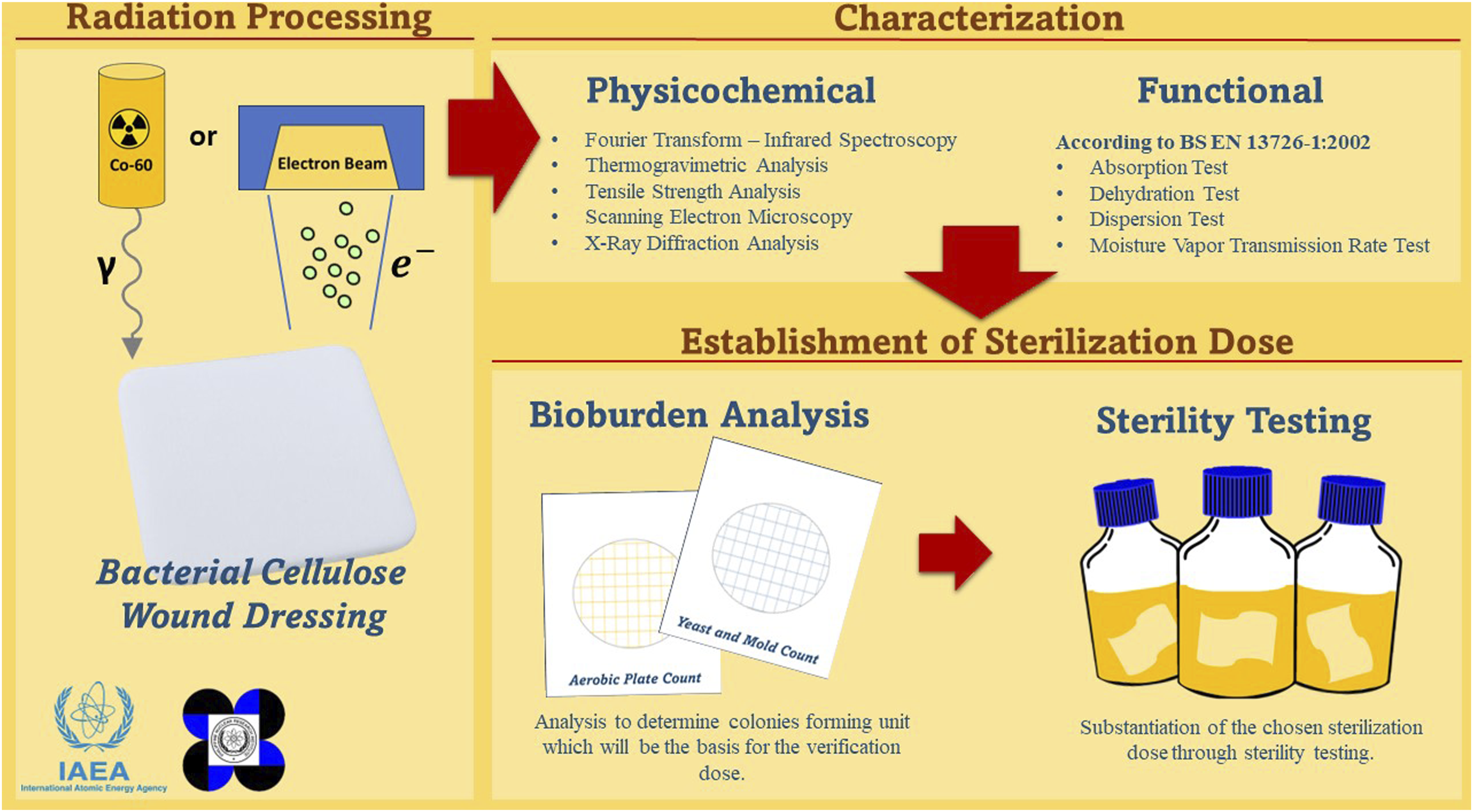
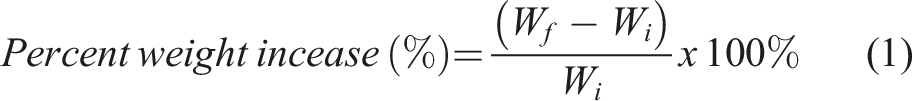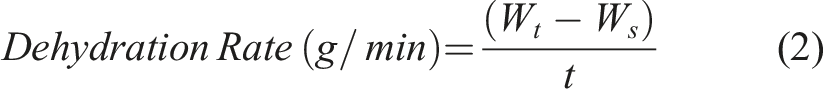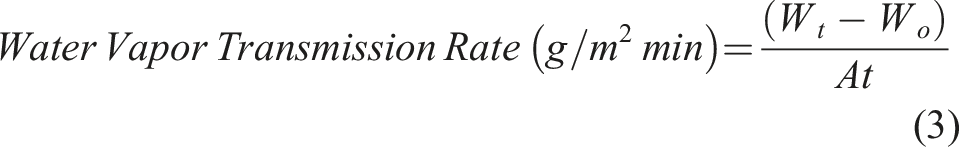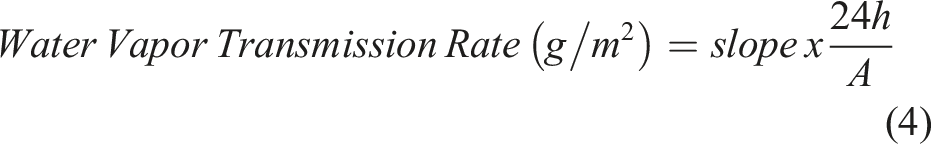Abstract
A commercial bacterial cellulose-monolaurin wound dressing was investigated for changes in the chemical structure, mechanical strength, thermal degradation, morphology, and functional swelling properties after exposure to gamma and electron beam radiations at doses 15-50 kGy. Radiation-induced oxidation occurred as seen in the FT-IR peaks at 1720-1750 cm−1. Degradation of the cellulosic network was observed in tensile strength reduction and shift in degradation temperature to lower values. The SEM cross-section images of the irradiated dressings revealed a less dense nanostructure network compared to the non-irradiated samples while the XRD diffractograms indicated a change in lattice direction/plane. Despite these changes, irradiation caused no significant effect on the functional properties especially at 15-25 kGy doses where most biomedical devices are sterilized. All irradiated wound dressings exhibited physical integrity, increased exudate absorption, and water vapor transmission rate - properties beneficial to wound-healing functionality. The pre-selected sterilization dose of 15 kGy for each ionizing radiation was successfully verified and substantiated following ISO 11137-2:2016, hence ionizing radiation is a suitable sterilization modality for the product.
Introduction
Bacterial cellulose (BC) is a well-known natural material of high purity due to the absence of hemicelluloses and lignin typically found in plant-derived cellulose. It is a glucose polysaccharide made up of units with (1-4)-glycosidic linkages naturally synthesized by various species of bacteria such as Acetobacter, Agrobacterium, Rhizobium, Sarcina using carbon sources like glucose, fructose, lactose, and others. 1 Bacterial cellulose has been used successfully in biotechnology and medicine. 2 It has excellent potential in wound-healing applications due to its unique properties, such as flexibility, high tensile strength, biocompatibility, excellent water retention, and absorption capacity.3,4 One characteristic that may limit its application as a wound dressing is the lack of antibacterial activity, which significantly prevents infection and promotes healing. 5
Monolaurin is a coconut lauric acid derivative that is well-studied for its antimicrobial properties. Lauric acid and monolaurin have a strong ability to destroy Gram-positive bacteria, especially S. aureus, fungi such as C. Albicans, and viruses including vesicular stomatitis virus (VSV), herpes simplex virus (HSV), and visna virus (VV). Lauric acid and monolaurin interact with specific functional groups located in the cell membrane and can cause damage to the cell. 6
Radiation sterilization contributes a significant share of about 45% in the industrial sterilization modalities of medical devices. Numerous advantages over chemical-based or heat-based sterilization techniques and safety concerns are driving factors to the widespread and increasing utilization of radiation sterilization. 7 Ionizing radiation such as gamma rays, electron beams, and X-rays constitute radiation sterilization techniques. Among these techniques, gamma irradiation is still the preferred method compared to e-beam and X-ray. Gamma rays have a higher penetrating ability than e-beam and X-rays; thus, many materials are sterilizable using gamma irradiation. 8 Conversely, the latter has been pursued increasingly due to the regulations surrounding the depleting supply of 60Co, the main source of gamma rays. 9 E-beam and X-rays should be suitable as an alternative to gamma irradiation based on both regulatory and scientific considerations. An example would be their higher energy output which effectively reduces irradiation time for mass sterilization of products. 7 Although radiation-based sterilization is already a mature technology that has been used for years, the lack of data on the resulting radiation effects on biomedical products impedes its adaptation. Therefore, documenting the effect of both modalities on a sample biomedical product, in this case, a bacterial cellulose-based wound dressing, will be beneficial to bridge this knowledge gap. While studies on the effect of ionizing radiation on bacterial cellulose-based materials have been published as early as 2009, 10 their suitability as a sterilization modality on these materials has yet to be studied thoroughly. Its effectiveness in reducing bacterial contamination while maintaining the wound-healing properties of BC must be established.
