Abstract
Prostate cancer (PCa) remains a leading cause of cancer-related morbidity and mortality among men worldwide. Multiparametric MRI (mpMRI) is currently the gold standard for PCa detection, diagnosis, and active surveillance. However, its reliance on dynamic contrast-enhanced (DCE) imaging introduces safety concerns, higher costs, and longer scan times. Biparametric MRI (bpMRI), which omits DCE, has emerged as a streamlined alternative that retains T2-weighted and diffusion-weighted imaging. This review critically examines the technical considerations, diagnostic performance, clinical applications, and limitations of bpMRI compared to mpMRI. We evaluate bpMRI’s sensitivity, specificity, and negative predictive value in detecting clinically significant prostate cancer (csPCa), highlighting its advantages in terms of patient safety, accessibility, and cost-effectiveness. Despite promising findings, the widespread clinical adoption of bpMRI is hindered by variability in imaging protocols, limited large-scale validation, and concerns over missed subtle lesions. Future directions include standardizing bpMRI protocols, integrating artificial intelligence and biomarkers, and conducting multi-centre trials to establish its role in PCa management. bpMRI holds significant potential as a reliable and efficient imaging tool that could complement or replace mpMRI in select clinical contexts.
Introduction
Prostate cancer (PCa) is the second most frequently diagnosed cancer and the fifth leading cause of death in men. 1 Multiparametric MRI (mpMRI) has emerged as the gold standard imaging modality for PCa detection, diagnosis, loco-regional staging, and surveillance. 2 MpMRI combines T2-weighted sequences (T2W) with diffusion-weighted imaging (DWI) and dynamic contrast-enhanced sequences (DCE) to optimize tumour detection. However, mpMRI requires the administration of gadolinium-based contrast agents, which have been linked to nephrotoxicity and other adverse reactions and result in longer scan times and increased costs. 3
Biparametric MRI (bpMRI) has been proposed as a streamlined alternative to mpMRI. BpMRI includes the same T2W, and DWI sequences used in mpMRI but omits DCE. BpMRI has several advantages over mpMRI including reduced scan time (20-30 minutes per case compared to 45-60 minutes for mpMRI), lower costs, and less patient discomfort. Studies have shown that bpMRI maintains good sensitivity and specificity in detecting clinically significant prostate cancer (csPCa), making it a promising alternative to mpMRI. 4
The implementation of bpMRI in clinical practice has certain challenges. Current studies on bpMRI efficacy are often limited by small sample sizes, retrospective designs, and variability in imaging quality. Most research relies on the Prostate Imaging Reporting and Data System (PI-RADS) v2.1 without strict radiologist blinding and inconsistency in MRI quality control protocols. Furthermore, many MR-fusion prostate biopsy protocols continue to depend on mpMRI findings, which may introduce bias when assessing the true effectiveness of bpMRI. 5
In this review, we aim to explore the comparative effectiveness of bpMRI and mpMRI. By evaluating diagnostic performance, clinical feasibility, and potential integration into prostate cancer management, we aim to determine whether bpMRI can serve as a reliable, more accessible, and cost-effective alternative to mpMRI.
Technical Considerations
T2-Weighted Imaging Parameters
T2-weighted imaging (T2W) is a fundamental component of bpMRI and plays a crucial role in prostate cancer detection. T2W sequences enhance visualization of prostate anatomy by depicting tumour foci as hypointense areas against the hyperintense background of normal glandular tissue. This contrast enables accurate lesion localization and characterization. User-selectable parameters such as Echo Time (TE), Repetition Time (TR), and Flip Angle can be adjusted to optimize image quality and lesion detection.
Diffusion-Weighted Imaging (DWI) Parameters
DWI provides functional information about prostate tissue by assessing water molecule movement within the gland. The effectiveness of DWI is influenced by key parameters such as b-values and apparent diffusion coefficient (ADC) calculations. Restricted diffusion, characterized by low ADC values, is a hallmark of csPCa, making DWI a crucial component of bpMRI.
