Abstract
Background
Postoperative pain following liver resection remains a clinical challenge, and the optimal analgesic strategy is still debated.
Objective
To determine whether a single intrathecal morphine injection provides superior analgesia and opioid-sparing effects compared with conventional systemic or regional techniques in adult patients undergoing liver surgery.
Methods
PubMed, Embase, Web of Science citation index, and the Cochrane Library were searched from inception to August 2025 for randomized controlled trials comparing intrathecal morphine with alternative analgesic regimens in liver resection. The primary outcome was pain intensity at rest 24 h after surgery (standardized mean difference). Secondary outcomes included pain intensity at 48 and 72 h and cumulative opioid consumption within 24 h postoperatively. Random-effects meta-analyses and I² statistics were used to assess pooled effects and heterogeneity.
Results
Eleven randomized controlled trials (n = 535) met the inclusion criteria. Intrathecal morphine reduced 24-h postoperative pain scores with a moderate effect (standardized mean difference = −0.64; 95% confidence interval: −0.84 to −0.44; p < 0.001; I2 = 55%) and decreased 24-h opioid consumption by 11.6 mg morphine equivalents (95% confidence interval: −19.3 to −3.9 mg; p = 0.003; I2 = 96%). Differences in pain intensity at 48 and 72 h were not statistically significant. Adverse-event profiles were comparable between groups.
Conclusion
A single dose of intrathecal morphine provides clinically meaningful early analgesia and opioid-sparing benefits after liver resection without increasing adverse events. Incorporating intrathecal morphine into multimodal analgesic protocols may accelerate recovery; however, further high-quality trials are warranted to refine dosing and identify optimal patient selection.
Keywords
Introduction
Hepatobiliary surgery is often associated with significant pain; therefore, adequate pain control is closely linked to postoperative recovery of patients. 1 Although the advent of minimally invasive techniques and multimodal perioperative analgesia has substantially reduced surgical trauma and improved the management of severe pain, postoperative pain control remains a significant challenge following hepatic surgery. Therefore, improving analgesia and the quality of postoperative recovery continue to be clinical priorities. 2 Previous studies have investigated various methods of postoperative pain management, including intravenous (i.v.) administration of systemic medications, local anesthesia at the surgical incision, and regional block anesthesia. 3 Adequate pain management is essential for early recovery and rehabilitation of patients undergoing hepatic surgery. 4 Opioids are considered the “gold standard” for the management of postoperative pain in clinical practice. 5 Evidence from animal studies suggests that epidural morphine sulfate provides analgesic effects and is superior to systemic morphine in alleviating postoperative pain. However, pain relief following a single epidural injection of morphine may not last for 24 h, and the placement of an epidural catheter can cause complications, particularly in patients receiving anticoagulants. 6 Clinical trials have shown that a single injection of intrathecal morphine (ITM) can produce strong analgesic effects lasting up to 24 h.7–9 Spinal opioid analgesia has a long history, beginning from the first report in 1901 and advancing through receptor-level mechanistic insights and early clinical series, which collectively established ITM as a viable neuraxial technique.10–13 Because the placement and removal of epidural catheters remain challenging after liver resection, single-shot ITM has been proposed as an alternative to patient-controlled i.v. analgesia or continuous epidural infusion.14–21 However, individual randomized controlled trials (RCTs) are underpowered and yield inconsistent results. Therefore, we conducted a systematic review and meta-analysis of RCTs to evaluate the efficacy and safety of ITM in adult patients undergoing liver surgery. We hypothesized that ITM provides superior analgesia within 24 h postoperatively compared with systemic opioids or regional blocks, with acceptable side-effect profiles.
Methods
The study has been reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 22 This meta-analysis was prospectively registered with the International Prospective Register of Systematic Reviews (PROSPERO; Registry Number: CRD42024552768).
Search strategy
We systematically searched electronic databases, including PubMed, the Cochrane Library, Web of Science citation index, and Embase, from inception to October 2024 to identify RCTs that met the predefined inclusion criteria. We also screened the reference lists of the identified articles. No language restrictions were applied during the search (Tables 1 and 2).1,23–32 Full database-specific search strategies are provided in Supplementary Table S1.
Summary of study characteristics.

N: number of patients.
Summary of intervention details.

