Abstract
Keywords
Background
In coronavirus-19 disease (COVID-19), acute respiratory distress syndrome (ARDS) represents the most common reason for admission to the intensive care unit (ICU) and is associated with high mortality.1–3
Several different mechanical ventilation modes are used for the treatment of patients with COVID-19-associated ARDS (CARDS). 4 While pressure-controlled low tidal volume lung protective ventilation (LTV) represents the most common ventilation mode, 4 some authors suggest airway pressure release ventilation (APRV) to be an effective alternative mode. 5 Beneficial effects might include less pulmonary inflammation, reduced lung injury and an improved preserving of the alveolar-capillary barrier.6–9 Clinically, APRV seems to be associated with less sedation requirement, reduced duration of mechanical ventilation, and lower hospital mortality in ARDS of other etiologies.10,11
However, there are only few heterogeneous studies examining the effect of APRV in CARDS.5,12–19 While APRV showed significantly improved gas exchange compared to conventional mechanical ventilation in a meta-analysis, 15 the first and only randomized controlled trial (RCT) was terminated early due to an interim analysis after the occurrence of a series of barotrauma, indicating a trend toward transient acidosis due to hypercapnia and higher mortality. 15
Against the backdrop of the partially contradictory results of available studies, we therefore analyzed the clinical outcome, complications, and longitudinal clinical course of ventilation parameters in patients with CARDS, who were ventilated using APRV. In addition, we compared these parameters with a propensity score–matched control group of patients, who were ventilated using LTV.
Methods
This study was conducted on 2 level II ICUs and 2 level III ICUs between April 2020 and January 2022. Prior to the start of the study, approval was obtained by the local ethics committee (approval number: 20-11729). Clinical outcome, complication rates as well as the longitudinal course of ventilation parameters, catecholamine requirements, laboratory values, and blood gas analyses on admission, 6 h post admission as well as on day 3 and day 7 were retrospectively obtained from patient charts as well as medical discharge reports. The data was captured in an online database (REDCap - Research Electronic Data Capture, v9.1.0, USA).
All ICU patients with confirmed COVID-19 were screened for the following inclusion criteria: Age ≥18 years; ARDS as defined by the Berlin criteria 20 and mechanical ventilation using APRV for more than 48 h. Patients were included either after endotracheal intubation at the reporting institutions or after being transferred to the reporting institutions due to progression of respiratory failure or for extracorporeal membrane oxygenation (ECMO) evaluation. Patients were excluded from the study, if mechanical ventilation lasted less than 48 h due to death or extubation, if the ventilation mode was changed for more than 2 h per day or if additional supportive devices, for example, ECMO were used.
Ventilation using APRV was implemented following previously described principles. 10 The high airway pressure (Phigh) was initially set to 30 cmH2O after endotracheal intubation or transferal to the reporting institution before attempting to reduce it gradually. The low airway pressure (Plow) was initially set at 5 cmH2O as previously described. 10 The duration of release phase (Tlow) was adjusted to achieve a termination of peak expiratory flow rate between 50% and 75% of peak expiratory flow rate. The duration of Phigh (Thigh) was initially calculated to achieve 14-17 release frequencies per minute and in the further course modified depending on the spontaneous respiratory activity of the patient. FiO2 was adjusted based on peripheral oxygen saturation and arterial blood gas analysis. In addition to pulse-oximetry and the results of repeated blood gas analyses, the intrapulmonary shunt 21 was used as a surrogate parameter to operate the gradual reduction of Phigh in the course of the ventilatory management. Airway pressure release ventilation settings as well as the dosages of sedative medications were successively reduced, aiming for a Richmond Agitation Sedation Scale 22 score between −2 (in severe ARDS) and 0 (during weaning), to allow for spontaneous respiratory activity of the patient.
To allow for a comparison with LTV, data of a control group of patients, who fulfilled the inclusion criteria and received LTV, were gathered. The ventilation using LTV was implemented according to the “ARDSnet”-protocol 23 and initially aimed for a tidal volume target of 6 mL/kg predicted body weight (PBW). The ventilatory settings were modified according to protocol to aim for the target oxygenation and target pH value maintaining lowest possible plateau airway pressure with an upper limit of 30 cmH2O. In both APRV and LTV, neuromuscular blocking agents were used only for intubation or in emergency situations, but not during regular treatment. Prone positioning was initiated according to current guidelines in the absence of contraindications in patients with PaO2:FiO2 ratio < 150 mm Hg and FiO2 ≥ 0.6 in both APRV and LTV patients.24,25 If indicated, prone positioning was conducted for at least 16 consecutive hours per day.
