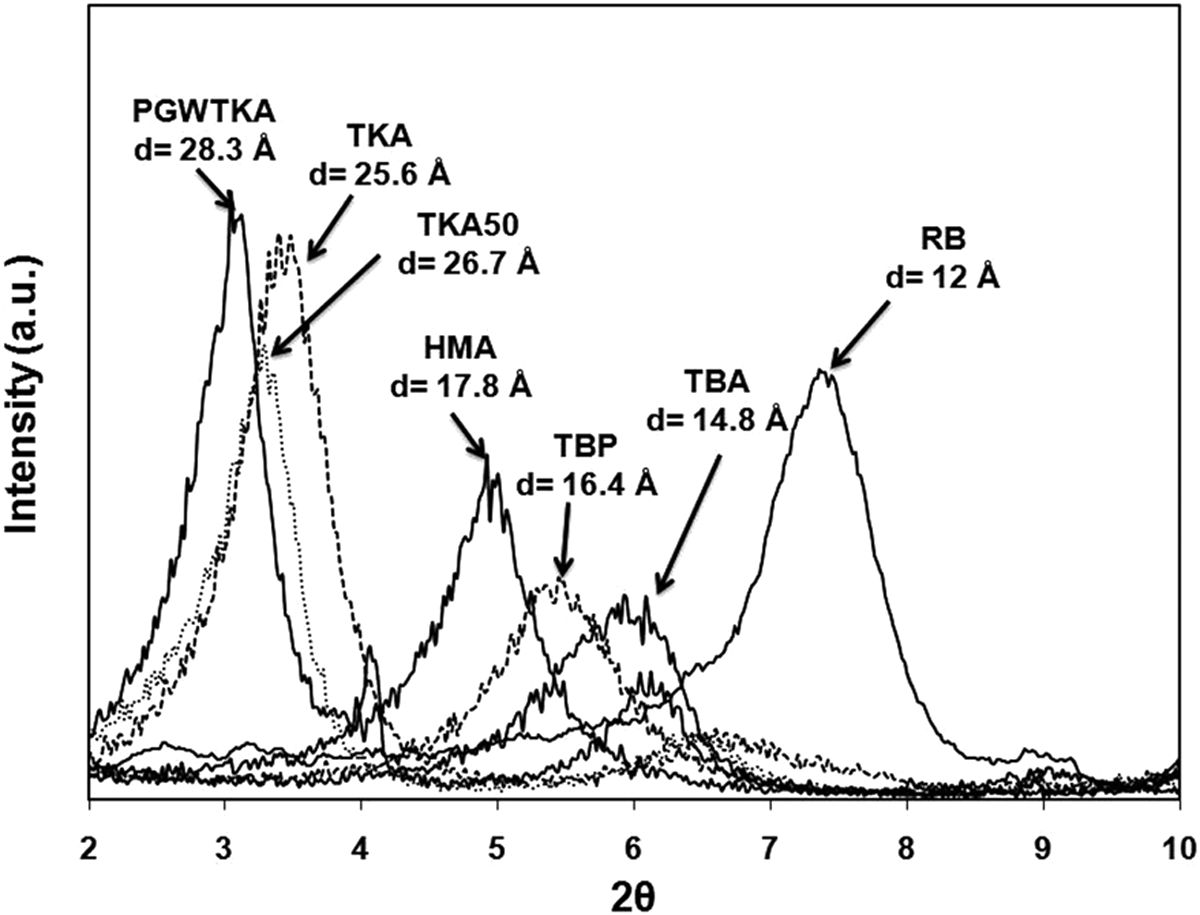
The d-spacing results are summarized in Table 3. All of the organoclays showed higher basal spacings in comparison to the unmodified bentonite. The increase in the basal spacing of the clays indicates that the organic modifiers had diffused between the silicate sheets and modification was effectively accomplished. Ion exchange between the Na+ ions of clay and ions of HMA is also inferred from the chemical analysis given in Table 1. The decrease in the Na+ content in RB after modification with HMA confirms these exchange reactions.
X-ray analysis results in Figure 1 show that the d-spacing of RB increases from 12 Å to 17.8 Å, 25.6 Å, 14.8 Å, and 16.4 Å after modification with 1.1 × CEC of HMA, TKA, TBA, and TBP, respectively. XRD of the organoclay prepared with excess (1.5 × CEC) of TKA shows relatively higher d-spacing (26.7 Å) compared with the one prepared with 1.1 × CEC, indicating that excess usage of surfactant gives rise to higher d-spacing, since more surfactant may intercalate through the clay layers. The highest basal spacing belongs to the organoclay prepared with TKA, which is a relatively bulkier cation in comparison to the other alkyl cations with four long alkyl tails. TBP and TBA with four short tails show similar and small d-spacing values, since they have the same number of carbon atoms, but they differ in the central atom. The basal spacing of the organoclay produced with HMA with one long tail was found to be 20.6 Å by Filho et al., 22 which is slightly different from the one found in the current study, mostly owing to the differences in the modification techniques. The basal spacing of the organoclay produced with TBP was found to be 16.8 Å by Hedley et al., 25 which is consistent with the result of the current study. To the best of our knowledge, no study was conducted with TKA.
High d-spacing results in lower physical interactions between the clay stacks, thus it is easier to delaminate the clay layers in the PCN production. Also, with high d-spacing it is easier for the polymer chains to intercalate through the clay layers. The d-spacing of PGWTKA is the highest among all the clays (d = 28.3 Å), which is most probably due to the high purity of PGW as shown by its high CEC value (145 mmol/100 g of clay). High CEC of the clay enables the clay to absorb high amounts of organic cation owing to its high exchangeable cation, that is, Na+, content.
Depending on the packing density, temperature, and chain length, the arrangement and confinement of alkyl chains between the galleries of clay are assumed to form mono- or bilayers or radiate away from the surface, forming extended (paraffin-type) mono or bimolecular arrangements. 26,27 Alkylammonium ions with short tails are arranged in monolayers, whereas alkylammonium ions with longer tails arrange themselves in bilayers with the alkyl chain axes parallel to the silicate layers. The monolayer arrangement has an approximate basal spacing of 14 Å, whereas the bilayer has an approximate basal spacing of 18 Å. Thus, the values of 14.8 Å for TBA and 16.4 Å for TBP are consistent with a monolayer arrangement of the quaternary alkylammonium ions in the interlayer space. Note that these surfactants have four short tails. The d-spacing of 17.8 Å for HMA (with one long tail) is consistent with the bilayer arrangement. 15,25 Three-layered structures of twirled alkyl chains are seen with highly charged smectites and/or long surfactant cations. 28 This pseudotrimolecular arrangement shows a basal spacing of approximately 22 Å. Thus, the values of 25.6 Å for TKA, 26.7 Å for TKA50, and 28.3 Å for PGWTKA indicate paraffin-type pseudotrimolecular arrangement.
TGA analysis
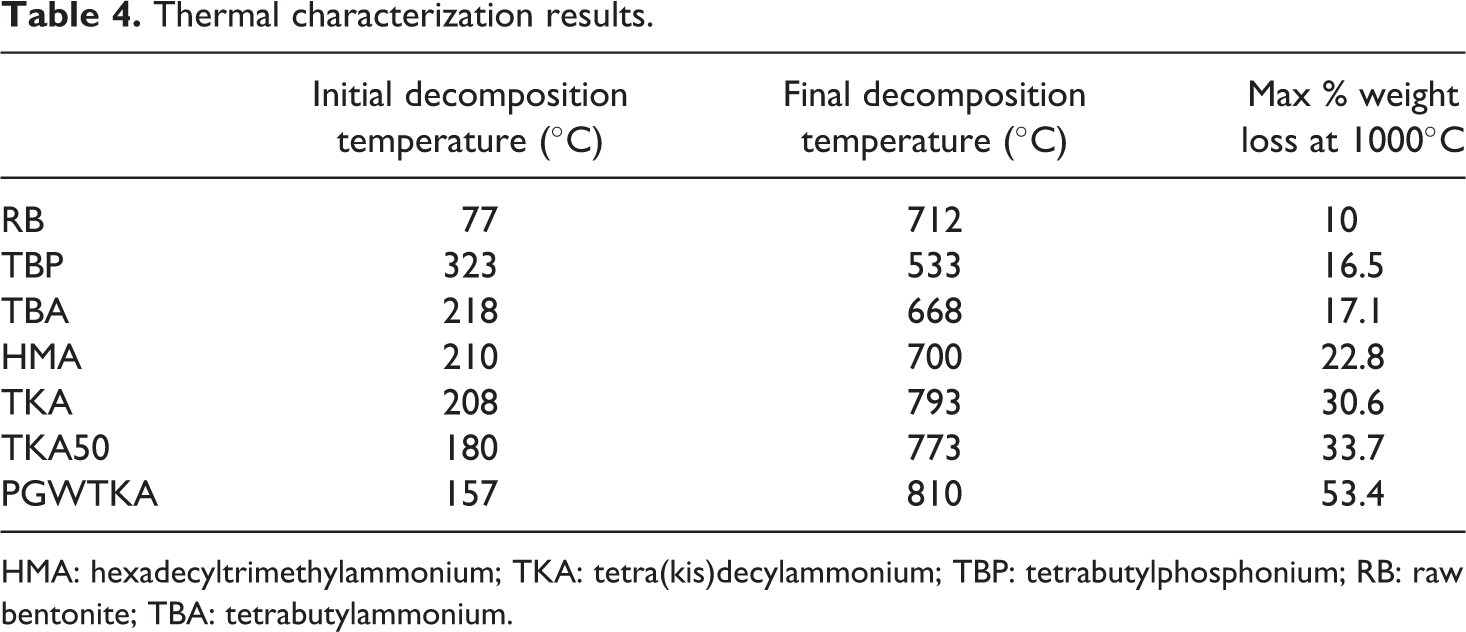
TGA analyses of RB and modified bentonites with RB (Figure 2 and Table 4) show that based on their first decomposition temperatures, the thermal stability of the organoclays can be classified as RB < PGWTKA < TKA50 < TKA < HMA < TBA < TBP. It can be seen from Figure 2 that the bentonite modified with phosphonium salt TBP has a higher thermal stability than the bentonite modified with ammonium salt TBA. These two surfactants have the same number of carbon atoms.
