Abstract
Low-density polyethylene (LDPE) nanocomposites with alumina as filler were prepared by melt intercalation process by using Haake Rheomix at 125°C for 12 min followed by compression moulding. Prior to blending of the alumina with LDPE, alumina particles were dispersed by using ultrasonication process. Surfactant was used to modify the alumina surface in order to enhance the interaction between alumina and LDPE. The melting temperature and crystallinity of nanocomposites were investigated by differential scanning calorimetry (DSC). Studies on dynamic mechanical analysis (DMA) were made to follow the changes in dynamic mechanical properties upon addition of nanoalumina to LDPE. Thermal stability of the nanocomposites was studied by using thermal gravimetric analysis (TGA). It was found that the crystallinity and melting temperature of LDPE/alumina decreased with the increase in alumina content. The results on TGA and DMA analysis revealed that the thermal stability of LDPE improved with the addition of alumina particle.
Introduction
Polymer composites are widely used in applications such as transportation, construction, electronics, and consumer products. Developing nanocomposites based on polymers and nanoscale fillers has been an attractive approach to achieve good properties. There are various nanoscale fillers that have been used to enhance the mechanical and thermal properties. 1 Organic–inorganic nanocomposite materials have been regarded as new generation of high performance materials since they combine the advantages of the inorganic materials (rigidity, high stability) and the organic polymers (flexibility, dielectric, ductility, and processability). 2
The great stiffness and hardness of ceramics can sometimes be combined with the toughness of polymers or metals by making composites. The properties of low-density polyethylene (LDPE) and alumina was opposite with each other, with the blending of both materials with expectation that produce a material balance in the properties. In the existed technology, nanotechnology has great potential application to improve the properties of existing materials or develop new materials with unique properties. So, high stiffness alumina was chosen as filler in the LDPE to maximize the properties of material produce.
The synthesis of nano α-Al2O3 was carried out with the simultaneous addition of sodium bis-2-ethylhexyl sulfosuccinate [Na(AOT)] and 1/3-benzened disulfonic acid disodium salt (SDBS) and rising the stirring time, to prevent extreme grain growth and aggregation of nanoparticles. This is a multistep process that involves the transformation of the aluminum alkoxide to an aluminum hydroxide [Al(OH)3] followed by the dehydration to form an oxo-hydroxide aluminum intermediate, AlO(OH) (bohemite), that will form the predecessor in the sol–gel process.
Thermal properties of the materials were significant for the fabrication temperature. In particular, thermal stability was considered as an important factor playing role in the nanocomposite structure and morphology formation. If the processing temperature is higher than the thermal stability of organic component, the decomposition will occur which can lead to variations between filler and polymer interaction. Thus, determination of onset temperature of degradation and stability of the polymer is essential. 3 Various investigation of the thermal stability of nanocomposites based on polymer matrix was done; one can see from these investigations that the incorporation of nano-sized mineral materials will affect the thermal stability of nanocomposites. 4
Sahebian et al. 5 reported that nano-sized calcium carbonate in high-density polyethylene (HDPE)/CaCO3 nanocomposites presents a significant effect on crystallinity, crystallization rates, melting point, and heat of melting of HDPE. Such a behavior was also observed by using SiO2 and montmorillonite (MMT) where the crystallization rates in both nanocomposites are higher compared with neat HDPE, but the degree of crystallinity was reduced. In view of this, it has been reported that nanoparticles can increase the crystallization rate of polymers that may cause a reduction on degree of crystallinity. 6
The dynamic modulus value and damping factor were used to quantify interfacial interaction in composites. Increased dynamic modulus values and low damping factor show the improved interactions between filler and the matrix. The higher the peak damping factor, the greater the degree of molecular mobility and the better the damping characteristics. 7 Higher damping factor is indicative of a material that has a high, nonelastic strain component, while a low value indicates one that is more elastic.
In this article, thermal properties of LDPE/alumina nanocomposites were investigated. Thermal gravimetric analysis (TGA) was performed to examine the effect of alumina on the thermal stability of LDPE nanocomposites. The effects of alumina on the melting point of LDPE nanocomposites have been studied through differential scanning calorimetry (DSC).