All data were captured in an online database (REDCap - Research Electronic Data Capture, v9.1.0). After exporting the data to Microsoft Excel (Version 16.48, Microsoft), the statistical analysis was performed using RStudio (version 4.1.2, R Foundation for Statistical Computing). Matching of APRV and LTV patients was performed with propensity-score estimation using a multivariate logistic regression model adjusting for the following factors: Patient characteristics (age, sex), arterial blood gas analysis (PaO2, PaCO2, PaO2/FiO2 ratio, bicarbonate, lactate, and pH) and “Acute Physiology And Chronic Health Evaluation II” (APACHE II) score at time of endotracheal intubation or transfer as well as the implementation of prone positioning. Matching was performed based on a 1:1 nearest neighbor matching procedure without replacement and a threshold for absolute standardized mean differences of 0.1 using RStudio and the MatchIt package. 26
To compare APRV and LTV, Wilcoxon rank sum test was used for continuous variables while Pearson's Chi-square test or Fisher exact test was used for categorical variables. Statistical significance was set at P ≤ .05 (2 sided).
Findings
Forty patients with CARDS, mechanically ventilated using APRV, were matched with 40 LTV patients. Results of propensity score–matching quality are shown in Supplementary Information Figure 1. Patient characteristics, status on admission, treatment data, clinical outcome, as well as complications are listed in Table 1. Regarding patient characteristics, comorbidities, and treatment, no significant differences