Abstract
Immunotherapy with checkpoint inhibitors (ICI) has revolutionized oncology by stimulating the immune system to target cancer cells. While effective in treating various malignancies, ICI presents unique challenges in radiological response assessment. Traditional criteria, such as RECIST 1.1, were designed for cytotoxic chemotherapy and fail to account for pseudo-progression—an immune-related phenomenon where tumour size transiently increases due to immune cell infiltration before eventual shrinkage. This occurs in a minority of patients and can lead to misclassification of treatment response. To address this, new assessment criteria have been developed. The immune-related response criteria (irRC) introduced a delayed assessment of new lesions, followed by immune-related RECIST (irRECIST), which sought to align with RECIST 1.1. However, inconsistencies in its application led to the development of iRECIST in 2016, a standardized framework integrating RECIST 1.1 with immunotherapy-specific modifications. Despite its potential to become the gold standard, iRECIST is complex and challenging to implement consistently. This review outlines key differences between RECIST 1.1 and iRECIST, explains their necessity, and provides comprehensive flowcharts and graphical representations to aid interpretation. By addressing common clinical scenarios and frequently asked questions, this article aims to enhance understanding and application of iRECIST in clinical practice.
Background
Immunotherapy with checkpoint inhibition (ICI) is a novel oncology treatment that acts by stimulating the immune system to target and destroy cancer cells. A number of clinical trials have proven its efficacy, leading to regulatory approval for use in multiple forms of cancer such as melanoma, lung cancer, and renal cell carcinoma.1-7
Radiological assessment of tumour burden plays a key role in guiding oncological management. Among the formal radiological response assessment methods,8-15 the most widely used is the Response Evaluation Criteria in Solid Tumours version 1.1 (RECIST 1.1). 11 RECIST 1.1 was developed in the era of cytotoxic chemotherapy prior to the introduction of immunotherapy. Response to cytotoxic chemotherapy is characterized by a reduction in tumour burden, without the development of new lesions. This is not always the case with ICI. Occasionally, an increase in tumour size can be seen in patients who are actually responding to treatment. This is termed pseudo-progression. This is a relatively rare phenomenon, for example, occurring in 2% to 8% of patients being treated with anti-PD-1/PD-L1 agents for non-small cell lung cancer and 9.7% of patients receiving ipilimumab for melanoma.16,17
Pseudo-progression occurs due to tumour infiltration by immune cells. This leads to a transient increase in tumour size, even though the patient may in fact be responding to treatment. This treatment response cannot be accurately captured by RECIST 1.1. Therefore, a number of revised assessment criteria have been devised to allow for the possibility of pseudo-progression.
The immune-related response criteria (irRC) were the first new criteria to be proposed. The major difference between irRC and RECIST 1.1 lies in the assessment of new lesions. In irRC, the development of new lesions is not automatically considered disease progression (as is the case with RECIST 1.1) and a 4 week interval is recommended for follow-up imaging to allow for possible pseudo-progression. The development of irRC was followed by the development of immune-related Response Evaluation Criteria In Solid Tumours (irRECIST).18,19 irRECIST aimed to provide greater alignment between irRC and the concepts of RECIST 1.1. 20 However, the recommendations of irRECIST have not always consistently been applied, leading to concerns regarding the consistency of results amongst various trials and suboptimal clarity on which concepts have been applied in each case. 21
In 2016, a subcommittee of the RECIST working group convened with the aim of standardising clinical trial data acquisition for immunotherapy. This led to the development of the Immune Response Evaluation Criteria in Solid Tumours (iRECIST), 21 encompassing the level of detail of RECIST 1.1 but allied to the principles of irRC and irRECIST. Currently, iRECIST is only in use in clinical trials. Although not yet fully validated for use in routine clinical practise, it is expected that iRECIST will become the gold standard for size based response assessment in the era of immunotherapy with checkpoint inhibition.
Given the detail and complexity of iRECIST, it can be challenging to use in practise. There are multiple potential eventualities when interpreting a case using iRECIST and this can lead to confusion for the interpreter. This review article firstly outlines the differences between RECIST 1.1 and iRECIST, while explaining why those differences are necessary. Secondly, comprehensive flow charts are provided, aiding the interpretation of cases at each step within iRECIST. This is followed by graphical representation of important clinical scenarios described in iRECIST, including scenarios that are commonly encountered as well as scenarios that interpreters may find challenging when using iRECIST. Finally, this article discusses frequently asked questions related to iRECIST, aiming to address any additional challenging concepts within the criteria.
RECIST 1.1 Versus iRECIST
iRECIST is in many ways similar to RECIST 1.1. Indeed, up to the point of progression there are few differences. With this in mind, the key areas for comparison between RECIST 1.1 and iRECIST are summarised below:
In general, many definitions and principles in RECIST 1.1 are carried over into iRECIST, particularly those that govern tumour assessment before progression has occurred.
Definitions of measurable and non-measurable disease; numbers and sites of target disease are unchanged
Definitions of complete response, partial response and stable disease are unchanged
Definition of progression of disease: The definition of the first episode of progressive disease is unchanged, but the principles of subsequent follow up are new in iRECIST (Figures 6-17).
Definition of new lesions: The definition of what denotes a new lesion is unchanged (for both target and non-target lesions). New lesions are followed up in iRECIST (if the patient is stable), but not in RECIST 1.1, in RECIST 1.1 they denote progressive disease (PD).
Principles of dealing with lesions that become too small to measure, lesions that split or coalesce, and cystic lesions are unchanged.
Principles of measurement and imaging techniques are unchanged.
The main point of difference occurs when patients develop progression of disease. In both RECIST 1.1 and iRECIST this is denoted by target lesion growth ≥20%, or unequivocal growth in non-target lesions, or new lesion development. At this point, assessment stops in RECIST 1.1.
However, in iRECIST, this scenario is termed unconfirmed progression (iUPD). Investigators have the option of performing a repeat assessment in 4 to 8 weeks, at which point there are 5 possibilities which can occur:
○ Confirmed progression (iCPD).
○ Continued iUPD.
○ Stable disease (iSD).
○ Partial response (iPR).
○ Complete response (iCR).
The reason the option for repeat assessment exists is to account for the possibility of pseudo-progression. It is important to emphasize that treatment beyond the initial designation of progression (iUPD in iRECIST) should only be considered in select cases of patients who are clinically stable or improving. Though it may be appropriate in select cases to treat beyond progression, inappropriate treatment beyond what is actually true progression may lead to reduced performance status and limit patients’ suitability for alternative treatment strategies. 19
iRECIST: A Practical Approach Summarised
Provided below are multiple flow charts, outlining the potential directions of a case at each potential response status within iRECIST (Figures 1-5). For example, for a patient at iUPD, the associated flow chart explains all the potential response status changes that can occur from that point. These flow charts can serve as a useful reference tool when reporting studies using iRECIST.
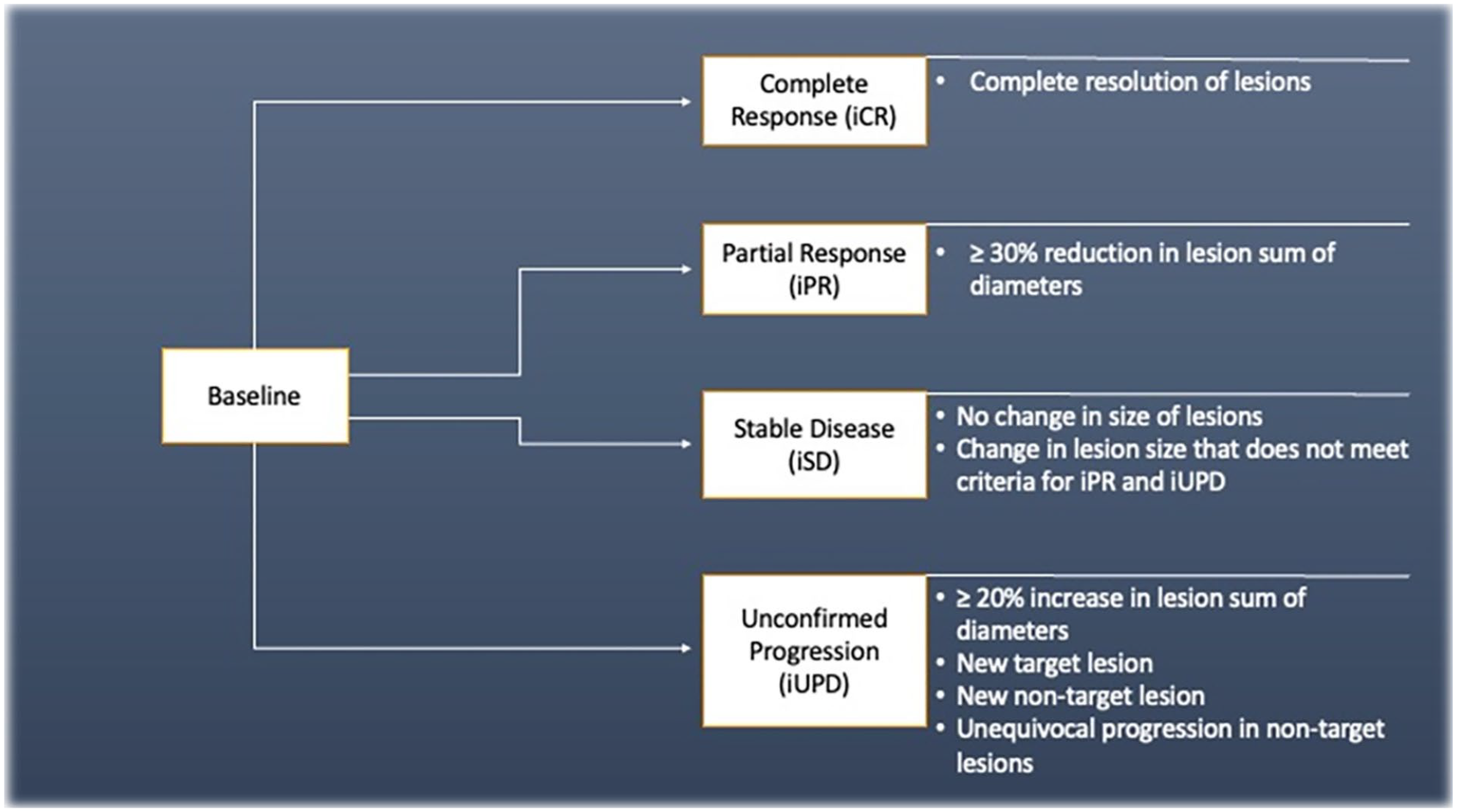
Displays the potential changes in status of a case from the baseline study, outlining the potential ways each outcome can occur.
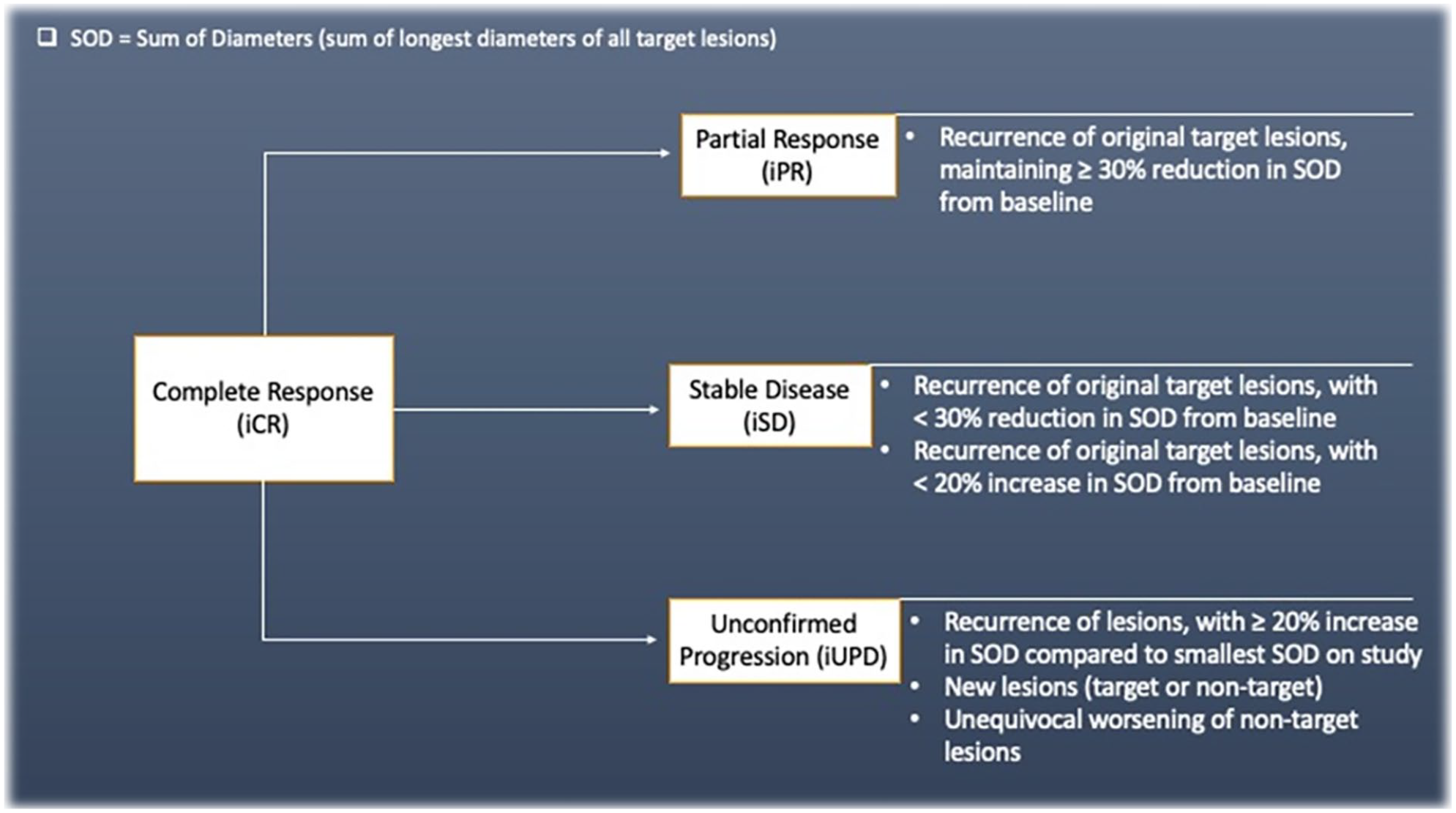
Displays the potential changes in status of a case following complete response (iCR), outlining the potential ways each outcome can occur. The status of iCR may also persist.