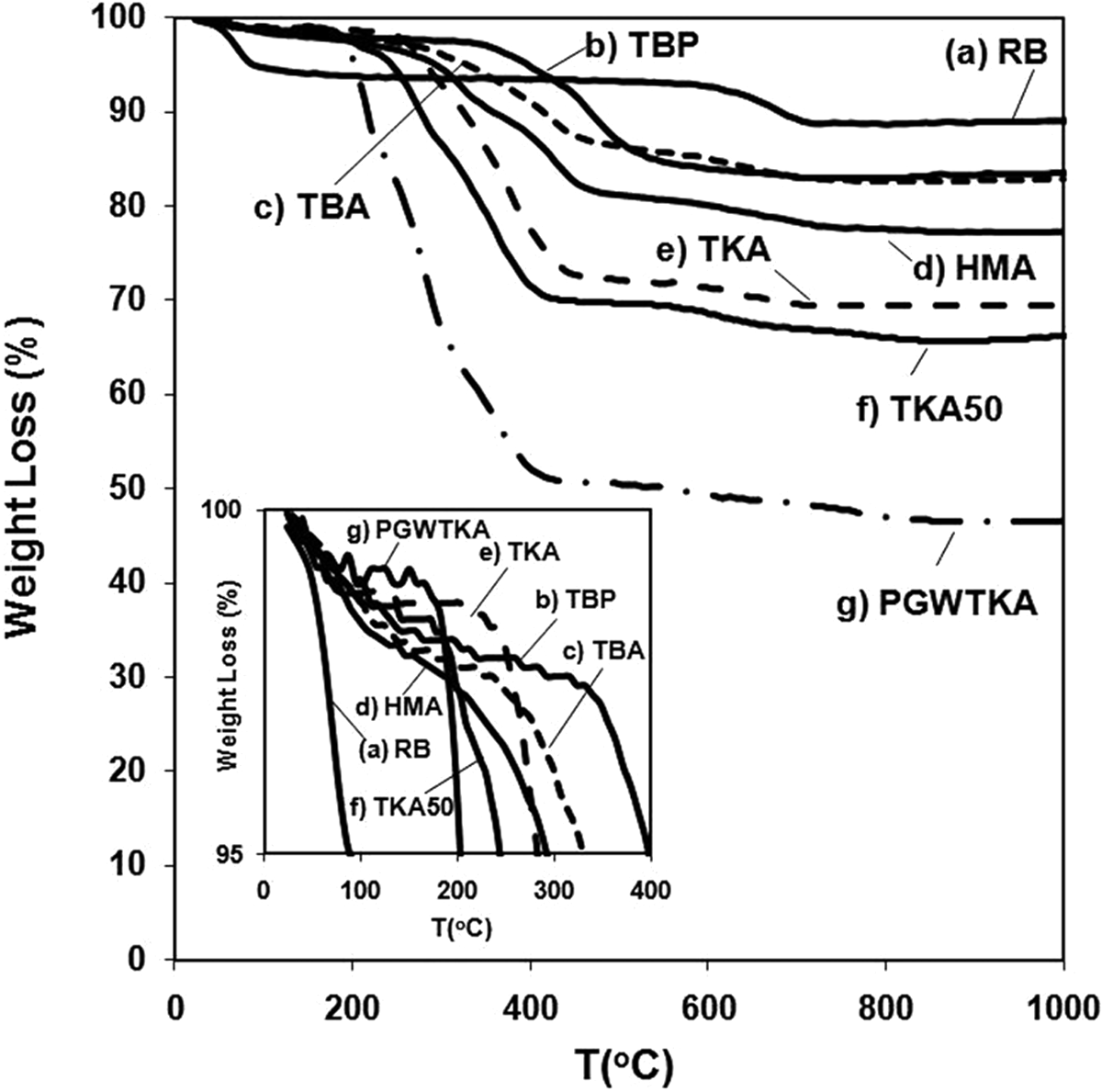
Thermogravimetric analysis results of (a) RB and organoclays, (b) TBP, (c) TBA, (d) HMA, (e) TKA, (f) TKA50, and (g) PGWTKA. HMA: hexadecyltrimethylammonium; TKA: tetra (kis)decylammonium; TBP: tetrabutylphosphonium; RB: raw bentonite; TBA: tetrabutylammonium.
Thermal characterization results.
HMA: hexadecyltrimethylammonium; TKA: tetra(kis)decylammonium; TBP: tetrabutylphosphonium; RB: raw bentonite; TBA: tetrabutylammonium.
Decomposition of bentonite manifests itself in two steps. The initial weight loss in the first step at 30–150°C is attributed to residual or free water evaporation, which is adsorped by the cations in the interlayer space of the bentonite. 12,18,22 The second loss is related to the dehydroxylation of structural water molecules from the crystal lattice of MMT. 12,16,25 The overall weight losses of all modified MMT’s are higher than that of RB, since these weight losses are due to decomposition of intercalated alkyl tails of the surfactants within the galleries of MMTs. The weight losses of the modified MMTs between 30°C and 700°C are due to the decomposition of the organic salts and dehydroxylation of the MMT layers. Beyond 700°C, residual carbonaceous product formation begins. 16,18,21,25 Xie and co-authors 21 claimed that the first and second decomposition steps of modified MMTs are the most important steps, since the formation of decomposition products may affect the interfacial interactions between the MMTs and polymer in PLSN. First, decomposition temperatures of modified bentonites in this study are greater than ∼200°C (Table 4) and thus permitting their use as organoclays in melt processing of polyolefins such as PP.
The maximum weight loss of the produced organoclays was calculated at 1000°C (Figure 2 and Table 4). This value is an indication of the organic content and the water present in the organoclays. RB has the least weight loss, since it does not have any organic modifier. The molecular weights of tetrabutylphosphonium tetrafluoroborate (M w = 346.2) and tetrabutylammonium tetrafluoroborate (M w = 329.3) are close to each other, thus the organoclays produced with these surfactants have almost the same organic + water content (∼17%).
The organoclay produced from (TKA)(Br) has a higher organic + water content (∼31 wt%) than the organoclay produced from hexadecyltrimethylammonium bromide ((HMA)(Br), ∼23 wt%). The M w’s of (TKA)(Br) and (HMA)(Br) are 659 and 364, respectively, thus the TKA organoclay has a higher organic content than the HMA organoclay. The TKA organoclay has a higher d-spacing than the HMA organoclay, indicating that the former organoclay has a higher organic content than the latter one.
In order to see the effect of the surfactant content, TKA and TKA50 can be compared. The organoclay prepared as TKA50 has a higher organic + water content (∼34 wt%) than the TKA organoclay (∼31 wt%).
The PGWTKA organoclay was produced from a commercial pure MMT with a CEC of 145 mmol/100 g of clay, whereas the TKA organoclay was prepared from a bentonite with a CEC of 67.5 mmol/100 g of clay. Owing to its higher CEC, the PGWTKA organoclay can absorb more surfactant than the TKA organoclay, thus it has a higher organic + water content (∼53 wt%) than the TKA organoclay (∼31 wt%).