ITM: intrathecal morphine; PCA: patient-controlled analgesia; IV-PCA: intravenous patient-controlled analgesia.
Study selection criteria
Inclusion criteria
Inclusion criteria were as follows.
Study design. RCTs comparing the efficacy and safety of ITM with other analgesic techniques in adult patients undergoing liver surgery; Participants. Adults (≥18 years old) undergoing any form of liver surgery, including hepatectomy or liver resection; Interventions. Studies evaluating single-shot ITM for postoperative analgesia; Comparators. Systemic opioids (e.g. i.v. morphine), regional blocks (e.g. epidural analgesia, erector spinae plane block (ESPB), and quadratus lumborum block (QLB)), or patient-controlled analgesia (PCA); Outcomes. Studies reporting one or more of the following outcomes: Primary. Postoperative resting pain scores at 24 h after surgery; Secondary. Opioid consumption at 24 h, postoperative nausea and vomiting (PONV), and other analgesia- or intervention-related complications.
Exclusion criteria
Exclusion criteria were as follows.
Study design. Nonrandomized or quasi-randomized studies (e.g. observational studies and cohort studies); Population. Studies including pediatric patients (<18 years old) or animal studies; Interventions. Studies using multiple neuraxial or fascial-plane blocks (e.g. combined ITM and epidural analgesia) or studies in which the specific effects of ITM could not be isolated; Data reporting. Studies that did not report, or from which data could not be extracted for, pain scores at 24 h post-surgery or opioid consumption; Abstract-only publications. Studies for which the full-text article was not available or that provided insufficient data for meta-analysis.
Two independent reviewers (Reviewer A and Reviewer B) screened all titles and abstracts. The interrater agreement for title and abstract screening was substantial, with a Cohen’s kappa of 0.85. Data were independently extracted by the same reviewers, with an excellent interrater agreement and a Cohen’s kappa of 0.90. Any discrepancies were resolved through discussion with a third reviewer (Reviewer C).
Data extraction
Data extraction included the following: authors, year of publication, analgesic regimen, type of surgical procedure, puncture location, type and dosage of local anesthetics, administration of opioids, postoperative pain scores, opioid-related side effects (primarily PONV), and complications associated with ITM. The included studies assessed the pain scores using either a visual analog scale (VAS) or a numeric rating scale (NRS). The primary outcome was postoperative resting pain scores at 24 h, as measured by VAS or NRS. Secondary outcomes included postoperative pain scores at 48 and 72 h, cumulative opioid consumption (morphine equivalent) at 24 h after surgery, PONV at 24 h, duration of hospitalization, and pruritus at 24 h postoperatively. When data were presented graphically, numerical values were extracted using WebPlot Digitizer. 33 As previously described, all reported perioperative opioid consumption was converted to i.v. morphine equivalents using a standardized conversion calculator. 34 Median (interquartile range) values were converted to the mean (SD) using established methods.35–38
Bias assessment
Methodological quality was independently assessed by two authors using the Cochrane Risk of Bias Tool, with any disagreements resolved by a third author (each article was rated as low risk, high risk, or unclear risk). 39 The Grading of Recommendations Assessment, Development, and Evaluation (GRADE) methodology was then used to assess the quality of evidence for each outcome. 40 Evidence quality for each outcome and unique comparison of interest was rated as low, moderate, or high. Each article was evaluated by two investigators, with a third investigator resolving any disagreements. The PRISMA flow chart illustrates the study selection process and provides reasons for exclusion of the screened records.
Statistical analysis
Review Manager, version 5.4 (Copenhagen: The Nordic Cochrane Centre), was used to perform all statistical analyses for this meta-analysis. This meta-analysis included studies with similar outcome indicators. Continuous data, including postoperative pain scores and opioid consumption, were recorded as mean and SD. Mean differences (MD) with corresponding 95% confidence intervals (CIs) were calculated for continuous data using a random-effects model. Dichotomous data, such as PONV and ITM-related complications, were analyzed as odds ratios (ORs) with 95% CIs using the Mantel–Haenszel method. 41 Heterogeneity among studies was estimated using I2 statistics. A random-effects model was used when I2 value was >50%, indicating high heterogeneity, whereas a fixed-effect model was used when I² value was ≤5042 For the primary and secondary outcomes, postoperative 24-h resting pain scores and postoperative 24-h opioid consumption, sensitivity analyses were performed using the leave-one-out approach to explore possible sources of heterogeneity. Subgroup analyses based on different levels of risk of bias were also conducted. The quality of individual studies was assessed using the Cochrane Risk of Bias Tool for RCTs. The quality was evaluated using the following potential sources of bias: sequence generation, allocation concealment, blinding of participants or outcome assessors, incomplete data, and selective reporting. The methodology for each study was graded as “high,” “low,” or “unclear,” reflecting the risk of bias.
