Abstract
The influence of alkaline aqueous solution treatment of groundnut shell–high density polyethylene composites consist of magnesium hydroxide as flame-retardant additives was investigated. FTT daul cone calorimetry and Perkin-Elmer Diamond differential scanning calorimetry (DSC) were used to characterize flammability and thermal properties of the fabricated composites. Quanta 200ESEM was used to investigate the effect of alkaline aqueous solution treatment on the microstructure of the composites. The result obtained revealed that the alkaline treatment of the shell enhanced filler–matrix compatibility. The cone calorimetry result revealed that Mg(OH)2 has good fire retardancy characteristics on the fabricated composites as properties such as heat release rate, effective heat of combustion, mass loss rate, total smoke produced, and specific extinction area were enhanced. DSC results suggested that the additives had little impact on the melting temperature of the composite.
Keywords
INTRODUCTION
Natural fiber–high-density polyethylene (HDPE) composites are the most recent area of interest for researchers [1,2]. Most of the material property requirement that are not present in the monolithic engineering materials are found in natural fiber–polymer composites [3,4]. Natural fiber–HDPE composites have found new applications in many sectors such as building and transport industries; therefore, the need to reduce material flammability is a key requirement [5].
Due to the unique properties of HDPE, such as good weathering resistance, it is also applied in many other fields of technology like the aerospace, automobile, and cable industries [6]. From chemical composition analysis, groundnut shell has been found to be flammable and so how to improve its flame retardancy is an important requirement. Research efforts have been made in fabricating flame-retardant olefin polymer composites, Zang and Horrock reviewed flame-retardant polypropylene–fiber composites [7].
Conventionally, halogens containing fire-retardant additives are the most widely used fire-retardant additives [8], but the environmental issues surrounding them became an impediment. The need for alternative flame-retardant material to replace the halogenated materials is now receiving considerable attention all over the globe [9].
It has been established through research that bromine-containing compounds are the most effective commercial fire retardants for reducing the flammability of HDPE [8,10,11]. But in order to sustain green technology, the search for an alternative (an environmental friendly fire-retardant additive that has the capability to replace bromine) is presently a target for researchers.
Unlike the halogen flame-retardant additives, magnesium hydroxide does not pollute the environment in any form. It had been used for thermosettic polymer composites in a semi-cross-linked system, but this study is designed to investigate its performance in a thermoplastic system.
Many research works have been conducted on the use of groundnut shell as reinforcing filler for different polymers [12–15], especially, the thermosettic family. The results obtained suggested that groundnut shells are highly hydrophilic in nature and as such, may not be very useful in many engineering applications [13]. This study was designed to address the hydrophilic nature of the shell by activating the hydroxyl group of the shell using 5%w/v of sodium hydroxide. From research, it has been found that fiber surface treatment enhances material properties [16].
Malachy et al (2002) suggested groundnut shell to have poor mechanical properties as a result of its hydrophilic characteristics in a hydrophobic HDPE matrix.
EXPERIMENTAL WORK
Materials
The groundnut shell was sourced locally in Zaria, Kaduna State, Nigeria. It was first washed, dried, and crushed into fine particles. ASTM – 11 standard sieves was used to separate the particulate into the required particle size of 300 µm (50 mesh).
The HDPE was supplied by SK Corporation Yuzuk in Korea. (Injection grade) and the magnesium hydroxide by BOD.
Sample Preparation
Many tasks were carried out to achieve the objectives of this study. First task was the alkaline aqueous solution treatment of the groundnut shell particulate and then the incorporation of the groundnut shell particulates into HDPE. Also, 30 wt% treated groundnut shell particulates were incorporated into the continuous phase (polymer matrix) with 10% Mg(OH)2 using Carvers Two-Roll Mill at a processing temperature of 160°C for 7 min to form system C while 30 wt% of untreated groundnut shell particulates was used to form system B. The resulting composites were then prepared according to the specified ISO standard specification for fire testing (ISO 5660-1-2002). This was performed using compression molding technique to produce 3 mm thick sheet of 100 mm2 for the cone calorimetry test.
Flammability Test
The flammability test experiment was performed via Fire Testing Technology dual cone calorimetry test using ISO 5660-1-2002 standard specification procedures on 3 mm thick 100 × 100 mm2. This was performed at a heat flux of 50 kW/m2 [17,18].
Thermal Characteristics
Differential scanning calorimetry (DSC) was employed to investigate the thermal properties of the composites such as the melting and crystallization temperatures together with other thermodynamic properties of the system.
Perkin-Elmer DSC was used at 10°C/min heating rate and the results obtained were represented in a thermograph.
Microstructure
Quanta 200 Environmental Scanning Microscope was used to examine the matrix–filler interaction of the composites under two magnifications, 500× and 1000×. The effect of alkaline treatment of the groundnut shell on the microstructure of the composites was investigated.
RESULTS AND DISCUSSION
Flammability Characteristic
Fire Testing Technology Dual Cone Calorimetry Test was carried out on system A (unfilled HDPE), system B (EGR + Mg(OH)2 composite) and System C (TEGR + Mg(OH)2 composite). The heat release rate (HRR) values of the various systems is shown in Figures 1–3. It is quite obvious that Mg(OH)2 act to reduce burning. HRR is an important parameter for characterizing the fire behavior of any polymer material. It is a measure of fire intensity and growth of any polymer system. Lower HRR indicates more effective fire retardancy. For system A, there was only one peak which coincided with the maximum mass loss rate (MLR) before a sharp decline to zero. Figure 2 is the HRR for system B; the result showed a peak which was sustained for a while and then a decline before 200 s. Figure 3 shows the effect of the alkaline treatment on the fire performance of system C. It showed a peak and decline after 200 s.
