Abstract
Background
Living donor hepatectomy is a major surgical procedure associated with significant postoperative pain. Effective analgesia is essential to enhance recovery and ensure donor safety. Traditional approaches such as epidural analgesia are effective but may raise safety concerns due to perioperative coagulopathy. Spinal analgesia using intrathecal morphine (ITM) has emerged as a potential alternative, providing long-lasting pain relief with a favorable safety profile.
Methods
This systematic review was conducted following PRISMA guidelines and registered in PROSPERO (CRD420251149887). PubMed and EMBASE were searched from January 2000 to September 2025 for randomized and observational studies evaluating spinal analgesia in living donor hepatectomy. Outcomes included pain intensity, opioid consumption, and complications. Study quality was assessed using the Oxford Centre for Evidence-Based Medicine (OCEBM) levels and the RoB 2 tool.
Results
The initial search revealed a total of 937 publications. After duplicate removal, 932 articles were eligible for screening from title and abstract, and 920 were excluded . The remaining 12 articles were then eligible for full-text review. Among these, 4 studies were excluded (abstract N = 1; letter to the editor or commentaries N = 1; no full text available N = 1; review N = 1). Eight studies involving 698 patients were included (seven randomized trials and one retrospective study). Spinal analgesia, mainly using 300–400 µg ITM, provided effective pain relief and reduced opioid consumption compared with intravenous patient-controlled analgesia, thoracic epidural analgesia, wound infiltration, and some fascial plane blocks. Adverse effects such as pruritus, nausea, and vomiting were common but mild and self-limiting; respiratory depression was rare.
Conclusions
Intrathecal morphine provides effective and durable postoperative analgesia in living liver donors, reducing opioid use and avoiding the risks of epidural catheterization. Despite promising results, evidence remains limited by small sample sizes and study heterogeneity. High-quality multicenter trials are needed to define optimal dosing and integrate spinal analgesia into multimodal enhanced recovery protocols for donor hepatectomy.
Keywords
Introduction
Living donor liver transplantation (LDLTx) is an established therapeutic option for patients with end-stage liver disease. Advances in surgical and anesthetic techniques have improved donor safety and perioperative outcomes. Nevertheless, donor hepatectomy, whether performed through open or laparoscopic surgery, and now also robotic approaches, remains a major surgical procedure associated with significant postoperative pain. 1
As with all major surgical procedures, adherence to Enhanced Recovery After Surgery (ERAS) protocols is recommended to optimize outcomes. Effective perioperative pain control is a key component of ERAS, as it facilitates early mobilization and rehabilitation, thereby reducing the risk of complications related to prolonged immobility. 2
In the context of living donor hepatectomy, the surgical approach itself has recently been recognized as an important factor influencing postoperative pain. In fact, donor open hepatectomy is associated with multiple sources of pain, including the large subcostal incision, rib retraction, diaphragmatic irritation, and visceral factors. 3 As a result, in recent years, laparoscopic donor hepatectomy has gained increasing popularity owing to its advantages, such as reduced intraoperative blood loss, lower postoperative opioid requirements, shorter hospital stay, and a lower incidence of complications. However, postoperative pain following laparoscopic donor hepatectomy remains a multifactorial issue, influenced by port-site placement, pneumoperitoneum, and the extent of abdominal dissection. 4
Moreover, living donors are usually healthy individuals with no concomitant diseases, making effective postoperative pain management a cornerstone of their perioperative care. Inadequate analgesia may compromise recovery and increase morbidity, highlighting the importance of safe and effective strategies in this population.
Selecting the most appropriate analgesic approach is therefore challenging, and requires a careful balance between efficacy, safety, and recovery outcomes. Commonly employed techniques include thoracic epidural analgesia (TEA), abdominal wall blocks, wound infiltration catheters, and intrathecal opioids.
Traditional strategies for postoperative pain control include intravenous patient-controlled analgesia (IV-PCA) and TEA. While epidural techniques provide effective pain relief, their safety in the context of donor hepatectomy is a matter of debate, particularly due to the risk of coagulopathy and epidural hematoma. 5 In this setting, spinal analgesia with a single-dose of intrathecal morphine (ITM) has emerged as an attractive alternative. A single preoperative dose of ITM can offer prolonged analgesia, reduce systemic opioid requirements, and avoid the risks associated with epidural catheterization. However, ITM is not without limitations, with adverse effects including nausea, vomiting, pruritus, and respiratory depression.6,7
Most available studies have investigated these techniques in patients undergoing non-donor hepatectomy or other major abdominal procedures. Evidence specifically addressing living liver donors, who represent a distinct population due to their young age, absence of comorbidities, and different pain thresholds, remains limited.