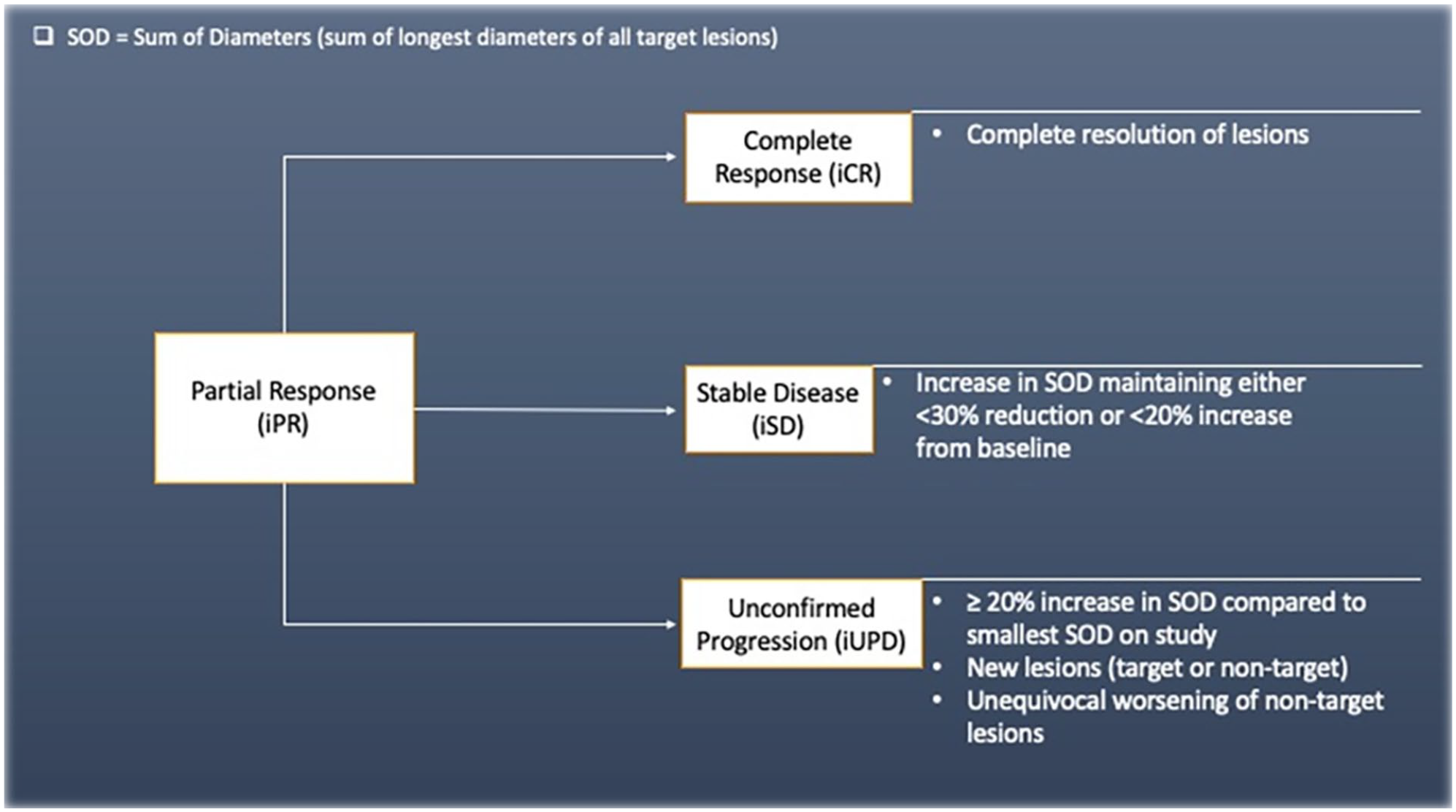
Displays the potential changes in status of a case following partial response (iPR), outlining the potential ways each outcome can occur. The status of iPR may also persist.
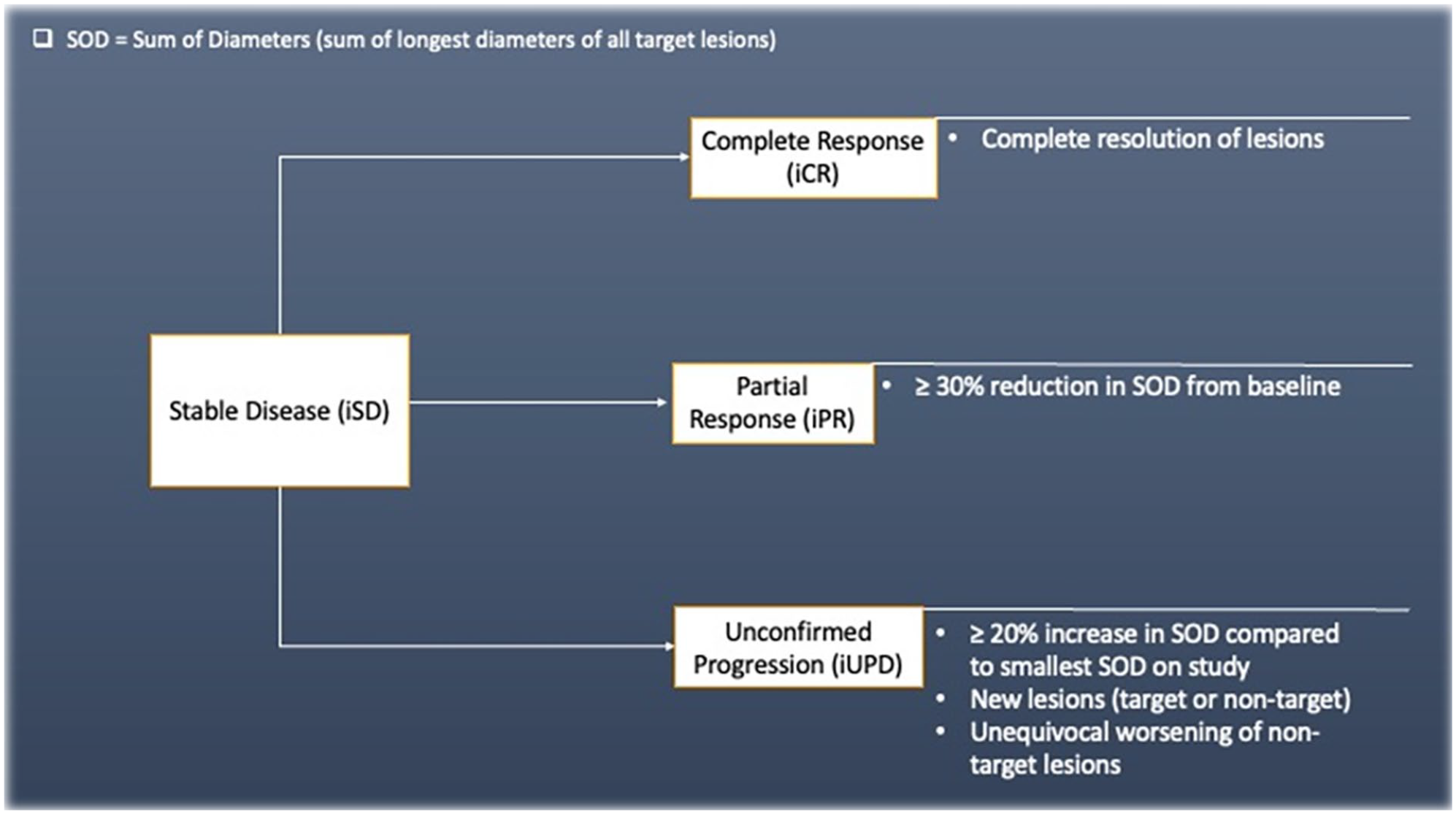
Displays the potential changes in status of a case following stable disease (iSD), outlining the potential ways each outcomes can occur. The status of iSD may also persist.
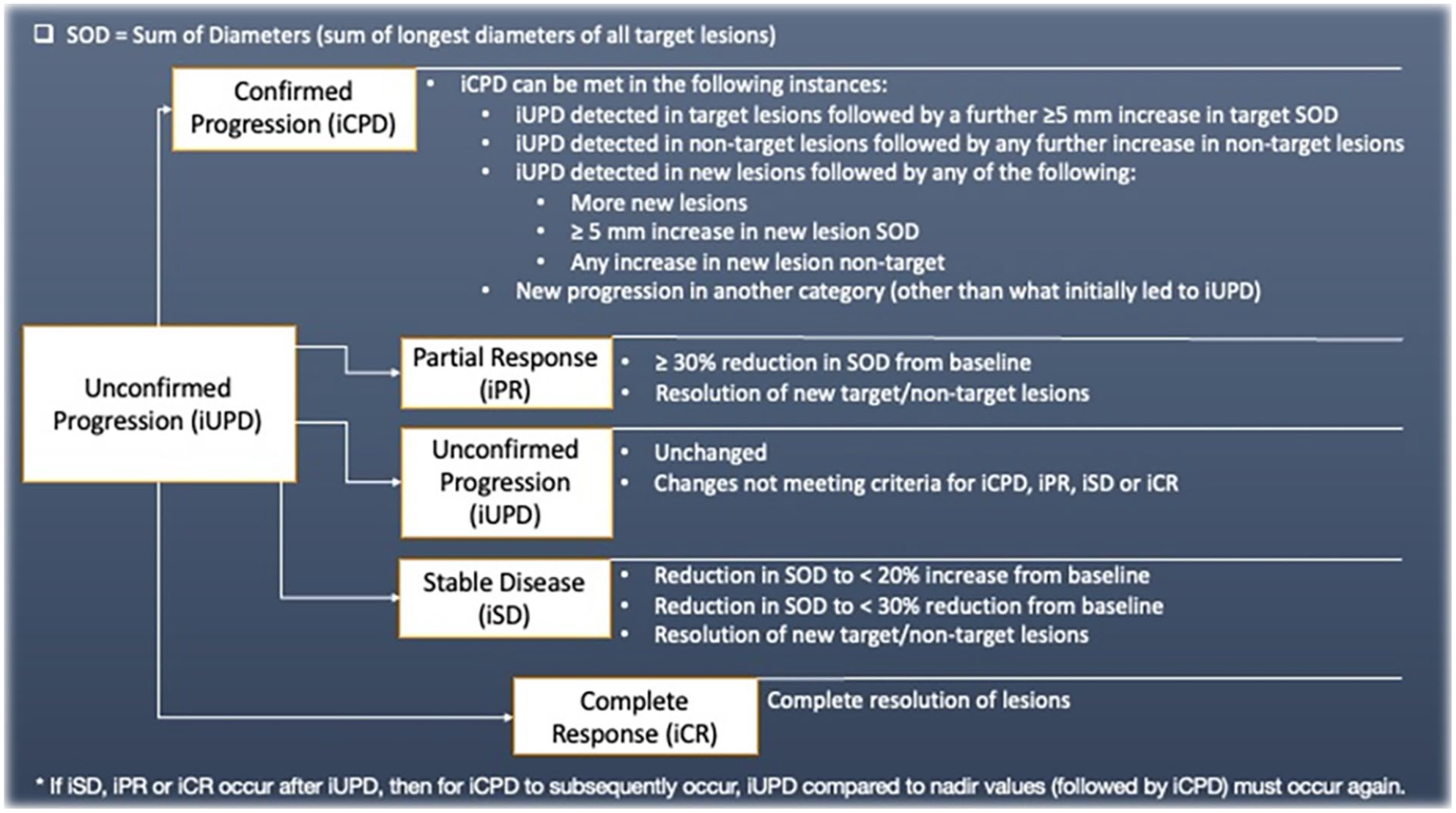
Displays the potential changes in status of a case following unconfirmed progression of disease (iUPD), outlining the potential ways each outcome can occur. If iPR, iSD, or iCR occur after iUPD, iRECIST introduces the concept of “re-setting the bar.” In this scenario iUPD needs to happen again and then be confirmed for iCPD to be deemed to have occurred.
iRECIST Scenarios
The use of iRECIST can be complex in practice, particularly following initial iUPD. There are multiple potential outcomes on follow up imaging after a designation of iUPD. The graphical representations provided below can be used as a tool for radiologists when reporting such cases and as a reference for clinicians when providing care for these patients (Figures 6-17). The iRECIST scenarios outlined here were chosen as they are either those which occur most commonly, or are those which are most challenging to deal with. It is important to note that not all potential scenarios are represented with graphical examples.
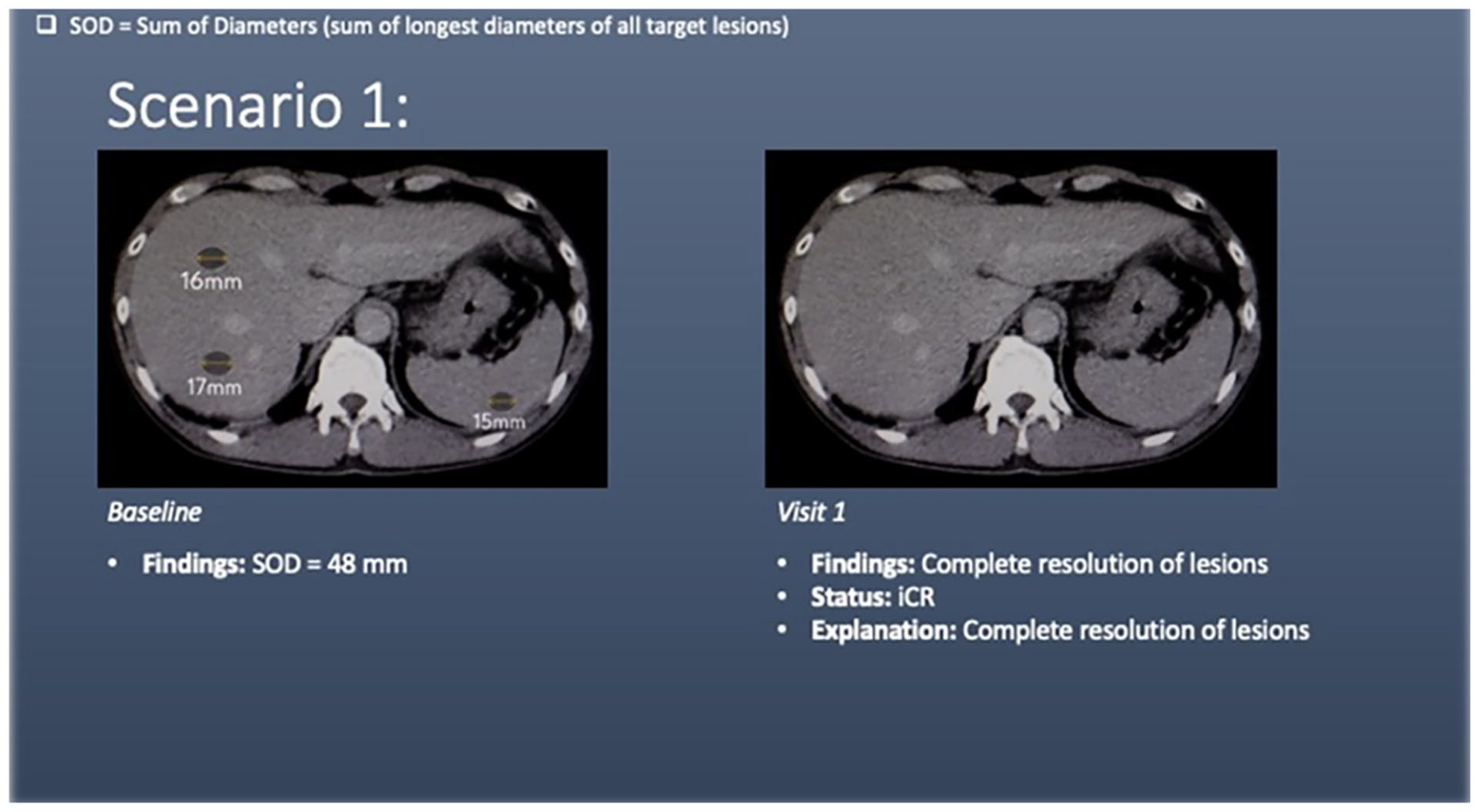
Scenario 1 shows liver and splenic lesions (target lesions) at baseline. At visit 1, there is complete resolution of the target lesions, meeting criteria for complete response (iCR).