Experiment
Materials
LDPE resin, grade TITANLENE®LDI 305YY, was supplied by Titan Group Sdn Bhd with density of 0.923 g/cm3. The melt flow index (MFI) was 6.0 g/10 min when conducted at 2.16 kg load and temperature at 190°C according to ASTM D1238. Sodium dodecylbenzenesulfonate (SDBS) in chemical grade, an anionic surfactant, was obtained from MERCK.
Alumina Nanoparticle Preparation
Alumina nanoparticles were synthesized by using sol–gel method with particle sizes between 10 and 40 nm. Aluminum isopropoxide Al(OC3H7)3 (Merck, German), aluminum nitrate Al(NO3)3.9H2O (Merck, German), Na(AOT) (Merck, German), and SDBS (Merck, German) were used as raw materials to prepare nano alpha-Al2O3. The starting solution was prepared through using aluminum isopropoxide and 0.5 M aluminum nitrate aqueous solution as a solvent. The molar ratio of alumina sol aluminum isopropoxide (ALP)/aluminum nitrate nonahydrate (ALN) was 3:1. The solutions were continuously stirred for 48 h. Then, the Na(AOT) and SDBS were added by adjusting the molar ratio between alkoxide and surfactants to be 0.1 and stirred for 1 h. It is expected that this time be the optimal time for the addition of surfactant and it occurs prior to the onset of the nucleation and growth step. The solutions were heated up to 60°C and stirred constantly for evaporation process. Viscosity and color changed as the sol turned into a transparent stick gel. The gels were then heat treated at 90°C for 8 h. Dried gels were calcined at temperature 1200°C. Finally, the calcined powders were crushed by using mortar and pestle. Phase identifications were performed by X-ray diffraction (XRD) using nickel filtered CuKα radiation in the range of 2θ = 10–80° with a scanning speed of 58/min. The XRD analysis in Figure 1 showed the most stable phase, α-Al2O3 occurred dominantly at 1200°C. The observation indicated that completion of the most stable phase, a-alumina occurs at this temperature. Although the thermodynamically stable α-Al2O3 phase can be obtained through a sequence of topotactic and reconstructive transformations (i.e., boehmite → γ →δ →θ→α), the morphology remains unchanged and the final products have the same shape as the initial phases. Alumina was dispersed using ultrasonic bath (Branson 1510) for 1 h at room temperature with the present of surfactant used for dispersion of alumina and surface treatment. Transmission electron microscopy (TEM) was performed to ensure the alumina produced were nano-sized and well dispersed (Figure 2).
