Abstract
Peritoneal disease can be classified as either benign or malignant in nature. Malignant peritoneal disease can be further considered as either primary or secondary in origin. Primary peritoneal malignancy includes peritoneal mesothelioma, serous carcinoma, and desmoplastic small round cell tumour. Peritoneal carcinomatosis is the most commonly encountered secondary malignant peritoneal disease, typically of ovarian, gastric, colorectal, pancreatic, small bowel neuroendocrine, or breast origin. Others include peritoneal lymphomatosis and sarcomatosis. Benign peritoneal pathology may mimic malignant disease. Differentiating benign from malignant peritoneal pathology can be challenging, but is critical to guide appropriate care and avoid unnecessary intervention. Cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) offers potentially curative treatment for patients with peritoneal carcinomatosis, pseudomyxoma peritonei, and peritoneal mesothelioma. For such patients, the radiologist provides crucial pre-operative information highlighting sites of disease involvement, particularly for sites which are challenging to assess at laparotomy or laparoscopy, including the hepatic dome, subdiaphragmatic space and mesenteric root. The radiologist is also essential to identify potential contraindications to surgery, as well as interpreting normal post-operative appearances, complications and assessing for disease recurrence.
Introduction
Historically, peritoneal malignancy carried significant morbidity and mortality, with limited effective treatment options for clinicians. 1 However, understanding of tumour biology has improved in recent years. This, coupled with new combination treatment strategies, namely cytoreductive surgery (CRS) with heated intraperitoneal chemotherapy (HIPEC) and or early post-operative intraperitoneal chemotherapy (EPIC) has led to improved survival when used for appropriately selected patients,2,3 although the use of EPIC does remain controversial internationally. 4
Alongside improvements in treatment, there have been significant improvements and advancements in imaging technology coupled with increasing availability.5-9 Given that CRS, HIPEC, and EPIC carry the potential for significant morbidity and mortality, this augments the role of the radiologist in the process of shared decision-making. Radiologists may identify patients for whom complete cytoreduction may not be feasible, as well as identifying potentially subtle areas of disease requiring closer inspection at surgery, which may be otherwise overlooked.
Anatomy
The peritoneum is the largest serous membrane in humans. The peritoneal cavity represents a potential space, formed between the thin visceral and parietal peritoneal membranes and covers the organs of the abdomen and pelvis either partially or completely. Under normal circumstances, trace of fluid within the peritoneal cavity facilitates smooth movement of the peritoneal layers. There are important sex-specific differences between males and females. In males, the peritoneal cavity is completely closed. In females, a connection exists between the peritoneal cavity and the fallopian tubes around the isthmus, which can be consequential as a potential pathway for disease spread in both benign and malignant conditions. The transverse mesocolon divides the peritoneal cavity into supramesocolic and inframesocolic compartments.
Peritoneal fluid flows in a predictable manner (Figure 1). Low subdiaphragmatic pressure caused by respiratory movement draws fluid into the upper abdomen, with gravity returning fluid to the pelvis. Fluid travels from the pelvis to the upper abdomen via the paracolic gutters, is redistributed via the falciform ligament to the greater and lesser omenta and also over the surface of the bowel. Fluid travels from the abdominal infracolic compartment to the pelvis. Fluid pools in dependent recesses and regions of stasis. Common sites of peritoneal disease include the retrovesical region, the ileocecal junction, the rectosigmoid, right paracolic gutter, Morison’s pouch, the right subdiaphragmatic space, the gastric antrum and the greater omentum.
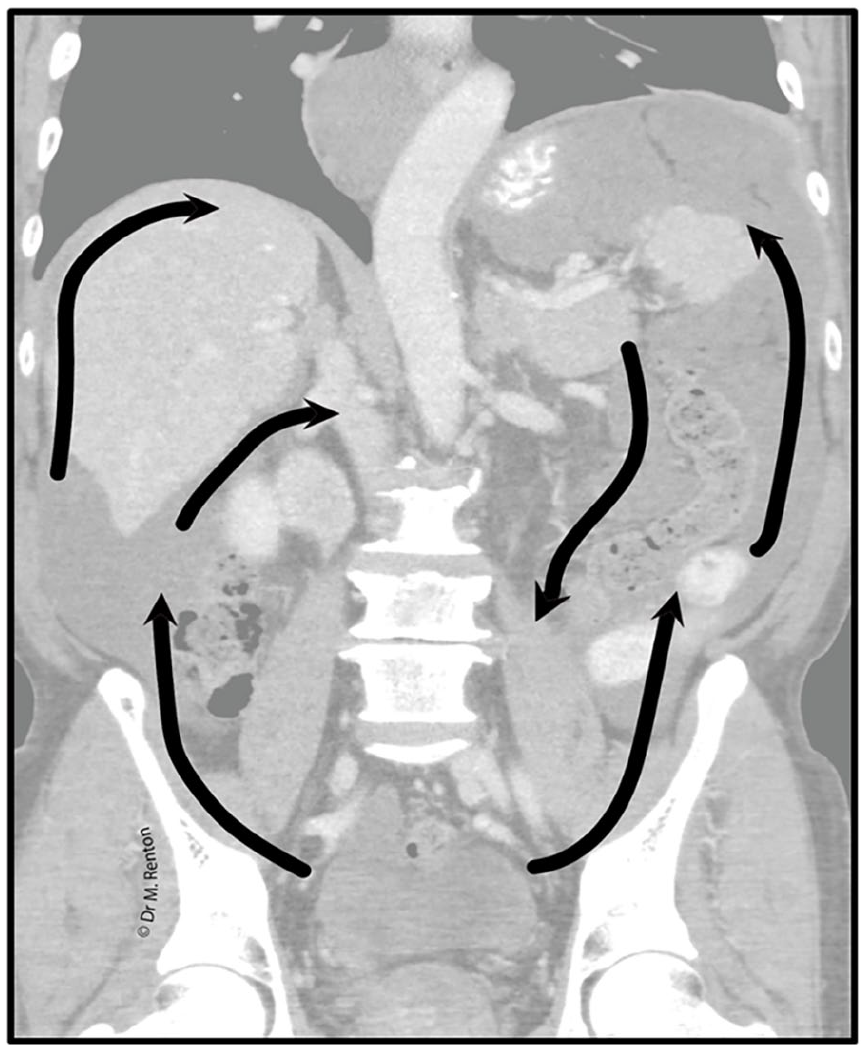
Coronal CT shows the typical flow of peritoneal fluid (black arrows) in a patient with pseudomyxoma peritonei with liver and splenic scalloping. © Dr. Mary Renton.
Mechanisms of Spread of Peritoneal Disease
Peritoneal disease may spread via a number of mechanisms, including
(i) Direct spread, from one organ to another: In the setting of gastric, pancreatic, colorectal, appendiceal, and ovarian carcinoma
(ii) Lymphatic spread, via sub-peritoneal lymphatics: In the setting of colorectal and ovarian carcinoma
(iii) Along the flow of ascitic fluid, that is, redistribution phenomenon: In the setting of gastric, ovarian, and colorectal carcinoma
(iv) Haematogenous spread: In the setting of disease arising from an extra-peritoneal primary, including breast cancer, lung cancer, and melanoma
Classification of Peritoneal Tumours and Imaging
As per the World Health Organization (WHO), tumours of the peritoneum in adults can be broadly categorized as either primary or metastatic (Table 1). 10
WHO Classification of Tumours of the Peritoneum. 10

Primary Peritoneal Tumours
Many tumours of primary peritoneal origin are recognized. While the vast majority of these are considered rare, we provide an outline of the features of some of the more commonly encountered entities below.
Peritoneal Mesothelioma
This is a rare entity, with a male predilection and more commonly seen in the sixth to seventh decades of life. Histologically, it may be categorized as: (a) epithelioid, the most common (b) sarcomatoid, most rare and aggressive subtype (c) biphasic/mixed type, second most common with a worse prognosis than epithelioid type. 11 Morphologically, it can manifest as (a) wet-type, with ascites and peritoneal plaques or nodules being the main features (b) dry-type, where peritoneal masses are seen often in the absence of ascites (Figure 2) (c) mixed-type, which has features of both wet and dry types. 12 Although not present in all cases, calcified pleural plaques may be observed and may be a useful imaging feature to help raise the index of suspicion of the reader. Typically, primary peritoneal mesothelioma manifests without distant supra-diaphragmatic disease or distant metastatic lymphadenopathy. This may be a useful imaging feature to help distinguish from metastatic disease manifesting with secondary involvement.
Axial and coronal images from CT abdomen and pelvis with IV contrast. There is plaque-like peri-hepatic soft-tissue thickening. No ascites. This was proven primary peritoneal mesothelioma (dry-type).
