Abstract
This article presents a comparative study on the effects of nano α-alumina on thermal degradation and dynamic mechanical properties of polypropylene (PP) nanocomposites. In this respect, two types of composites were prepared, with dispersant and without dispersant. Thermogravimetric analysis of both composites showed a drastic shift in the weight loss curve toward higher temperature, which led to reduction in the PP heat release rate in both composites. Differential scanning calorimetry results showed that the crystallinity of the nanocomposites was increased especially in the presence of dispersant. The storage modulus of the nanocomposites was found to be higher than that of pure PP, because nanofiller increased the stiffness of the nanocomposites. The glass transition temperatures of the nanocomposites were not significantly changed and damping factor of the nanocomposites was decreased.
Introduction
Polymeric nanocomposites have been recently established as an existing new class of materials filled with particles of at least one dimension in the nanosized range (1–100 nm). These nanocomposites exhibit superior mechanical performance and improve the barrier properties at very low loading levels compared to conventional filler composites. Improvement on mechanical properties, such as stiffness and toughness, dimensional, barrier and thermal properties, are usually observed.1,2 However, there are several key issues in the fabrication of polymer nanocomposites that should be carefully taken into account. These issues include uniform dispersion of nanoparticles against their agglomeration due to Vanderwaals bonding, alignment of nanofiller in the matrix, volume fraction, manufacturing rate and cost. 3 To improve the dispersion and interfacial properties of the polymer nanocomposites, there are at least two ways in practice. The first one is to treat the surface of nanoparticles and the second one is to modify the surface properties of polypropylene (PP). 4 Most of the oxide fillers are hydrophilic in nature, whereas many polymers including PP are hydrophobic. Dissimilarity of the surface chemistry between the filler and polymer contributes to the decrease in the tensity of filler–polymer systems. Widely used reactive coupling agents are silans and titanates categories. In general, titanium oxide can be represented as O=T=O, and this unsaturated characteristic is expected to react with hydroxyl groups on the surface of inorganic fillers to give hydrophobic polymer–compatible monomolecular layers. 5 Accordingly, they promote adhesion, improve dispersion, lower viscosity, and prevent phase separation. The introduction of active groups onto nanoalumina surfaces was achieved by the reaction shown in Figure 1.
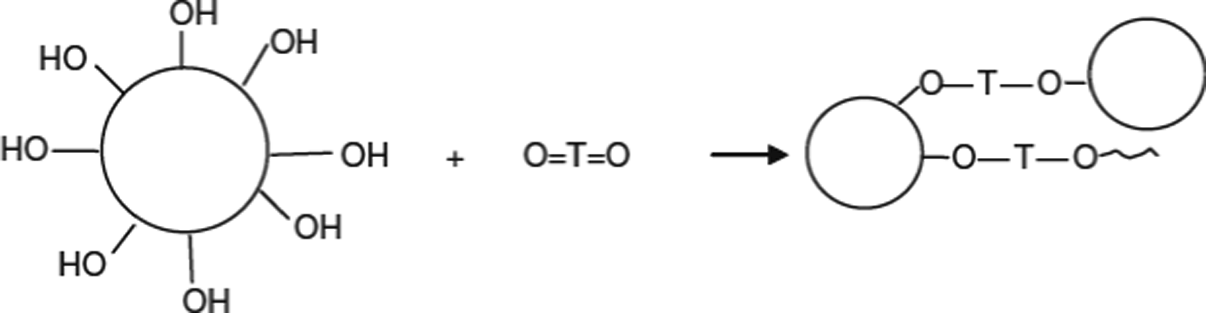
Schematic illustration of the reaction between titanium oxide and the hydroxyl groups of nanoalumina.
Among commodity thermoplastics, PP is characterized by light weight, low cost, easy processing, high mechanical strength, excellent chemical stability and excellent electrical properties. 6 In this research nano α-Al2O3 was chosen as the filler due to its special combined chemical and physical properties such as excellent resistance to heat and wear, high specific strength and good oxidation resistance. In composite systems, dynamic properties such as damping behavior and thermal properties such as thermal stability and thermal expansions and crystallization kinetics are very important factors that affect the quality of the final products. 4
In this work, sodium dodecyl benzenesulfonate was used as dispersant for the surface treatment of nano α-Al2O3.The effects of dispersant on the thermal and dynamic behavior of PP/nano α-Al2O3 composites were investigated.
Materials and methods
Materials
The PP grade 600G (melting temperature and melt flow rate are 165°C and 11 g/min, its density is 900 kg/cm3) used in this work was supplied by Petronas Polymers Marketing and Trading Division Malaysia. Nano α-Al2O3 with the average particle size of 20–30 nm and density of 3106 kg/m3 was used. These nanoparticles were made in the laboratory by the procedure discussed by Mirjalili et al. 7 Titanium dioxide (TiO2) powder with a minimum assay of 98% used as a coupling agent was supplied from Fisher Chemicals Sdn. Bhd., Malaysia. Sodium dodecylbenzene sulfonate (SDBS) purchased from Merck (Germany) was used as a dispersant.
Surface treatment of nano α-Al2O3
Ultrasonication (KQ2200DE Ultrasonic Cleanser, 100 W, Kunshan of Jiangsu Equipment Company, China) was used for preparation of miscellaneous aqueous nano-suspensions, which is a conventional method for dispersing the extremely entwined or aggregated nanoparticle samples. About 0.1 g alumina nanoparticle and water solution (99.8 g) with an anion surfactant (0.1 g SDBS) were directly mixed in a 150-ml beaker. The suspension was sonicated for 1 h and was dried at 80°C for 4 hours.
Preparation of PP/nano α-Al2O3 composites samples
All materials used for preparation of PP/nano α-Al2O3 were weighted and listed in the Table 1.
Formation of PP/nano α-Al2O3 composites
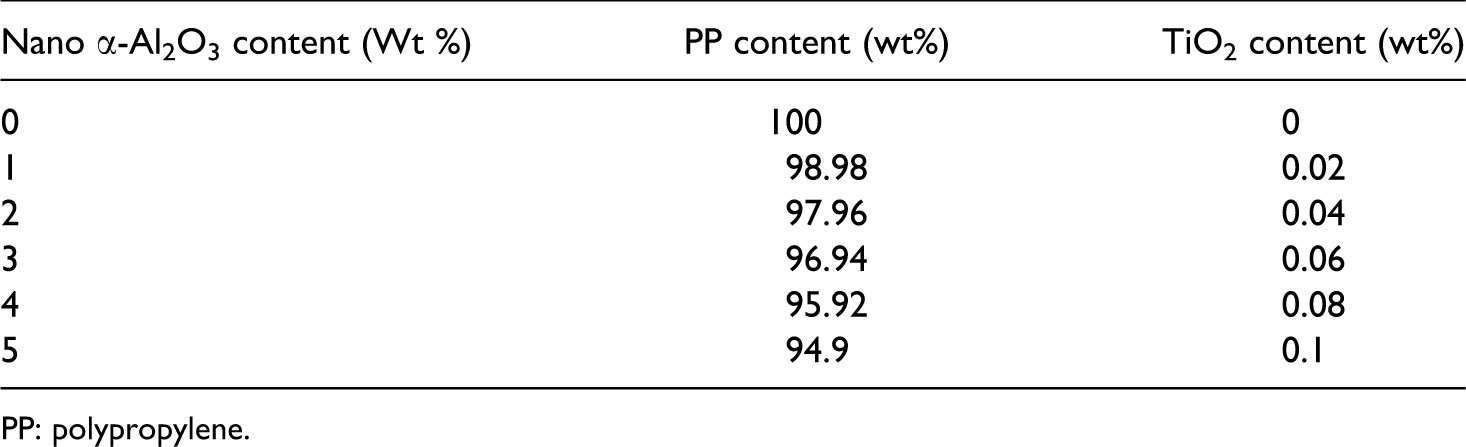
PP: polypropylene.