XRD spectra of aluminum oxide obtained under various conditions: (a) XRD spectrum of bohemite formed without surface stabilizer before calcined, (b) XRD spectrum of bohemite formed after the addition of Na(AOT) before calcined, (c) XRD spectrum of bohemite, formed after the addition of SDBS before calcined, (d) XRD spectrum of α-Al2O3, formed without surface stabilizer, after calcined at 1200°C, (e) XRD spectrum of α-Al2O3, formed after the addition of Na(AOT) after calcined at 1200°C and (f) XRD spectrum of α-Al2O3, after the addition of SDBS after calcined at 1200°C. TEM image of dispersed nanoalumina particles.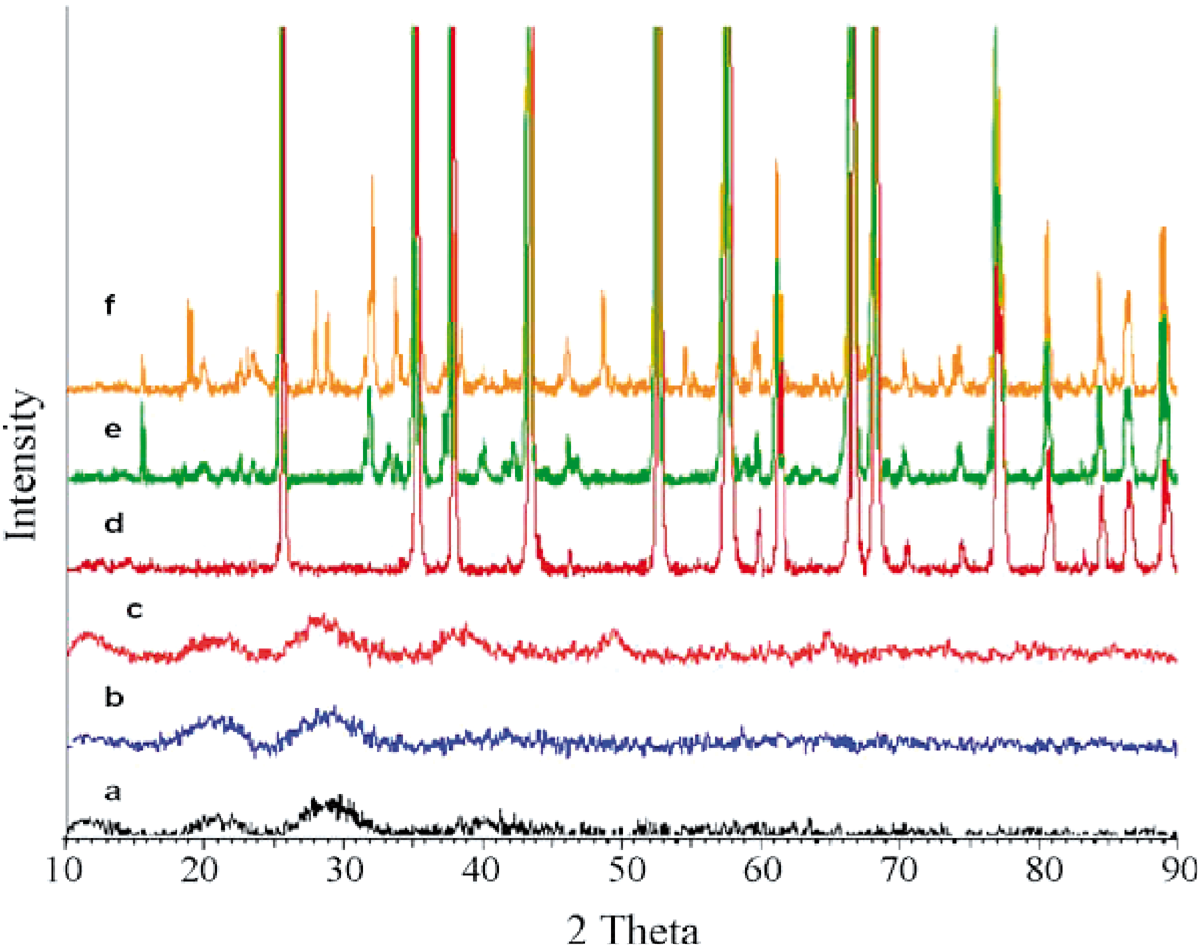

LDPE/Alumina Nanocomposites Preparations
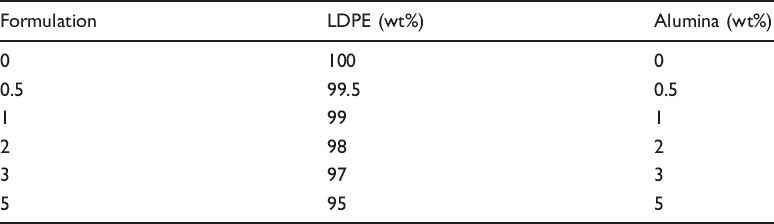
Formulation for LDPE/alumina nanocomposites.
Thermal Gravimetric Analysis
The thermal stability of alumina/LDPE nanocomposites and pure LDPE were investigated by TGA using a Perkin-Elmer TGA 7 thermal analyser under nitrogen flow (10 mL/min) from 30°C to 600°C at the heating rate of 10°C/min.
DSC Analysis
The influences of the alumina nanoparticles on the nonisothermal crystallization behavior of the alumina/LDPE nanocomposites were evaluated by using DSC. The specimens were excised from the extruded strands and each sample is between 5 and 10 mg. All DSC analyses were performed with Mettler Toledo DSC 823e under nitrogen atmosphere. Each sample was performed under same process, which is heated from 10°C to 150°C at a rate of 10°C/min for heating history, then cooled to 10°C at 10°C/min, and finally heated back to 150°C at 10°C/min. All the exothermic and endothermic curves were recorded. The crystallinity degree of the pure LDPE and the matrix of the alumina/LDPE composites can be calculated automatically using software at DSC, which was determined by integrating the heat flow from 40°C to 120°C and enter the heat of fusion of the purely crystalline form of LDPE, that is, 289.9 J/g. 8 The heats of crystallization ΔHc were determined by integrating cooling scans from 40°C to 120°C.
