Abstract
Background:
Hepatocellular carcinoma (HCC) has an uneven global distribution and a higher incidence in males. The Liver Imaging Reporting and Data System (LI-RADS) is a standardized imaging framework for HCC diagnosis. It is unclear whether the evidence used to inform LI-RADS reflects the global burden of HCC.
Purpose:
To determine whether the geographic and sex distribution of studies assessing LI-RADS and those included in the LI-RADS individual participant data (IPD) database reflect the global burden of HCC.
Materials and Methods:
We conducted a cross-sectional meta-research study comparing the country- and sex-specific HCC prevalence from GLOBOCAN 2022 with the distribution of studies eligible for and included in the LI-RADS IPD database. Studies were identified through a systematic search of 4 databases. Study Protocol:
Results:
We identified 470 eligible studies including 98 014 patients; of these, 76 studies comprising 11 924 patients were included in the IPD database. Asian and African countries, excluding Republic of Korea, were underrepresented in both the eligible and the IPD dataset. North America was overrepresented. Female patients were under-represented in LI-RADS eligible studies (Z = −21.95, P < .0001) and in studies included in the IPD (Z = −9.02, P < .0001) compared to the global prevalence of HCC in females.
Conclusion:
LI-RADS research is disproportionately reported from some countries relative to HCC burden. Asia, Africa, and female patients remain underrepresented. This may affect the generalizability and diagnostic equity of the LI-RADS system and underscore the need for improved global inclusivity in LI-RADS research.
Introduction
Hepatocellular carcinoma (HCC) is the most common primary liver malignancy, accounting for 906 000 new cases and 830 000 deaths globally in 2020. 1 Common risk factors for HCC include patients with chronic liver disease such as cirrhosis or chronic hepatitis B virus (HBV) and patients with a prior history of HCC. HCC occurs more frequently in male patients (3:1 M/F) and is unevenly distributed globally, with 76% of cases found in Asia, compared to 10.0% in Europe and 5% in the United States. 2 In the Eastern hemisphere, cirrhosis secondary to chronic HBV is the primary risk factor for HCC, while in the Western hemisphere, the leading risk factors include cirrhosis due to chronic hepatitis C virus (HCV), alcohol-associated liver disease (ALD), and metabolic dysfunction-associated steatotic liver disease (MASLD). 3 Among patients with HCC in the United States, 50% to 60% are infected with HCV, 20% to 25% have ALD, and 20% to 30% exhibit features of the metabolic syndrome, including diabetes and obesity, which are closely linked to MASLD. 3
The Liver Imaging Reporting and Data System (
The LI-RADS
A potential source of bias that may impact generalizability of the literature is an imbalance between the global burden of HCC and the geographic distribution of published LI-RADS studies. For instance, if most LI-RADS research originates from regions with a high proportion of HCC secondary to HBV infection, applying LI-RADS in populations with different predominant risk factors, such as MASLD, may impact different diagnostic performance. In addition, low- and middle-income countries which are often under-represented in randomized trials and biomedical research more broadly, and females who are historically underrepresented due to sex-based exclusions in study design may also impact the generalizability of published LI-RADS studies.7,8 The purpose of this study is to determine whether the geographic and sex distribution of studies assessing LI-RADS and those included in the IPD database reflect the global burden of HCC.
Methods
Study Design
We performed a cross-sectional meta-research study examining the global burden of HCC in relation to the geographic and sex representation of studies eligible for or included in the LI-RADS Living Systematic Review and IPD Meta-Analysis Project. The study protocol is available on the
The LI-RADS living systematic review project has approval from the institutional review board (IRB) at The Ottawa Hospital. The LI-RADS IPD project has assessed numerous accuracy measures pertaining to LI-RADS; this study represents a unique analysis with no overlap with prior work.9-19 No personal health information was disclosed to the project team. This study integrated data from
LI-RADS IPD Search and Inclusion Criteria
Search, screening, and inclusion methods are detailed on our
All CT, MRI, and CEUS studies reporting the percentage of HCC for LI-RADS categories 1 to 5 in patients at high risk (hepatic cirrhosis, chronic HBV infection, current or prior HCC) were eligible for inclusion. CT, MRI, and CEUS techniques were evaluated for each study to determine concordance with the LI-RADS technical imaging guidelines.
Data Collection Process and Definitions for Data Extraction
Data from eligible studies included in the LI-RADS IPD were stored in DistillerSR, with information extracted on country of origin, sample size, and reported sex distribution. While the analysis focused on sex due to its availability in study demographics, the influence of gender could not be assessed because it was not reported in the included studies. The prevalence of primary liver cancer (ICD-10 C22), was obtained from the GLOBOCAN 2022 database for 185 countries and territories, as well as the male to female global proportion of HCC. 20 Data were extracted in duplicate by 2 assessors with disagreements resolved by consensus and when necessary, discussion with a third reviewer.
