Abstract
Background
The World Health Organization declared the coronavirus disease 2019 (COVID-19) a pandemic on March 11, 2020. Currently, there have been over 500 million confirmed cases and 6 million deaths worldwide. 1 Critical illness from COVID-19 has increased the need for inpatient beds and intensive care unit (ICU) resources. Strain on the healthcare system and ICU have been associated with increased mortality. 2 Due to scare resources and the complexity of COVID-19 management, an interdisciplinary approach to patient care may be beneficial.
In 2001, the Society of Critical Care Medicine recognized critical care pharmacists as an important part of an ICU interdisciplinary team. 3 Patients in the ICU often receive multiple high-risk medications and are subjected to frequent changes in therapeutic plans. 4 Clinical pharmacist services have been shown to decrease medication-related adverse events in the ICU5–8 and contribute to better patient outcomes.9–14 Multiple studies have demonstrated that pharmacists increase compliance with the ICU Liberation Bundle also known as the ABCDEF (ie, Assess, prevent, and manage pain; Both spontaneous awakening and breathing trials; Choice of analgesia and sedation; Delirium-assess, prevent, and manage; Early mobility and exercise; and Family engagement and empowerment) Bundle.15–22 Finally, pharmacists have been shown to play an integral role in decreasing costs in the ICU6,23–26 and the emergency department. 27
Data on pharmacists’ interventions in critically ill patients with COVID-19 is limited. A retrospective analysis of pharmacist interventions in critically ill patients with COVID-19 in Saudi Arabia showed that pharmacists made multiple interventions related to drug procurement, drug information, and drug dosing. 28 Apart from this study, the pharmacist role in the management of critically ill patients with COVID-19 has been underrecognized and poorly described. We aim to quantify the different types of interventions made by clinical pharmacists in critically ill patients with COVID-19 in the United States.
Materials and Methods
Study Population and Design
A single center, retrospective, observational study was conducted at Vanderbilt University Medical Center (VUMC) in Nashville, Tennessee. All adult patients admitted to the COVID-19 and Medical ICUs (MICU) with confirmed COVID-19 between March 1, 2020 and June 30, 2021 were included in the study.
VUMC's pharmacy practice model consisted of clinical pharmacy specialists and decentralized clinical pharmacists who provide clinical and operational medication services to patients. During the COVID-19 pandemic, our primary MICU pharmacist provided services to both COVID-19 ICU and MICU patients. The MICU pharmacist provided clinical support services every day from 7 AM to 3:30 PM. She rounded with 2 teams daily providing verbal recommendations. Following rounds, she met with the other 2 teams in person to make verbal recommendations. Additionally, a unit-based pharmacist covered the MICU from 3:30 PM until 11:00 PM. Central-based pharmacists performed order entry overnight (11:00 PM until 7 AM) and made interventions via telephone when indicated. During that time, inventions are made in-person or via telephone. The pharmacist to patient ratio during this extremely busy time ranged between 1:35 and 1:72 patients.
All interventions made by pharmacists on patients admitted to the COVID-19 ICU or MICU, only while in the ICU, were collected, and subsequently categorized and analyzed. Interventions were counted as any medication or ICU liberation bundle recommendation made to the covering team. The team could defer if they did not think it was indicated. There were no specific protocols followed by the covering pharmacists other than a pharmacist to dose protocol for heparin anti-factor Xa levels and bivalirudin. The institution did have COVID-19 therapy guidelines, a pain/agitation/delirium orderset, and a panel that indicated what the results were of each of the letters of the ICU Liberation Bundle daily. Interventions were documented as ivents (document reporting function of EPIC, VUMC's electronic health system) as part of standard pharmacist practice that existed prepandemic. All pharmacists at VUMC were encourage to document all interventions affecting patient care as ivents
The primary outcome of this study was the median number of interventions by pharmacists per patient. The secondary outcome was quantification of the different types of interventions performed. The institutional review board at VUMC approved this retrospective observational study via an exemption.