Solitary Fibrous Tumour of the Peritoneum
Solitary fibrous tumours (SFT) are known to occur throughout the body. Imaging features are not specific and tissue sampling is often required to confirm diagnosis. Neither small nor large size of the lesion can help confirm the diagnosis, as many SFTs may be quite large at initial presentation. Depending on the internal composition of the lesion, their appearance on unenhanced imaging can be variable. Lesions may appear similar in attenuation or signal intensity to adjacent muscle if there is a high cellular content. However, intralesional foci of low density or cystic appearing change may be seen if there are internal myxoid or cystic components. They often appear as solid lesions which display avid post-contrast enhancement, both on CT and MRI. They may be surrounded by prominent surrounding vessels. Displacement and compression of surrounding vascular structures may be seen. Direct infiltration with invasion into surrounding vasculature is atypical for SFTs. 13
Desmoid Fibromatosis
Fibromatosis is known to affect many parts of the body. Regardless of location, imaging features tend to be relatively non-specific. Although technically considered a benign entity, they can demonstrate locally aggressive imaging features and thus may present with acute complications, which occasionally may be the first presentation of disease. Morphologically, fibromatosis exists on a spectrum. It may be either well-defined or ill-defined with the appearance of local infiltration. It typically appears as soft tissue attenuation on CT and more commonly tends to be relatively hypoenhancing. Although historically thought to be predominantly markedly hypointense on T1- and T2-weighted MRI, it has become increasingly recognized that this may display heterogenous signal intensity as the composition of the lesion changes over time with variation between cystic or myxoid type components, which may be replaced by fibrous tissue over time. 14 Due to the infiltrative pattern of growth which can be seen, fibromatosis may encase adjacent structures such as vasculature or bowel loops, leading to acute complications such as bowel obstruction.
Peritoneal Inclusion Cyst
Most commonly, peritoneal inclusion cysts are identified in women of reproductive age, although they may also be seen in post-menopausal women or in male patients. A supporting history of pelvic or peritoneal inflammation is often present, for example a history of endometriosis, inflammatory bowel disease or in patients with previous pelvic surgery. Typically, this manifests as a relatively homogenous cystic structure which may contain a few thin internal septations. It should conform to surrounding structures, for example insinuating along the pelvic sidewalls, but typically it does not displace surrounding structures. In female patients, a typical finding is to identify a normal ovary which can be located either along the lateral margin of the cyst, or within the centre of the cyst itself. Findings such as nodules along the wall, thickened septations with internal enhancement or evidence of vascularity, generalized ascites or enlarged lymph nodes should raise suspicion for an alternative diagnosis and should prompt further work-up.
Secondary Peritoneal Tumours
Peritoneal metastasis accounts for the vast majority of peritoneal disease. This encompasses peritoneal carcinomatosis, peritoneal sarcomatosis, pseudomyxoma peritonei, and peritoneal gliomatosis. Overall, peritoneal carcinomatosis accounts for the vast majority of peritoneal metastatic disease. The presence of peritoneal metastasis inherently implies more advanced disease and typically portends a worse prognosis. Peritoneal carcinomatosis as a term was often historically used incorrectly, as it only refers to peritoneal malignancy of epithelial origin. Peritoneal malignancy is a preferred term, encompassing all cell lineages.
Ascites, unless otherwise clearly explained, must be regarded as an indicator of potential underlying peritoneal disease. Differentiation between serous and mucinous ascites may be feasible on imaging. Mucinous ascites should be considered suggestive of pseudomyxoma peritonei, potentially related to gastric signet ring cell carcinoma or mucinous appendiceal neoplasms. Radiologically, this is manifest by fluid with increased density and or septations. Eventually, the fluid typically exerts mass-effect, displacing the mesentery, omentum, and bowel and indents the surface of solid organs resulting in a classic “scalloped” appearance. Serous ascites typically lacks this mass-effect, although may become loculated.
Solid components of peritoneal malignancy are typically seen as either single or multiple nodules of varying size along the peritoneum. If large enough for interrogation, the nodules may display contrast enhancement. Over time, the soft tissue nodules may ultimately become calcified. Although macronodular disease is typically readily detectable, micronodular disease can prove evasive, even for experienced radiologists and regardless of imaging modality being used.
An “omental cake” is seen when there is advanced disease involvement of the greater omentum, manifest by a conglomerate soft tissue mass, which may be sizeable even at baseline imaging.
Although more discrete, nodular forms of disease may initially be more readily visible, close inspection of bowel and vascular pedicles is required in all cases to assess for the presence of laminar type disease infiltration, which can manifest as a predominantly linear pattern of thickening and tissue retraction.
Gravity dependent areas and areas of stasis require close inspection, in particular.
Specific entities and imaging features are described in further detail below.
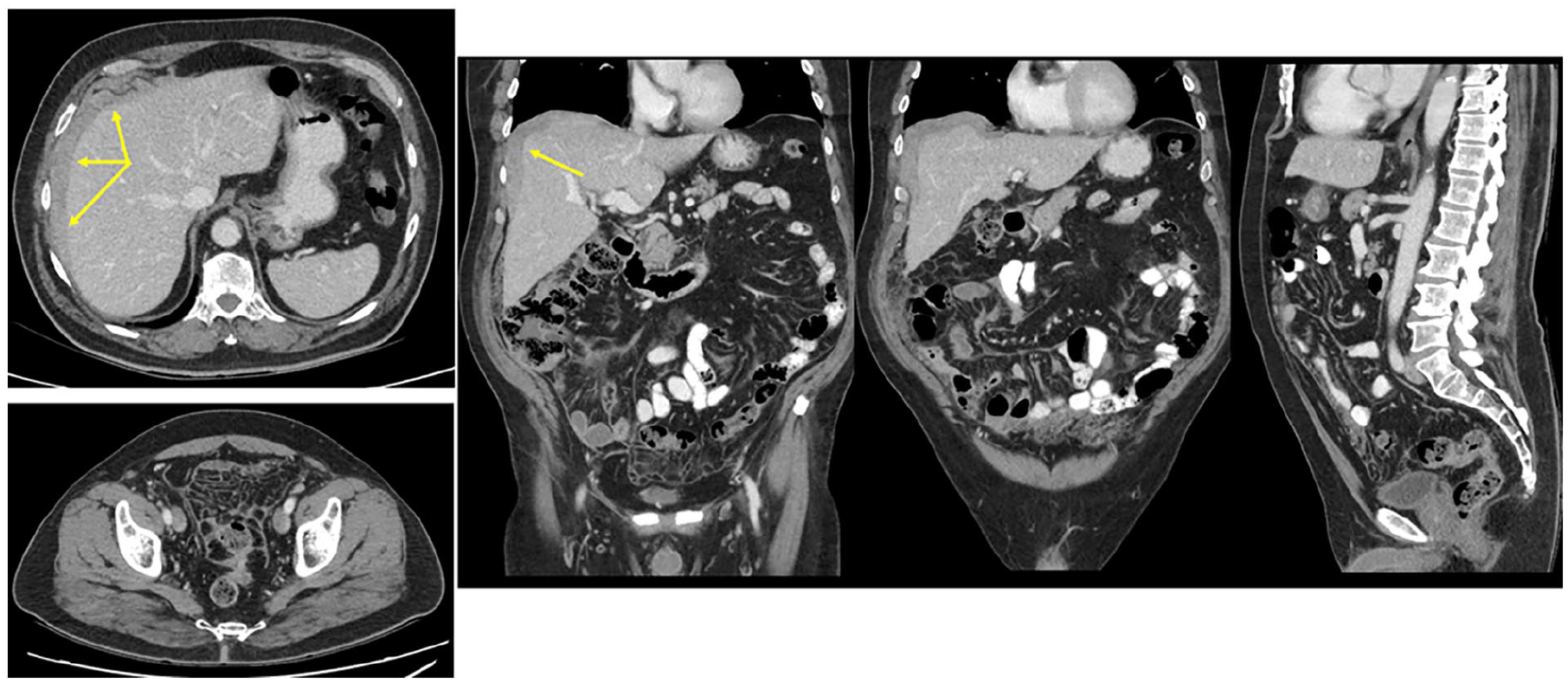
Pseudomyxoma Peritonei
Pseudomyxoma peritonei (PMP) at its origin is caused by a mucin-producing tumour, typically of appendiceal origin. Radiologically, it is characterized by the development of mucinous ascites, which often has a somewhat loculated appearance and is associated with multiple implants, which eventually results in a “scalloped” morphology of solid organs such as the liver (Figures 3 and 4). It is typically rare to see metastatic extension of disease beyond the confines of the abdomen or pelvis. The stage and grade of PMP is dependent on whether epithelial cells are identifiable in the extra-cellular mucin and, if so, by the abundance of such cells. Imaging is unreliable in detecting and quantifying the presence of extra-cellular mucin.
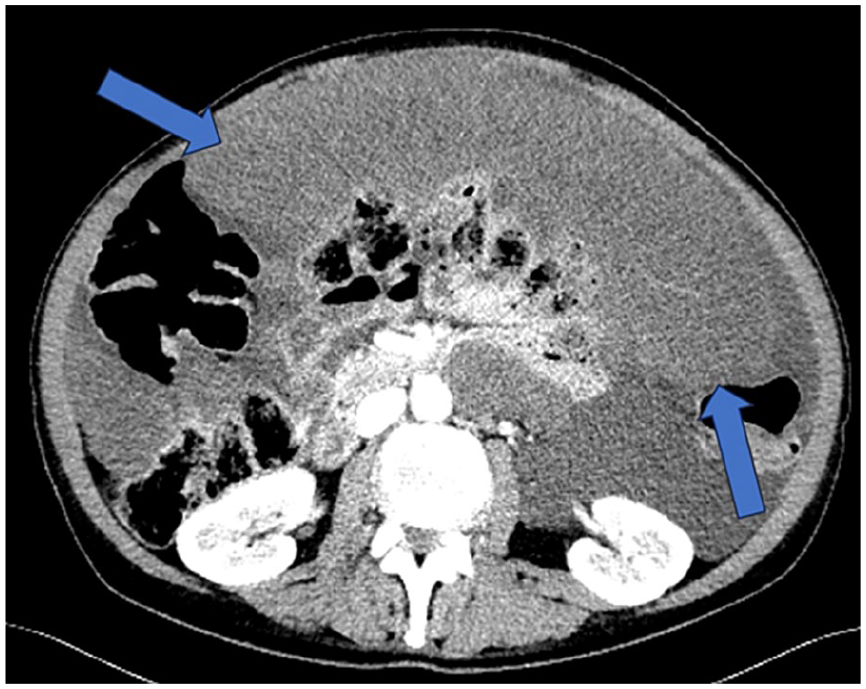
Axial image from CT abdomen and pelvis, illustrating a large solid omental cake (blue arrows) and mucinous ascites in a patient with pseudomyxoma peritonei.