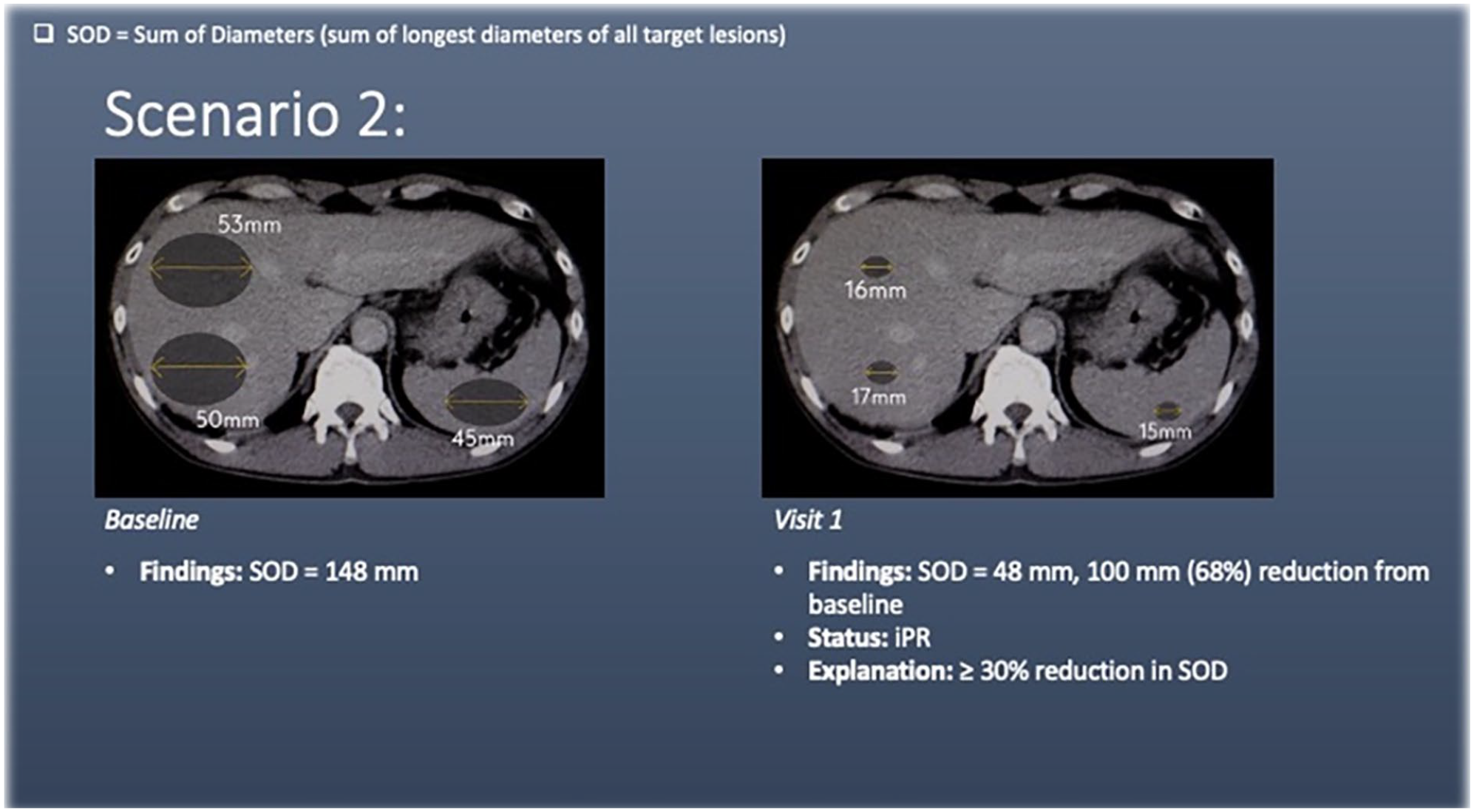
Scenario 2 shows liver and splenic lesions (target lesions) at baseline. At visit 1, there is a ≥30% reduction in the sum of diameters of the target lesions, meeting the criteria for partial response (iPR).
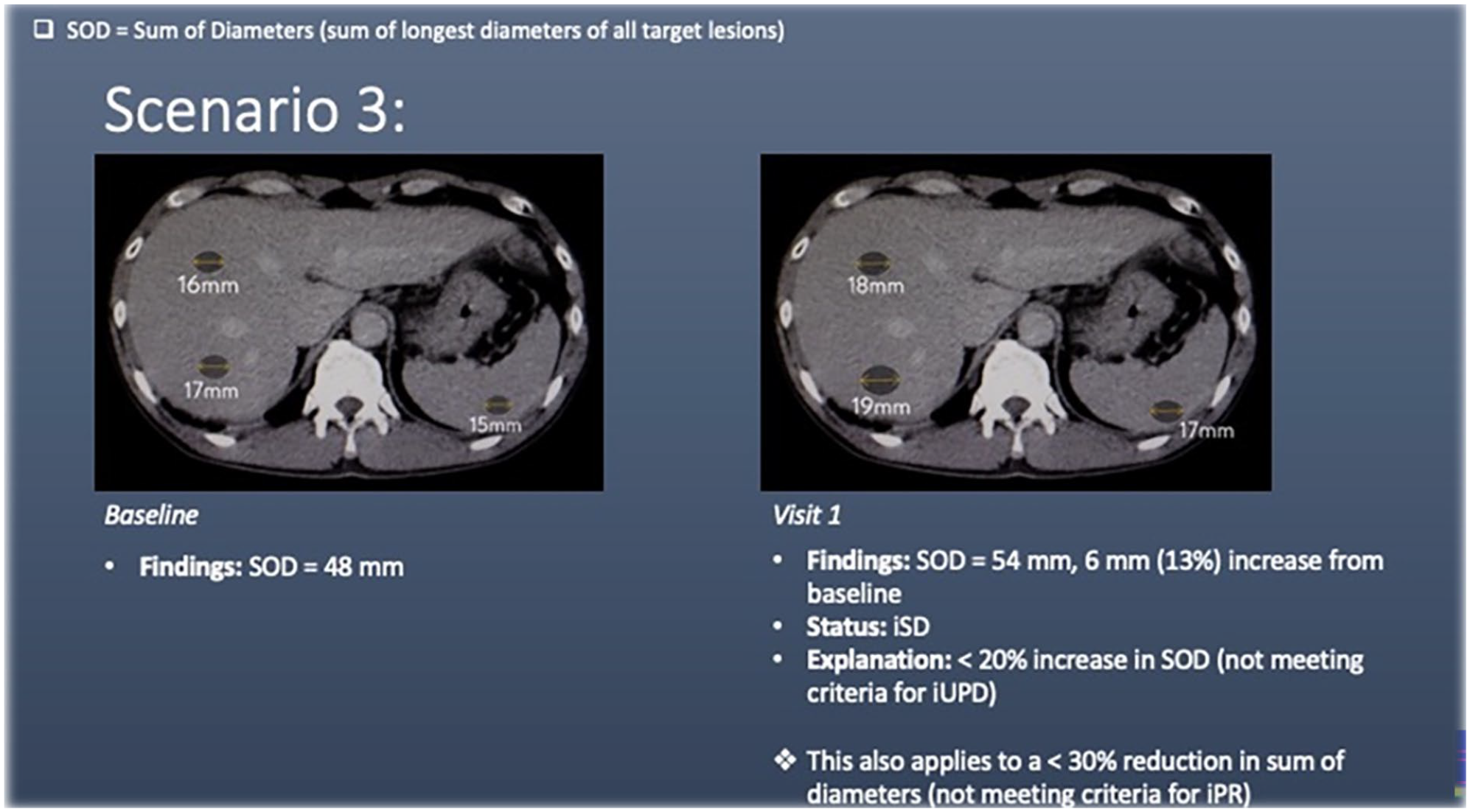
Scenario 3 shows liver and splenic lesions (target lesions) at baseline. At visit 1, there is <20% increase in the sum of diameters of the target lesions. This does not meet the criteria for disease progression and falls into the stable disease (iSD) category.
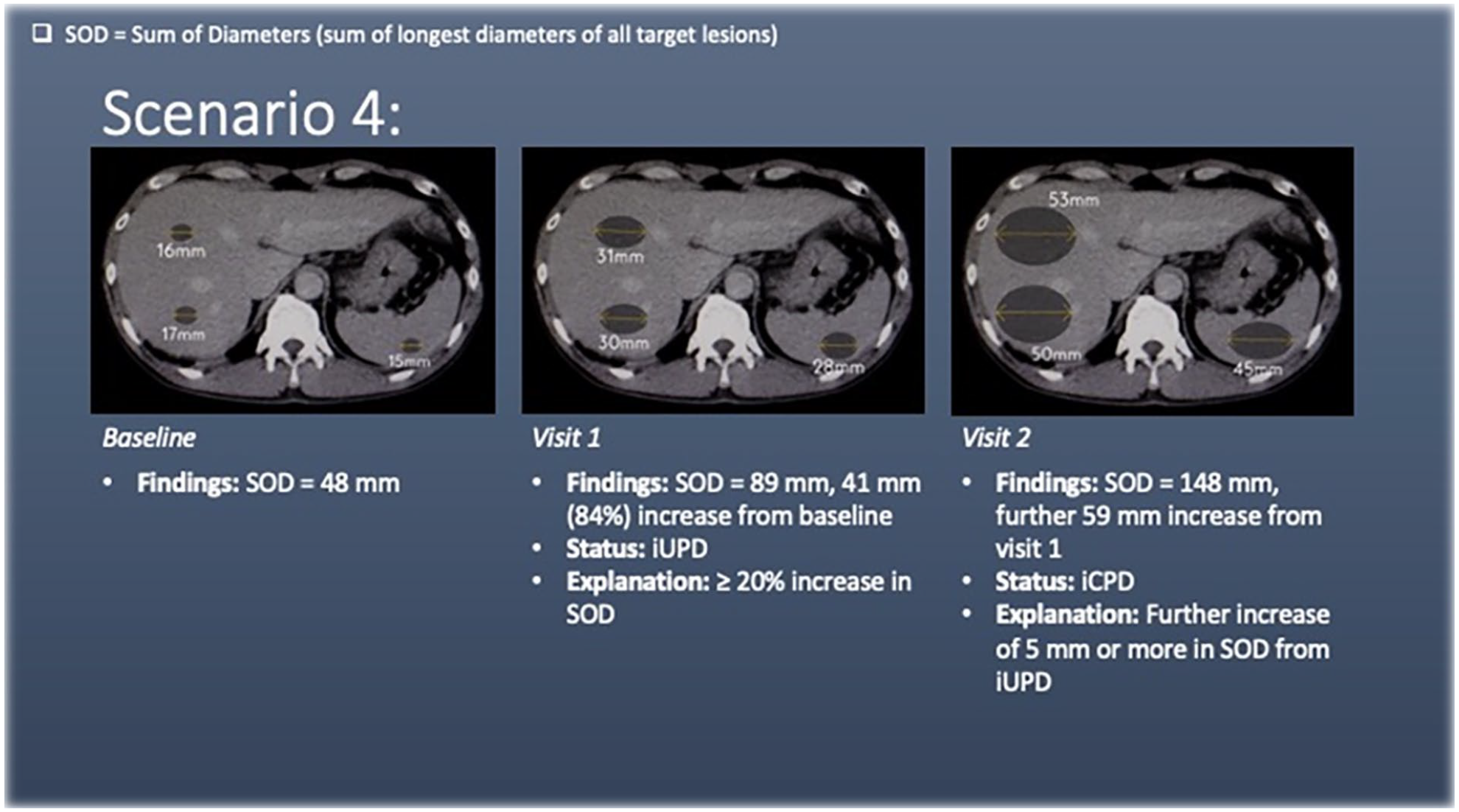
Scenario 4 has liver and splenic lesions (target lesions) at baseline. At visit 1, there is a ≥20% increase in the sum of diameters of the target lesions, meeting the criteria for unconfirmed progression of disease (iUPD). As with RECIST 1.1, iUPD is calculated with reference to the smallest SOD on study (in this case the baseline SOD).
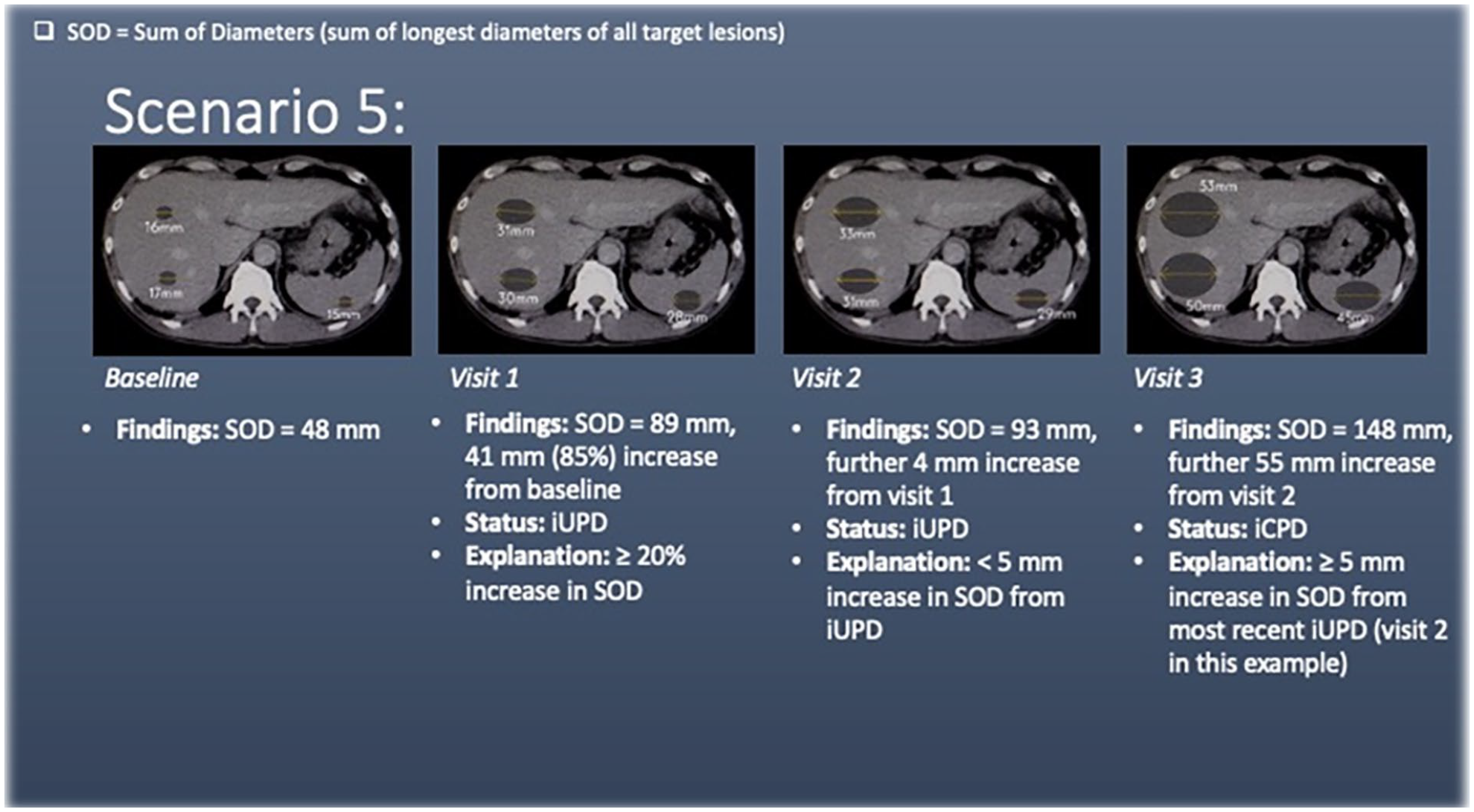
Scenario 5 has liver and splenic lesions (target lesions) at baseline. At visit 1, there is a ≥20% increase in the sum of diameters of the target lesions, meeting the criteria for unconfirmed progression of disease (iUPD).