Statistical Methods
Categorical data are reported as frequencies and percentages. Continuous data are reported as medians with interquartile ranges (IQR).
Results
A total of 768 patients with confirmed COVID-19 were admitted to the participating study ICUs during the study period. Baseline characteristics are listed in Table 1 and oxygen requirements during the hospitalization are listed in Table 2. Of 768 patients included, a pharmacist performed an intervention on 655 (86%). Those without an intervention had a shorter median hospital length of stay of 6 (IQR 3-11) versus 12 days (IQR 7-21); shorter median ICU length of stay of 1 (IQR 0-2) versus 5 days (IQR 1-11), and lower 60-day mortality (27% vs 49%) compared to patients with at least 1 pharmacist intervention. Pharmacists performed a total of 7027 interventions with a median of 6 pharmacist interventions per patient (IQR 3-14). Figure 1 shows the types of interventions pharmacists delivered. A total of 1680 medication discontinuations were performed (Figure 2), of which 359 (21%) were for antimicrobial medications, 335 (20%) for cardiac medications, and 295 (17.5%) for analgesic, sedative, or paralytic medications.
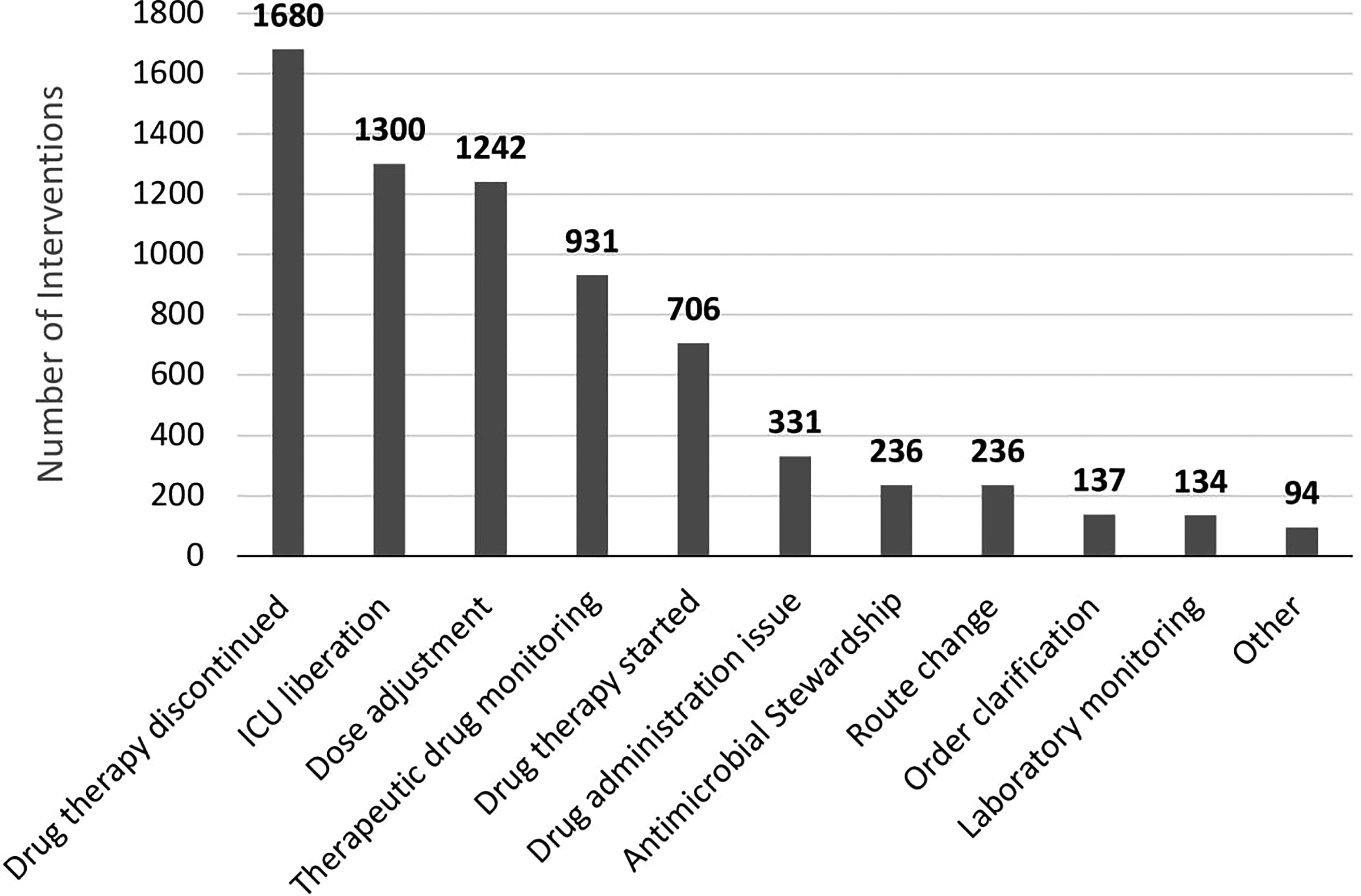