Axial, coronal, and sagittal images from IV contrast-enhanced CT abdomen and pelvis in a 63 year-old patient with a history of a mucinous appendiceal neoplasm. There are multiple lobulated cystic attenuation masses throughout the abdomen and pelvis, compatible with pseudomyxoma peritonei.
Peritoneal Carcinomatosis
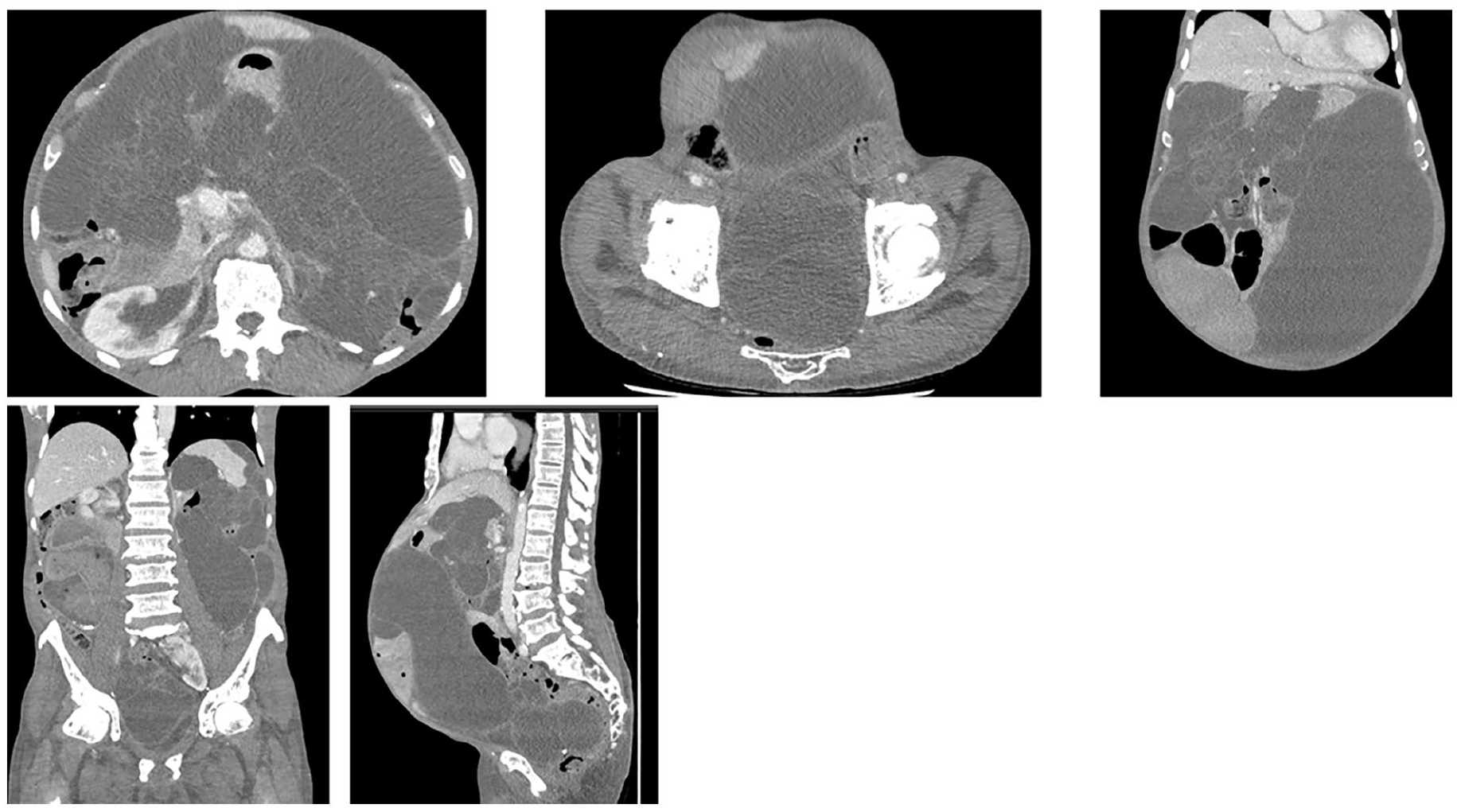
The imaging features are non-specific and there may be overlapping features. The most commonly encountered signs of disease include ascites, discrete peritoneal nodules or implants, larger more confluent mass-like soft tissue including omental cakes (Figures 5 and 6), infiltration along the mesentery or serosal deposits along the gastrointestinal tract.
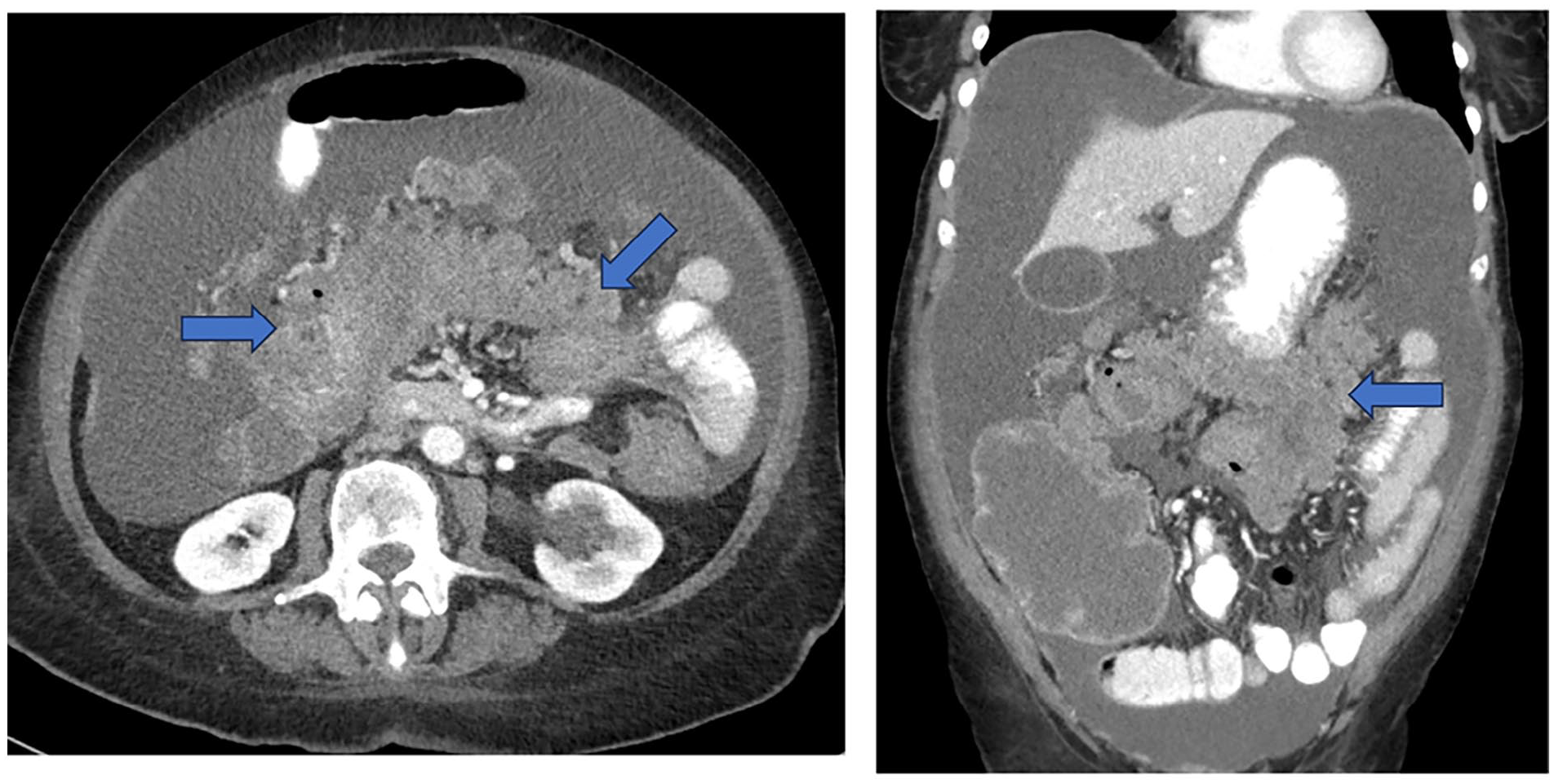
Axial and coronal contrast enhanced images from CT abdomen and pelvis, illustrating nodular and mass like disease (blue arrows) from an ovarian primary malignancy with large volume ascites, compatible with peritoneal carcinomatosis.