In this research, we investigated the effects induced by gamma and e-beam radiations on the chemical structure, mechanical strength, thermal degradation, morphology, and functional swelling properties of a commercial bacterial cellulose-monolaurin wound dressing and determined the suitability of radiation sterilization for this medical device. No studies conducted so far have specifically examined the impact of irradiation on the functionality of bacterial cellulose as a wound dressing nor its effect on the sterility of the biomedical device. Consequently, this study aims to provide valuable insights into the sterilization of emerging biomedical devices through radiation and make it an essential guide for future research in this field. This research gives an excellent opportunity to collaborate with the local company and to give evidence-based recommendations in considering radiation methods of sterilization, either by gamma or e-beam. This study also motivates to increase radiation sterilization activities in the Philippines; hence, it will open doors to future research collaboration with the first locally owned commercial e-beam irradiation facility on the radiation effects on polymeric materials commonly used in medical devices.
Methods
Bacterial cellulose-monolaurin wound dressing
The non-sterilized and retort-sterilized bacterial cellulose-monolaurin (BC-M) wound dressings, sold under the brand name CocoPatch, were provided by the local company PatchMed Cosmetic Trading. The dressings came in foil pouches packed in 3.5” × 3.5” sizes (Supplemental Figure S1). All reagents used were analytical grade, and all aqueous solutions were prepared using purified water.
Radiation processing
Gamma irradiation was performed in the Multipurpose Irradiation Facility of DOST-PNRI using the Ob Servo Sanguis 04-Gamma Irradiator with an activity of 8.3 kCi. Triplicate samples were irradiated with doses 15, 25, 35, and 50 kGy at a dose rate of 2 kGy/h. The absorbed dose mapping and process monitoring were done using an ethanol-chlorobenzene (ECB2021A) dosimetry system. The dose uniformity ratio for the process ranged from 1.077 to 1.095.
Electron beam irradiation was performed in the Electron Beam Irradiation Facility of DOST-PNRI. Samples were placed in conveyor carts and exposed to an e-beam of 2.0 MeV energy at currents ranging from 6.2 to 21.2 mA to deliver the doses 15, 25, 35, and 50 kGy. Samples were passed twice under the beam at once per side. The absorbed dose mapping and process monitoring were done using a B3 radiochromic dosimetry system. The dose uniformity ratio for the process ranged from 0.98 to 1.04. All irradiation was performed at ambient temperature.
Physicochemical characterization
Fourier-transform infrared spectroscopy
Freeze-dried wound dressings were directly examined via spectroscopic characterization using a Perkin Elmer Spectrum One FTIR spectrophotometer. Samples were pressed down against a diamond substrate and the infrared spectrum (600-4000 cm−1) was measured using Fourier-Transform-Infrared Spectroscopy-Attenuated Total Reflectance (FT-IR ATR). Principal component analysis (PCA) was performed on the spectral data (4000-500 cm−1) to quantify the variation in the spectra caused by the irradiation.
Thermogravimetric analysis
Five to 15 mg of freeze-dried samples were prepared, and their thermal properties were investigated using Netzsch STA449 F3 Jupiter. Thermogravimetric analysis (TGA) was performed over the temperature range of 30°C–1000°C with a heating rate of 10°C/min under a nitrogen atmosphere.
Tensile strength analysis
Tensile strength was measured using a Zwick Roell Universal Testing Machine equipped with a flat grip. The dressings were cut into dumbbell shapes (63 mm in length, 3 mm gauge width) before being subjected to 10 mm/min cross speed from the initial 25 mm gauge length. Five measurements were recorded for each sample.
Scanning electron microscopy analysis
The surface and cross-section morphologies of selected samples were analyzed using a Hitachi SU3800 microscope. The samples were attached to a sample stub with carbon tape for security while allowing the electrons to move to the ground.
X-ray diffraction analysis
The crystalline property of selected samples was identified using Shimadzu LabX-6000 with Cu radiation running at 40.0 kV and 40 Ma. The samples were continuously scanned over a range of 2° – 60° at 2θ and a rate of 1° per min.
Functional characterization
Absorption test
The absorption analysis was performed according to the BS EN 13726-1:2002, Part 1: Aspects of absorbency, Section 3.2: free swell absorptive capacities with slight modification.
11
The test solution, which served as pseudo wound exudate, was prepared by dissolving 8.298 g of NaCl and 0.367 g CaCl2 in 1.0 L Milli-Q water. The BC-M wound dressing was cut to 2 cm × 2 cm and weighed before submerging it in the test solution (1:100 w/v ratio). The submerged sample was incubated at 37°C and was periodically removed and weighed to determine the weight increase over time. The percentage of weight increase was calculated using equation (1), where Wi is the mass of the sample before immersion and Wf is the mass of the sample collected at a specific time.
Dehydration test
The 2 cm × 2 cm cut BC-M wound dressing was immersed in the test solution for 30 min and then removed and placed in the incubator at 37°C. It was weighed periodically to determine the weight loss caused by dehydration. The rate of dehydration was calculated using equation (2), where W
s
is the mass of the swollen sample before incubation and W
t
is the mass of the sample collected at time t (min).
Dispersion test
The dispersion analysis was performed according to the BS EN 13726-1:2002, Part 1: Aspects of absorbency, Section 3.6: dispersion characteristics with slight modification. 12 The 2 cm × 2 cm cut dressing was immersed in the test solution and vigorously swirled for 1 min using an Erlenmeyer flask. The solution in the flask was visually inspected to ensure the physical integrity of the sample and to check the presence of dispersed fibers from the dressing in the solution. Moreover, the UV absorbance characteristic of the solution was also measured at 200 nm to 400 nm using the Shimadzu UV-1800 UV/Vis spectrophotometer.
Moisture vapor transmission rate test
The water vapor transmission rate analysis was performed according to the BS EN 13726-2: 2002, Part 2: moisture vapor transmission rate of permeable film dressings with slight modifications.
13
A cylindrical vial with an inner diameter of 130 mm was filled with 20 mL test solution. For the vials with BC-M samples, the orifice of the vial with test solution was excessively wrapped by a 3 cm × 3 cm size dressing and the edge of the bottle was further sealed parafilm. In addition, the covered and uncovered vials with test solutions were used as positive and negative controls, respectively. All the vials were then incubated at 37°C and weighed at different periods for 12 h. The water vapor transmission rate (WVTR) was calculated using equation (3) where Wt and Wo are the initial weight of sample vials with test solution at time t and time before incubation, respectively, and A is the sample surface area. Alternatively, WVTR was calculated using equation (4) using the slope generated from the regression line of the weight loss versus time plot. The effective transfer area A is calculated using the inner diameter of the vial which is also equal to its orifice diameter.
Establishment of sterilization dose
Bioburden analysis
Thirty (30) dressings, 10 from each of the three independent production batches were used. Each sample was aseptically transferred to Butterfield’s phosphate buffer (pH 7.4), vigorously mixed for 60 s then serially diluted 10-folds and plated on Rapid Assay Kit Petrifilm™ (3M™ Microbiology Products, USA) to quantify the microbial load in the products. Incubation was done following the standard conditions: APC, 35°C for 48 h [15], and MYC, 20-25°C for 3–5 days.14,15 Colony-forming units (CFU) were counted, and the bioburden of the samples was evaluated. The overall average bioburden was calculated from the average batch bioburden values.
Recovery efficiency
Freshly prepared culture of Bacillus pumilus 1040 was adjusted using 0.5 McFarland standard (approximately 1.5 × 108 CFU/ml). Serial dilution of inoculum suspension was prepared such that two (2) mL aliquots contained approximately 15 to 150 CFU. Two (2) mL of inoculum suspension were inoculated to three (3) samples of BC-M and allowed to dry under a biosafety cabinet for 2 min. Then, 145 mL of phosphate buffer was aseptically transferred to samples and mixed thoroughly. Afterward, 1.0 mL of the dilution was transferred onto aerobic plate count (APC) rapid assay kits (Petrifilm™, 3M Microbiology Products, USA). The plates were incubated at 35°C for 24-48 h. The recovery efficiency was calculated by comparing the colony count of the organisms recovered to the positive control (pure isolates). Analyses were done in triplicate.
Substantiation of 15 kGy as sterilization dose by VDmax 15 method for multiple production Batches 16
Substantiation of 15 kGy as radiation sterilization dose (RSD) was done using the Verification Dose Maximum or VDmax 15 method, as provided by ISO 11137-2:2016. The bioburden-based verification dose setting was obtained from Table 10 of ISO 11137-2:2016. Two sets of ten (10) BC-M samples from one of the three batches were irradiated based on the required verification dose. After irradiation of each set at the verification dose, samples were analyzed for sterility.