Interventions made by pharmacists. The intervention category and the number of total interventions is displayed on the x and y axis, respectively. Abbreviation: ICU, Intensive care unit.
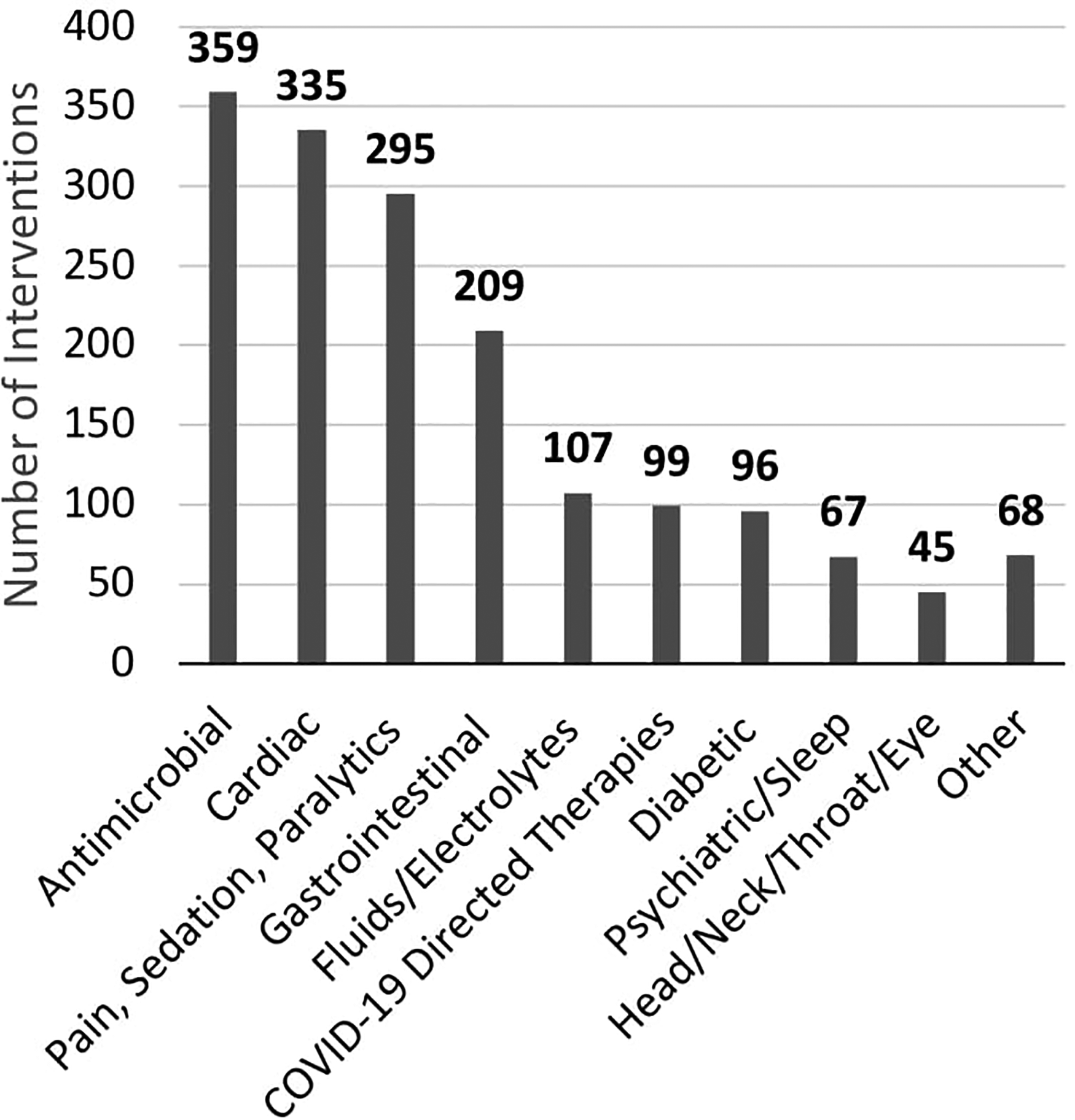
The most common intervention made was drug therapy discontinuation (n = 1680), categorized by therapeutic class. COVID-19-directed therapies include remdesivir, tocilizumab, baricitinib, and dexamethasone.
Baseline Characteristics.

Abbreviations: ICU, Intensive care unit; IQR, interquartile range; LOS, length of stay; SOFA, Sequential Organ Failure Assessment.
Oxygen Requirements.
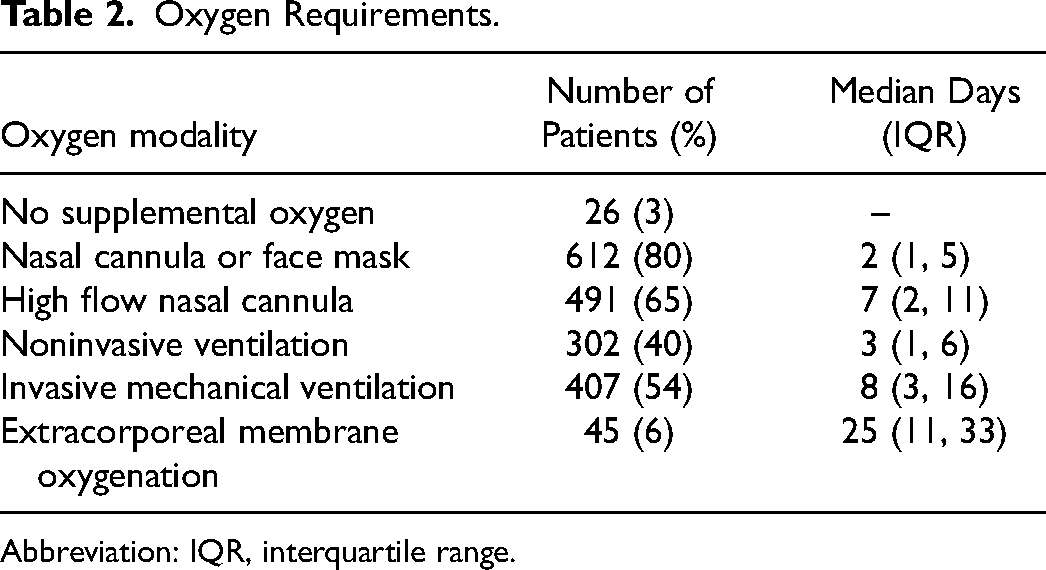
Abbreviation: IQR, interquartile range.