HRR of system A. HRR of system B. HRR of system C.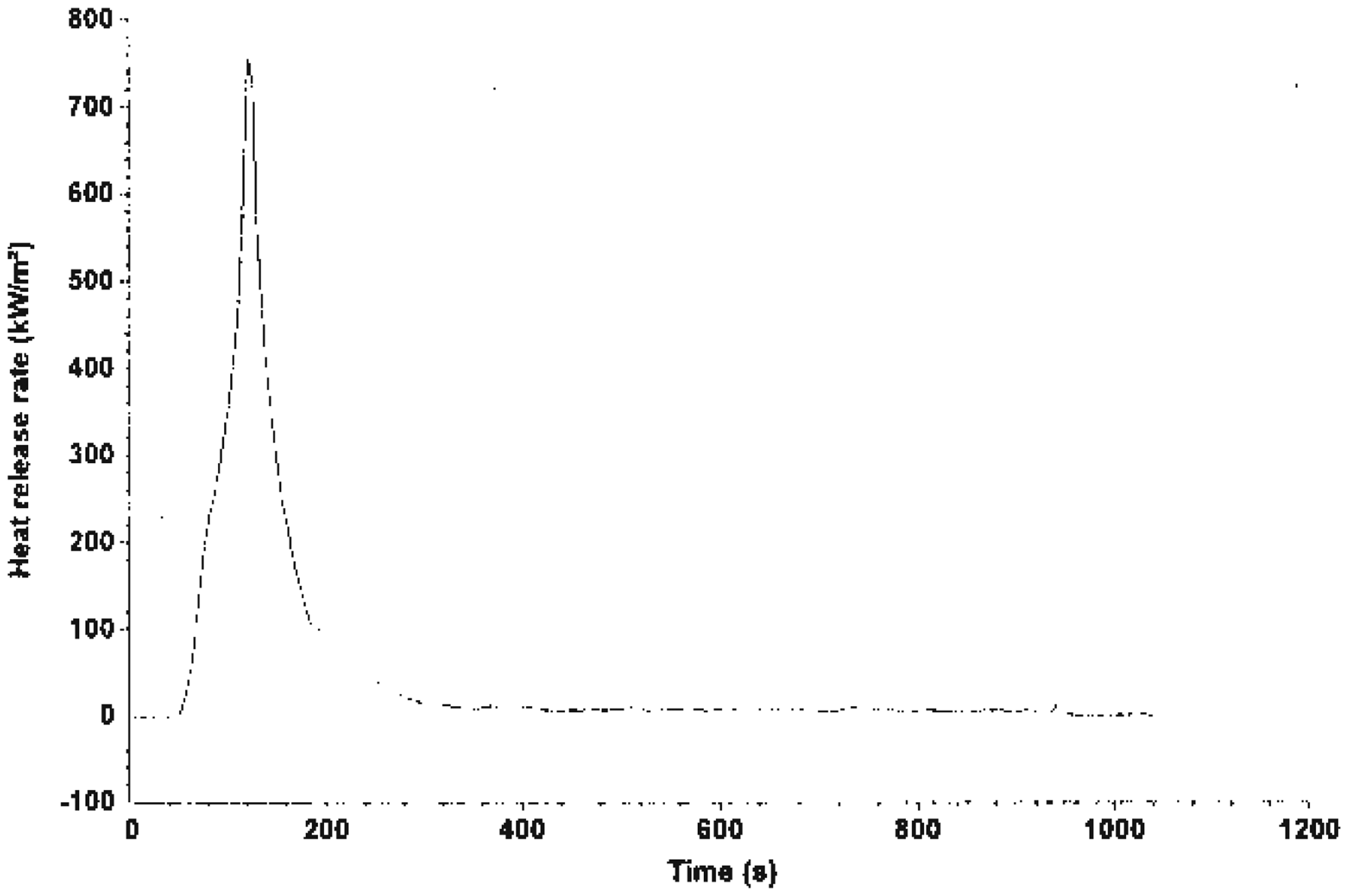
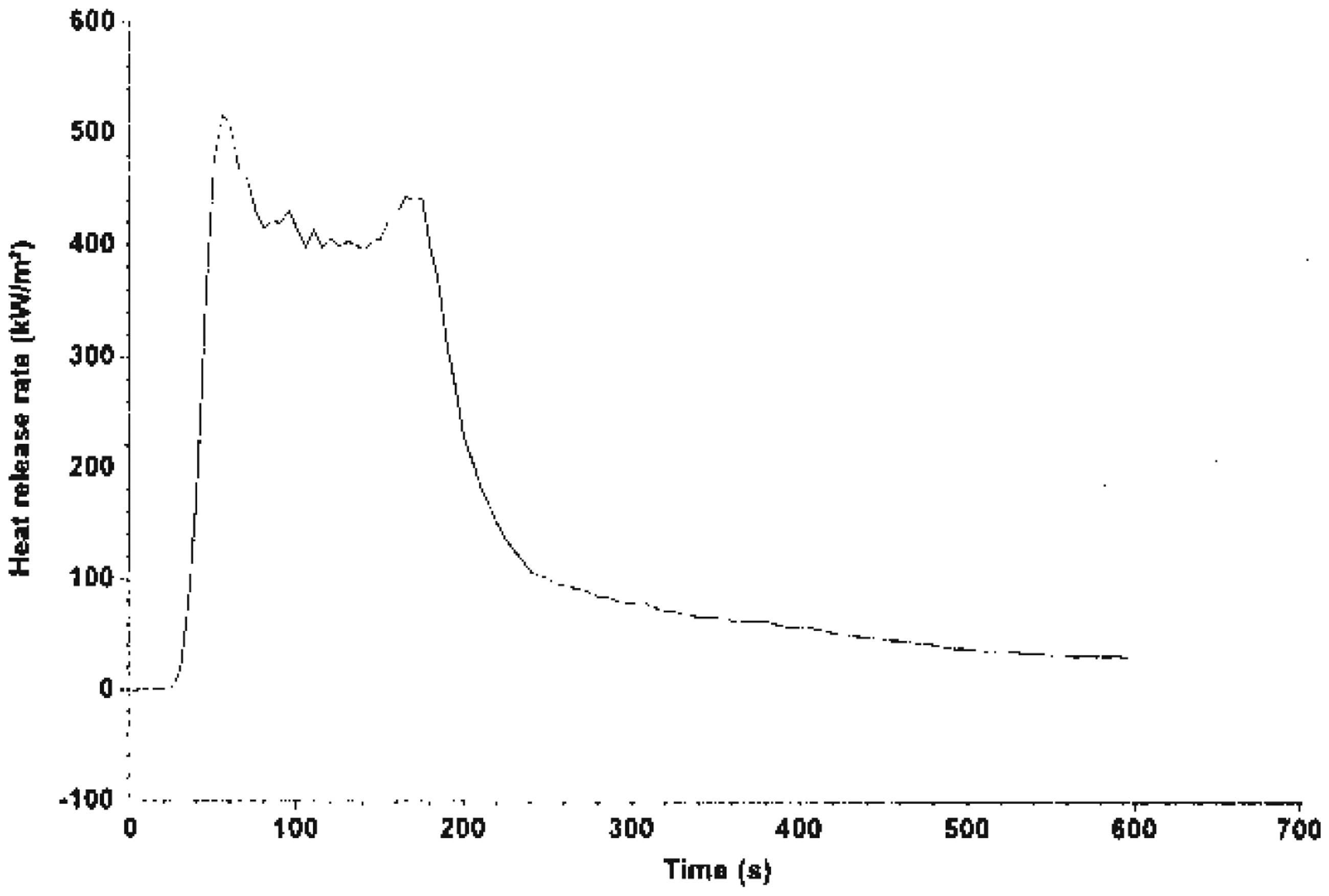
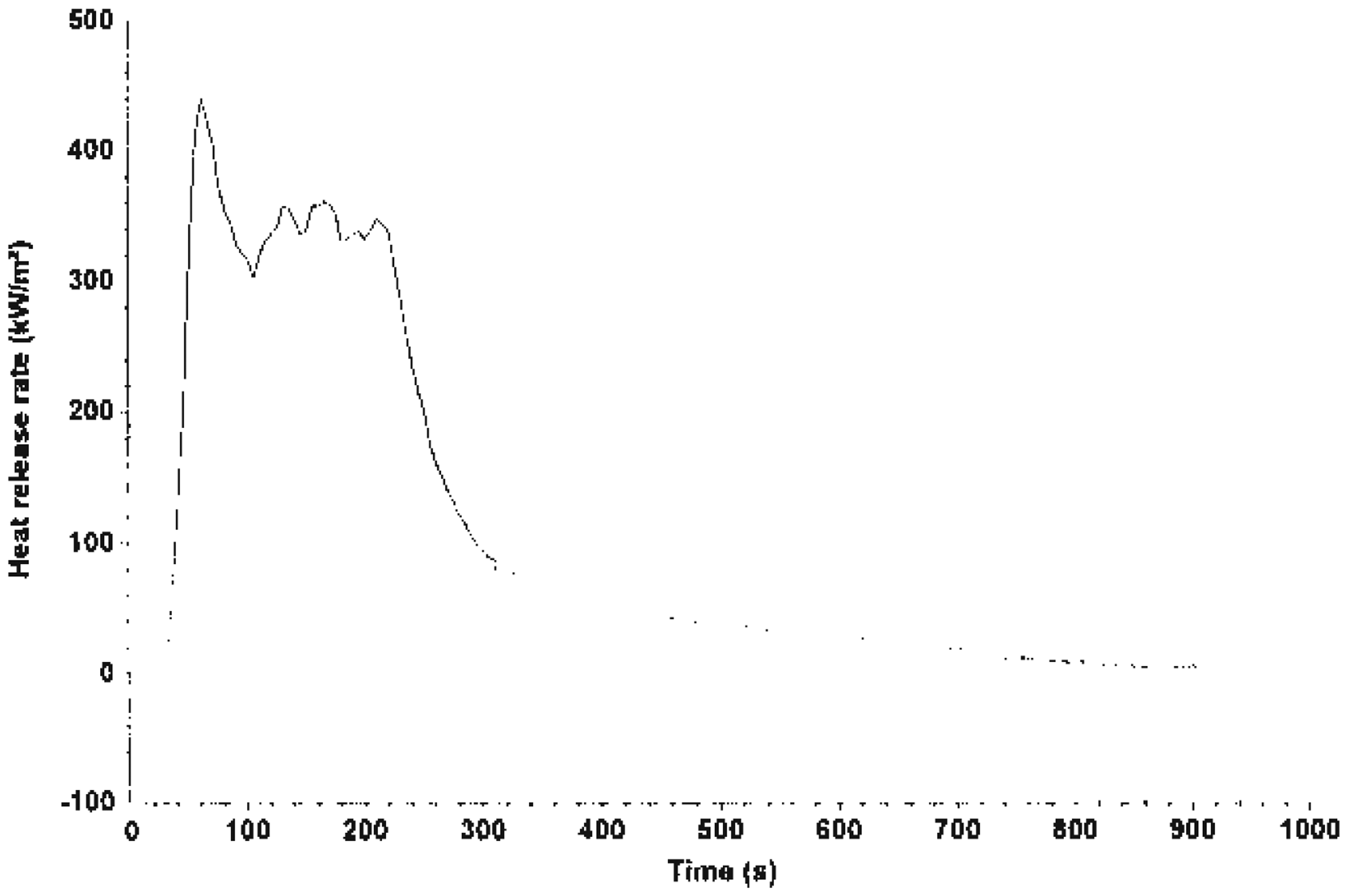
Thermal degradation of any polymer system leads to the evolution of a volatile substance, which ignites and sustains burning. HRR increases when there is sufficient quantity of combustible gases until a peak occurs, which coincides with the maximum MLR.