FTIR analysis
FTIR spectra of RB, and modified bentonites were obtained using KBr pellets. In the FTIR spectra of RB (Figure 3), the band near 3620 cm−1 is attributed to the OH group bound with Al+3 in bentonite, which is a characteristic peak of MMTs with high amounts of Al in the octahedra. 29 Free water of bentonite gives bands at 3440 cm−1 and 1639 cm−1, and are attributed to stretching and bending vibrations of the OH groups of water. The characteristic band of MMT at 1114 cm−1 shows Si–O out-of-plane stretching vibration. The band at 1033 cm−1 is attributed to Si–O in-plane stretching and the band at 529 cm−1 is due to Si–O bending vibrations. Bending vibration of hydroxyl groups with (aluminum hydroxide) is given by the band at 910 cm−1, while the band at 871 cm−1 is attributed to AlFe–OH. Moreover, the band at 696 cm−1 shows the presence of quartz in the bentonite. 19,30,31
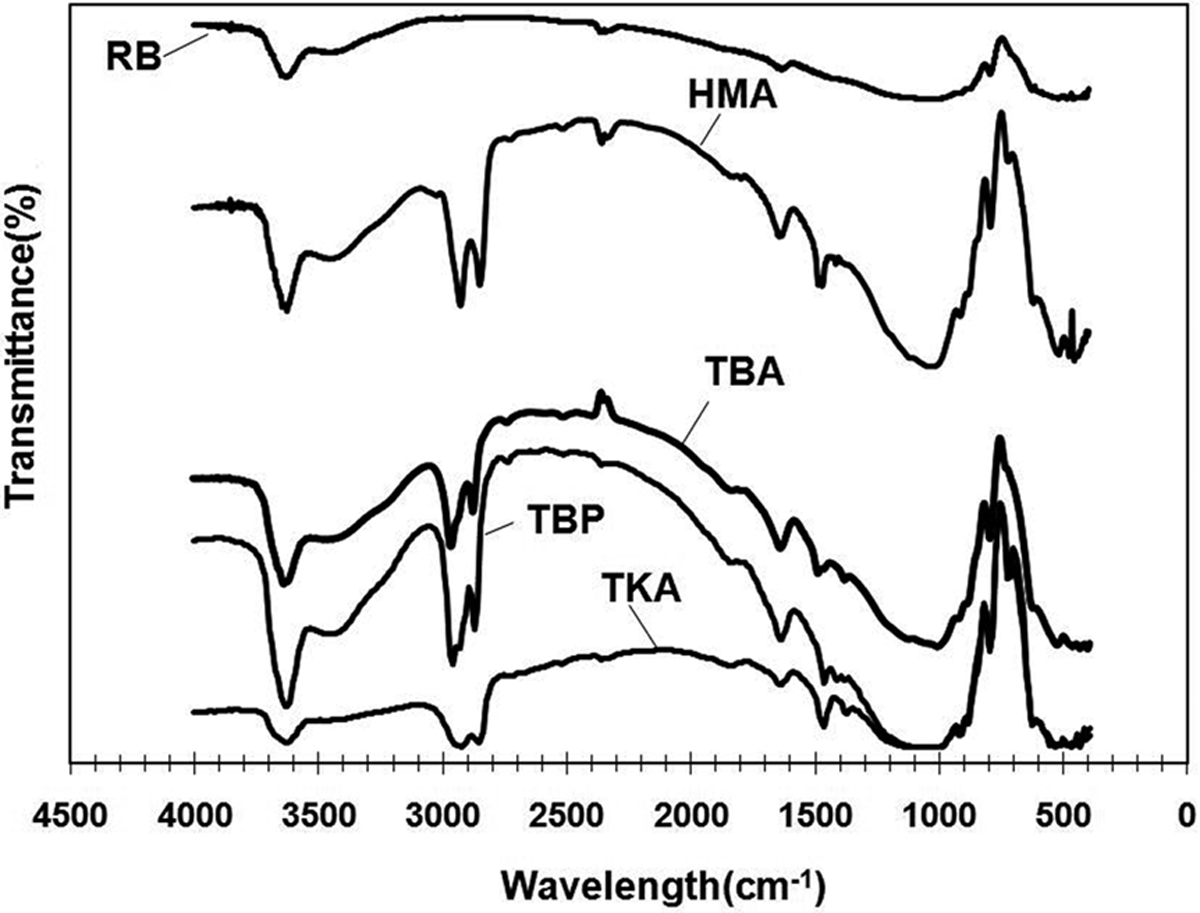
Fourier transform infrared spectroscopy results of (a) RB and organoclays, (b) TBP, (c) TBA, (d) HMA, and (e) TKA. HMA: hexadecyltrimethylammonium; TKA: tetra(kis)decylammonium; TBP: tetrabutylphosphonium; RB: raw bentonite; TBA: tetrabutylammonium.
In the FTIR spectra of alkylammonium/phosphonium cations, C–H stretching usually gives bands between wave numbers of 3020 cm−1 and 2800 cm−1.31 Thus, the FTIR spectra of modified bentonites (Figure 3) show absorption bands near 2931 cm−1 (υ as(CH2), asymmetric stretching of CH2) and 2854 cm−1 (υ s(CH2), symmetric stretching of CH2) due to cations intercalated between the MMT layers. 32 New absorption band in modified bentonites at 1487 cm−1 is attributed to flexural vibrations of CH3 (δ as(C–H)), which arises due to the cation (CH3)4N+. 32 FTIR analysis indicates intercalation of salts within the bentonite layers with these new bands observed in the modified bentonites.
XRD and transmission electron microscopy of nanocomposites
X-ray analysis of the composites prepared with 2 wt% of organoclay and 2 wt% of MAPP are given in Figures 4 to 7. The figures show that the shift of the peak to lower angles can be seen only in the case of TKA organoclay that has the longest alkyl chain length compared to the other three salts. The original d-spacing of TKA did not change significantly in this clay, as it increased from 2.56 nm to 2.65 nm following compounding. Transmission electron microscopy (TEM) analysis of the same composition in Figure 8 indicates intercalated layers with TKA as well. X-ray analysis of HMA, TBA, and TBP show no shifts in the d-spacing following compounding. These results may indicate the possibility of insufficient mixing of organoclay, MAPP, and PP. There have been several studies on the effects of the processing route and the effects of shear on the dispersion of organoclays. Hong et al. 33 compounded a masterbatch (composed of organoclay and MAPP) and then diluted it with PP. Their XRD results revealed that the clay dispersion took place in the first masterbatch compounding and no improvement was observed after additional shear force was exerted in the second compounding process. This was attributed to the possible polarity difference between the PP and MAPP. However, Quintanilla et al. 34 claimed that the two-step mixing is better than the one-step mixing to obtain dispersed clay structures in a PP matrix. Touati et al. 35 showed that a four cycle shear application resulted in high quantities of delaminated organoclay layers in the PP matrix. However, the mechanical and thermal properties were deteriorated due to the negative effects of degradation.
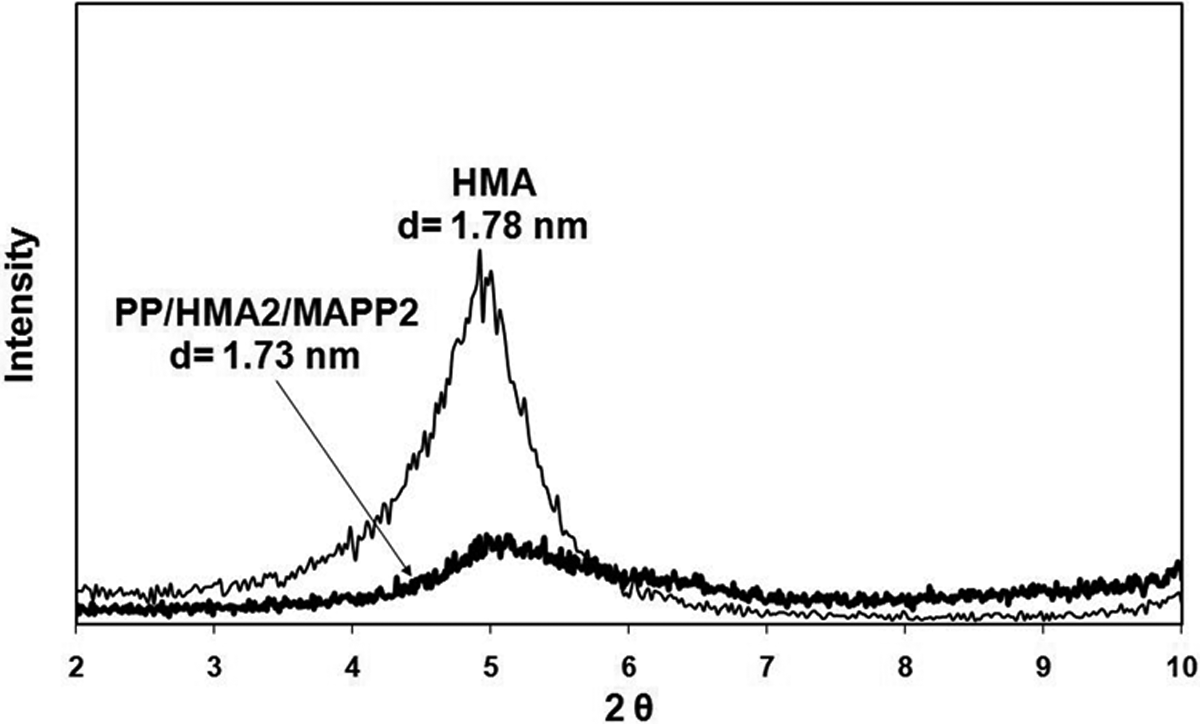
X-ray diffraction diagram of HMA and PP/HMA2/MAPP2. HMA: hexadecyltrimethylammonium; PP: polypropylene; MAPP: PP-grafted maleic anhydride.
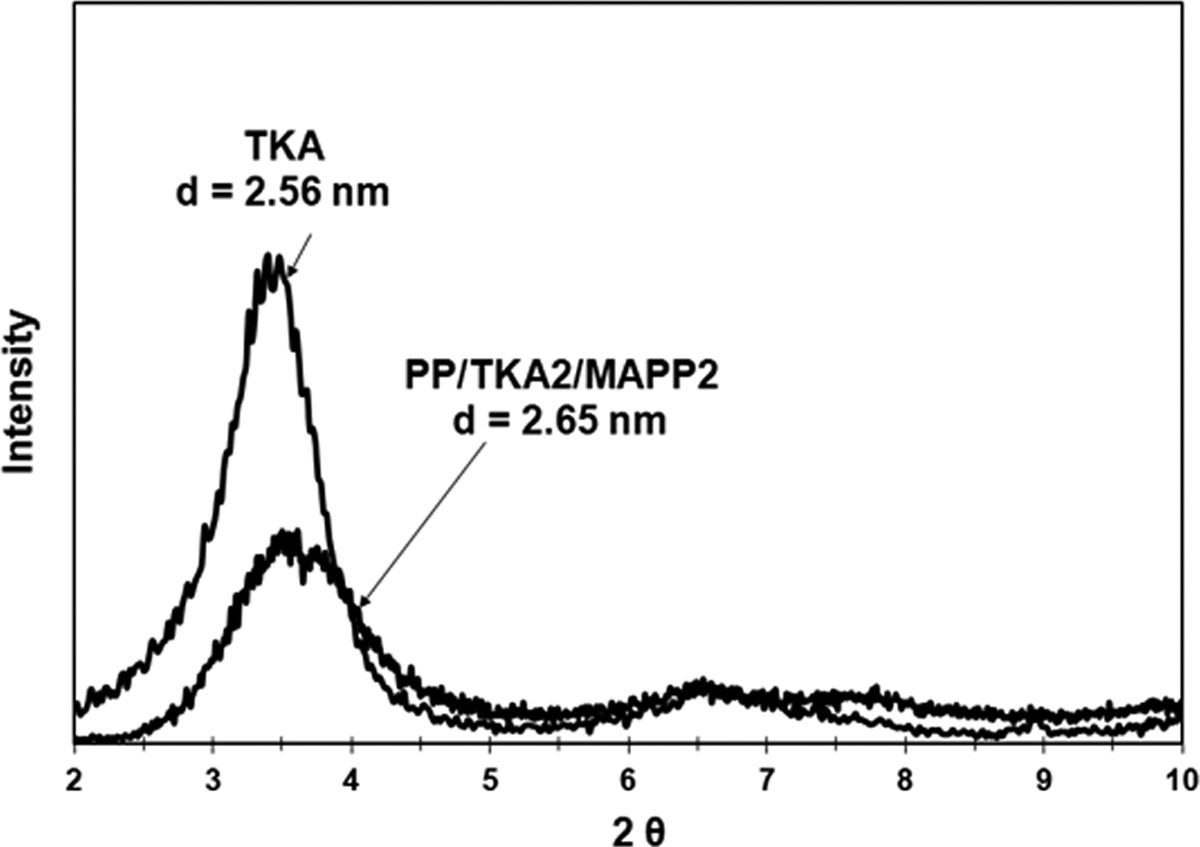
X-ray diffraction diagram of TKA and PP/TKA2/MAPP2. TKA: tetra(kis)decylammonium; PP: polypropylene; MAPP: PP-grafted maleic anhydride.