Predrying process was carried out on the nano α-Al2O3 at 80°C for 4 h. Existence of moisture in the filler inclined to cause defects such as voids, pinholes and so on, in the manufactured composites. The optimal content of coupling agent (TiO2) is 2 wt% of the filler. The systems with 2 wt% of TiO2 showed positive results. 5
Compounding and processing
The melt blending of PP and nano α-Al2O3 powder was carried out using Thermo Haake Poly Drive with RheomixR600/610 blending machine at 175°C, with rotor speed of 50 rotations per minute (rpm) with a total evacuated chamber capacity of 40 g. The first step of mixing involved the PP preheating for about 4 min. After preheating, the speed was maintained at 50 rpm for another 8 min to ensure uniform heat distribution throughout the batch. Then, nano alumina filler was included and 2 min later TiO2 powder was added. The rotor was stopped at the 12th min and the melted compound was taken out for sheeting. The melt compound of PP/nano α-Al2O3 of the size 15 × 15 cm were then formed by Hsinchu hot press machine. The compound was preheated for 2.5 min and hot pressed for another 3 min, under the pressure of 150 kg/cm2 at 180°C, with 10 times of compression bumping. The sheet obtained was directly cooled with the cold press for 2.5 min of cooling cycle.
Thermogravimetric analysis
The TGA analysis was performed by using Perkin Elmer TGA/SDTA 7 Model (Champaign, IL 61822). The weight of the sample used was about 10 mg. The samples were heated from 35°C to 600°C at a heating rate of 10°C/min in a dynamic inert nitrogen atmosphere.
Different scanning calorimetry analysis
The different scanning calorimetry analysis (DSC) was performed using the Mettler Toledo TA Instruments DSC 823 Model (Swindon, SN2 1EU). The weight of sample used was about 5–6 mg. The samples were encapsulated in aluminum pans and were heated from 35°C to 250°C at a heating rate of 10°C/min in a dynamic nitrogen atmosphere with a flow rate of 10 ml/min. Each sample was held for 1 min at 35°C to eliminate the trapped moisture content.
Dynamic mechanical analysis
The dynamic mechanical properties were measured by dynamic mechanical analyzer model Perkin Elmer TE. The samples from compression molded plaques were cut into rectangular shape with the dimension of ∼60 mm in length; ∼13 mm in width and ∼3 mm in thickness. The measurement was carried out using three points bending flexural test. The samples were subjected to an oscillating frequency of 1 Hz and oscillating amplitude of 10 μm in the temperature ranges of −30–80°C at a heating rate of 2°C/min.
Results and discussion
Thermal properties
It is essential to understand the thermal tolerance of polymer composites because of the variety of working environments of this composite in several engineering requests. The majority criterion for plastic material employed in the requests like electronic packaging, casing for electrical apparatus, external part of automotives and numerous more, is unpredictability and elevated temperature of materials which used in operational environment. Thermal stability of plastic materials can be improved by introducing fillers with the thermoplastic matrix. 8
Thermogravimetric analysis
TGA is used to measure the change in mass of a sample as a function of temperature or time or both. The samples were heated from 35°C to 600°C.
TGA curves of PP and PP/nano α-Al2O3 composites with different filler loading, both in the presence and absence of dispersants are given in Figure 2. Both the TGA curves exhibit an extreme shift of weight loss toward higher temperature. The heat initiated the degradation procedures as it breaks down the fillers and matrix structure by causing molecular chain ruptures or scission.
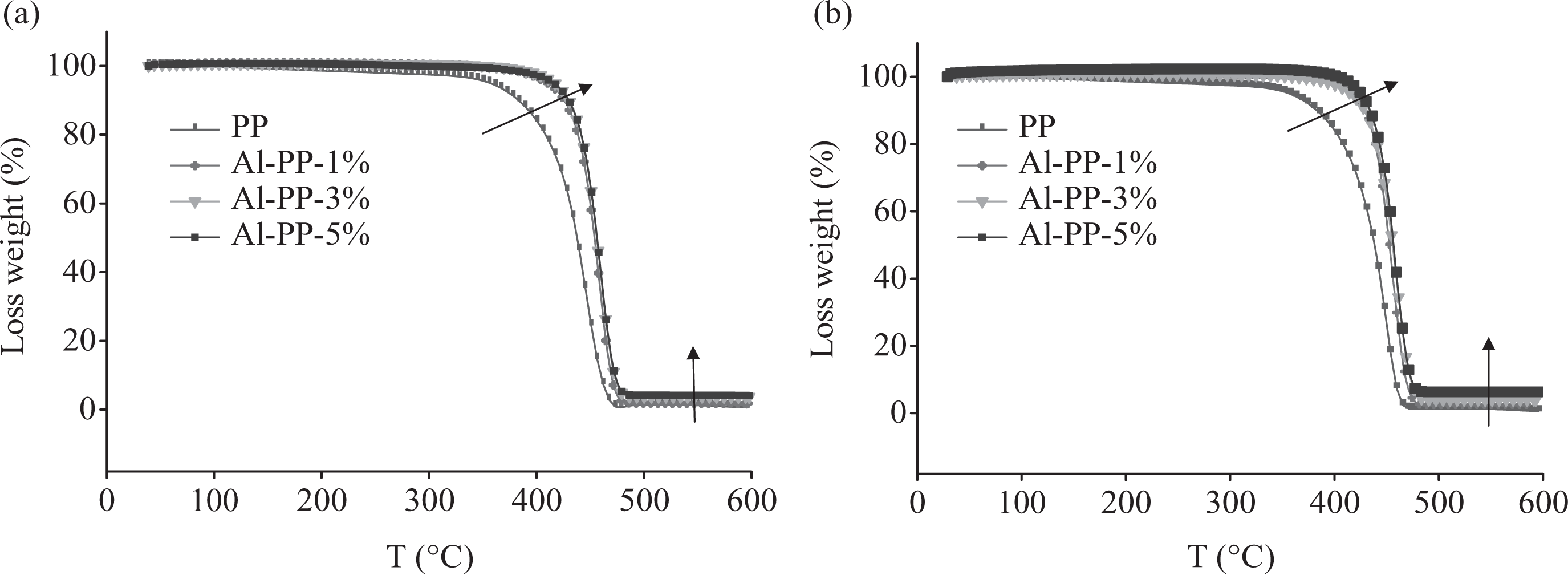
Thermogravimetric analysis (TGA) curves of polypropylene (PP) matrix and PP/nano α-Al2O3 composites with different filler contents (a) in the absence of dispersant and (b) in the presence of dispersant.