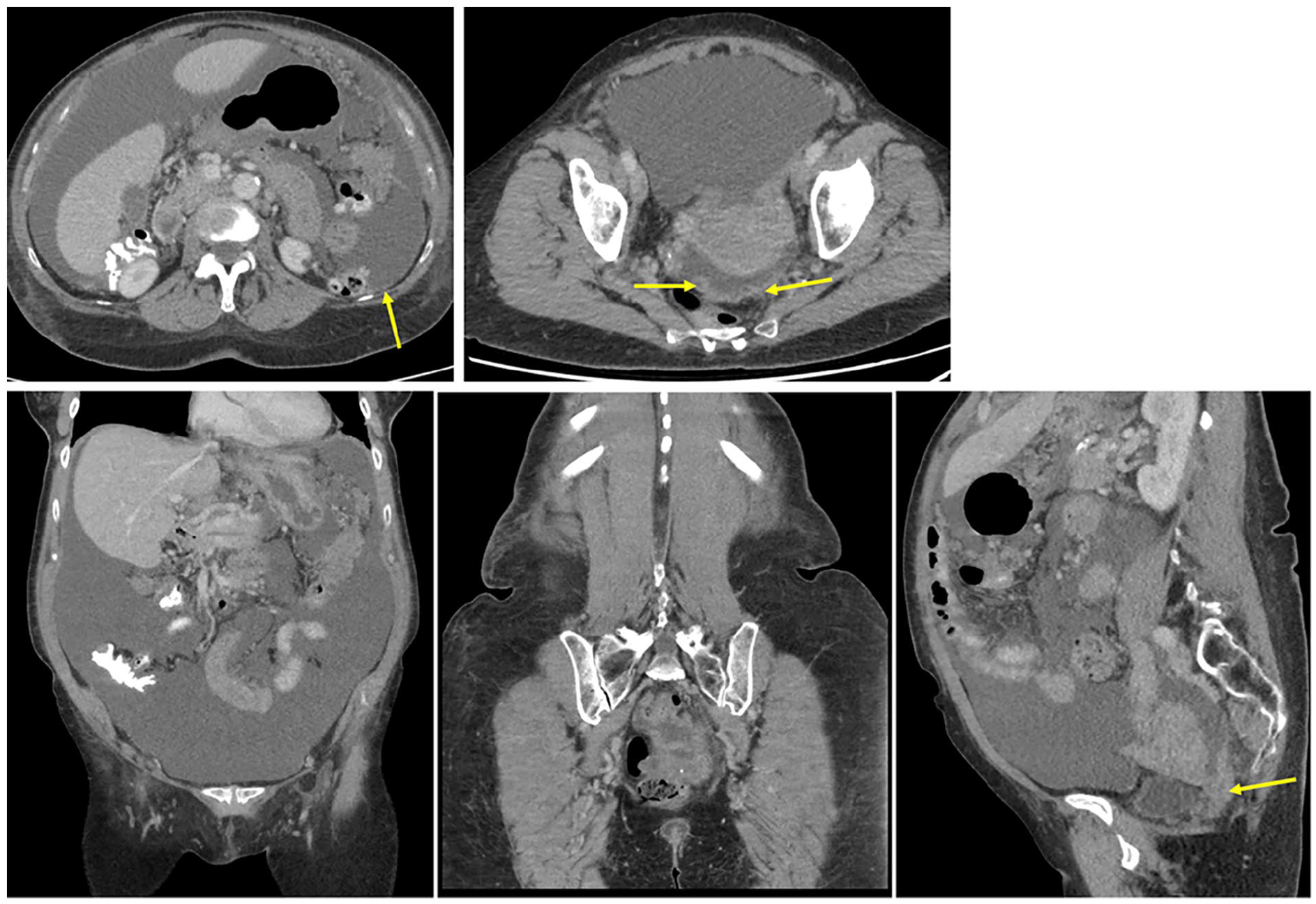
Axial, coronal, and sagittal images from contrast-enhanced CT abdomen and pelvis in a patient with a history of a serous ovarian neoplasm. There is large volume ascites and nodular thickening of the peritoneum (yellow arrows) with mesenteric injection. Findings are consistent with peritoneal carcinomatosis.
Distinguishing between other forms of peritoneal malignancy
Identifying the primary site of disease is considered key to ensuring the optimal treatment regime in these patients. However, this can be challenging both clinically and radiologically, particularly in cases of advanced disease, as there may often be overlap of imaging features and the primary site of disease may be somewhat masked by surrounding disease. Histological and immunohistochemical analysis can help to advance the diagnosis, although complementary clinical and radiological input is often needed to determine the primary site of the offending tumour.
Although not specific, some imaging features may help to point toward a more likely primary site of disease. For example, patients with peritoneal carcinomatosis secondary to pancreatic or ovarian cancer tend to have a greater burden of omental disease in the earlier stages of disease. Peritoneal carcinomatosis tends to mimic the morphology of the primary tumour, particularly in the earlier stages of disease. For example, in the setting of gastric adenocarcinoma, peritoneal disease tends to have a more solid and spiculated appearance.
Patients with peritoneal sarcomatosis may have a known history of, or an identifiable, extra-abdominal sarcoma with secondary metastatic involvement which often has a more conspicuous dominant soft tissue mass.
Ultimately, however, it should be noted that overlapping features of peritoneal disease can make accurate identification of the primary site of disease challenging in many cases.
Mimics of Peritoneal Tumours
There are a number of potential mimics of peritoneal tumours, most commonly of infectious or inflammatory etiology. A discussion of the most common entities is provided below.
Infectious Etiology
Tuberculous peritonitis is a key differential consideration of peritoneal malignancy. The imaging features can be challenging to differentiate and therefore at initial presentation, it may be reasonable to describe peritoneal carcinomatosis as a differential for tuberculous peritonitis and vice versa. From a clinical perspective, certain features from the history may help to point toward a diagnosis of tuberculous peritonitis. For example, if the patient is from a region where tuberculosis is endemic. It should also be a consideration in patients who are immunosuppressed, either primarily related to underlying disease or secondarily as a result of medications used to treat disease. For example, in patients taking immune modulating agents in the setting of chronic conditions such as rheumatoid arthritis, amongst others. With an aging population, it is important to recall that patients may have had a very remote exposure to tuberculosis many years previously and therefore re-activation disease following initiation of immune modulating therapy is a possibility. A detailed exposure history from the patient may help to clarify this. Furthermore, if the patient presents predominantly with a clinical picture suggesting infection, the possibility of infectious etiology should be higher among the differential considerations. From an imaging perspective there are certain, albeit non-specific, findings which may help to point toward the diagnosis. Features to specifically scrutinize include the presence of low-attenuation lymphadenopathy (Figure 7), the presence of calcified lymph nodes or granulomas which hint toward prior granulomatous disease or involvement of the gastrointestinal tract with wall thickening and stranding around the cecal pole and ileocecal valve in particular. Involvement of other organ systems such as the kidneys, manifest by wedge-shaped areas of hypodensity or urothelial thickening may also be seen. Despite these findings, however, this diagnosis ultimately requires multi-disciplinary input. Feasibly, findings such as low-attenuation lymphadenopathy can be seen in the setting of metastatic necrotic lymphadenopathy. Furthermore, the ileocecal valve is an area known to be prone to disease involvement in the setting of peritoneal carcinomatosis and therefore positive evidence of involvement is not reliable in distinguishing either entity.
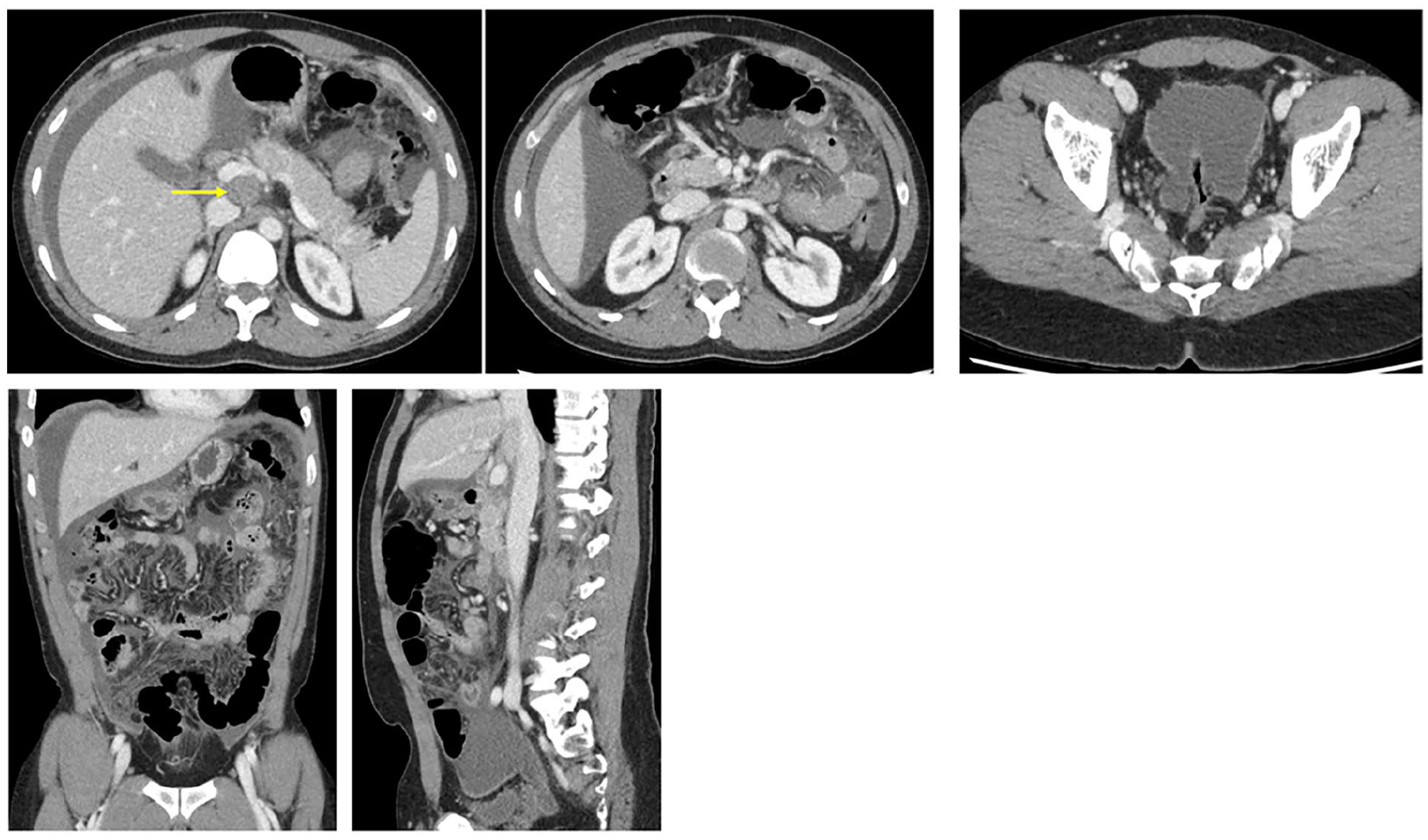
Axial, coronal, and sagittal images from contrast-enhanced CT abdomen and pelvis in a 29 year-old male patient. There is low-attenuation lymphadenopathy (yellow arrow), ascites and mild diffuse peritoneal thickening and enhancement with mesenteric injection and stranding. Findings were shown to be related to tuberculosis.
