Abstract
Purpose:
The precentral cortex normally demonstrates lower signal intensity compared to remainder of the neocortex on 2D fluid attenuated inversion recovery (FLAIR) images. Loss of this normal hypointensity bilaterally can be seen in patients with adult hypoxic-ischemic brain injury (HIBI). We have named this the ‘FLAIR motor’ sign (FMS). The performance of this sign for detection of HIBI is evaluated in this case-control study.
Methods:
MRI studies of 74 consecutive patients with clinical evidence of HIBI following cardiac arrest formed the ‘case’ group. Controls comprised of normal MRI studies of an equal number of age and gender matched patients. Two fellowship-trained neuro-radiologists reviewed the MRI studies in a blinded randomized fashion and recorded the presence or absence of ‘FLAIR motor’ sign.
Results:
Average time from cardiac arrest to MRI was 7.12 days (range: 1-25 days). The average sensitivity and specificity of ‘FLAIR motor’ sign for HIBI was 86.49% and 100% respectively. The sign demonstrated excellent inter-reader agreement (kappa >0.8).
Conclusion:
The loss of the normal hypointensity in bilateral pre-central cortex on 2D-FLAIR images is a specific and reliable MRI sign of HIBI in the subacute phase following cardiac arrest in adults.
Introduction
MRI is commonly performed for diagnosis and prognostication when adult hypoxic-ischemic brain injury (HIBI) is clinically suspected. HIBI is frequently characterized by restricted diffusion and high signal on T2-weighted and fluid attenuated inversion recovery (FLAIR) images. The cerebral cortex (in particular perirolandic and medial occipital cortex), deep gray structures, hippocampi, and cerebellum are commonly involved. 1 The nature of the hypoxic-ischemic insult and the timing of imaging relative to insult onset, affect the pattern of signal changes.1,2 Cortical involvement has been described as a poor prognostic predictor in HIBI, 3 emphasizing the need of identifying cortical involvement. Diffuse cortical and deep gray structure involvement can make cortical signal abnormality less conspicuous for the reader. 4 The MR signal characteristic of cortex, which typically exhibits higher signal than white matter on T2-weighted sequence, makes it difficult to identify cortices with slightly elevated T2 signal indicative of injury. Pseudo-normalization of ADC can also result in challenges in detecting abnormalities on DWI.5,6 Furthermore, lack of adequate clinical history in the setting of altered level of consciousness can create diagnostic dilemmas. Therefore, there is a need to identify unique imaging finding which may aid in the diagnosis of HIBI.
The precentral cortex or primary motor cortex displays a lower signal intensity on 2D fluid-attenuated inversion recovery (FLAIR) images compared to other regions of the neocortex 7 (Figure 1A). Our study is based on the hypothesis that loss of normal hypointensity of the pre-central cortex bilaterally on 2D-FLAIR images is a useful radiological sign of HIBI. The purpose of our study is to evaluate the performance of this ‘FLAIR motor sign’ (FMS) in detecting global hypoxic-ischemic brain injury.
