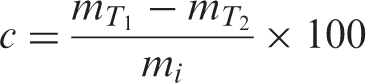Abstract
Thermogravimetric analysis is always used to study the pyrolysis process of polymers and composites and can reflect their thermal degradation properties. In the article, an exfoliated nanocomposite was synthesized by vinyl acetate and organic montmorillonite through different processes. Linear macromolecular chains of polyvinyl acetate were formed in the layers of organic montmorillonite, and no chemical bond existed between them, but only physical effect. Organic montmorillonite was exfoliated into layers or sheets of nanoparticles, randomly dispersing in the matrix of polyvinyl acetate, so the different synthesis processes and the addition of organic montmorillonite had little effect on the structure of the exfoliated nanocomposite, but some certain effect on the pyrolysis properties. The pyrolysis was found to consist of eight phases, and those of polyvinyl acetate and the exfoliated nanocomposite were similar. The presence of organic montmorillonite had no obvious effect on the pyrolysis temperature, but mainly delayed the thermal degradation process. In addition, their pyrolysis kinetics was firstly analyzed by the Agrawal integral equation. The kinetic parameters, including the kinetics mechanism function, the activation energy, the frequency factor, the reaction order, the reaction rate constant, the form factor of heat flow curve, and the kinetic compensation effect equation, were obtained in detail.
Introduction
Generally, materials and their products are used at a certain environmental temperature, and in their use, they will reflect to different temperatures and show their different thermal physical properties, these properties are usually called asthermal properties or pyrolysis properties. The pyrolysis properties of polymers or composites are very important in their forming and application processes, so theyshould be investigated in detail. Thermogravimetric analysis (TGA) is always used to do this. Recently, montmorillonite (MMT) or organic montmorillonite (OMMT) has been applied in many polymers, such as polyacrylate ester,1,2 poly (methyl methacrylate),3,4 polyurethane,5,6 epoxy,7,8 polycarbonate,9,10 polyethylene,11,12 polyvinyl acetate (PVAc)13–15 etc., to improve their chemical, physical, and mechanical properties, but few of them are specifically related to the pyrolysis properties, and till now, no systematic study on this subject has appeared.
Therefore, in the article, based on our previous work about PVAc,16–19 OMMT,20–24 and some exfoliated nanocomposites of PVAc and OMMT,25–27 a new OMMT was prepared by MMT and octadecyl trimethyl ammonium bromide (STAB) according to refs 20 and 21, the intercalated nanocomposite of OMMT was obtained. STAB is a long alkyl chain quaternary ammonium salts, its cations arrange with the half paraffin-type molecular in the layers of MMT. Then to simplify the synthesis process, under the conventional conditions in the lab, with no ultrasonic dispersion and no radiation, PVAc-OMMT was polymerized by VAc and OMMT through different synthesis processes. In the synthesis, OMMT was 2% of VAc25–27; it was a reasonable amount and would bring good effect on the pyrolysis properties. PVAc and PVAc-OMMT were homogeneous amorphous linear polymers; their structure was mainly investigated by X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), and dispersion in water, then their TGA tests were also carried out, their pyrolysis processes were studied, their kinetics was also firstly analyzed by the Agrawal integral equation in detail.
Experimental
Samples
A total of 1.00 g of OMMT was immersed into 25.00 g of VAc for 24 h. It was then mixed with 70.00 g of 10% polyvinyl alcohol solution, 0.50 g of alkylphenol polyoxyethylene (10) ether, 3.75 g of 10% ammonium persulfate solution, 6.25 g of sodium lauryl sulfate, and 250 g (200 g, 150 g, 100 g, 50 g) of water while stirring vigorously for 8 h. When the mixture became a homogeneous emulsion, the temperature rose to 70°C. While stirring vigorously, 3.75 g of 10% ammonium persulfate solution and 25.00 g of VAc were gradually added into the homogeneous emulsion in 6 h (5 h, 4 h, 3 h, 2 h) for polymerization. Subsequently, the temperature rose to 85–90°C, the emulsion further polymerized for 0.5–1 h. After polymerization, when the temperature dropped to below 50°C, the emulsion was washed bywarm deionized water for several times to clean the residue of emulsifier, stabilization agent, initiator, and others, so the pure polymer was obtained with a high yield of 80%–90%. Then 4.00 g of ethanol, 3.00 g of water, 0.30 g of sodium benzoate, 0.18 g of sodium bicarbonate, and 6.00 g of Di-n-butyl phthalate were added, and finally PVAc-OMMT-A (PVAc-OMMT-B, PVAc-OMMT-C, PVAc-OMMT-D, PVAc-OMMT-E) was obtained. Here, we used A, B, C, D, and E to respectively correspond to the different synthesis processes with different polymerization time and different proportion of water. PVAc-A, PVAc-B, PVAc-C, PVAc-D, and PVAc-E were also synthesized by the same processes as PVAc-OMMT, butwithout OMMT. From our previous work,26,27 their molecular weights were 1.0 × 10 2 (PVAc-A), 5.0 × 10 2 (PVAc-B), 4.0 × 10 3 (PVAc-C), 8.1 × 10 3 (PVAc-D), 1.4 × 10 4 (PVAc-E), 3.8 × 102 (PVAc-OMMT-A), 1.2 × 103 (PVAc-OMMT-B) 8.1 × 103 (PVAc-OMMT-C), 2.0 × 104 (PVAc-OMMT-D), and 4.0 × 10 4 (PVAc-OMMT-E).
XRD
The samples were tested by DX-2000 of X-ray diffraction under Cu Kα of radiating, 40 kV of tube voltage, 30 mA of tube current, scanning from 0.5° to 15° at a rate of 0.02°ċs−1, and λ = 1.54184 Å of wavelength. The large value of d(001) forMMT or OMMT is calculated by Bragg law of λ = 2dsinθ, where λ is the wavelength representing the intensity of X-ray, d is the distance between layers of MMT, and θ is the diffraction angle.
FTIR
The samples mixed with kalium bromatum powder were pressed into plates, and then tested by the NICOLET 380 of FTIR.
Dispersion
About 3 to 5 drops of PVAc or PVAc-OMMT were added to 20 mL of water in a glass dish (diameter 90 mm). When they became homogeneous, the dispersion could be observed with water as reference.