Inflammatory Etiology
A number of inflammatory type conditions can result in sterile, benign peritonitis. Among the more common causes include sclerosing peritonitis which occurs as a result of chronic, low-grade foreign-body type reaction. This is most commonly encountered among peritoneal dialysis patients, although is also recognized among patients with longstanding ventriculoperitoneal or lumboperitoneal shunts. It is important to review any historical imaging available to determine the presence of previously-sited catheters which have subsequently been removed. For example, in renal transplant patients who may have previously been treated with peritoneal dialysis more remotely. Typical imaging findings include relatively diffuse but smooth thickening involving both the visceral and parietal peritoneal reflections (Figure 8). Calcifications may also be present. Relative stability in size and morphology across serial studies or at minimum slow evolution of changes helps to support the diagnosis, although as in the case of infectious causes of peritoneal disease, an appropriate clinical history is most helpful in establishing the diagnosis.
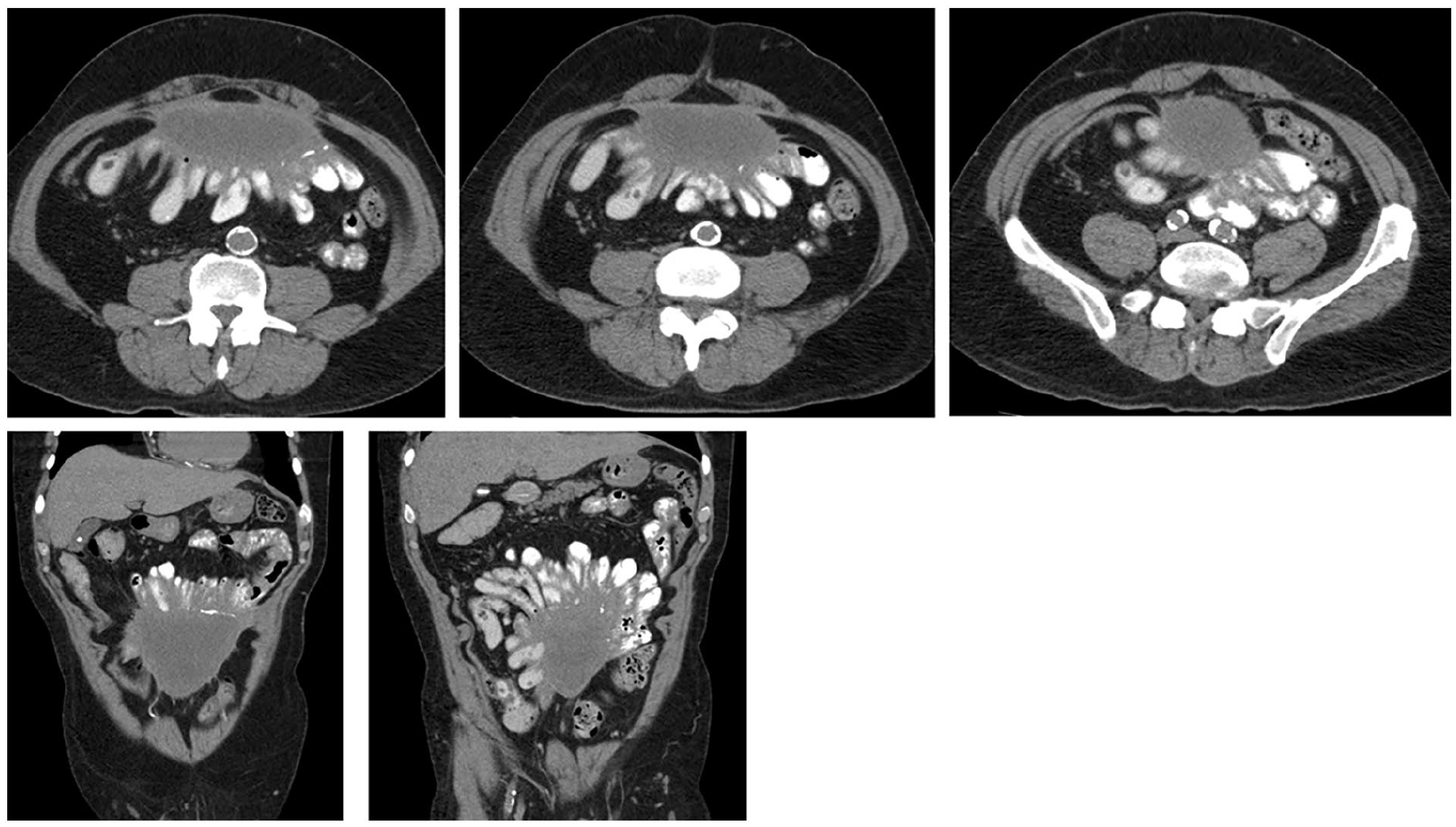
Axial and coronal images from CT abdomen and pelvis in a 61-year-old patient, who had a history of peritoneal dialysis and tuberculosis. There is a predominantly cystic attenuation mass in the ventral mid-lower abdomen with peripheral calcification and soft tissue thickening. This involves both the visceral and parietal peritoneal reflections. Bowel loops are tethered to the surface. Imaging findings were stable over long-term follow-up and consistent with manifestations of sclerosing peritonitis.
Surveillance Imaging in Peritoneal Malignancy
The frequency of follow-up imaging and to a degree the choice of modality will be influenced by multiple factors, including the extent of cytoreduction achieved if surgery has been undertaken, as well as the underlying disease process. Even for experienced radiologists with a selection of multi-modality imaging, it is recognized that differentiation between expected post-treatment findings versus residual or recurrent disease can be challenging. Nodular or more laminar thickening of the bowel wall or mesentery secondary to scar and granulation tissue formation may display post-contrast enhancement as well as increased metabolic activity on functional imaging such as PET/CT. Moreover, the morphology of recurrent peritoneal disease may differ significantly from the baseline imaging appearances. 15 Regardless of imaging modality, surveillance imaging in patients with peritoneal metastasis requires a significant time investment by the reporting radiologist.
The Role of the Radiologist
The role of the radiologist is multi-faceted in the context of caring for patients with peritoneal disease. Across various stages of the patient care pathway, the radiologist plays a key role in the following areas:
Suggesting or confirming the presence of peritoneal disease on the basis of imaging and, where possible, suggesting a possible primary site of disease
Identifying a suitable site for percutaneous fluid or tissue sampling ± performing biopsy
Assessing the extent of tumour spread
Identifying sites of disease considered unfavourable for surgery (outlined below)
Assessing response to systemic and surgical therapy and potential complications thereof
Assessing sustained treatment response or identifying local or distant disease recurrence
Unfavourable sites of disease include extensive involvement of the small bowel mesentery, disease involvement around the porta hepatis, extensive diaphragmatic disease involvement, extensive pancreatic disease, extensive pelvic side-wall disease, or retroperitoneal disease involvement.
The PAUSE acronym can serve as a useful reporting framework for radiologists, which may help to identify patients for whom complete cytoreduction may not be technically feasible or may be potentially futile. It may also serve as a useful template for those radiologists less-experienced in reporting peritoneal disease to formulate a more focused and clinically relevant report. It focuses on the following key elements 16 :
P: Primary tumour and peritoneal carcinomatosis index (PCI)
A: Ascites and abdominal wall involvement
U: Unfavourable sites of involvement
S: Small bowel and mesenteric disease
E: Extra-peritoneal metastases
Cytoreductive Surgery and Patient Selection
Given the potential associated morbidity and mortality with cytoreductive surgery, it is imperative that only those patients for whom complete cytoreduction may be feasible are selected for surgery.17,18 A number of key domains must be considered in assessing patients for potential surgery: (i) Clinical performance status (ii) histopathological assessment (iii) peritoneal cancer index (PCI) (iv) completeness of cytoreduction (CC) score.19,20
Clinical/Performance Status
Using the Eastern Co-Operative Oncology Group (ECOG) scoring system, patients require a score <2 to be considered candidates for cytoreductive surgery.