Variations in Acquisition Techniques and Sequences
Variations in imaging acquisition techniques can impact the diagnostic performance of bpMRI. Differences in scanner hardware, protocol standardization, and sequence optimization influence image quality and lesion detectability. Advanced acquisition strategies, including optimized gradient strengths and high spatial resolution imaging, are continually being explored to refine bpMRI effectiveness. 6
Image Interpretation and Reporting
PI-RADS v2.1 serves as the standardized framework for interpreting prostate MRI, including bpMRI. Although PI-RADS was originally developed for mpMRI, adaptations for bpMRI are being evaluated. The omission of DCE imaging in bpMRI necessitates modifications in lesion scoring, with growing research indicating the ability of PI-RADS to maintain high diagnostic performance. 7
Importance of Standardized Reporting
Standardized reporting ensures consistency in prostate MRI interpretation. Using uniform PI-RADS category cutoffs allows bpMRI readings to be comparable to mpMRI findings. This standardization facilitates communication among radiologists, urologists, and oncologists and improves clinical decision-making. 8
Hardware and Software Considerations
The strength of the MRI magnet significantly influences image quality and resolution. Higher field strengths, such as 3 Tesla (3 T), are preferred due to higher signal-to-noise ratios compared to 1.5 Tesla (1.5 T) systems. Ultra-high-field MRI systems, like 7 Tesla (7 T), are being explored for their potential to provide even greater detail, though their clinical application is currently limited. 9
Advancements in MRI software, including the integration of artificial intelligence (AI), have shown promise in standardizing image acquisition, interpretation, and reporting. AI-assisted tools can enhance diagnostic accuracy, reduce inter-reader variability, and streamline workflow in prostate MRI evaluations. 10
Diagnostic Performance
Several studies have compared bpMRI to mpMRI and histopathology, offering insights into the reliability and validity of bpMRI for risk stratification, biopsy decision-making, and active surveillance.
Sensitivity, Specificity, and Accuracy
The diagnostic accuracy of bpMRI has been assessed by comparing its performance to mpMRI and histopathology results. Studies have reported that bpMRI achieves sensitivity ranging from 80% to 95% for detecting csPCa, with specificity ranging from 70% to 90%.7,11,12 While these values are slightly lower than those of mpMRI, using a histopathologic Gleason >6 the sensitivity and specificity of bpMRI have been found to be 92% and 48%, respectively. 13
Several meta-analyses have demonstrated that while the absence of DCE in bpMRI results in a minor reduction in sensitivity, the specificity remains comparable to mpMRI.4,10 The clinical trade-off is that bpMRI reduces scan time and cost while maintaining sufficient accuracy for csPCa detection.