A total of 1300 ICU Liberation interventions were performed by pharmacists (Figure 3). These represent pharmacist-driven interventions on rounds in which the pharmacist assisted in completion of components of the ICU Liberation Bundle beyond medications. Of the 1300 ICU Liberation interventions made by the pharmacist, 665 (51%) involved discussion of whether or not it was safe to perform daily spontaneous awaking trials (SAT), 600 (46%) involved dialoging and updating Richmond Agitation Sedation Scale goals, and 35 involved recommendations of putting in a physical therapy or occupational therapy consult if the physician or advance practice provider thought clinically indicated.
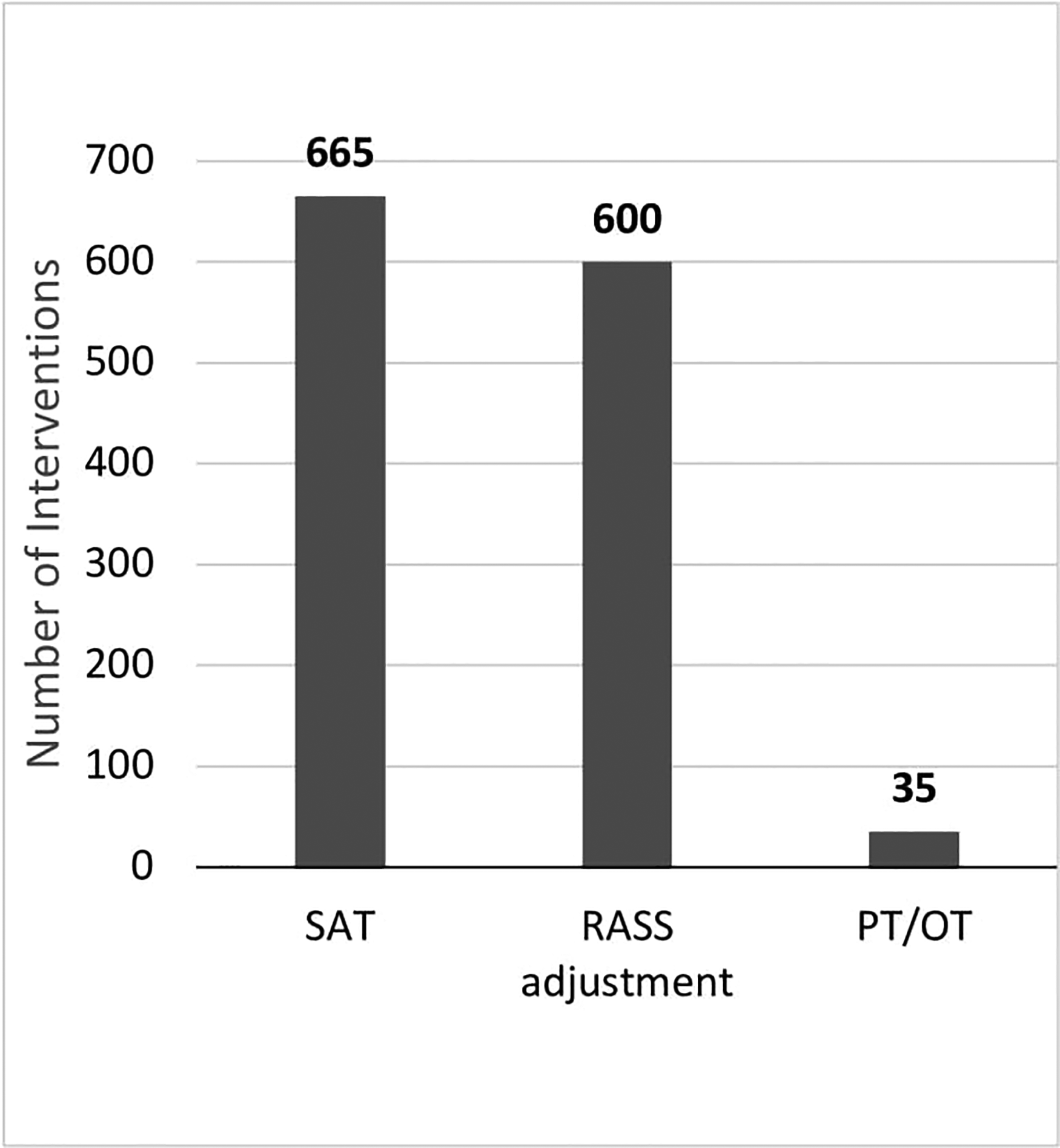
The second most common intervention made was ICU liberation (n = 1300). These interventions are categorized by type of ICU Liberation Bundle element that the intervention relates to. Abbreviation: ICU, Intensive care unit.