Outcome Measures
To quantify the available LI-RADS evidence, 2 measures of published LI-RADS evidence were used: the number of published LI-RADS studies eligible for inclusion into the LI-RADS IPD, and the number of studies for which data are available within the LI-RADS IPD Redcap database. Location of study was determined by identifying the country of the corresponding authors as well as the description of setting in the methods. For studies with patients from multiple countries, the study “counted” for each of these countries. Both measures of LI-RADS evidence base were calculated as a percentage of the number of studies from each country, divided by the total number of studies from all countries.
The estimated burden of disease for HCC was calculated by the number of individuals with HCC in a country, divided by the total number of individuals with HCC globally. To assess the representation of females in the eligible studies, including IPD, and the GLOBOCAN database, we calculated the proportion of female patients within each dataset. Specifically, we determined the number of females in each eligible study and in the GLOBOCAN database.
Statistical Analysis
To compare the burden of disease for HCC in each country to the percentage of LI-RADS studies eligible for inclusion to the LI-RADS IPD, and to the percentage of the studies from each country that is available within the IPD, we calculated the differences between percentages of prevalence in each country reported in GLOBOCAN, and eligibility percentage in our database. The possible range of outputs in differences ranged from −100% to +100%, representing maximum under-representation to maximum over-representation respectively. A z-test was conducted to assess for differences in sex proportions between LI-RADS-eligible studies and studies included in the IPD database to those reported in GLOBOCAN. All analyses were performed using R (version 4.3.1) and visualization using the ggplot2 package and mapmychart visualization software.21-23
Results
The study selection process is summarized in the PRISMA flow diagram in Figure 1.
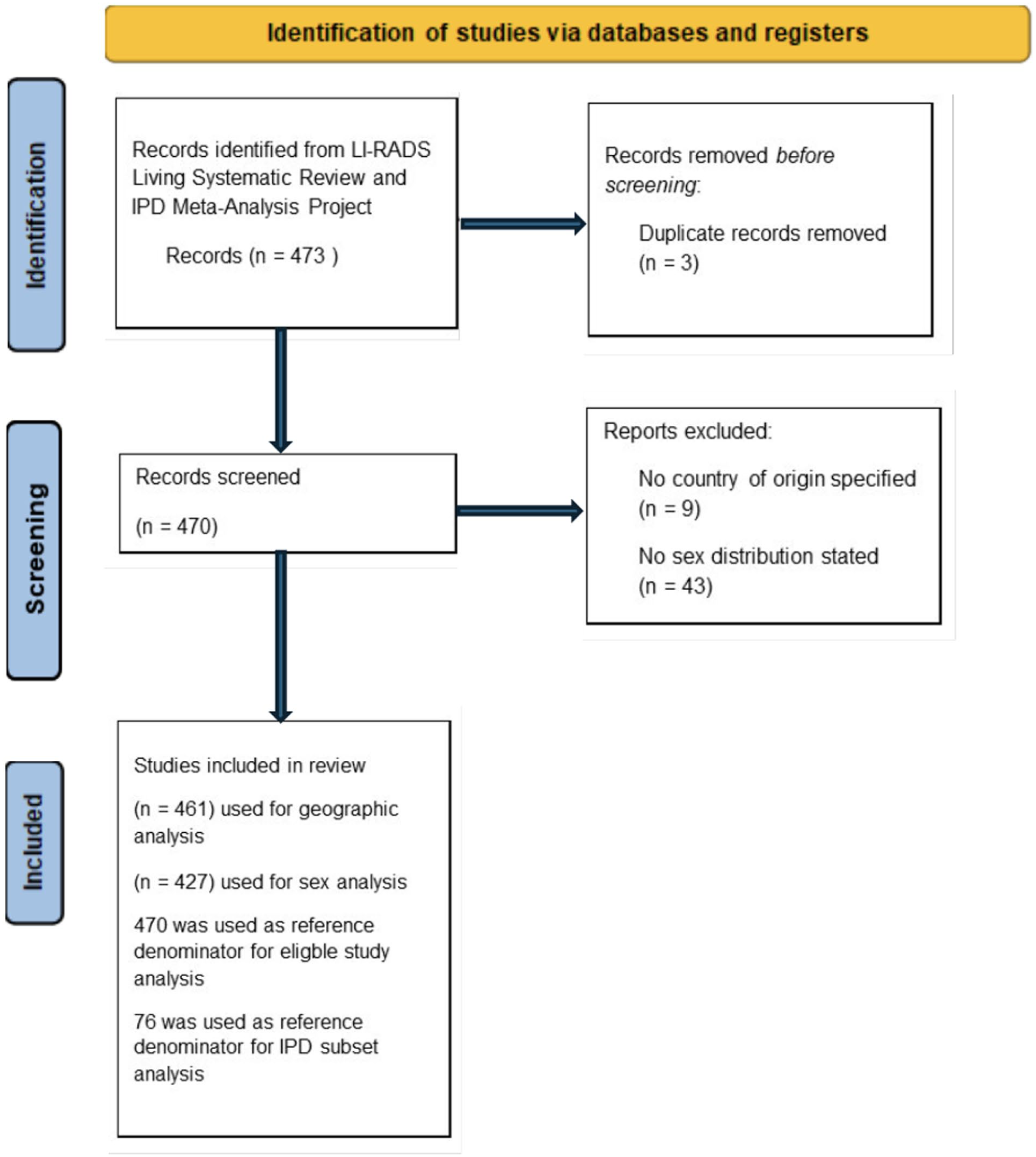
PRISMA 2020 flow diagram of study selection for geographic, sex-based, and IPD subset analyses.