Histopathological Assessment
Pre-operative histopathological assessment is an important tool in decision making for surgery. Depending on the primary disease process, some patients with peritoneal disease are known to have a better prognosis than others. For example, patients with peritoneal disease secondary to epithelial ovarian cancer, certain patients with peritoneal metastasis from colorectal cancer and patients with pseudomyxoma peritonei have more favourable outcomes.21-23 This is contrasted with patients who have peritoneal disease from a primary gastric cancer who are known to have a worse prognosis. 24
Peritoneal Cancer Index (PCI)
The PCI is a well-established tool used as an aid to defining the extent and volume of peritoneal disease. 25 The score ranges from 0 to 39, with lower scores typically associated with resectable disease and a better prognosis. If the radiologic PCI permits progression to attempted cytoreductive surgery, the radiologic findings should be correlated with findings at direct surgical inspection and palpation. Despite advances in medical imaging, it is recognized that discrepancy in findings between radiologic and intra-operative scores can occur26,27 and therefore, patients deemed surgical candidates on the basis of imaging may be inoperable at the time of surgery. Such patients thus undergo an “open and close” surgery.
To calculate the radiologic PCI, the abdomen is scored in 13 distinct regions (Figure 9). 25 There are 9 separate regions in the abdomen—right, centre, and left in the upper, mid, and lower abdomen. Further, the small bowel is divided into 4 parts—upper and lower jejunum, upper and lower ileum. Each region requires methodical inspection and a score of 0 to 3 assigned to each:
0: No measurable disease/absence of cancer
1: Metastatic deposit measuring <0.5 cm diameter
2: Metastatic deposit measuring 0.5-5 cm diameter
3: Metastatic deposit measuring >5 cm
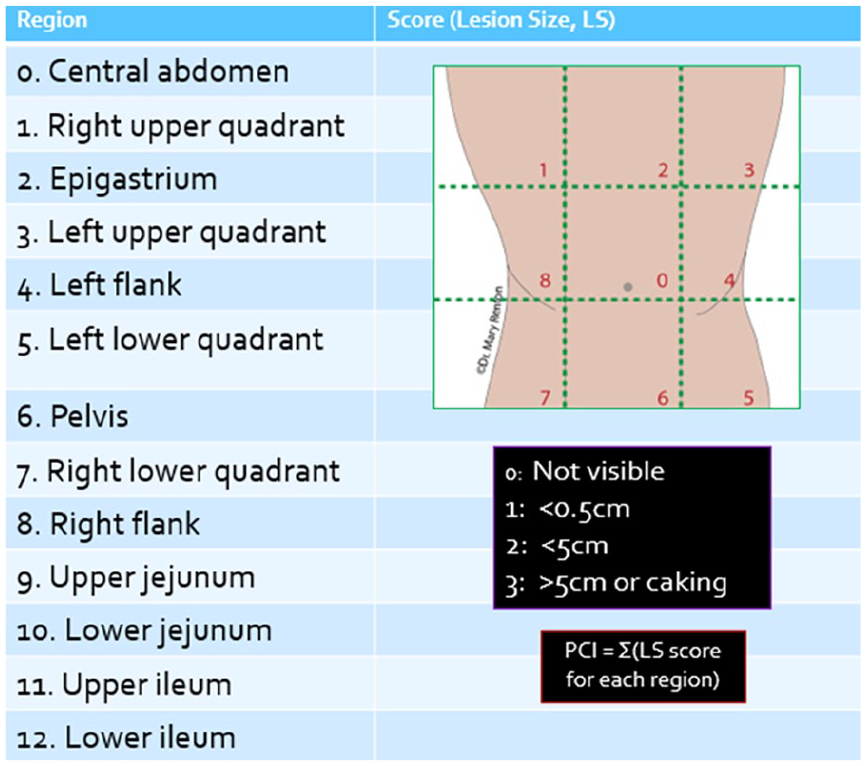
Calculation of the Peritoneal Cancer Index (PCI). Modified from van Driel et al. 23
Many centres adopt a threshold PCI score of less than 20 to determine patients who may be surgical candidates, although this is variable depending on the local practice and the specific patient characteristics. Some centres will, in certain circumstances, determine patients to be surgical candidates with a PCI of <24. Ultimately, decision making regarding suitability for surgery should be made as part of a multi-disciplinary team.
Although used as a benchmark scoring system in helping to guide patient selection, the PCI is not without limitations. As with many scoring systems, it is limited by the subjectivity related to the radiologist reading the study. Furthermore, there is a lack of standardization of the PCI to account for tumours of differing origin and tumour biology/pathology. 28 Therefore, its adoption and use in daily practice is variable across institutions. At our institution, which is a large quaternary referral centre, the practice is to not provide a formal PCI score in the radiology report. Rather, readers are encouraged to refer to it as an aid for reporting. At our institution, patients who are deemed potential surgical candidates will ultimately undergo diagnostic laparoscopy to determine if cytoreductive surgery is feasible.
Completeness of Cytoreduction Score
The completeness of cytoreduction (CC) score is calculated by the surgical team following the completion of cytoreductive surgery and is informed by findings of visual inspection and examination.
Broadly, the CC score is considered as either complete cytoreduction or incomplete cytoreduction.
Complete cytoreduction: CC-0, CC-1 CC-0: No residual macroscopic disease CC-1: Residual macroscopic disease <0.25 cm
Incomplete cytoreduction: CC-2, CC-3 CC-2: Residual macroscopic disease, 0.25-2.5 cm CC-3: Residual macroscopic disease, >2.5 cm
The radiologist should be mindful of the CC score when reporting surveillance studies, paying close attention in particular to those areas with a CC score ≥1.
Other Pre-Operative Scoring Tools
Peritoneal Surface Disease Severity Score (PSDSS)
The PSDSS is used in assessment of patients with peritoneal metastatic disease from a colorectal primary. The score is multi-faceted, taking into account the radiologic PCI, the patient’s clinical picture (pain, ascites, weight loss) and tumour biology (pathology, degree of differentiation, nodal status). Patients with PSDSS III-IV have worse life expectancy than those with PSDSS I-II. 29
Imaging Modality of Choice
Plain Film Radiography
Although abdominal radiographs should not be routinely relied upon to assess for the presence of peritoneal disease, radiologists should be familiar with signs that can be detected by plain film radiography. These include the hepatic angle sign, the flank stripe sign, centralization of bowel loops, and a generalized increased density over the abdomen and pelvis (Figure 10A). 30
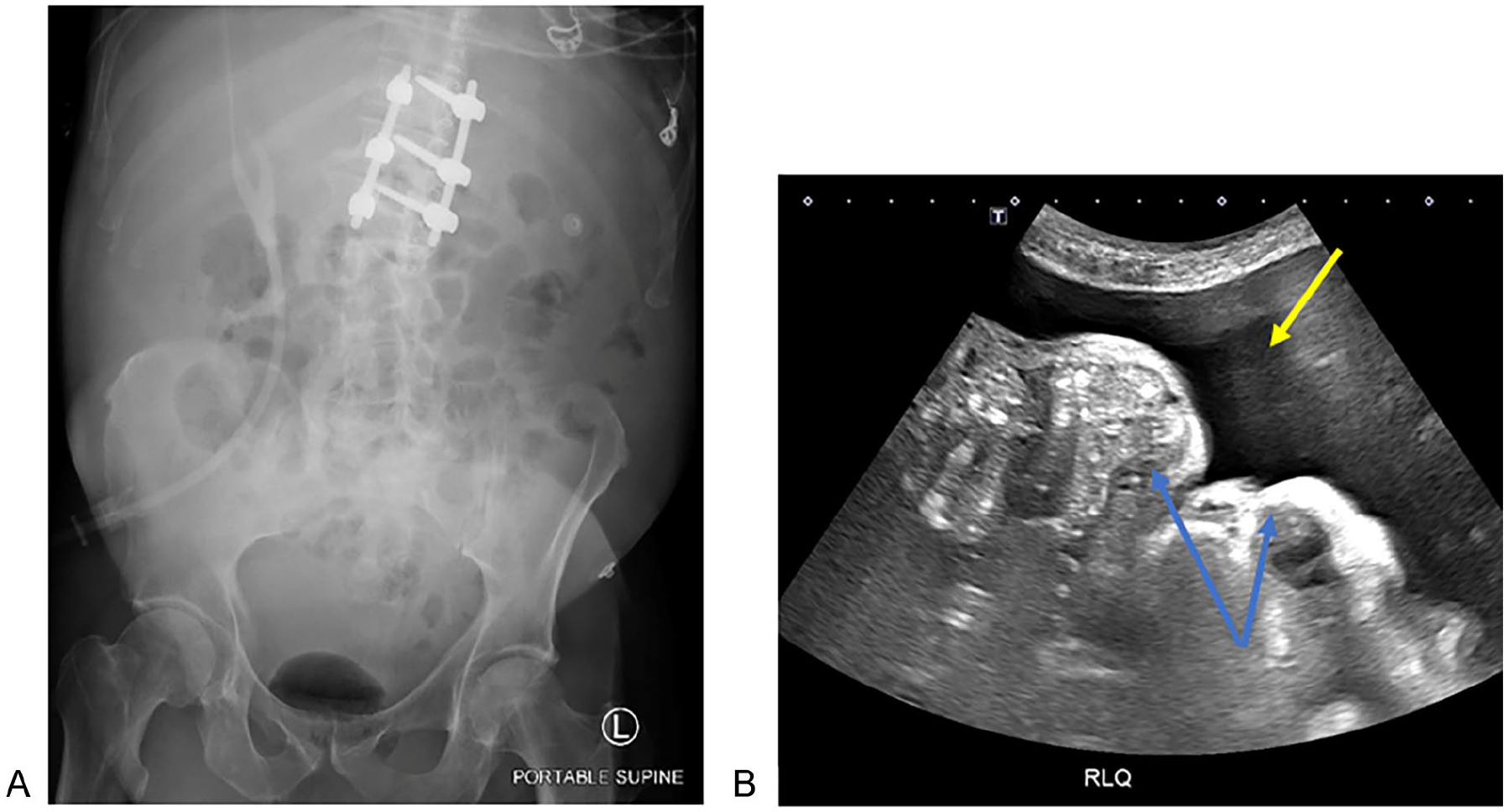
(A) Portable AP supine radiograph in a male patient. There is generalized increased density projected over the abdomen, clumping of bowel loops centrally and obliteration of the pro-peritoneal fat line. (B) A corresponding single image from ultrasound abdomen from the same patient, with image acquired from the right lower quadrant (RLQ), confirms the presence of ascites (yellow arrow) with displacement of bowel loops (blue arrows).