Table 1 shows the results of TGA for both nanocomposites, with and without dispersants. The initial degradation temperature (T5%) for PP was approximately 350.2°C. Adding nano α-Al2O3 to PP (5 wt%) shifted the T5% to around 417.6°C. It means that T5% increased to about 68°C with the addition of nano α-Al2O3. It was due to the increasing the absorbed heat that causes higher temperature degradation of the composite. Therefore, as the nanofiller loading was increased (Figure 2 and Table 1), there was an increase in the degradation temperature. The addition of nano α-Al2O3 loading can increase the end degradation temperature (T95%) of PP matrix. For example, the incorporation of 3 wt% of nano α-Al2O3 into PP enhanced the end degradation temperature to about 472–476°C. This is caused by the integration of filler into the matrix of PP that decreased the chain mobility in the absorption and boundary layers. This leads to a reduction in the tension induced to the carbon–carbon chain in polymer by thermal excitation. Since greater part of bond rupture is made by this mode, it was anticipated that less degradation occurred and thermal stability would be enhanced (Figure 2). 8
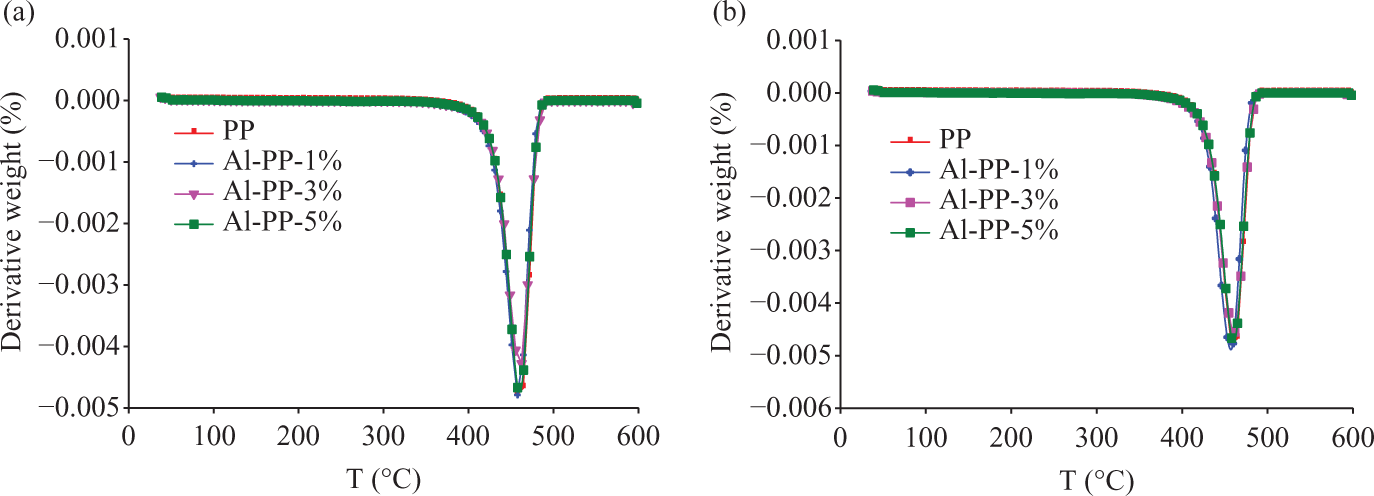
Derivatives of the thermogravimetric curve (DTG) of polypropylene(PP) /nano α-Al2O3 composites with different filler content (a) In the absence of dispersant (b) In the presence of dispersant.
From Figure 3 (a and b), it is evident that the maximum degradation temperature (Td) was increased. This exhibited the PP/nano α-Al2O3 composites are more thermally stable than the corresponding pure PP system.9,10
The percentages of composite residue were presented in Figure 4. The char residue of the composites was enhanced by increasing the nano α-Al2O3 loading. This is contributed to the elevated inorganic content in the filled materials. 8
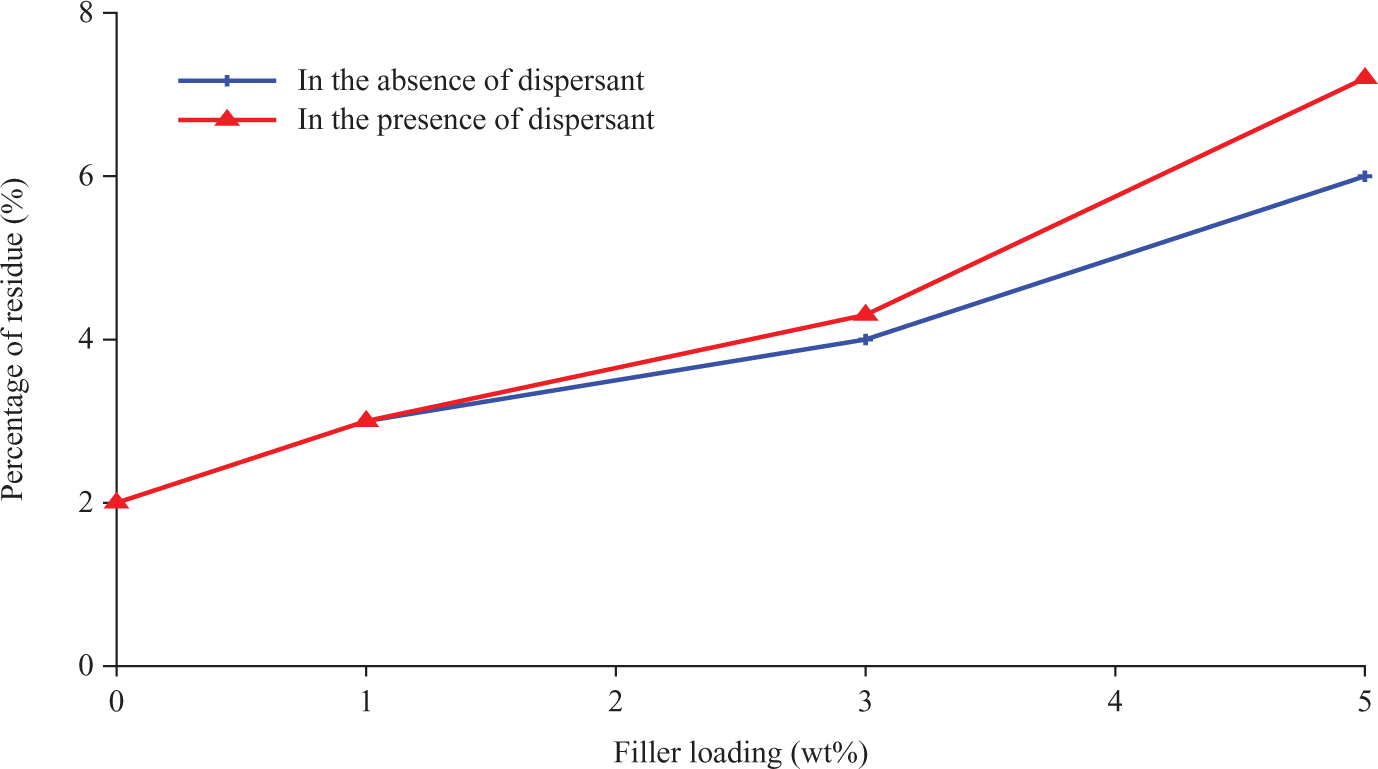
Percentage of residue of polypropylene (PP)/nano α-Al2O3 composites at different filler loadings.
Overall, the results for both PP/nano α-Al2O3 composites in the absence and in the presence of dispersants proved that the presence of the SDBS increased the thermal stability, because the aggregates shift to the primary particles with the dispersants. Hence, better wetting of filler enhances the thermal properties of the nanocomposites. 11
Different scanning calorimetry analysis
The investigation of the crystallinity was carried out using DSC.