This systematic review aims to evaluate and synthesize the current evidence on spinal analgesia and other perioperative pain management strategies in living donor hepatectomy, with a focus on their efficacy, safety, and impact on short-term postoperative outcomes.
Methods
Data source and search strategy
This systematic review was conducted according to the recommendations of Preferred Reporting Items for Systematic Review and Meta-Analyses (Prisma). 8 It was prospectively registered with the International Prospective Register of Systematic Reviews (PROSPERO – number CRD 420251149887).
Search and selection strategy
Two independent reviewers (P.C and G.M) conducted a systematic review of the PubMed and EMBASE databases. The search strategy included the following key words and/or MeSH terms according to the controlled vocabulary of the databases sought: (((spinal anesthesia) OR (intratechal morphine)) AND (living liver donor)) OR (living liver hepatectomy).
Publications were retrieved from January 2000 to September 2025. The two independent reviewers (P.C., G.M.) selected the studies for inclusion. Any discrepancies regarding the relevance of the papers to include were resolved through discussion and consensus with an additional author (G.B). Nonetheless, there was agreement in the selection of papers between the two authors.
Eligible studies were randomized trials and observational studies in adult patients in which the spinal analgesia (Intervention) was used for pain management strategies in living donor hepatectomy (Population), assessed according to standard scales such as the Visual Analogue Scale (VAS), the Numeric Rating Scale (NRS) and other (Outcome), with or without comparison with other analgesic techniques or strategies (Comparator). Studies describing the use of spinal analgesia for non-donor hepatectomy or other major abdominal procedures were excluded, as were animal or cadaveric studies or studies in languages other than English. We also used a snowballing method, checking the bibliographic references or citations of included studies, but no additional records were found. From each study, the following data were obtained: authors’ names, year of publication, type of study, number of patients, spinal analgesia technique, drugs used, pain intensity in the postoperative course, and complications related to the intervention.
Quality assessment
The quality of evidence was evaluated using the Oxford Centre for Evidence-Based Medicine (OCEBM) 2011 Levels of Evidence methodology. 9
For each selected study, the risk of bias was evaluated by two authors (P.C. and G.M.), independently using the RoB 2 tool for randomized trials. 10 Certainty of evidence was assessed by four principal factors (risk of bias, inconsistency, indirectness, and imprecision) using the Grading of Recommendations Assessment, Development and Evaluations (GRADE) method. The certainty of the evidence was rated from high (i.e., we are very confident that the true effect lies close to that of the effect estimate) to very low (i.e., we have very little confidence in the effect estimate: the true effect is likely to be substantially different). Any discrepancies in bias assessment between the assessors were recorded.
Evidence synthesis and results
The initial search revealed a total of 937 publications. After duplicate removal, 932 articles were eligible for screening from title and abstract, and 920 were excluded . The remaining 12 articles were then eligible for full-text review. Among these, 4 studies were excluded (abstract N = 1; letter to the editor or commentaries N = 1; no full text available N = 1; review N = 1). The inclusion/exclusion process is reported in Figure 1.