When all the combustible gas has been exhausted, the combustion reaction stops. At this point, there is always an abrupt decline to zero with 100% mass loss.
Flammability characteristics of the various composites using fire testing technology dual cone calorimeter.
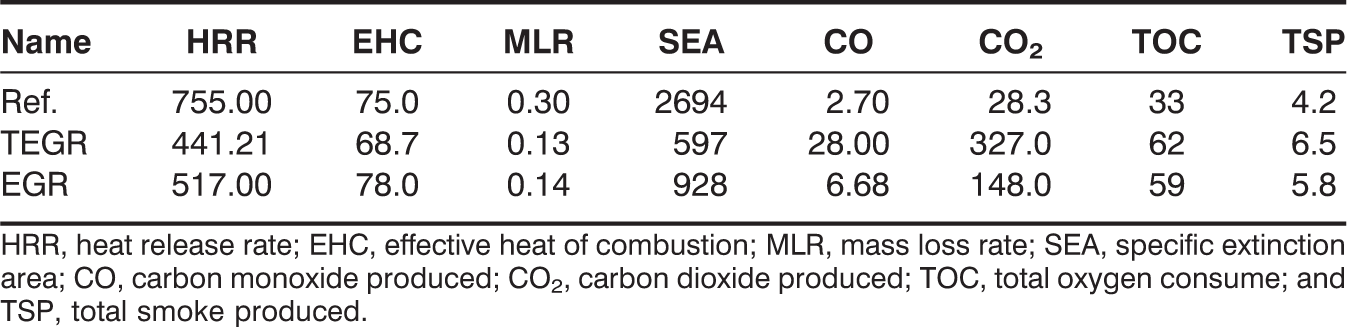
HRR, heat release rate; EHC, effective heat of combustion; MLR, mass loss rate; SEA, specific extinction area; CO, carbon monoxide produced; CO2, carbon dioxide produced; TOC, total oxygen consume; and TSP, total smoke produced.