Results
Results of literature search
Our study selection process is shown in the PRISMA flow diagram. The literature search initially identified 89 citations across four electronic databases. After full-text review and application of exclusion criteria, 11 RCTs comprising 535 patients met the inclusion criteria.1,23–32 Table 1 presents the characteristics and details of the included studies. All 11 articles were published between 2006 and 2023 and were written in English. Notably, one study compared the effects of different doses of morphine administered via intrathecal injection. The assessment of risk of bias for the included studies was performed using the Cochrane risk of bias tool, and the results are presented in Figure 1.

Risk of bias summary: authors’ judgment of each risk of bias item for all included studies. Green, red, and yellow circles indicate low, high, and unclear risk of bias, respectively.
24-, 48-, and 72-h postoperative resting pain scores
According to the results of the meta-analysis, compared with i.v. morphine, epidural catheterization, ESPB, and QLB, ITM significantly reduced postoperative resting pain scores within 24 h (SMD = −0.64; 95% CI: −0.84 to −0.44; p < 0.00001; I2 = 55%). However, ITM had no significant effect on resting pain scores at 48 h (SMD = 0.44; 95% CI: −0.35 to 1.23; p = 0.27; I2 = 89%) and 72 h (MD = 1.07; 95% CI: −0.34 to 2.48; p = 0.14; I2 = 96%) postoperatively (Figure 2). Given that I² values were >50%, a random-effects model was used. Sensitivity analyses were conducted by excluding individual studies to determine whether heterogeneity was reduced; no clear source of heterogeneity was identified.

Postoperative resting pain scores at 24, 48, and 72 h.
To assess potential publication bias for the primary outcome (postoperative 24-h pain scores), a funnel plot was generated (Figure 3). The plot showed slight asymmetry, suggesting a potential risk of publication bias. However, due to the limited number of included studies (fewer than 10), this result should be interpreted with caution.