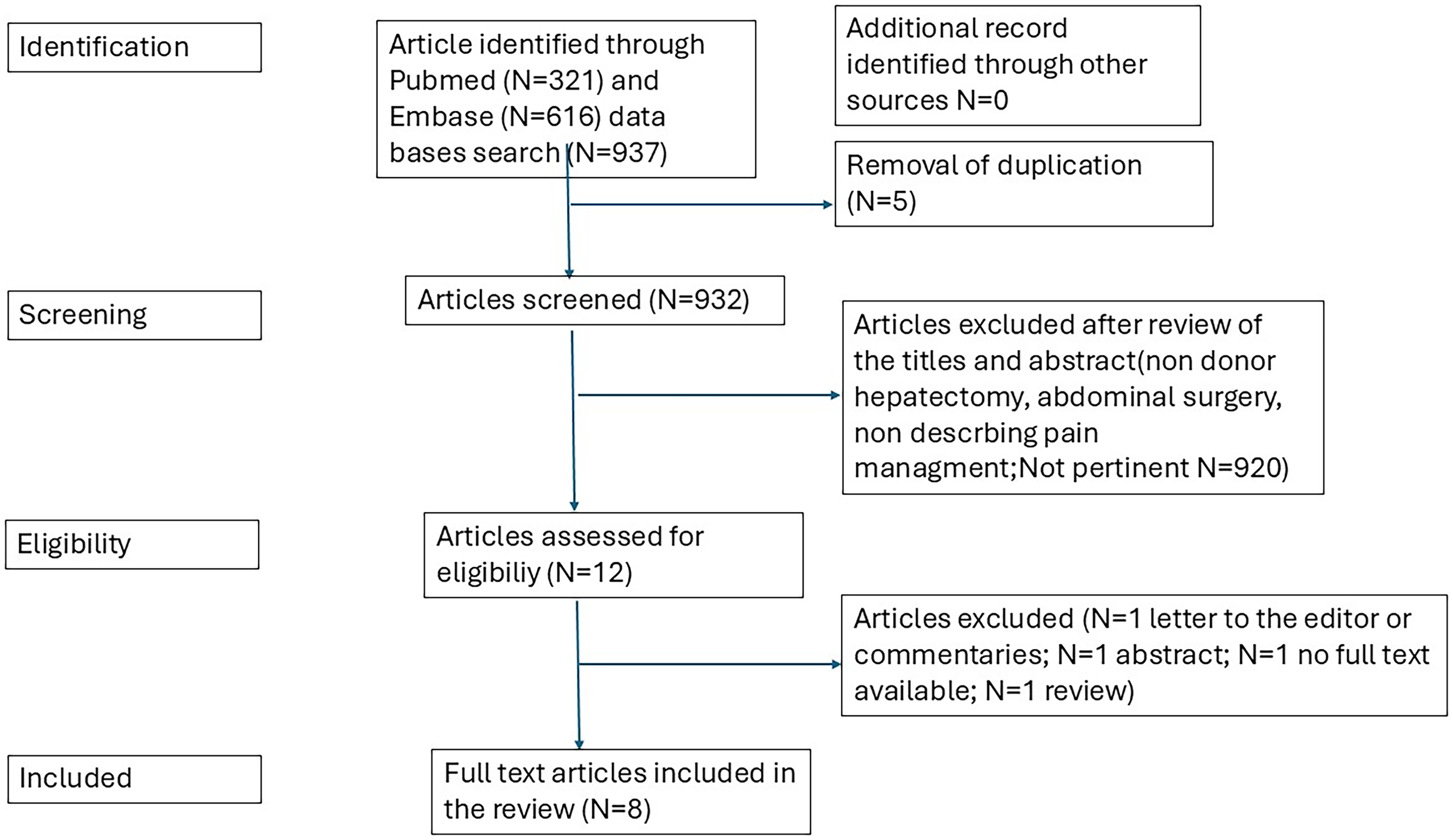
Flow chart selection process.
The overview and main findings of the included studies are summarized in Table I.
Summary of Studies Included in the Systematic Review.