TGA
PVAc and PVAc-OMMT were tested by NETZSCH TG209 F1 under 7–10 mg ofsample, 25–800°C at a heating rate of 10°Cċmin−1, and a nitrogen flow rate of 60–80 mLċmin−1. In the pyrolysis, the phase thermal weight loss (c, %) is calculated by:
Results and discussion
Structure
In the article, different nanocomposites of PVAc-OMMT were prepared throughdifferent synthesis processes, all of them were exfoliated nanocomposites shown in Figure 1. From the figure, we can see that the diffraction peaks of MMTand OMMT appeared, their 2θ were 7.00° and 4.18°, their d(001) were 1.263 nm and 2.114 nm, respectively.20,21 After organic modification, OMMT was an intercalated nanocomposite; its XRD pattern still had the diffraction peak. Butfor PVAc-OMMT, as shown in the figure, including PVAc-OMMT-A, PVAc-OMMT-B, PVAc-OMMT-C, PVAc-OMMT-D, and PVAc-OMMT-E, no diffraction peaks appeared within 0.5–5° of θ on their XRD patterns.28,29 In the emulsion polymerization, on the basis of the first intercalation of STAB into MMT, the linear macromolecular chains of PVAc were formed in the layers of OMMT, the formed chains made the second intercalation. The distance between MMT layers was greatly stretched, the original ordered crystal layers were completely destroyed, MMT layers were fully exfoliated, and they independently dispersed in the emulsion with nanometer-thick layers. In other words, OMMT wasgreatly exfoliated into layers or sheets of nanoparticles, and they randomly dispersed in the matrix of PVAc. The exfoliated nanocomposite of PVAc-OMMT was obtained.
X-ray diffraction (XRD) patterns.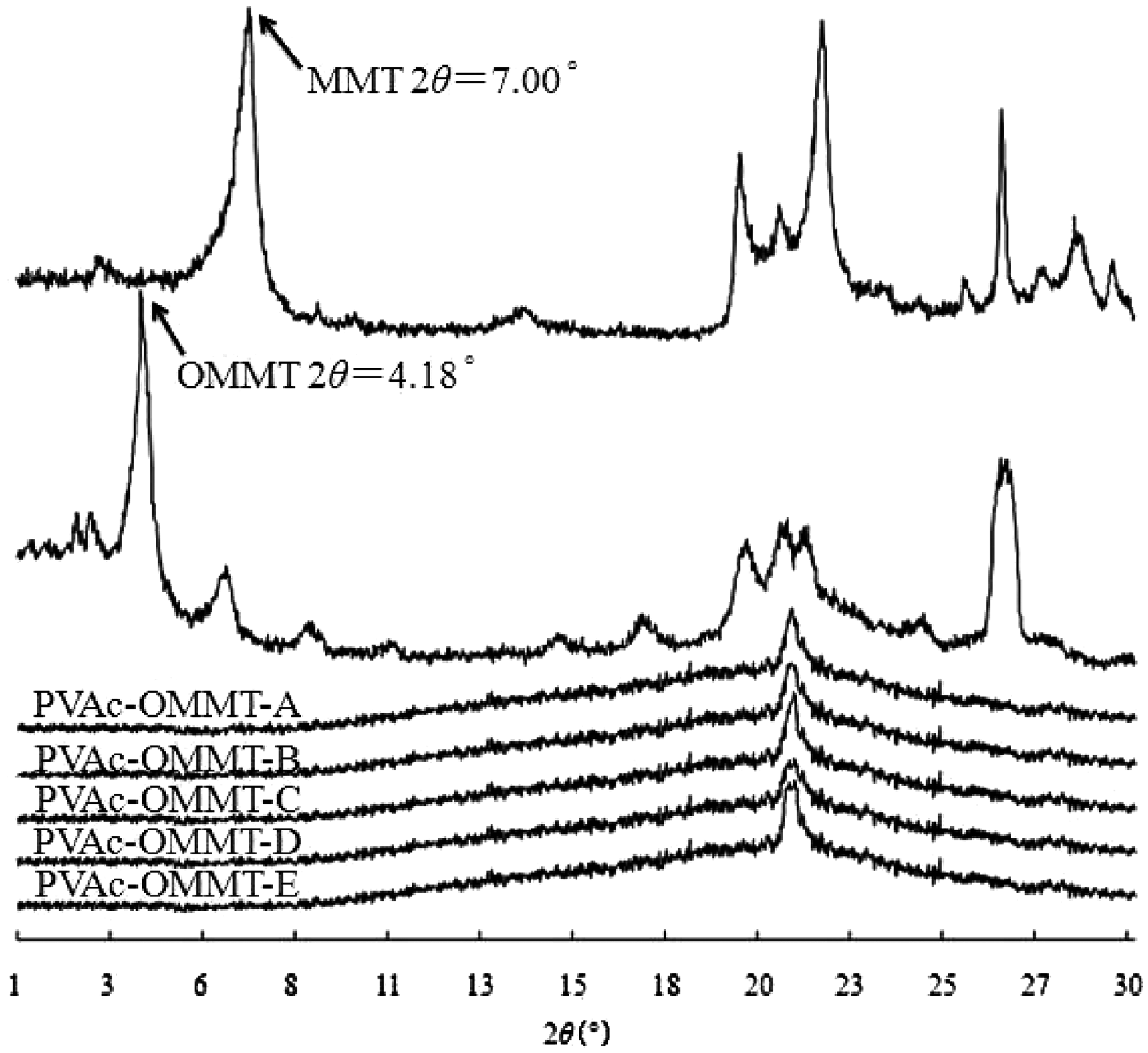
Then, to further study the interaction between PVAc and OMMT, they werealso tested by FTIR. The results are shown in Figure 2. From the figure, we can see that no chemical bond was found between OMMT and PVAc, but only physical adsorptions or the secondary bonds. The absorption bands of PVAc-OMMT were stacked by that of OMMT and PVAc. Neither were new absorption bands formed nor did existing absorption bands disappear. The different synthesis processes had no effect on the interaction. FTIR spectra of PVAc-A to PVAc-E were the same, and those of PVAc-OMMT were the same, too. In PVAc-OMMT, PVAc and OMMT were connected or absorbed together by physical effect. It was precisely because of these physical adsorptions or the secondary bonds; OMMT could still keep its good stability in the relatively harsh emulsion intercalation process and guarantee its structural continuity and stability.