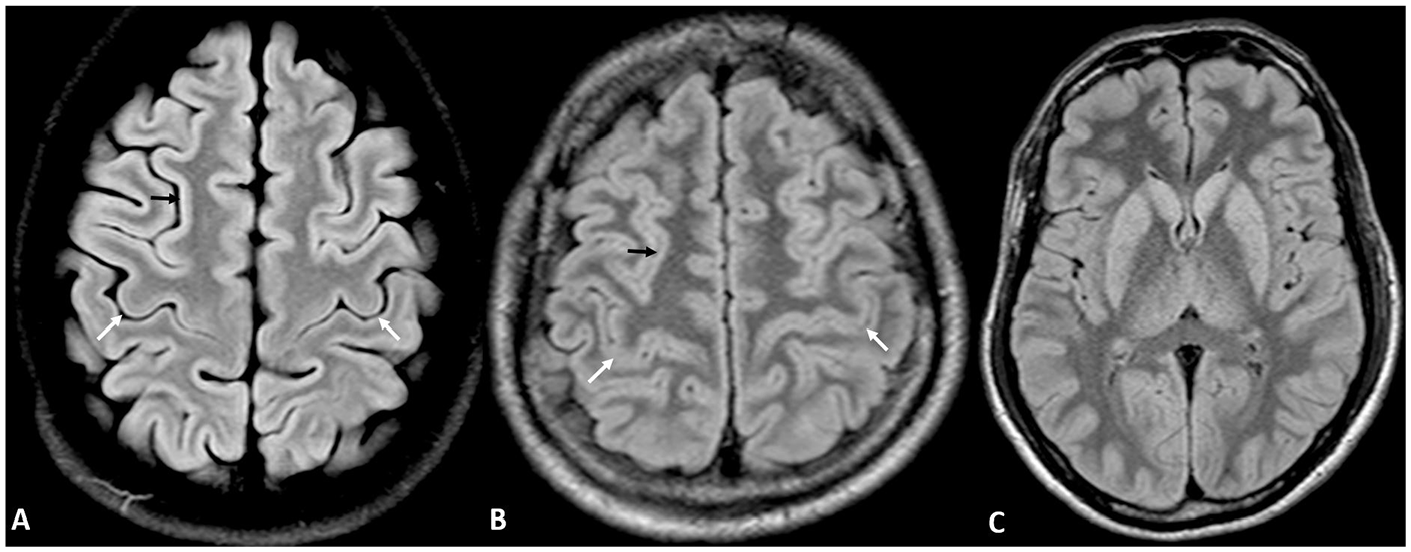
(A) 2D FLAIR image showing normal hypointensity of bilateral precentral cortex (white arrows) in relation to superior frontal cortex (black arrow) in a 20 year old male. (B) Loss of normal FLAIR hypointensity in bilateral precentral cortex (white arrows) in relation to superior frontal cortex (black arrow) consistent with positive ‘FLAIR motor sign’ in a 47 year old male with hypoxic ischemic brain injury post cardiac arrest. (C) 2D FLAIR image of the same patient at the level of basal ganglia demonstrates diffuse involvement of the cortex and deep gray structures highlighting the utility of the sign in diffusely abnormal or ‘supernormal’ scan.
Materials and Methods
Institutional review board approval was obtained for this case control study.
Patient Cohort
A retrospective review of imaging records was carried out to identify 108 consecutive patients who underwent an MRI for clinical suspicion of HIBI following a cardiac arrest at our institution from February 2005 to September 2020. Out of these, based on review of clinical and imaging records, 34 patients who were subsequently found to have no clinical or imaging evidence of HIBI were excluded. Patients with unclear history of cardiac arrest associated with HIBI were not included. The remainder 74 patients formed the ‘case’ group. Time between cardiac arrest and MRI was recorded and cerebral performance category 8 was also determined based on chart review.
For the ‘control’ group, consecutive age and gender matched patients who had brain MRI for other indications (headache, dizziness) and had final reports of normal studies were included.
Patients with history of substance use, on hyperbaric oxygen therapy, ALS, uncontrolled seizures or status epilepticus, and on medications known to cause encephalopathy (eg, cyclophosphamide) were excluded from the study.
Imaging Technique
MRI scans for both groups were acquired using an institutional MRI brain protocol on a 1.5 T Philips Achieva (Philips Healthcare, Best, the Netherlands) or a 3 T Siemens Skyra (Siemens Healthineers, Erlangen, Germany). All studies included sagittal T1 and axial T2, FLAIR and DWI (diffusion weighted imaging) sequences. On the 1.5 T scanners, the two-dimensional acquisition for FLAIR sequence was performed with 232 × 141 matrix (0.86 × 1.26 × 5 mm3 acquired voxel size, 0.45 × 0.45 × 5 mm3), 5-mm section thickness, TR = 11 000 ms, TE = 140 ms, TI = 2800 ms, flip angle = 90°, NEX = 1, and section gap = 0.5 mm. On the 3 T scanners, the two-dimensional acquisition for FLAIR sequence was performed with 320 × 224 matrix (0.89 × 0.63 × 5 mm3 acquired voxel size, 0.31 × 0.31 × 5 mm3), 5-mm section thickness, TR = 9000 ms, TE = 81 ms, TI = 2500 ms, flip angle = 150°, NEX = 1, and section gap = 0.5 mm.
