Abstract
Theranostic represents a transformative approach in precision medicine, integrating diagnostic imaging with targeted radiopharmaceutical therapy to enhance individualized disease management. Rooted in nuclear medicine, this approach uses molecular targeting agents labelled with diagnostic radioisotopes for imaging and therapeutic radioisotopes for treatment, ensuring a seamless transition from diagnosis to therapy. The field has evolved significantly in the last decade, with prostate-specific membrane antigen (PSMA)-targeted radioligand therapy transforming prostate cancer patient management and somatostatin receptor (SSTR)-targeted agents revolutionizing neuroendocrine tumour (NET) treatment. Considering its interdisciplinary nature, collaboration between nuclear medicine specialists, oncologists, radiologists, and other healthcare professionals is critical to refining clinical applications and improving patient outcomes. As prominent members of the theranostic team, radiologists play a pivotal role, from patient selection and imaging-based eligibility assessment to response evaluation and long-term monitoring. In this regard, advanced imaging modalities facilitate the precise evaluation of disease characteristics, guiding treatment decisions. Hence, as theranostics becomes increasingly integrated into patient management, radiologists face the need to be well-versed in both the technical aspects and clinical implications. In this review, we aimed to provide a primer for radiologists to gain a general insight into the theranostic field and its basic principles.
Introduction
“Theranostic” is a term that emerged in the early 2000s, blending the words “
Today, the modern theranostic paradigm has gained significant traction with the development of new radiolabelled molecules to target various diseases, enabling therapeutic radiopharmaceuticals to precisely treat what is visualized on diagnostic imaging using the same molecular target. Current research focuses on improving targeting mechanisms, developing next-generation radiopharmaceuticals, and expanding theranostics into new indications. With its broad applicability, theranostics now plays a critical role in personalized medicine, where the goal is to move beyond a “one-size-fits-all” approach and deliver targeted, individualized treatments.