Fourier transform infrared spectroscopy (FTIR) spectra.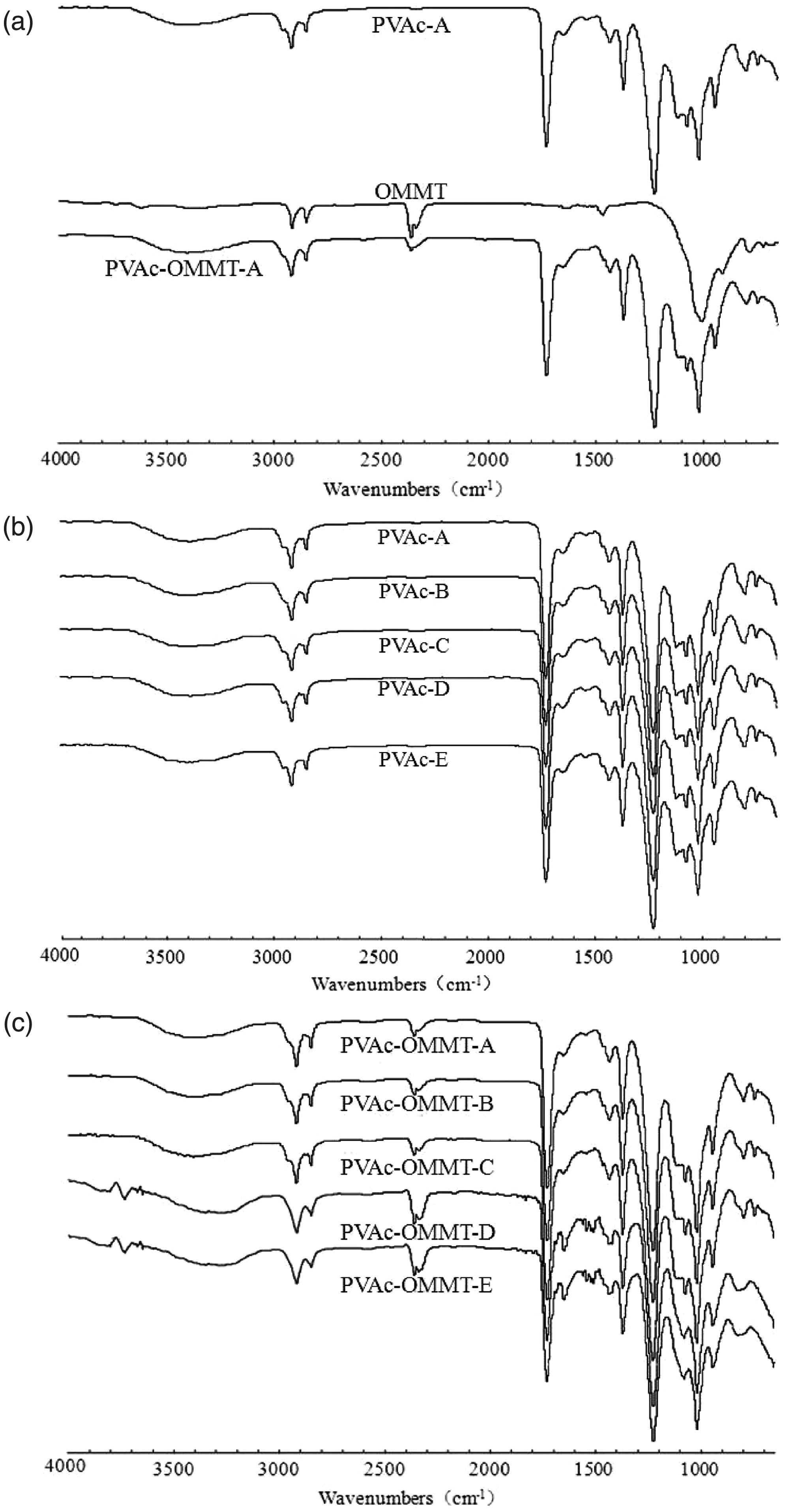
Besides these, their dispersion in water was also investigated. From the visual, all PVAc emulsions, including PVAc-A, PVAc-B, PVAc-C, PVAc-D, and PVAc-E, and some PVAc-OMMT emulsions, including PVAc-OMMT-A, PVAc-OMMT-B, and PVAc-OMMT-C, were the same. They were viscous, milk-white, homogeneous, fine emulsions. All had no coarse particles, no foreign bodies, and no delamination. Butfor PVAc-OMMT-D and PVAc-OMMT-E, they were greatly different. There appeared delamination, especially for PVAc-OMMT-E, its delamination was very severe. Actually, PVAc-OMMT-D was also viscous, milk-white, homogeneous, fine emulsions, but it had many large or small coarse particles. These particles deposited at the bottom, formed the white precipitate. Oppositely, for PVAc-OMMT-E it was not the case. Its delamination was very obvious. The top part was colorless, transparent liquid, like water, not viscous. The bottom part was viscous, milk-white floccus. Moreover, when some were dropped in water, their dispersion was also different to that shown in Figure 3. Comparing with water, the figure shows no coarse particles were found in the dispersion of all PVAc, PVAc-OMMT-A, PVAc-OMMT-B, and PVAc-OMMT-C, their dispersion was much better. But for PVAc-OMMT-D and PVAc-OMMT-E, their dispersion was another case, which was apparently different from others. As shown in the figure, the top sample of PVAc-OMMT-D dispersed in water appeared to be more viscous, its color was much deeper than others, like milk. The bottom sample dispersing in water showed some coarse particles, some were small and some were large; other part liquid was also like milk. From PVAc-A to PVAc-E and from PVAc-OMMT-A to PVAc-OMMT-D, we can know that the dispersion color became much deeper one than one;this phenomenon may be caused by the different synthesis processes that resulted in different emulsions with different viscosity and different molecular weight. Of course, PVAc-OMMT-E was an exception; it was completely different from all the others. Its dispersion with top sample was colorless, transparent liquid, like water, while there were white floccus and coarse particles in the dispersion withbottom sample. This may be also caused by its special synthesis process.
Dispersion.
The dispersion was also further studied in our previous work25–27 by transmission electron microscope. We found that the particle diameter of PVAc was from 250 nm to 500 nm while OMMT’s was from 50 nm to 100 nm, they randomly dispersed together, and the smaller OMMT particles were adsorbed around thelarger PVAc particles to form the ‘Strawberry’ structure, ‘Swallow’ structure, ‘Tactoids’ structure, ‘Half Core-Shell’ structure, or ‘Similar Core-Shell’ structure. And a kind of emulsion often contained one or more than one structures, this was due to most particles individually, randomly dispersed, and many of them were absorbed, gathered, or aggregated together to form different structures, and they all showed a good dispersion in the matrix of PVAc.