Medication dose adjustments were the third most common pharmacist intervention, with 1242 dose adjustments made over the course of the 16-month trial (Supplemental Figure 1). Among medication dose adjustments, the most common interventions were for diabetic medications (23%), antimicrobial medications (22%), and gastrointestinal medications (21%).
The fourth most common intervention documented was therapeutic drug monitoring (Supplemental Figure 2). Overall, there were 931 consults for pharmacist-led therapeutic drug monitoring with 453 (49%) for vancomycin or aminoglycosides, 253 (27%) for warfarin, 189 (20%) for argatroban or bivalirudin, and 36 (4%) for medication reconciliation or monitoring of other high-risk medications.
Discussion
This retrospective observational study describes pharmacist interventions in the care of critically ill adults with confirmed COVID-19. Pharmacists made interventions on the majority of patients ranging from medication initiation, dose adjustment, and discontinuation to interdisciplinary engagement in the ICU Liberation Bundles. Patients with longer ICU and hospital courses were more likely to receive pharmacy interventions.
Data on pharmacist interventions in critically ill patients with COVID-19 is limited. A single center, retrospective cohort study in Saudi Arabia which evaluated pharmacist interventions in critically ill patients with COVID-19 over a 3-month time frame, found pharmacists made 470 interventions on 79 patients. Most interventions were related to drug storage or procurement (40%) or correction of errors in dosing (32%). The most common pharmacologic category of involved medications upon which pharmacists intervened were antibiotic (16.8%) and electrolyte and mineral-related medications (11.7%). 28
In the present study, the most common intervention made was drug therapy discontinuation, emphasizing the role pharmacists play in medication reconciliation during the ICU stay and at the time of transfer out of the ICU. Previous studies in critically ill patients without COVID-19 have shown that medications started in the ICU for acute indications are frequently continued past discharge, even if no longer indicated. A prospective cohort study by Morandi et al assessed the frequency of potentially inappropriate medications and actually inappropriate medication (AIMs) prescribed at discharge to patients after an ICU stay. Common types of inappropriate medications were anticholinergic medications, opioids, and antipsychotics. Of the medications classified as AIMs, 67% of anticholinergics were initiated in the ICU, 76% of the opioids were initiated in the ICU, and 4 of every 5 atypical antipsychotic classified as AIMs were initiated in the ICU. Other studies have similarly shown that acid suppressant therapies and bronchodilators initiated in the ICU are commonly continued after discharge from the hospital, despite no continued indications for use.29–31
The second most common intervention made in this study was interventions related to ICU Liberation such as initiating discussion about performance of SATs and ensuring appropriate sedation goals were addressed daily. As one of the most consistent parts of an interprofessional ICU team, 3 pharmacists are well-equipped to help with implementation and maintenance of the different components of the ICU Liberation Bundle. A previous study evaluating a pharmacist-driven SAT quality improvement program found that pharmacists’ involvement led to improving compliance from 20% to 97% of patient-ventilator days. 15 This pharmacist involved program led to a higher SAT pass rate and enhanced compliance to a core measure in the treatment of critically ill patients. 15 Pharmacists also play an important role in selection and monitoring of sedative agents. In 2 observational studies evaluating the impact of a pharmacist-led sedation protocol, ensuring appropriate sedation levels through validated methods led to a reduction in the percentage of discomfort scores on a modified Ramsay scale and shorter duration of mechanical ventilation.16,21
Pharmacists did not make interventions in 14% of patients. Possible reasons why patients did not have an intervention performed included limited pharmacist care on the weekends. The pharmacist only working every fourth weekend fully covering the MICU while someone covered pharmacokinetics consults only during her absence on other weekends. Another reason could be learned behavior. Given the similar characteristics of these patients, practitioners anticipated what interventions were going to be made and performed them beforehand.