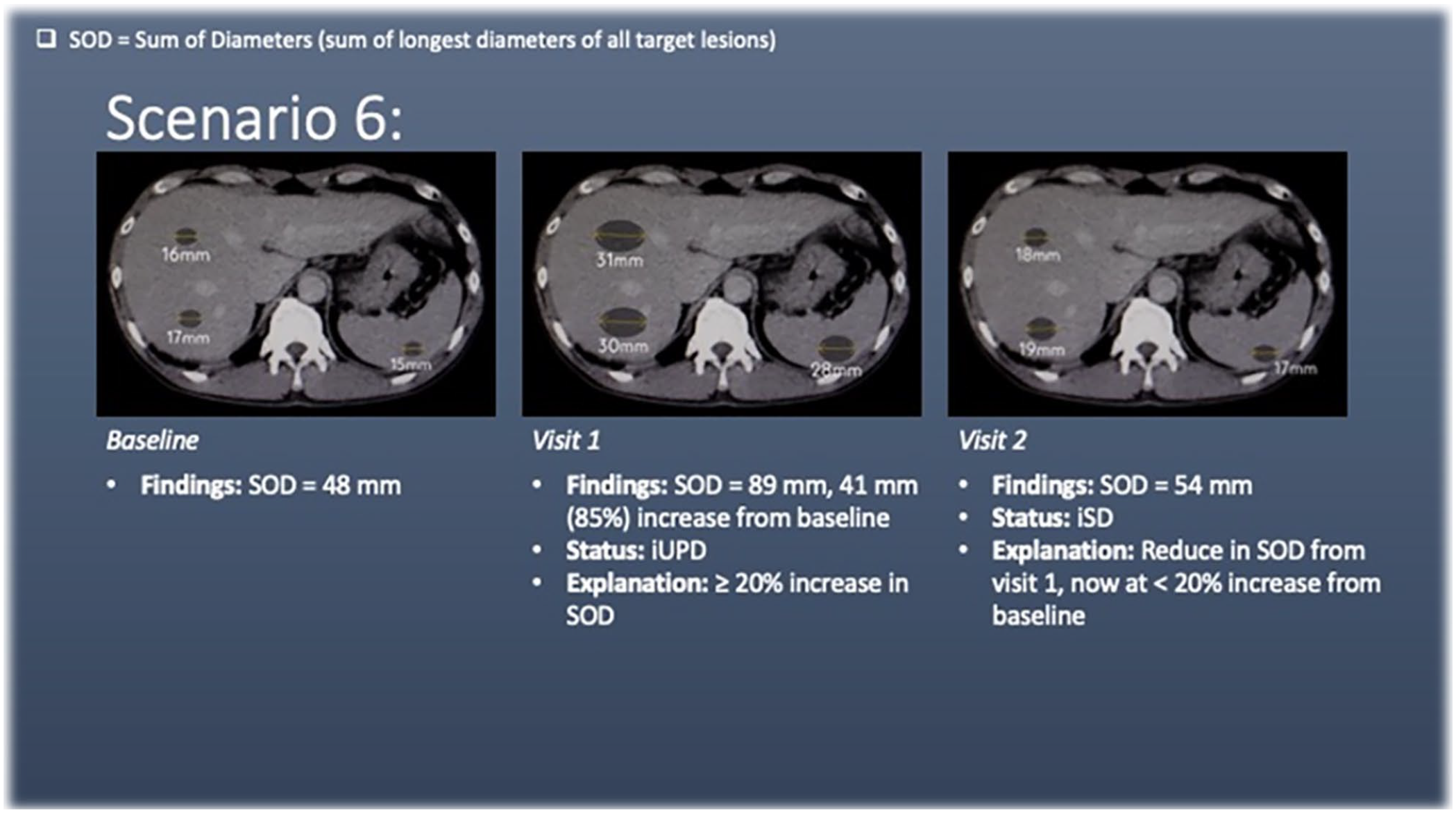
Scenario 6 has liver and splenic lesions (target lesions) at baseline. At visit 1, there is a ≥20% increase in the sum of diameters of the target lesions, meeting the criteria for unconfirmed progression of disease (iUPD).
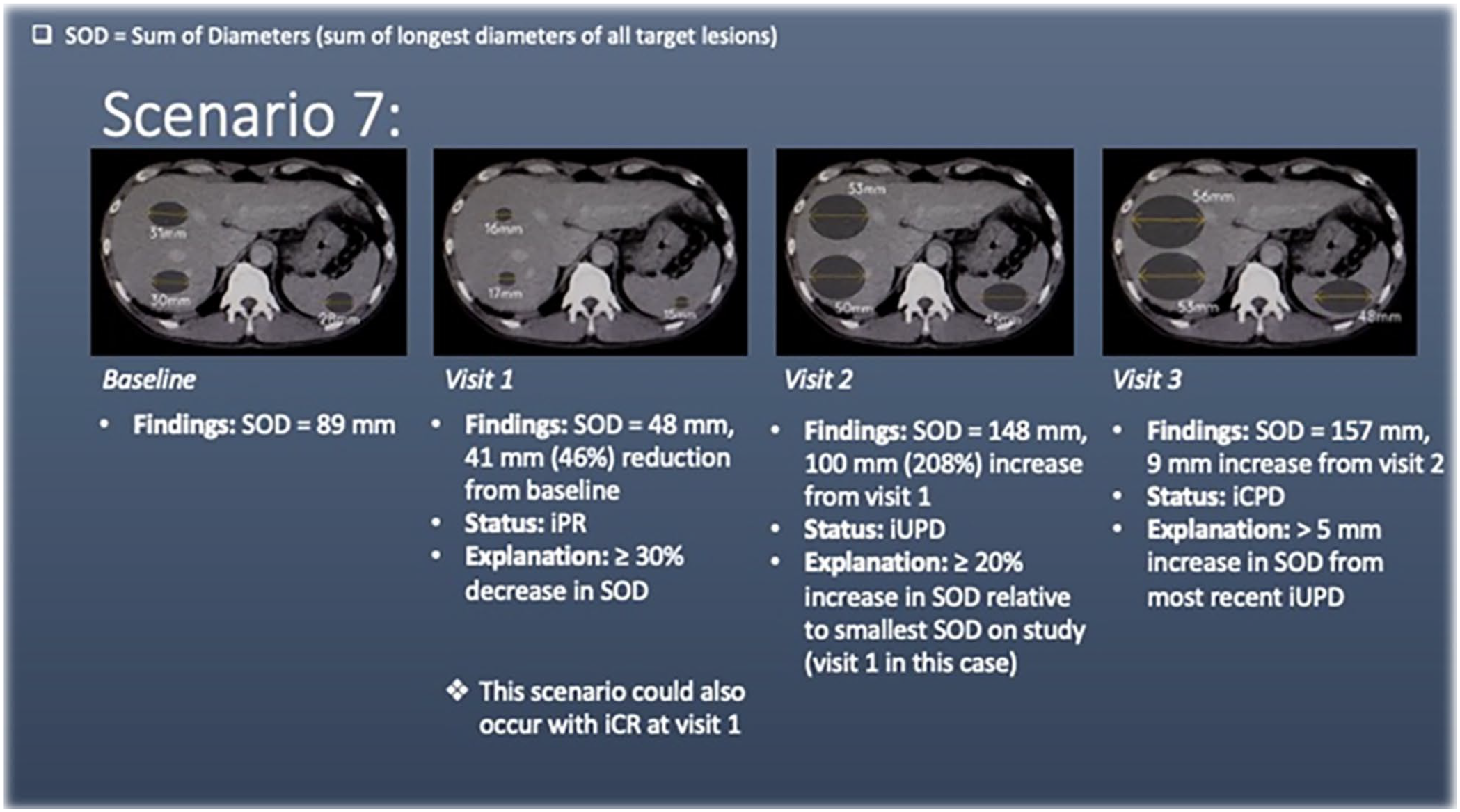
Scenario 7 has liver and splenic lesions (target lesions) at baseline. At visit 1, there is a ≥30% decrease in the sum of diameters of the target lesions, meeting the criteria for partial response (iPR).
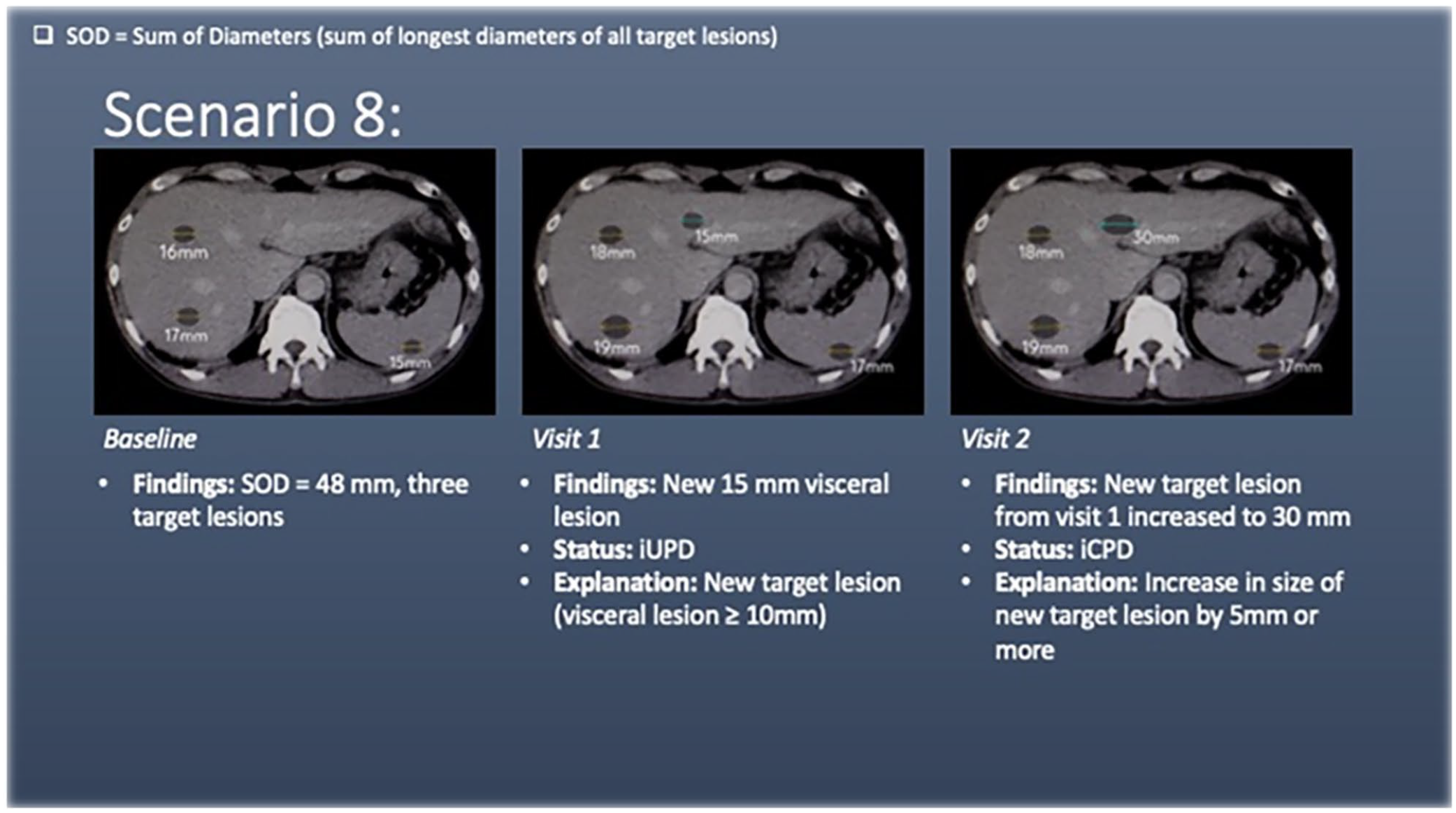
Scenario 8 has liver and splenic lesions (target lesions) at baseline. At visit 1, there is a new target lesion (liver) that measures ≥10 mm in diameter. This meets the criteria for unconfirmed progression of disease (iUPD).
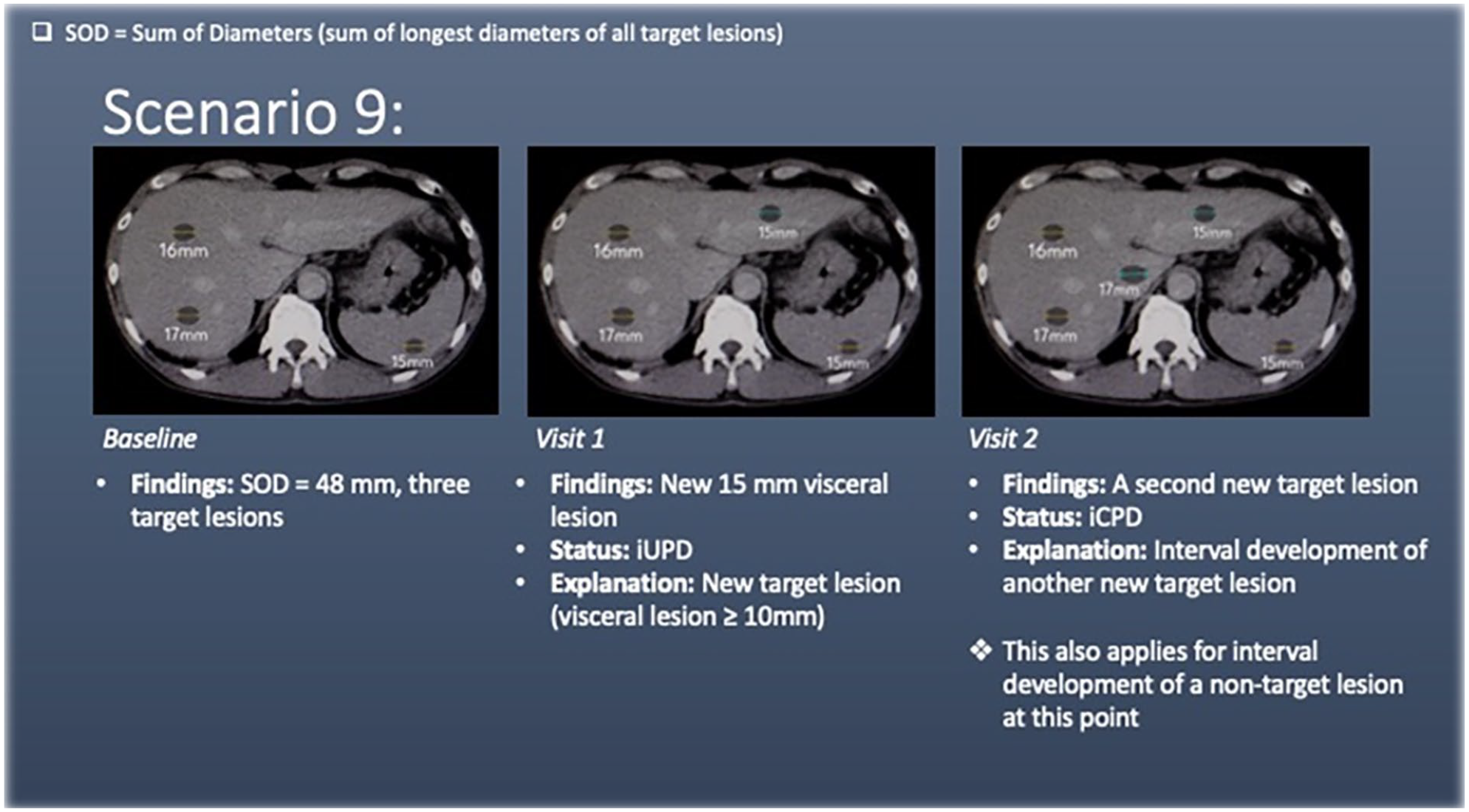
Scenario 9 has liver and splenic lesions (target lesions) at baseline. At visit 1, there is a new target lesion (liver) that measures ≥10 mm in diameter. This meets the criteria for unconfirmed progression of disease (iUPD).