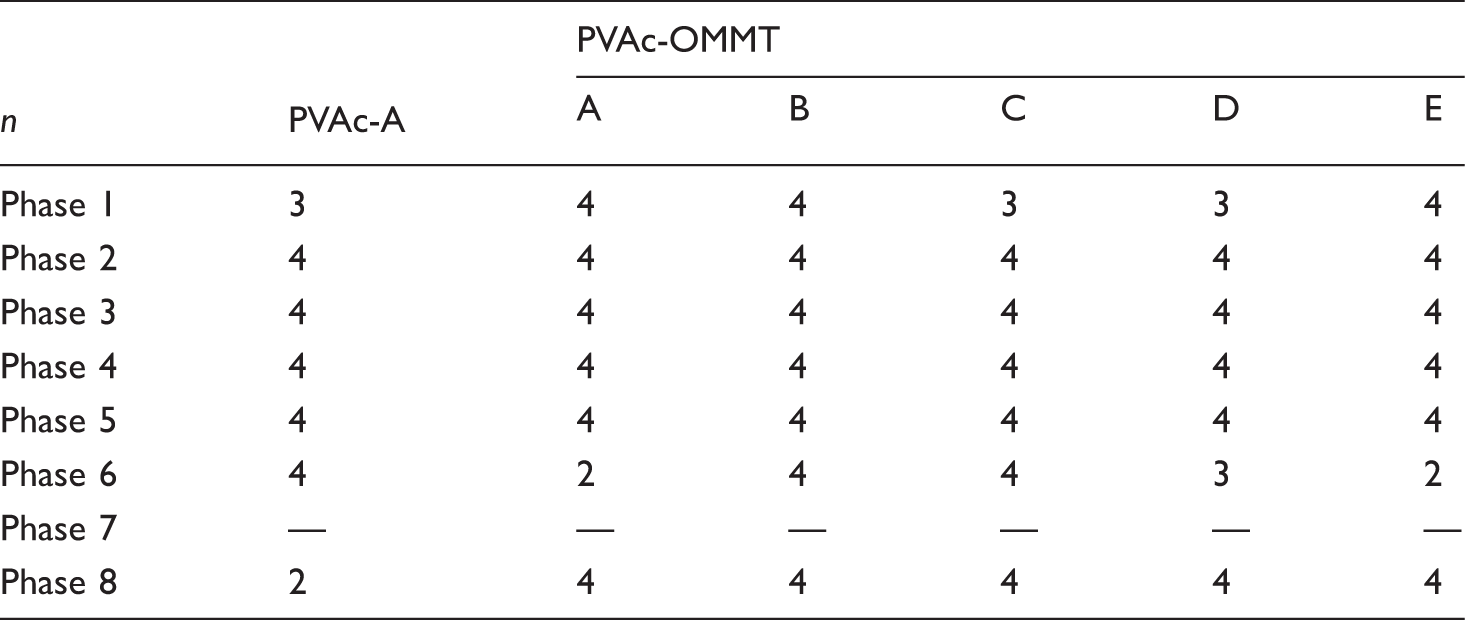
Pyrolysis properties
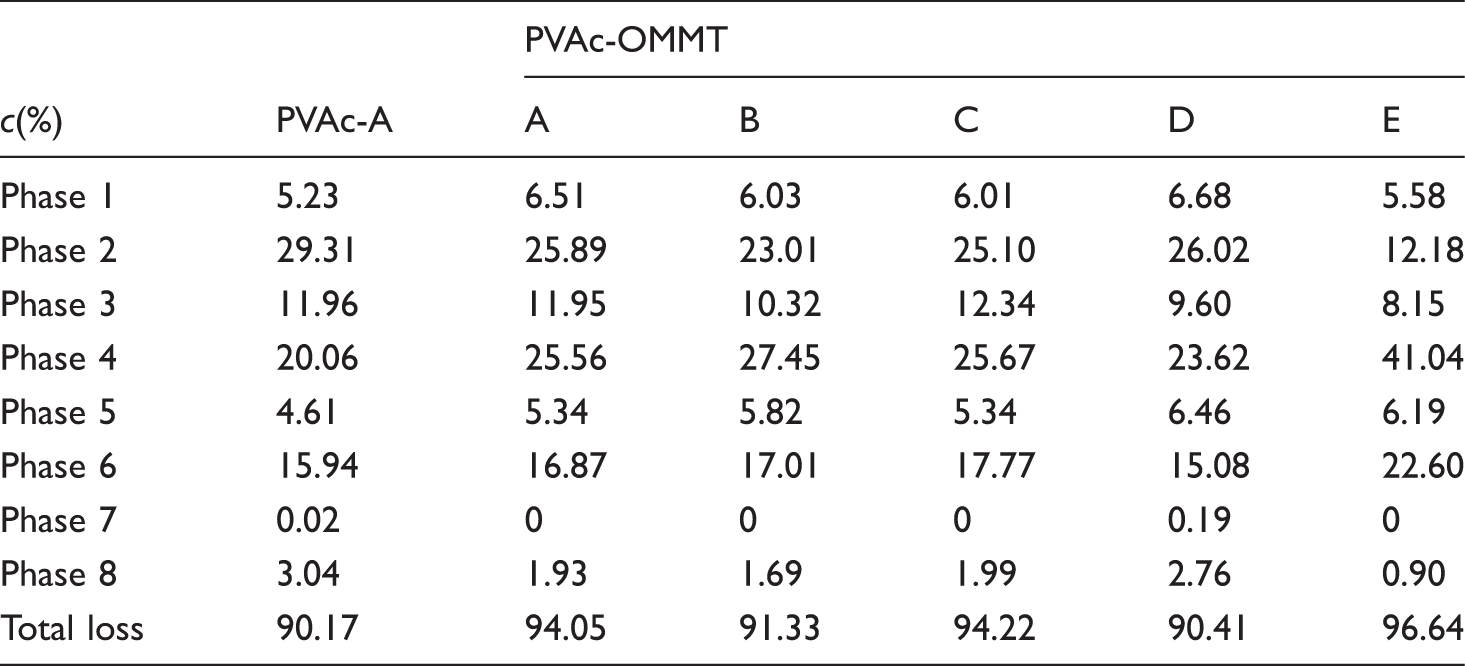
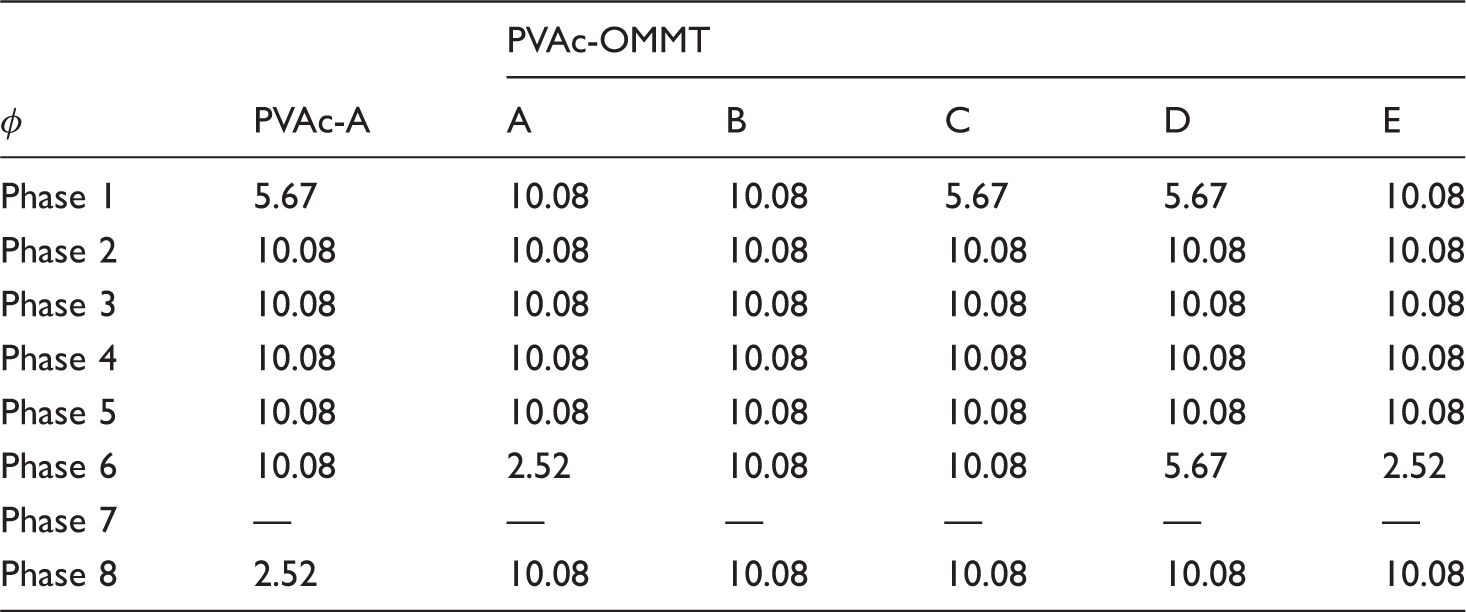
From the results described above, we can see that the different synthesis processes had little effect on the structure of PVAc and PVAc-OMMT; PVAc-OMMT wasan exfoliated nanocomposite, from PVAc-A to PVAc-E and from PVAc-OMMT-A to PVAc-OMMT-E, their FTIR spectra were almost the same. Butfor the dispersion in water, all PVAc, PVAc-OMMT-A, PVAc-OMMT-B, and PVAc-OMMT-C, their dispersion was much better, while those of PVAc-OMMT-D and PVAc-OMMT-E were great different. Therefore, in this part, on behalf of all PVAc, PVAc-A was chosen and tested by TGA, and all PVAc-OMMT, including PVAc-OMM-A, PVAc-OMMT-B, PVAc-OMMT-C, PVAc-OMMT-D, and PVAc-OMMT-E were tested by TGA. Their TGA spectra are shown in Figure 4. In the figure, each one contains three curves; the curve of weight loss is the thermogravimetric (TG) curve, the curve of weight loss rate is the derivative thermogravimetric (DTG) curve, and the curve of heat flow is the differential thermal analysis (DTA) curve. They will describe the pyrolysis process in detail. As shown in Figure 4, the pyrolysis was found to consist of eight phases, phase 1 from 25°C to 150°C, phase 2 from 150°C to 230°C, phase 3 from 230°C to 290°C, phase 4 from 290°C to 350°C, phase 