Sterility test
The sterility tests were conducted in accordance with ISO 11737-2:2016. 16 All media glassware media bottles and forceps were autoclaved prior to the analysis. Each of the irradiated samples (SIP = 1) was transferred aseptically to a sterile media bottle with 200 mL soybean casein digest broth (TSB) and incubated at a temperature of 35 ± 2°C. Daily monitoring was done to record the number of positive results for a period of 14 days. The verification dose was accepted if no more than two (2) positive results from the ten (10) samples were obtained.
Statistical analyses
Statistical differences between the data from the irradiated samples and that of non-irradiated samples were calculated and evaluated. Tukey’s test was used at a 95% confidence level together with an analysis of variance at p < .05.
Results
The BC-M dressings were exposed to the same doses of gamma and e-beam radiation in the sterilization dose range commonly used in industry. The minimum and maximum doses were 15 kGy and 50 kGy, respectively. The effects of ionizing radiation were assessed based on changes in the chemical structure, mechanical strength, thermal degradation, morphology, and functional swelling properties.
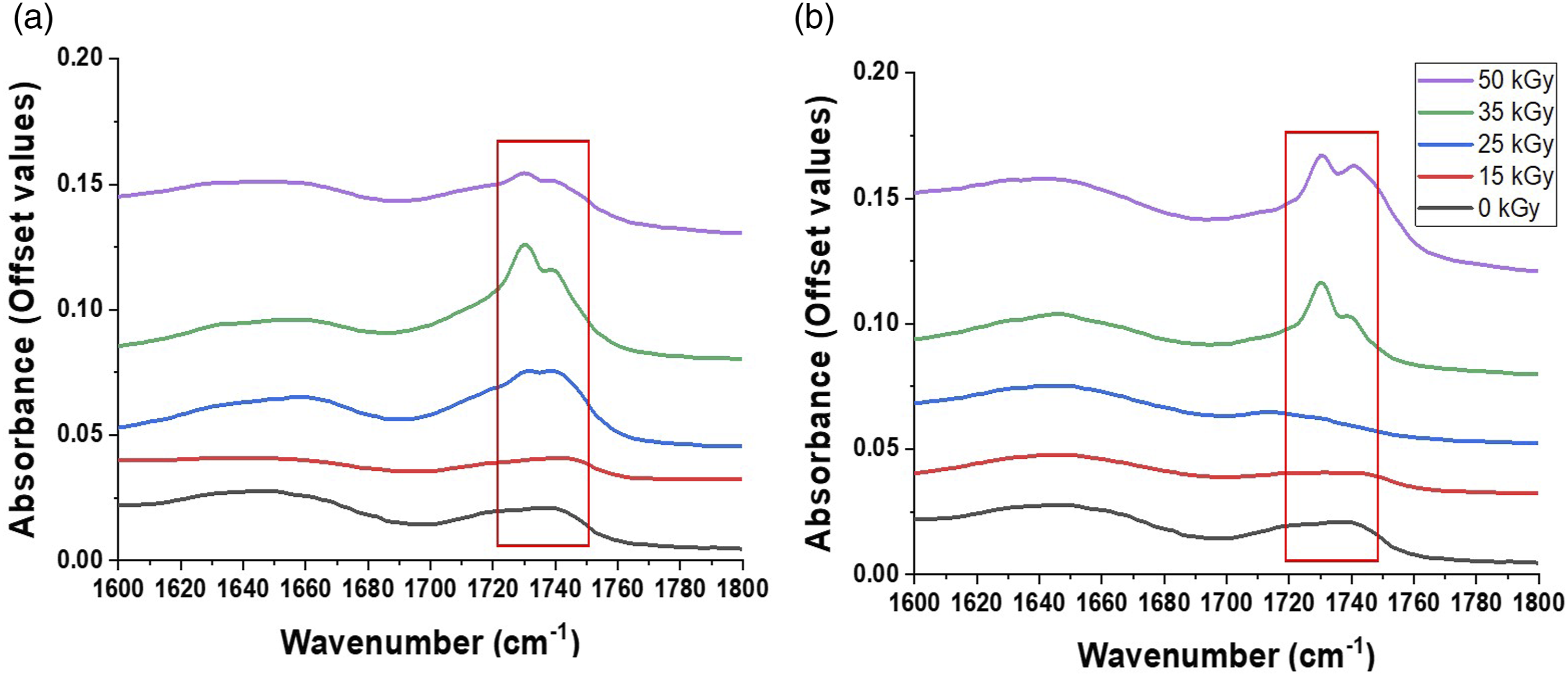
Analysis of the FT-IR spectrum of the non-irradiated freeze-dried BC-M sample showed the characteristic bands for cellulose, similar to published reports: 3346 cm−1 for O-H stretching, 2920 cm−1 for -CH2- asymmetric stretching, 1644 cm−1 for bound H2O bending, 1428 and 1109 cm−1 for C-OH, 1336 cm−1 for plane O-H, and 1057 cm−1 for C-O-C and C-OH elongation (Figure 1).17,18 The spectra of irradiated BC-M showed similar bands to that of the unirradiated sample except for the peak present within 1700–1750 cm−1 (Figure 2). The band in this range corresponds to the stretching vibrations of carbonyl groups that formed due to radiation-induced oxidation.