between APRV and LTV were identified. Airway pressure release ventilation patients had a significantly higher rate of prior mechanical ventilation at external ICUs before being transferred to the reporting institutions due to progression of respiratory failure or for ECMO evaluation (38% in APRV, 15% in LTV; P = .022). At admission, both patient groups presented with similar “Sequential Organ Failure Assessment” (SOFA) scores and APACHE II scores without significant differences.
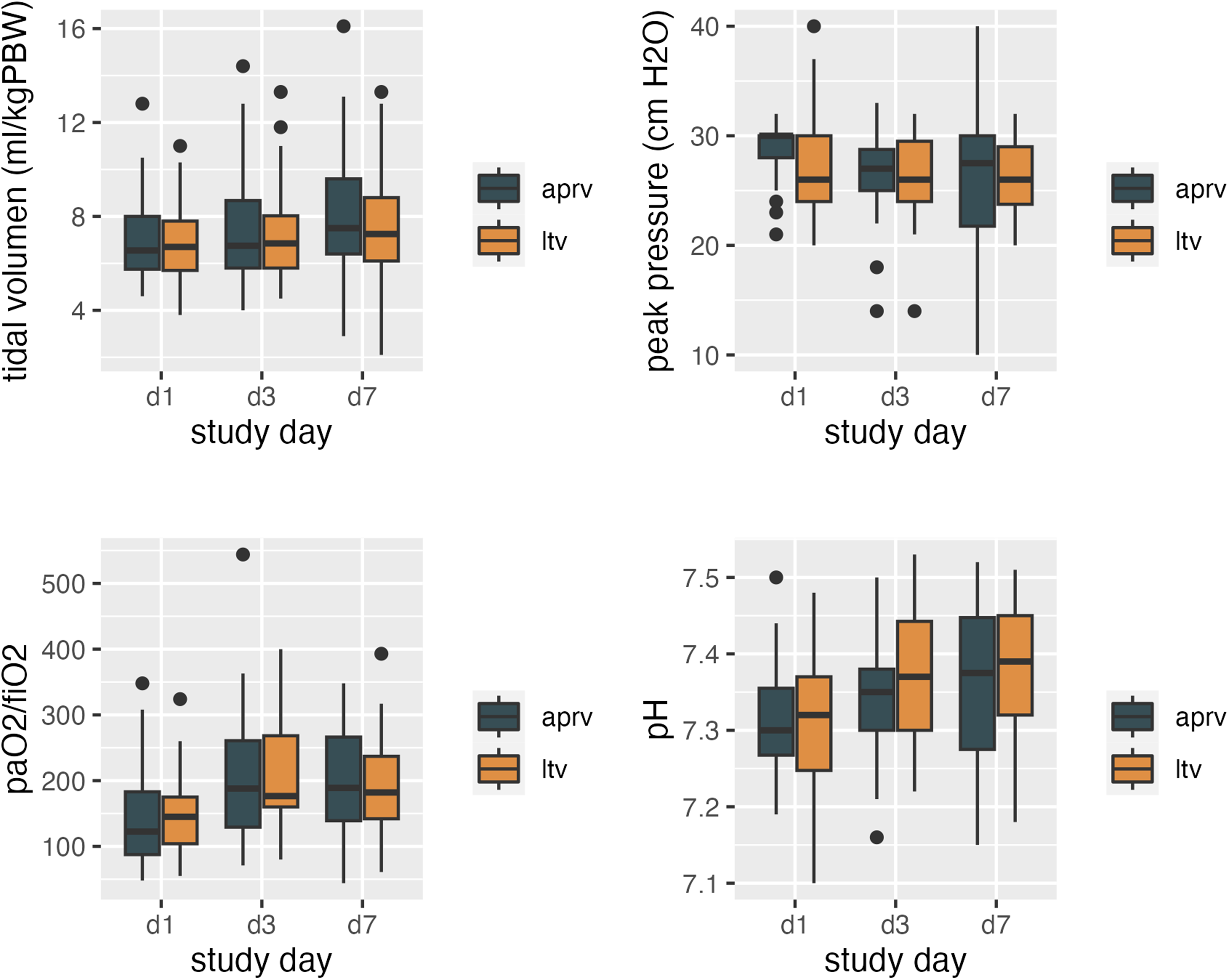
Respiratory mechanics and gas exchange. Boxplots of peak pressure, tidal volume per predicted body weight (PBW), PaO2/FiO2 ratio, and pH values in patients with COVID-19-induced ARDS. Patients were mechanically ventilated either using airway pressure release ventilation (APRV) or using pressure-controlled low tidal volume ventilation (LTV). Data were gathered at admission as well as on day 3 and 7 of mechanical ventilation. d1: Day 1 (at the time of endotracheal intubation or admission to the reporting ICU); d3: Day 3 of mechanical ventilation; d7: Day 7 of mechanical ventilation.
Patient Characteristics.
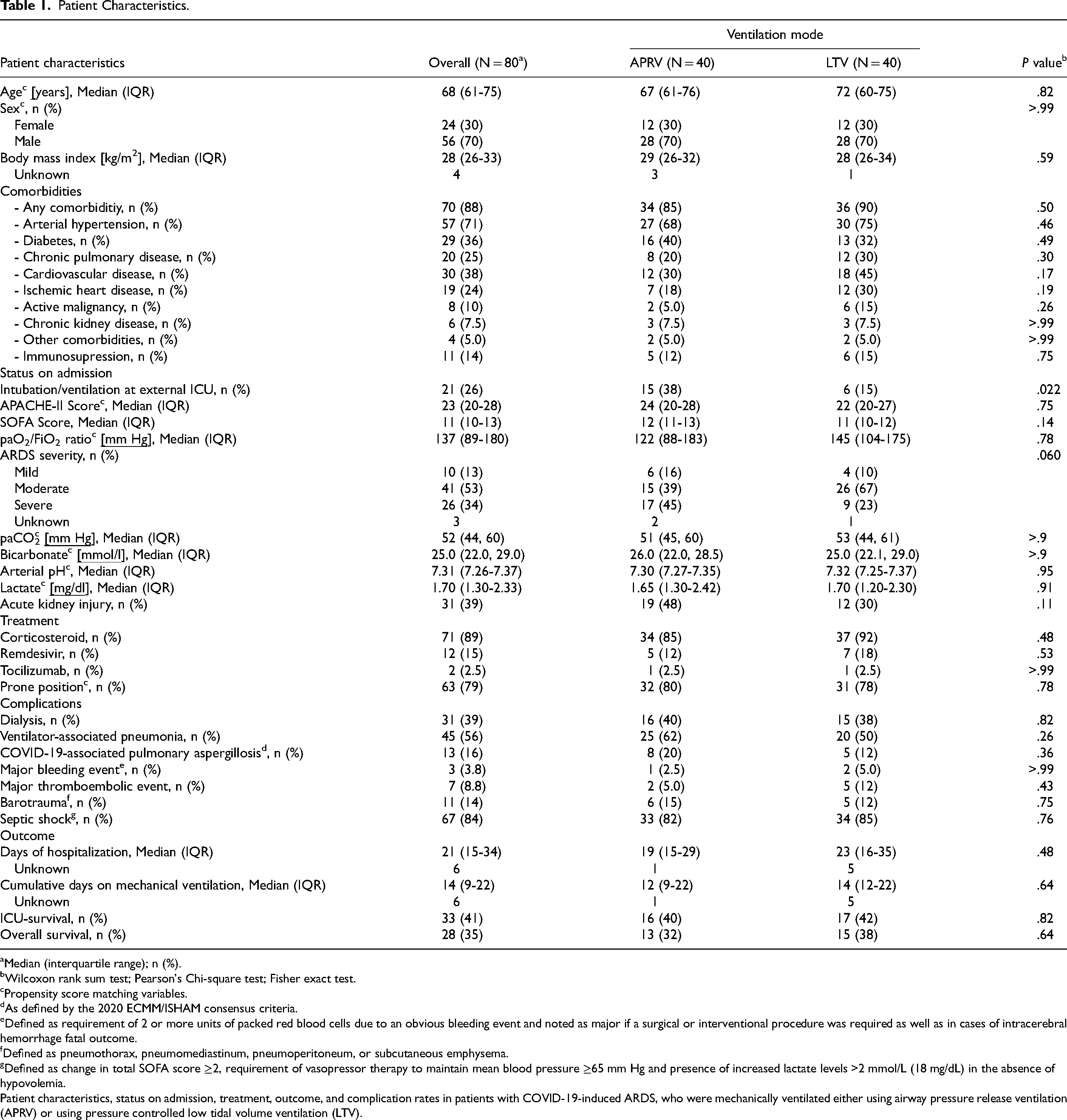
Median (interquartile range); n (%).
Wilcoxon rank sum test; Pearson's Chi-square test; Fisher exact test.
Propensity score matching variables.
As defined by the 2020 ECMM/ISHAM consensus criteria.
Defined as requirement of 2 or more units of packed red blood cells due to an obvious bleeding event and noted as major if a surgical or interventional procedure was required as well as in cases of intracerebral hemorrhage fatal outcome.