Dynamic Mechanical Analysis
The sample was analyzed by dynamic mechanical analysis (DMA) (TA Instrument Q800). Single cantilever method was used for the test and subjected to an oscillatory deformation with a frequency 1 Hz while being heated from −150°C to 70°C under controlled scan rates of 10°C/min, and temperature was maintained by using liquid nitrogen.
Results and Discussion
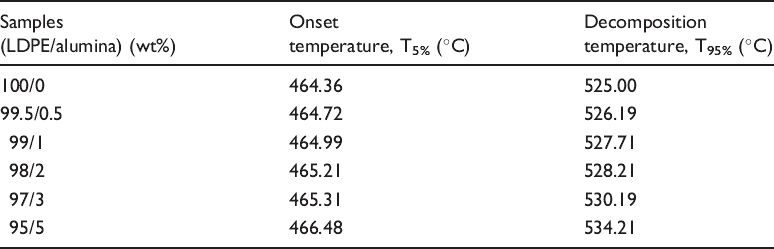
Onset and decomposition temperature of LDPE/alumina nanocomposites.
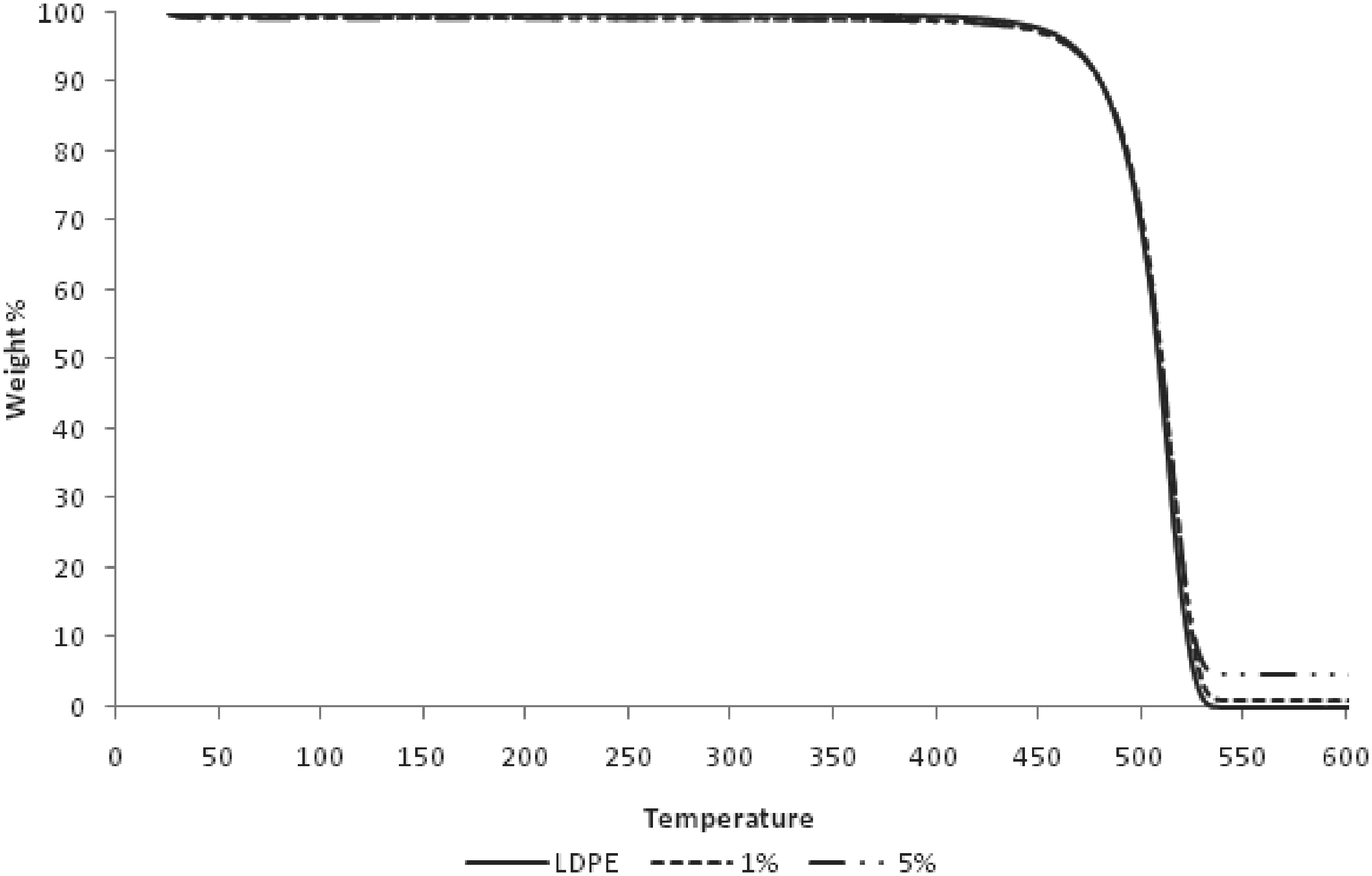
TGA curves for LDPE and LDPE/alumina nanocomposites.