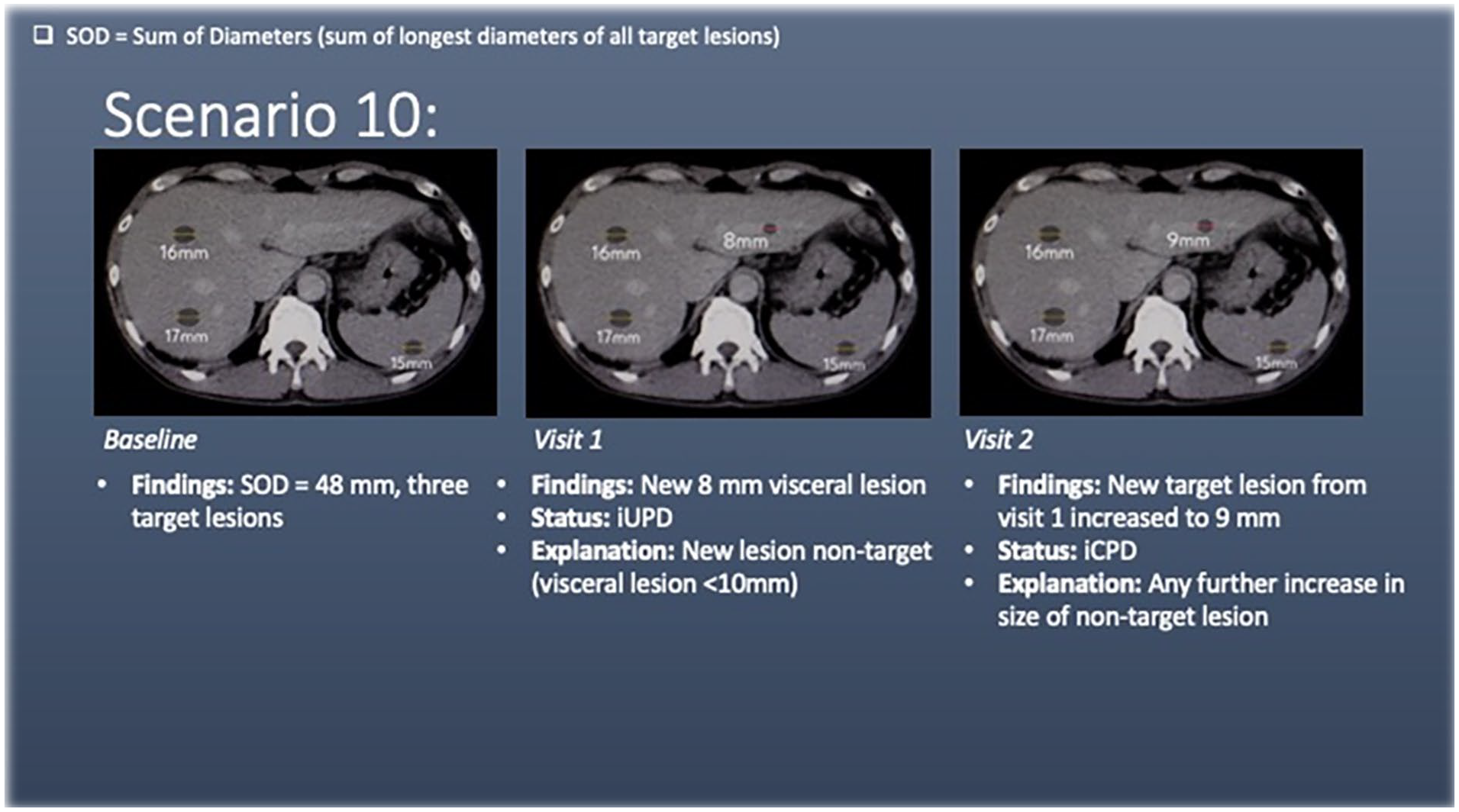
Scenario 10 has liver and splenic lesions (target lesions) at baseline. At visit 1, there is a new liver lesion. As this lesion measures <10 mm, it is not a target lesion and is termed “new lesion non-target.” This meets the criteria for unconfirmed progression of disease (iUPD).
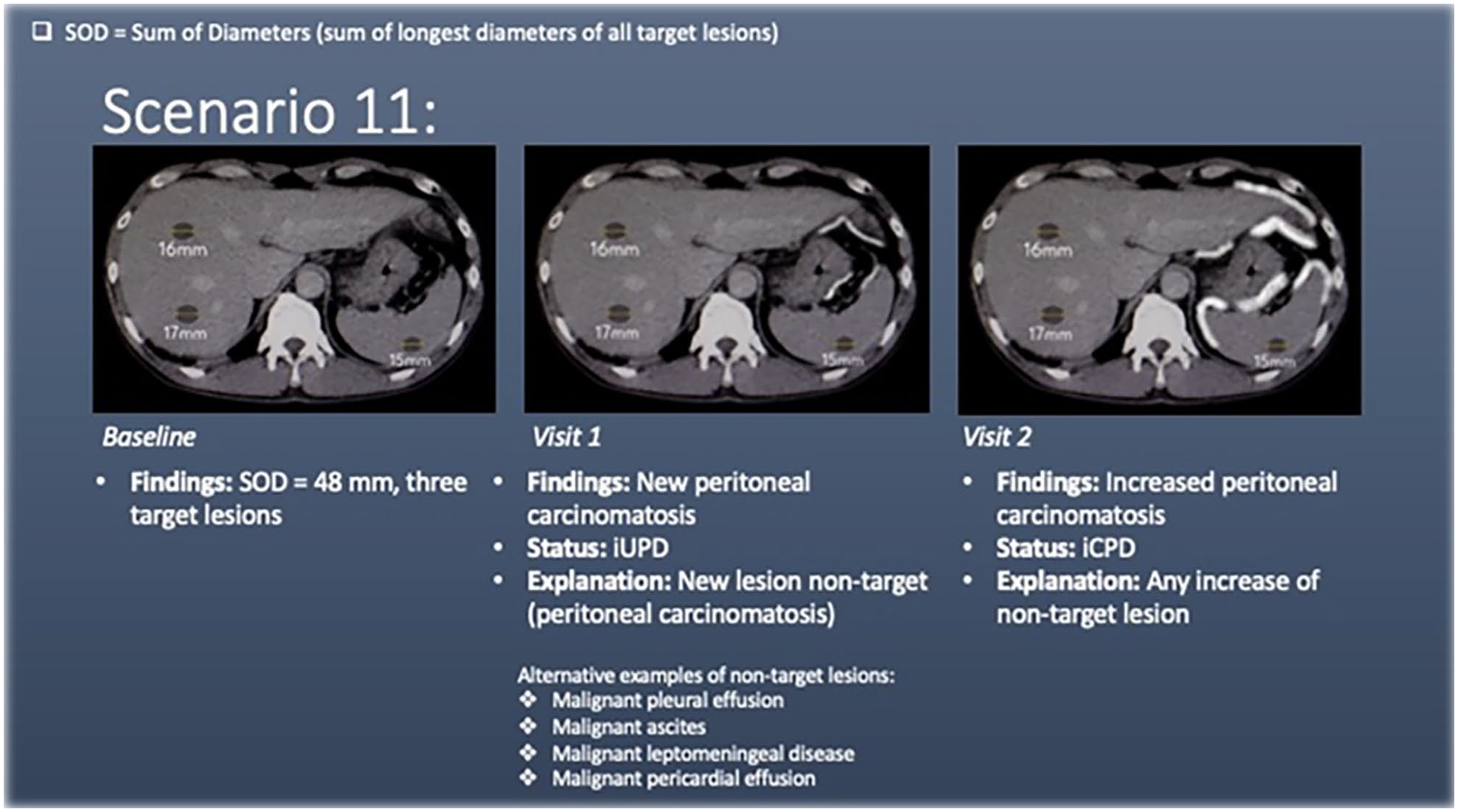
Scenario 11 has liver and splenic lesions (target lesions) at baseline. At visit 1, there is development of peritoneal carcinomatosis (new non-target lesion; denoted by white lines adjacent to the stomach spleen and liver), meeting the criteria for unconfirmed progression of disease (iUPD). Other examples of non-measurable non-target lesions include, but are not limited to, malignment pleural effusion, malignant ascites, malignant leptomeningeal disease, and malignant pericardial effusion.
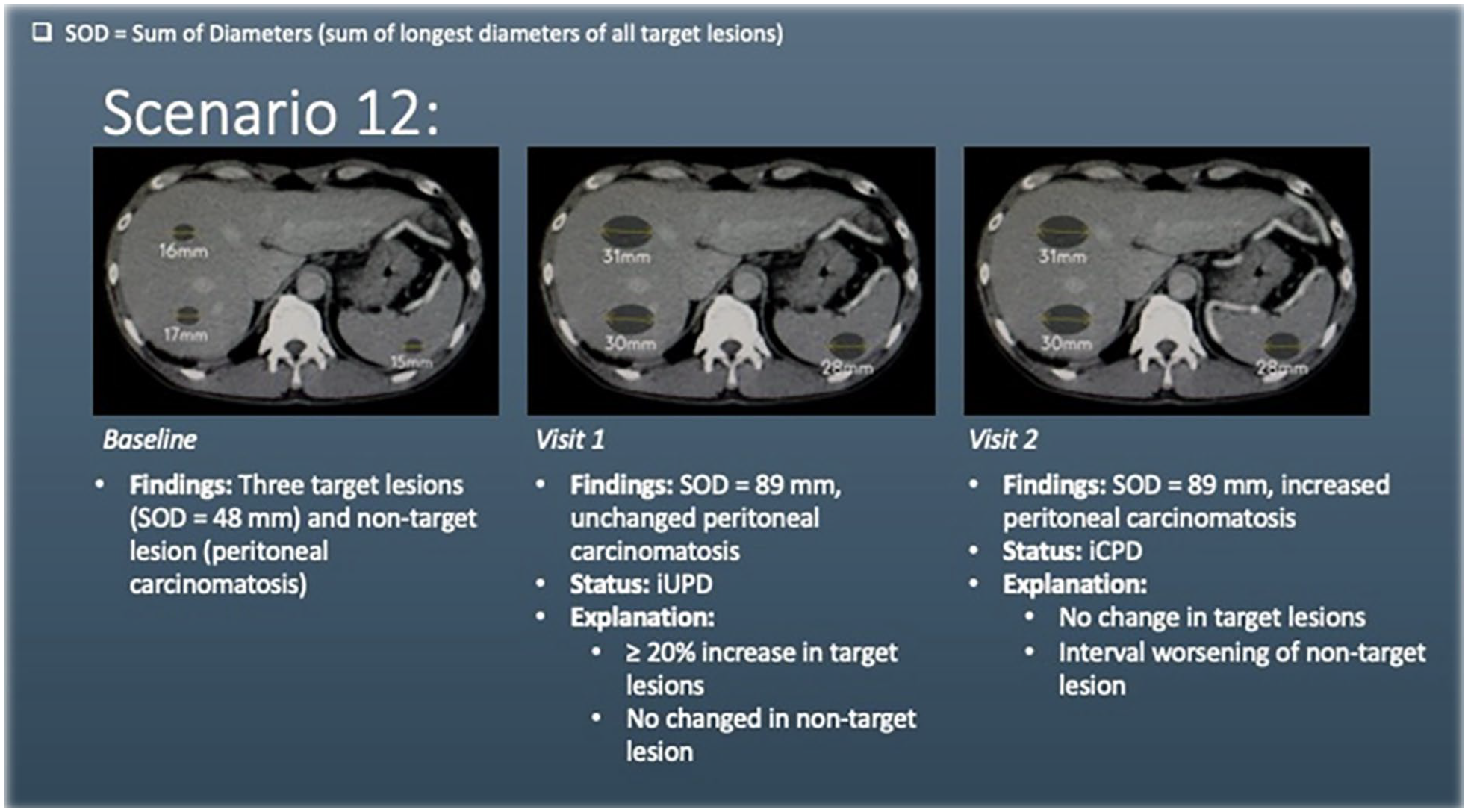
Scenario 12 has liver and splenic lesions (target lesions) and peritoneal carcinomatosis (non-target lesion) at baseline. At visit 1, there is ≥20% increase in the sum of diameters of the target lesions, with no increase in the peritoneal carcinomatosis. This meets the criteria for unconfirmed progression of disease (iUPD).
Thirteen common scenarios are provided. An anonymised image of an axial abdominal Computed Tomogram (CT) was used as the template for representing the scenarios. Target and non-target lesions of various sizes were digitally sketched onto the image. The images represent many of the concepts of iRECIST but they do not cover the multiple ways in which each concept can occur. The images in all scenarios are displayed on the same representative CT slice with the aim of maintaining simplicity and allowing direct comparison between scenarios. As a result, particular types of target and non-target lesions were used (ie, Liver/splenic lesions and peritoneal carcinomatosis). The scenarios displayed could also be represented in a number of other ways, using different types of target or non-target lesions.