Imaging Review
MRI studies were reviewed on 2D-FLAIR images in a blinded randomized fashion by 2 fellowship-trained neuroradiologists and the FMS sign was recorded as positive or negative. The readers were blinded to each other’s observations and clinical history. The readers called the sign positive when the entire thickness of bilateral precentral cortex on the axial image at the level of the hand knob was equal or greater in intensity on 2D-FLAIR images compared to the internal reference (superior frontal cortex). The readers were allowed to use their judgment to take into account noise, volume averaging, motion, and other artifacts just like they would do in routine clinical practice. After completion of the initial set of observations, the studies with disagreements were reviewed again by the readers and consensus readings and reason of disagreement were noted.
Statistical Analysis
The measures of performance (sensitivity, specificity, accuracy, positive and negative predictive values) of the FLAIR motor sign for diagnosis of HIBI were calculated for the 2 observers. Cohen κ coefficient was calculated to measure inter-observer agreement. Values of κ of 0.21 to 0.4, 0.41 to 0.6, 0.61 to 0.8, and 0.81 to 1 were considered fair, moderate, substantial, and excellent agreement respectively. All statistical analyses were performed by using R, version 4.1.0 (R Foundation, http://www.r-project.org/).
Results
Average time from cardiac arrest to MRI was 7.12 days (range: 1-25 days). The distribution of cerebral performance category 1, 2, 3, 4, and 5 in the cases were 0% (n = 0), 6.76% (n = 5), 6.76% (n = 5), 9.46% (n = 7), and 77.03% (n = 57) respectively. No significant age and gender difference was present between the cases (mean age 55.64 years, age range: 19-95 years, 22% females) and controls (mean age 56.43 years, age range: 20-89 years, 22% females).
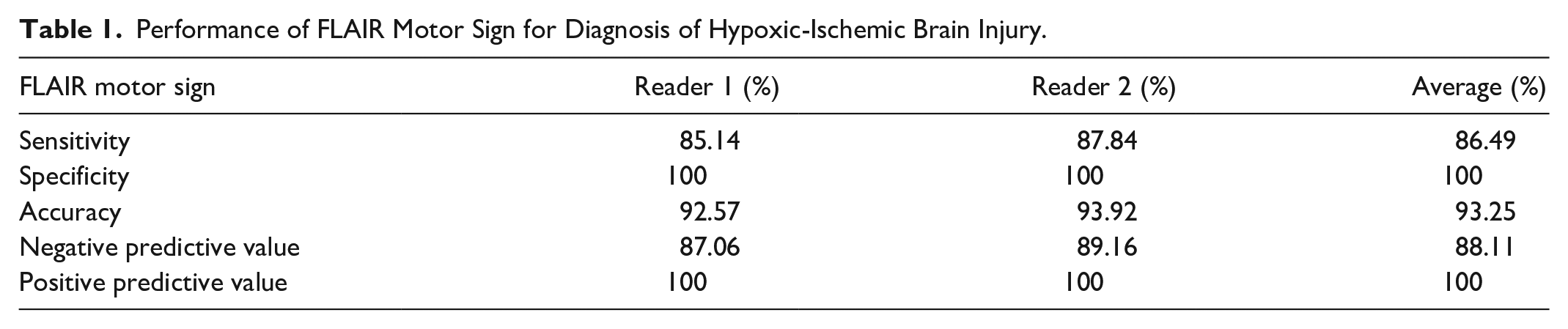
The diagnostic performance of the sign for HIBI is summarized in Table 1. Readers 1 and 2 reported the absence of the sign in 11 and 9 patients in the ‘cases’ group respectively (Figures 1B, C and 2). Both readers correctly noted absence of the sign in all 74 controls. The average sensitivity and specificity of the sign was 86.49% and 100% respectively.