Defined as pneumothorax, pneumomediastinum, pneumoperitoneum, or subcutaneous emphysema.
Defined as change in total SOFA score ≥2, requirement of vasopressor therapy to maintain mean blood pressure ≥65 mm Hg and presence of increased lactate levels >2 mmol/L (18 mg/dL) in the absence of hypovolemia.
Patient characteristics, status on admission, treatment, outcome, and complication rates in patients with COVID-19-induced ARDS, who were mechanically ventilated either using airway pressure release ventilation (APRV) or using pressure controlled low tidal volume ventilation (LTV).
The time-dependent course of respiratory mechanics and ventilation parameters are displayed in Figure 1. Average tidal volumes per PBW, peak pressure values as well as blood gas analyses (PaO2, PaCO2, PaO2/FiO2 ratio, bicarbonate, lactate, pH) on admission, 6 h post-admission as well as on day 3 and day 7 were similar for both APRV and LTV. No significant difference in the levels of C-reactive protein, ferritin, and interleukin-6 as well as the neutrophil-to-lymphocyte ratio was observed between both groups.
No significant differences between APRV and LTV patients were identified for ICU survival, duration of mechanical ventilation, as well as complication rates. A Kaplan-Meier estimator is illustrated in Figure 2, showing no significant differences between APRV and LTV. Similar complication rates with respect to ventilator-associated pneumonia, necessity for hemodialysis, thromboembolic events, and barotrauma were detected for both APRV and LTV patients.