Table 1 presents the global distribution of primary liver cancers based on age-adjusted prevalence data from the GLOBOCAN 2022 database.
Global Distribution of Hepatocellular Carcinoma (HCC) by Region, with Total Number of Male and Female Cases.
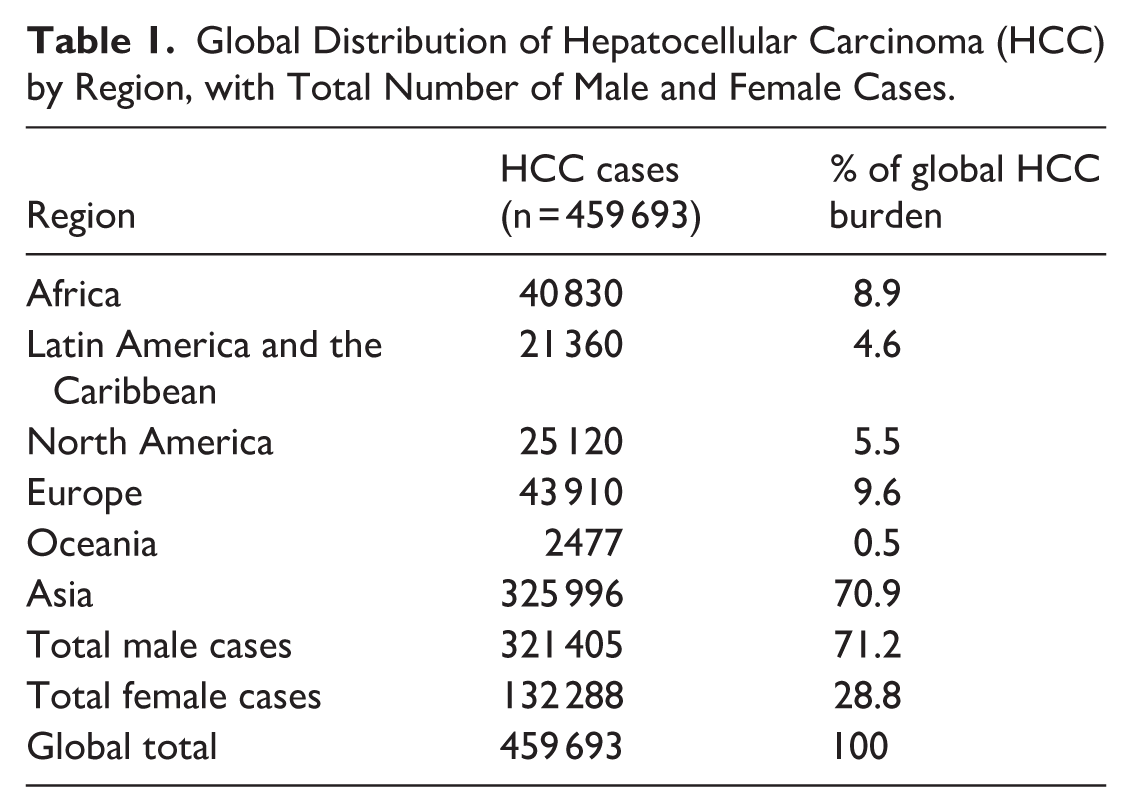
Table 2 presents the number and percentage of LI-RADS eligible studies and IPD studies contributed by each country. Of the 470 eligible studies, the largest proportion originated from China (n = 165; 35.1%), followed by the Republic of Korea (n = 115; 24.5%) and the United States (n = 69; 14.7%).
Name and Proportion of LI-RADS Eligible and Individual Participant Data (IPD) Studies by Country of Origin (Countries with ≥1 Eligible Study Only).

Figure 2 presents a forest plot comparing the difference between the percentage of global HCC burden per country and the percentage representation of each country in the LI-RADS eligible studies and in studies included in the IPD database. Most countries cluster tightly around the zero-difference mark, reflecting relatively proportional representation. However, a subset of countries demonstrates substantial divergence.