Factors Influencing Diagnostic Performance
Multiple factors impact the diagnostic performance of bpMRI:
Field Strength: Higher magnetic field strengths (3 T vs 1.5 T) improve image resolution and lesion conspicuity, enhancing diagnostic confidence. 14
Radiologist Experience: More experienced radiologists achieve higher sensitivity and specificity in interpreting bpMRI. 15
Use of AI and Automated Analysis: Emerging artificial intelligence (AI)-based tools help improve consistency in lesion detection and PI-RADS scoring. 16
Detection of Clinically Significant Prostate Cancer (csPCa)
The primary goal of prostate MRI is to differentiate between indolent and clinically significant cancers (Gleason score >6). The literature suggests that bpMRI retrains a strong ability to detect high-grade tumours (Gleason score >6), with detection rates closely mirroring those of mpMRI. 6 A systematic review reported that although bpMRI and mpMRI are comparable in terms of overall performance, bpMRI has increased specificity 17 and DCE only marginally improves the identification of smaller, less conspicuous lesions. The PI-RADS v2.1 system has been adapted to bpMRI interpretations, ensuring that lesion classification remains standardized. 18 Additionally, bpMRI demonstrates fewer false-positive results, reducing unnecessary biopsies (Figures 1 and 2). 6
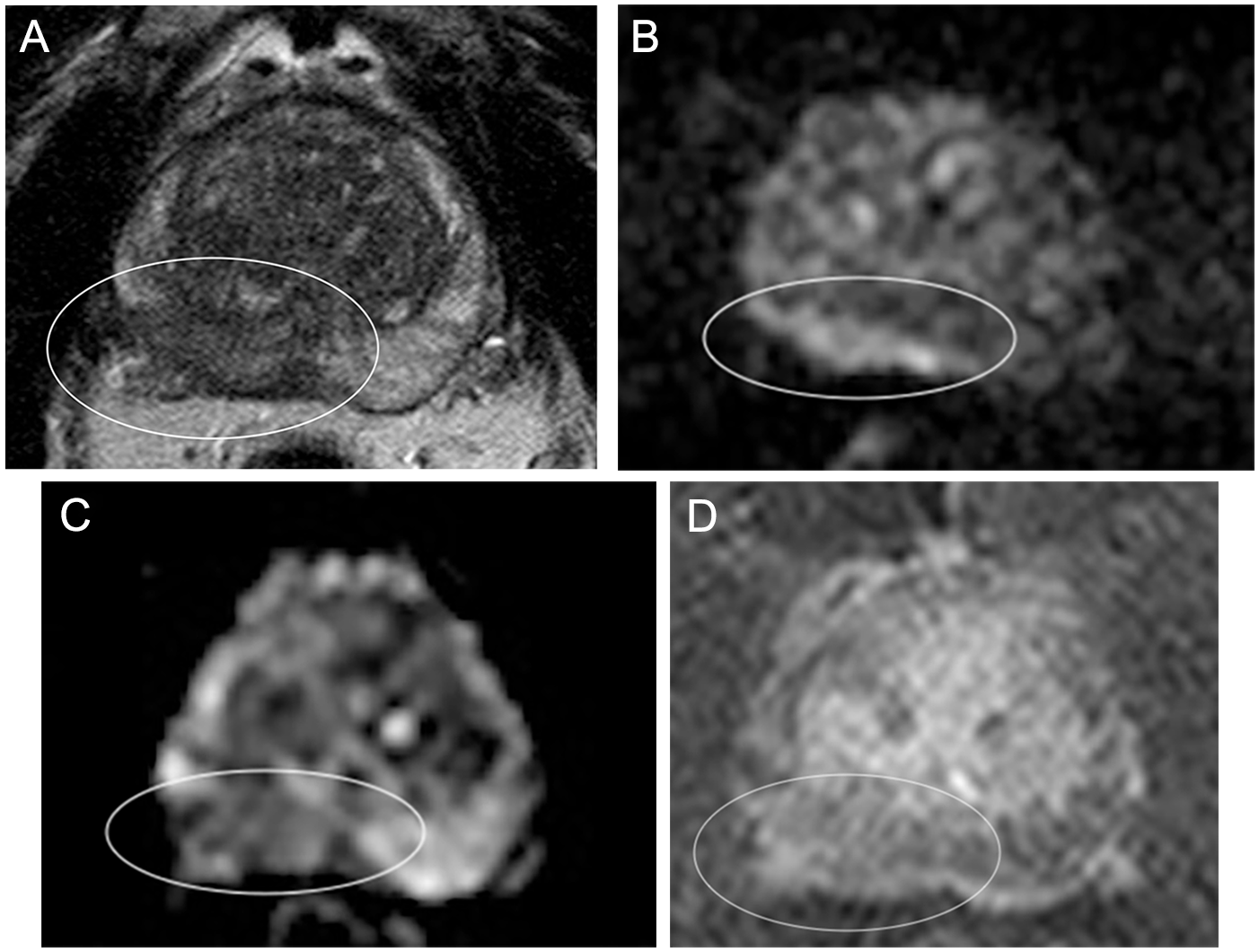
Sixty-five-year-old: (A) Axial T2W image shows a large area of abnormal low signal measuring approximately 3.7 cm within the right peripheral zone with extraprostatic tumour extension and possible involvement of the right neurovascular bundle (white circle). High signal is seen on the DWI image (B) and low signal is seen on the ADC map (C) in keeping with restricted diffusion in the right peripheral zone (white circles). The findings are in keeping with PIRADS 5 disease. The DCE image (D) shows minimal equivocal early enhancement and offers no additional information to contribute to the final diagnosis (white circle).
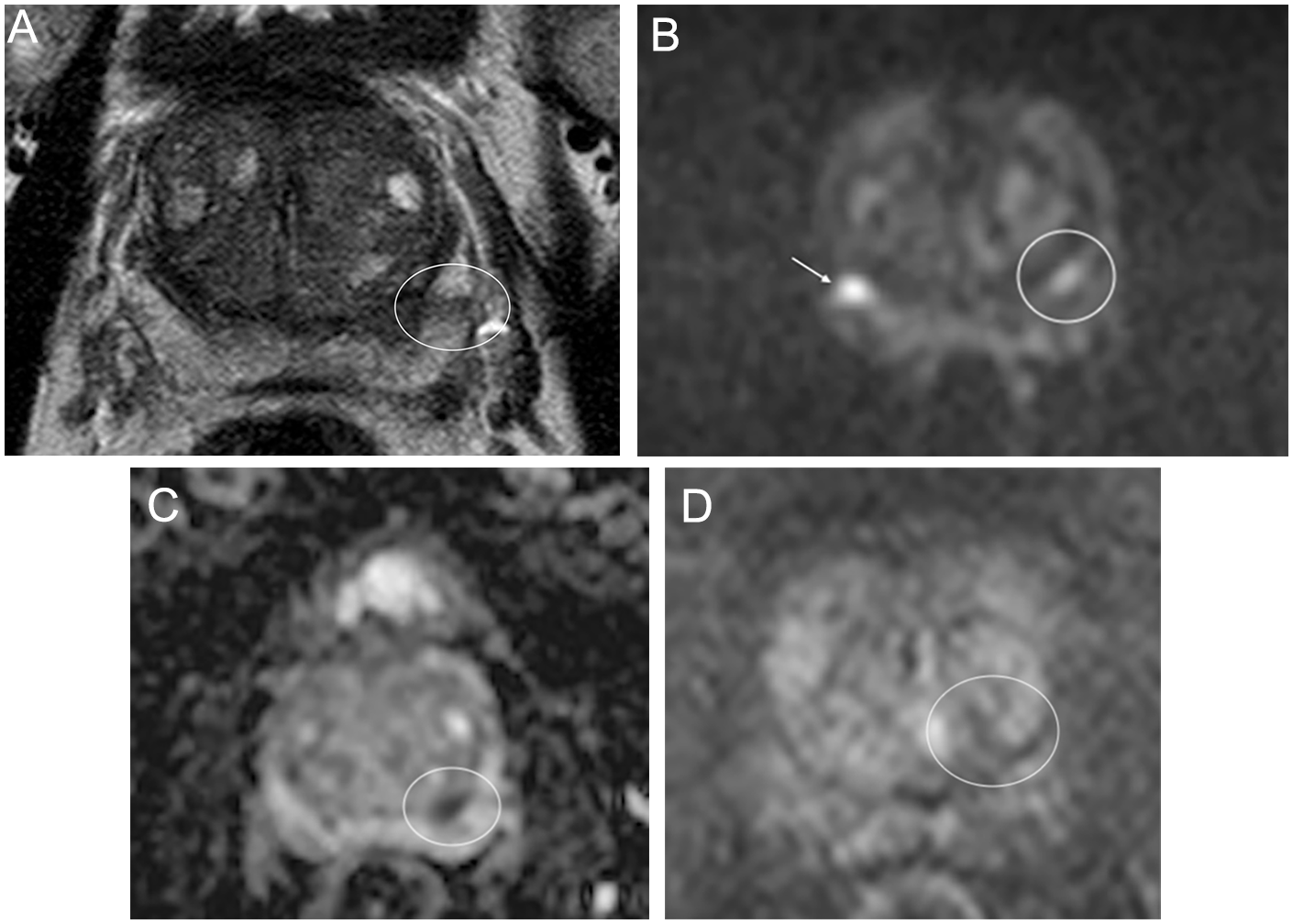
Fifty-eight-year-old: (A) Axial T2W image shows a 9 mm area of abnormal low signal within the left peripheral zone (white circle). High signal is seen on the DWI image (B) and low signal is seen on the ADC map (C) in keeping with restricted diffusion in the left peripheral zone (white circles). The findings are in keeping with PIRADS 4 disease. The DCE image (D) shows no early enhancement and offers no additional information to contribute to the final diagnosis (white circle). Incidentally, the DWI image (B) shows an area of high signal within the right peripheral zone with no associated signal changes on the ADC map or T2W images, likely artifactual (white arrow).