It has been reported by other researchers 4 that the thermal stability of the polymer/nonmetal nanocomposites will be improved as the content of the nonmetal nanoparticles increases. This is because the inorganic material can prevent the heat to expand quickly and limits further degradation. 9 Table 2 shows that the thermal decomposition temperatures of the nanocomposites are higher than pure LDPE, and it increases gradually with the content of alumina nanoparticles in nanocomposites. The thermal decomposition temperature increased due to the retardant effect of the alumina nanoparticles in the LDPE. Thus, the diffusion of volatile decomposition products within the higher alumina particles content in nanocomposites hindered by the alumina nanoparticles was slower than that within the lower alumina particles content in nanocomposites. The onset temperature was reduced with the higher alumina particles content in nanocomposites, which explained by lower mass losses in sample with higher alumina particles content. It’s also proven by the higher char yield with the higher alumina particle in nanocomposites. In summary, the alumina has the barrier effect which would improve the thermal stability.
The crystallization and melting behavior are shown in Figure 4. The DSC melting curves of pure LDPE and LDPE/alumina show only one endothermic peak, with slight decrease in the melting peak temperature when alumina is added to LDPE. Percent crystallinity of sample was measured as the area under sample melting curve and compared with 100% crystalline polymer of the same sample. The melting point and the crystallinity of the LDPE/alumina nanocomposites were found to be lower than those of the pure polymer. The decreasing of crystallinity of the LDPE with the increase in nano alumina