18
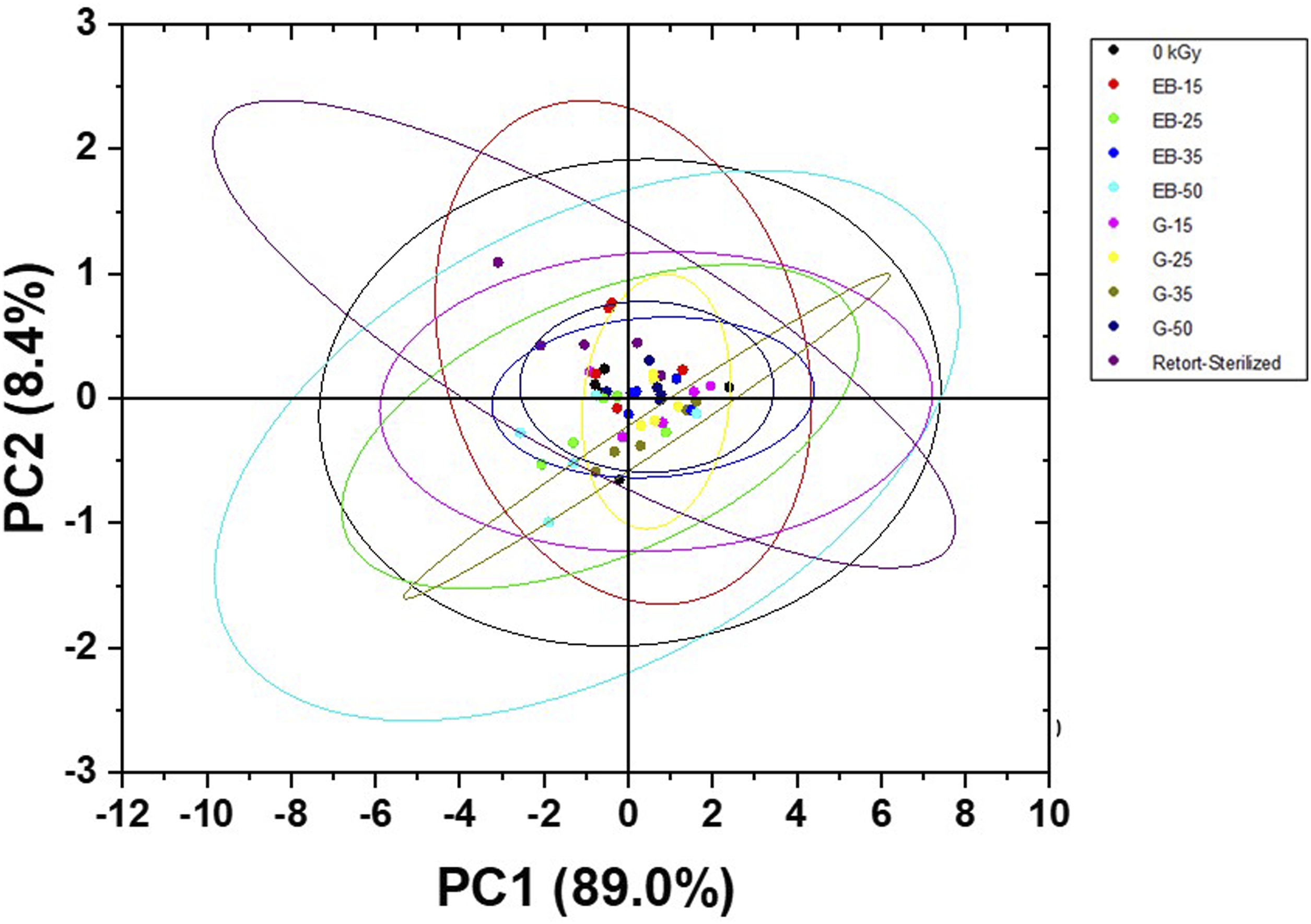
The intensity of the carbonyl peaks was more prominent beginning at 25 kGy in gamma-irradiated (Figure 2(a)) and at 35 kGy in e-beam irradiated samples (Figure 2(b)). The FT-IR spectra were further evaluated through a chemometric method called Principal Component Analysis (PCA). The score scatter plots (Figure 3) show percentage variability where the PC1s (89.0%) represent the most variability between spectra while the PC2s (8.4%) are for the set of characteristics with the second most variability. FT-IR spectrum of non-irradiated bacterial cellulose-monolaurin wound dressing. FT-IR spectra of irradiated BC-M samples at different absorbed doses of gamma ray (a) and electron beam (b) showing peaks of carbonyl functional group. Score scatter plots from the FT-IR spectrum of BC-M samples at different absorbed doses of gamma-ray and electron beam.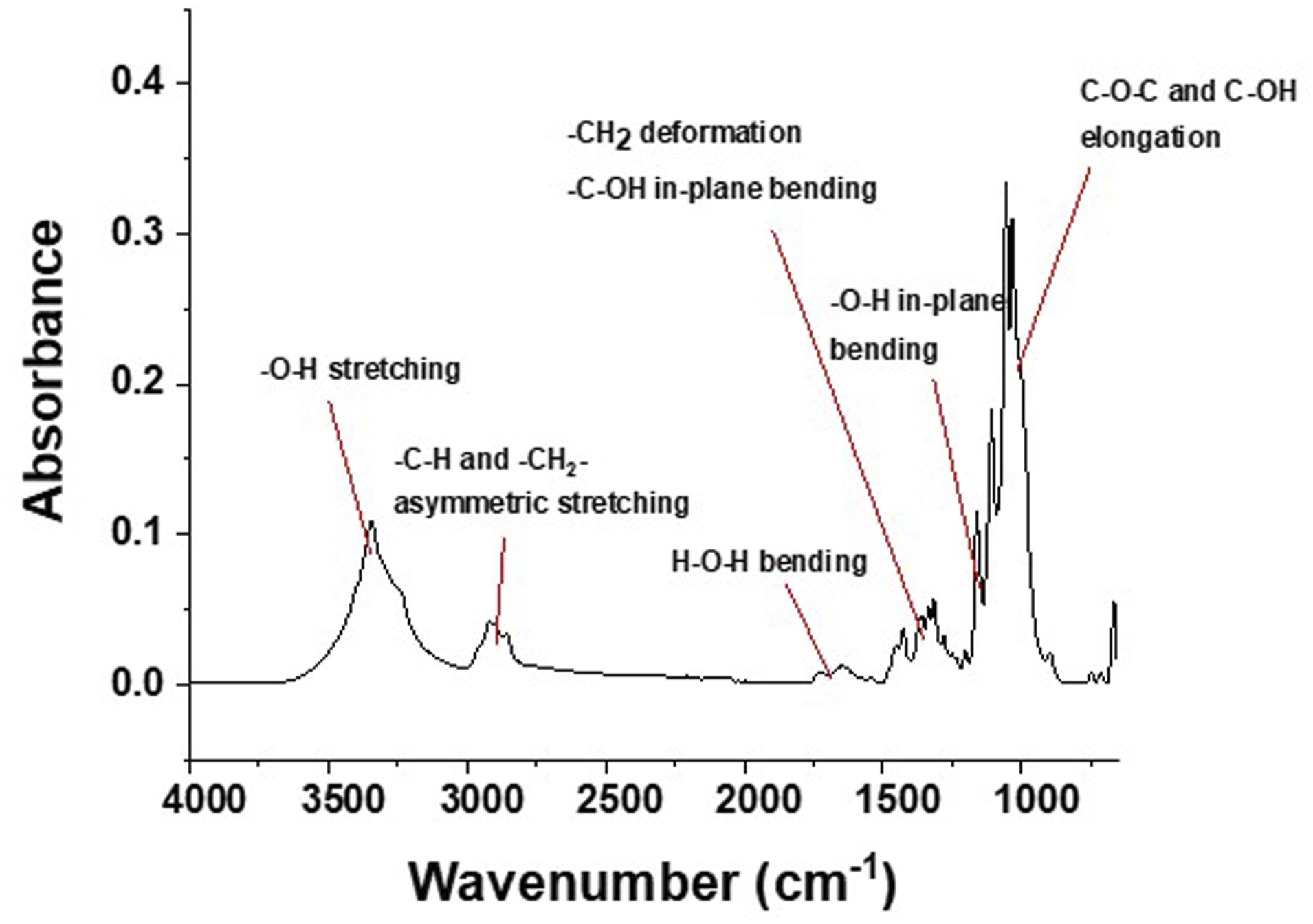
The tensile strength (Fmax) and elongation at break (%) properties are depicted in Figure 4. There was an observed decrease in the tensile strength of samples exposed to both ionizing radiations, but the decrease was less for e-beam treatment from 15 to 35 kGy compared to gamma treatment. Tensile strength and percentage elongation at break of BC-M samples which include retort-sterilized, non-irradiated (0 kGy), gamma ray-irradiated, and electron beam-irradiated at 15-50 kGy.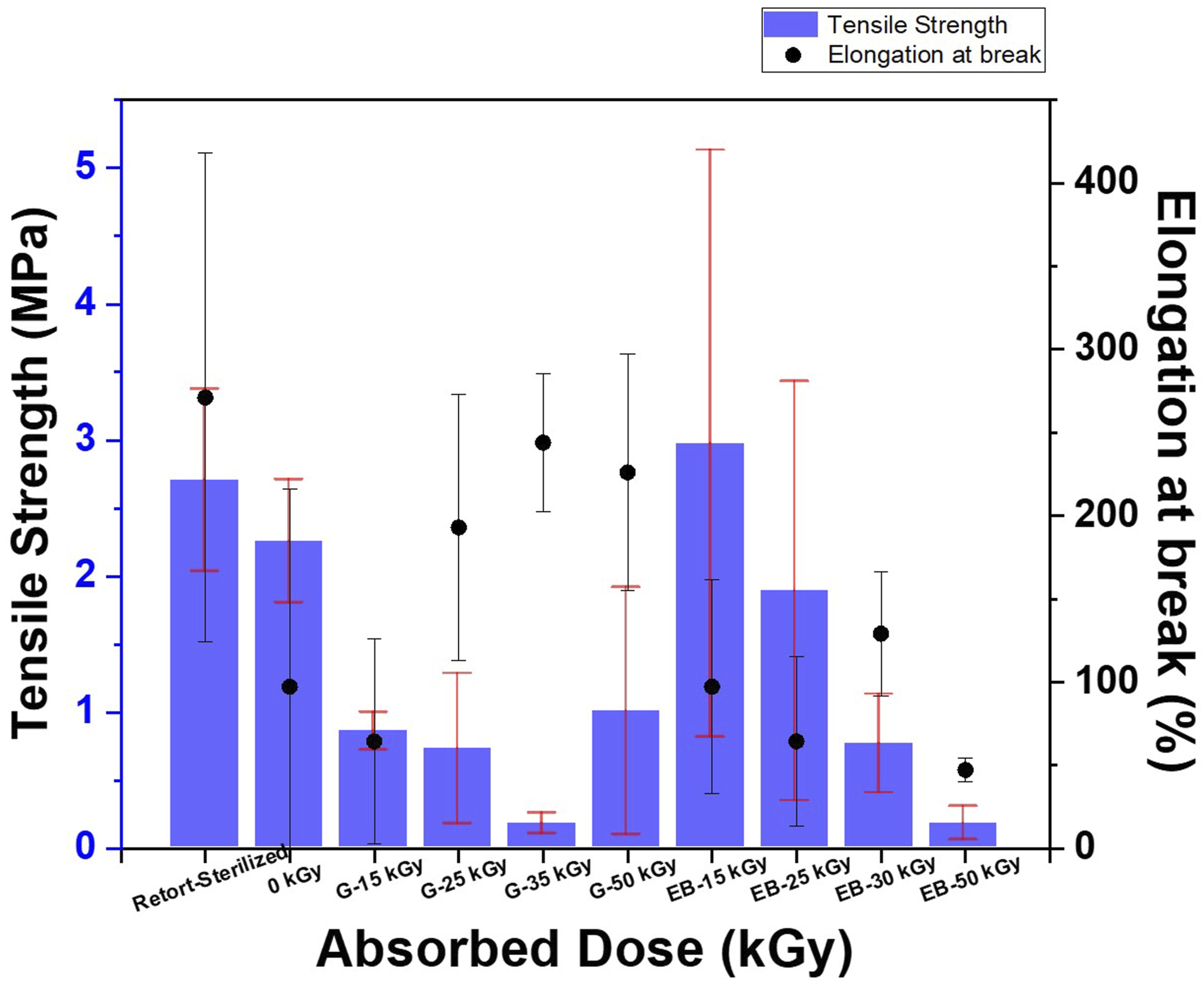
The TG curves of samples in Figure 5 showed regions of thermal degradation attributed to water evaporation (35-260°C), cellulose depolymerization and decomposition (260-400°C), and charred residue formation.