Case Example: Background of Bladder Transition Cell Carcinoma
Visit 1: Baseline
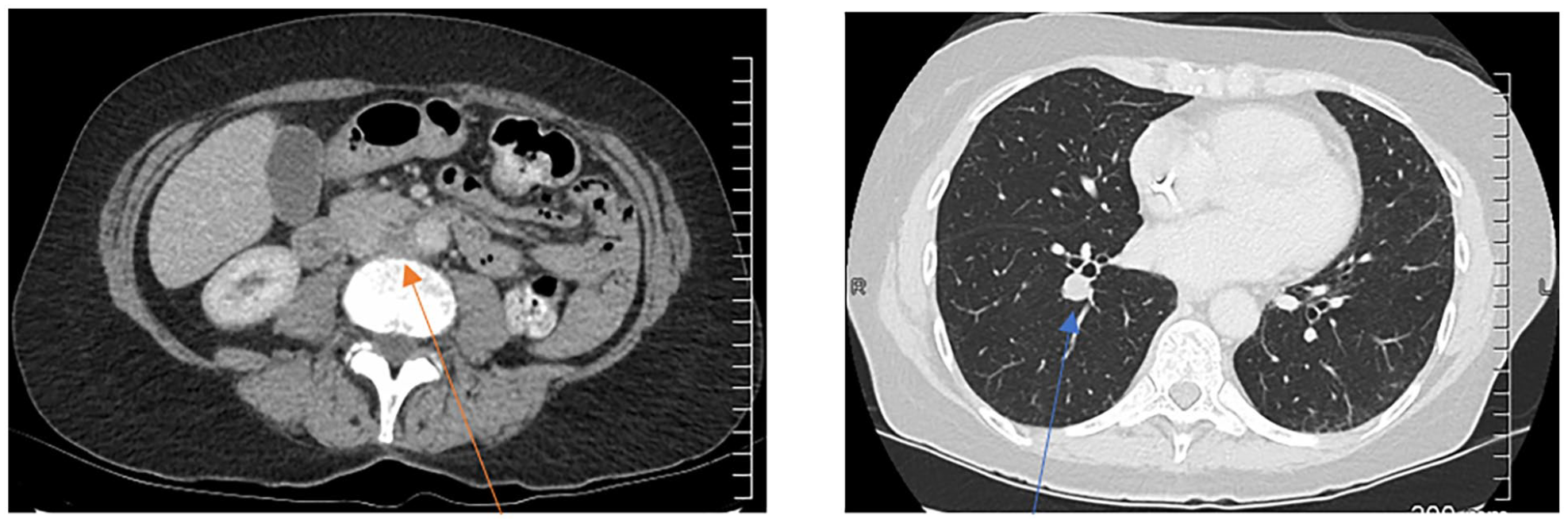
Aortocaval lymphadenopathy (orange arrow) and a right lung metastasis (blue arrow)
SOD = 24 mm
Visit 2
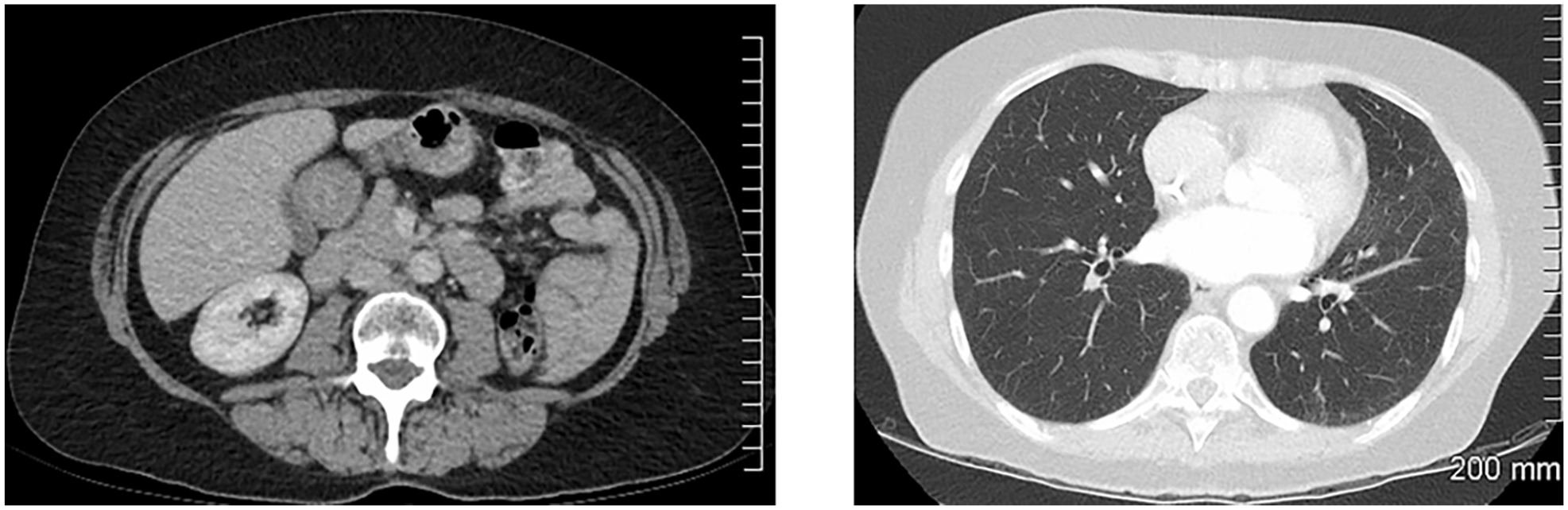
Resolution of the aortocaval lymphadenopathy and lung metastasis
SOD = 0
Status: iCR (complete resolution of lesions)
Case Example: Background of Bladder Transition Cell Carcinoma
Visit 1: Baseline
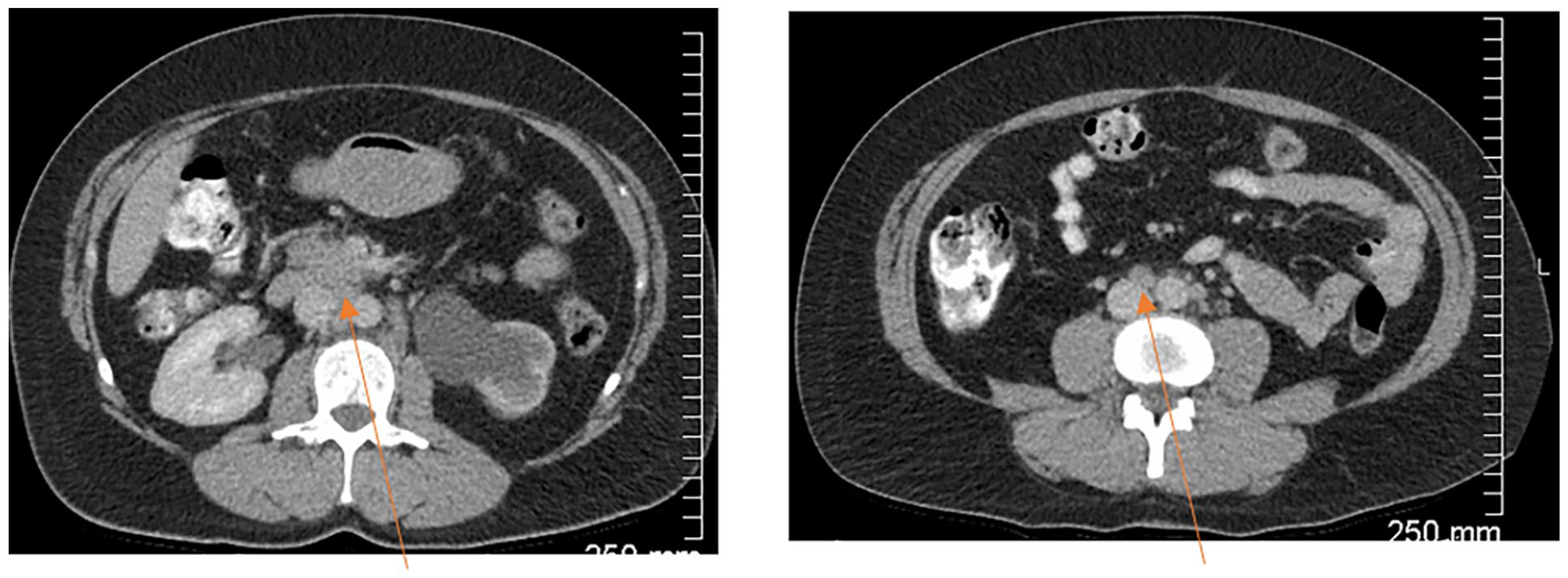
Retroperitoneal lymphadenopathy (orange arrows). Additional retroperitoneal lymphadenopathy not shown.
SOD = 340 mm
Visit 2
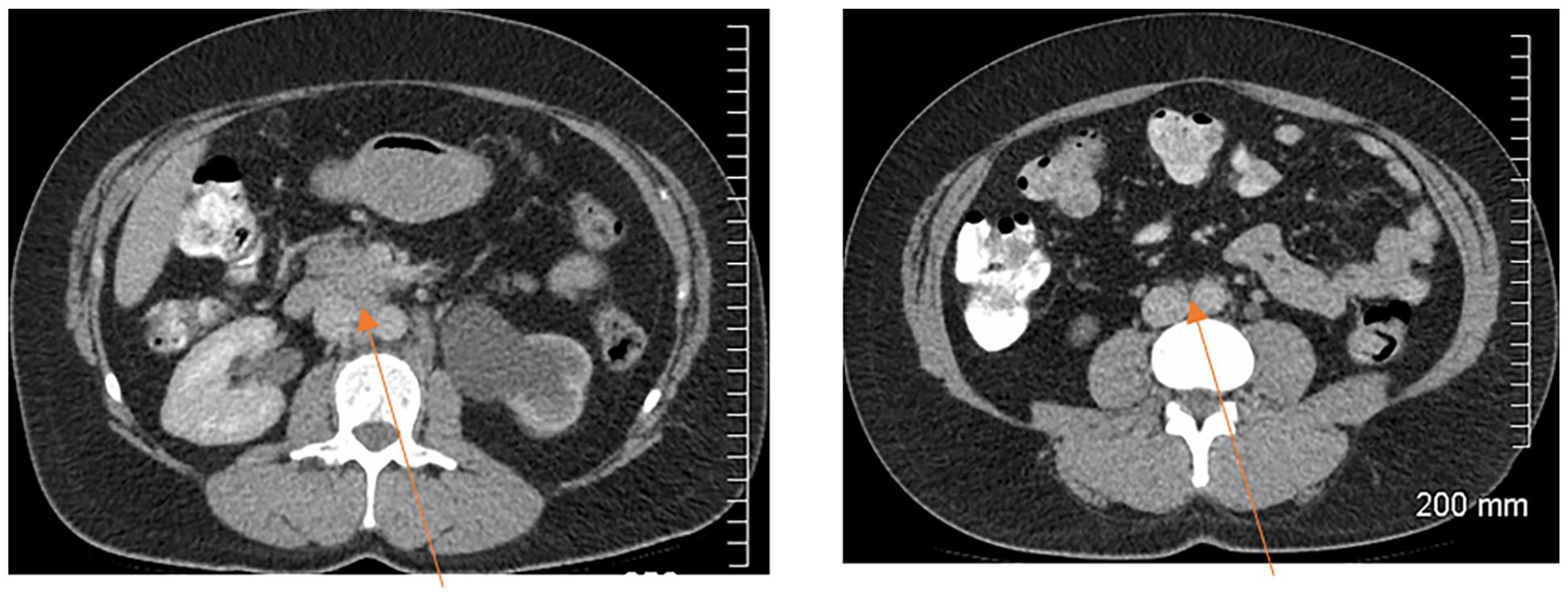
Reduced retroperitoneal lymphadenopathy (orange arrows)
SOD = 120 mm
Status: iPR (>30% reduction in SOD compared to baseline)
Visit 3
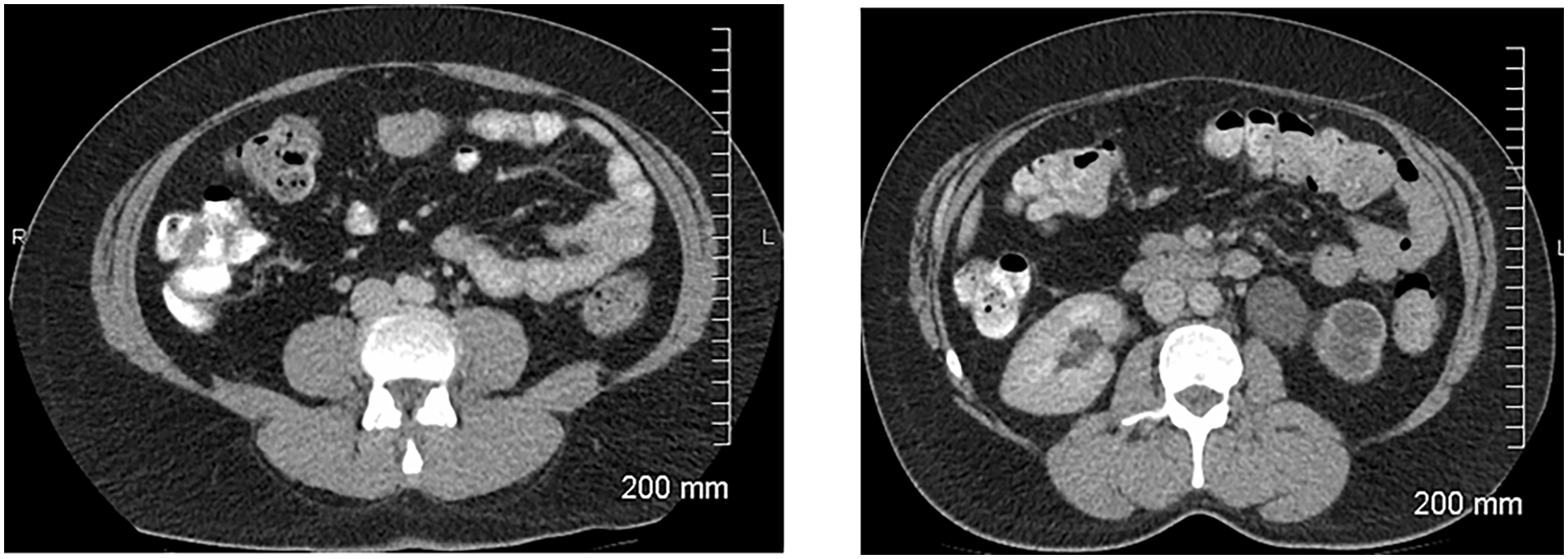
Resolved retroperitoneal lymphadenopathy
SOD = 0
Status: iCR (complete resolution of lymphadenopathy)
A similar scenario can occur when there is a reduction in the sum of diameters of the target lesions. If the reduction is <30%, this does not meet the criteria for partial response (iPR) and falls into the stable disease (iSD) category.
Case Example: Background of Small Cell Lung Carcinoma. Selected Images Showing a Perihilar Mass With Right Hilar Lymphadenopathy (Partially Shown) and a Small Right Pleural Effusion
Visit 1: Baseline
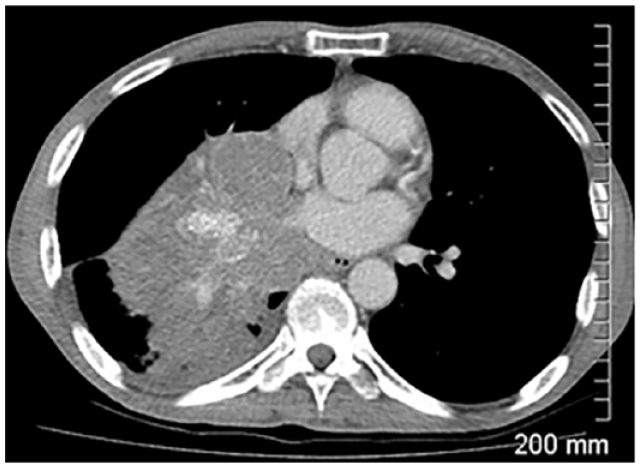
SOD = 121 mm
Visit 2
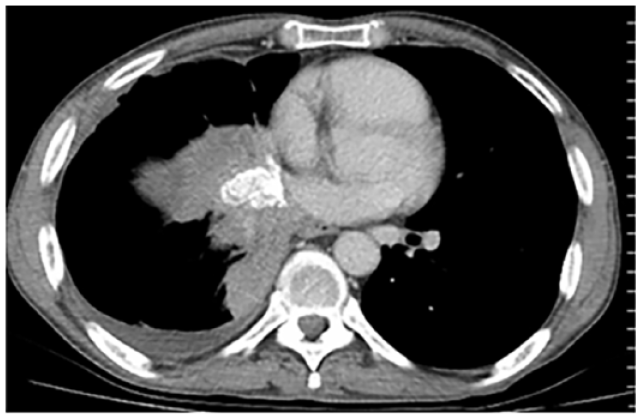
SOD = 99 mm
Status: Stable disease
<30% reduction in SOD from baseline
Visit 3
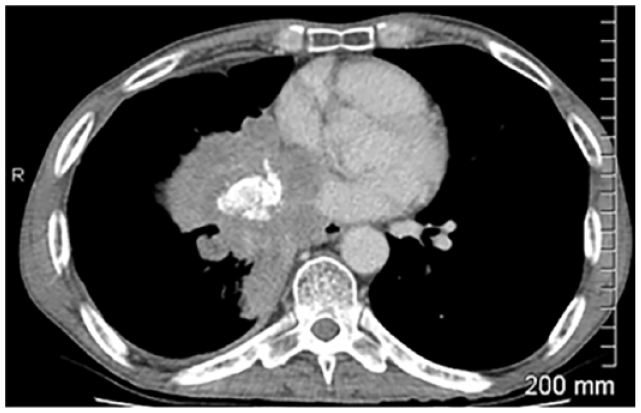
SOD = 108 mm.
Status: Stable disease.
<20% increase in SOD from baseline.
At visit 2, there is a further increase of 5 mm in the sum of diameters of the target lesions, meeting the criteria for confirmed progression of disease (iCPD).