5 from 350°C to 410°C, phase 6 from 410°C to 510°C, phase 7 from 510°C to 710°C, and phase 8 from 710°C to 800°C. TG curves of PVAc and PVAc-OMMT were similar. From Table 1, we can see that all the total weight loss was more than 90%, only less than 10% was left, these burnt residue may be some fire-retardant materials, such as MMT, which is a mineral, it cannot be burnt, as well as some other inorganic additives added in the polymerization. We can also find out that the pyrolysis mainly occurred in phase 2, phase 3, phase 4, and phase 6. In these phases, small molecules and macromolecules all were completely burnt, that is to say that with the temperature rising, PVAc as the polymer matrix was gradually pyrolysized, and finally was completely burnt. For PVAc-OMMT, besides the pyrolysis of PVAc, it also included the pyrolysis of OMMT, the organic content of OMMT here we used was 40%–60%, and OMMT was mainly pyrolysized during 300–400°C.22–24 Even so, from our above description about structure, PVAc and PVAc-OMMT were synthesized through five different processes, but their structures were similar, so their TG curves and weight loss were bound to be similar.
Thermogravimetric analysis (TGA) curves. Phase weight loss.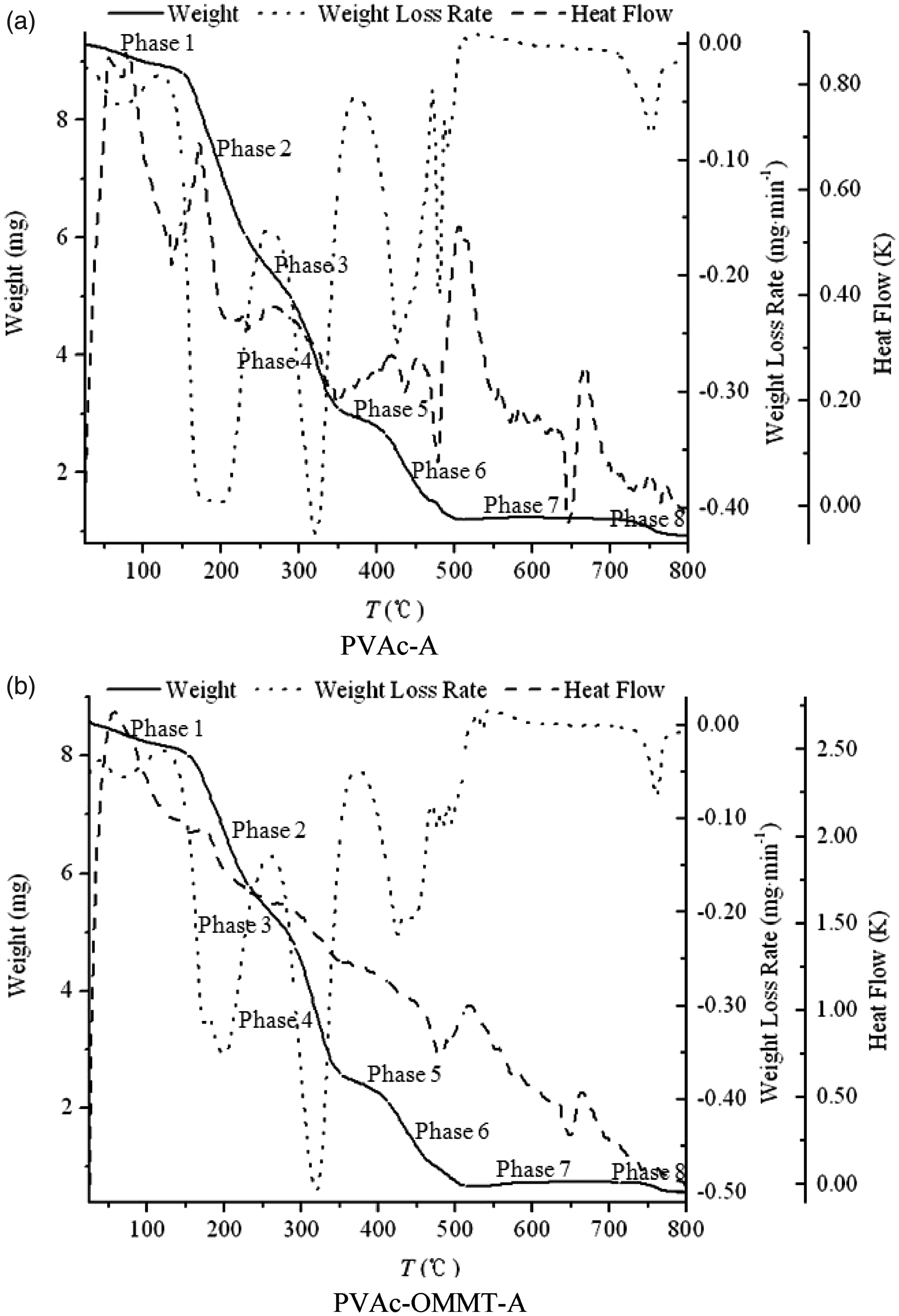
Pyrolysis valley characteristics of differential thermal analysis (DTG) curve.