12
DSC analysis is usually one of the most suitable methods for analyzing the first-order transition such as melting and crystallization. Detailed DSC analysis on the nano α-Al2O3-filled PP composites was performed in order to assess the possible changes in the crystallite structure and overall degree of crystallinity of the matrix. From the recorded melting and crystallization patterns, the thermal parameters such as melting temperature (Tm), crystallization temperature (
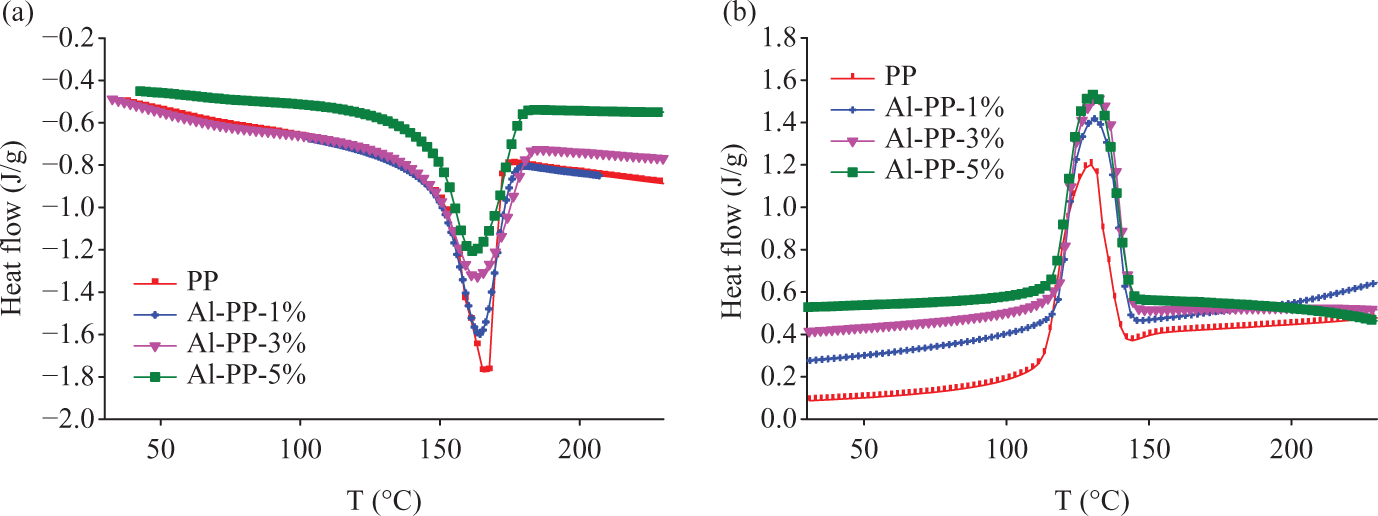
Crystallization and melting graphs of polypropylene (PP)/nano α-Al2O3 composites with different filler contents in the absence of dispersant (a) heating and (b) cooling modes.
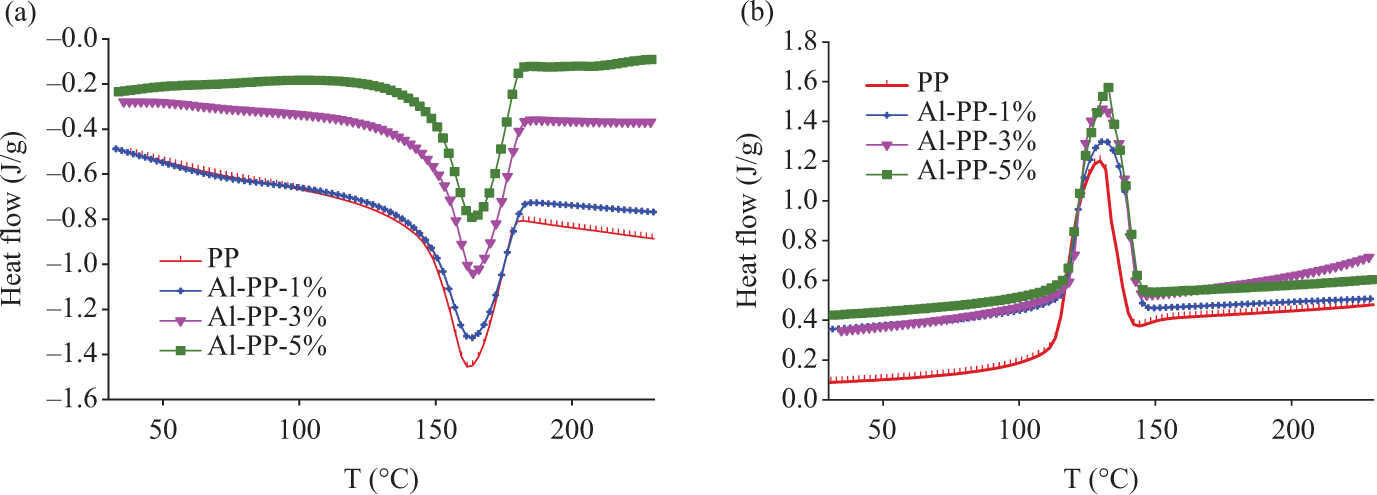
Crystallization and melting graphs of polypropylene (PP)/nano α-Al2O3 composites with different filler contents in the presence of dispersant (a) heating and (b) cooling modes.
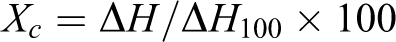
Where ΔH is the crystallization heat of the sample analyzed (J/g) and ΔH100 is a reference value that characterizes the heat of crystallization for a 100% crystalline polymer.8,12 As an example, ΔH100 is 207.1 J/g for PP. Derived from Table 2, heat of crystallization (ΔHc) and the percentage of crystallinity (Xc) for PP composites were increased by increasing the nano α-Al2O3 loading. They designated that the nano α-Al2O3 particles are effectual nucleation agent for PP. The melting and crystalline temperatures for nano α-Al2O3 filled PP composites are more when compared to the pure PP. Additionally, as shown in Figures 5(b) and 6(b), the crystallization peak of PP is shifts progressively toward the higher temperature due to the addition of nano α-Al2O3 particles. At the same time, the width of crystallization peak becomes more and more narrow. Nano α-Al2O3 particles served as the effectual nucleating agents that assist in the crystallization of PP, by initiating heterogeneous crystallization. A further effect of being a nucleation factor is an increase in the polymer crystallization temperature. As a consequence of the higher crystallization temperature, the crystals have a higher melting temperature than the unseeded polymer.