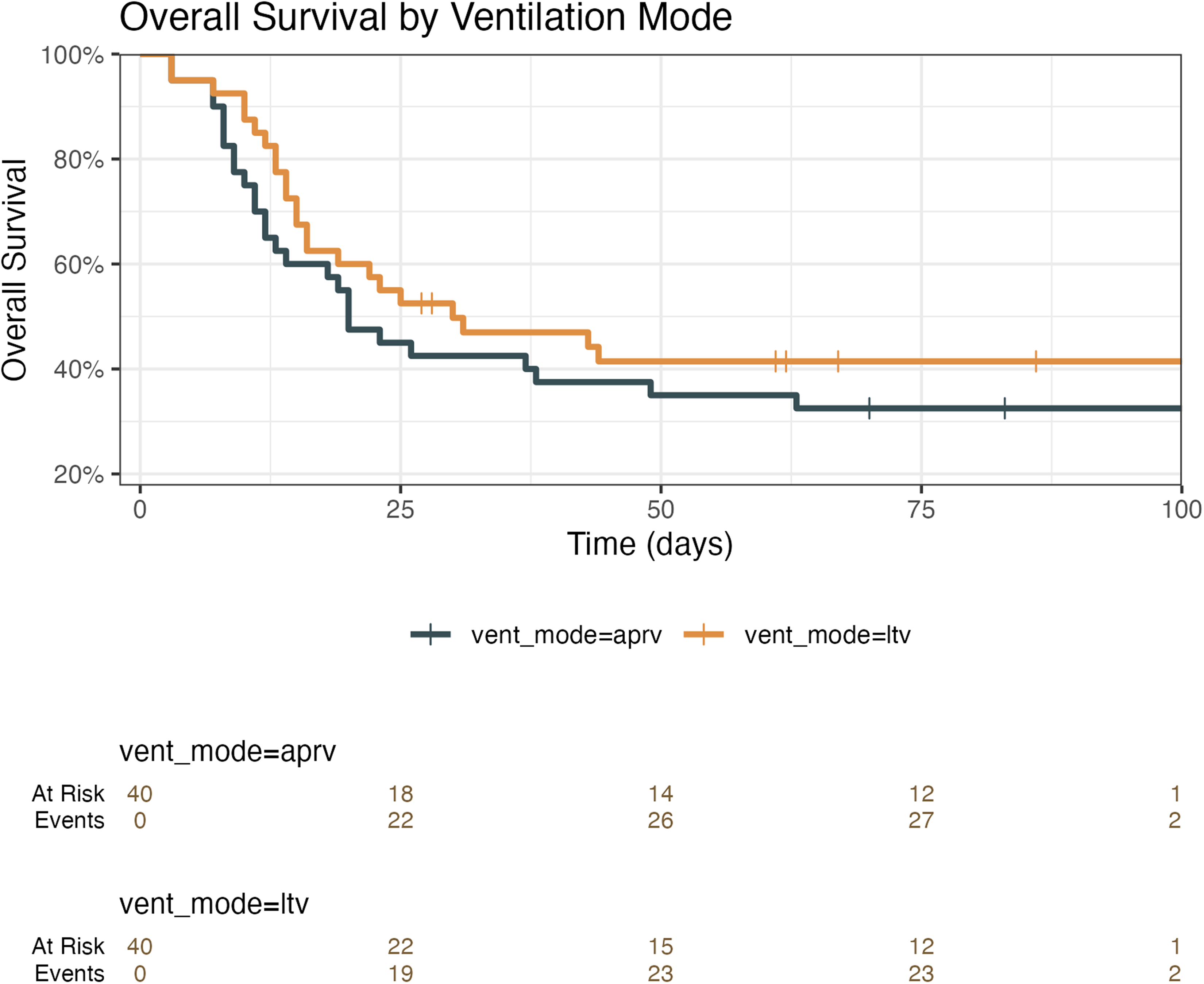
Kaplan-Meier estimator. Kaplan-Meier estimator of patients with COVID-19-induced ARDS that were either mechanically ventilated using airway pressure release ventilation (APRV) or using pressure-controlled low tidal volume ventilation (LTV).
In a Cox proportional hazards regression analysis, including the factors age, sex, external ventilation, septic shock, ARDS severity, prone position, and ventilation mode, only age (hazard ratio [HR] 1.08, 95% confidence interval [CI] 1.04-1.1; P < .001), severe ARDS (HR 2.62, 95% CI 1.02-6.7; P = .046), and the occurrence of septic shock (HR 17.18, 95% CI 2.06-143.2; P = .009) were independent adverse prognostic factors, but not the ventilation mode (compare Figure 3).