Risk Stratification and Patient Selection
Role of bpMRI in Identifying Patients Who Require Biopsy
BpMRI plays a critical role in stratifying patients for the purposes of biopsy. Patients with PI-RADS 4 or 5 lesions on bpMRI warrant biopsy. In contrast, PI-RADS 1 or 2 lesions are associated with a low likelihood of cancer and allow patients to avoid unnecessary biopsy. 19
Use of bpMRI in Active Surveillance
For men on active surveillance, bpMRI provides a non-invasive means to monitor disease progression. Studies indicate that bpMRI can reliably track tumour changes over time, helping to identify when a biopsy or treatment escalation is necessary. 20
Negative Predictive Value (NPV) of bpMRI
The negative predictive value (NPV) of bpMRI is crucial in determining whether patients can safely forgo biopsy. BpMRI has a reported NPV of 88% to 95% for ruling out csPCa, meaning that when no suspicious lesions are detected, the likelihood of missing a clinically significant tumour is low. 21 This high NPV supports the role of bpMRI in reducing unnecessary interventions and improving patient management.
Advantages and Limitations
Advantages of bpMRI
Reduced Acquisition Time and Cost
BpMRI significantly shortens scan duration compared to mpMRI. Standard mpMRI requires 45 to 60 minutes depending on the scanner, whereas bpMRI can typically be completed in 20 to 30 minutes, or as little as 15 minutes 22 resulting in increased efficiency in high-volume centres. 22 Essentially, approximately 3 bpMRIs can be completed for every mpMRI, significantly improving patient access to these studies. 23
The removal of contrast-enhanced sequences further reduces costs, making MRI more accessible for routine screening and surveillance. 22 Porter et al found that when compared to mpMRI, the average bpMRI had a lower average reimbursement and gross profit of $558.61 and $510.44 USD, respectively. This lower cost equated to a 59% decrease in labour and material costs for the providers. 23
Increased Accessibility
Because bpMRI reduces both cost and scan time, it can be more widely implemented in healthcare systems with limited resources. In regions where access to prostate MRI is constrained due to financial or logistical reasons, bpMRI offers a feasible alternative. 15
Less Need for Contrast Agents
Unlike mpMRI, which requires the use of gadolinium-based contrast agents for DCE imaging, bpMRI relies only on T2W and DWI. This eliminates concerns regarding allergic reactions, risk of gadolinium deposition in the brain, and long-term nephrotoxic effects, making bpMRI a safer option for repeat imaging.24,25 This makes bpMRI particularly beneficial for patients with renal insufficiency, those who experience discomfort during contrast administration, and those who require frequent repeat imaging.