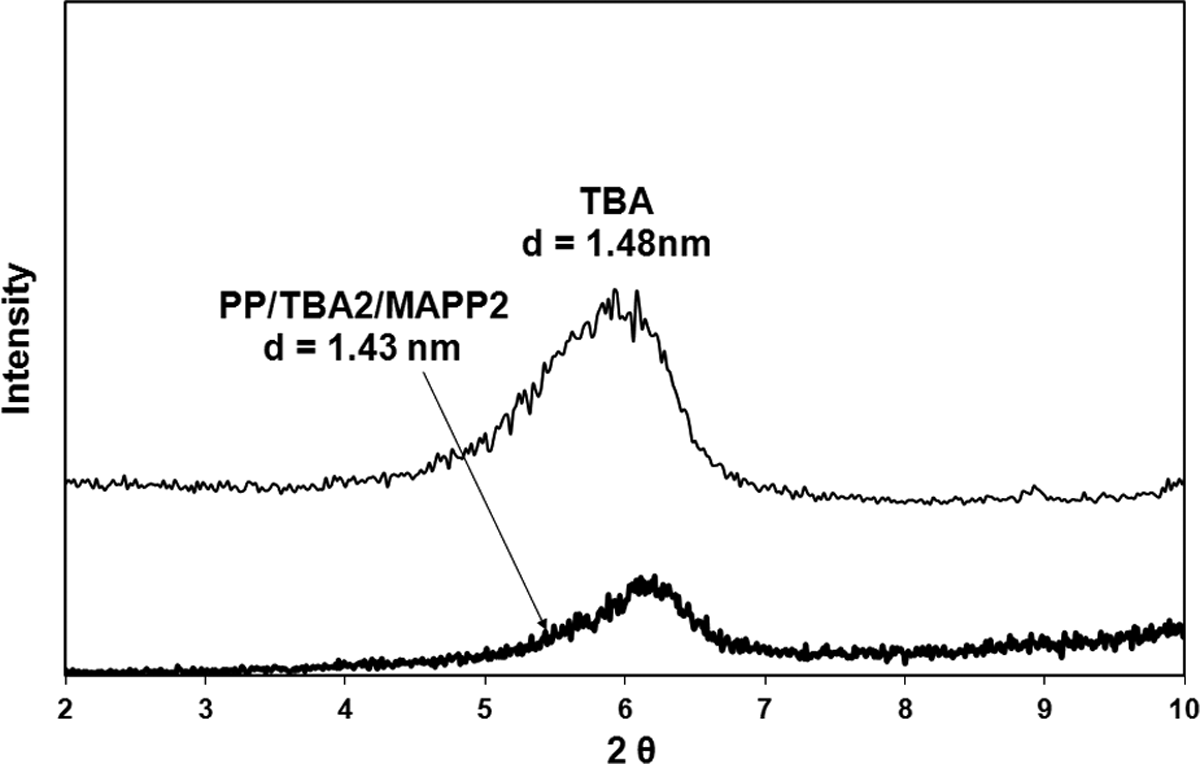
X-ray diffraction diagram of TBA and PP/TBA2/MAPP2. TBA: tetrabutylammonium; PP: polypropylene; MAPP: PP-grafted maleic anhydride.
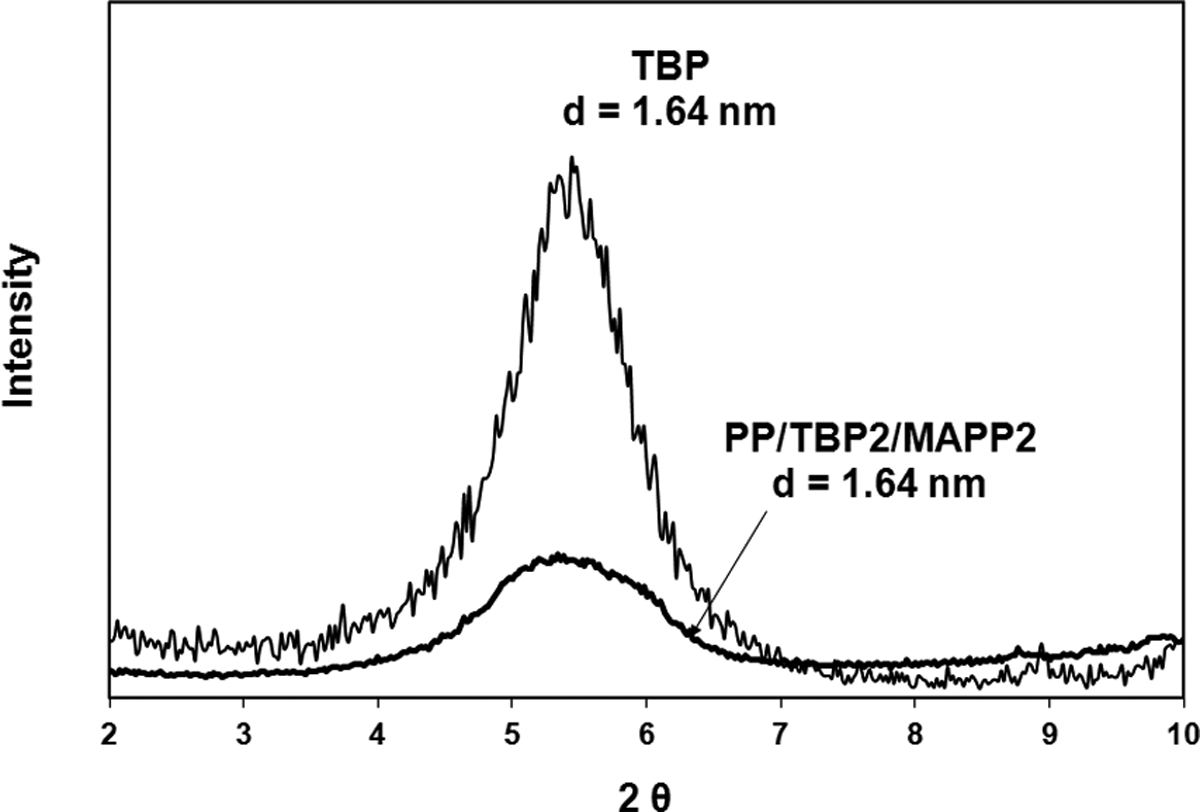
X-ray diffraction diagram of TBP and PP/TBP2/MAPP2. TBP: tetrabutylphosphonium; PP: polypropylene; MAPP: PP-grafted maleic anhydride.

Transmission electron micrograph image of PP/TKA2/MAPP2. TKA: tetra(kis)decylammonium; PP: polypropylene; MAPP: PP-grafted maleic anhydride.
In our study, a two-step direct melt mixing was applied. Possibly, the short alkyl chain lengths of TBA and TBP could not enhance the intercalation of organoclay layers. Although HMA has a longer alkyl chain length in comparison to TBA and TBP, it also did not aid the dispersion of clay layers within the polymer matrix. These results indicate that several elements contribute to the formation of delaminated clay structures in polyolefins. Some of these factors are the matrix–filler interactions, type, and weight content of the modifier, the amount of the MAPP compatibilizer used, and the processing conditions.