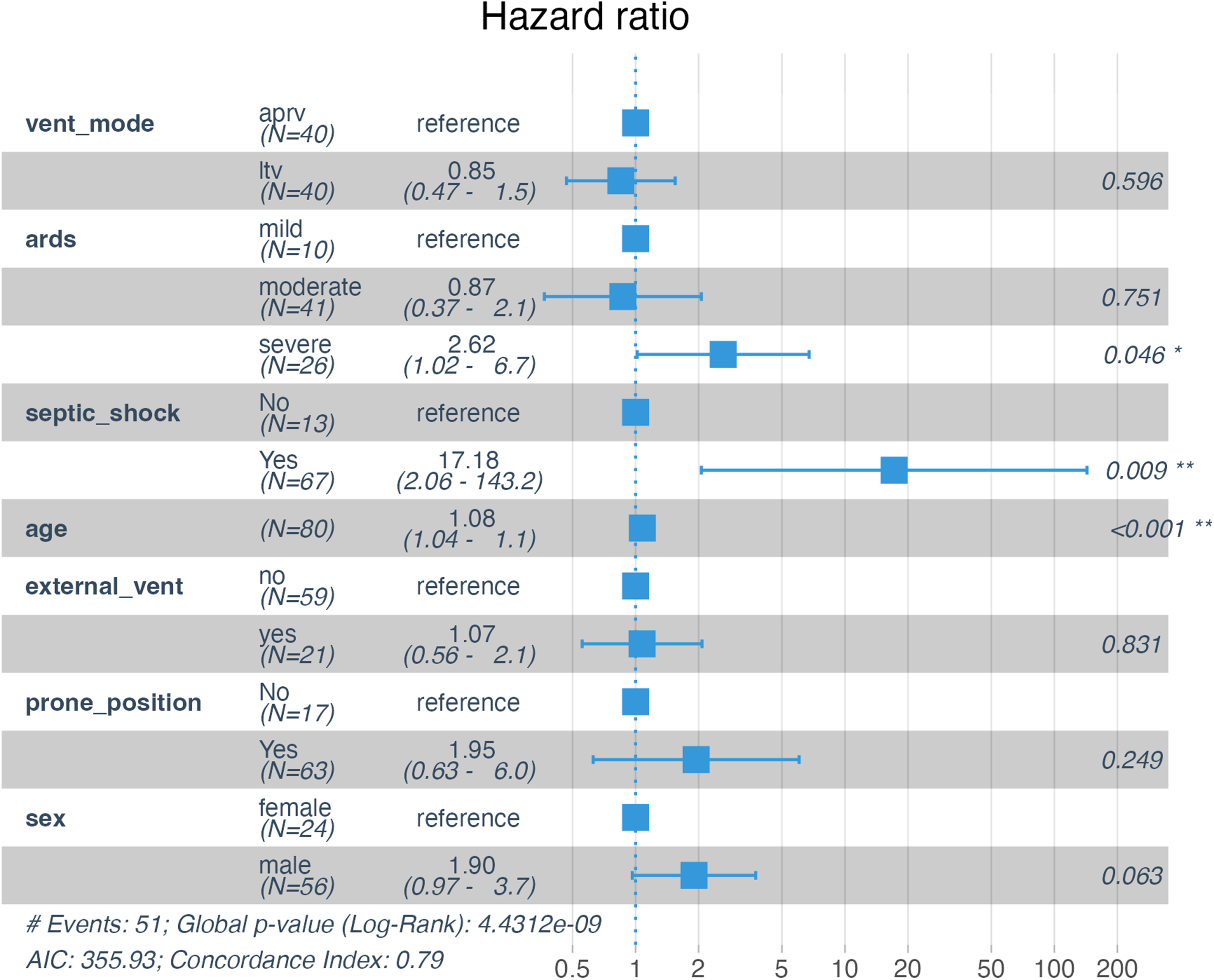
Cox proportional hazards regression analysis. Forest plots summarize the results of the Cox proportional hazards regression analysis in patients with COVID-19-induced ARDS. Patients were mechanically ventilated either using airway pressure release ventilation (APRV) or using pressure-controlled low tidal volume ventilation (LTV). Shown are the adjusted hazard ratios for overall survival and the 95% confidence intervals.
Interpretation
As key findings of our study, we found similar clinical outcomes with respect to survival and length of mechanical ventilation in APRV patients and in the matched LTV control group. Moreover, rates of ventilation-associated and nonventilation-associated complications did not significantly differ between both ventilation modes.
These results contribute to the database of APRV in COVID-19 but must be interpreted in the framework of several heterogeneous studies, which are examined in more detail hereinafter: Notably, most available data were derived from retrospective, single-center analyses with limited sample sizes. Two retrospective analyses, one single-center analysis including 60 CARDS patients from New York (USA) 12 and one 2-center analysis including 17 patients from Nancy (France), 16 investigated the short-term effect of APRV on ventilation parameters after converting from a conventional ventilation mode to APRV. Switching to APRV resulted in significantly improved oxygenation in both studies, but in the analysis from New York increased APRV release volumes were noted compared to LTV tidal volumes (7.9 vs 6.6 mL/kg PBW). In a prospective pilot study of 10 APRV patients, improved oxygenation as well as decreased vasopressor and sedation requirements were noted. 5 In contrast to the studies,12,14,19 demonstrating significantly higher tidal volumes beyond the limits of lung-protective ventilation in APRV, the present study showed similar tidal volumes per PBW as well as peak pressure values compared to the matched LTV control group that followed ARDSnet recommendations. 23 However, regardless of the ventilation mode, the aim of lung protective ventilation less than or equal to the “magic number” 27 of 6 mL/kg PBW was missed in a considerable number of patients (compare Figure 1). However, there is an ongoing debate whether the ARDSnet tidal volume limitations should be applied to the APRV release volume or whether concepts of a mandatory mode of ventilation (ARDSnet) are mistakenly transferred to the paradigm of a spontaneous ventilation mode. 28 The effect of APRV on improving oxygenation that has also been shown in a recent meta-analysis 15 was not reproducible in the present study.