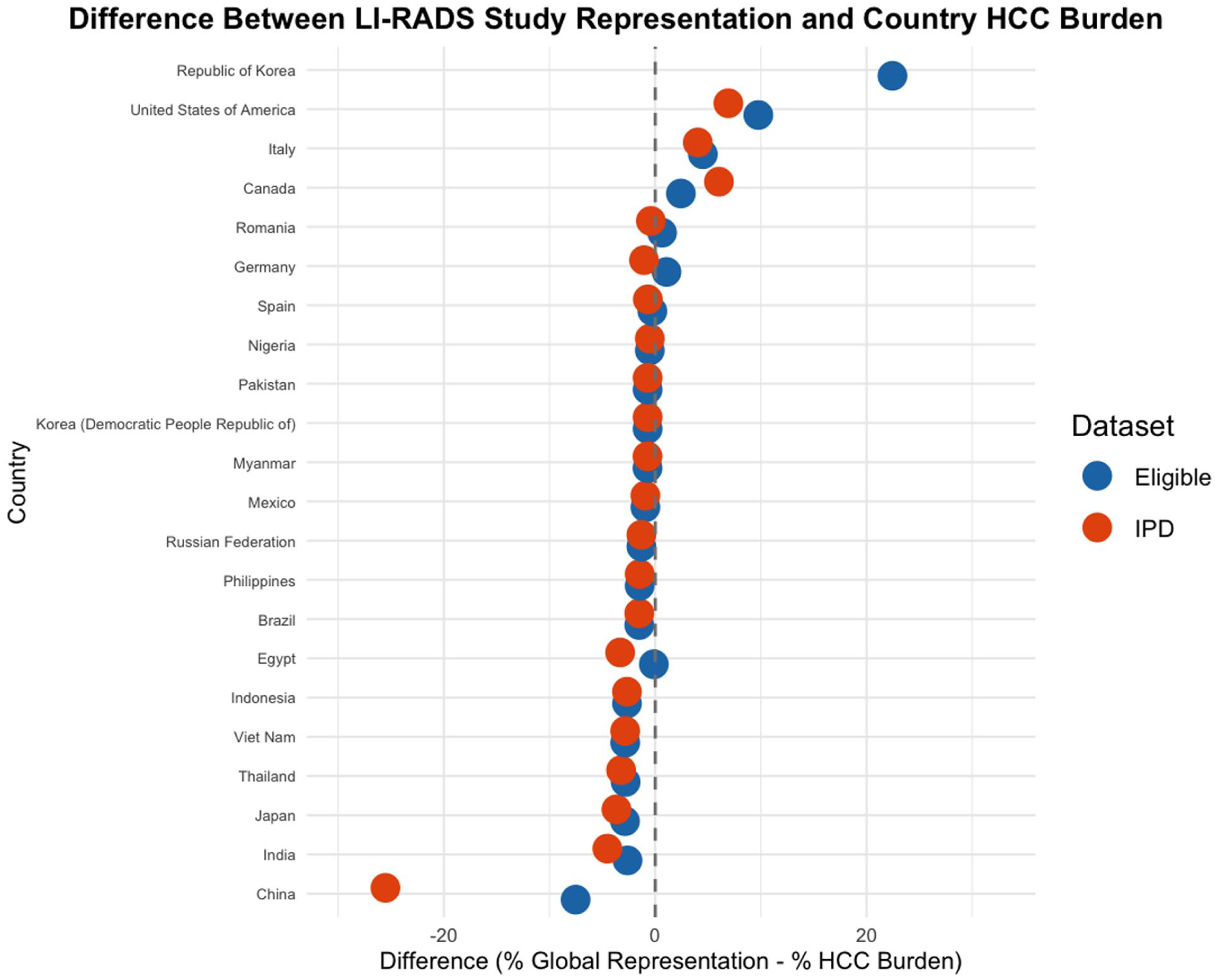
Forest plot depicting discrepancies between Global HCC burden and country representation in LI-RADS research. This forest plot illustrates the difference between each country’s representation in LI-RADS research and contribution to the global hepatocellular carcinoma (HCC) burden. Blue dots represent the difference between the proportion of eligible studies originating from each country and that country’s share of the global HCC burden. Red dots represent the difference between the proportion of individual patient data (IPD) studies and the global burden. The x-axis indicates the percentage point difference (% Representation − % HCC Burden), with a dashed vertical line at 0 marking proportional alignment. Countries with a difference of 0.5% ± of 0 were excluded for readability purposes. Positive values reflect overrepresentation, while negative values indicate underrepresentation. Only countries with data available for at least one study type are shown. Select countries with extreme discrepancies are labeled.
Figure 3 illustrates the difference between each country’s share of the HCC burden and its representation in the LI-RADS database using a global heatmap.