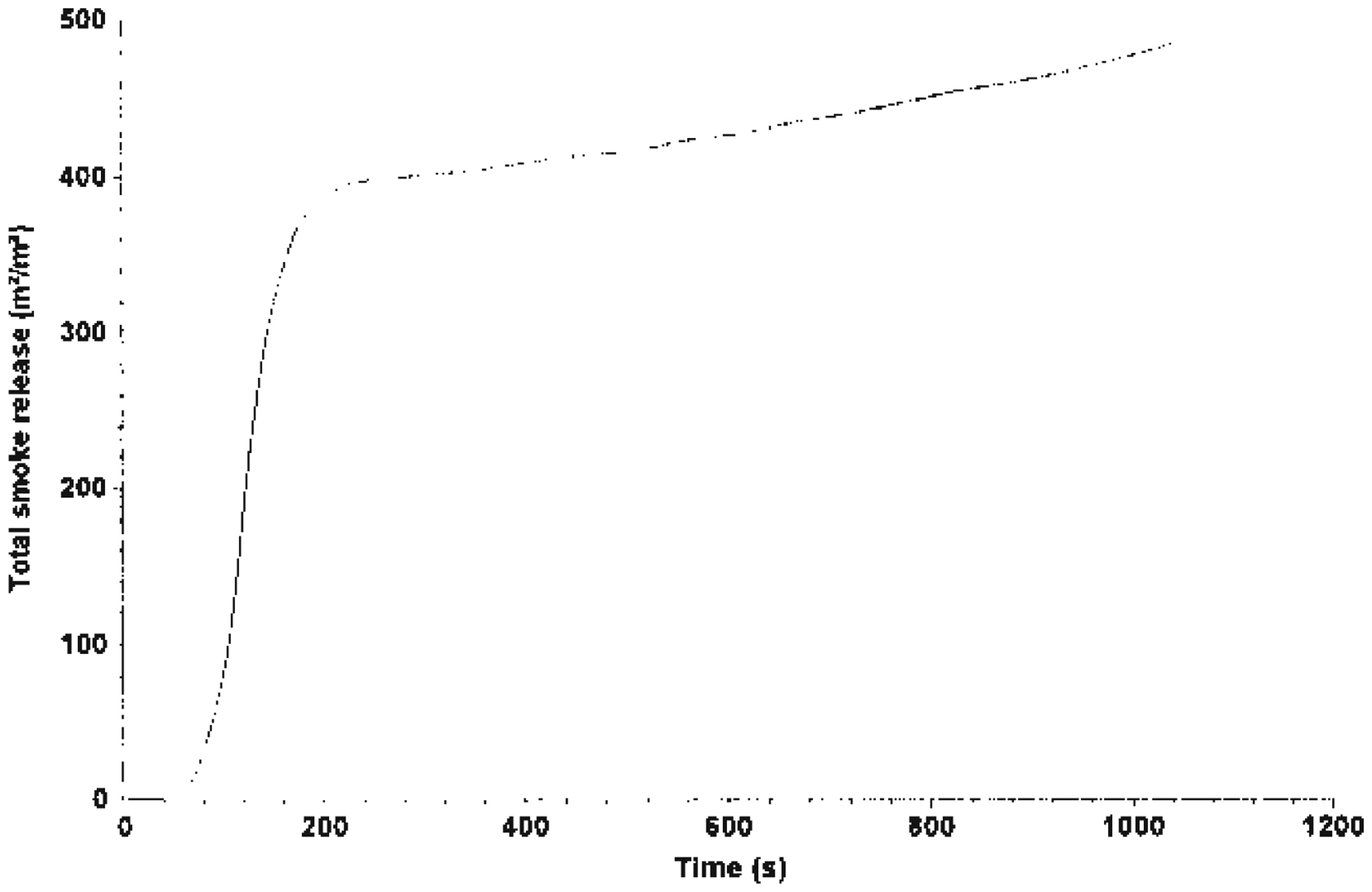
Total smoke release by system A.
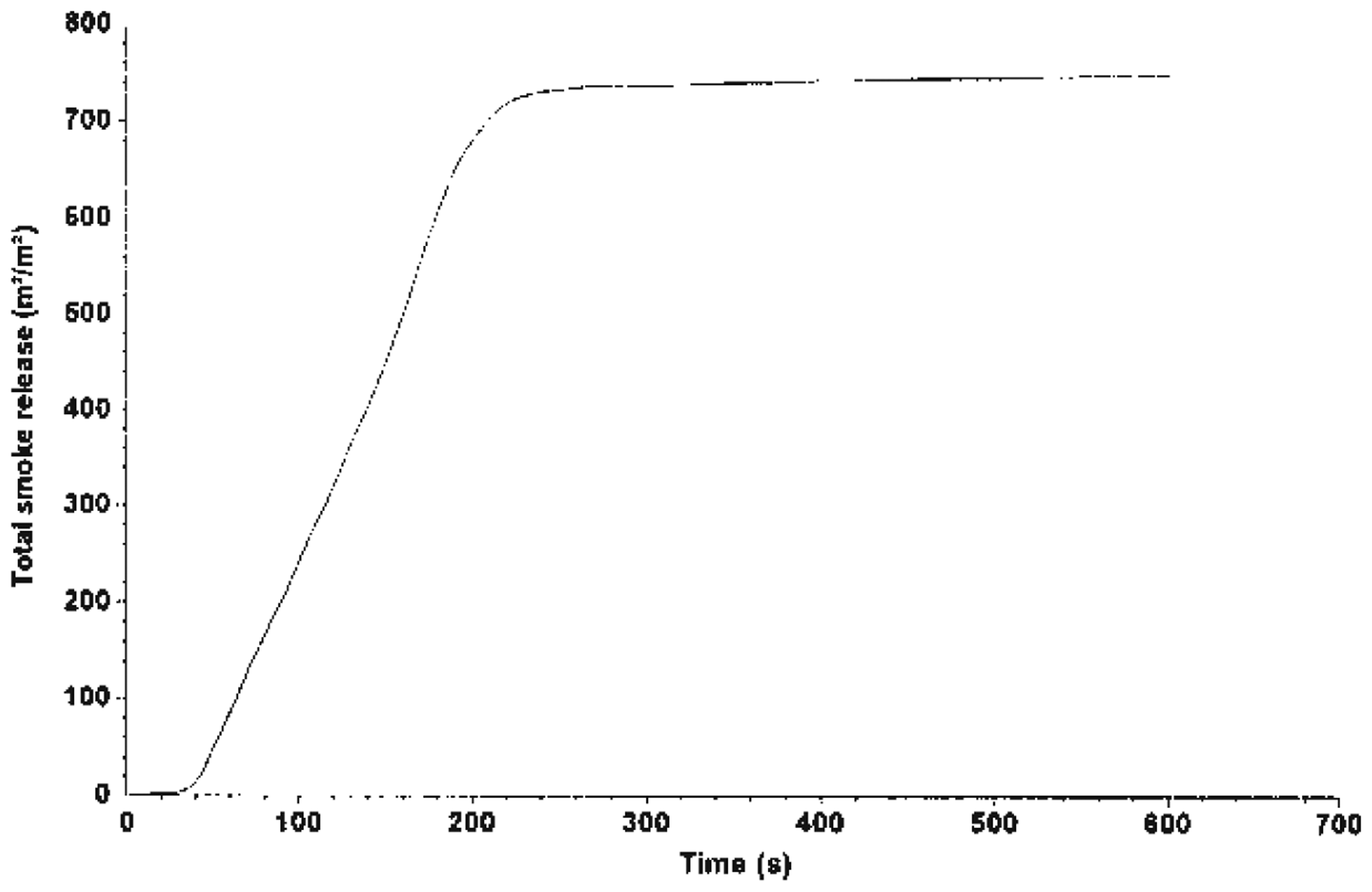
TSP by system B.