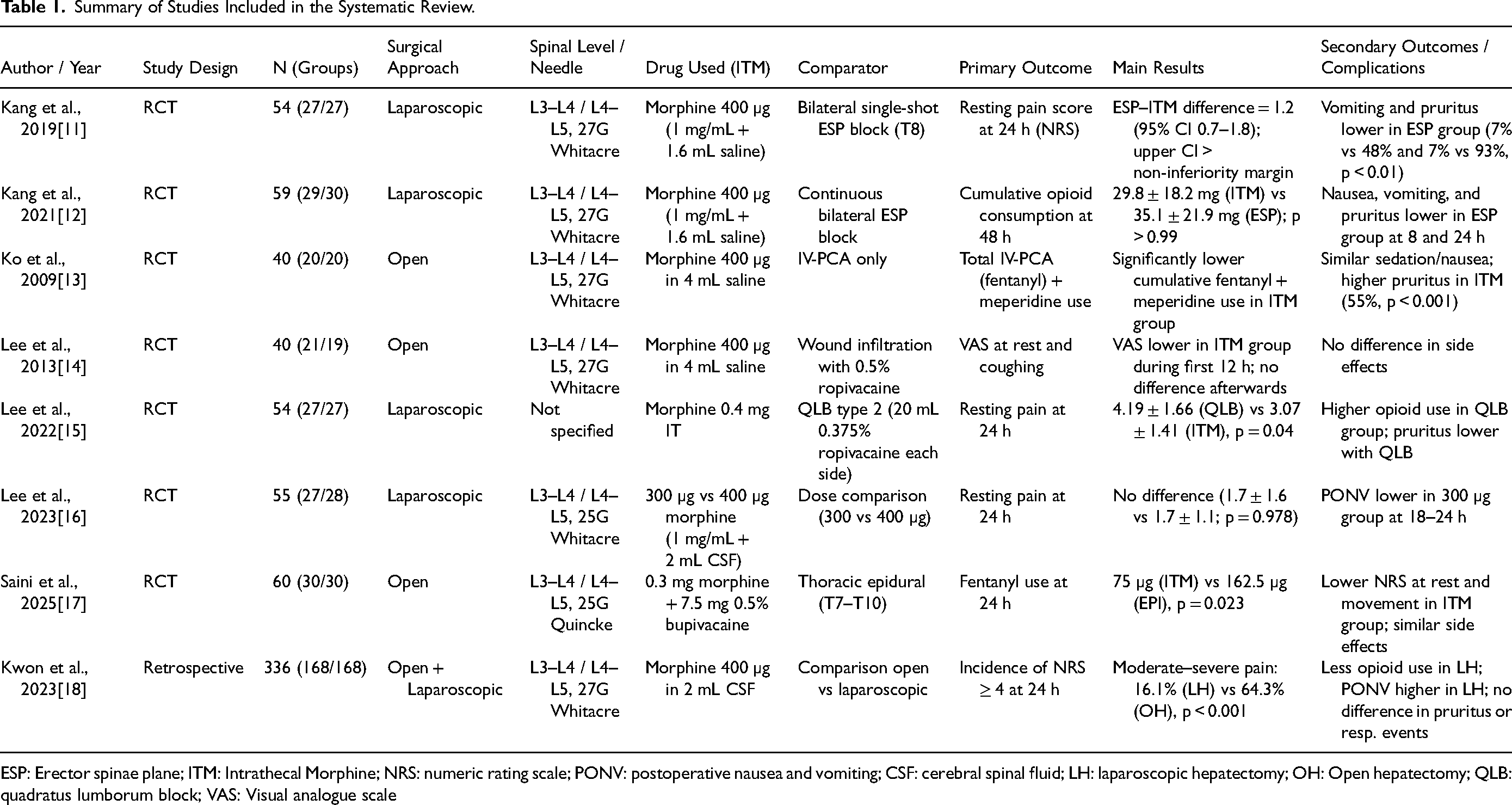
ESP: Erector spinae plane; ITM: Intrathecal Morphine; NRS: numeric rating scale; PONV: postoperative nausea and vomiting; CSF: cerebral spinal fluid; LH: laparoscopic hepatectomy; OH: Open hepatectomy; QLB: quadratus lumborum block; VAS: Visual analogue scale
Studies and risk of bias assessment
In accordance with OCEBM, level 2 of evidence was assigned to the seven randomized trials included in this review,11–17 and retrospective studies were classified as level 3. 18 Due to the clinical heterogeneity of the studies included, a meta-analysis was not feasible, so a systematic review of the studies was undertaken.
Figure 2 summarizes the assessment of risk of bias made for the seven randomized controlled trials included in this systematic review, using the RoB 2 tool. Overall, five studies (11-14,17) were judged to have some concerns in at least one domain.. The remaining two trials (15-16) were assessed as having a low risk of bias across all domains. A detailed domain-by-domain evaluation for each study is provided in Supplemental File 1.