Case Example: Background of Renal Cell Carcinoma
Visit 1: Baseline
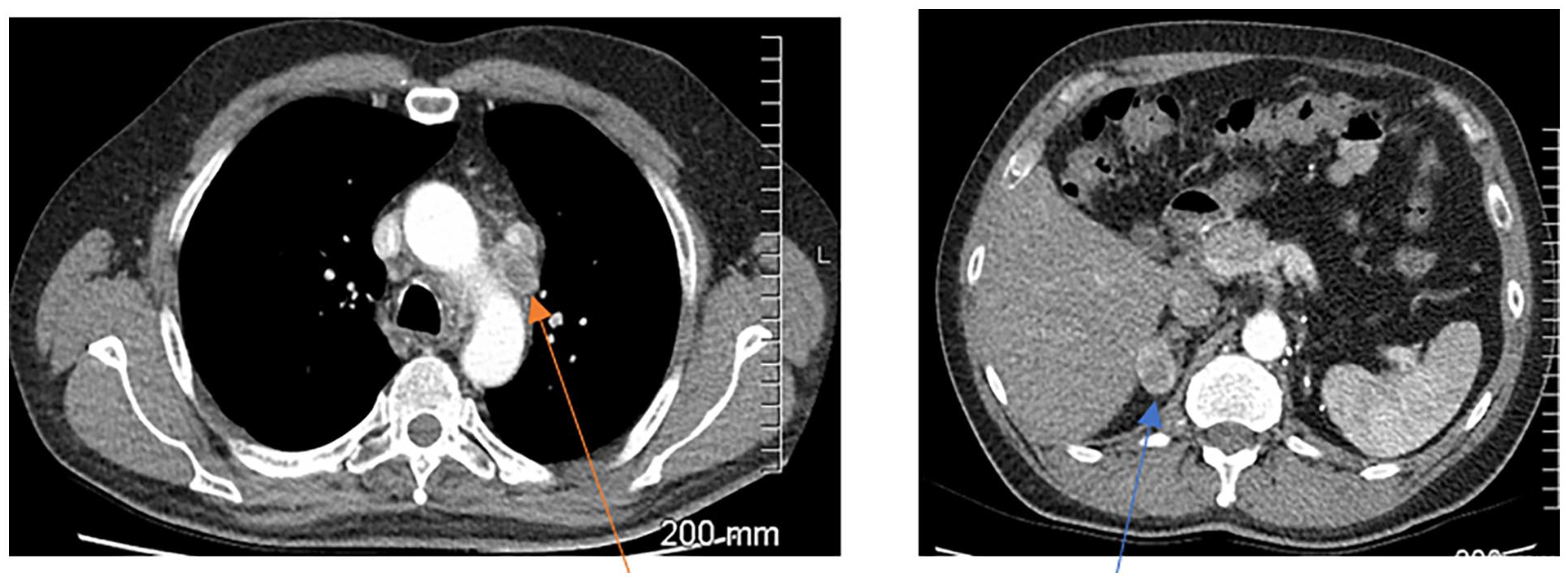
Mediastinal lymphadenopathy (orange arrow) and a right adrenal metastasis (blue arrow)
SOD = 151 mm
Visit 2
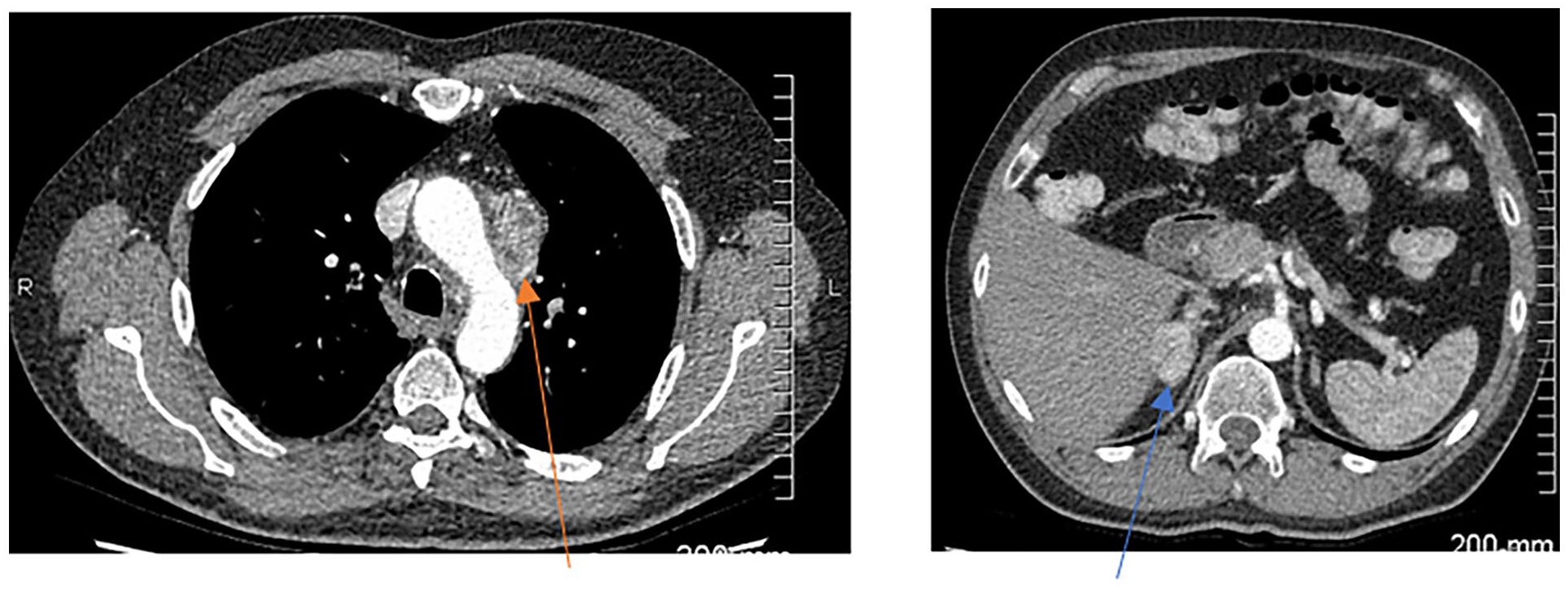
Increased mediastinal lymphadenopathy (orange arrow) and right adrenal metastasis (blue arrow)
SOD = 184 mm
Status: iUPD (>20% increase in SOD)
Visit 3
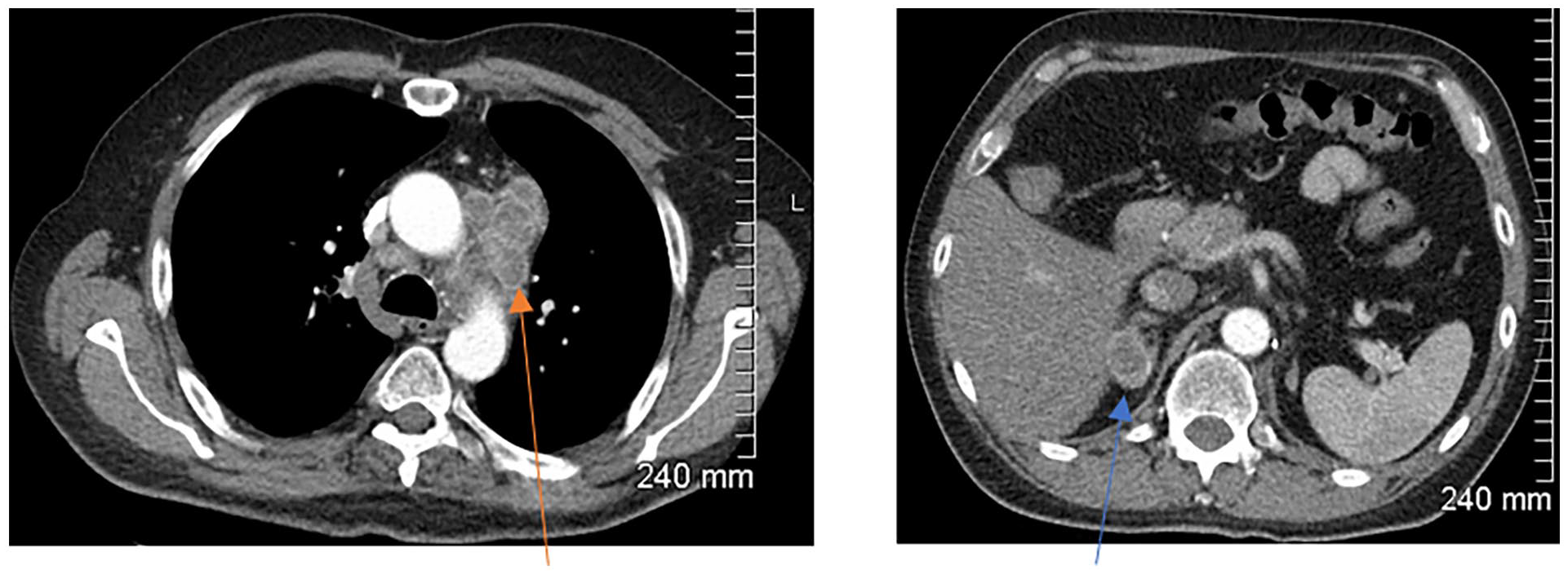
Further increased mediastinal lymphadenopathy (orange arrow). Stable size of the right adrenal metastasis (blue arrow) with interval development of necrosis.
SOD = 200 mm
Status: iCPD (further increase of >5 mm in SOD)
At visit 2, there is <5 mm increase in the sum of diameters of the target lesions. This does not meet the criteria for confirmed progression of disease (iCPD) and remains iUPD.
At visit 3, there is ≥5 mm increase in the sum of diameters of the target lesions from the most recent study (visit 2), meeting the criteria for confirmed progression of disease (iCPD).
At visit 2, there is a reduction in SOD compared to visit 1, but the SOD remains <20% increase compared to baseline. Progression of disease has not been confirmed. Compared to baseline, the current SOD does not meet the criteria for iPR and therefore falls into the stable disease category (iSD). In this scenario, as discussed above, iUPD would need to occur again and then be confirmed for iCPD to be deemed to have occurred.
At visit 2, there is a ≥20% increase in the sum of diameters of the target lesions compared to the smallest SOD on study, meeting the criteria for unconfirmed progression of disease (iUPD).
At visit 3, there is a ≥5 mm increase in the sum of diameters of the target lesions, meeting the criteria for confirmed progression of disease (iCPD).
This case highlights that patients can go from partial response (iPR) directly to unconfirmed progression of disease (iUPD).
At visit 2, this new target lesion increases in diameter by a further 5 mm, meeting the criteria for confirmed progression of disease (iCPD).
Case Example: Background of Bladder Transition Cell Carcinoma
Visit 1: Baseline
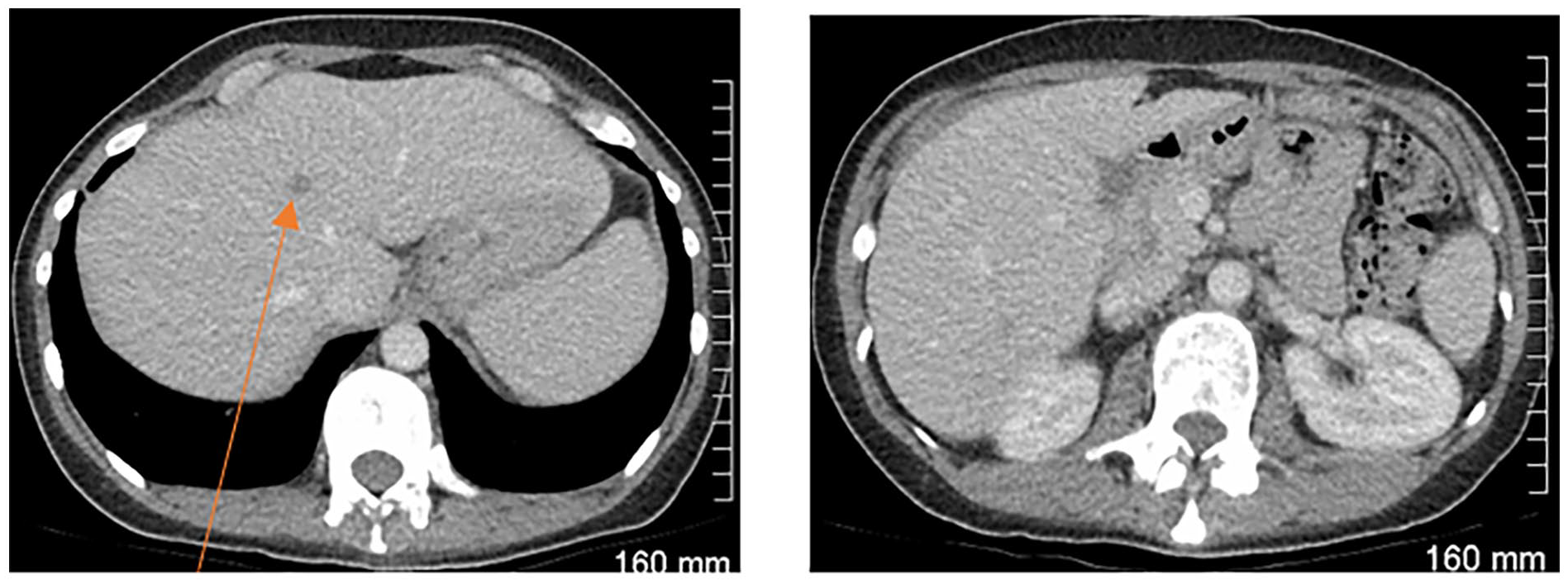
Single liver metastasis in segment 8 (orange arrow) of the liver. Second image contains no lesions (for comparison of subsequent visits)
SOD = 10 mm
Visit 2
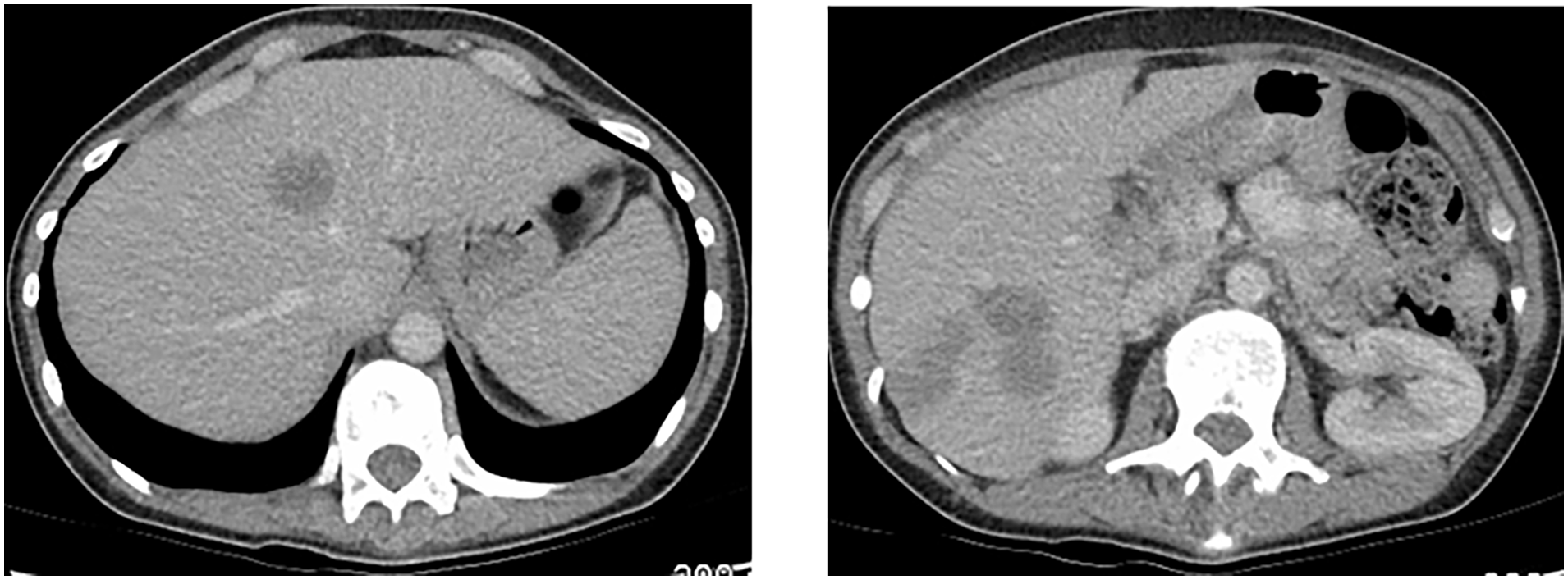
Increased metastasis in segment 8/4a of the liver. New lesions in segment 6.
SOD = 80 mm. New liver lesions
Status: iUPD (new visceral lesions >10 mm). The pre-existing liver lesion has also increased in size.