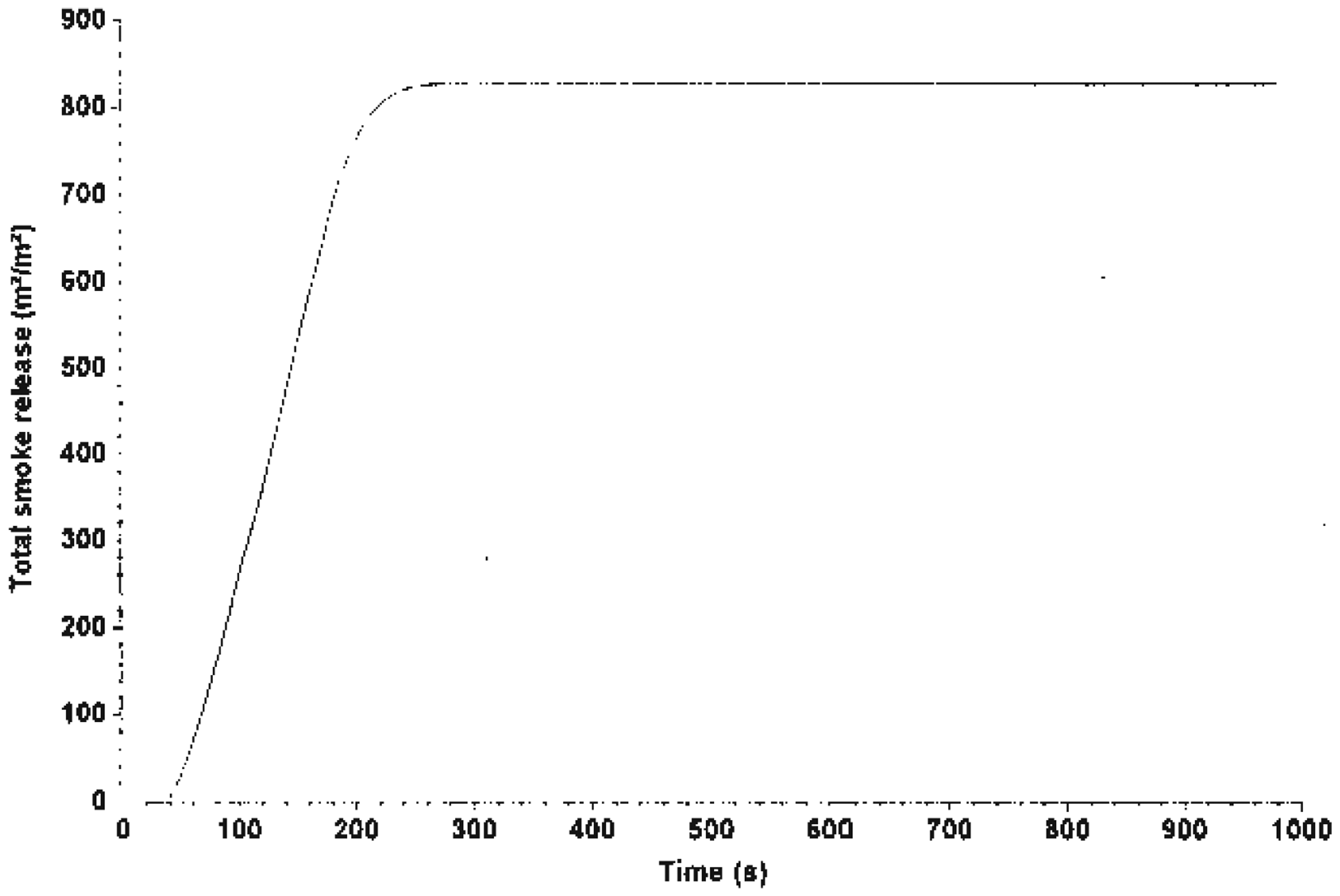
TSP by system C.
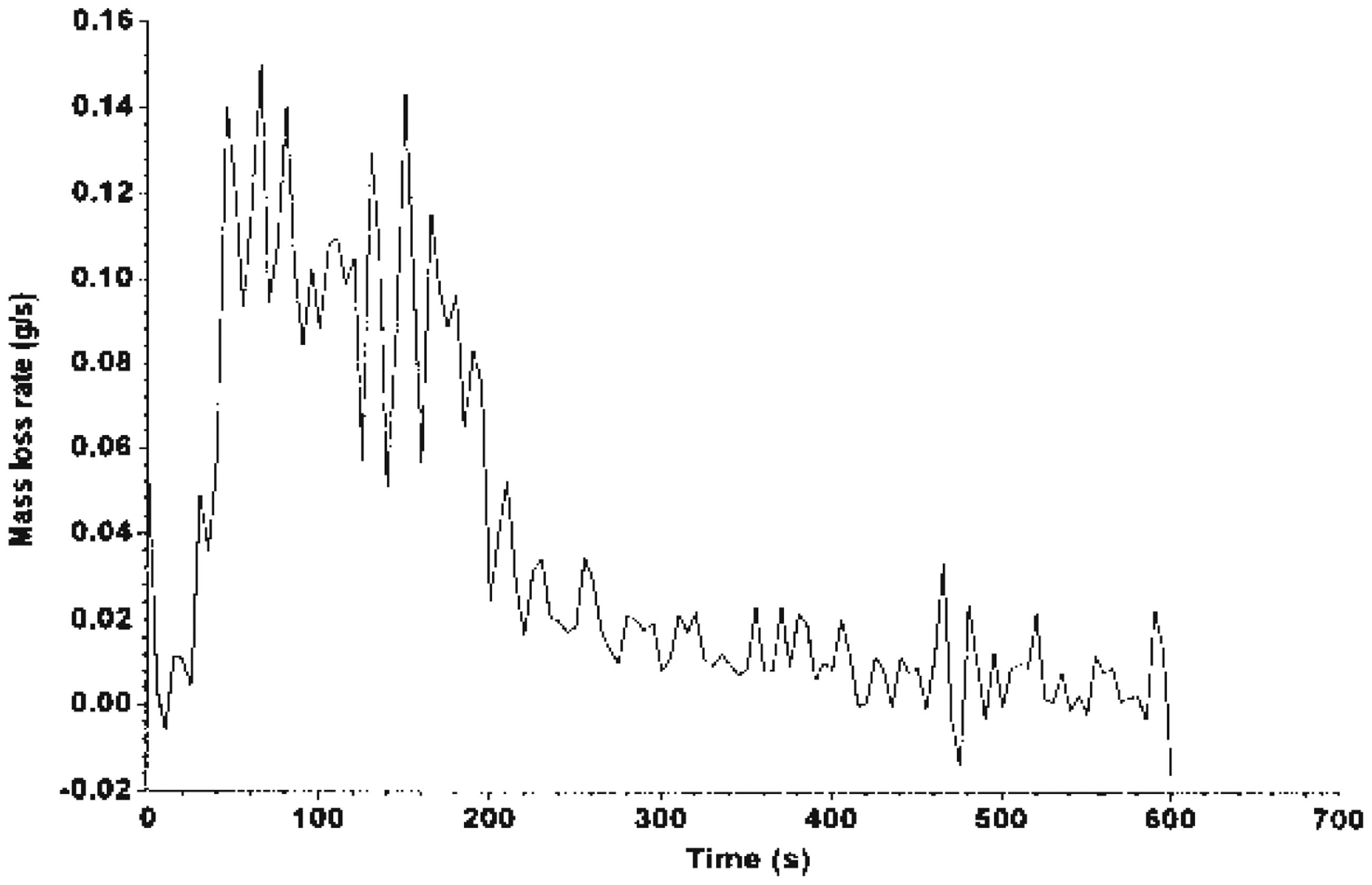
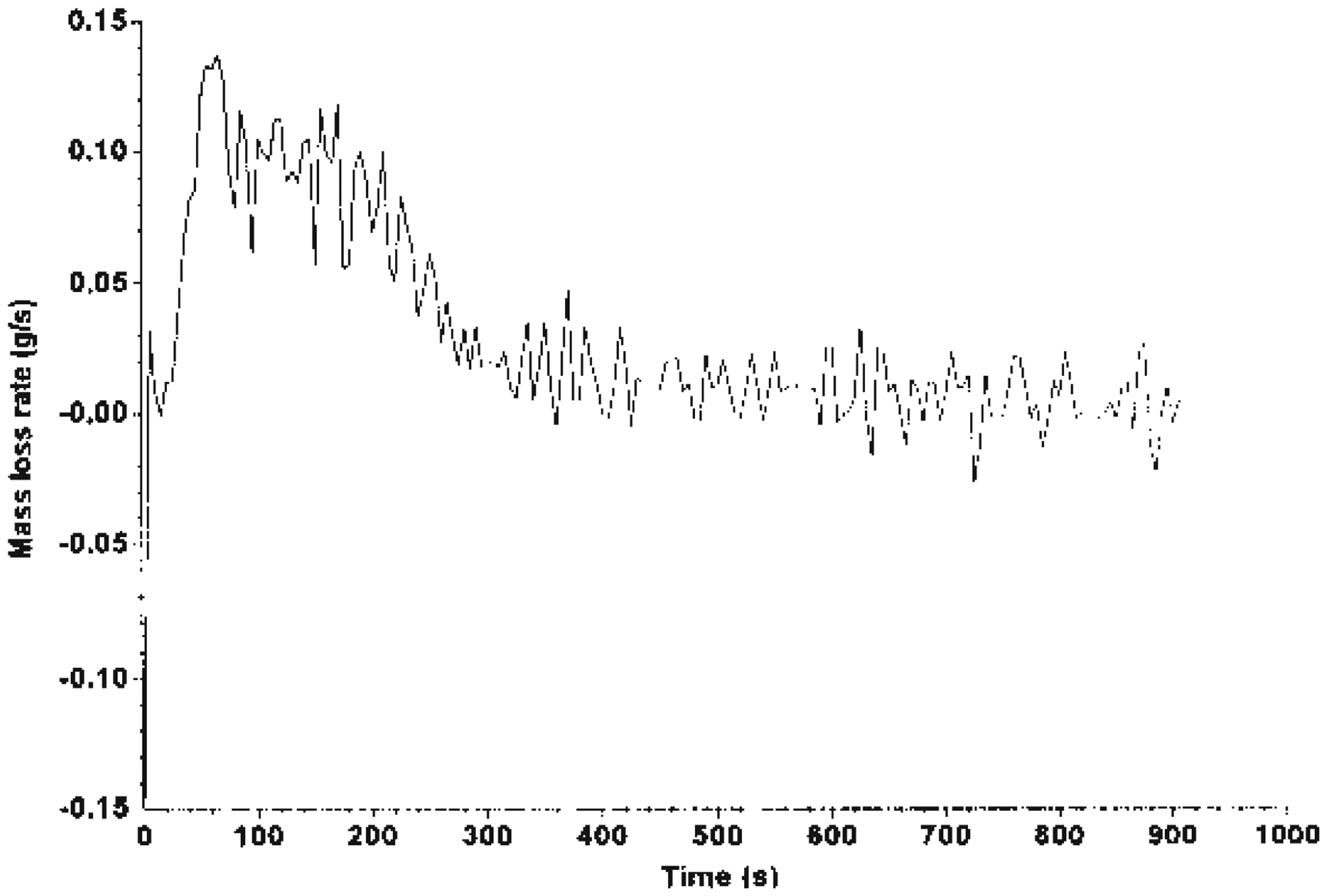
The MLR curve is illustrated in Figures 7–9, which shows that Mg(OH)2 decreased the rate of mass loss of the system. System C shows an enhancement with alkaline treatment. Figures 10 and 11 show the photographs of the residues left after burning the EGR and TEGR (systems B and C). System A (unfilled HDPE) burnt and left no residue.