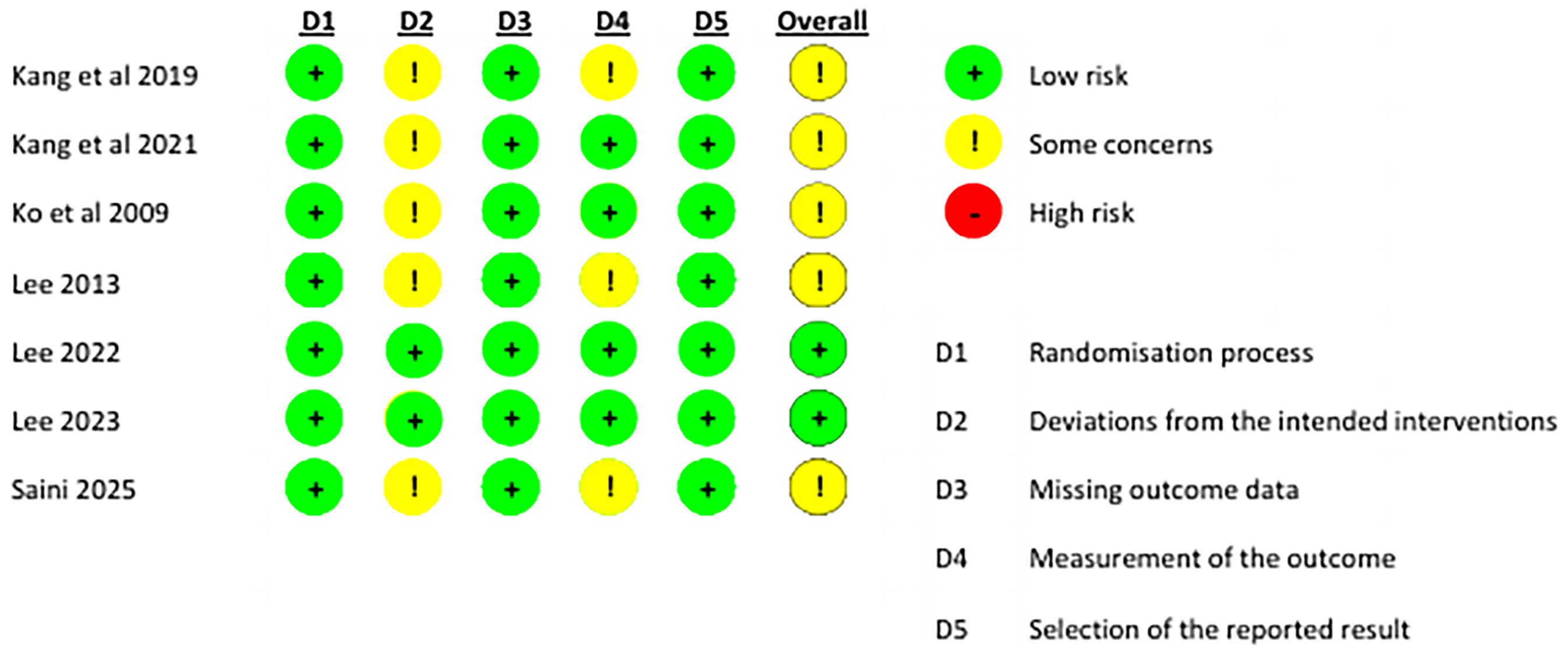
RoB 2 tool risk of bias assessment.
Study characteristics
Among the eight included studies, seven were randomized controlled trials (RCTs)11–17 and one was a retrospective study, 18 for a total of 698 patients. Of these, 545 received spinal analgesia, while the remaining 153 were managed with alternative techniques, including bilateral single-shot ESP block (n = 27), 11 continuous bilateral ESP block (n = 30), 12 QLB type 2 (n = 27) [15], epidural analgesia (n = 30), 17 wound infiltration (n = 19), 14 and conventional intravenous/PCA analgesia (n = 20). 13
In all included studies, spinal analgesia was performed at either the L3–L4 or L4–L5 interspace. A 27G Whitacre spinal needle was used in five studies,11–18 while two studies reported the use of a 25G spinal needle, Whitacre and Quincke, respectively.16–17 One study did not specify either the interspace level or the type of needle employed. 15
Regarding dosage, six studies reported the administration of 400 µg of intrathecal morphine, one study described the use of 300 µg, and one study compared two groups receiving 300 µg and 400 µg of intrathecal morphine, respectively.
Four studies reported the use of spinal anesthesia for living donor laparoscopic hepatectomy, two trials described its use for the open approach, and one trial compared the efficacy of ITM for the laparoscopic and open donor hepatectomy.