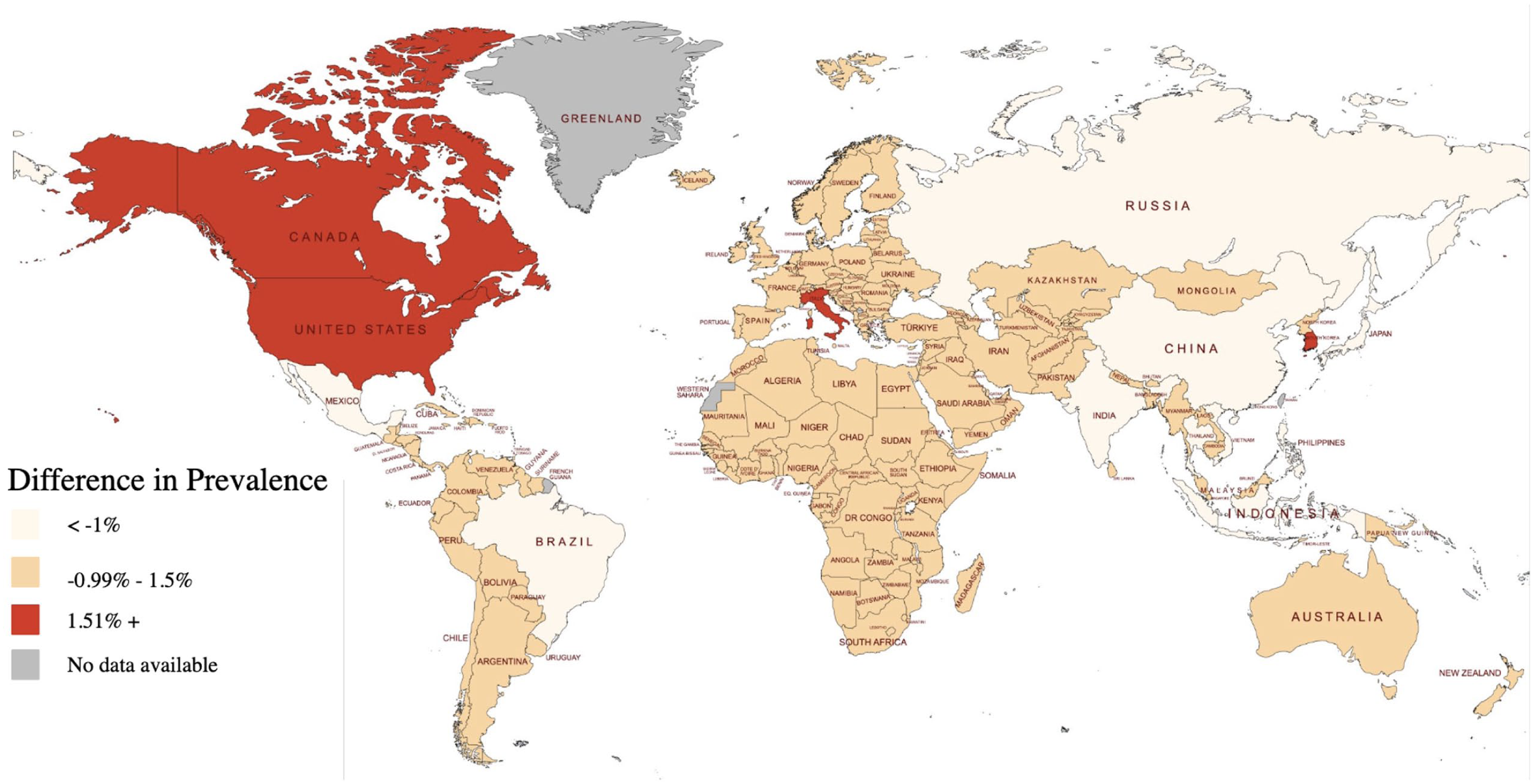
Geographic visualization of disparities between eligible study representation and global HCC burden by country. Global map displays the difference between each country’s share of global HCC burden and the proportion of eligible studies it contributed. Countries shaded in dark red are those that contributed far more studies than expected based on their disease burden (>1.51% overrepresentation). Countries shaded in orange represent a −0.99% to 1.51% difference. Beige shades indicate a <−1% difference. Gray areas indicate missing data. Image created with mapmychart.net.
Of the 76 studies included in the LI-RADS IPD database, the Republic of Korea accounted for 50.0% (n = 38), China for 17.1% (n = 13), and the United States for 11.8% (n = 9). Canada, Italy, and Poland contributed 5 (6.6%), 4 (5.3%), and 2 (2.6%) studies respectively.
Supplemental Table 1 presents a summary of primary liver cancer prevalence (both sexes, ages 15-85+) based on GLOBOCAN 2022 data, alongside the proportion of LI-RADS studies attributed to each country.
Several countries such as Bangladesh, Australia, and Angola were absent from the LI-RADS eligible study database. Albania, Armenia, Azerbaijan, Austria, Bahamas, Bahrain, Barbados, and Belize each had reported HCC cases ranging from 10 to 534, yet none were represented in the LI-RADS eligible studies or in studies included in the IPD database.
India accounted for 4.53% of the global HCC burden and contributed to 1.91% of eligible studies.
China, which carries the highest global burden of hepatocellular carcinoma (42.6%), was underrepresented in both the LI-RADS eligible study dataset and in studies included in the IPD database. The proportion of studies from China comprised only 35.1% of the eligible dataset and 17.1% of studies included in the IPD database, yielding a difference of −7.5% and −25.5%, respectively, when compared to its share of global HCC burden.