MLR of system A. MLR of system B. MLR of system C. Residue of system B left after testing using FTT dual cone calorimeter. Residue of system C left after testing using FTT dual cone calorimeter.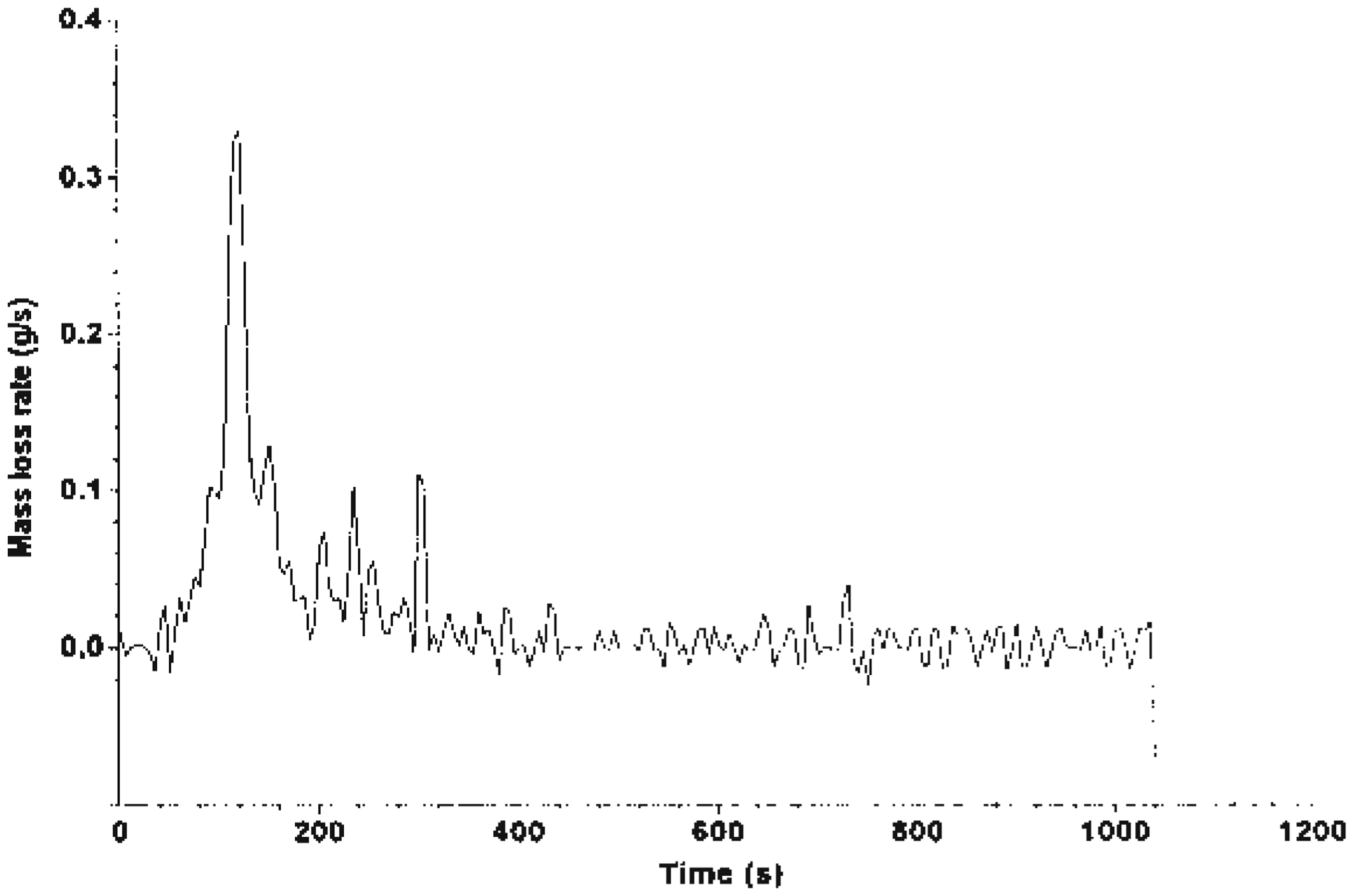


Thermal Analysis
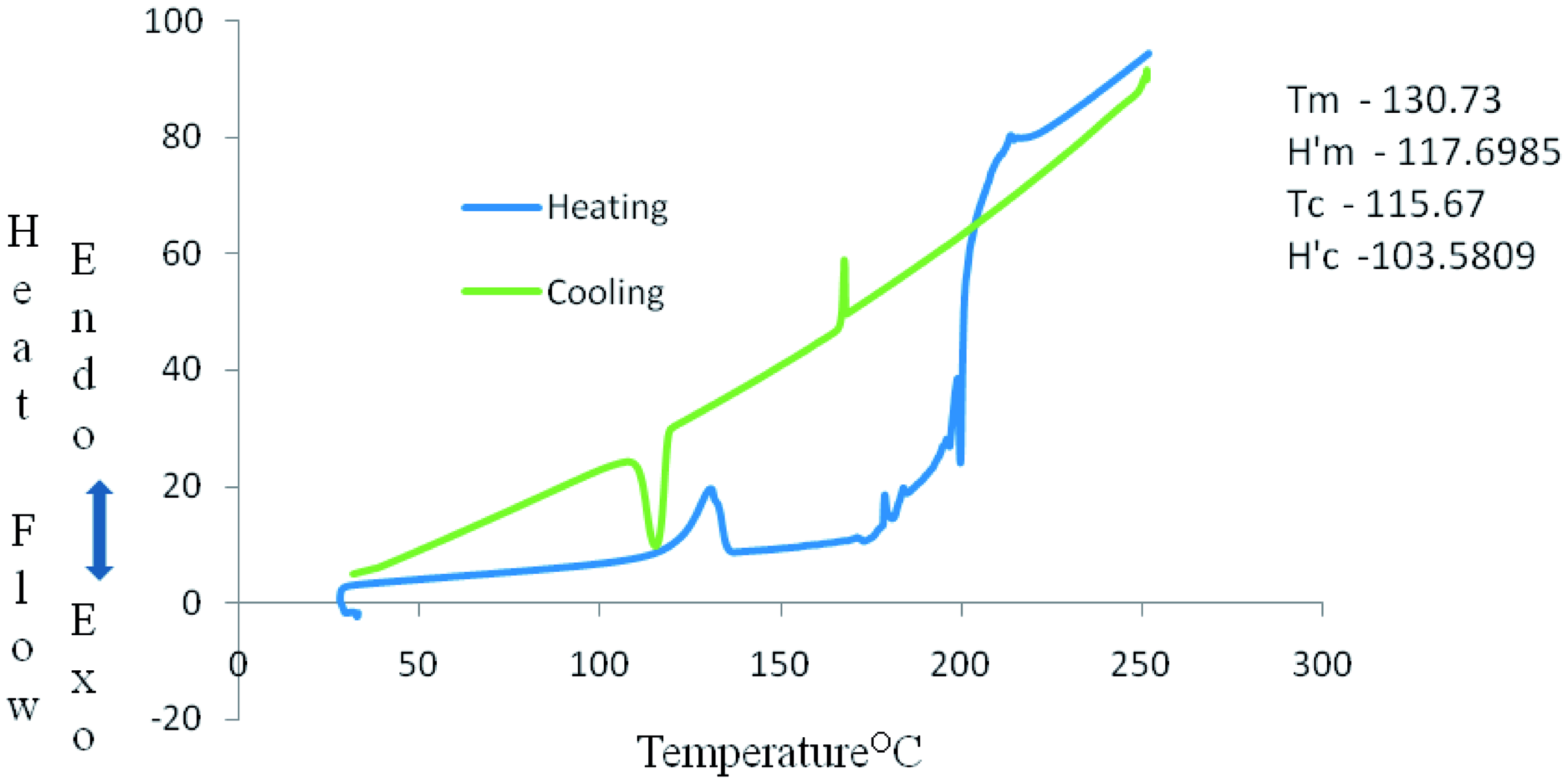
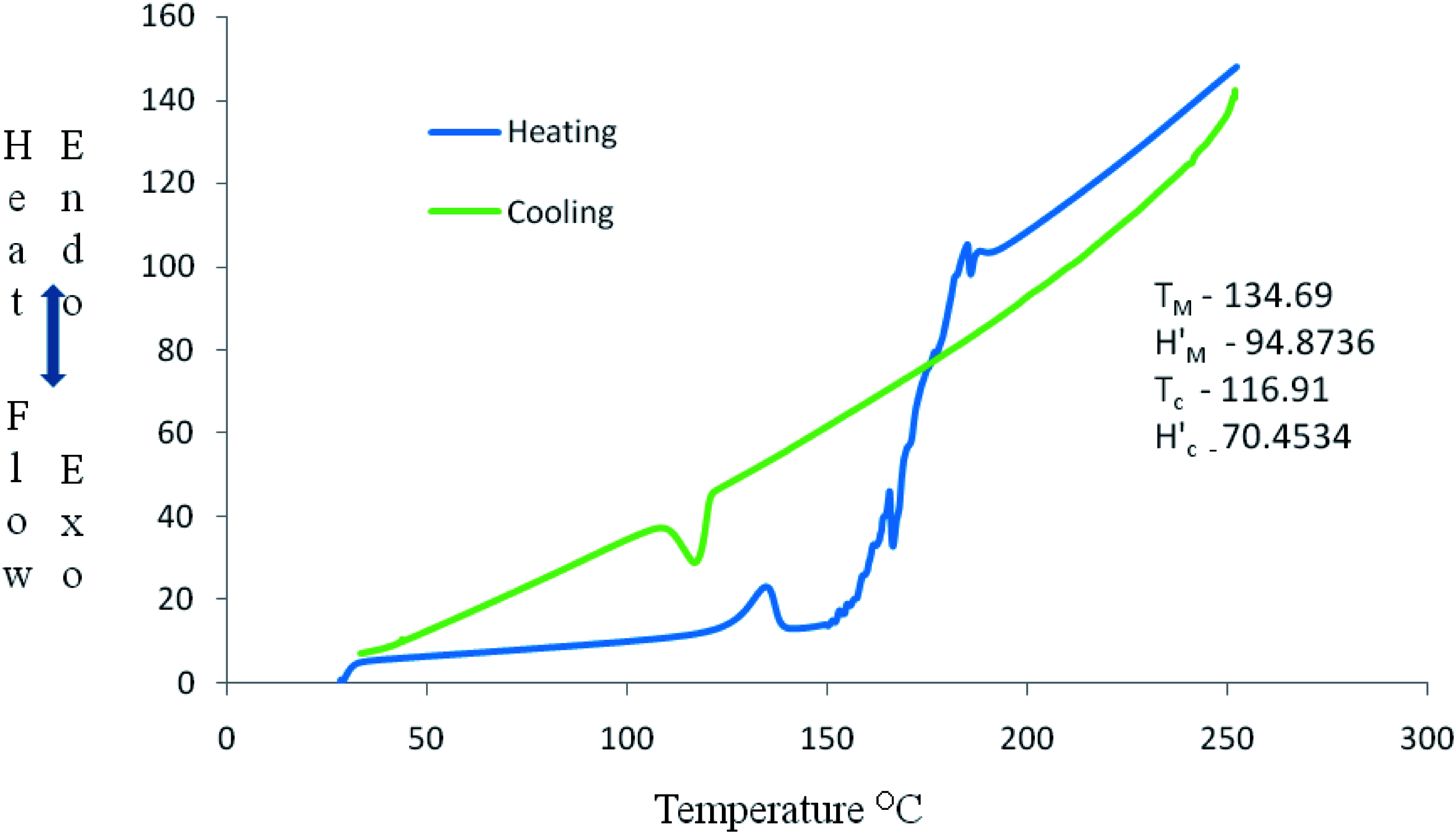
DSC curve in Figures 12–14 show some thermal parameters for the various systems. Figure 12 shows melting temperature of the unfilled system as 130°C and 180 J/g heat of fusion while Figures 13 and 14 show melting temperatures and heat of fusion of 138.73°C, 103 J/g, and 134.69°C, 67 J/g, respectively. Alkaline treatment of the shell increased the melting temperature of the compound and reduction in the heat of fusion of system B and C.