loading is believed to be caused by the nanoparticles that had disturbed the configuration of the matrix. It also indicates that the presence of alumina restricts the mobility of the polymer chains during crystallization.
DSC curves of heating for LDPE and LDPE/alumina nanocomposites.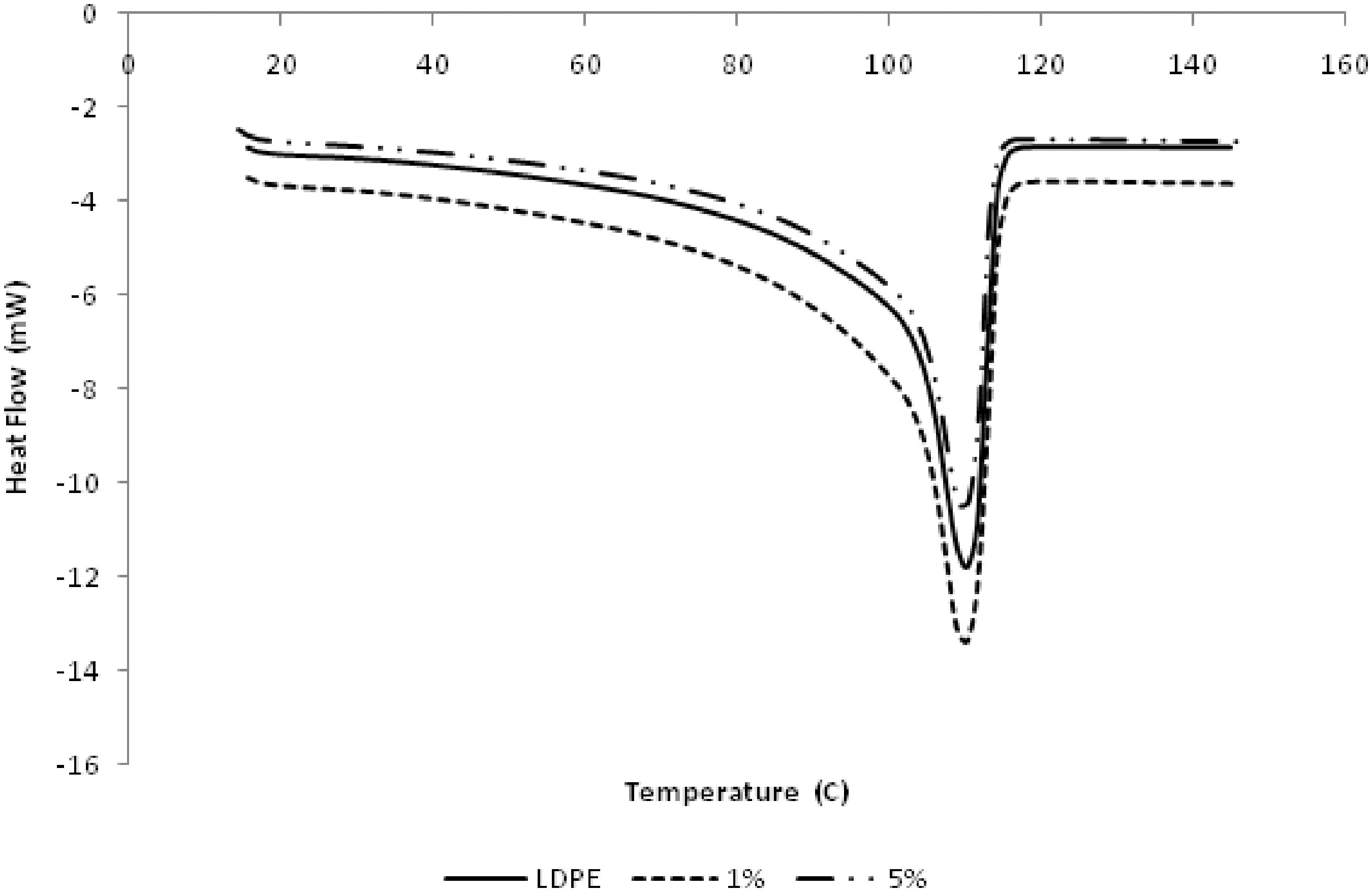
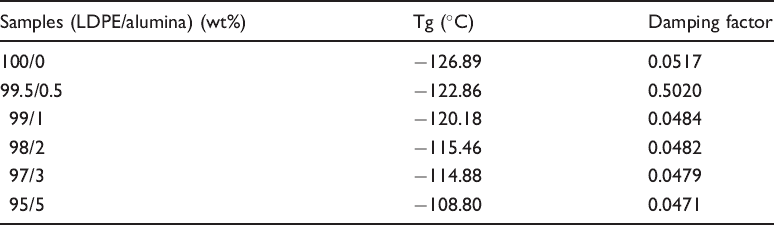
Tg and damping factor of LDPE and nanocomposites.
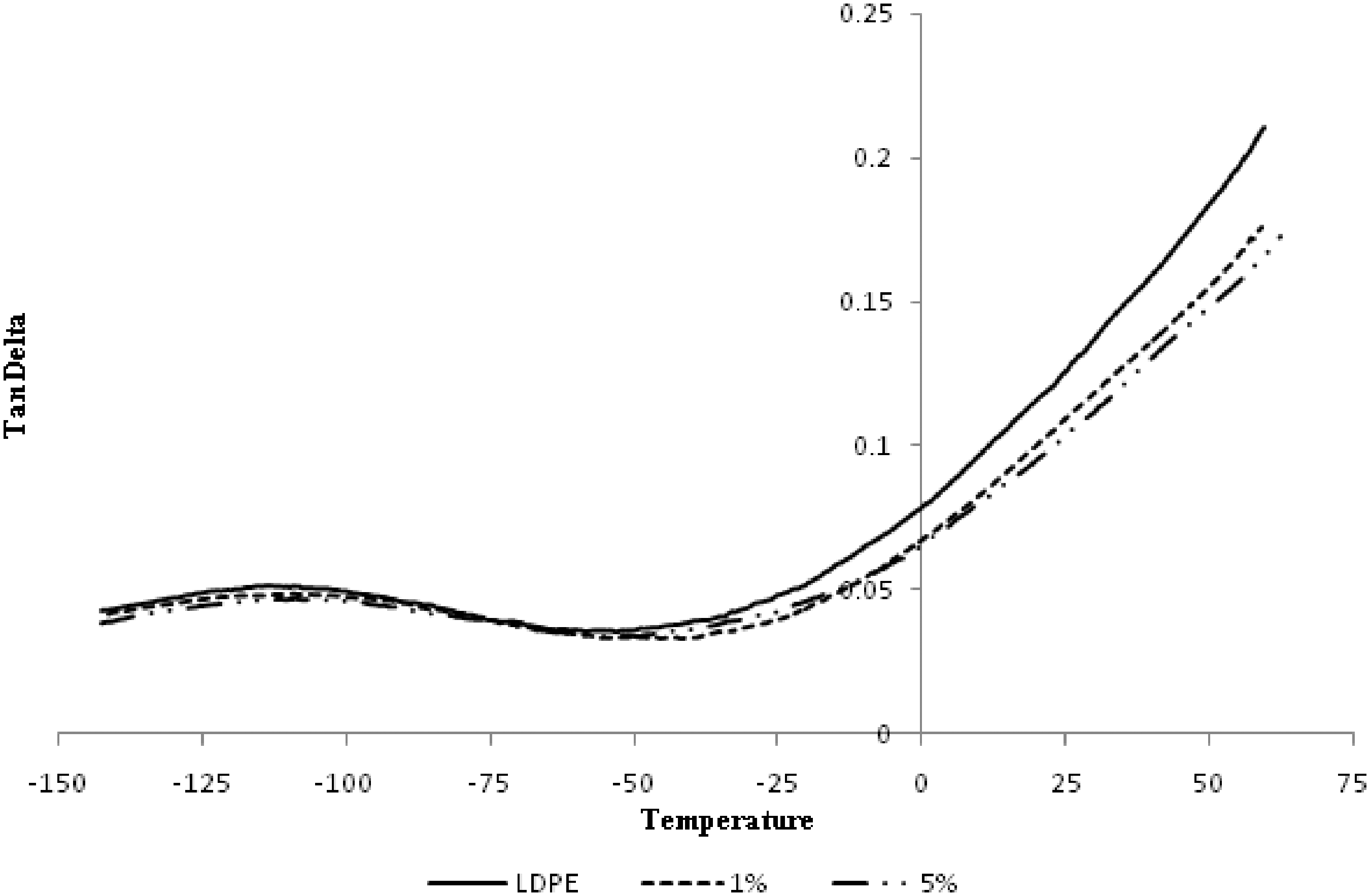
Damping factor (tan delta) of LDPE/alumina nanocomposites at different alumina percentages.
Figure 6 presents the temperature dependence storage modulus of LDPE at 0, 1, and 5% nano alumina loading. It shows that the storage modulus of the nanocomposites are lower than that of the neat LDPE at lower temperatures from −150 to about 20°C, while above 20°C the values of the storage modulus for all the nanocomposites are greater than LDPE.