Republic of Korea contributed 115 eligible studies (24.47%) and 38 of IPD studies, accounting for 50.00% of all IPD studies, despite a global HCC burden of only 2.0%, yielding a difference of +24.5% (eligible studies) and +48.0% (studies included in the IPD database).
To visualize broader regional patterns, we plotted the difference between each region’s contribution to LI-RADS studies and its share of the global HCC burden. Positive values reflect overrepresentation, while negative values indicate underrepresentation. Differences in region-level representation can be seen in Figure 4.
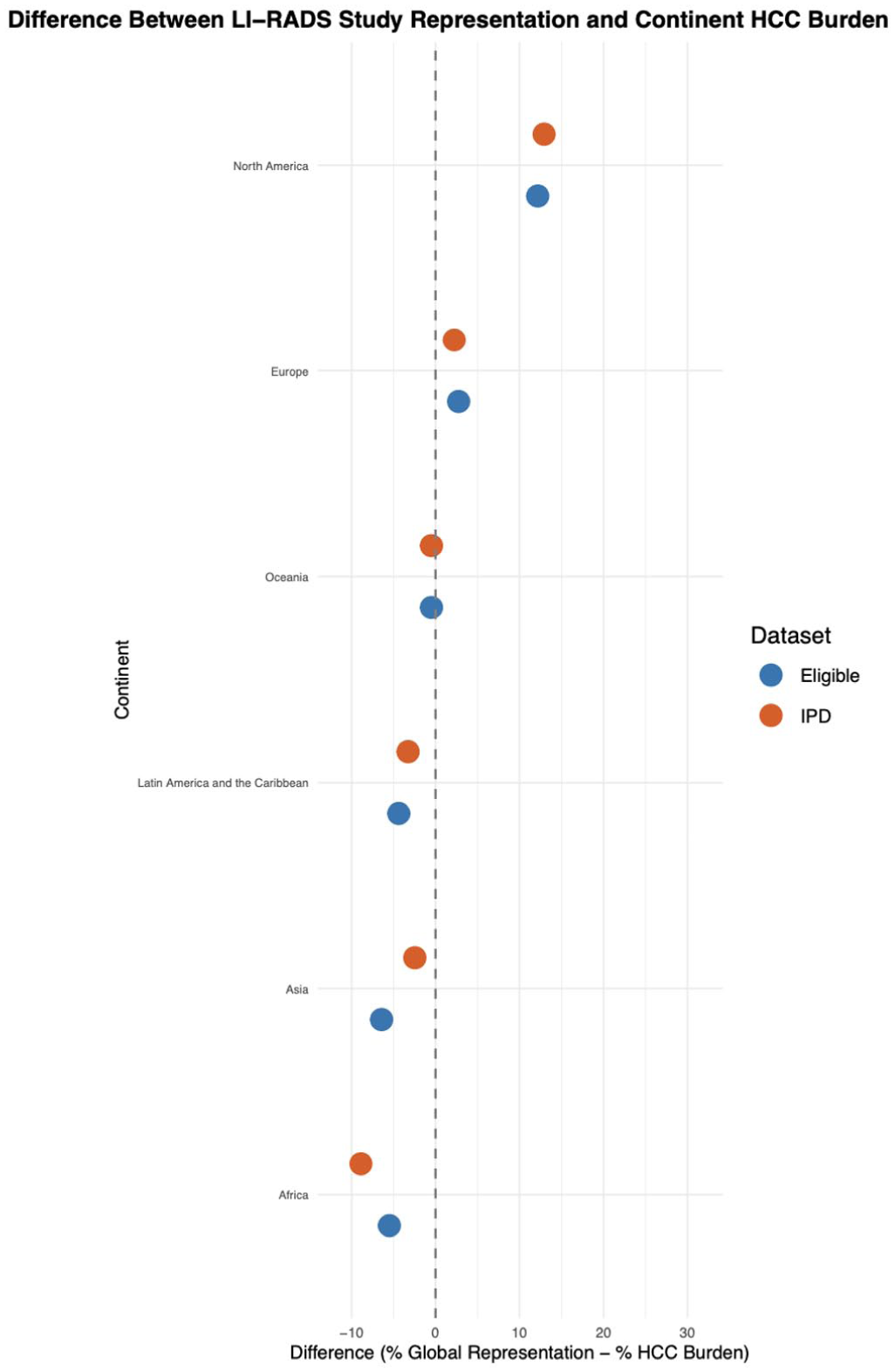
Difference between LI-RADS study representation and global hepatocellular carcinoma (HCC) burden by region. This plot displays the percentage point difference between each region’s representation in LI-RADS studies and its share of the global HCC burden. Blue points represent eligible studies; orange points represent studies contributing individual patient data (IPD). The vertical dashed line at 0% indicates proportional representation. Positive values reflect overrepresentation in LI-RADS research relative to disease burden, while negative values indicate underrepresentation.