Effect of filler loading on thermogravimetry analysis (TGA) of properties of PP/nano α-Al2O3 compositesa
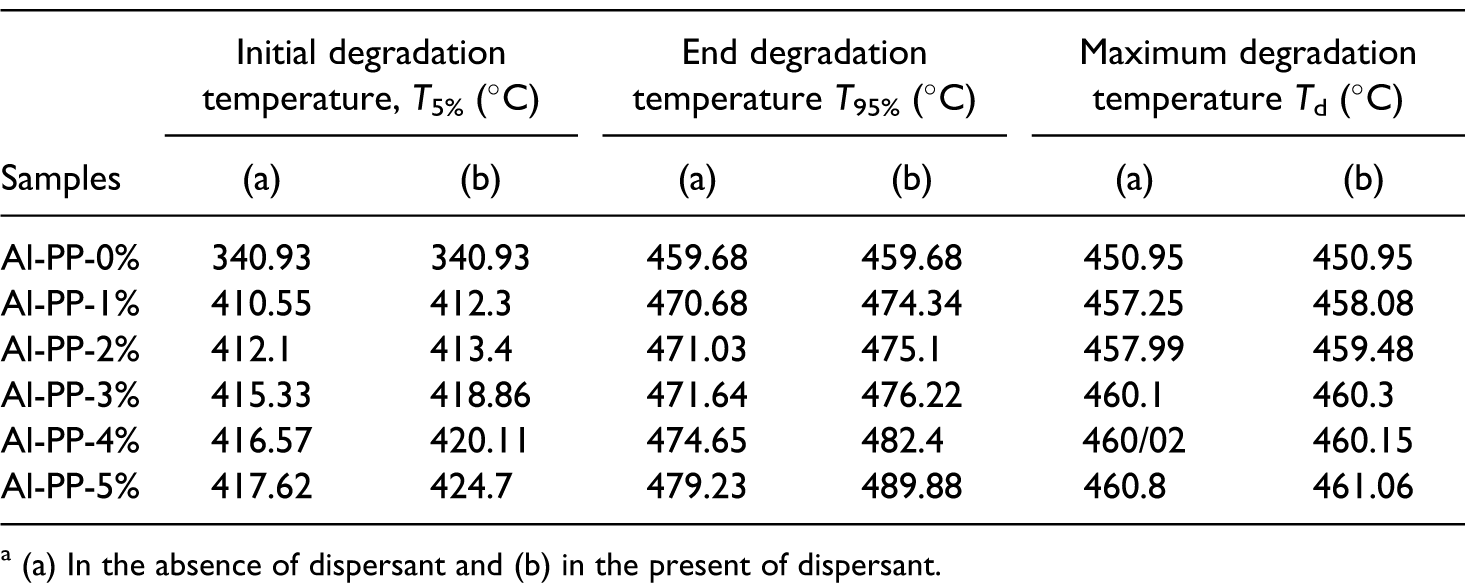
(a) In the absence of dispersant and (b) in the present of dispersant.
Comparatively, these properties are more obvious in the systems containing dispersants due to good dispersion and progressed particle orientation that allowed improved rearrangement of the particles (refer to Figure 7; Table 3).13,3
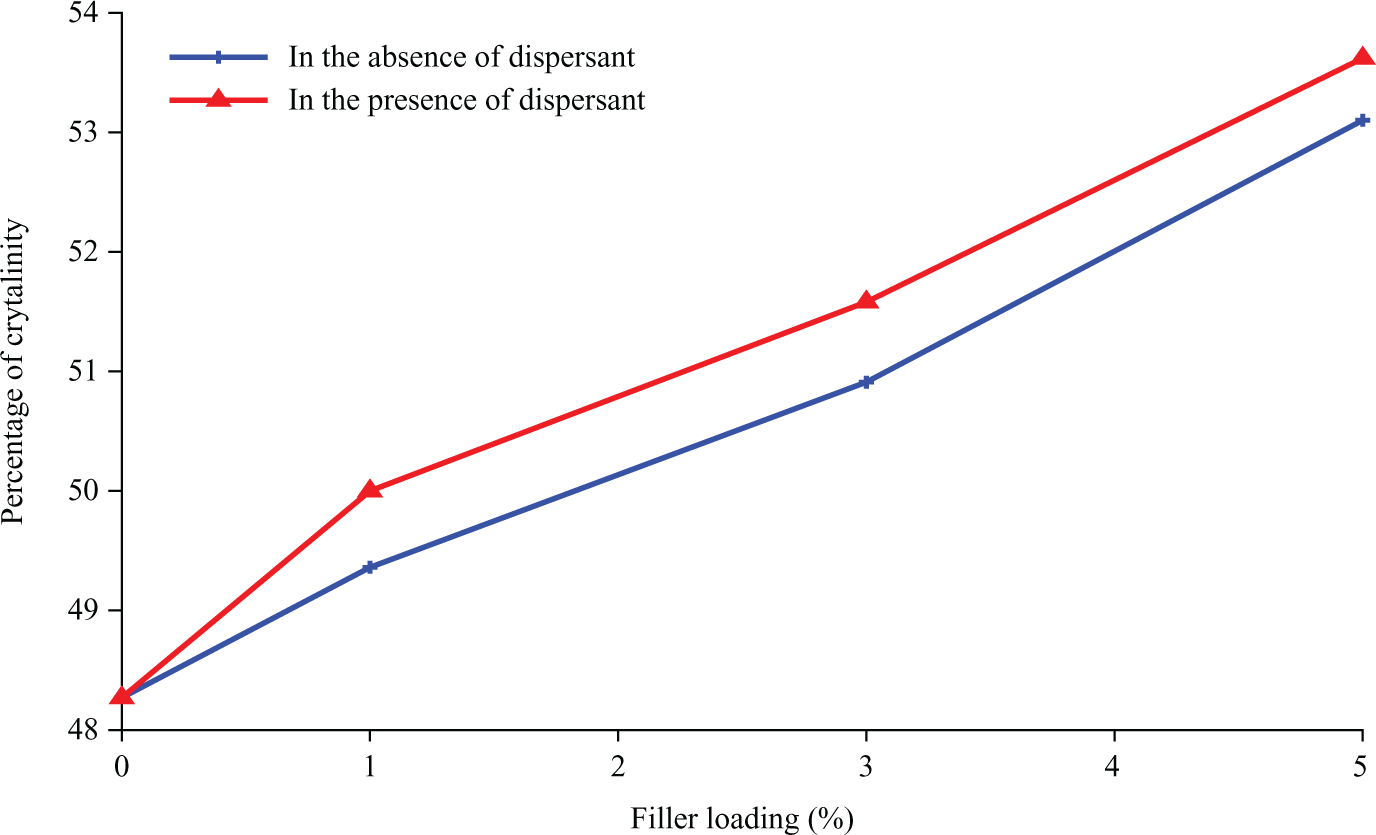
Percentages of crystallinity of polypropylene (PP)/nano α-Al2O3 composites at different filler loadings.
Effect of filler loading on differential scanning calorimetry (DSC) analysis of PP/nano α-Al2O3 composites a
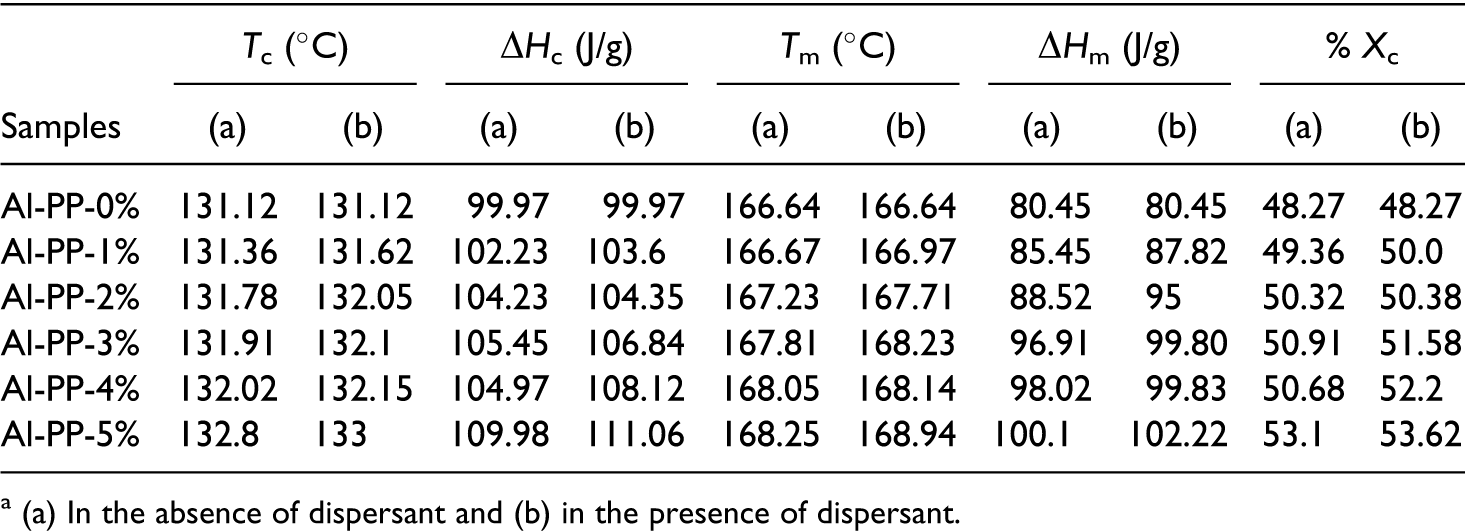
a (a) In the absence of dispersant and (b) in the presence of dispersant.