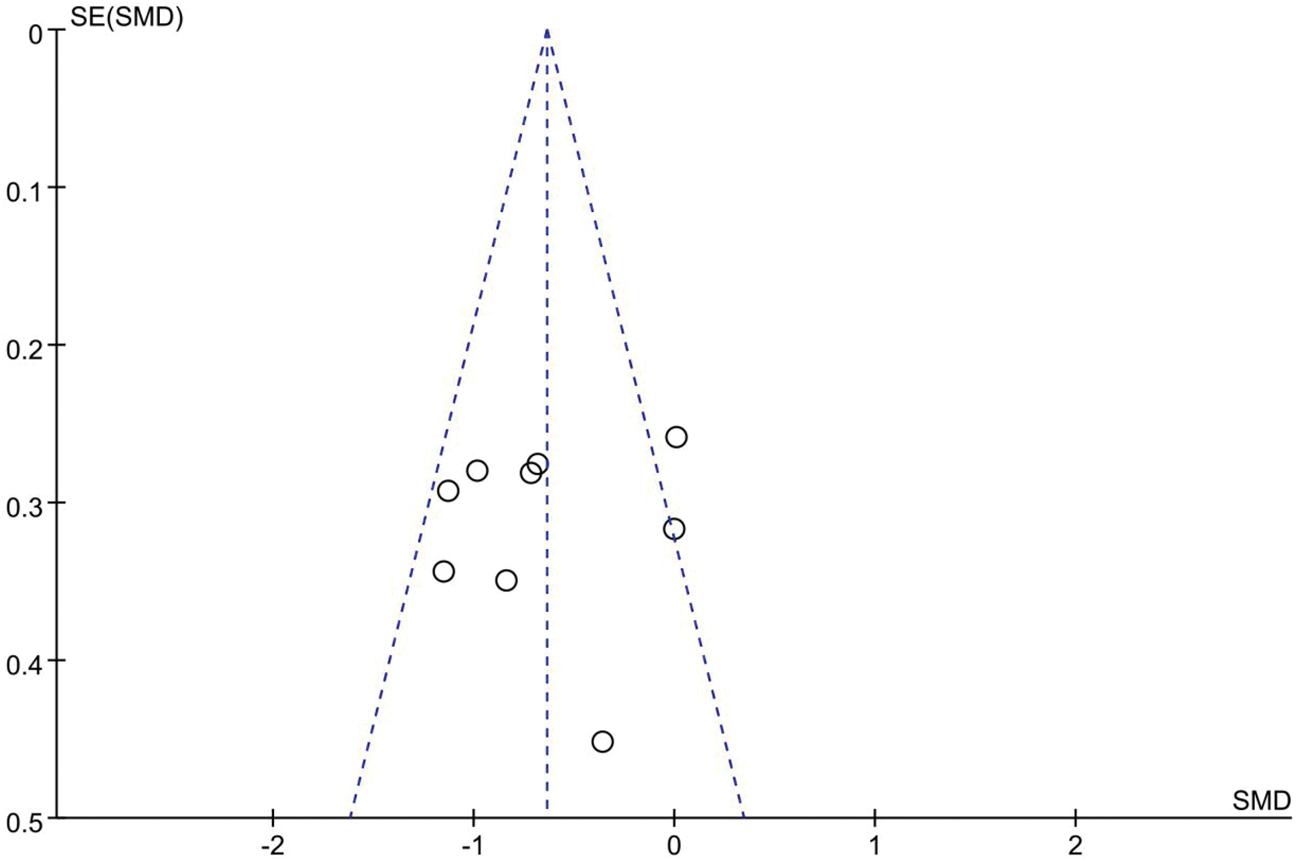
Funnel plot for assessing publication bias of the primary outcome (24-h postoperative pain scores).
Random-effects meta-regression was performed to explore sources of heterogeneity in postoperative 24-h pain scores. Using surgical approach (open vs laparoscopic) as a moderator did not materially reduce heterogeneity (R2 = 0%, β = +0.36 SMD for laparoscopic vs open; 95% CI: −0.43 to +1.15; p = 0.37). ITM dose (continuous) explained 13% of the between-study variance (R2 = 13.3%), with a nonsignificant trend toward diminishing analgesic benefit at higher doses (β = −2.48 SMD per mg; 95% CI: −6.61 to +1.64; p = 0.24). A multivariable model including both moderators (surgical approach and ITM dose) did not further improve model fit (overall p = 0.60; R2 = 0%).
Postoperative cumulative opioid consumption at 24 h postoperatively
The pooled data from six RCTs showed that, compared with the control group, ITM significantly reduced cumulative postoperative opioid consumption at 24 h (MD = −11.58; 95% CI: −19.31 to −3.86; p = 0.0003; I2 = 96%). Considering that different doses of morphine may contribute to the high heterogeneity among the included studies, a subgroup analysis for the cumulative opioid consumption at 24 h postoperatively was conducted. This analysis demonstrated that insufficient morphine dosing in one study may have led to the need for additional postoperative morphine supplementation (Figure 4). Sensitivity analysis, conducted by removing individual studies to examine changes in heterogeneity, did not identify a clear source of heterogeneity.

Forest plot of cumulative opioid consumption (intravenous morphine equivalents) at 24 h postoperatively in the ITM and control groups, with subgroup analysis for different doses of morphine.
Length of hospital stay
The pooled data from four RCTs showed that, compared with the control group, ITM did not significantly reduce hospital length of stay (MD = −0.34; 95% CI: −0.82 to 0.13; p = 0.16; I2 = 32%). Because I2 value was <50%, a fixed-effect model was used (Figure 5).