Sex Representation in LI-RADS Eligible Studies Versus Global HCC Estimates
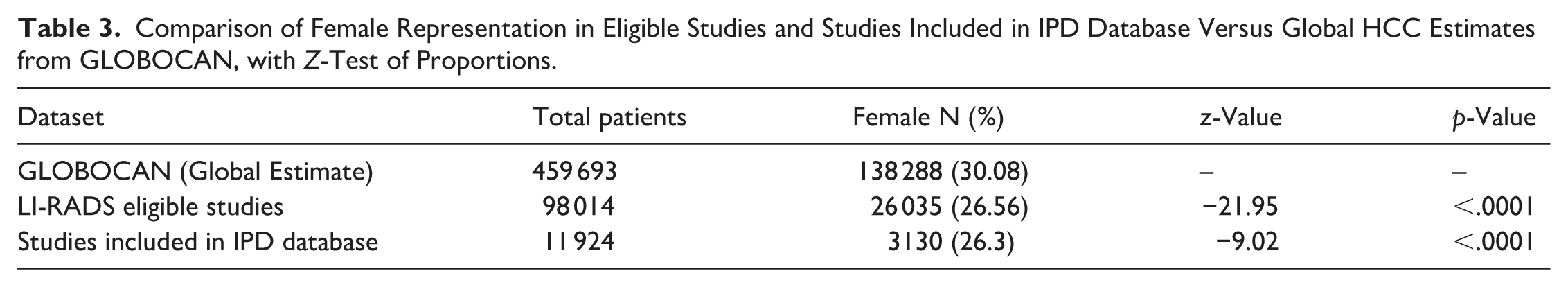
Among the 98 014 patients included in LI-RADS eligible studies, 26 035 were female, corresponding to 26.6% of the sample. Z tests of proportions are shown in Table 3.
Comparison of Female Representation in Eligible Studies and Studies Included in IPD Database Versus Global HCC Estimates from GLOBOCAN, with Z-Test of Proportions.
Discussion
This study identifies underrepresentation of patients from regions with the highest global disease burden, most notably China, India, Japan, Egypt, and other countries across Asia and Africa in LI-RADS research. Our study also identified a significant underrepresentation of female patients in LI-RADS-eligible studies relative to global HCC prevalence.
In East and Southeast Asia, HCC is predominantly driven by HBV, often presenting in younger patients and associated with higher alpha-fetoprotein (AFP) levels. 24 Conversely, in Western nations such as the United States and Canada, which are overrepresented in the IPD database, HCC is more often associated with HCV, ALD, and MASLD. 3 These etiologic differences may influence tumor biology and vascular profiles, and thereby impact imaging characteristics and LI-RADS classification. 25 HBV-related HCCs for example (common in East and Southeast Asia) present more frequently as well-differentiated, non-hypervascular tumors in early stages, which may not meet LR-5 criteria due to absent or subtle enhancement features. 26
The underrepresentation of China, a nation where nearly half of all global patients with HCC reside, is of particular importance. China’s health system has made substantial investments in liver cancer screening, particularly through ultrasound surveillance programs in HBV-infected populations, making it uniquely positioned to benefit from optimized imaging strategies. 27 Yet, the lack of Chinese IPD in LI-RADS research may limit the system’s performance in this setting, potentially reducing its value in clinical practice where it is most needed. LI-RADS is not yet widely adopted in China, in part due to variation in imaging standards and the use of national frameworks such as the China National Liver Cancer (CNLC) guidelines, updated in 2024. 28 Differences in contrast agent emphasis and inclusion of subcentimeter HCC may pose challenges for standardizing diagnostic criteria, underscoring the importance of including Chinese IPD in future efforts.
Another pattern in our results is the sharp drop in country representation from the eligible study level to the IPD level. For instance, Egypt, India, and Germany all had eligible studies but no representation in the IPD subset. Possible contributing factors, such as language, infrastructure, and data-sharing logistics, may prevent the inclusion of valuable participant-level data from these regions. 29 Overreliance on data from a narrow set of countries risks limiting the generalizability of LI-RADS toward specific populations.