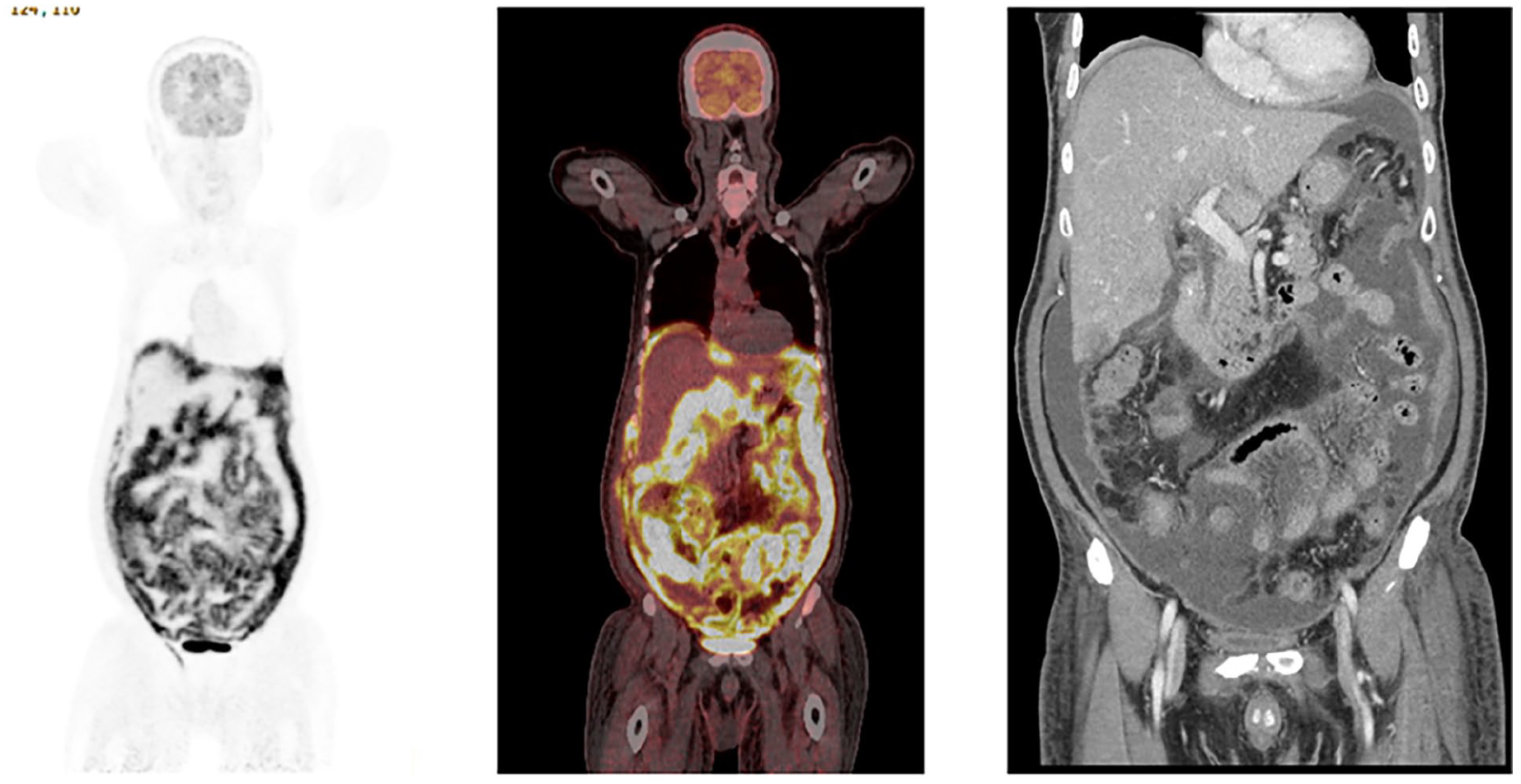
Coronal maximum intensity projection (MIP) image, fused coronal image from F18-FDG PET/CT and coronal CT abdomen and pelvis with IV contrast. There is diffuse intense increased metabolic activity related to the peritoneum, with diffusely increased metabolic activity also noted related to loops of small and large bowel. There is moderate—large volume ascites with thickening of bowel loops. Findings are consistent with peritoneal lymphomatosis in this patient with a history of diffuse large B-cell lymphoma (DLBCL).
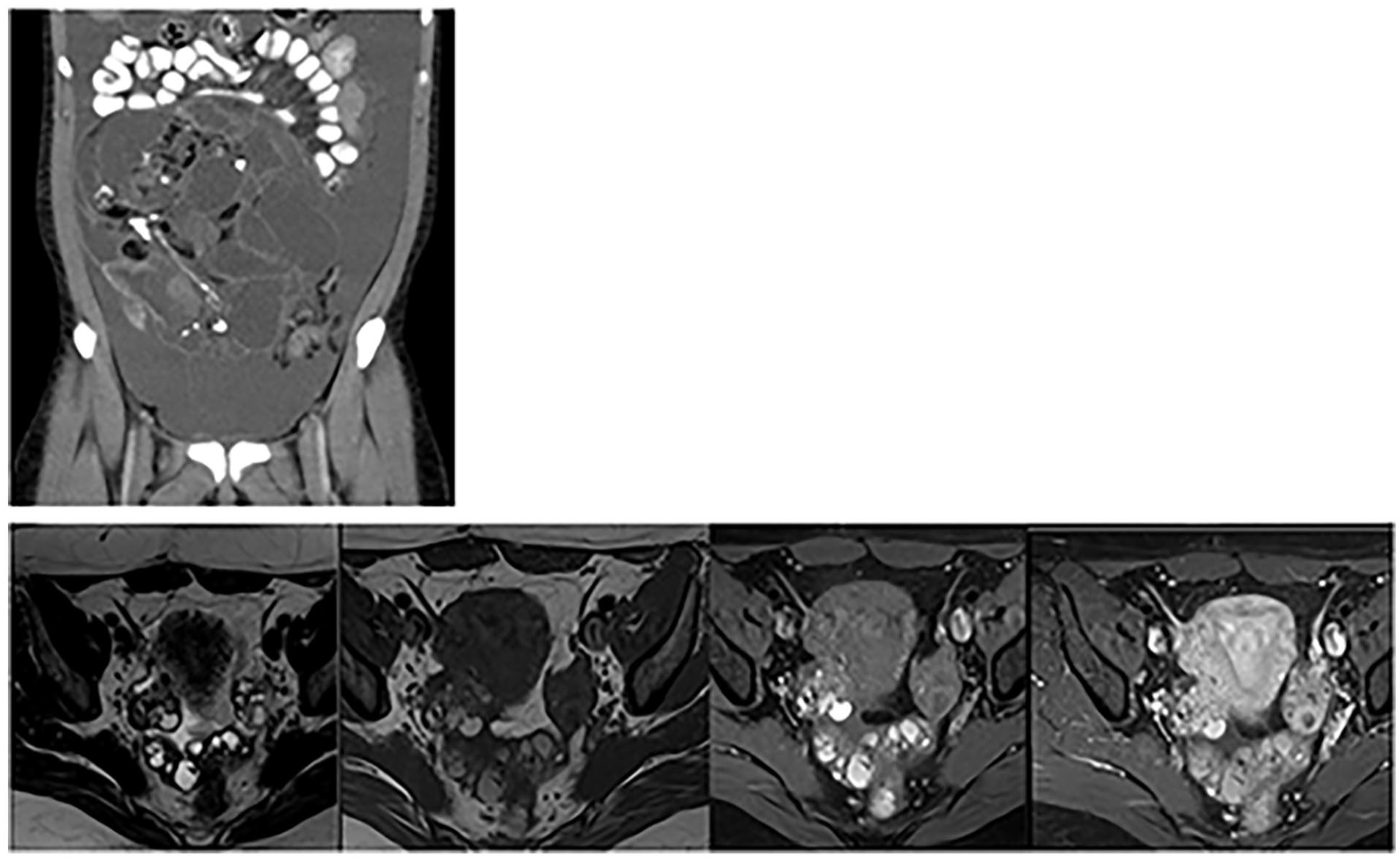
Top—Coronal image from oral and IV contrast-enhanced CT abdomen and pelvis in a 28 year-old female. There is large volume ascites. Heterogenous mass in the central abdomen, containing cystic spaces, macroscopic fat and areas of tooth-like calcification. Pathology confirmed immature teratoma. Bottom—In order—Axial T2 and T1 non fat-saturated, T1 fat-saturated pre-contrast, T1 fat-saturated post-gadolinium images acquired at 4 minutes post injection from the same patient, 3 years post-surgery. There are multiple peritoneal implants in the pelvis with heterogenous signal intensity. These were proven to be peritoneal implants related to spillage at the time of resection of the teratoma. Findings are consistent with gliomatosis peritonei.