Spinal analgesia vs. IV-PCA analgesia
In a randomized trial, Ko et al. 13 compared the efficacy of ITM combined with IV-PCA with IV-PCA alone. They enrolled 40 patients undergoing donor right hepatectomy. The primary outcome measure was consumption of IV-PCA (fentanyl) and supplementary meperidine. Secondary outcomes were evaluation of pain at rest and when coughing using the VAS.
The required fentanyl was significantly less in the ITM IV-PCA group during the first 24 hours (407.3 [62.7] vs. 594.8 [209.3] mcg; p < 0.001), together with a lower consumption of meperidine 0 [0] vs.175 [90] mg; p < 0.001). In addition, the cumulative postoperative consumption of meperidine and IV-PCA were significantly less in the ITM IV-PCA group up to 72 hours.
Regarding secondary outcomes, patients in the ITM IV-PCA group had significantly less pain at rest and when coughing during the first 24 hours (p < 0.001).
The frequency of sedation, dizziness, or nausea did not differ between groups, but the authors reported a higher incidence of pruritus in the ITM IV-PCA group during the first 24 hours after surgery (55%; p = 0.001).
Spinal analgesia vs. epidural analgesia
In a randomized trial, Saini et al. 17 compared the postoperative analgesia provided by ITM and TEA in open living donor hepatectomy.
They enrolled 60 patients (ITM group n = 30; TEA group n = 30). The postoperative fentanyl consumption for the first 24 hours was significantly lower in the ITM group compared to the EPI group (75 mcg vs.162.5 mcg, respectively; p = 0.023). NRS scores up to 12 hours in the postoperative period at rest, on movement, and for shoulder pain were significantly lower in the ITM group compared to the EPI group (p = 0.000).
No significant differences in incidence of nausea and vomiting and pruritus were reported.
Spinal analgesia vs. fascial plane blocks
Spinal analgesia was compared to fascial plane blocks in three randomized trials.11–12,15 Kang et al. compared the analgesic efficacy of spinal anesthsia with both bilateral single shot and continuous ESPB in living donors undergoing laparoscopic hepatectomy.11–12
In the 2019 trial, 11 they reported lower resting pain scores 24 hours postoperatively in the ITM group compared with the single shot ESPB group (1.3 ± 1.1 for the ITM group (95% CI 0.8 to 1.7) and 2.5 ± 1.0 for the ESP group (95% CI 2.1 to 2.9). The highest and the average pain scores at PACU stay, as well as the resting pain scores at 8 and 24 hours postoperatively were significantly higher in the ESP group than in the ITM group, with no differences at 48 and 72 hours postoperatively. No differences were reported regarding opioid consumption at at 24, 48, and 72 hours postoperatively, while it was lower in ITM group during PACU stay. The authors reported lower incidences of postoperative vomiting (p = 0.002) and pruritus (p < 0.001) in the ESPB group.
The same group, in 2021, 12 compared ITM with continuous ESPB (CESPB) in another randomized trial. They reported similar cumulative 48-hour opioid consumption between the ITM and ESPB groups (29.8 ± 18.2 vs. 35.1 ± 21.9 mg, mean difference (ESP-ITM) (95% CI), 5.3 (11.5 to 22) mg; p > 0.99). Resting pain scores at 48 and 72 hours postsurgery were significantly lower in the ESPB group (0 [0–2] vs. 3 [1.5–3], and 0 [0–2] vs. 3,1–3 respectively, both p < 0.001), Also in this study, the ESPB group had significantly lower incidences of postoperative nausea, vomiting, and pruritus at all timepoints.
Lee et al. compared QLB type 2 with spinal analgesia for laparoscopic donor hepatectomy. 15 They reported higher resting pain score at 24 hour postsurgery in the QLB group (4.19 ± 1.66 in the QLB group and 3.07 ± 1.41 in the ITM group (n = 27, p = 0.04). Moreover, cumulative opioid consumption was significantly higher in the QLB group at 24 hours and 48 hours postsurgery. Though the ITM group exhibited a higher incidence of postoperative pruritus at all time points, there were no differences in other recovery outcomes.
Spinal analgesia vs. wound infiltration
Lee et al., in a randomized trial, analyzed the efficacy of ropivacaine wound infusion versus intrathecal morphine with intravenous fentanyl in living open hepatectomy. 14 The authors reported lower VAS scores in the first 12 postoperative hours in the ITM group while, at other times, VAS scores were comparable between the groups. In the wound infusion group, rescue IV fentanyl and meperidine use was significantly reduced 24 to 48 hours and 48 to 72 hours after surgery compared with the first 24 postoperative hours. Regarding complications, no differences were observed between groups, including PONV and pruritus.