Kim et al. 36 studied the effect of MAPP concentration and the ratio of MAPP/organoclay in PP-based organoclay nanocomposites, and the results revealed that high MAPP concentration and high MAPP/organoclay ratio leads to exfoliation mechanism owing to enhanced attraction of MAPP on the silicate layers, permitting easy delamination of the clay layers during shearing. Rodriguez-Llamazares et al. 37 showed that the amount of compatibilizer plays an important role in producing polymer–organoclay nanocomposites with a high degree of dispersion. The ratio of the compatibilizer to organoclay for direct melt mixing process has been chosen as 3:1 in order to obtain highly dispersed PP/clay nanocomposites. 38,39 Thus, in our study PP/HMA1/MAPP3 composite with an HMA/MAPP ratio of 1/3 was also prepared to increase the delamination of the clay layers. Figure 9 shows the XRD of this composition and indicates that it has a higher delamination and insertion of clay layers compared with the composites prepared with an HMA/MAPP ratio of 1. The XRD peak in Figure 9 has a lower intensity and it is broader than the one in Figure 4, pointing out to a more delaminated and disordered silicate layer structure than the one in Figure 4. 40 This result confirms that increased MAPP/organoclay ratio enhances the exfoliation of organoclay layers in the PP matrix. Transmission electron micrograph of PP/HMA1/MAPP3 (Figure 10) shows some intercalated clay layers as well as tactoids. The same ternary composition ratio was prepared with TKA (PP/TKA1/MAPP3) and its transmission electron micrograph (Figure 11) exhibits few intercalated and exfoliated single layers of TKA organoclay in the PP matrix. The nanocomposite prepared with commercial organoclay Cloisite 25A also shows low XRD peak intensity (Figure 12), indicating possible delamination of the organoclay through compounding with PP/MAPP.
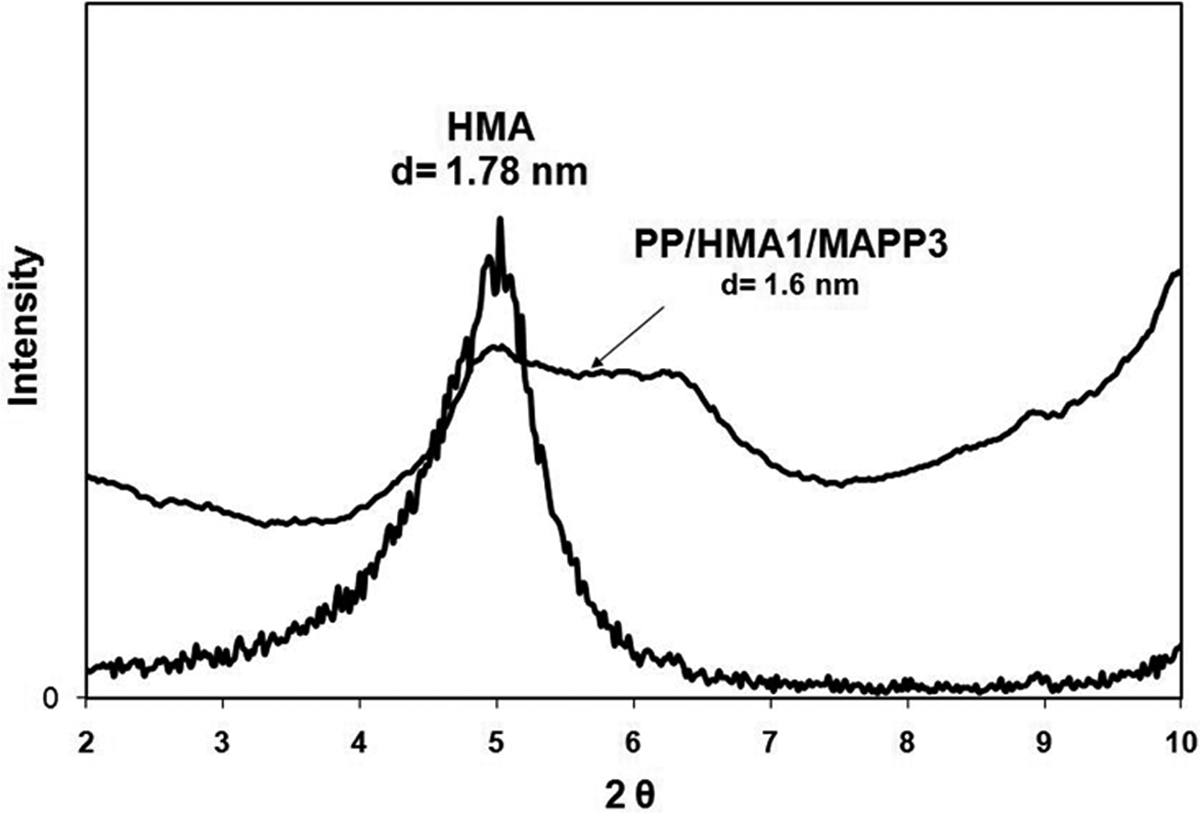
X-ray diffraction diagram of HMA and PP/HMA1/MAPP3. HMA: hexadecyltrimethylammonium; PP: polypropylene; MAPP: PP-grafted maleic anhydride.
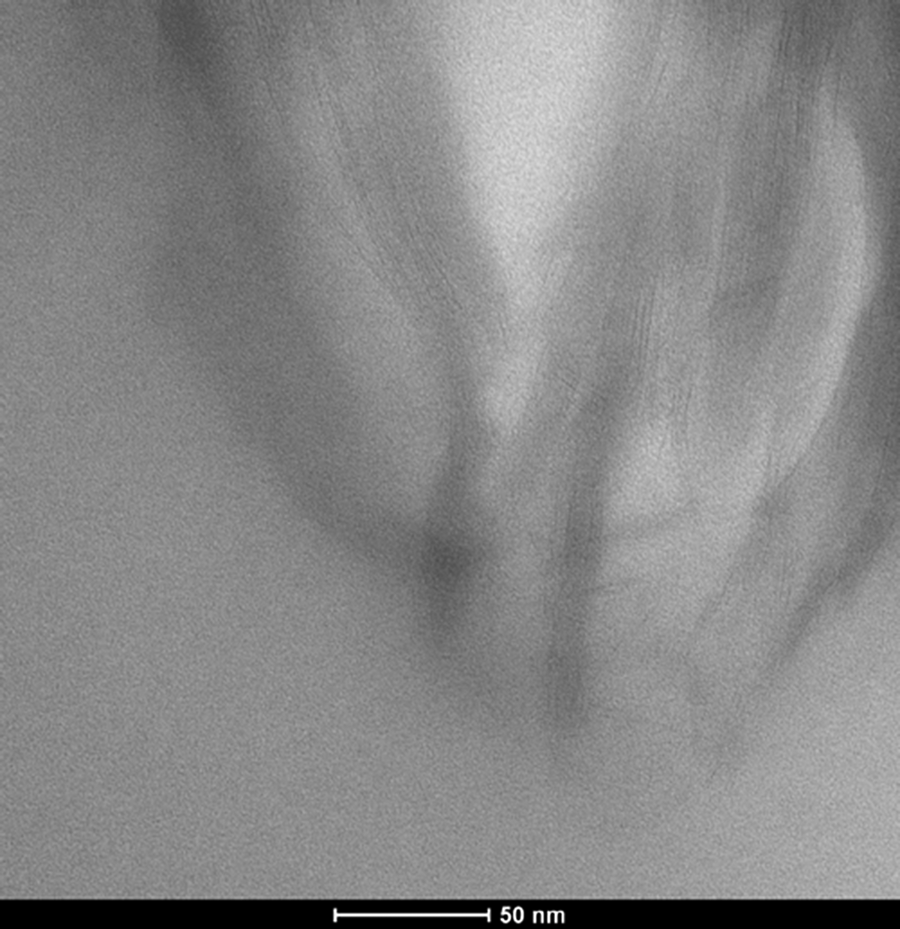
Transmission electron micrograph image of PP/HMA1/MAPP3. HMA: hexadecyltrimethylammonium; PP: polypropylene; MAPP: PP-grafted maleic anhydride.
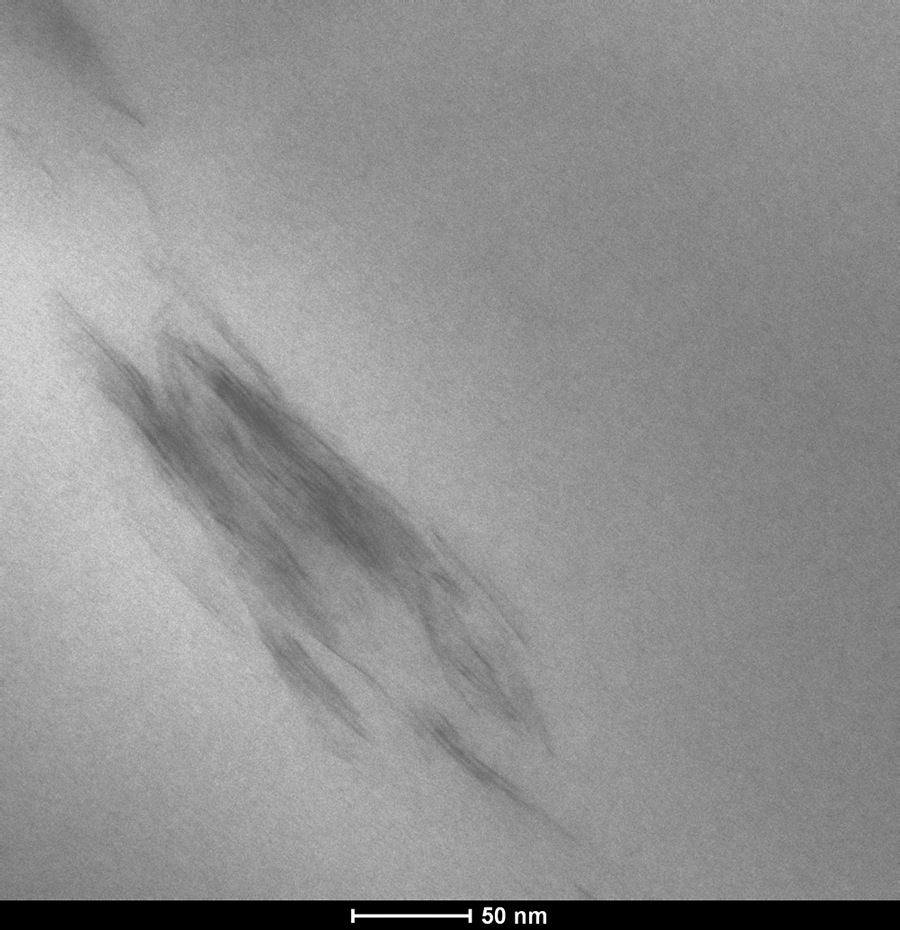
Transmission electron micrograph image of PP/TKA1/MAPP3. TKA: tetra(kis)decylammonium; PP: polypropylene; MAPP: PP-grafted maleic anhydride.