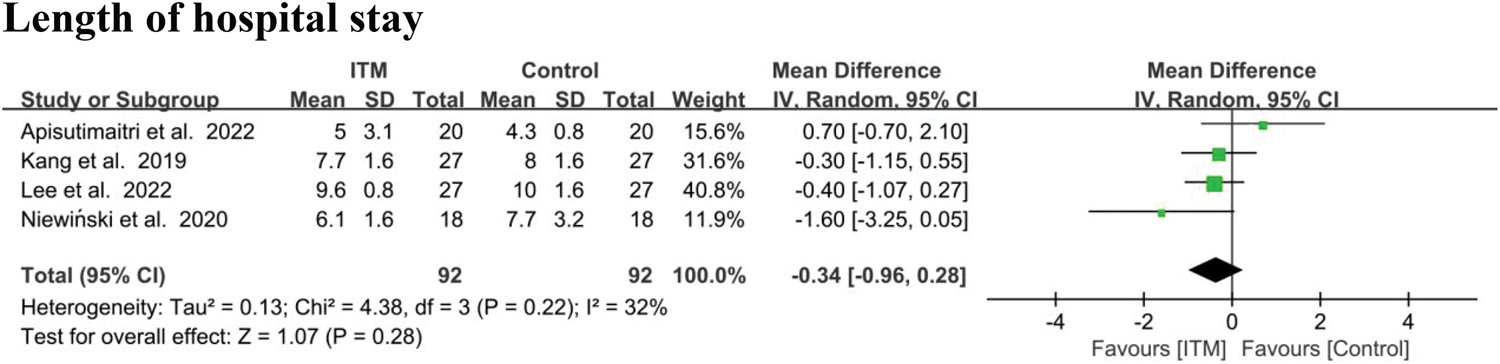
Forest plots for the length of postoperative hospital stay in the ITM and control groups.
Side effects and complications associated with ITM
Notably, compared with the control group, patients in the ITM group experienced a significant increase in postoperative pruritus (risk difference (RD) = 0.51; 95% CI: 0.21 to 0.80; p = 0.0008; I2 = 95%) (Figure 6). The incidence of opioid-induced pruritus varies depending on the type of opioid and the route of administration, with mechanisms that can be broadly categorized as peripheral or central, the latter being particularly complex. 43 Conversely, ITM did not significantly affect the incidence of PONV (RD = 0.07; 95% CI: −0.12 to 0.26; p = 0.45; I2 = 79%] (Figure 6).
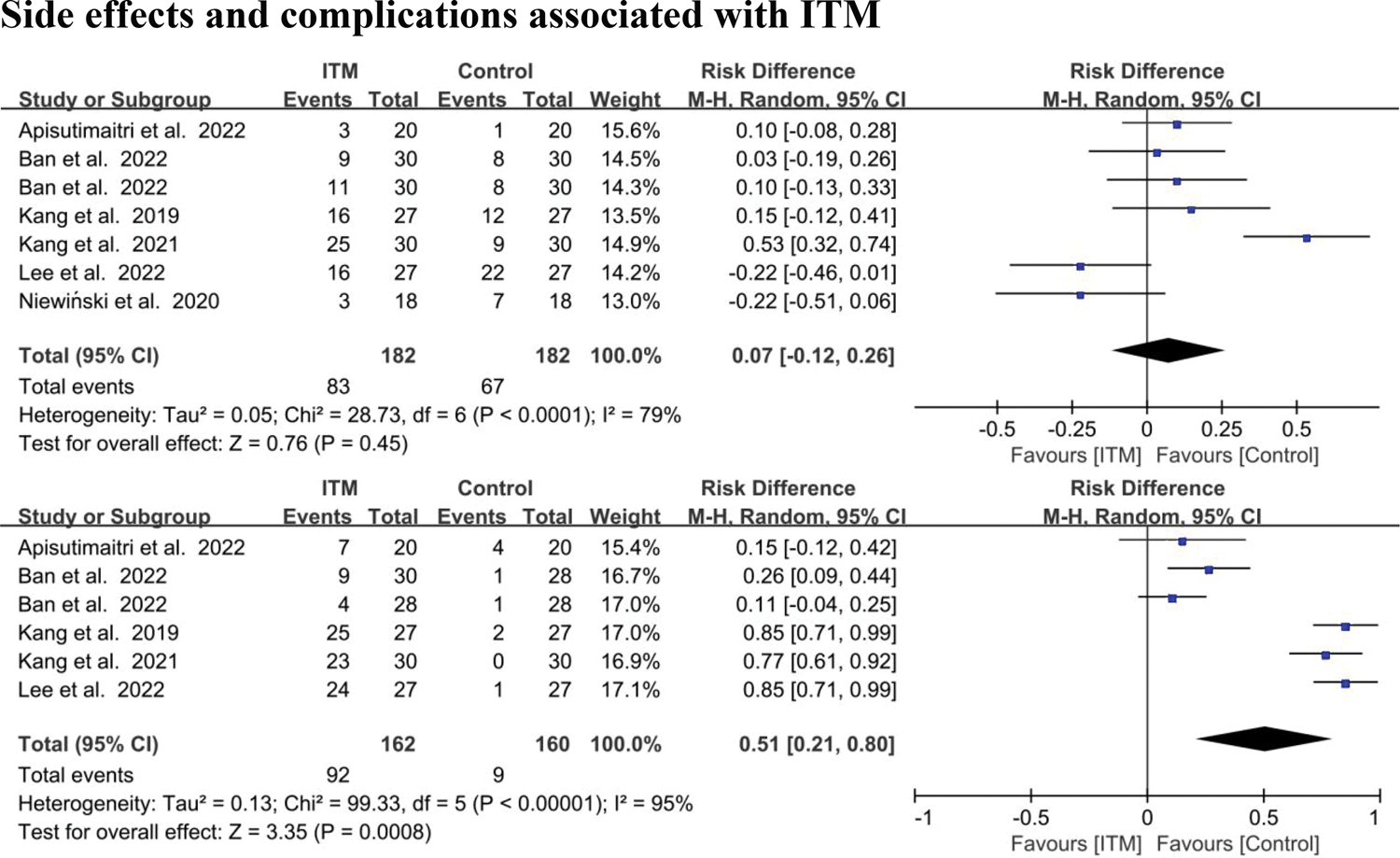
Forest plots of postoperative nausea and vomiting and pruritus in the ITM and control groups.
Publication bias
According to the meta-analysis, the results showed high heterogeneity (I2 > 50%). Sensitivity analysis using the leave-one-out method showed no significant changes in the pooled effect size. Publication bias was assessed using the Cochrane Risk of Bias Tool (Figure 1). The quality of evidence for the primary outcomes, including postoperative resting pain scores at 24 h, opioid consumption, incidence of postoperative pruritus, incidence of PONV, and hospital length of stay, was evaluated using the GRADE methodology. The quality of evidence for these outcomes was rated as high (Table 3).
Quality of evidence assessment for main results.

CI: confidence interval; IV: intravenous; MD: mean difference; SMD: standardized mean difference.
Discussion