Visit 3
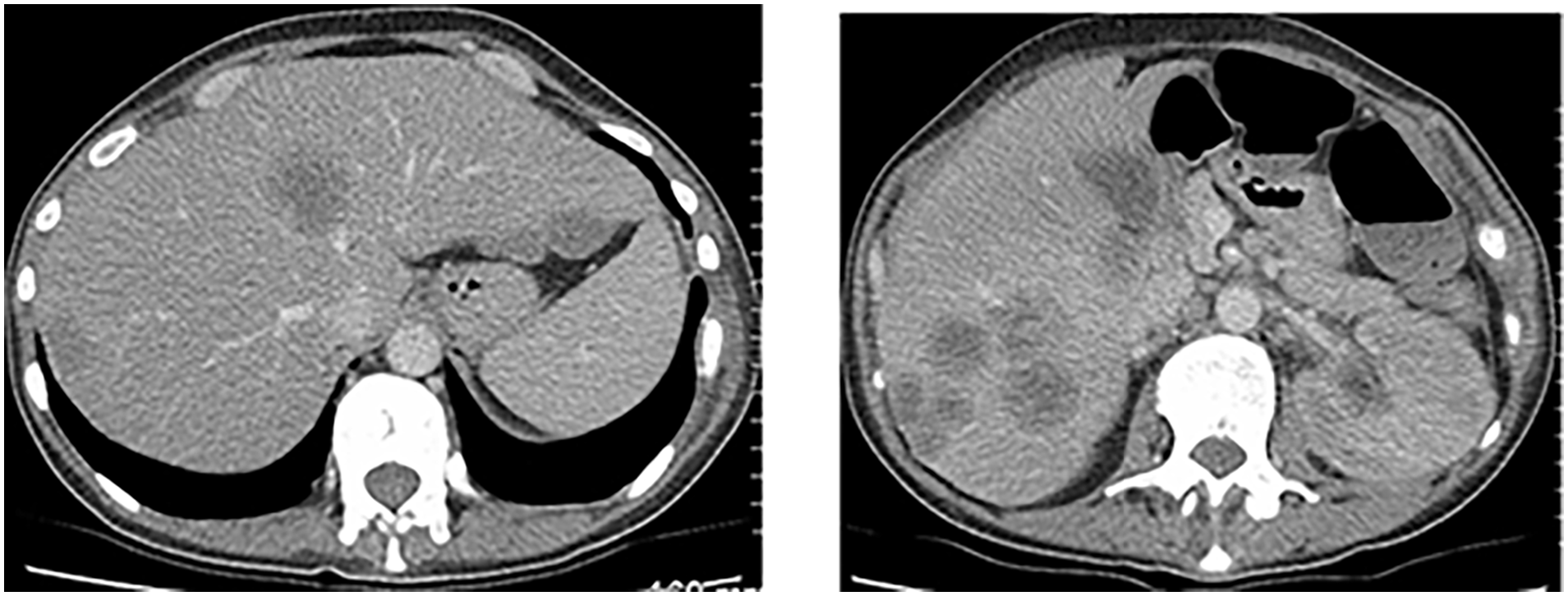
Further increased lesion in segment 8/4a and increased lesions in segment 6.
SOD = 120 mm
Status: iCPD (increase in size of new target lesions by more than 5 mm)
At visit 2, there is development of a new target lesion (≥10 mm) in the liver. This meets the criteria for confirmed progression of disease (iCPD). It is worth noting that development of a non-target lesion at this point would also result in iCPD.
Case Example: Background of Bladder Transition Cell Carcinoma. Images Show New and Evolving Liver Metastases. The Patient Also Had Pelvic Lymphadenopathy (Not Shown)
Visit 1: Baseline
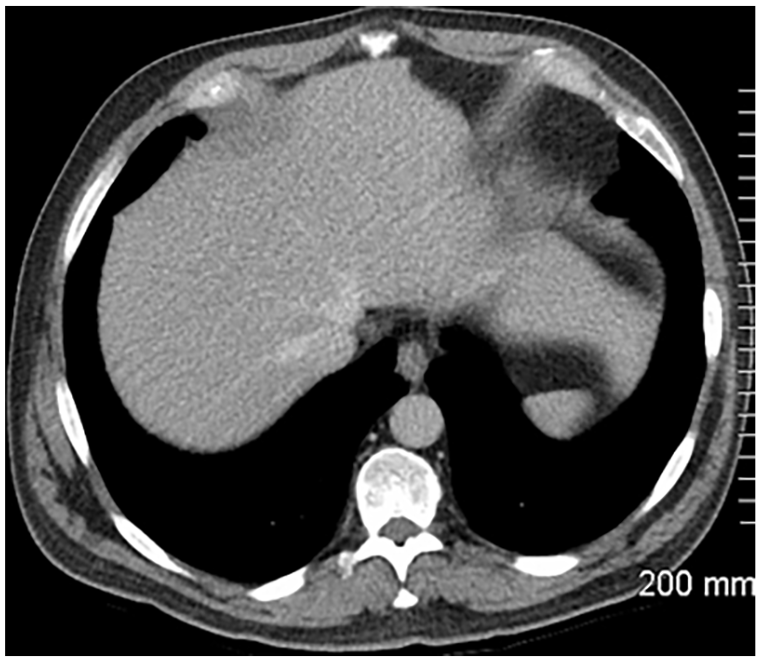
No liver lesions
Visit 2
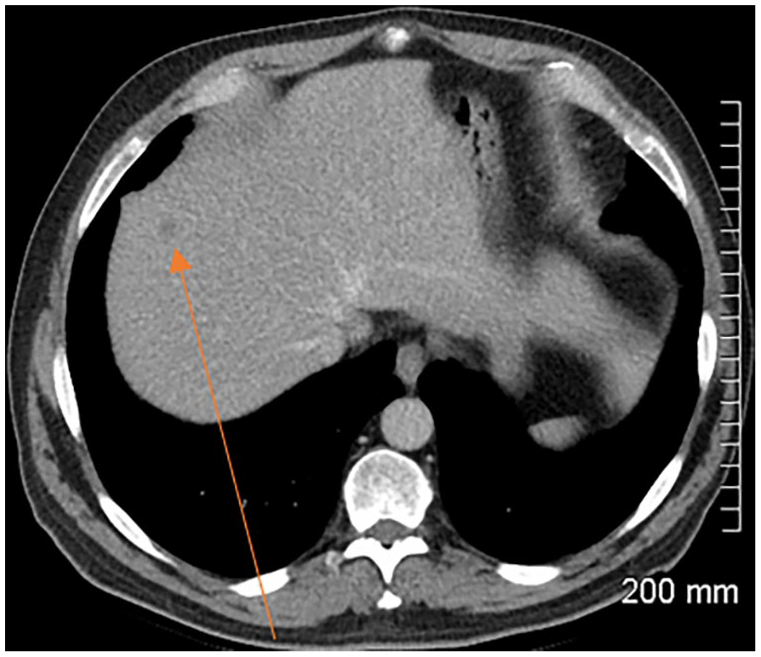
New 10 mm liver lesion (orange arrow).
Status: iUPD (new target lesion)
Visit 3
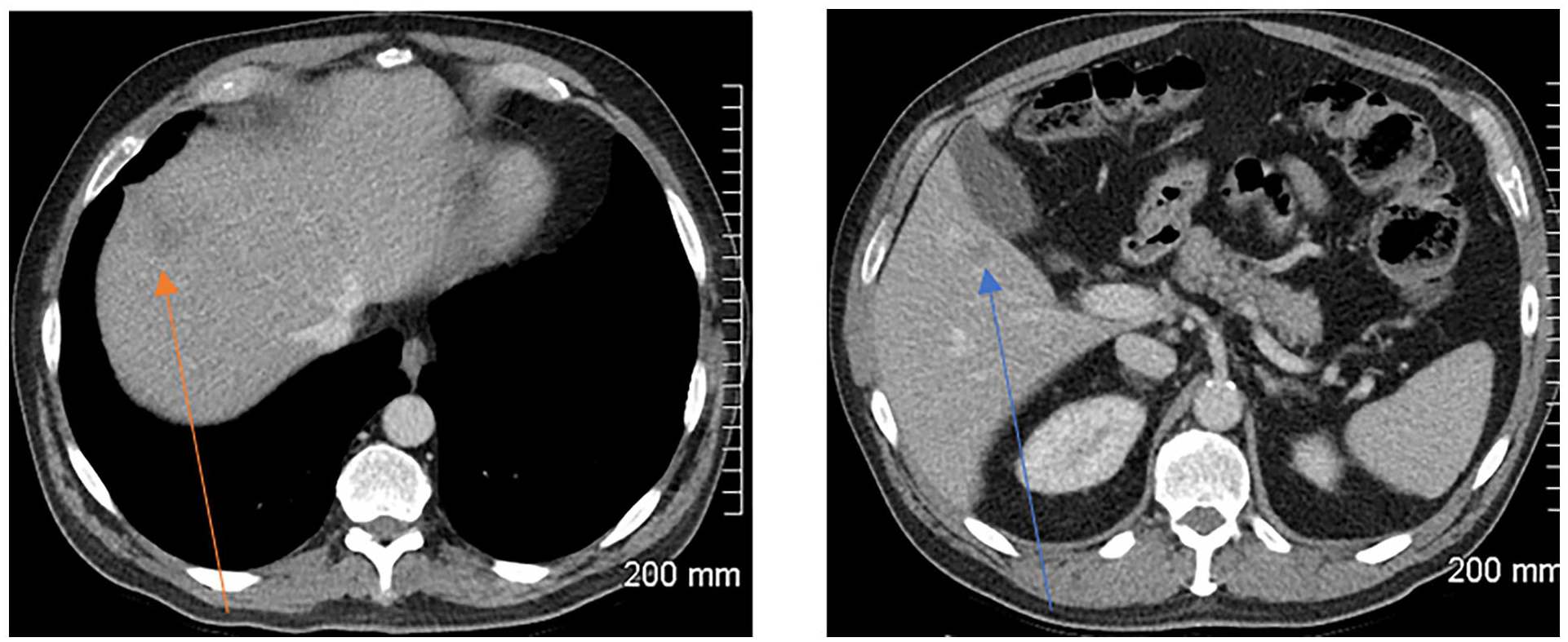
Development of another target liver lesion (blue arrow), confirming progression of disease (iCPD). The original liver lesion also increased by more than 5 mm (orange arrow), another factor that confirms progression.
Development of a non-target lesion at visit 3 would have also have confirmed progression of disease.
At visit 2, the non-target lesion increases further in size, meeting the criteria form confirmed progression of disease (iCPD). It is worth noting that any further increase in size of new non-target lesions results in confirmed progression of disease (iCPD).
At visit 2, there is increase in the amount of peritoneal carcinomatosis, meeting the criteria for confirmed progression of disease (iCPD). It is worth noting that any increase of a non-target lesion at this point would result in iCPD.
Case Example: Background of Renal Cell Carcinoma
Visit 1
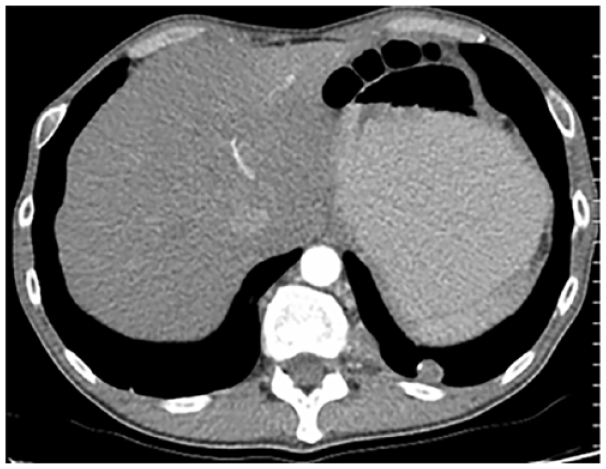
Lung metastasis in the left lower lobe. Additional pulmonary metastases not shown.
Visit 2
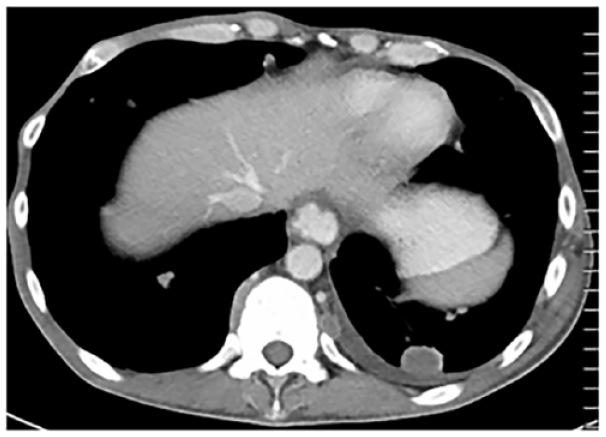
Development of a small left pleural effusion with a parietal pleural nodule, consistent with a malignant effusion. This is a new non-target lesion.
Status: iUPD
Visit 3
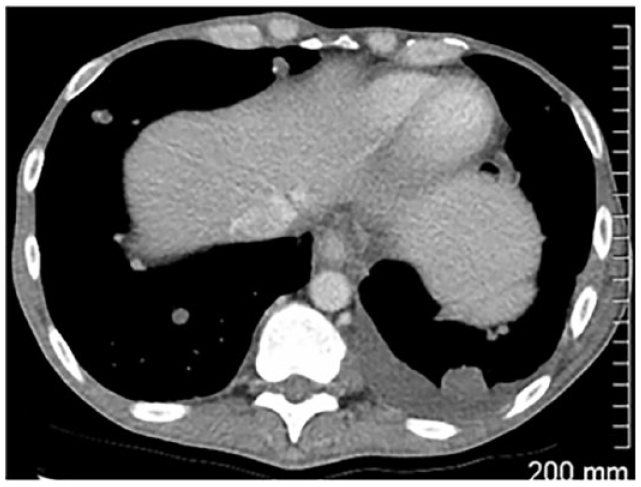
Further increase in size of the malignant left pleural effusion.
Status: iCPD
At visit 2, there is no change in the target lesions. However, there is unequivocal increase in the amount of peritoneal carcinomatosis. This meets the criteria from confirmed progression of disease (iCPD). It is worth noting that for iCPD to be confirmed progression can occur in a lesion category that had not caused iUPD (in this case the initial progression was in target lesions, and the sequent progression was in non-target lesions).
iRECIST: Frequently Asked Questions
If the patient does not meet the criteria for iUPD, can the radiologist simply follow the principles of RECIST 1.1? Yes, if the criteria for iUPD have never been met, then follow RECIST 1.1.
If the patient progresses to iUPD in one lesion category (eg, target lesions), at the 4 to 8 week follow-up scan does the patient have to demonstrate progression in the same category to be classified as iCPD? No. If iUPD occurred due to progression in one category (target, non-target, or new lesion) then progression in another lesion category can also confirm iCPD.
Can a patient stay in iUPD indefinitely? Yes, unless the criteria for iCPD, iSD, iPR, or iCR are met, the patient can stay in the iUPD category indefinitely. iUPD can be assigned multiple times (not necessarily consecutively).
If iCPD is not confirmed at the 4 to 8 week follow-up, when should the patient next be imaged? Imaging should revert to the originally planned schedule. For example, if scans were scheduled for 10, 20, and 30 weeks and the patient had a scan to confirm progression at 15 weeks (but iCPD did not occur), then the next scan should occur at 20 weeks (unless deemed clinically necessary).
What date should be used as the date of progressive disease in order to calculate progression free survival (PFS)? This should be the date on which iUPD occurred, provided that iCPD occurs at the subsequent assessment. If iUPD occurs but is followed by iSD, iPR, or iCR, then that date of iUPD should not be used to calculate PFS.
If the patient undergoes iUPD and then improves to iSD, iPR, or iCR, are they classified as iCPD the next time they progress? No. The patient must return to a status of iUPD before iCPD can occur.
Can patients be designated as iCR even if they have previously been classified at iUPD? Yes. A prior designation of iUPD does not preclude subsequent iCR.
Can the best overall response (BOR) be classified as iSD, iPR, or iCR if the patient has previously had a designation of iUPD? Yes, this is possible. A prior designation of iUPD does not preclude a subsequent best overall response of iSD, iPR, or iCR.
If a patient has a status of iUPD, can a scan obtained with 4 weeks be used to confirm progression of disease (iCPD)? Yes, this is possible if the scan was performed for worsening clinical symptoms or signs and the scan reveals additional new lesion not previously identified. However, if this scan only shows an increased size of lesion but no new lesions, it should not be used to confirm progression of disease as pseudoprogression could still account for these findings. In this case, a follow-up scan should be performed within the standard 4 to 8 week window to assess for confirmed progression.
Conclusion
The use of novel response assessment criteria such as iRECIST are important in the management of patients receiving immunotherapy. It is also important that these criteria are used with upmost accuracy to ensure patient are managed appropriately. iRECIST are a detailed, complex set of criteria and this can lead to challenges with their use and implementation. This review article can be used as a reference tool by radiologists and physicians when using iRECIST and aims to help radiologists accurately and efficiently use the criteria and to help physicians understand the rationale behind the status given to their patients.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