Limitations of bpMRI
Potential Loss of Diagnostic Accuracy Compared to mpMRI
While bpMRI performs well in detecting high-grade tumours, it may have reduced sensitivity for subtle or small lesions compared to mpMRI. 26 The absence of contrast may limit the ability to distinguish between aggressive and indolent tumours. Additionally, bpMRI relies on DWI sequences, which are susceptible to distortions and artifacts caused by motion, field inhomogeneity, and low signal-to-noise ratio (SNR). These artifacts may reduce image quality and diagnostic confidence, particularly in patients with large prostates or prior treatment.27,28
Variability in Protocols and Reporting
Differences in MRI scanners, acquisition protocols, and radiologist experience contribute to variability in bpMRI interpretations. Utilization of PI-RADS v2.1 aims to reduce inter-reader variability, but challenges remain in ensuring consistency across institutions. 29
Comparative Analysis: bpMRI Versus mpMRI
Direct Comparisons in Clinical Studies
Several clinical studies have sought to compare the diagnostic performance of bpMRI and mpMRI in the same patient cohorts to determine whether bpMRI could serve as a viable alternative to mpMRI while maintaining diagnostic accuracy. A meta-analysis by Bass et al 30 found that while mpMRI demonstrated slightly higher sensitivity in identifying csPCa, bpMRI maintained comparable specificity and reduced the rate of unnecessary biopsies. Another systematic review 31 analyzed 14 studies and concluded that bpMRI had a sensitivity of 85% and specificity of 91%, compared to 88% sensitivity and 89% specificity of mpMRI. These findings suggest that while mpMRI may detect a slightly higher number of csPCa cases, bpMRI still provides robust diagnostic performance, particularly in patients where contrast administration is contraindicated.
Kuhl et al 27 assessed the efficacy of bpMRI protocols in men with elevated PSA levels and found that bpMRI can detect clinically significant lesions with an accuracy similar to mpMRI, but with a significantly shorter imaging time. The ability of bpMRI to maintain strong diagnostic capabilities while streamlining the imaging process is a key advantage in clinical settings. These meta-analyses collectively suggest that bpMRI offers diagnostic performance comparable to mpMRI in detecting both PCa and csPCa, supporting its potential as a less invasive and more cost-effective imaging modality (Table 1). Additionally, the lack of gadolinium given to patients was found to have no significant effect on outcome, with bpMRI having comparable PI-RADS ⅗ scoring rates, specificity, sensitivity, and number of biopsies performed. 32
Meta-Analysis Comparison of bpMRI With mpMRI in Terms of Accuracy, Negative Predictive Value, and Average Sensitivity and Specificity for Both PCa and csPCa.
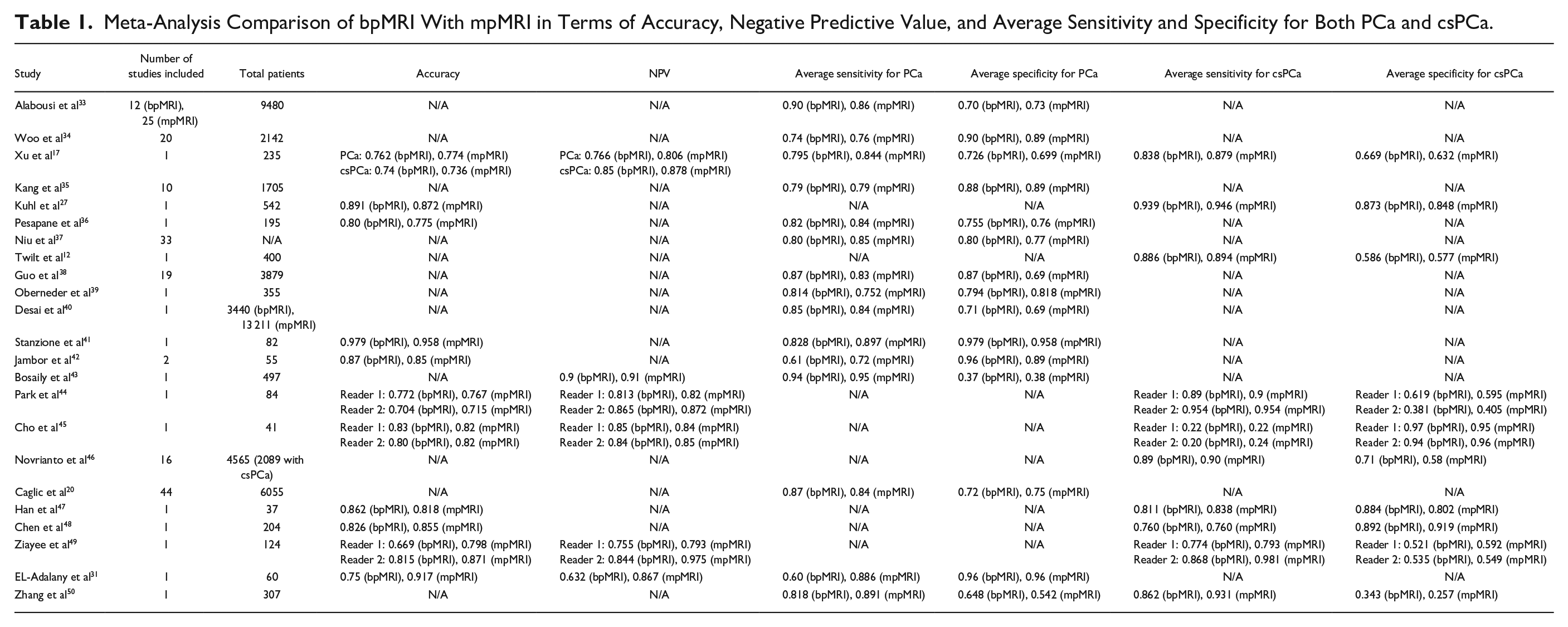
Despite these promising results, some limitations have been noted. One of the main concerns with bpMRI is its reduced ability to detect subtle lesions that may be more apparent with the use of DCE in mpMRI. This limitation could impact early detection of smaller, high-grade tumours, which is particularly relevant for patients undergoing active surveillance. 28
Analysis of Differences in Diagnostic Performance and Clinical Outcomes
The diagnostic accuracy of bpMRI and mpMRI varies based on factors such as field strength, radiologist experience, and standardized reporting criteria. Variability in the application of PI-RADS v2.1 for bpMRI may influence diagnostic performance, making it essential to refine interpretation guidelines specifically for biparametric imaging. 29