In this meta-analysis, compared with i.v. morphine, epidural catheterization, ESPB, and QLB, ITM significantly reduced postoperative 24-h resting pain scores in patients undergoing liver surgery (p < 0.0001). However, there was no significant reduction in pain scores at 48 and 72 h postoperatively (p = 0.27, p = 0.14). Furthermore, compared with the control group, ITM significantly reduced the cumulative consumption of opioids within the first 24 h postoperatively (p = 0.003). The incidence of PONV and hospital length of stay in patients receiving ITM were not statistically different from those receiving other analgesic methods (p = 0.45, p = 0.28). However, most patients receiving ITM experienced postoperative pruritus (p = 0.008), which may affect patient recovery to some extent. The recent RCT by Rousseleau et al. 44 provides additional evidence that a single ITM dose confers early analgesic benefits after liver resection, consistent with the direction of our pooled effect estimates. As this study was published after the prespecified search cutoff date, it was not included in the current quantitative synthesis; however, it will be incorporated in the next update of our meta-analysis. There are various treatments for opioid-induced pruritus; however, most treatments remain controversial and results are conflicting. 45 Opioids remain the gold standard for managing moderate to severe acute pain as well as cancer-related pain. Opioid-induced pruritus is well-recognized to be typically unresponsive to conventional antipruritic treatments, thereby posing a challenge for clinicians in pain management. These inconsistent findings highlight the need for further high-quality RCTs.