19
Irradiation shifted the decomposition onset temperature and temperature at maximum degradation rate (Tmax) to lower values (Supplemental Table S1) as well as increased the amount of charred residue formed in contrast to the non-irradiated sample.20,21 Thermograms of BC-M at all radiation doses of gamma (a) and electron beam (b).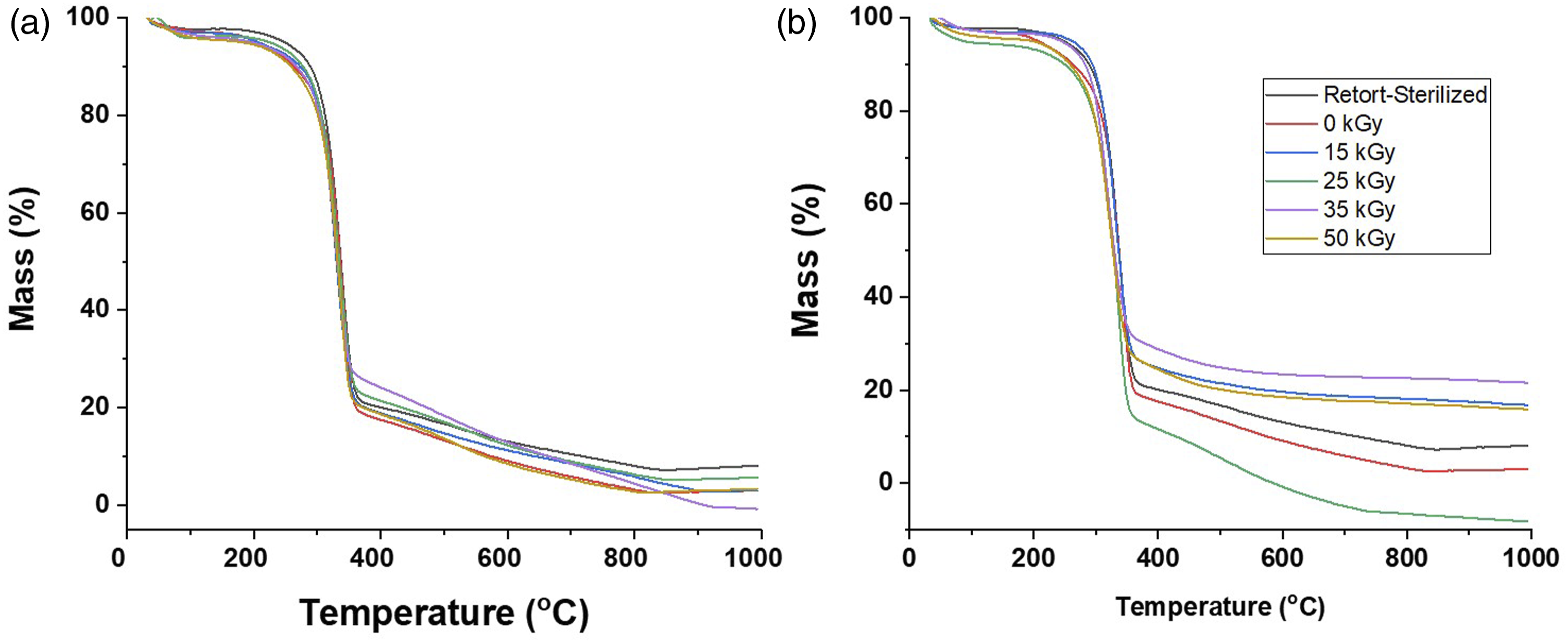
The SEM cross-sectional images of BC-M samples that were non-irradiated and irradiated at 15 and 25 kGy are shown in Figure 6. The alternating layers of bundled and loose nanofibrils, representing the crystalline and amorphous regions respectively, were clearly visible. SEM photos of non-irradiated (a) and irradiated BC-M samples at 15 kGy of gamma ray (b), 15 kGy of electron beam (c), 25 kGy of gamma ray (d), and 25 kGy of electron beam (e).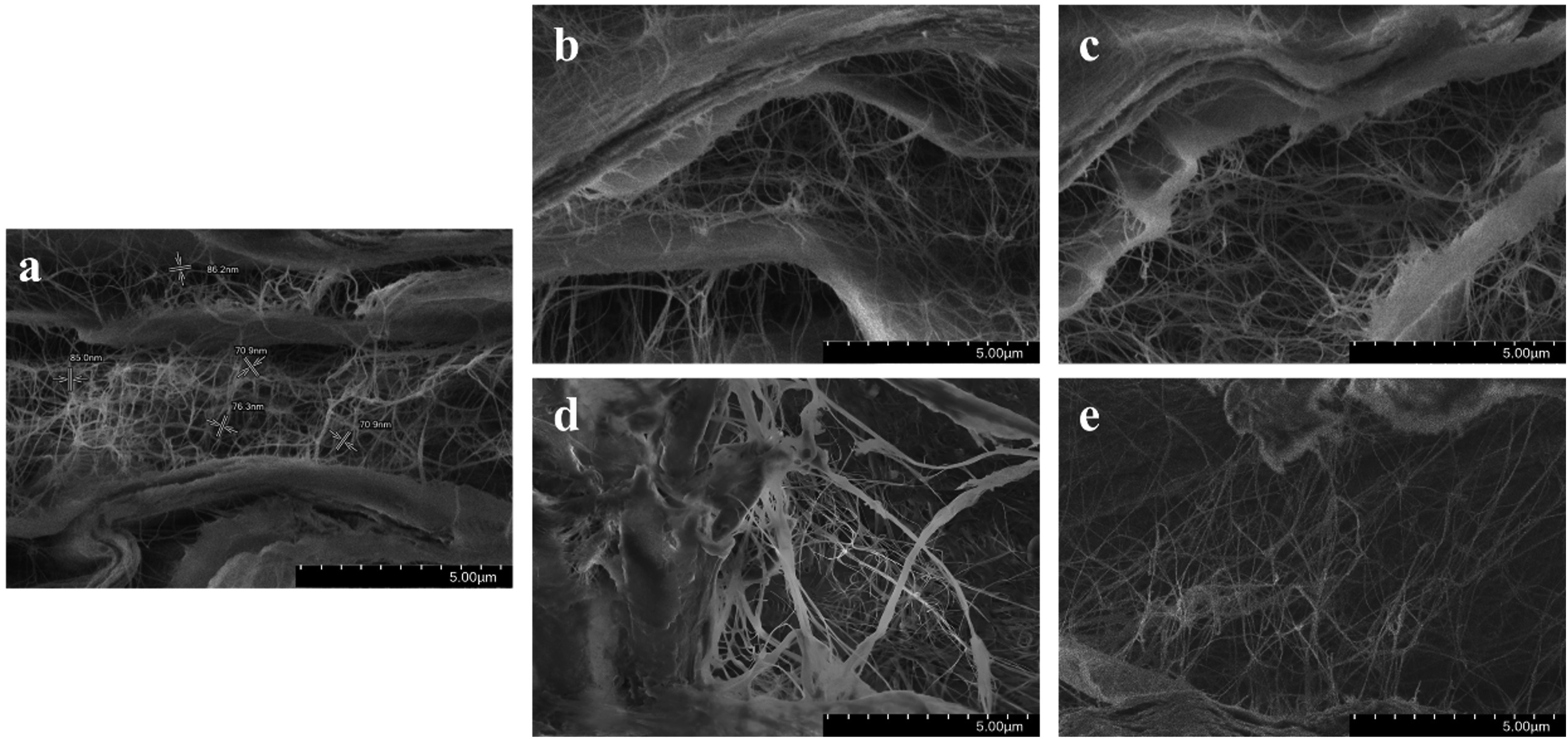
The XRD diffractogram pattern shown in Figure 7 corresponds to the typical peak profile of cellulose I allomorph.22,23 The prominent XRD peaks centered at 14.1°, 16.4°, and 22.5° are assigned to (1 0 0), (0 1 0), and (1 1 0) of Iα plane (triclinic) and/or (1 XRD diffractograms of selected BC-M samples: non-irradiated (0 kGy), gamma-irradiated (G-15 kGy, G-25 kGy), and e-beam irradiated (EB-15 kGy, EB-25 kGy).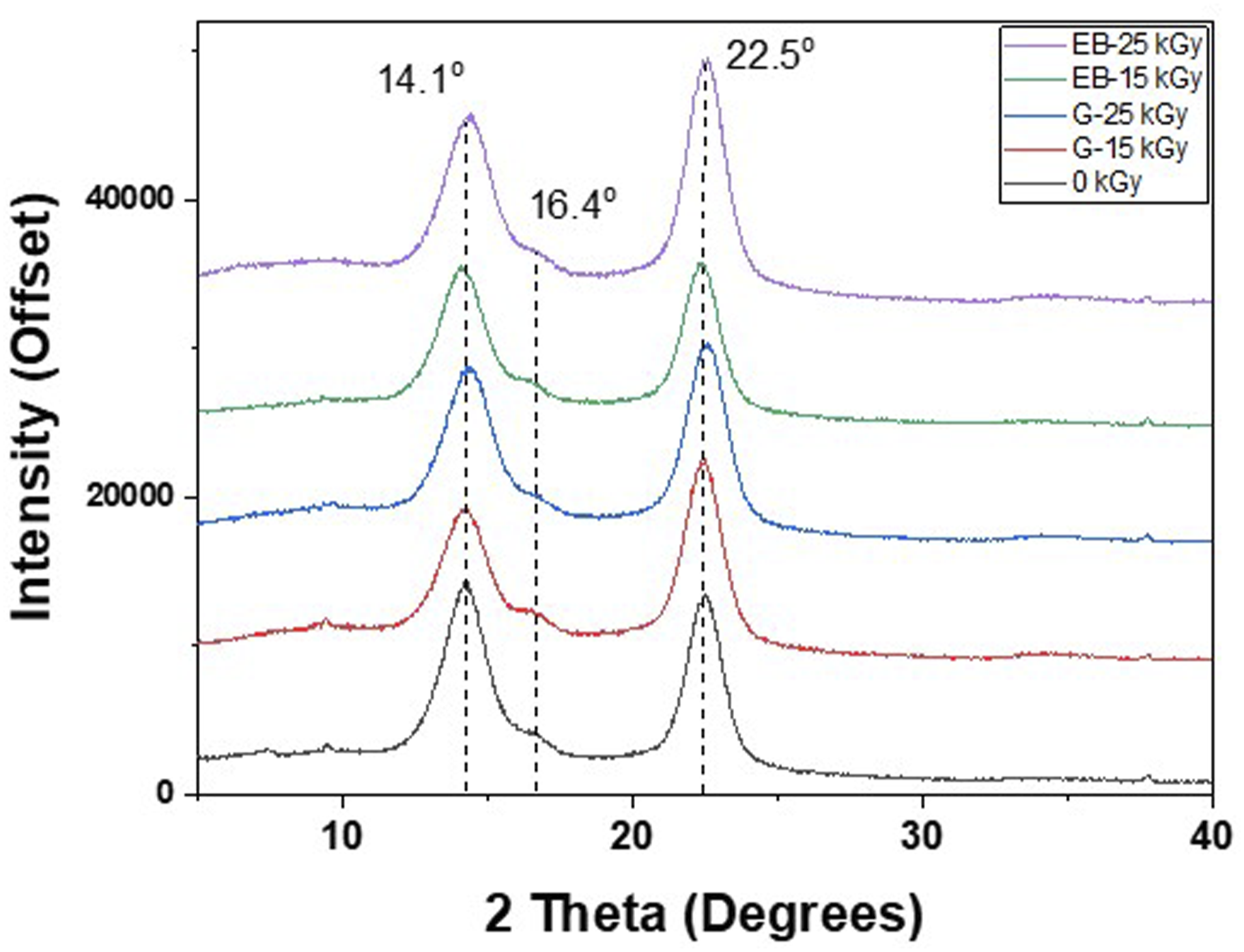
In addition to conducting physicochemical assessments, the functional properties of the dressings were also examined before and after the irradiation process. Sterilization procedures, while essential for preventing infections, can potentially alter the characteristics of the dressing. Therefore, it was imperative to assess and confirm that the dressing's ability to support the wound healing process remained unaffected after exposure to radiation. This approach aimed to guarantee that the dressing would continue to serve its intended purpose effectively in clinical settings, ensuring optimal patient outcomes and safety. Several functional properties were assessed including absorption and dehydration capacity, physical integrity (through dispersion analysis), and water vapor transmission rate.
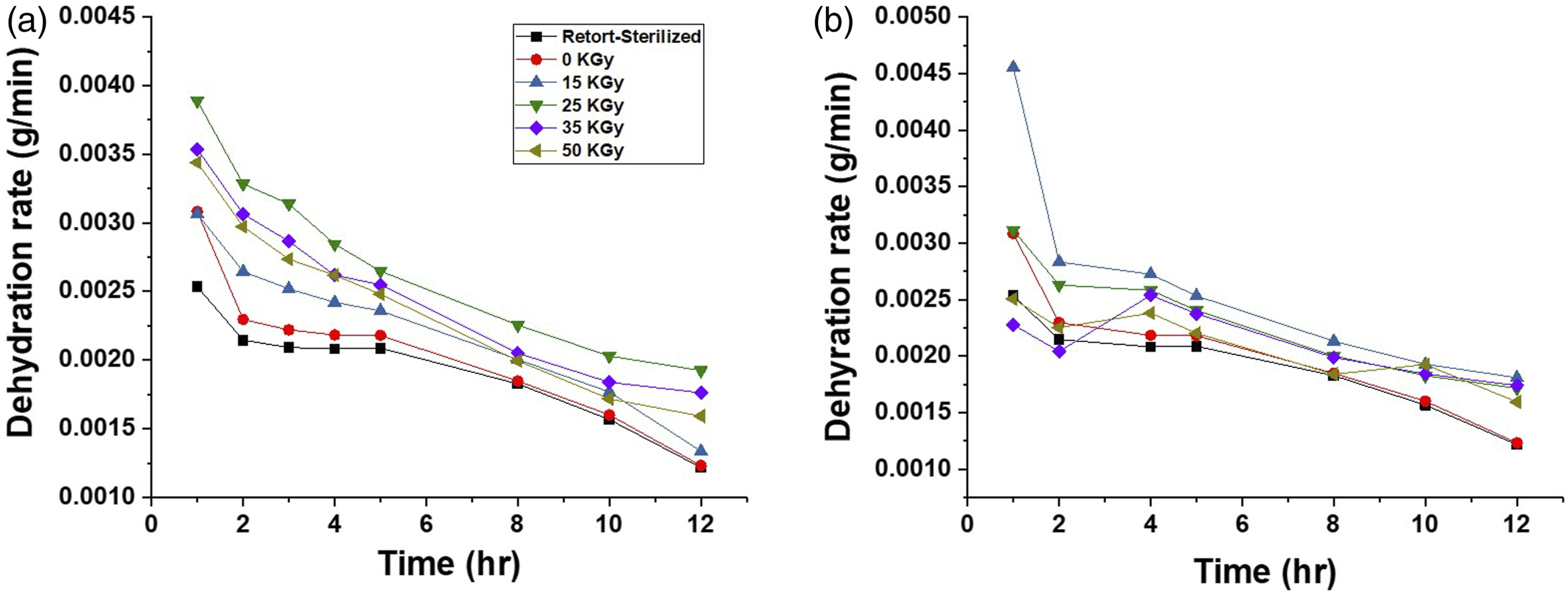
The non-irradiated sample had a 124% weight increase after 20-h immersions in the pseudo-wound exudate (Figure 8). Gamma and electron beam-irradiated samples, on the other hand, had higher weight increases with values between 161% and 268%. The absorption property of the BC-M dressing irradiated with the electron beam showed higher values compared to the gamma-treated and retort-sterilized samples. The dehydration rate also increased for all absorbed doses of gamma and electron beam compared to the control (Figure 9). The percentage weight increases of BC-M samples irradiated at different doses of gamma ray (a) and electron beam (b). The dehydration rates of BC-M samples irradiated at different doses of gamma ray (a) and electron beam (b).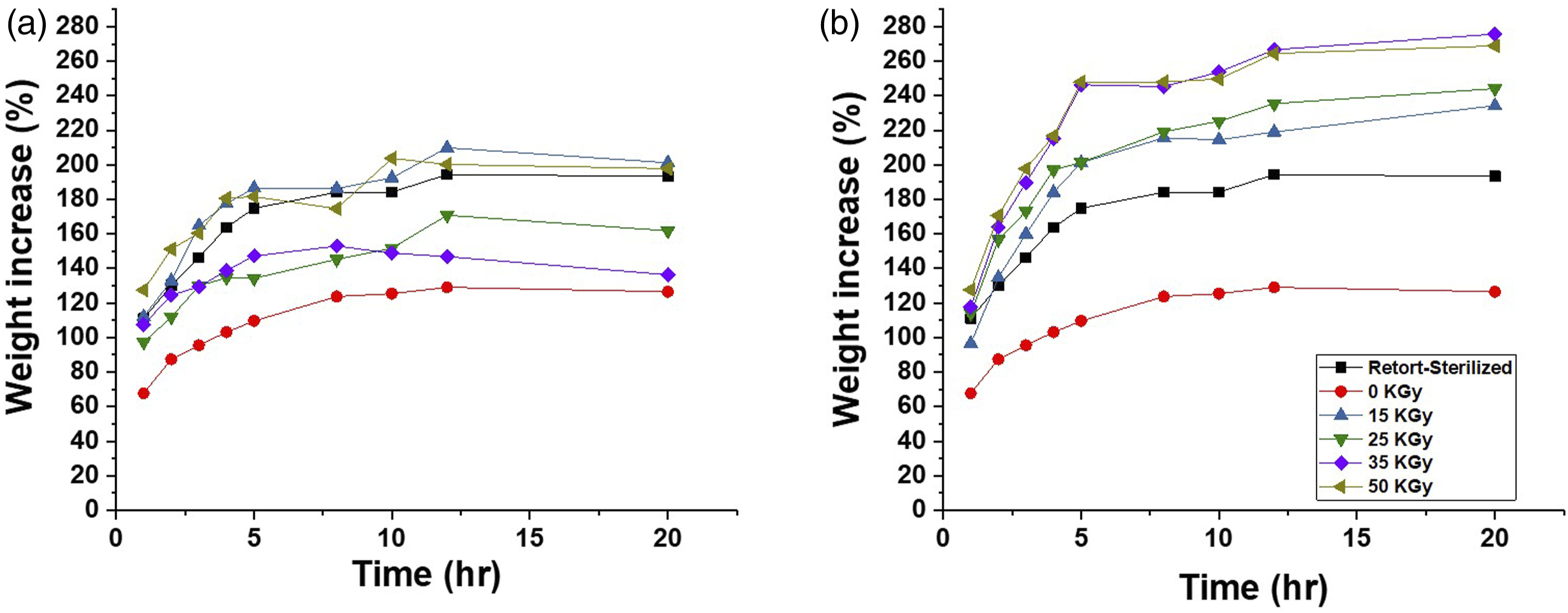