Perkin-Elmer Diamond DSC curve of system A. Perkin-Elmer Diamond DSC curve of system B. Perkin-Elmer Diamond DSC curve of system C.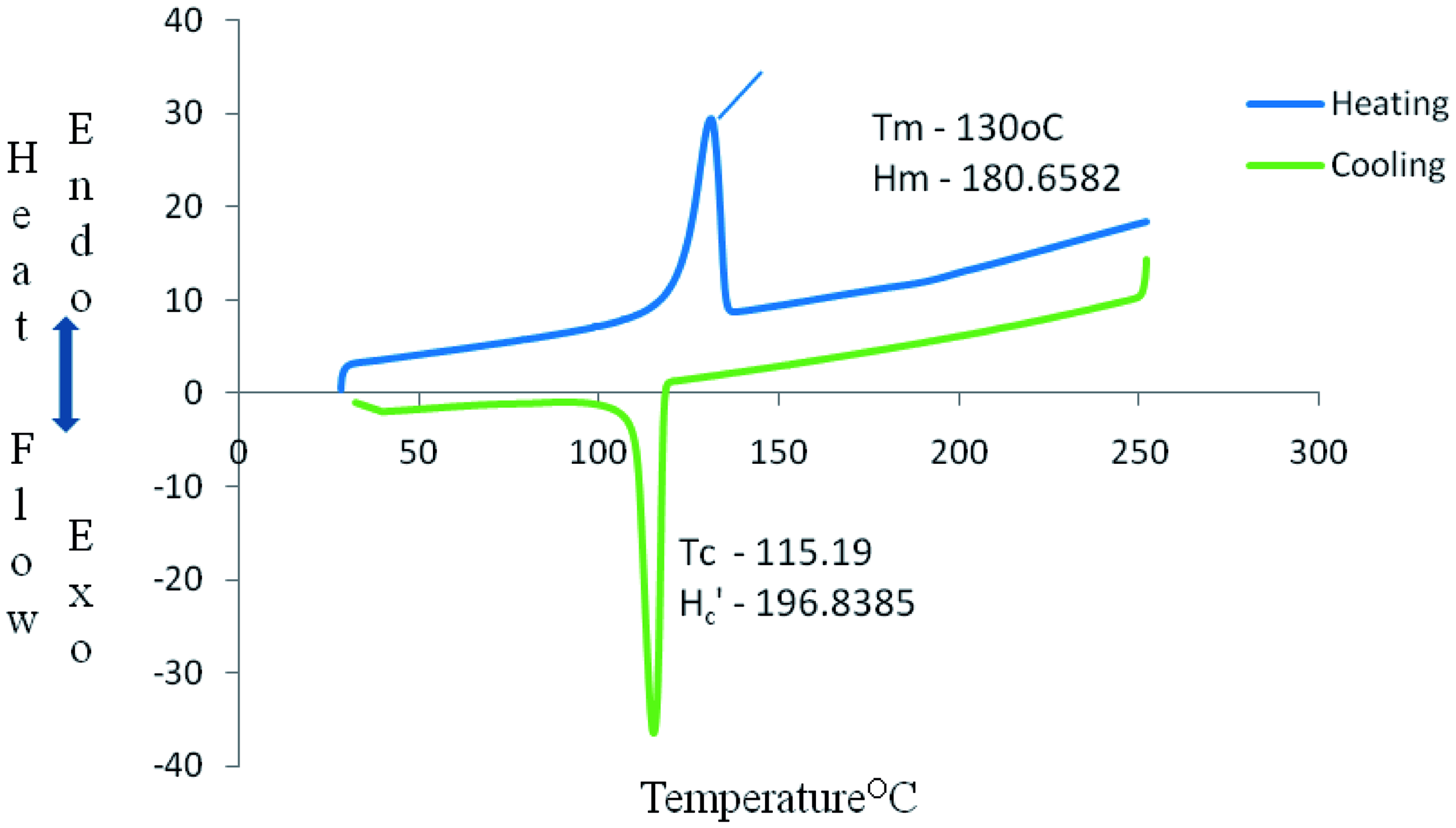
Microstructure
Figure 15(a) and (b) show the morphology of systems B and C. The micrograph showed that the fillers are uniformly dispersed in the HDPE matrix. Both the 500× and 1000× magnifications showed good filler–matrix interaction. The micrograph of system C showed in Figure 16 reveals good filler–matrix interaction. There was a coarse structure like void formation which may be the effect of the alkaline treatment of the shell particulates of the fabricated composite.
Magnifications of system B at (a) 1000× and (b) 500×. Magnifications of system C at (a) 1000× and (b) 500×.

CONCLUSION
The flammability and thermal characteristics of alkaline aqueous treated groundnut shell–HDPE composites were studied and the following conclusions drawn: Alkaline treatment of the groundnut shell reduced the effective heat of combustion of the composites when compared with the untreated shell. Other properties affected are specific extinction area (SEA), HRR, and TSP Mg(OH)2 fire-retardant additives have an impact on the system flame retardancy characteristics. Systems B and C burnt and left residues, as shown in Figures 10–11, while System A burnt and left no residue. The melting and crystallization temperatures of the various systems were marginally influenced by the alkaline treatment as systems B and C had melting and crystallization temperatures of 130.73°C, 115.67°C and 134.69°C, 116.91°C, respectively. DSC investigation showed that filler incorporation into HDPE does not affect the thermal properties of the fabricated composites significantly. SEM results showed good fillers–matrix compatibility. Micrograph obtained revealed a positive impact of alkaline aqueous treatment of filler in the fabricated composites.