Performance of FLAIR Motor Sign for Diagnosis of Hypoxic-Ischemic Brain Injury.
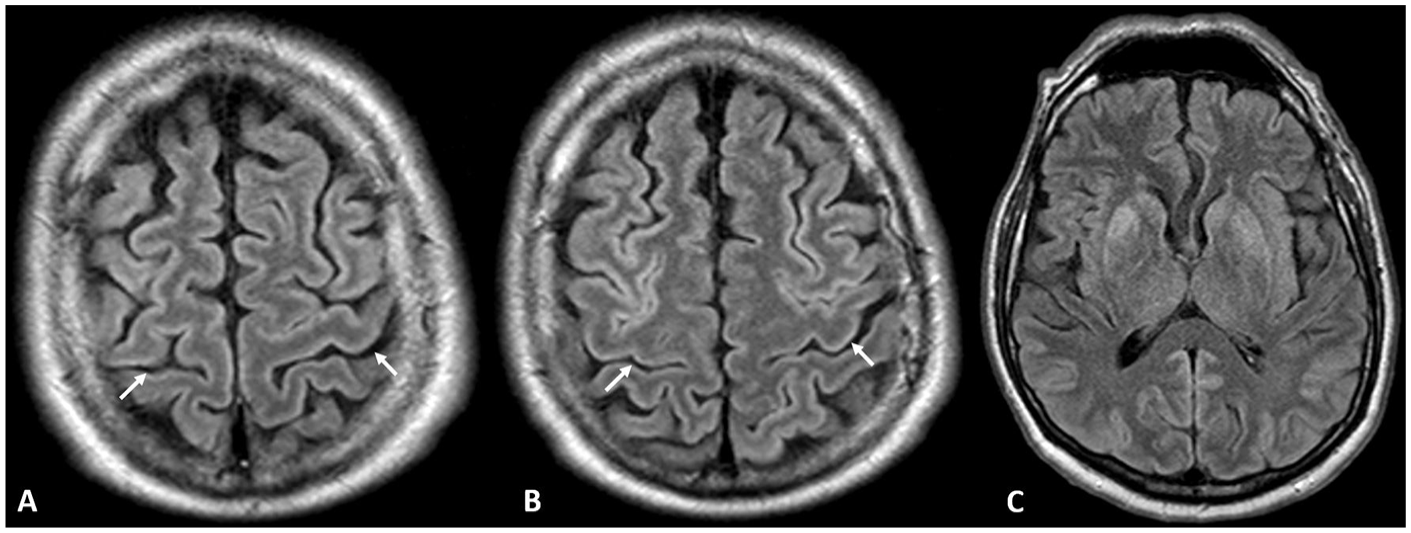
(A) Fifty year old male with history of cardiac arrest and clinical features of hypoxic ischemic brain injury. Preserved normal hypointensity in precentral cortex (white arrows) consistent with negative ‘FLAIR motor sign’. (A, B) There is patchy FLAIR hyperintensity in the remainder of the cerebral cortex and (B) basal ganglia.