As far as the pyrolysis of PVAc and PVAc-OMMT were concerned, their pyrolysis kinetics needed to be further studied. For the kinetics, the mechanism function is always expressed as:
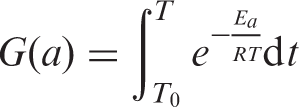
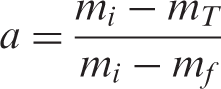
The Agrawal approximate equation is
34
:
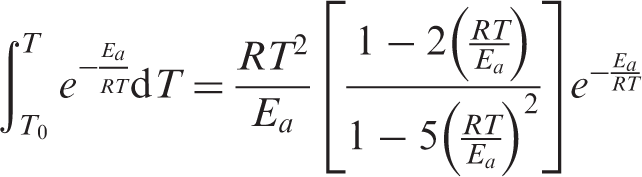
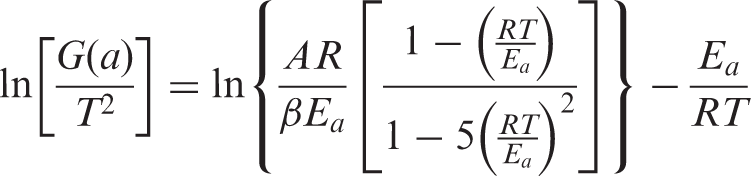
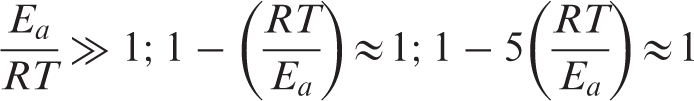
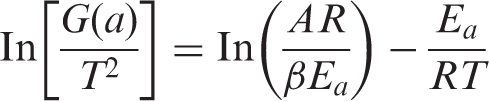
The relationship between ln
Reaction order.
Pyrolysis kinetics.
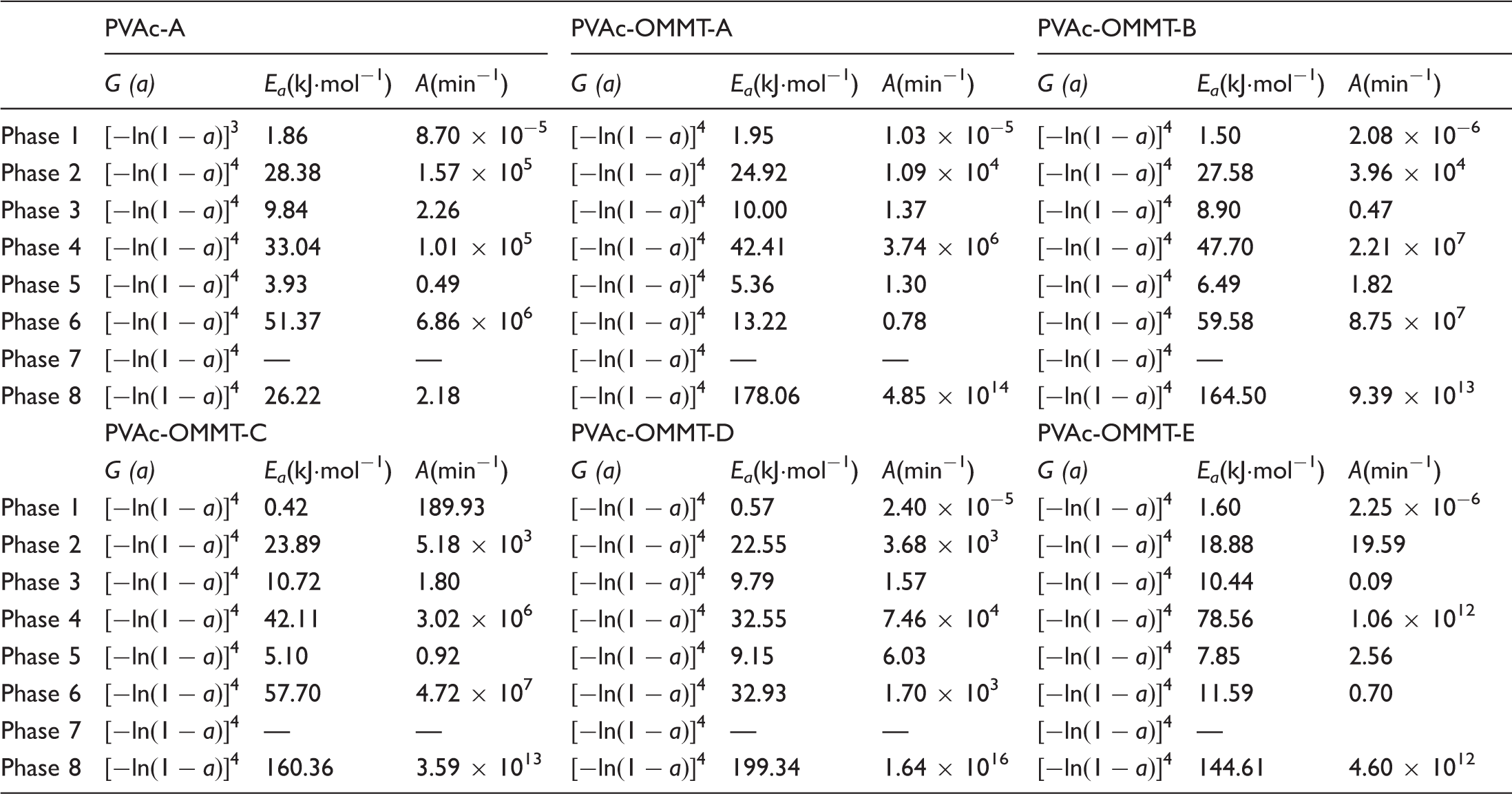
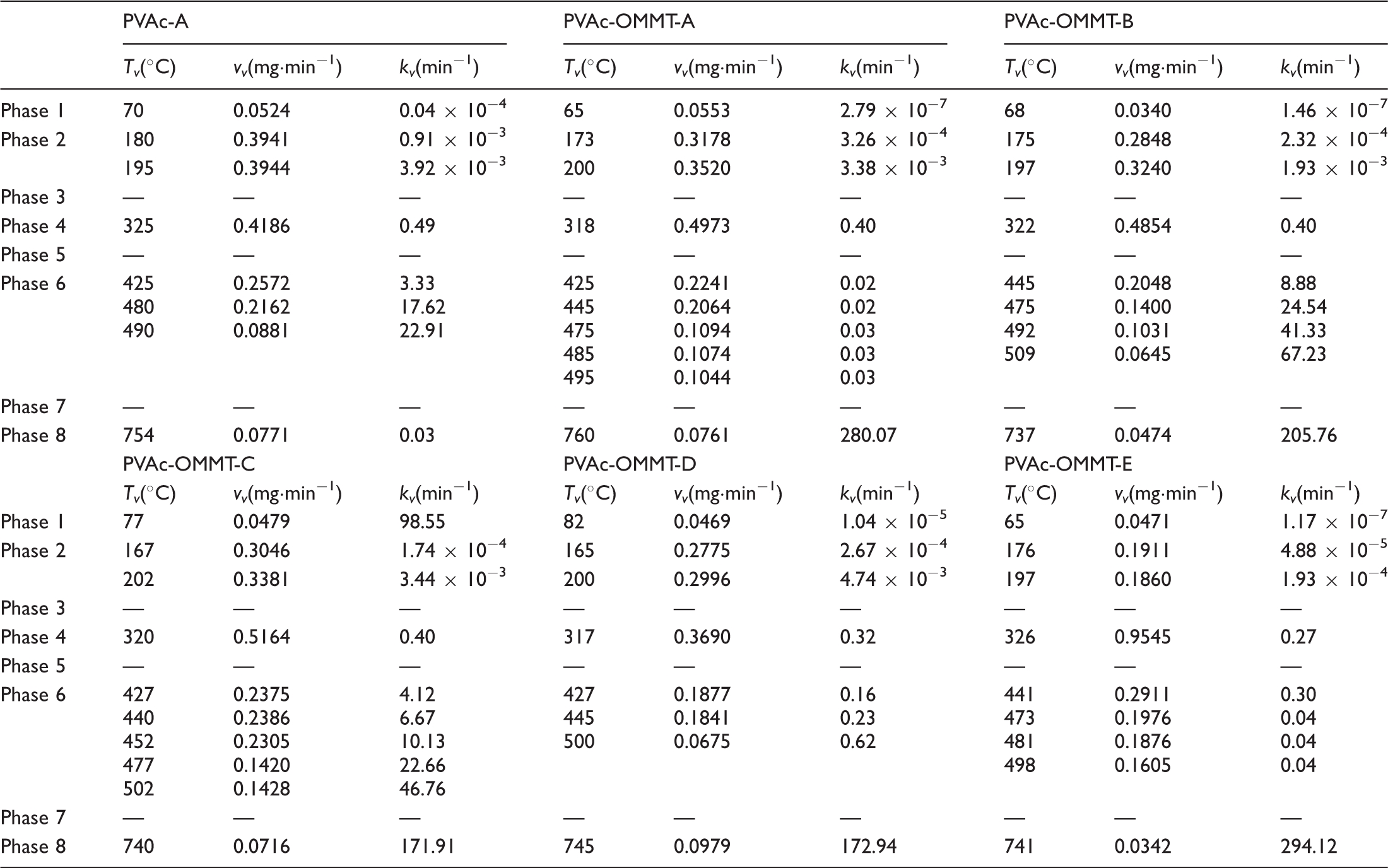
In the pyrolysis kinetics, the reaction rate constant (k) is always to quantitatively explain the reaction rates, it is usually related to A, Ea, and T, and calculated by Arrhenius equation:
k=A