Ultrasound (US)
The role of ultrasound in imaging peritoneal disease is reasonably limited. It can be helpful in confirming the presence of ascites where peritoneal disease is suspected (Figure 10B) and also in assessing for superficial peritoneal tumour deposits. It is limited, however, by the fact that it is operator-dependent and also by the field of view. Ultrasound may be employed for image-guided percutaneous tissue or fluid sampling, as well as in therapeutic procedures such as placement of drainage catheters.
Computed Tomography (CT)
Globally, CT remains the main-stay first line imaging modality for staging of peritoneal disease. Particular advantages are ease of access, relatively rapid scan acquisition, high spatial resolution and the ability to produce multi-planar reformats. Furthermore, CT abdomen/pelvis can be combined with CT thorax as part of the same study acquisition to assess rapidly for the presence of extra-peritoneal disease, in particular assessing for signs of pleural disease involvement. The provision of high-quality multi-planar reformat images improves assessment of anatomically challenging regions such as the subphrenic spaces.
Many centres advocate for the use of oral contrast to improve detection of small bowel deposits in particular. Negative contrast agents are preferred in many centres. Positive contrast agents may be used, although require specific caution to ensure that the contrast is adequately dilute so as not to obscure peritoneal disease. At our institution, the preference is to administer positive oral contrast agents before CT studies. However, we are also mindful of the fact that this is not always feasible. For example, if patients with a known history of extensive peritoneal disease present with a clinical picture of bowel obstruction, oral contrast may be omitted if the patient simply cannot tolerate ingestion or if it would cause a potential delay in operative management. The authors are also mindful that patients with peritoneal disease are more likely to have ongoing nausea that may be difficult to control with medical therapy. Therefore, for patients presenting for routine outpatient re-staging imaging, while oral contrast is preferred, it is not an absolute if the patient is unable to tolerate the agent.
Anatomic sites recognized as pitfalls in imaging include the porta hepatis, ureters, small bowel, mesentery, and the subphrenic regions and thus special attention to these sites is required. 31
Even with advances in thin-slice image acquisition and multi-planar reformats, CT remains limited in the detection of small lesions, with sensitivity of as low as 11% quoted among some studies for sub-5 mm lesions. 26 The radiologist should pay close attention to the liver for assessment of possible intra-parenchymal metastases versus possible serosal/capsular implants. The presence of peritoneal metastatic disease typically upstages the patient’s M category as part of their TNM staging. This can sometimes prove challenging and reviewing imaging findings across multiple modalities may be helpful.
Magnetic Resonance Imaging (MRI)
The overall diagnostic sensitivity in detecting peritoneal disease is somewhat comparable for both CT and MRI. However, MRI holds particular advantage in helping to augment sensitivity in detecting micronodular and laminar patterns of peritoneal disease. This is largely related to the addition of diffusion-weighted imaging (DWI) more routinely into protocols. This is further augmented by the addition of multiplanar fat-suppressed T1-weighted contrast-enhanced imaging, which, ideally, should be performed in both early and late phases (5-10 minutes) following injection of contrast. Peritoneal deposits typically display a pattern of delayed contrast-enhancement and should be more conspicuous against normal background peritoneum. Despite these benefits and improved access globally, access to MRI remains limited by cost and the duration of the study protocol. 15 Furthermore, given the longer time of the study, patients may experience difficulties with breath-holding for examination, particularly in the setting of concurrent pleural disease with pleural effusions, for example. Even with significant technical advances in recent years, MR imaging may be significantly degraded by artifact for example from hip prostheses. Susceptibility artefact related to prior abdominal surgery may also result in some limitations of assessment of the peritoneum and abdominal wall.
PET/CT (Positron Emission Tomography-CT)
18F-FDG PET/CT has been shown to have improved diagnostic accuracy in the detection of peritoneal disease when compared with either PET or CT performed in isolation. 32 The use of PET/CT does require special consideration, however, and is not routinely recommended in the assessment of mucinous peritoneal disease given the relatively low metabolic activity. Furthermore, PET/CT is limited by spatial resolution and it should be recognized that peritoneal deposits smaller than 1 cm may not be reliably detected, although this is likely to improve over time with newer generation scanners.
Increased peritoneal uptake should be interpreted with caution in the setting of previous intervention such as biopsy or drain placement. This generally leads to increased metabolic activity locally, either in a focal or more diffuse manner, in the immediate—short term following intervention. This is compounded, however, by the fact that peritoneal disease may seed along these tracts and therefore accurate differentiation of benign versus malignant versus combined pathology can be challenging.
PET/CT can add particular value in cases where extra-peritoneal findings may be equivocal on conventional cross-sectional imaging, either helping to support or refute a suggestion of extra-peritoneal metastatic disease. This may prove particularly helpful, for example, in the setting of a basal subpleural pulmonary nodule which may be technically challenging to biopsy percutaneously.
Comparatively higher radiation exposures when compared with conventional CT, cost of the study and the longer study duration are all factors which may be potentially limiting.
There is a growing body of evidence that novel radiotracers such as 68-Ga FAPI (Gallium-68 labelled fibroblast activation protein inhibitor) may be a superior radiotracer to use in the setting of peritoneal disease. A recent systematic review and meta-analysis comparing its use with 18F-FDG suggested that 68-Ga FAPI may be substantially superior in the detection of peritoneal disease and thereby helping to improve patient selection for surgery, although further studies are required to validate this interpretation. 5
Dual-Energy CT (DECT) and Spectral Photon-Counting CT (SPCCT)
The availability and experience with dual-energy CT continues to grow worldwide, although DECT scanners are not readily available in all radiology departments as of yet. Comparatively speaking, there is a reasonably small but growing body of evidence that DECT may augment the detection of peritoneal carcinomatosis and may allow detection of disease at earlier stages. A recent study has found the addition of material density images yields a higher contrast-to-noise ratio, leading to an earlier and an improved rate of detection of peritoneal carcinomatosis. 33
Although not yet widely available and with no large-cohort studies to confirm findings, early small-scale case studies have suggested a potential benefit of SPCCT to help improve the detection of peritoneal metastatic disease. This relies on the significantly improved spatial resolution of SPCCT to enable detection of smaller deposits. 34 Applicability to routine practice remains limited at present, however.
PET/MRI
The application of PET/MRI is broadly speaking limited to predominantly research capacity at present, although it is anticipated it may become increasingly used in clinical practice over the coming years. To date, evidence has been somewhat conflicting in terms of added benefit that PET/MRI can provide above conventional MRI for the assessment of peritoneal disease. However, while various workgroups generally find consensus that conventional MRI with DWI and PET/MRI compare well at global assessment of peritoneal disease, PET/MRI may offer additional benefit in assessing for the presence of small bowel disease. 35
Potential Imaging Pitfalls in the Post-Operative Period
In the immediate-early post-operative period, imaging may be challenging to interpret, particularly in the context of the patient with pyrexia of unknown origin or with general clinical deterioration. Organ inflammation under the peritonectomy is expected, typically affecting the stomach, ureters, and bladder which can show non-specific wall thickening and hyper-enhancement. Unless there is specific clinical, biochemical, or microbiological evidence suggesting organ-related sepsis in the acute setting, this should be considered a normal post-operative appearance.
Venous thrombus may also be encountered and although not considered “normal,” it is often expected, for example in the splenic or ovarian veins and is related to extensive vessel ligature. Infarcts can develop in the affected vascular bed as a result. There are a myriad of potential solid and hollow visceral as well as vascular complications which can arise as a result of cytoreductive surgery, although detailed discussion is beyond the scope of this paper.
Detection of residual disease in the immediate post-operative period can be challenging due to the extent of post-operative inflammatory change that may be seen. Communication with the operating surgeon and correlation with operative findings including the extent of cytoreduction achieved are important. Specific attention is required to areas where the surgeon felt complete cytoreduction was not achieved. Comparison across serial pre-operative studies may help to detect sites of residual disease by assessing for stability or progression in size and morphology at sites of concern. Mucinous implants, for example, can be easily confused for small volumes of non-organized ascites in the post-operative period if pre-operative imaging is not reviewed carefully. In the immediate post-operative period, it is expected to see extensive post-surgical changes including non-organized fluid, fat stranding, and mesenteric congestion. At surveillance studies, while it may take a number of months for these changes to fully resolve, local or metastatic recurrence must be questioned if these changes persist, fail to show signs of improvement, if there is clear progression or obvious new sites of disease. Taking extra-abdominal findings into account may also help to improve reader confidence in determining recurrent intra-abdominal disease. For example, if there is frank progression of intra-thoracic or supraclavicular metastatic disease, the index of suspicion for recurrent disease in the abdomen should be much higher. Generally speaking, colleagues in surgical, medical, and radiation oncologic disciplines appreciate that establishing a temporal pattern of findings is required in these cases to clarify any concerns for disease recurrence.
Conclusion
In summary, although there is a seemingly increasing choice of imaging modalities to consider as further evidence emerges, core concepts in the radiologic assessment of patients with peritoneal disease remain little changed over the years. Ultimately, the role of the radiologist as part of the multi-disciplinary team is to accurately and precisely describe the morphology and extent of disease, identify extra-peritoneal disease that should preclude surgery as well as identifying sites where the surgeon may potentially overlook at the time of attempted cytoreduction. The time investment by the radiologist in reading such studies is not insignificant, particularly in the context of more aggressive treatment strategies with improving clinical therapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