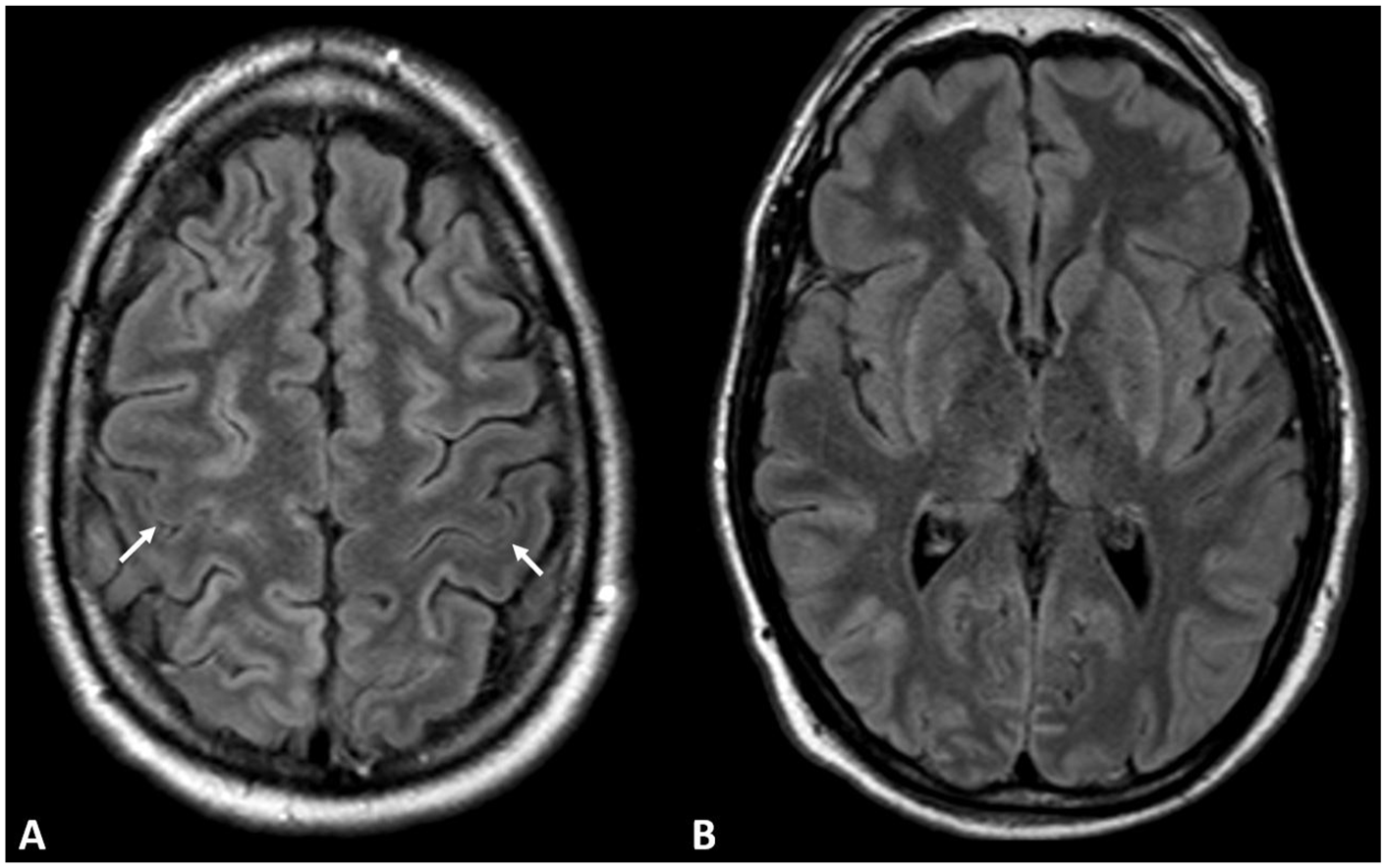
The sign demonstrated excellent inter-reader agreement (kappa >0.8). Disagreement amongst the readers occurred in 2 cases, where patchy involvement of the precentral cortex was felt to be the cause by the 2 readers (Figure 3). In addition, in one of these cases, artifacts were also suspected resulting in challenges with interpretation.
Sixty-seven year old male with hypoxic ischemic brain injury post cardiac arrest. FLAIR images show asymmetric loss of normal hypointensity in bilateral precentral cortex (white arrows), more noticeable on the superior slice (A) compared to the inferior slice (B) at the level of the hand knob. This was called positive ‘FLAIR motor sign’ by one of the readers and negative by the other reader. On the FLAIR image at the level of the basal ganglia, there is patchy involvement of the cerebral cortex and deep gray structures (C).