Although HCC has a known male predominance (3:1 M/F) this does not account for the underrepresentation of female patients in diagnostic studies. 2 Although recent IPD evidence suggests that LI-RADS major feature performance is consistent across sex, these findings are derived from datasets with underrepresentation of female patients and several high-burden regions, and therefore may not fully capture potential effects of sex or geography on diagnostic accuracy in globally representative populations. Biological sex may influence the presentation and detection of HCC; for example, Liou et al reported that females were more likely to be diagnosed via surveillance (37.9% vs 29.6%, P = .003), resulting in detection at earlier stages with smaller tumors (median 30 vs 39.5 mm, P = .038) and lower rates of portal vein invasion and metastasis. 30 These differences suggest that sex-specific variations, whether biological, behavioral, or systemic, can influence tumor characteristics and imaging outcomes. 30 If diagnostic systems like LI-RADS are primarily developed and validated in male-predominant cohorts, they may not be applicable for female patients, increasing the risk of indeterminate or false-negative classifications. Despite this potential risk, a recent study by Adamo et al found no significant difference in the association of LI-RADS major features with HCC between males and females, which reduces concern related to the observed underrepresentation found in females in eligible studies and studies included in the IPD database. 14 However, these conclusions remain dependent on the representativeness of the underlying dataset, underscoring the importance of ensuring global inclusivity in future LI-RADS research. In addition, longstanding issues regarding sex bias in radiologic research emphasize the need for sex-stratified validation of imaging criteria. 31 In many regions, particularly North America and the Middle East, the incidence of MASLD related HCC is increasing, while the contribution of viral hepatitis is declining. As the etiologic drivers of HCC continue to transition away from viral causes toward MASLD, the traditional male predominance of HCC may attenuate over time, potentially narrowing the observed sex gap in disease incidence. These trends demonstrate the importance of ensuring that diagnostic systems such as LI-RADS are validated in contemporary and evolving patient populations, including those with MASLD-related liver disease. 32
Our study emphasizes an important message to the LI-RADS community and imaging researchers in general. Given that the LI-RADS IPD database is used to guide updates to the LI-RADS system, the observed imbalances call for methodological reforms and improved representation, both in study recruitment and in the curation of datasets used for meta-analysis. Furthermore, future research should adopt stratified sampling approaches when designing validation studies to ensure balanced representation across sex, geographic regions, and underlying disease etiology. Such representation is critical for developing diagnostic tools that are effective and generalizable across patient populations. In regions where HCC presents with distinct etiologic and phenotypic characteristics, such as HBV-driven disease in East Asia or aflatoxin-related HCC in sub-Saharan Africa, regionally adapted or etiology-specific modifications of LI-RADS may enhance diagnostic accuracy and clinical utility. Finally, journals and funding agencies can play an important role by recommending disaggregated reporting of sex and geography in diagnostic accuracy studies and incentivizing inclusion of underrepresented populations.
This study has several limitations. Our analysis relied on global HCC burden estimates from the GLOBOCAN 2022 database, which, while widely used, may be subject to inaccuracies. Notably, the GLOBOCAN classification groups HCC and intrahepatic cholangiocarcinoma (ICC) under the broader category of “liver cancer” (ICD-10 code C22), limiting the specificity of burden estimates for HCC alone. This may result in overestimation of true HCC prevalence in countries where ICC constitutes a higher proportion of “liver cancer” cases. However, HCC comprises approximately 90% of all primary liver cancers globally, minimizing the impact of this limitation. 33 Because GLOBOCAN reports liver cancer under a combined category that influences HCC and intrahepatic cholangiocarcinoma (iCCA), sex-based estimates may be slightly affected. iCCA has a sex distribution of 1.1:1 M/F, compared to 3:1 for HCC, which may increase the proportion of females in the global estimate. 34
Third, variability in cancer registration systems, diagnostic capacity, and healthcare infrastructure across countries introduces additional limitations. For example, the validity of GLOBOCAN estimates has been assessed for other cancer types, with studies showing that accuracy varies by cancer site, prognosis, and data availability. 35 It was found that cancers with low mortality or those detectable by screening (eg, thyroid, prostate, breast) had less accurate estimates compared to cancers with poor survival, highlighting the method’s limitations. 35
Conclusion
This study highlights major geographic and sex-based disparities in LI-RADS research, raising concern about its global applicability and diagnostic equity. High-burden countries are underrepresented while low-burden countries are overrepresented. Female patients are also significantly underrepresented compared to global HCC demographics. Addressing these gaps will likely require investment in data infrastructure and inclusive recruitment strategies to ensure LI-RADS reflects the diversity of the global HCC population.
Supplemental Material
sj-docx-1-caj-10.1177_08465371261429344 – Supplemental material for Geographic and Sex Distribution of LI-RADS Research Globally: A Cross-Sectional Meta-Research Study
Supplemental material, sj-docx-1-caj-10.1177_08465371261429344 for Geographic and Sex Distribution of LI-RADS Research Globally: A Cross-Sectional Meta-Research Study by Hoda Osman, Eric Lam, Christian B. van der Pol, Hala Talal Mahdi, Rawan Awad, Nabil Islam, Mostafa Alabousi, Jean-Paul Salameh, Mustafa R. Bashir, Andreu F. Costa, Haresh Naringrekar, Justin Presseau, An Tang and Matthew D. F. McInnes in Canadian Association of Radiologists Journal
Footnotes
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Canadian Institutes of Health Research (CIHR) Operating Research Grant (#469052) to Matthew DF McInnes, Radiological Society of North America (RSNA) R&E Foundation Research Scholar Grant to Christian van der Pol, and Fonds de recherche du Québec en Santé (FRQ-S) Merit research scholarship (#376472) to An Tang.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