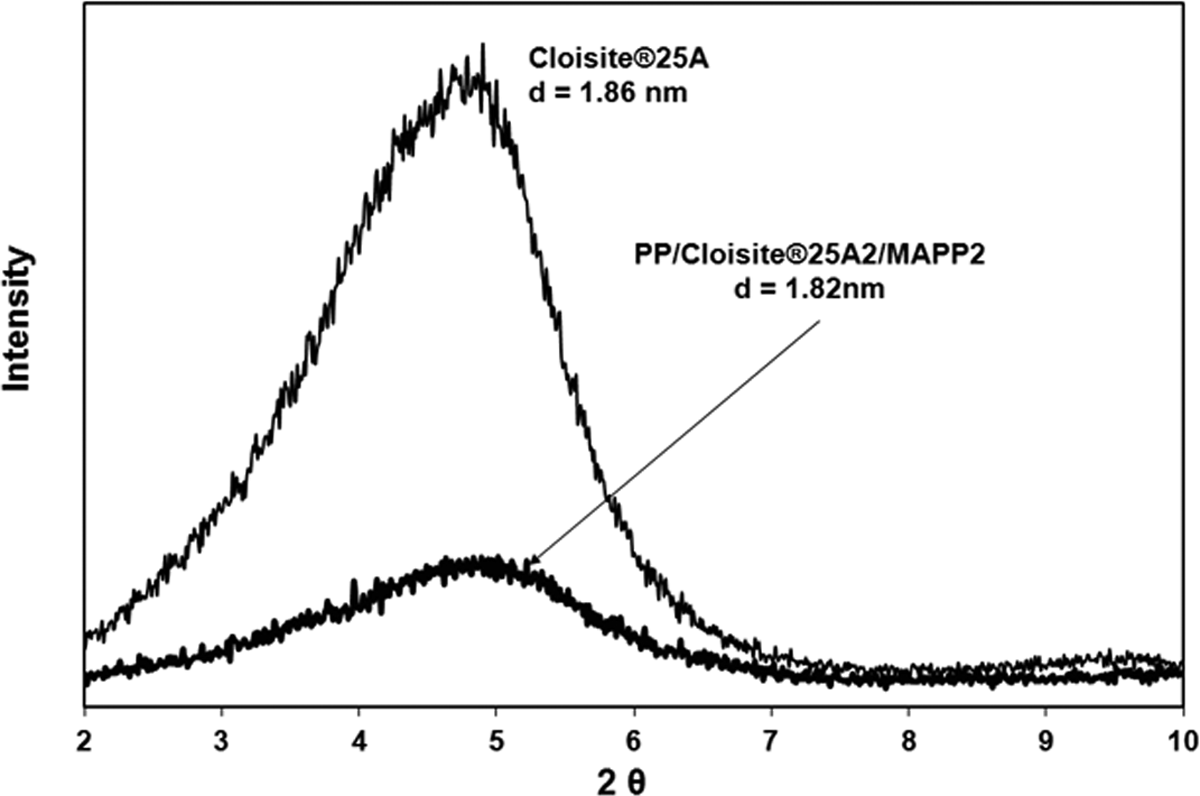
X-ray diffraction diagrams for Cloisite® 25A and PP/Cloisite 25A2/MAPP2.
DSC analysis of the nanocomposites
Thermal characteristics of PP, PP/MAPP blends, and PP/organoclay/MAPP composites were investigated using DSC. Table 5 summarizes the DSC results of the PP-based composites in terms of melting temperature, T m, heat of fusion of the sample (ΔH (J/g)), and percentage crystallinity. In the binary blend, addition of MAPP to PP slightly increased the crystallinity of the PP. Ternary composites of PP/organoclay2/MAPP2 with an MAPP/organoclay ratio, α, of 1 generally showed a slight increase in the crystallinity. This could be due to the nucleation effect of the organoclay. When the clay content was lower, as in PP/TKA1/MAPP3, no significant increase was observed in the crystallinity, and this composition did not show any dispersion as observed by XRD. However, in the PP/HMA1/MAPP3 ternary nanocomposite, the clay exhibited intercalation/exfoliation and owing to the nucleation effect of the clay, the crystallinity is significantly higher than that of PP.
Differential scanning calorimetry results of PP and PP/MAPP-based composites.
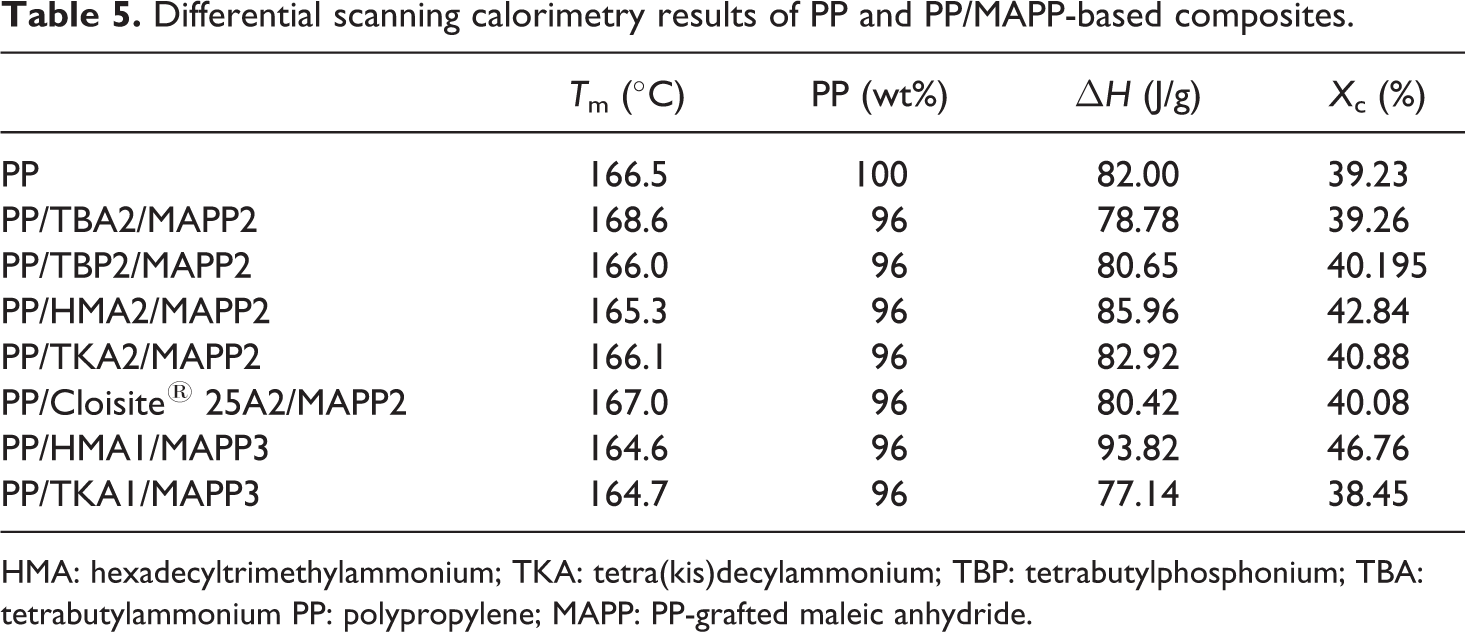
HMA: hexadecyltrimethylammonium; TKA: tetra(kis)decylammonium; TBP: tetrabutylphosphonium; TBA: tetrabutylammonium PP: polypropylene; MAPP: PP-grafted maleic anhydride.
Mechanical properties of the nanocomposites
Typical stress–strain diagrams of PP-based composites is shown in Figure 13. PP shows yield followed by a ductile behavior. Neat PP that is extruded twice has tensile strength (TS), Young’s modulus (YM), and percentage elongation at break values of 39 MPa, 1512 Mpa, and 422, respectively. Addition of MAPP into the PP decreased its YM, since MAPP has a lower modulus than PP. At the same time, MAPP enhanced the elongation at break of PP.