As a consequence, a better dispersion of the filler enhances the crystallization. Consequently, dispersion of nano α-Al2O3 in PP matrix, in presence of the dispersant (SDBS), leads to the enhancement of crystallization temperature and degree of crystallinity.
Dynamic mechanical analysis
Dynamic mechanical analysis (DMA) is a sensitive system that characterizes the mechanical responses of materials by monitoring property changes with respect to temperature and/or frequency of oscillation. The method separates the dynamic response of materials into two different parts: an elastic process (E′) and a viscous or damping (E″) process. The elastic process describes the energy stored in the system, while the viscous component describes the energy degenerate during the process. 14
Storage modulus (E′)
Storage modulus determines the ability of the material to absorb or store energy, and high storage modulus indicates more rigid material. 15 Figure 8 displays the variation in storage modulus (E′) for PP/nano α-Al2O3 composites as a function of temperature. Comparing the E′ spectra of the composites with the unfilled PP, it was found that the E′ improved for both composites by increasing the nano α-Al2O3 loading. This is because of the reinforcement imparted by the nano α-Al2O3, allowing greater stress transfer at the interface from the PP to nano α-Al2O3. These results indicate that the incorporation of the filler has increased the stiffness of the nanocomposites.
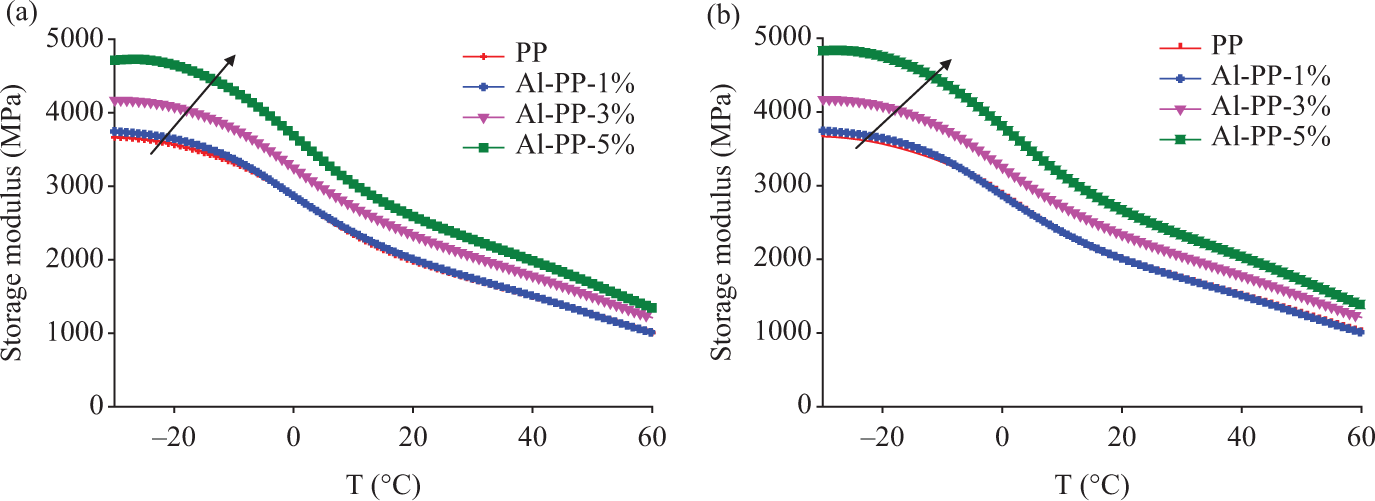
Storage modulus of unfilled polypropylene (PP) and PP/nano α-Al2O3 composites with different filler loadings (a) in the absence of dispersant and (b) in the presence of dispersant.
This is supported by the results reported by Zhang and Zhang 16 that storage modulus is higher while the molecular movement is limited, thus causing the storage of mechanical energy to increase. As the temperature of the composite samples was increased, the storage modulus decreased regularly.6,14,17
By analyzing the storage modulus for both composites in the absence and presence of dispersants, it can be seen that the composites consisting of SDBS possess higher value of storage modulus (refer to Figure 9). SDBS can disentangle large agglomerates of nanoparticles and improve the dispersion of particles in the polymer matrix, thus enhancing the interfacial adhesion.
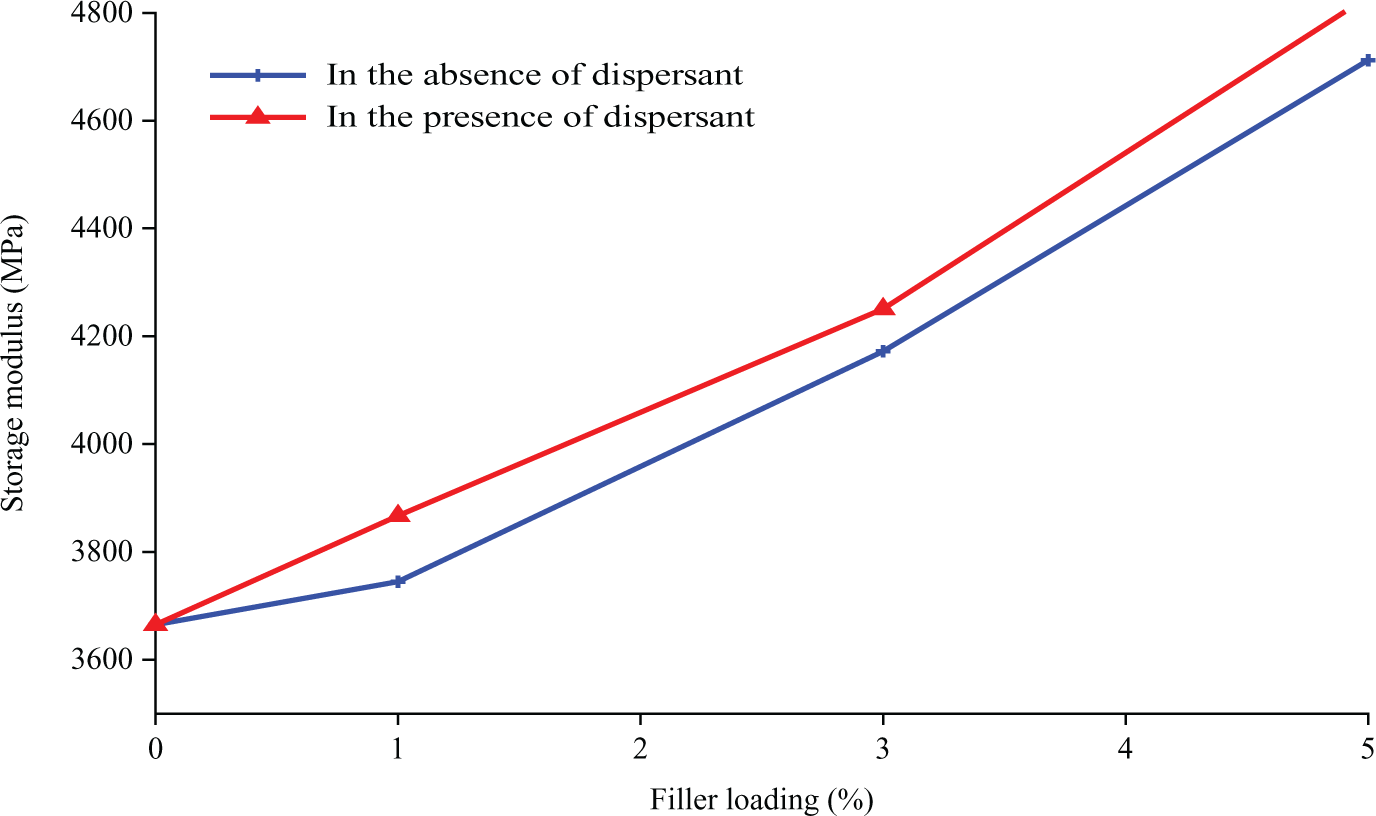
Storage modulus of polypropylene (PP)/nano α-Al2O3 composites at different filler loadings.