Discussion
The principal finding of this systematic review is that spinal analgesia for pain management in living liver donors appears to be effective in providing satisfactory pain relief, even when compared with other techniques. Compared with TEA, fascial plane blocks, wound infiltration, and traditional intravenous analgesia, ITM provides equivalent or superior pain control, along with a reduction in cumulative opioid consumption. The evidence is derived primarily from randomized trials, though the total number of patients enrolled was small. However, there was considerable clinical heterogeneity regarding the techniques and comparator drugs, which represents a major limitation. This substantially reduces the feasibility of performing a meta-analysis, and limits the strength of conclusions regarding the effectiveness of this technique in this patient population. Moreover, with the exception of two trials (15, 16), some concerns remain following the risk of bias assessment using the RoB 2 tool for the randomized studies included in this review.
Living donor hepatectomy is a unique surgical event: it involves healthy individuals who voluntarily undergo a major surgical procedure solely for altruistic purposes. 19 Consequently, perioperative management must prioritize both patient safety and comfort, minimizing the physical and psychological burden associated with the intervention. 20 From this perspective, spinal analgesia offers several advantages.
A single-dose intrathecal injection of morphine provides reliable and long-lasting analgesia without the need for continuous infusions or catheter management. This simplifies perioperative care and may enhance patient mobility and satisfaction. Furthermore, the avoidance of epidural catheterization eliminates potential complications associated with coagulopathy, which can occur transiently after hepatic resection. 21 This concern is particularly relevant in living donors, where even minor risks must be minimized.
Importantly, the reviewed trials showed that spinal analgesia resulted in lower cumulative opioid consumption and better early pain control, both at rest and during mobilization. These findings support the incorporation of ITM-based analgesia within ERAS pathways for donor hepatectomy, where optimal pain control plays a crucial role in promoting early ambulation, reducing pulmonary complications, and facilitating prompt discharge. 2
Nevertheless, the use of intrathecal morphine is not without limitations. Adverse effects, especially pruritus, nausea, and vomiting, were frequently reported, though they were generally mild and self-limiting. Respiratory depression, a potentially serious complication, was rarely observed but remains a concern that justifies rigorous postoperative monitoring, particularly during the first 24 hours. 13 Thus, while ITM can replace or complement other analgesic strategies, its use must be coupled with structured monitoring protocols and staff expertise in managing opioid-related side effects.
Compared with TEA, ITM showed equivalent or superior pain control in several trials, with lower postoperative opioid consumption and comparable side-effect profiles. 17 The elimination of the risks associated with epidural catheter placement, including hematoma formation, hypotension, and technical failures further reinforces the value of ITM in this specific surgical population. Given that epidural analgesia requires meticulous management, and may be contraindicated in patients at risk of coagulopathy, spinal analgesia represents a pragmatic and potentially safer alternative. 22
Compared with fascial plane blocks, the results were less consistent and controversial. While ITM generally provided superior analgesia in the immediate postoperative period, one study found that CESPB could achieve similar analgesic efficacy, with fewer opioid-related adverse effects. 11 These findings suggest that the choice between ITM and fascial plane blocks should be individualized based on patient characteristics, institutional experience, and available resources. The potential combination of ITM with regional techniques also warrants exploration as part of a multimodal strategy to further optimize analgesic balance, and minimize side effects.
Wound infiltration and traditional intravenous analgesia were consistently less effective, providing higher pain scores and requiring greater opioid supplementation. This aligns with previous evidence that purely systemic analgesic approaches are insufficient in controlling the multimodal pain generated by donor hepatectomy, which includes somatic, visceral, and diaphragmatic components.23–25
Despite the overall encouraging results, this review identified several important methodological limitations that must be considered when interpreting the findings. A major limitation is the considerable heterogeneity among the included studies. There was variability in terms of the surgical approach (open vs. laparoscopic hepatectomy), ITM dosing (ranging from 300 to 400 µg), comparator techniques, and outcome measures used to assess pain and recovery. Such differences substantially limited the possibility of carrying out a meta-analysis, and hindered the formulation of definitive conclusions on the comparative effectiveness and safety of spinal analgesia in this context.