In terms of clinical outcomes, mpMRI remains the preferred imaging modality in high-risk cases where comprehensive lesion characterization is critical. However, in low- to intermediate-risk patients, bpMRI has shown promise as an effective screening tool that reduces unnecessary biopsies while maintaining a high NPV. 15 The ability of bpMRI to rule out non-clinically significant cancer while accurately detecting csPCa is crucial in reducing over treatment.
Cost-Effectiveness Analysis
Studies have consistently shown that bpMRI is more cost-effective than mpMRI due to its shorter acquisition time and the absence of contrast agents. According to a cost analysis by Belue et al, 14 bpMRI reduces overall imaging costs by approximately 30% to 40% compared to mpMRI. 51 The elimination of contrast administration not only reduces direct costs associated with gadolinium-based agents but also decreases the need for additional medical staff and monitoring.
Another economic analysis conducted by Turkbey et al 52 evaluated the cost-benefit of bpMRI versus mpMRI and found that the adoption of bpMRI in routine clinical practice could significantly reduce overall healthcare expenses without compromising diagnostic outcomes. By avoiding unnecessary biopsies and follow-up imaging, bpMRI contributes to cost savings while maintaining an acceptable level of diagnostic performance.
Impact on Healthcare Resource Utilization
The implementation of bpMRI has the potential to improve healthcare efficiency by reducing imaging time, increasing patient throughput, and minimizing resource allocation for contrast administration. In settings where MRI availability is limited, bpMRI offers a feasible alternative. A study by Stabile et al 15 highlighted that bpMRI could increase MRI accessibility for a broader patient population, particularly in community hospitals and lower-income regions where cost and resource constraints are significant barriers.
Additionally, reducing the need for contrast-enhanced imaging minimizes potential risks associated with gadolinium in patients with renal impairment. This is particularly beneficial in elderly populations, who are at higher risk for chronic kidney disease and other comorbidities. 52
Current Clinical Applications and Future Directions
Clinical Implementation of bpMRI
Although bpMRI has been increasingly integrated into clinical practice, implementation varies widely across institutions and healthcare systems.
Current Adoption Rates and Clinical Practice Guidelines
While mpMRI remains the gold standard for prostate imaging, bpMRI can serve as an effective alternative in the appropriate clinical settings such as low- to intermediate-risk patients. 53 Alternatively, mpMRI is more helpful in patients with indeterminate lesions and for loco-regional staging of csPCa. Moreover, while experienced readers perform well using both bpMRI and mpMRI, less experienced readers with <300 cases have been found to perform significantly worse in bpMRI. 54
Huebner et al noted that bpMRI is a viable solution in centres where contrast administration is not feasible or unnecessary. 55 Kortenbach et al assessed disease progression at confirmatory staging biopsies after 1 year of active surveillance. The study compared patients who underwent pre-biopsy bpMRI before their initial diagnostic transrectal ultrasonography-guided biopsies with those who did not. Results indicated that patients who had pre-biopsy bpMRI exhibited a reduced risk of reclassification and disease progression after 1 year, suggesting that pre-biopsy bpMRI enhances the selection process for men entering active surveillance programs. 56
Though these studies underscore the growing interest in integrating bpMRI into routine clinical workflows, particularly for active surveillance and pre-biopsy evaluations, they also emphasize the necessity for further validation through large-scale, multi-centre trials to confirm bpMRI’s efficacy compared to mpMRI.