Pain-free removal is one of the most desired characteristics of wound dressings and removal of wound dressing can be eased if the physical integrity is maintained
26
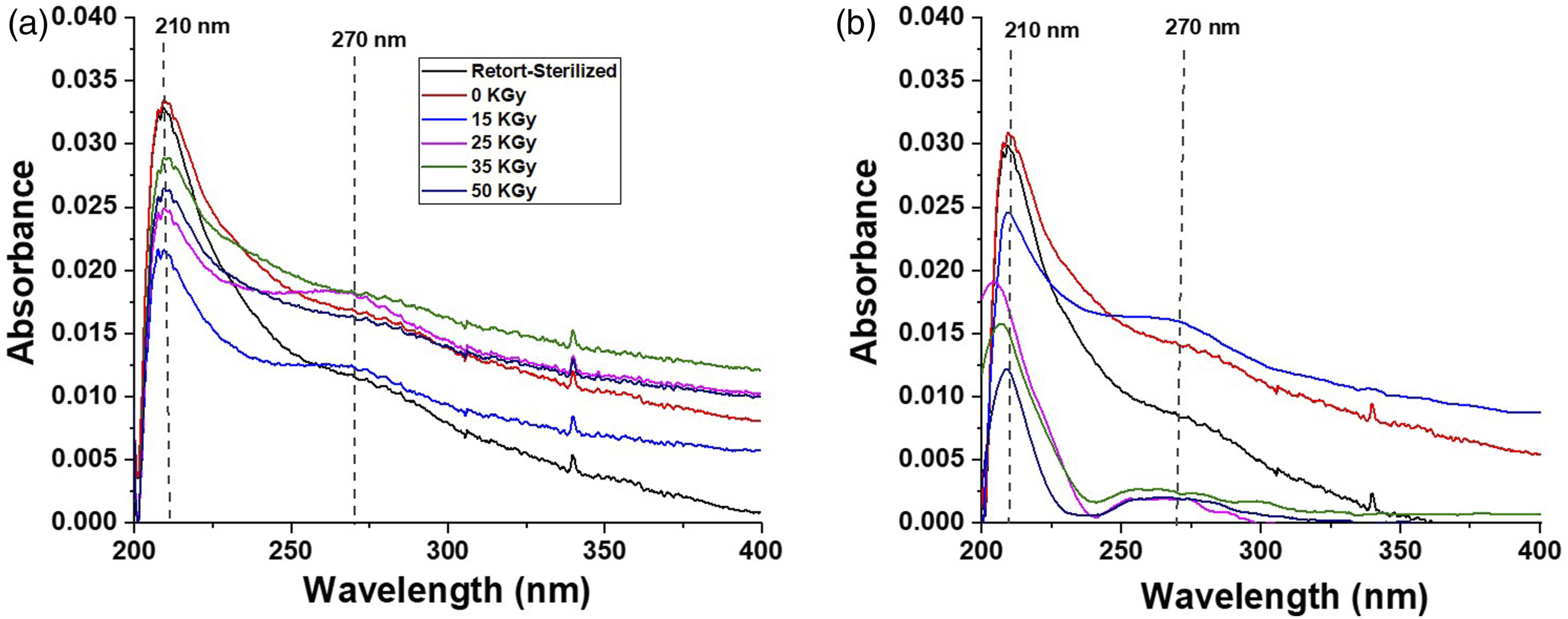
after the sterilization process. After being vigorously shaken, all BC-M samples remained physically intact and did not shred any visible fibers dispersed in the solution (Figure 10). UV analysis was used to show that the dispersing solution did not contain any peaks corresponding to the dressing. The UV spectra of all samples showed a strong absorption peak at 210 nm (Figure 11). Interestingly, a weak absorption peak at 270 nm was present in both the BC-M dressings irradiated with 15 and 25 kGy gamma rays, as well as for all electron beam-treated samples. Photographs showing the physical integrity of BC-M samples before and after vigorous shaking in the test solution. Samples include retort-sterilized, non-irradiated (0 kGy), gamma ray-irradiated at 15-50 kGy (a), and electron beam-irradiated at 15-50 kGy (b). UV spectra at 200 to 400 nm of BC-M samples irradiated at different doses of gamma ray (a) and electron beam (b).
An important function and requirement for wound dressings is keeping the wound at optimal moisture. Water vapor transmission rate (WVTR) forms an important part of the fluid handling properties of a dressing. Supplemental Figures S2 and S3 show how the WVTR of BC-M samples changed over the 12-h incubation period. All the samples exhibited higher WVTR compared to negative and positive controls. Moreover, e-beam irradiated dressings showed higher vapor transmission rates than the gamma irradiated samples.
The characterization results were employed to ascertain the absorbed dose at which the dressing can continue to function without significant deviation from the control. Based on the results, 15 kGy was chosen as the potential dose. To establish this as the radiation sterilization dose (RSD), a series of microbial assessments were performed according to ISO 11137-2:2016. 16
The bioburden analysis of 10 selected BC-M product items from three independent batches (a total of 30 samples) resulted in an overall average bioburden of 0.3 CFU/g. Based on this value, the verification dose (from Table 10 of ISO11137-2:2016) obtained was 1.3 kGy. Product items from one batch, 10 each for gamma rays and e-beam, were irradiated at 1.3 kGy and tested for sterility. All gamma-treated samples passed the sterility tests while one out of ten e-beam-treated samples turned positive for sterility. From these results, the verification dose was accepted and the 15 kGy dose was substantiated as the sterilization dose.