Loss modulus (E″)
Loss modulus determines the ability of a material to dissipate energy. 15 As shown in Figure 10, the loss modulus of unfilled PP improved with the addition of nano α-Al2O3 loading. It seems that a higher viscosity influences the molecular movement due to the presence of the fillers. Joseph et al. 17 reported that the amplitude of the loss modulus increased in the presence of fillers. This indicates that higher viscosity because of the presence of the fillers leads to a restriction in the molecular movement. The higher the nano α-Al2O3 content, the higher the viscosity, which ultimately needs extra energy for dissipation.
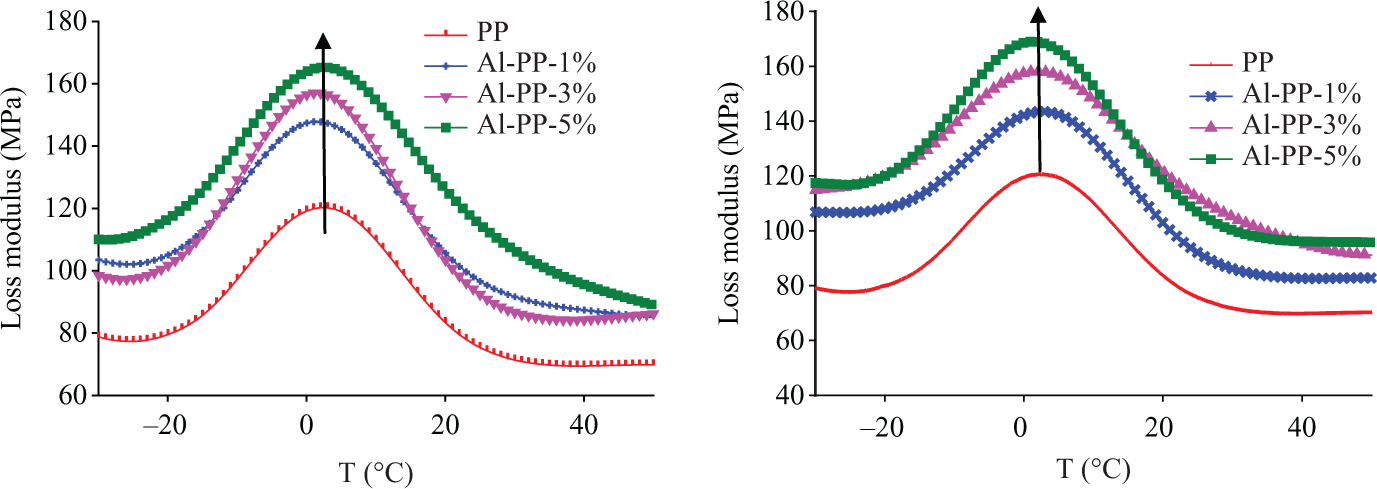
Loss modulus of unfilled polypropylene (PP) and PP/nano α-Al2O3 composites with different filler loadings (a) in the absence of dispersant and (b) in the presence of dispersant.
Based on Figure 11, treatment in the presence of the dispersant SDBS increased the active surface area of the nano α-Al2O3, ensuing improvement in wetting of the nano α-Al2O3 during compounding, and therefore, superbonding between filler and matrix.
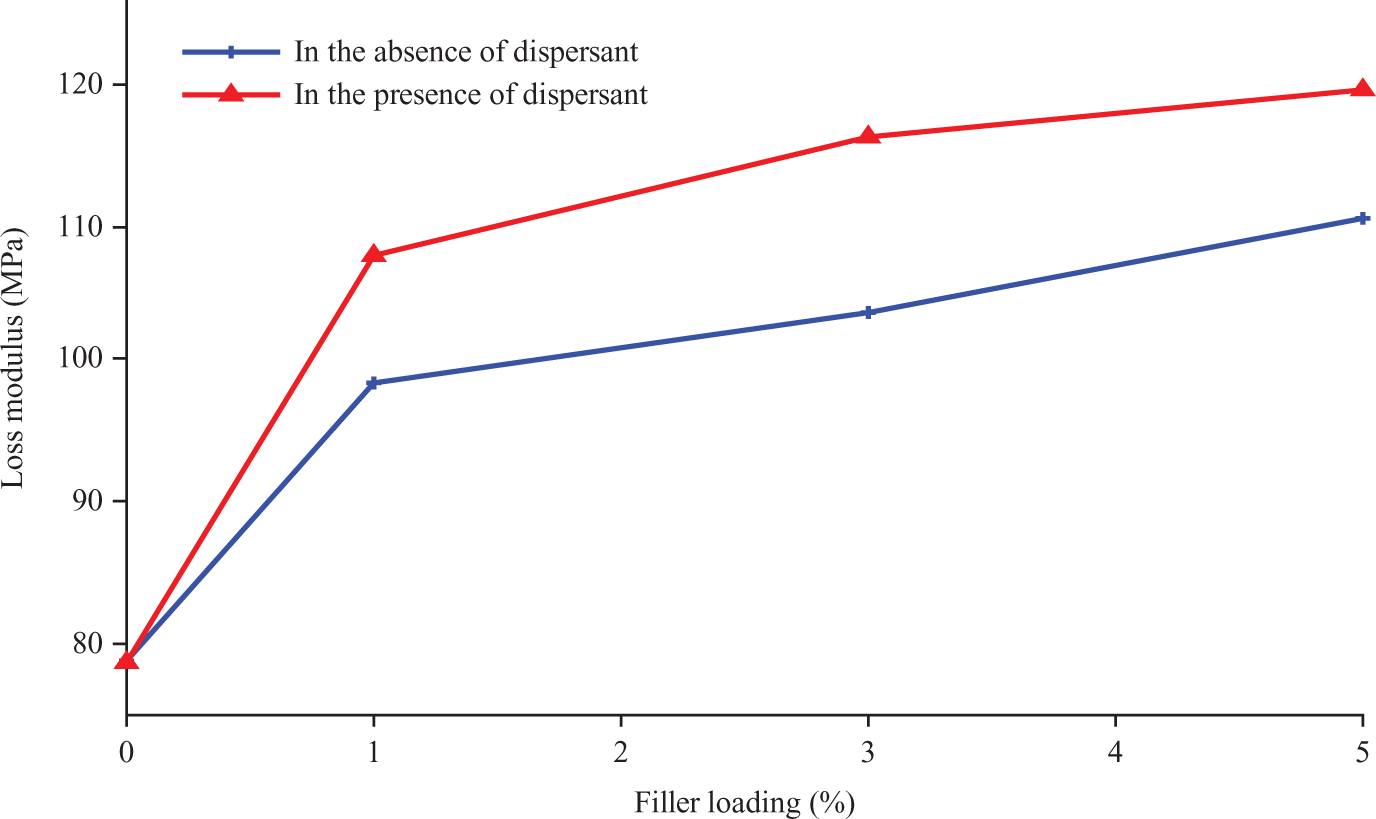
Loss modulus of polypropylene (PP)/nano α-Al2O3 composites at different filler loadings.