k in each pyrolysis phase of PVAc-A and PVAc-OMMT was obtained as shown in Table 2. The variation trend of k was similar to those of Tv and vv. From phase 1 to phase 8, k was gradually getting greater and greater, from some minimum k to some maximum k. As we know, k is closely related to the reaction temperature, reaction medium (or solvent), catalyst, and so on, even to the shape and characteristics of reactors, so during the pyrolysis, with the pyrolysis temperature rising, k increased.
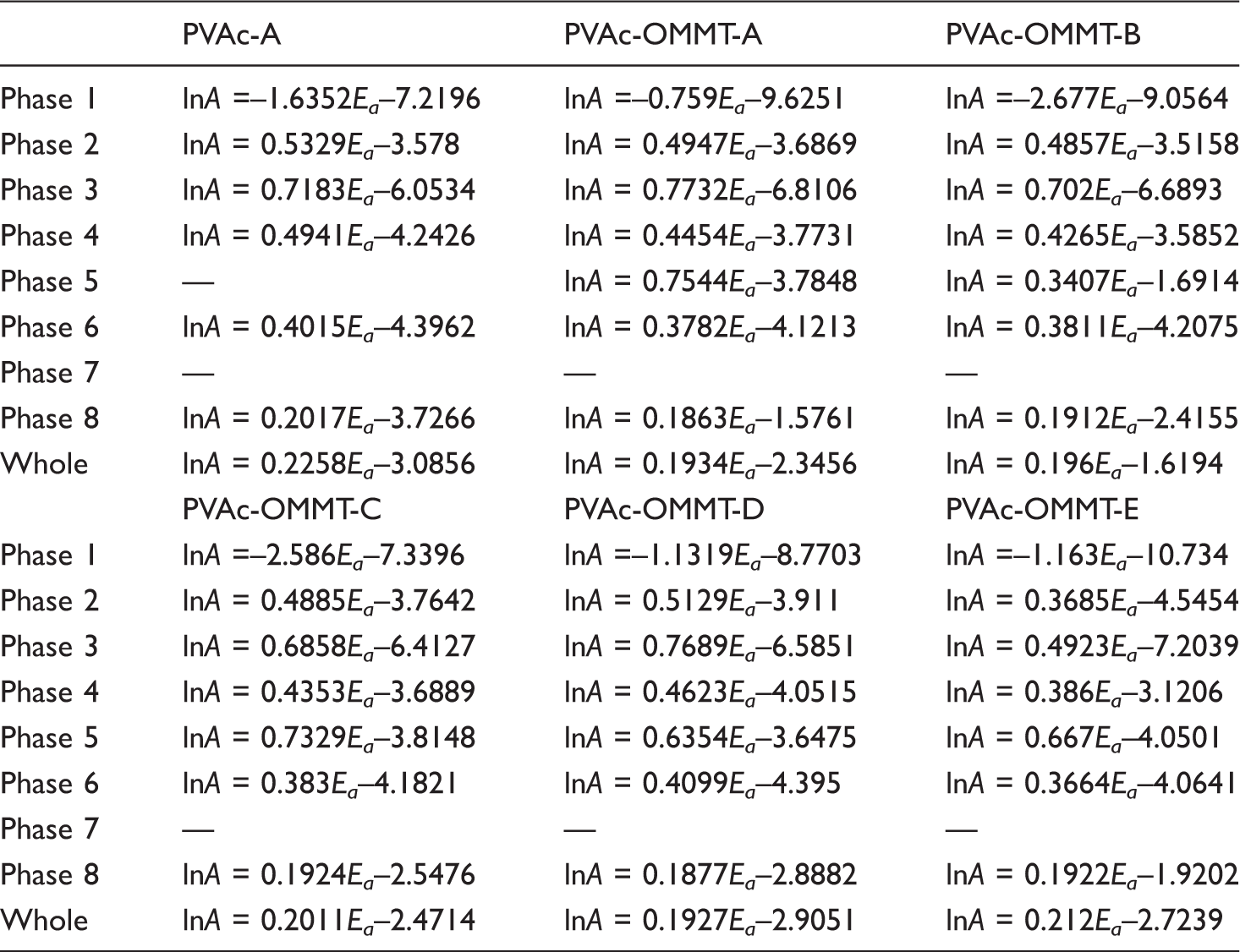
In addition, the linear relationship between lnA and Ea is named as the kinetic compensation effect. It is a main characteristic in kinetics, explaining that A partly compensates to the change of Ea. The equation 35 is:
lnA = KEa +Q
Kinetic compensation effect equations.