Discussion
It is well established that the predominant action of ionizing radiation on polysaccharides is degradation.10,27,28 The formation of carbonyl groups, possibly aldehydic or carboxylic form, was a consequence of the glycosidic bond cleavage and cellulose depolymerization.28–31 To determine whether this change was statistically significant, the FT-IR spectra were further evaluated through a chemometric method called Principal Component Analysis (PCA). This technique aims to find a small set of principal components (PC) that can describe the most variability in the spectra. PCA creates new variables called PCs derived from the recognition and highlighting of the physicochemical properties of the sample from the spectra, in this case, properties such as band diversity. 32 The score scatter plots (Figure 3) demonstrated clear grouping inside the confidence ellipse of the control; indicating that even after irradiation, the chemical changes were not significant to cause variation in the whole spectra. However, three replicates of the EB-50 kGy spectra were found outside of the control and this can be attributed to the presence of more carbonyl groups in the bacterial cellulose polymer.
Ionizing radiation also induced changes in the elasticity and mechanical strength of the samples. An increase in elasticity of the cellulosic membrane was apparent in gamma-irradiated BC-M from 25 to 50 kGy giving higher % elongation at break values compared to the unirradiated. Meanwhile, e-beam irradiation at doses 35 kGy and 50 kGy resulted in lower flexibility as the samples broke easily during the tension test. The reduction in the mechanical property of cellulosic materials has been established to be caused by main chain scission in both crystalline and amorphous regions in addition to the weakening and breaking of the hydrogen bond network during irradiation.18,33 Although the values decreased from 2.25 MPa (control, non-irradiated) to as low as 0.17 MPa, the tensile strengths of irradiated samples were still comparable to similar commercial wound dressings (0.04-0.53 MPa) reported by Minsart et al. (2022). 34 The result also suggested that e-beam irradiation at doses 15 and 25 kGy has the potential for BC-M dressing sterilization with no significant difference in the mechanical strength from the control.