Another relevant aspect concerns the sample size of the included studies. Most trials were conducted on relatively small cohorts, reducing the statistical power to detect differences in less frequent but clinically relevant outcomes, such as respiratory complications or delayed recovery. Furthermore, though most randomized trials were classified as level 2 evidence according to OCEBM criteria, the quality assessment using the RoB 2 tool revealed “some concerns” in several domains, particularly regarding allocation concealment and blinding procedures. These methodological limitations emphasize the need for more rigorously designed trials to strengthen the evidence base in this field.
Insight for future research
Future investigations should aim to address the limitations identified in this review by implementing standardized protocols for analgesic administration, outcome measurement, and adverse event reporting. Large, multicenter randomized controlled trials are needed to confirm the observed benefits of ITM, and define the optimal dosing regimen that maximizes analgesic efficacy while minimizing adverse effects.
Moreover, comparative studies exploring multimodal approaches that integrate ITM with other regional or systemic analgesic strategies could conceivably offer valuable insights into more balanced and individualized pain management. The incorporation of objective recovery metrics, such as time to ambulation, length of hospital stay, and patient-reported quality-of-recovery scores, would also allow for a more comprehensive assessment of the true clinical impact of spinal analgesia in living donors.
In addition, with the growing popularity of fascial plane blocks in the context of major surgery, 26 large randomized controlled trials are warranted to evaluate their efficacy both in comparison with spinal analgesia and in combination with it as part of multimodal analgesic regimens. Given the specific characteristics of living liver donors, who may experience transient hepatic dysfunction and related coagulation abnormalities after hepatectomy, fascial plane blocks could represent an even safer alternative from a hematologic standpoint.
Though the risk of serious complications such as neuraxial hematoma is markedly lower with single-shot spinal analgesia compared to thoracic epidural analgesia,27–30 this risk is theoretically precluded with fascial plane blocks, which are performed in more superficial and compressible tissue planes.31–33 This fact is of particular importance in the setting of living donation, where the patient is a healthy individual undergoing a high-risk procedure solely for altruistic purposes, and where even minimal iatrogenic risk must be avoided. Consequently, future studies comparing these techniques should not only focus on analgesic efficacy, but also on safety endpoints, including the incidence of bleeding or neurological complications, to better define the most appropriate and ethically sound strategy for this unique patient population.
Finally, long-term follow-up studies are needed to evaluate the effect of perioperative analgesic strategies on donor well-being, satisfaction, and overall quality of life. Given the ethical and psychological implications of living donation, optimizing every aspect of perioperative care, including pain management, is essential to ensure donor safety, comfort, and confidence in the transplantation process.
Conclusion
Spinal analgesia using intrathecal morphine appears to provide effective and durable postoperative pain control in living liver donors, with an overall favorable safety profile. Compared with alternative techniques, ITM achieves equal or superior analgesia, and reduces overall opioid consumption. Nevertheless, the current evidence base is limited by small sample sizes and significant clinical heterogeneity.
Given the increasing use of minimally invasive donor hepatectomy, future high-quality randomized studies are essential for establishing the optimal analgesic strategy for this population.
Supplemental Material
sj-docx-1-jic-10.1177_08850666251406212 - Supplemental material for Efficacy of Spinal Analgesia for Pain Management in Living Donor Hepatectomy: A Systematic Review
Supplemental material, sj-docx-1-jic-10.1177_08850666251406212 for Efficacy of Spinal Analgesia for Pain Management in Living Donor Hepatectomy: A Systematic Review by Paolo Capuano, Gennaro Martucci, Yaroslava Longhitano, Raymond M. Planinsic and Gaetano Burgio in Journal of Intensive Care Medicine
Supplemental Material
sj-docx-2-jic-10.1177_08850666251406212 - Supplemental material for Efficacy of Spinal Analgesia for Pain Management in Living Donor Hepatectomy: A Systematic Review
Supplemental material, sj-docx-2-jic-10.1177_08850666251406212 for Efficacy of Spinal Analgesia for Pain Management in Living Donor Hepatectomy: A Systematic Review by Paolo Capuano, Gennaro Martucci, Yaroslava Longhitano, Raymond M. Planinsic and Gaetano Burgio in Journal of Intensive Care Medicine
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