After intrathecal administration, all opioids may produce analgesia at least in part through spinal mechanisms.5,46 The deposition of opioids within the spinal canal follows a multicompartmental pattern and occurs simultaneously. Opioids move cephalad via the cerebrospinal fluid (CSF); undergo spinal diffusion; bind to nonspecific receptors in the white matter and specific receptors in the gray matter; and clear into the epidural space, where they bind to lipophilic structures. From the latter two compartments, opioids are subsequently redistributed into the bloodstream via vascular reuptake. 47 The clinical profile of each opioid results from the sum of these distribution pathways, which determine its bioavailability and spinal effect. 48 Intrathecal opioid administration is an attractive analgesic technique because the drug is injected directly into the CSF, close to the central nervous system structures where opioids act. The procedure is simple and rapid, with a relatively low risk of technical complications or failure. ITM without local anesthetics is typically administered as a single preoperative dose in conjunction with general anesthesia to prevent pain after major surgery. 49 In summary, among patients undergoing major surgery under general anesthesia who receive systemic opioid treatment for postoperative breakthrough pain, the addition of ITM reduces pain intensity and systemic morphine consumption but does not reduce the risk of morphine-related adverse reactions. The optimal dose of ITM when used alone remains uncertain. 50
In our meta-regression analysis, we explored potential sources of heterogeneity. The analysis found that neither surgical approach (open vs laparoscopic) nor higher doses of ITM (>0.4 mg) explained a significant proportion of the variability of 24-h postoperative pain outcomes. Specifically, the type of surgery did not materially reduce heterogeneity (R2 = 0%; β = +0.36 SMD; 95% CI: −0.43 to +1.15; p = 0.37), and ITM dose as a continuous variable explained only 13% of the between-study variance (R2 = 13.3%; β = −2.48 SMD per mg, 95% CI: −6.61 to +1.64; p = 0.24). These findings suggest that other unmeasured factors, such as patient characteristics, perioperative care, or the timing of ITM administration, may influence outcomes.
Moreover, although ITM doses of 0.3–0.4 mg significantly reduced pain by approximately 0.65 SD and spared approximately 12 mg morphine equivalent compared with the control, the effect of doses higher than 0.4 mg did not provide additional benefits. This finding is consistent with the Enhanced Recovery after Surgery (ERAS) and American Society of Anesthesiologists (ASA) guidelines, which recommend limiting ITM to ≤0.4 mg for optimal postoperative analgesia.
The remaining unexplained heterogeneity suggests that future studies should focus on standardizing interventions and reporting additional variables that can affect pain outcomes, such as multimodal analgesia protocols and patient comorbidities. Further high-quality studies are needed to confirm the optimal dose and to examine the long-term safety and efficacy of ITM in liver surgery.
Clinical takeaway
Key takeaway from the study are as follows.
Recommended dose. 0.3–0.4 mg single-shot ITM; When to use. As part of a multimodal analgesia protocol in laparoscopic or robotic liver resection when epidural analgesia is contraindicated or impractical; Expected benefit. Reduced 24-h postoperative pain scores (≈ 0.6 SD) and reduced 24-h postoperative opioid consumption (≈ 12 mg i.v. morphine equivalents); Key risk. Increased pruritus (∼8% absolute increase), monitored and managed with antihistamines or low-dose naloxone when troublesome; Practical tips. Certain practical tips are as follows: Avoid ITM doses >0.4 mg to reduce the risk of respiratory depression; Combine ITM with paracetamol/nonsteroidal anti-inflammatory drugs and local-infiltration analgesia for additional opioid-sparing effects; Consider avoiding ITM in patients with obstructive sleep apnea or severe obesity.
Because bedside recommendations are most persuasive when they align with established practice standards, we next assessed how our results correspond with recent consensus guidelines. Our findings reinforce and elaborate upon current recommendations from ERAS and ASA. The 2022 ERAS Society consensus for liver surgery advises epidural analgesia for open resections but allows single-shot ITM ≤0.4 mg as part of multimodal analgesia when epidural placement is impractical or contraindicated.
1
Our pooled estimate (SMD = −0.60 for 24-h pain; −11.6 mg i.v. morphine-equivalent reduction) confirms that a 0.3–0.4 mg ITM dose provides clinically meaningful early analgesia without excess respiratory depression, thereby supporting ERAS recommendation, particularly for laparoscopic or robotic procedures.
51
Consistently, the 2016 ASA practice guidelines on neuraxial opioids recommend limiting ITM to ≤0.3–0.4 mg for major abdominal surgery to balance analgesia against pruritus and delayed respiratory depression.