Discussion
The cerebral cortex (neocortex) consists of 6 layers: I—molecular, II—external granular, III—external pyramidal, IV—inner granular, V—internal pyramidal, VI—multiform. 9 The cerebral neocortex has also been divided into 5 types according to Von Economo and Kosinkas: Granular, agranular, frontal, parietal, and polar. 10 The primary motor cortex of the human brain is unique in that it is thick despite high myelin content and has the unique Betz cells.11,12 Although previously characterized as agranular, the primary motor cortex has been recently categorized as Eulaminate III. 13
The primary motor cortex has been described by Kim et al using double inversion recovery sequences at 3 T as having a ‘triple-layer’ appearance made up of an inner hyperintense layer, a middle hypointense layer, and an outer hyperintense layer. 14 The central hypointense line of the triple-layer appearance corresponds with myelin bands on a myelin-stained brain specimen which would correspond with the high myelin content of the primary motor cortex.
Additional putative explanations for this appearance have been proposed. Properties of the extracellular matrix and the amount of perineuronal nets have been proposed to account for the difference owing to abundance of these complexes in primary motor cortex compared to other areas such as prefrontal cortex.7,15 Magnetic susceptibility and resulting T2 shortening due to of iron deposition as a normal aging process in the primary motor cortex may also contribute to the low signal intensity. However, magnetic susceptibility has limited effect on the FLAIR sequence and this does not explain why normal younger individuals also demonstrate this low signal intensity.7, 16
The absence of this sign in multiple patients in our study cohort is expected as perirolandic sparing is known to occur in HIBI. Muttikkal and Wintermark found diffuse cortical involvement with perirolandic sparing in 8% of patients with cardiac arrest and 10.94% of all the patients in their study. 1 The 2 instances of inter-reader disagreement in our study due to patchy involvement of precentral cortex may suggest a spectrum of involvement of the precentral cortex.
The FLAIR motor sign could be particularly useful when there is diffuse cortical and deep gray structure involvement (so called ‘supernormal’ scan) and also in the presence of less common MRI pattern of HIBI (such as paucity of cortical involvement; Figure 4). Although all the patients in our study had known history of cardiac arrest, this sign could prove especially useful when clinical history is limited, such as in ‘found down’ patients.