As of March 2025, the European Association of Urology (EAU) and the American Urological Association (AUA) have not established definitive guidelines advocating for bpMRI as a first-line diagnostic tool for prostate cancer. However, emerging evidence supports its role in select patient populations. The AUA’s 2023 guideline on the early detection of prostate cancer emphasizes the use of mpMRI in specific clinical scenarios, such as prior to repeat biopsies. It does not, however, provide specific recommendations regarding the routine use of bpMRI. 57
Role in Screening, Diagnosis, and Active Surveillance
In the context of active surveillance, bpMRI plays a crucial role in monitoring disease progression. The Prostate Cancer Radiological Estimation of Change in Sequential Evaluation (PRECISE) recommendations underscore the importance of MRI in baseline assessments and ongoing evaluations during active surveillance. While current guidelines do not specify MRI protocols or frequency, the use of bpMRI aligns with efforts to minimize patient exposure to contrast agents without compromising the detection of clinically significant prostate cancer. 20
Emerging Techniques and Research
Artificial Intelligence and Machine Learning in bpMRI Interpretation
The integration of artificial intelligence (AI) and machine learning (ML) into bpMRI interpretation is a rapidly evolving field with the potential to enhance diagnostic accuracy and efficiency. AI-driven tools are being developed to assist radiologists in lesion detection, risk stratification, and automated PI-RADS scoring. These technologies aim to improve inter-reader agreement, reduce variability in interpretation, and enhance the diagnostic accuracy of bpMRI.
Recent studies have demonstrated promising results in this area. A study by Hosseinzadeh et al 58 showed that deep learning models trained on bpMRI datasets could match or even surpass the performance of experienced radiologists in detecting csPCa, highlighting its potential to assist in clinical decision-making. Furthermore, Debs et al evaluated a deep learning pipeline for csPCa detection using bpMRI. Their findings indicated that AI-assisted interpretation performed on par with radiologists, suggesting its potential to standardize readings and reduce inter-observer variability. 59
As AI-driven decision-support systems continue to advance, they may facilitate the broader adoption of bpMRI by addressing concerns regarding its diagnostic performance compared to mpMRI. However, further large-scale, multi-centre studies are necessary to validate these findings and integrate AI-based solutions into routine clinical practice.
Standardization of bpMRI Protocols and Reporting
One of the key challenges in the widespread adoption of bpMRI is the lack of standardized imaging protocols and reporting guidelines across institutions. Unlike mpMRI, which follows well-defined criteria under PI-RADS v2.1, bpMRI protocols vary in aspects such as b-value selection, DWI parameters, and interpretation criteria. This variability affects diagnostic consistency and complicates the comparison of results across studies and clinical settings. 60
To address these discrepancies, organizations like the European Society of Urogenital Radiology (ESUR) and the European Association of Urology Section of Urologic Imaging (ESUI) are working toward standardizing prostate MRI acquisition and interpretation. While current guidelines primarily focus on mpMRI, efforts are underway to refine recommendations that account for the growing use of bpMRI in clinical practice. 61
The establishment of uniform bpMRI protocols will be crucial for ensuring consistent diagnostic performance, facilitating large-scale multi-centre studies, and enabling broader clinical implementation. Future guideline updates may integrate bpMRI-specific recommendations to optimize its role in prostate cancer detection and management.
Combining bpMRI With Other Biomarkers
An emerging area of research focuses on combining bpMRI findings with prostate cancer biomarkers to enhance diagnostic accuracy. Prostate-specific antigen density (PSAD) has been identified as a valuable parameter for improving lesion characterization and risk stratification. A study published in the American Journal of Roentgenology demonstrated that PSAD could aid in distinguishing between benign and malignant lesions, particularly in biopsy-naïve men with negative bpMRI results. 62 Another study concluded that combining semi-quantitative and quantitative bpMRI parameters with PSAD enhances overall diagnostic performance. 63
In addition to PSAD, circulating tumour DNA (ctDNA) has gained attention as a promising biomarker, 64 with further studies needed to establish standardized protocols for incorporating it with bpMRI.
Future Research Needs
Despite the growing body of evidence supporting bpMRI, several critical areas require further investigation before it can be fully integrated into clinical practice as a standalone alternative to mpMRI.