52
Our dose-subgroup analysis showed no additional benefit with ITM doses above 0.4 mg, whereas the incidence of pruritus increased by ∼8%. These findings quantitatively support the ASA dose ceiling. Thus, the present meta-analysis provides level-I evidence that complements existing guideline statements and may encourage broader, guideline-concordant uptake of ITM in ERAS pathways for minimally invasive liver resection.
Limitations
When interpreting our findings, several caveats should be considered. Only 11 RCTs, each of moderate size (the largest intrathecal-morphine arm enrolled 86 patients), met our eligibility criteria, and most were conducted in high-volume Asian centers. None of the included studies stratified outcomes by incision type or surgical approach; therefore, procedure-specific effects of ITM may have been masked, limiting external validity. Clinical heterogeneity was considerable. ITM doses ranged from 0.2 mg to 0.6 mg, whereas control regimens varied from patient-controlled i.v. opioids to epidural catheters and fascial-plane blocks, each embedded within different multimodal protocols. Pain intensity was measured with subjective VAS or NRS scales, and assessor blinding was rarely reported, introducing potential detection bias. Two trials reported more than 10% loss to follow-up, and only three studies were prospectively registered, raising the possibility of selective outcome reporting. Statistically, substantial residual heterogeneity persisted despite subgroup and dose analyses (I2 = 55%–96%). Additionally slight funnel-plot asymmetry with an Egger test p value of 0.08 suggests that positive small studies may be overrepresented. Collectively, these limitations temper confidence in the precise magnitude of benefit, although sensitivity analyses confirmed the direction of effect. Larger, well-designed RCTs that standardize ITM dosing, classify surgical technique, and report core outcome sets, including long-term pain and opioid-related adverse events, are needed to refine perioperative analgesic guidelines.
Conclusions
ITM appears to be an effective analgesic strategy for early postoperative pain management in liver surgery, significantly reducing pain and opioid consumption within the first 24 h. However, these analgesic benefits diminish over time, and further high-quality studies are required to confirm these findings and assess long-term safety.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605261416668 - Supplemental material for Early postoperative pain and opioid use after liver surgery: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605261416668 for Early postoperative pain and opioid use after liver surgery: A systematic review and meta-analysis by Yatao Liu, Zhaohui Gao, Xiaodong Su, Chengying Ji and Qian Fu in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605261416668 - Supplemental material for Early postoperative pain and opioid use after liver surgery: A systematic review and meta-analysis
Supplemental material, sj-pdf-2-imr-10.1177_03000605261416668 for Early postoperative pain and opioid use after liver surgery: A systematic review and meta-analysis by Yatao Liu, Zhaohui Gao, Xiaodong Su, Chengying Ji and Qian Fu in Journal of International Medical Research
Footnotes
Acknowledgments
We sincerely thank all the staff members of the Department of Anesthesiology and Operation at the First Hospital of Lanzhou University for their support during the study. We also appreciate the valuable suggestions from our colleagues at the First School of Clinical Medicine of Lanzhou University.
Author contributions
Zhaohui Gao (First author): Conceptualization, literature search, data extraction, and manuscript drafting.
Yatao Liu (Corresponding author): Study design, data analysis, supervision, and critical revision of the manuscript.
Xiaodong Su: Data collection, validation, and statistical support.
Chengying Ji: Literature review and methodology validation.
Qian Fu: Preparation and editing of figures and tables.
All authors reviewed and approved the final version of the manuscript.
Data availability statement
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request. All data extracted from published studies are included in this article and its supplementary information files.
Declaration of competing interest
Authors declare no any conflict of interest.
Ethical approval
This is a meta-analysis article with no ethical requirements.
Funding
This work was supported by the Key Talent Project of Gansu Province for 2022, organized by the Organization Department of the Gansu Provincial Party Committee (Project Number: 23JRRA1496); the High-End Foreign Project Plan for the 2023–2024 academic year, sponsored by the Ministry of Science and Technology (Project Number: G2023175006L); and the Joint Research Fund Project of Gansu Province for 2023 (Project Number: 23JRRA1496).
Supplemental material
Supplemental material for this article is available online.
Trail registry number
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