Several other studies additionally focused on the intermediate-term outcome of APRV in the form of ICU-mortality as well as duration of ICU-treatment. A retrospective single-center study from Detroit (USA) compared 18 APRV patients with 160 LTV patients. 17 While no significant effect on ventilator-free days and in-hospital mortality (APRV 60% vs LTV 50%) was observed, a reduction of vasopressor and sedation requirements was noted. A similar reduction of vasopressor requirements and significantly improved oxygenation were identified in another retrospective study of 14 APRV patients; however, the 28-day mortality in APRV patients was 86% (vs 52%). 19 Notably, in this study the mean tidal volume in APRV was 9.8 mL/kg PBW and APRV was discontinued in the majority of patients when hypoxemia improved. A retrospective multicenter study from Australia examined a total of 25 CARDS patients, 11 of whom were ventilated with APRV, while 14 were treated with other ventilation modes including LTV. 13 At day 90, LTV patients showed a significantly lower mortality rate compared to APRV patients (46% in APRV, 7% in other ventilation modes), but APRV patients were defined as those who received APRV at any time during the entire clinical course, and no standardized protocol was used. Most comparable in terms of the patient population to the present study was a study from Jacksonville (USA), that retrospectively analyzed CARDS patients that were transferred for ECMO evaluation. 18 Twenty of 30 patients were switched from LTV to APRV as rescue strategy and managed without ECMO, showing a mortality of 45% in APRV patients compared to 70% in patients that received ECMO, but were, according to the reference, mostly in the extremis of respiratory failure. Similarly, in the present study, significantly more APRV patients were ventilated at external hospitals prior to being transferred to the reporting institutions due to progression of respiratory failure or for ECMO evaluation, suggesting a high percentage of critically ill patients failing to previous conventional LTV that were subsequently ventilated using APRV as a rescue strategy. In the APRV group, the presence of severe ARDS was almost twice as common as in the LTV group (45% vs 23%). Nevertheless, the duration of ventilation and ICU stay tended to be lower in the APRV group than in the LTV group. With regard to the described mortality data, the results of the present study are consistent with available literature on mechanically ventilated patients with COVID-19,29,30 and no significant increase in ICU mortality was noted in APRV patients. Additionally, a Cox proportional hazards regression analysis showed that the ventilation mode is not an independent adverse prognostic factor. The rate of ventilation-associated complications such as barotrauma did not differ between the groups similar to previous studies.10,14
In 2022, the first and only RCT comparing APRV with LTV in CARDS was published. 14 However, the single-center study from Guadalajara (Mexico) was terminated early due to an interim analysis that included 45 APRV patients after the occurrence of a series of barotrauma in the APRV group and a trend toward higher mortality (78% in APRV, 60% in LTV). Furthermore, severe transient respiratory acidosis with pH < 7.15 and PaCO2 ≥ 55 mm Hg occurred twice as often in APRV patients than in LTV patients (42% in APRV, 15% in LTV). The authors named patient-related factors, prone positioning, and inconsistent APRV implementation strategies as potential causes for the severe hypercapnia. Regarding the management of hypercapnia, the protocol recommended either favoring a decrease of Thigh (to increase mandatory ventilation) over prolonging Tlow (to increase the release volume) or allowing for spontaneous breathing. However, spontaneous breathing was only permitted when Phigh was less than or equal to 24 cmH2O to prevent ventilator-induced lung injury. For this reason, patients might have been exposed to higher amounts of sedation and received 57 h of neuromuscular blocking on average. At the same time, the authors stated that the clinicians were “reluctant” to set the Thigh below 4 to 6 s, which in consequence without spontaneous breathing compensation predisposed to hypoventilation. In the present study, no differences regarding severe acidosis were noted. An opposite effect was even shown in the aforementioned study from New York. 12 After switching from a conventional ventilation mode to APRV, PaCO2 dropped significantly, while the minute ventilation decreased at the same time. The authors attributed this finding to improved alveolar recruitment and reduction of dead space ventilation. Similar observations were made when applying APRV in ARDS of other etiology. 10