Storage modulus of LDPE/alumina nanocomposites.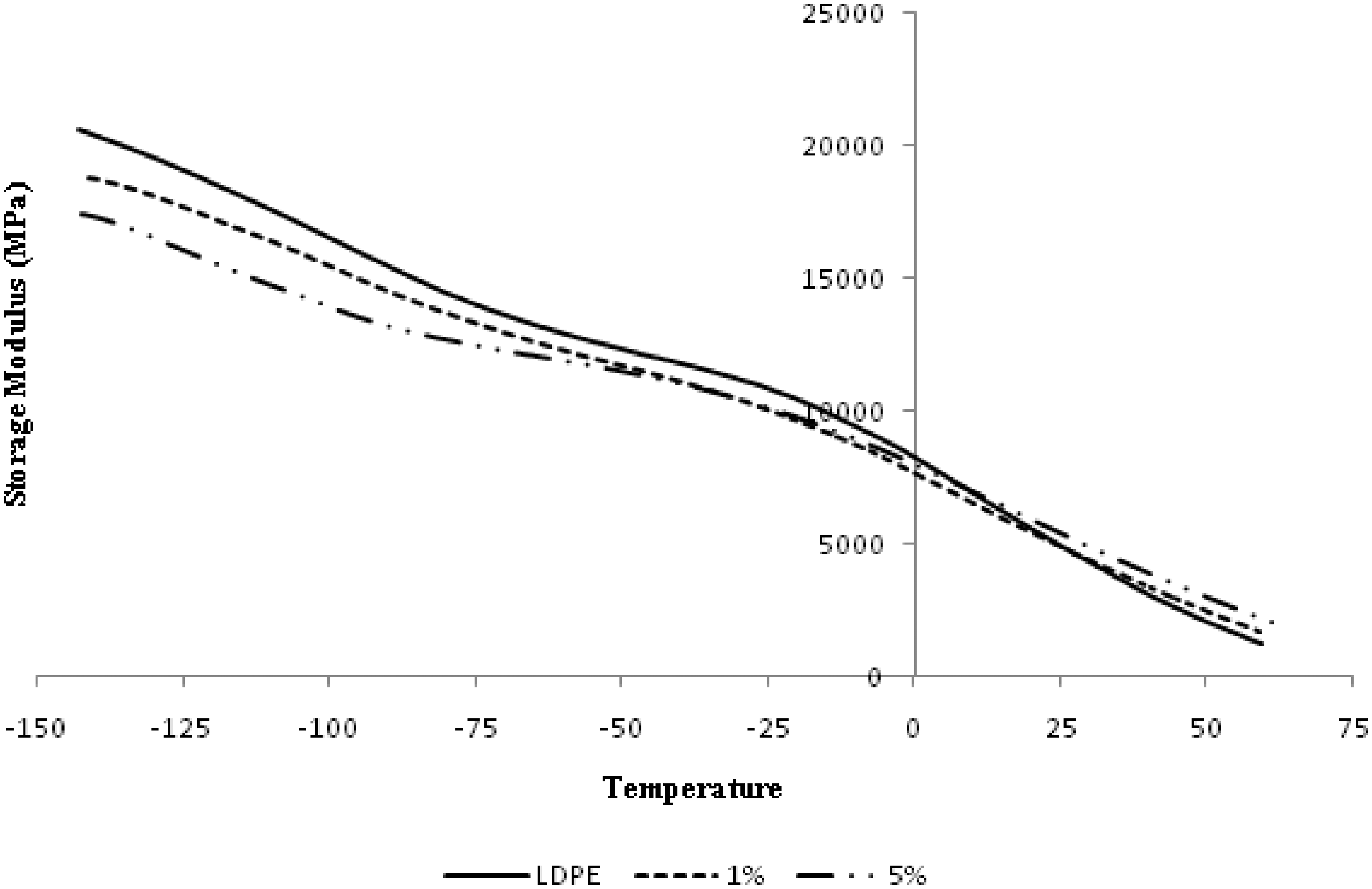
From Huang et al.’s
2
study, the temperature dependence storage modulus at low temperature range was affected by three factors. First, storage modulus and Tg increase in particle loading that were caused by the movement of macromolecular chains of interphase was restricted by nanocomposites. Second, voids and pores that were formed by nanoparticles agglomeration during sample preparation can decrease the storage modulus. Finally, the nanoparticles loading may decrease the crystallinity of the matrix due to the presence of imperfect crystal nucleated by the particles.
10
Figure 7 shows the SEM micrograph of the surface of LDPE/nano α-Al2O3 composites in the absence of dispersant. The agglomeration of nanoparticles in nanocomposites produces the voids in nanocomposites which may weaken the storage modulus.
SEM micrograph of the surface of LDPE/nano α-Al2O3 composites in the absence of dispersant with (a) 100 X and (b) 500 X of magnification.
At lower temperatures, the contribution of the interphase to the storage modulus is less than that of the others because of the high modulus of the matrix, and therefore the storage modulus of polymer matrix is higher than those of the composites. At higher temperature, the increase of the storage modulus for composites was due to the relaxation of LDPE.
Conclusions
Thermal properties of LDPE/alumina were studied using melt intercalation process. The addition of alumina nanoparticles enhanced the thermal stability of LDPE. The onset temperature of LDPE/alumina nanocomposites decreased with increasing filler contents. The crystallinity and melting temperature of LDPE/alumina nanocomposites decreased with the increase in alumina content. DMA studies indicate that the Tg of the LDPE shift to higher temperature with the addition of the alumina nanoparticle.