Damping factor
Tan (δ) measurements give practical information related to Tg and provide an indication of the strength of a material’s ability to store and dissipate energy. Tan δ indicates both viscous and elastic behaviors of the materials; tan δ < 1 demonstrates more elastic behavior of the material, that is, the material may perform like solid. On the other hand, tan δ > 1 reveals more viscous behavior and such a material behaves more like a liquid. As shown in Figure 11, the <1 range of tan δ indicates the fabricated composites behave similar to solid. In addition, the integration of nanoalumina reduces the peak height of tan δ. It is due to the incorporation of stiff filler that restricts the movement of polymer molecules. Khalid et al. 14 also stated that the poorer interfacial adhesion of PP and filler leads to the higher damping at the interface (Table 4).
Tg and Tan delta maximum (damping maximum) values of PP/ nano α-Al2O3 composites.
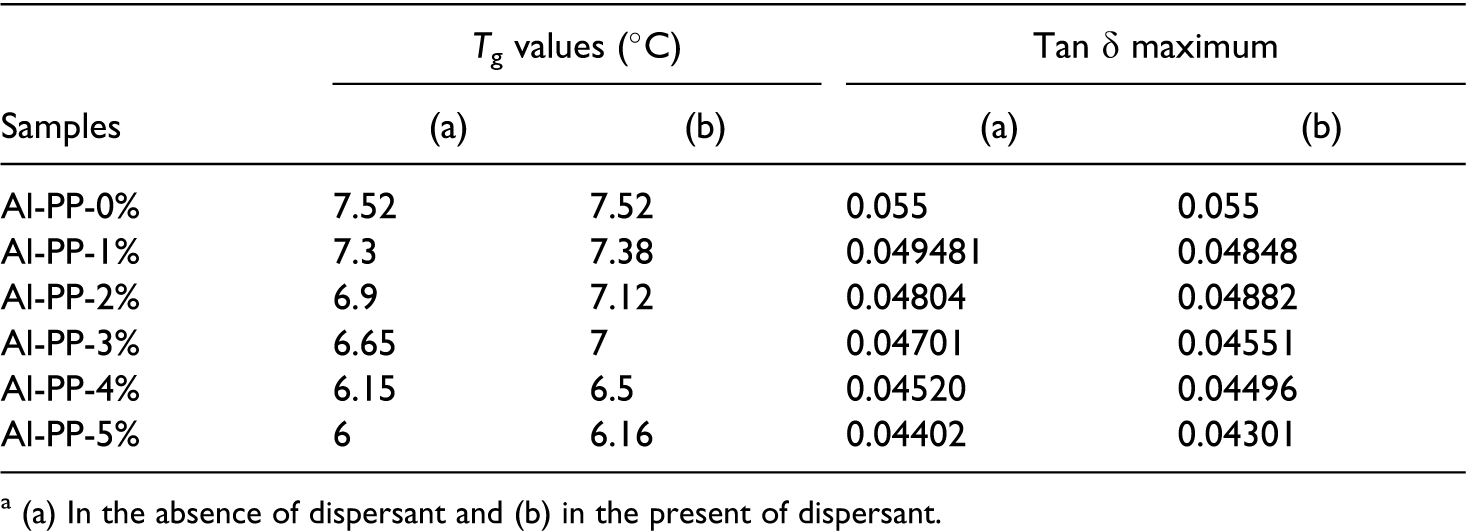
(a) In the absence of dispersant and (b) in the present of dispersant.
From the damping factor curves, Tg of the composites can be determined by the tan δ peak temperature. Vishkaei et al. 15 , showed that Tg of the nanocomposites improved by increasing the loading of nanoparticles because the nanoparticles limit the thermally induced segmental motions of the polymer chains in the composites resulting in a higher Tg. From Figure 12 and Table 4, the β-transition (glass-rubbery transition) between ∼6–8°C for the composites and pure PP can be obviously observed and the glass transition (Tg) of nanocomposites did not give any significant change as the filler loading was increased at 5 wt % nano α-Al2O3 loading. This is due to the shifting of the peak related to the glass-rubbery transition, and it is attributed to the free molecular motions unrestricted by amorphous PP (Figure 13).
Tan δ profiles of unfilled polypropylene (PP) and PP/nano α-Al2O3 composites with different filler loadings (a) in the absence of dispersant and (b) in the presence of dispersant.
Damping factor (tan δ) of polypropylene (PP)/nano α-Al2O3 composites at different filler loadings.
Comparing tan (δ) for PP/nano α-Al2O3 composites in the absence and presence of dispersants shows that the peak height of tan δ decreases in the presence of a dispersant. Effective dispersion will promote the transfer of stress from the matrix to the reinforcement fillers, preventing the formation of stress concentrations.
Conclusion
The Al2O3 nanoparticles were dispersed in the PP matrix using the internal mixing method.
Thermal decomposition curves of the PP/nano α-Al2O3 composites showed that the temperature for the weight loss in the composites is increased by adding the amount of nano-Al2O3, which significantly reduced the rate of heat release for PP, making these composites a good option for effective fire retardant. Moreover, the presence of dispersant in PP/nano α-Al2O3 composites has a little effect on the thermal stability.
DSC curves show that the crystallinity is deeply influenced by the presence of the nano α-Al2O3 in the matrix, owing to the influence of filler as the nucleating agent. As a result, better dispersion of the filler enhances the crystallization temperature and the degree of crystallinity.
Storage modulus and loss modulus of nanocomposites improved with increasing loading percentages of nano α-Al2O3. These observations show improvement in the stiffness and energy dissipation of the composites deformed under the periodic stress. Addition of nano α-Al2O3 also reduces the tan δ peak height and glass temperature. Poorer interfacial adhesion of PP nanocomposite leads to the higher damping at the interface and decreased Tg because of the molecular motions related with unrestricted amorphous PP. Comparing the DMA (storage modulus, loss modulus and tan (δ)) of PP/ nano α-Al2O3 composites in the absence and presence of dispersants shows that all these properties improved in the presence of dispersants.
Footnotes
Acknowledgments
The authors wish to thank the University Putra Malaysia, which supported this work.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