This study has many strengths. The results of this study echo many studies documenting the important role of critical care pharmacists in treating and preventing medication-related problems. However, this is only the second study to describe the role of pharmacist in making pharmacy interventions in critically ill patients with COVID-19. This study adds additional valuable information to the literature in that it is in a much larger patient population over a longer period of time in a different geographical setting. Additionally, while physicians and nurses have received appropriate recognition for their tremendous role of caring for patients during the COVID-19 pandemic, this study highlights the critical role pharmacists played during that time period.
Our study has several limitations. First, this was a retrospective study which did not compare care for patients with COVID-19 with and without a pharmacist. However, previous studies have shown that similar interventions by pharmacists have led to significant improvement in patient outcomes.6,9,10,23,25 Second, multiple pharmacists delivered interventions, which could have permitted variability in how each pharmacist delivered and documented their intervention. For example, de-escalating from cefepime to cefazolin could have been documented as antimicrobial stewardship, medication discontinuation, or medication initiation. Pharmacoeconomic data was not collected in our study. However, a multicenter, prospective, observational study of pharmacy interventions made by 215 ICU pharmacists at 85 centers showed a cost avoidance of $23,404,089 through performance of 55,926 interventions. 26 It is hypothesized that the number of interventions performed in this study also resulted in significant cost savings. Lastly, we do not have information on how much time was spent per intervention. This could vary diversely from for example: 30 s to change a medication from intravenous to oral to 10 min to write a complex pharmacokinetic consult for vancomycin dosing. Given this was a single-center study with majority of interventions conducted by 1 individual, it is unknown if the results of this study would be reproducible at other institutions.
Conclusion
This study found that critically ill adults with COVID-19 received a median of 6 pharmacist interventions, most commonly medication initiation, dose adjustment, or discontinuation or assuring compliance with ICU Liberation Bundle elements. Future research should examine the impact of medication-related interventions made in patients with COVID-19 on pharmacoeconomic and clinical outcomes.
Supplemental Material
sj-pdf-1-jic-10.1177_08850666231156551 - Supplemental material for Quantifying Critical Care Pharmacist Interventions in COVID-19
Supplemental material, sj-pdf-1-jic-10.1177_08850666231156551 for Quantifying Critical Care Pharmacist Interventions in COVID-19 by Jack C. Pluenneke, PharmD, Matthew W. Semler, MD, MSCI, Jonathan D. Casey, MD, MSCI, Edward T. Qian, MD, Todd W. Rice, MD, MSc, and Joanna L. Stollings, PharmD, FCCM, FCCP in Journal of Intensive Care Medicine
Footnotes
Authors Contributions
JCP and JLS contributed toward atudy concept and design, acquisition of the data, analysis and interpretation of the data, and drafting of the manuscript. Critical revision of the manuscript for important intellectual content by JCP, JLS, MWS, JDC, ETQ, TWR. All the authors took the responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MWS was supported in part by a grant from the NIH (K23HL143053). ETQ was supported by the National Heart, Lung, and Blood Institute award No. T32HL087738.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