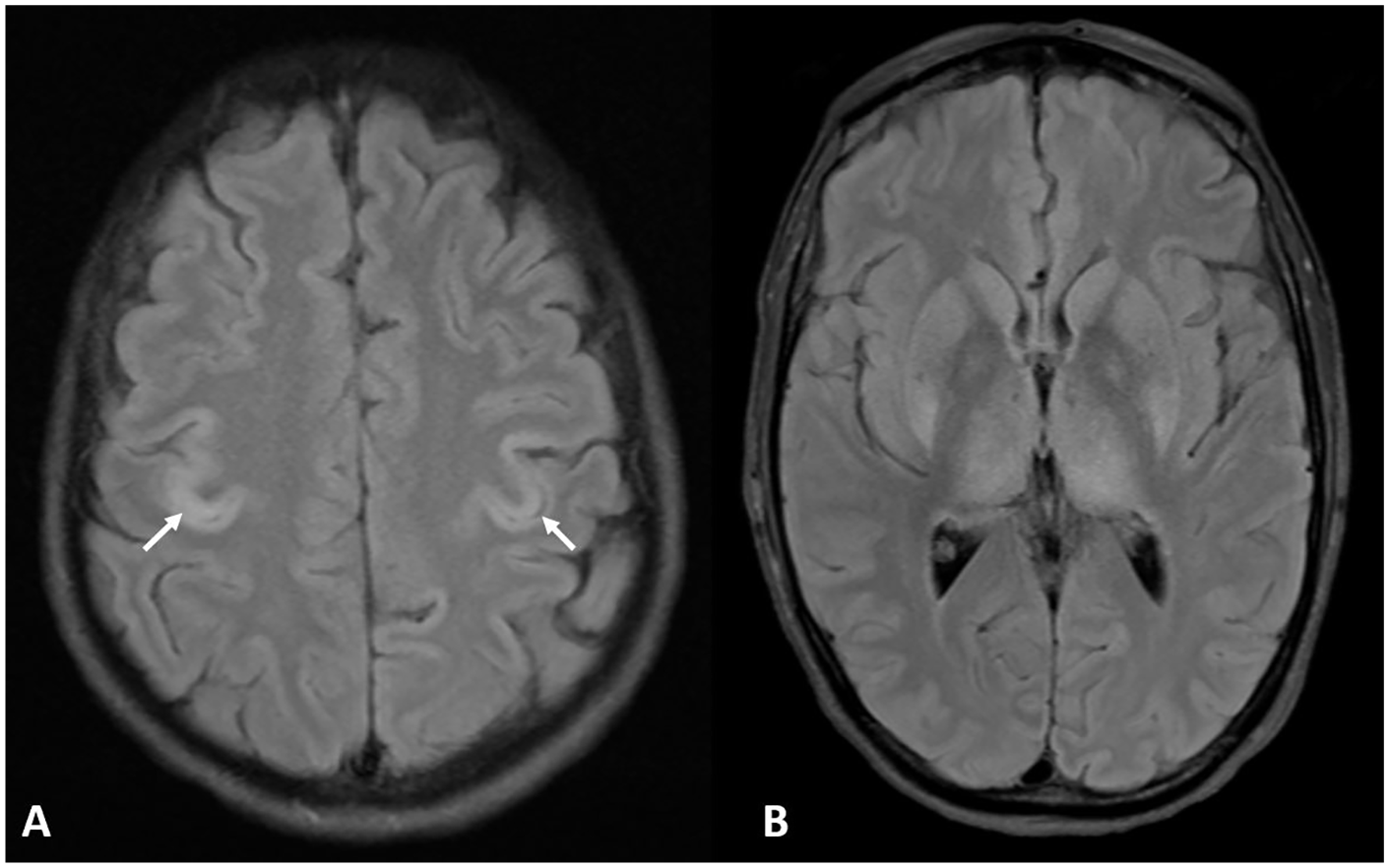
(A) A 43 year old male unresponsive post cardiac arrest. FLAIR hyperintensity in bilateral precentral cortex (white arrows) suggests hypoxic ischemic brain injury despite paucity of cortical involvement. (B) It also supports hypoxic-ischemic cause for hyperintensity in bilateral thalami and posterior putamina, which in isolation could suggest raise other diagnostic possibilities such as atypical PRES.
Our study had limitations. The FMS sign is limited to the 2D FLAIR sequence. We have anecdotally observed that the sign is less obvious on 3D FLAIR sequence. The study is limited by small sample size and retrospective single center design. The study was limited to the subacute period post arrest, as no scans were performed in the acute (<24 hours) period. Controls included MRI studies done on clinically symptomatic patients. Although no major change for the 2D FLAIR sequence was found in our records, some optimization and minor changes in parameters may have occurred during the long study period. We could not statistically correlate the sign with cerebral performance category due to small number in most of the categories. The study is also limited by contextual bias, as the readers may have been influenced by the presence of other features of HIBI in the imaging studies, potentially leading to an artificially increased diagnostic accuracy. However, we chose to present the entire image stack rather than selected images in order to replicate routine clinical practice as closely as possible.
Conclusion
The loss of the normal hypointensity in bilateral precentral cortex on the 2D-FLAIR images is a specific and reliable MRI indicator of post cardiac arrest subacute global hypoxic-ischemic brain injury in adults.
Footnotes
Acknowledgements
The authors express their sincere gratitude to Dr. Lyne Noel de Tilly for teaching them the sign described in this paper.
Author Note
Presented in part at the Canadian Association of Radiologists (CAR) 2023 Annual Meeting. Won the best (1st place) scientific research project award at this meeting.
Ethical Considerations
Institutional research ethics board approval was obtained.
Consent to Participate
Waived for this retrospective study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