Areas That Need Further Investigation
Large-Scale, Multi-Centre Trials—While preliminary studies suggest that bpMRI can effectively detect csPCa, large-scale, multi-centre trials are needed to validate its diagnostic accuracy across diverse patient populations. Standardized protocols must be implemented to ensure reproducibility and consistency in bpMRI performance across different institutions. The PRECISION trial 65 was a landmark multi-centre study that demonstrated the efficacy of mpMRI in pre-biopsy risk stratification for prostate cancer. This trial involved 500 men across 25 centres in 11 countries, highlighting the feasibility and impact of large-scale, collaborative research in this domain. A similar multi-centre study focusing on bpMRI could provide definitive evidence regarding its utility, cost-effectiveness, and clinical impact.
Standardization of Imaging Protocols—Unlike mpMRI, which follows well-defined PI-RADS v2.1 guidelines, bpMRI protocols remain inconsistent. Further research is needed to establish standardized acquisition parameters and interpretation criteria to enhance diagnostic reliability.
Longitudinal Studies on Active Surveillance—BpMRI has shown promise in active surveillance programs for prostate cancer, but long-term studies are required to determine its effectiveness in monitoring disease progression. Research should focus on whether bpMRI alone can reliably detect subtle changes in tumour characteristics over time compared to mpMRI.
Integration with Artificial Intelligence (AI) and Machine Learning—AI-driven diagnostic tools have demonstrated potential in assisting radiologists with lesion detection and risk stratification in bpMRI. Future research should explore the development and validation of AI-based decision support systems to enhance diagnostic accuracy, reduce inter-reader variability, and streamline imaging workflows.
Comparison with Emerging Biomarkers—The role of bpMRI in combination with prostate cancer biomarkers, such as PSAD, ctDNA, and genomic risk scores, requires further study. Research should investigate how integrating imaging with liquid biopsy and genetic profiling could improve early detection, risk stratification, and treatment planning.
Diagnostic Limitations and False Negatives—Some concerns remain regarding bpMRI’s ability to detect smaller or subtler lesions without dynamic contrast-enhanced (DCE) imaging. Additional research is needed to evaluate whether bpMRI alone can reliably identify all clinically significant tumours or whether certain patient subgroups may still require mpMRI for optimal detection.
By addressing these research gaps, bpMRI could become a more robust and reliable tool for prostate cancer detection, risk assessment, and disease monitoring, ultimately improving patient outcomes and clinical decision-making.
Conclusion
BpMRI has emerged as a promising alternative to mpMRI for PCa detection, risk stratification, and active surveillance. By eliminating the need for dynamic contrast-enhanced (DCE) imaging, bpMRI offers a safer, more accessible, and cost-effective imaging approach, particularly benefiting patients with contraindications to contrast agents. Studies have demonstrated that bpMRI achieves comparable sensitivity and specificity to mpMRI in detecting PCa, particularly when standardized reporting systems such as PI-RADS v2.1 are used. Additionally, its shorter acquisition time and reduced cost improve accessibility and workflow efficiency, making it an attractive option for widespread clinical implementation.
Emerging research suggests that bpMRI can be further enhanced through integration with biomarkers such as PSAD and circulating tumour DNA (ctDNA), potentially improving diagnostic accuracy, and enabling personalized treatment strategies. Furthermore, advancements in AI and machine learning offer significant promise for enhancing the consistency and reliability of bpMRI interpretation, reducing inter-reader variability, and streamlining diagnostic workflows.
Despite these promising developments, challenges remain before bpMRI can be widely adopted as a standalone alternative to mpMRI. Standardization of imaging protocols and reporting criteria and validation through large-scale multi-centre trials are crucial next steps. While bpMRI has demonstrated high negative predictive value (NPV), concerns persist regarding its ability to detect smaller or more subtle lesions compared to mpMRI, which may impact risk stratification and treatment planning in certain patient populations. Variability in acquisition protocols, radiologist expertise, and scanner field strength further contribute to the need for continued refinement of bpMRI techniques.
BpMRI has the potential to become a cost-effective, widely accessible imaging modality, complementing or even replacing mpMRI in select clinical settings. Its ongoing development will be instrumental in improving prostate cancer diagnosis, reducing unnecessary biopsies, and enhancing patient outcomes worldwide.
Abbreviations
PCa: Prostate cancer
mpMRI: Multiparametric MRI
MRI: Magnetic Resonance Imaging
T2W: T2-weighted imaging
DWI: Diffusion-weighted imaging
DCE: Dynamic contrast-enhanced sequences
bpMRI: Biparametric MRI
csPCa: clinically significant prostate cancer
PI-RADS: Prostate Imaging Reporting and Data System
TE: Echo Time
TR: Repetition Time
ADC: Apparent Diffusion Coefficient
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