Finally, the form factor of DTA curve (ϕ) should also be taken into account, it indicates the symmetry of DTA curve, the symmetry will be worse when n decreases. ϕ is defined by Kissinger
36
as shown in Figure 5; it is only closely related to n as:
ϕ of differential thermal analysis (DTA) curve.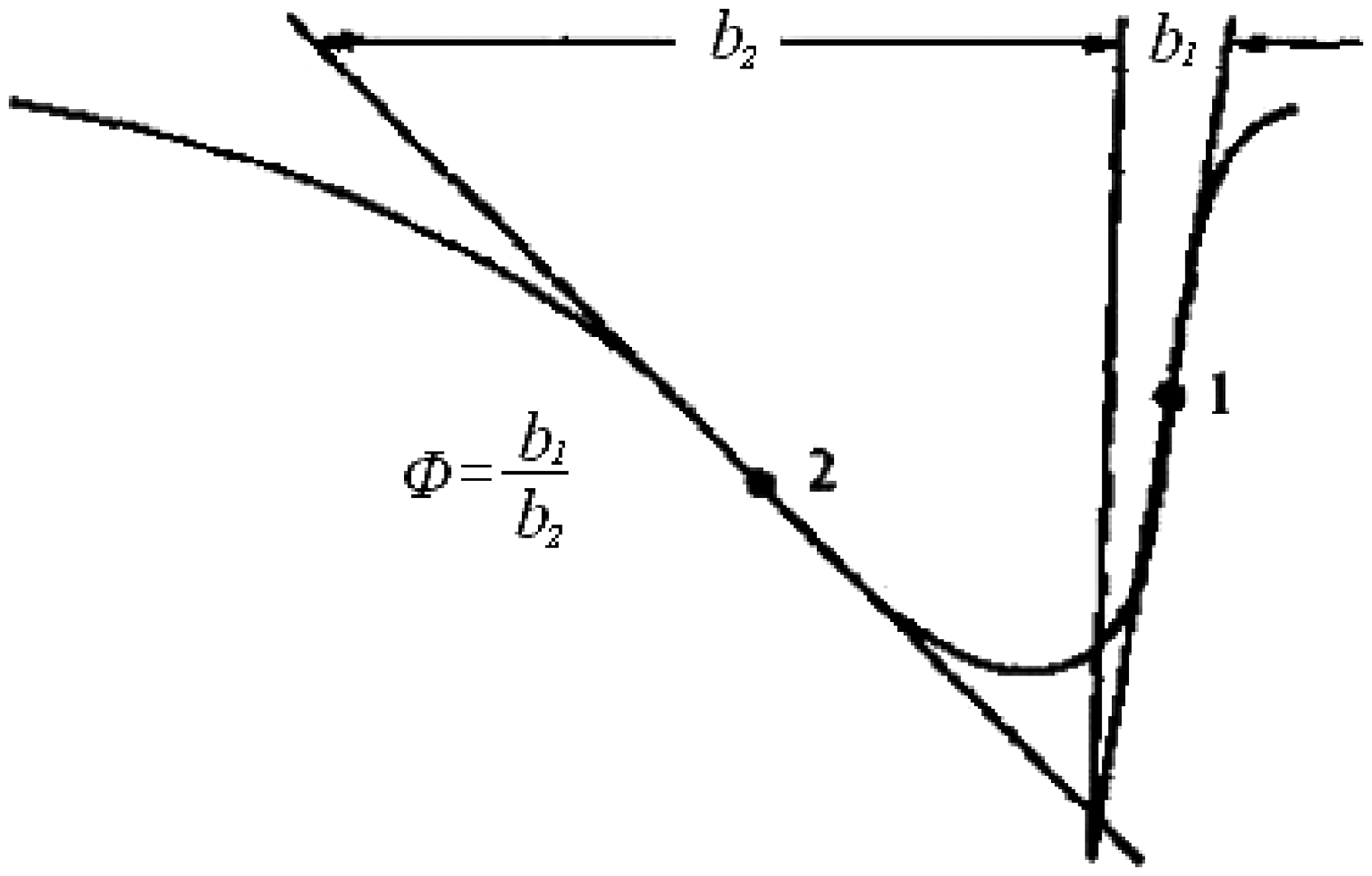
ϕ = 0.63n 2
ϕ of differential thermal analysis (DTA) curve.
Conclusions
In the article, an exfoliated nanocomposite was prepared by VAc and OMMT through different processes. Linear macromolecular chains of PVAc were formedin the layers of OMMT, and no chemical bond existed between OMMT and PVAc, but only physical effect. OMMT was exfoliated into layers or sheets ofnanoparticles, randomly dispersing in the matrix of PVAc, so the different synthesis processes and the addition of OMMT had little effect on the structure of PVAc-OMMT, but some certain effect on the pyrolysis properties. The pyrolysis was found to consist of eight phases, and those of PVAc and PVAc-OMMT weresimilar. The presence of OMMT had no obvious effect on the pyrolysis temperature, but mainly delayed the thermal degradation process. In addition, their pyrolysis kinetics was firstly analyzed by the Agrawal integral equation. The kinetic parameters, including G (a), Ea, A, n, k, ϕ, and the kinetic compensation effect equation, were obtained in detail.
Footnotes
Acknowledgement
The authors are grateful for the financial support of the Program for New Century ExcellentTalents in University of the Ministry of Education of China (NCET-06-0825).