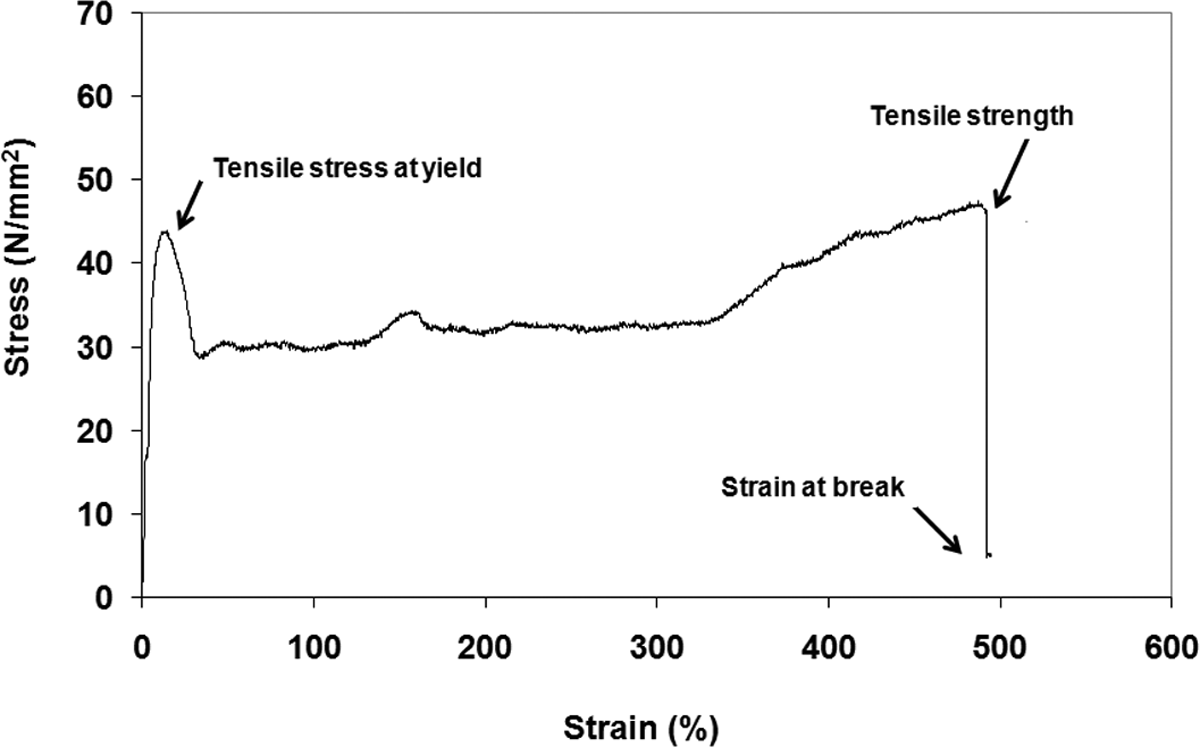
Typical stress–strain diagram of PP. PP: polypropylene.
Effects of the organoclay type and ratio of MAPP/organoclay (α) on TS can be seen in Figure 14. The first three bars in the figures represent the neat PP, extruded PP, and PP98-MAPP2 mixture, respectively. The following bars with the same shade effect show the composites prepared with the same organoclay but with different α values, where the first one represents the composite of PP/Organoclay2/MAPP2 (α = 1) and the second one represents the composite of PP/Organoclay1/MAPP3 (α = 3). For comparison purposes, the composite with the commercial organoclay, Cloisite 25A, was also prepared. TBA and TBP had little increase in the d-spacing compared to TKA and HMA, therefore samples with α = 3 were prepared only with TKA and HMA. X-ray data of the composites (Figures 4 to 7 and 9) show limited dispersion of the clays in the PP matrix, and this effect can be seen in the tensile and yield strength properties as well (Figures 14 and 15). If the clay particles had been exfoliated, larger surface area would have been formed between the clay particles and the polymer, making the stress transfer to the clay layers more effective, and resulting in higher tensile properties. 41 Figure 16 shows enhancement in the YM of the nanocomposites regardless of the clay type. For the samples with α = 1, YM of the PP increased by melt mixing with MAPP and the organoclays TBA, TBP, and HMA. However, it did not increase much with TKA. YM enhancements obtained with α = 1 may be attributed to the orientation of the clay layers, in addition to other factors such as the concentration of clay, and adhesion between the clay–polymer–compatibilizer interfaces. Also, tactoids of clay particles residing in the matrix would lead to an increase in the modulus. The organoclays with small d-spacing (obtained with surfactants with short alkyl tail, i.e. TBA and TBP) showed higher modulus values when compared with the organoclays with TKA and HMA whose d-spacings are much higher (Figure 16).
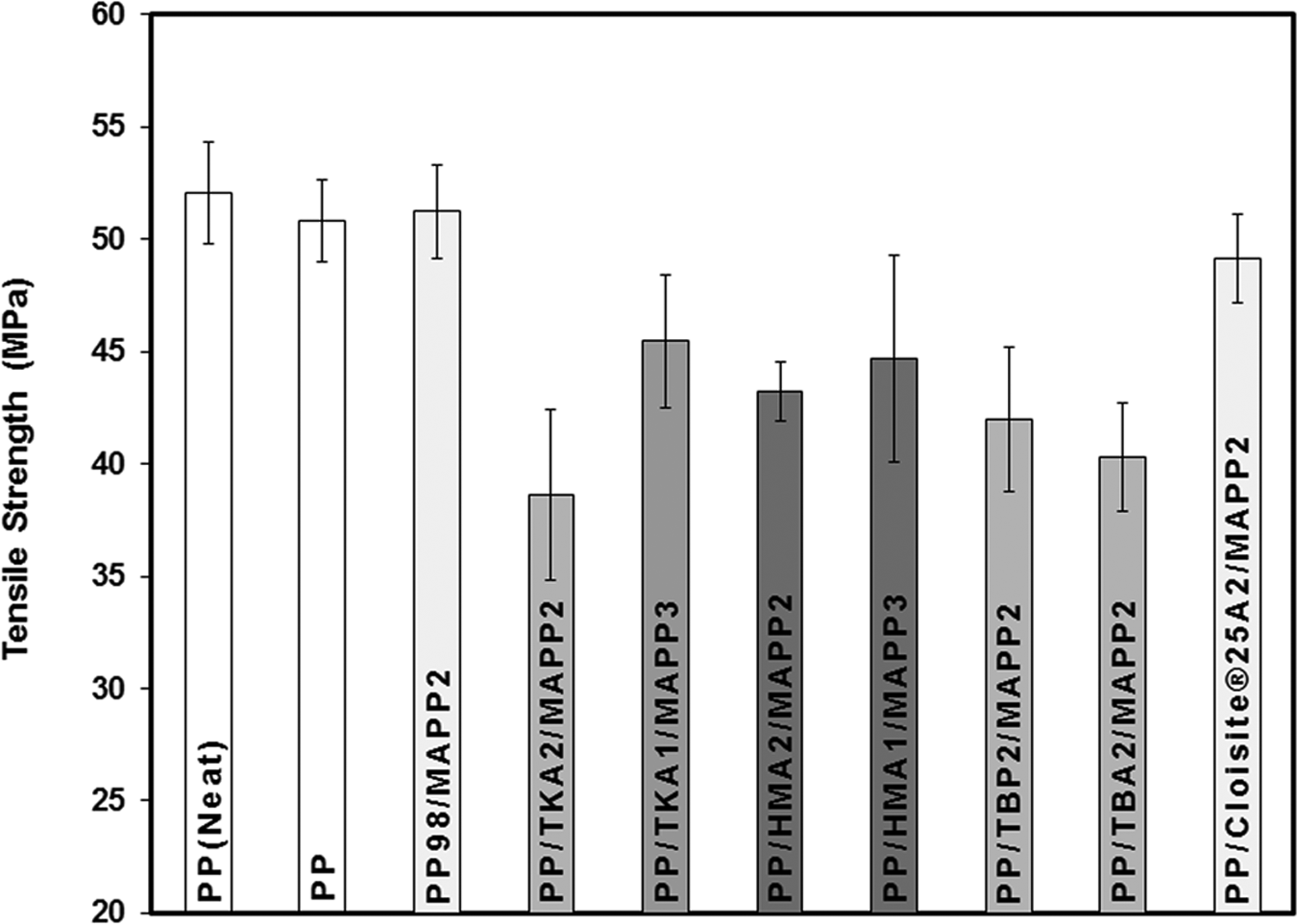
Effect of organoclay type on tensile strength of PP/Organoclay2/MAPP2 and PP/Organoclay1/MAPP3 composites. PP: polypropylene; MAPP: PP-grafted maleic anhydride.
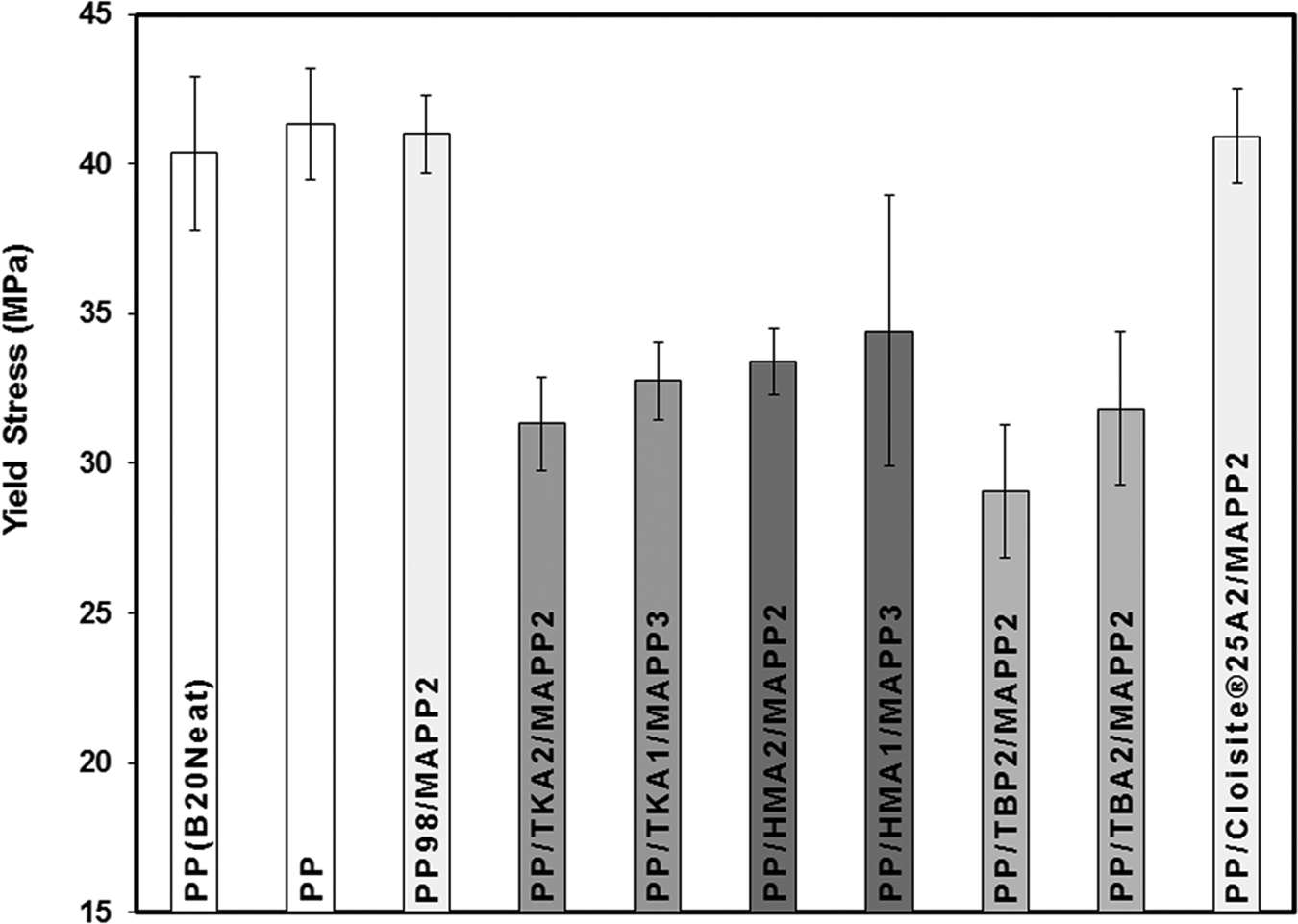
Effect of organoclay type on tensile stress at yield of PP/Organoclay2/MAPP2 and PP/Organoclay1/MAPP3 composites. PP: polypropylene; MAPP: PP-grafted maleic anhydride.