The weakening of the BC-M polymeric network was also observed in both TGA analysis and SEM images. The decrease in Tonset and Tmax, shown by the thermograms (Figure 5), indicated that the depolymerization and decomposition of the cellulose dressing was initiated faster for irradiated samples. Irradiated BC-Ms also showed less dense structures and more separated cellulosic nanofibrils compared to the non-irradiated BC-M (Figure 5(a)). The loosening of the network (Figure 6) due to radiation may also be the cause of the change in the crystalline form of irradiated BC-M. As observed in the diffractogram the relative intensity of the 22.5 increased for all treatments but decreased for 14.1° (Figure 7). This is indicative of the change in lattice direction/plane from (1 0 0) to (1 1 0).
While these observations seem to signify an adverse effect of radiation on the samples, it is important to note that wound dressings are commonly used at room temperature; thus, it is also necessary to discuss the changes at the working temperature and the storage conditions. The change in the mass percentages of the dressings from 30 to 50°C was negligible (Supplemental Table S1) and these minimal mass losses were attributed only to water evaporation. This is evidence showing the feasibility of using ionizing radiation in processing BC-M wound dressings. To further substantiate this claim, the functional properties of the dressing were also characterized.
Two of the important functional properties of a wound dressing in the form of hydrogel or membrane are its exudate absorption capacity and dehydration rate. High and controlled absorption of exudates is a crucial factor to manage faster wound healing 35 but this should match with its dehydration rate to provide an optimal moist environment to induce wound healing. Since the BC-M dressing is a novel formulation, we can only compare the properties of irradiated samples against the control dressing provided by the company, PatchMed Cosmetic Trading. To determine how highly energetic radiation affects the absorption property of the bacterial cellulose-based wound dressing, the irradiated samples were immersed in the simulated exudate solution (Figure 8). The exudate absorption capacity of the BC-M dressings was enhanced due to the altered network structures caused by irradiation. This change in the structure also caused the higher dehydration rate of irradiated samples (Figure 9). The spaces created in the network induced by the radiation caused the easier absorption of water and it also weakened the capacity to hold the water; thus, increasing the dehydration rate.
The physical integrity of the dressing upon contact with wound exudate was assessed through exposure accompanied by shaking. Upon subjecting the BC-M samples to vigorous shaking, we observed a noteworthy outcome. These dressings remained entirely intact, without any visible shedding of fibers into the surrounding solution, as depicted in Figure 10. The UV spectral peaks of the solution were similar to those observed in earlier studies by Trabelsi et al. (2009) and Plermja et al. (2010).36,37 The weak absorbance at 270 nm in the UV/Vis spectrum of certain irradiated samples (Figure 11) was due to the presence of carbonyl groups and conjugated systems in the oxidized polysaccharides. 38 Polysaccharides, including cellulose, undergo radiolysis upon exposure to high-energy radiation, which leads to the formation of carbonyl groups and conjugated unsaturated systems. 29 Even though the physical structure of the dressings was visibly maintained, minimal shedding was observed through the spectra.
Studies have proven that a desirable microenvironment is essential for wound healing where an ideal moisture content is present. A moist wound environment has several benefits that result in faster and better quality of healing. The presence of moisture increases autolytic debridement, cell migration re-epithelialization, and collagen synthesis. It also decreases pain, inflammation, and scarring.39,40 WVTR plays a crucial role in assessing the fluid-handling capabilities of a dressing. It impacts not only the hydration of the wound environment but also affects the moisture levels in the adjacent tissues, which can be a concern, particularly in cases where maceration is a potential issue. 41
The calculated WVTR values based on the constructed regression line are presented in Supplemental Table S2. Compared to the unirradiated dressing, the WVTR was the same at 15 kGy but was lower from 25 kGy to 50 kGy for samples exposed to gamma radiation. Meanwhile, the values for the electron beam-irradiated samples were all higher relative to the non-irradiated BC-M samples. Normal intact skin has a WVTR between 200 and 300 g/m2 per day, while the water vapor loss for injured skin can range from 279 to 5138 g/m2 per day depending on the type of wound. 40 It was suggested that materials for wound dressing should have a higher WVTR value than the normal skin to avoid dehydration and accumulation of exudates between the wound and dressing which may result in infection. 42
Sterilization is a fundamental process in the manufacture of medical devices. It involves the removal or inactivation of all microorganisms present on the surface or in the product to achieve an acceptable SAL or sterility assurance level. A SAL of 10−6 is considered the standard for medical devices.43,44 Radiation sterilization relies on ionizing radiation to inactivate microorganisms and the radiation sterilization dose (RSD) must represent a sterility assurance level of 10−6, which defines a probability of 1 in 1000 000 that a device is not sterile. 45
In establishing RSD, a dose substantiation method for 25 or 15 kGy as a sterilization dose to achieve a 10−6 SAL is used. The method for 25 kGy applies to products with an average bioburden less than or equal to 1000 CFU/g, whereas the method for 15 kGy applies to products with a bioburden less than or equal to 1.5 CFU/g. 16
A bioburden analysis was performed to determine the population of viable microorganisms present on or in the bacterial cellulose-monolaurin wound dressings. This pre-sterilization assessment helped in establishing the required sterilization dose for achieving a 10−6 SAL. Aerobic Plate Count, as well as Yeast and Mold Count, were used to determine the number of bacterial and fungal colonies present. The entire product (Sample Item Portion or SIP = 1) was used to test thirty (30) BC-M dressings, ten (10) from each of the three independent production batches. The average bioburden per batch and the overall bioburden are given in Supplemental Table S3. A total of 9 CFU/g was obtained from the 30 samples, resulting in a calculated overall average bioburden of 0.3 CFU/g. The low bioburden values may be attributed to factors such as the GMP process, the boiling step involved in the processing, and the presence of monolaurin. Monolaurin has an antimicrobial ability due to its capacity to alter the bacterial cell envelope. 46 No correction factor was applied since the spore inoculation method has no recovery count from the samples. Considering that the average bioburden of the BC-M wound dressings was below 1.5 CFU/g and the influence of ionizing radiation on the physicochemical and functional properties was found to be inconsequential at 15 kGy, this dose was verified and substantiated through the VDmax 15 method.
Since each of the batch averages was less than twice the overall average, 16 0.3 CFU/g was used for determining the VDmax 15 . Referring to Table 10 in ISO 11137-2:2016, a verification dose of 1.3 kGy was prescribed. Subsequently, ten (10) samples were irradiated at this verification dose, and each sample underwent individual sterility testing. To meet the criteria for success, it was required that no more than one out of the 10 samples would test positive after a 2-weeks analysis period. 16 The outcomes of the sterility test are presented in Supplemental Table S4, revealing that only one out of the 10 dressings was found to be unsterile, thereby substantiating 15 kGy.
To prove further the effectivity of the established RSD for each radiation modality, 10 more sample dressings were irradiated at 15 kGy dose and tested for sterility. As expected, none of the irradiated dressings showed any signs of viable microorganisms during the 2-weeks incubation period (Supplemental Table S5). The pieces of evidence provided by this study showed the effectiveness of gamma and e-beam in sterilizing BC-M dressings, which should push manufacturers to consider radiation as an alternative sterilization modality.
Conclusion
Both gamma and e-beam radiations caused degradation of the BC-M cellulosic network. This was evident in the reduction of tensile strength values and loosening of the nanofibrils. The FT-IR showed the occurrence of radiation-induced oxidation at varying intensities, however, the PCA method revealed that oxidation to a carbonyl-containing group was not a significant structural change. Moreover, the reduction in the physical strength of the samples was not manifested in the functional properties, especially at doses 15 and 25 kGy. The irradiated samples exhibited improved exudate absorption and WVTR at the specified doses. Both properties can benefit the wound-healing ability of the dressing. The establishment of sterilization dose was accomplished following the VDmax 15 method. The average bioburden of three batches of BC-M dressings was determined to be 0.30 CFU/g. The dressings irradiated both by gamma and e-beam at the verification dose of 1.3 kGy passed the sterility tests. Hence, the VDmax 15 method effectively confirmed the pre-selected 15 kGy as the appropriate RSD. These encouraging findings from our study suggest that ionizing radiation, whether gamma or electron beam, may be considered a viable sterilization modality for the commercial bacterial cellulose-based wound dressings.
Supplemental Material
Supplemental Material - Radiation processing of bacterial cellulose-monolaurin wound dressing: Physicochemical effects, functional analysis, and sterilization
Supplemental Material for Radiation processing of bacterial cellulose-monolaurin wound dressing: Physicochemical effects, functional analysis, and sterilization by Charito Tranquilan-Aranilla, Jeric Flores, Alvin Kier Gallardo, Gina Abrera, Franklin Pares in Journal of Biomaterials Applications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the International Atomic Energy Agency (CRP F23035 Contract No. 24422).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.