In summary, the interpretation and assessment of the present study on the effect of APRV in CARDS is challenging. The current study situation is characterized by great heterogeneity regarding the indication, timing, and implementation of APRV, the experience of the medical staff, the investigated patient populations as well as the overall setting. A variety of APRV protocols, approaches, and weaning strategies are at hand. Furthermore, the different clinical application of effective treatment strategies such as prone positioning or corticosteroids further aggravates comparability. As successful ventilation using APRV depends on experience, close and frequent patient monitoring by the staff as well thorough titration of sedation and ventilation settings, caution should be exercised in cumulating data and drawing conclusion both pro and con APRV. The present study illustrated that the clinical outcome as well as complication rates can be equivalent using APRV compared to conventional LTV in patients with CARDS, while at the same time showing similar tidal volumes per PBW and peak pressure values. As a considerable number of patients in the APRV group failed to previous conventional LTV or were transferred for ECMO evaluation, APRV seems to be a safe rescue strategy and worth trying given the experience and manpower to use APRV. However, the present study does have its limitations. The largest limitation results from the small sample size. Due to the retrospective study design, residual confounding and selection bias cannot be excluded. The selected matching parameters, which included a variety of prognostically relevant factors instead of a pure focus on ventilation parameters such as the PaO2/FiO2 ratio, can also be seen critically. Despite thorough propensity score matching, there may be additional unaccounted confounders that could affect the results. Supplementary data regarding additional ventilation parameters, including information on compliance, resistance, and the intrinsic PEEP as well as data on sedation requirements cannot be provided. Due to the selected patient population with a high percentage of ventilated patients from external hospitals, positive effects from early application of APRV, including improved alveolar recruitment and reduced lung injury,6–9 could not take effect. In previous studies in ARDS of other etiologies, advantages of APRV such as reduced duration of mechanical ventilation and ICU stay were shown especially when early application of APRV was performed. 10 However, due to its multicenter design, a detailed description of APRV implementation following previously described principles 10 as well as thorough propensity score matching including important prognostic factors this study contributes needed evidence for the effect of APRV as rescue strategy in CARDS.
Conclusion
Intensive care unit survival, duration of mechanical ventilation, and ICU treatment as well as ventilation-associated complication rates were equivalent using APRV compared to conventional LTV in patients with CARDS.
Supplemental Material
sj-docx-1-jic-10.1177_08850666231207303 - Supplemental material for Airway Pressure Release Ventilation in COVID-19-Associated Acute Respiratory Distress Syndrome—A Multicenter Propensity Score–Matched Analysis
Supplemental material, sj-docx-1-jic-10.1177_08850666231207303 for Airway Pressure Release Ventilation in COVID-19-Associated Acute Respiratory Distress Syndrome—A Multicenter Propensity Score–Matched Analysis by Jan-Hendrik Naendrup, Jonathan Steinke, Jorge Garcia Borrega, Sandra Emily Stoll, Per Ole Michelsen, Yannick Assion, Alexander Shimabukuro-Vornhagen, Dennis Alexander Eichenauer, Matthias Kochanek and Boris Böll in Journal of Intensive Care Medicine
Footnotes
Acknowledgments
The authors would like to acknowledge the hard work and devotion to patient care of all ICU physicians and nurses, enabling this study.
Authors’ Note
Prior to the start of the study, approval was obtained by the local ethics committee (approval number 20-11729). Given the noninterventional retrospective nature of the study, no informed consent had to be obtained from the included patients. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors’ Contributions
JHN, ASV, DAE, MK, and BB conceived the study. JHN, JS, JGB, SES, POM, and YA conducted the chart reviews of patient and checked for enrollment. JHN, JS, and BB analyzed the data. JHN, JS, JGB, POM, YA, ASV, DAE, MK, and BB interpreted the data. JHN and BB wrote the first draft of the manuscript. JS, JGB, SES, POM, YA, ASV, DAE, and MK critically revised the manuscript. BB organized the study as an overall supervisor. All the authors reviewed the final draft of the manuscript and agreed on submitting it to the Journal of Intensive Care Medicine.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BB received scientific grants and honoraria not related to the submitted manuscript from Novartis, Kite/Gilead, Miltenyi, Roche, and Janssen & Janssen. JGB received scientific grants from Kite/Gilead not related to the submitted manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