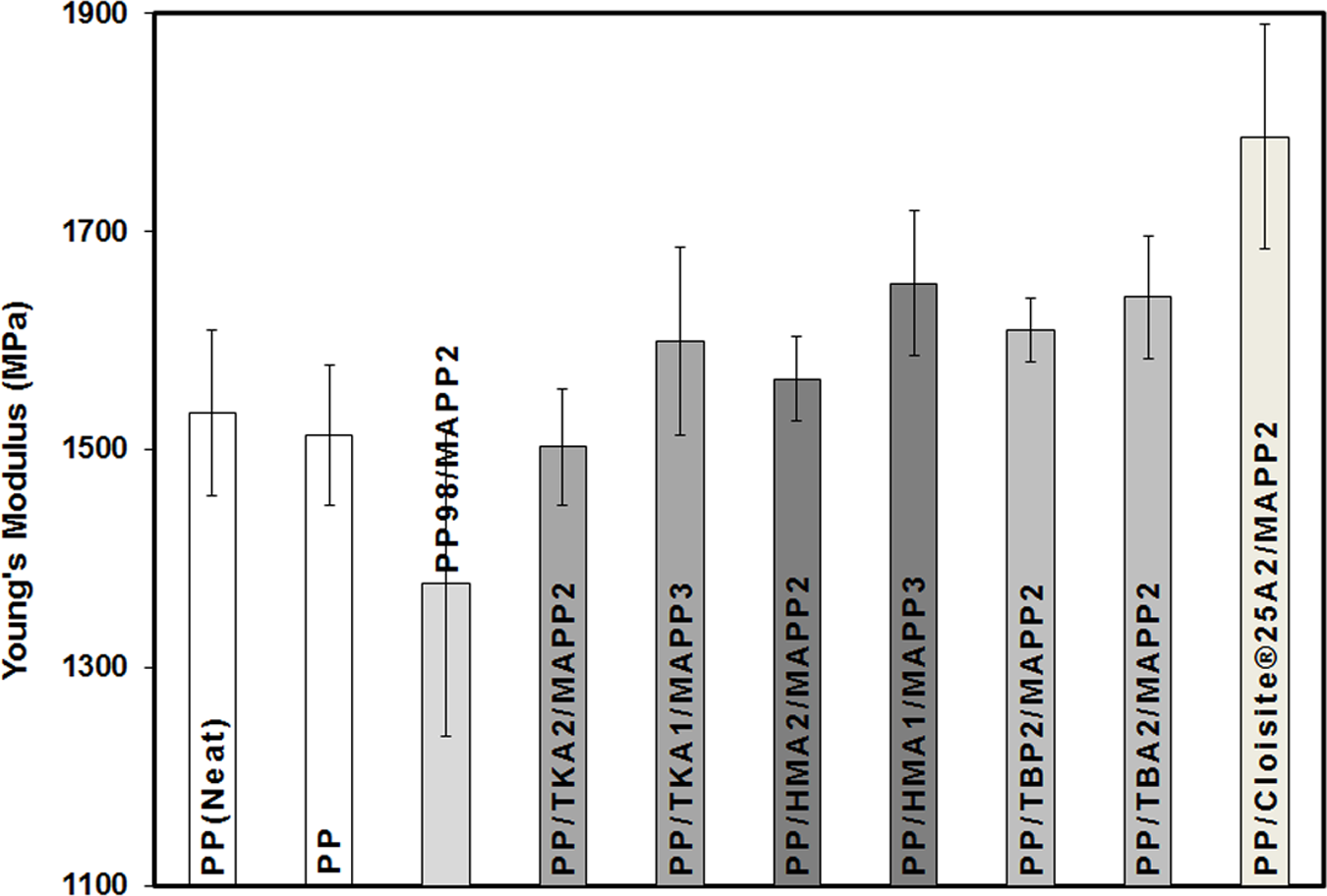
Effect of organoclay type on Young’s modulus of PP/Organoclay2/MAPP2 and PP/Organoclay1/MAPP3 composites. PP: polypropylene; MAPP: PP-grafted maleic anhydride.
YM of the samples with α = 3 are higher in comparison to the YM of the samples with α = 1, owing to the high concentration of MAPP used. Increase in the MAPP concentration was found to increase the dispersion of clay layers due to increased adhesion between the compatibilizer and organoclay. 36 Kim et al. 36 studied PP/organoclay/MAPP nanocomposites by TEM using different MAPP/organoclay ratios and observed that the degree of dispersion evidently increased as the MAPP/organoclay ratio increased, regardless of the total clay concentration. High concentration of MAPP leads to better clay dispersion and adhesion resulting in increase in the modulus, as seen in Figure 16. The XRD of the composite prepared with HMA with α = 3 (Figure 9) showed broader and reduced intensity of the clay also proving the presence of increased adhesion and higher number of delaminated clay layers. The improved adhesion due to MAPP may be explained in terms of the imide bond formation between the nucleophilic ammonium/phosphonium groups of the clay and MA groups. Organoclay surfaces exist in an acid–base equilibrium, which is ready to react as a nucleophile with the carbonyl groups of the MAPP. 34 These interactions increase the adhesion. In the case of HMA organoclay, this enhanced interaction may be attributed to the increase in the degree of crystallinity of the composites observed by the addition of organoclay. Figure 5 shows that percentage crystallinity of PP increased from 39 to 46.76 in the ternary composite of the HMA organoclay when α was equal to 3.
XRD of the composites showed limited dispersion of the organoclay in the matrix, and the tensile strength of the ternary composites were lower compared with that of pure PP, most probably due to weak interfacial attraction between the silicate layers and the matrix. Also, the organoclays of this study were prepared from unpurified bentonite samples that contain nonclay impurities. In the absence of impurities and flaws, the mechanical properties of the nanocomposites depend on the degree of forces that binds the atoms together. However, in this case, the RB used contains contaminants such as quartz, silica, feldspar, sodium carbonate, and chlorite. 42 These particles probably caused poor adhesion of the polymeric matrix to the clay particles, forming holes at the interphase, that act as defects and stress concentrators in the matrix assisting the failure mechanism. 42 Also, the plasticization effect that may arise from the unintercalated alkyl surfactants may cause contamination. As also mentioned previously in the XRD part, modifiers with bulky alkyl tails (TKA and HMA) may locate between the particles of clay layers and not in between the galleries inducing plasticization effect at the processing temperature and decreasing the composite properties.
Samples prepared with the commercial organoclay Cloisite 25A show higher modulus compared with the one prepared with modified organoclays prepared in this study as well as pure PP. Comparison of the structure of Cloisite 25A with HMA shows that Cloisite 25A with two long alkyl tails may exhibit higher intercalation owing to its higher d-spacing. Also, highly pure MMT used in Cloisite 25A would result in smaller degree of stress concentration effect.
Conclusions
Bentonite was effectively modified with QP and QA salts to produce organoclays that can be used in PLSN. Surfactants with different alkyl tails were used in order to see their effects in the powdery organoclay form and in nanocomposites with a PP matrix. XRD, TGA, and FTIR analyses confirmed the intercalation of surfactant molecules through the MMT layers. XRD showed increased basal spacings of the bentonite after modification indicating intercalation. FTIR analysis of the organoclays showed new absorption bands differing from the FTIR of the unmodified bentonite, also implying intercalation. Although XRD of the composites indicated microcomposite formation, TEM images of the same materials showed that there were intercalated organoclay regions with HMA and TKA organoclays in the PP matrix. Results showed that the organoclays produced with the bentonite in this study can be used to obtain nanocomposites with PP matrix.
Footnotes
Acknowledgement
The authors thank Karakaya Bentonit A.S., Ankara, Turkey for the bentonite.
Funding
The experimental part of this research work was financially supported by Middle East Technical University under contract number BAP-2006-07-02-00-01.