As a prominent example, radioiodine therapy revolutionized thyroid cancer management several decades ago. More recently, theranostic pathways have been transformed mainly by 2 indications. In prostate cancer, the introduction of Prostate-Specific Membrane Antigen (PSMA) agents has significantly improved both diagnosis and treatment. These agents can be labelled with isotopes such as 68Ga, 18F, or 99mTc (68Ga-/18F-/99mTc-PSMA) for diagnostic imaging and with 177Lu or 225Ac (177Lu-/225Ac-PSMA) for therapeutic purposes. 2 Similarly, in selected neuroendocrine tumours (NETs), radiopharmaceuticals such as 68Ga-DOTATATE (amongst other DOTA agents) and 177Lu-DOTATATE have proven to be invaluable counterparts for diagnosis and treatment, respectively. 3
Radiologists play a key role in theranostics, leveraging their expertise in a wide variety of therapy-relevant imaging modalities. In certain cases, interventional radiologists can also help with the radiotracer administration. As theranostic becomes increasingly integrated into patient management, radiologists face the need to be well-versed in both the technical aspects and clinical implications. Moreover, the interdisciplinary nature of this field requires close collaboration with nuclear medicine physicians, oncologists, and other specialists, such as urologists, for treating prostate cancer.
In this review, we provide a primer for radiologists, offering a general introduction to the field of theranostics and its basic principles. References and relevant terminology are included for those seeking further in-depth reading and exploration.
Principles of Theranostics
The hallmark of theranostics is its dual capability by developing radiopharmaceuticals for both diagnostic and therapeutic purposes, targeting the same specific molecules expressed by tumours. When labelled with
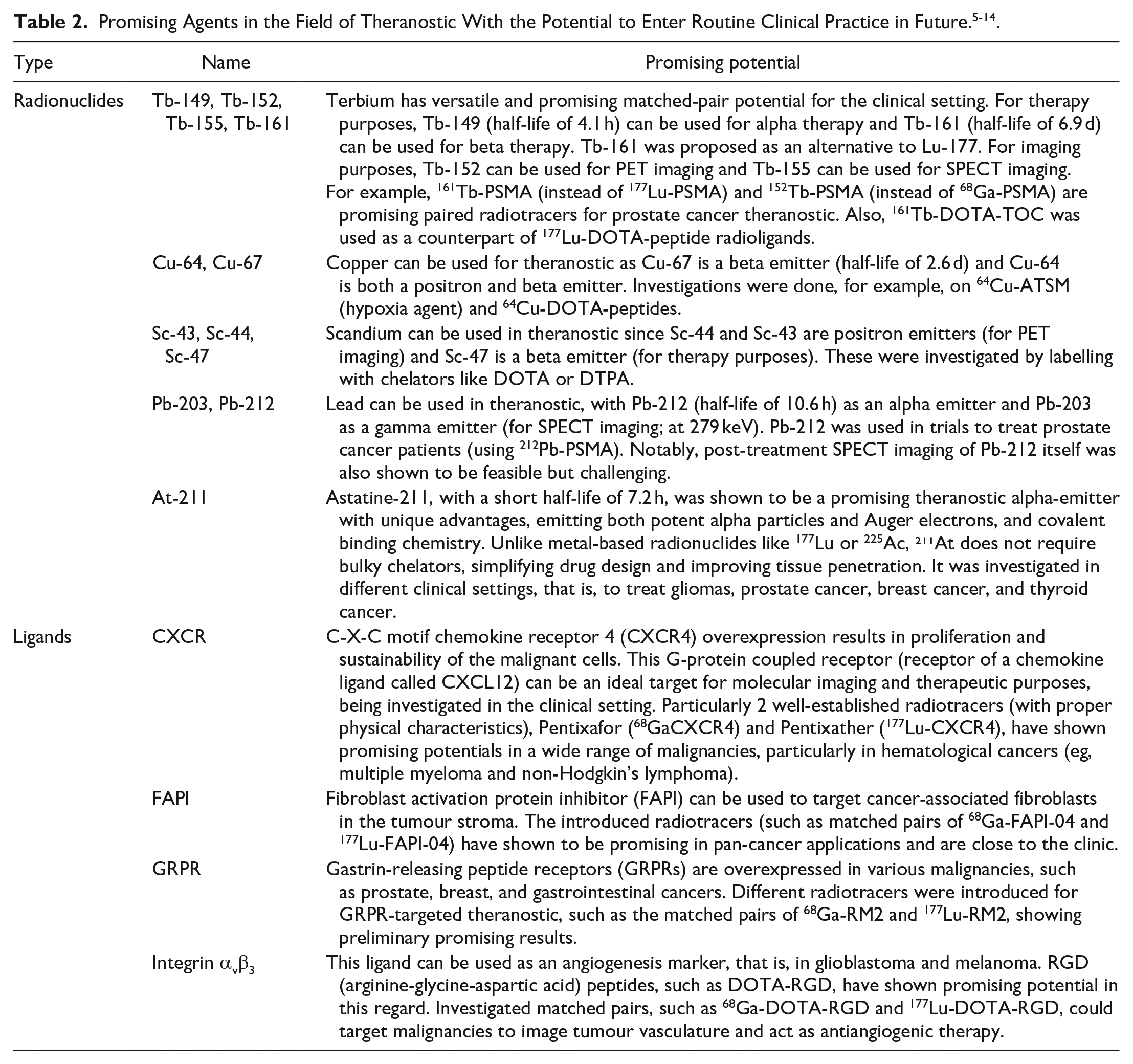
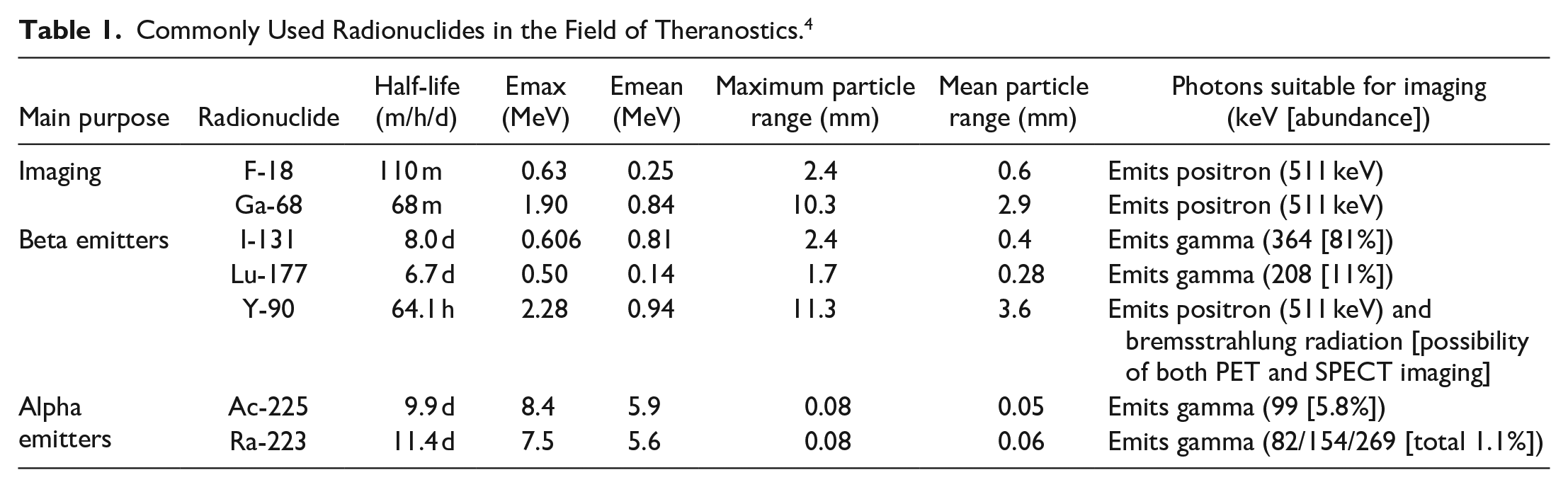
Theranostic agents typically function by binding to receptors or surface/transmembrane proteins that are overexpressed on tumour cells. Once administered, the diagnostic isotope emits positrons (as in the case of 18F or 68Ga) or gamma rays (as with 123I), which are detected by PET or single photon emission computed tomography (SPECT). Therapeutic isotopes bind to target expressed on tumour cells, and release beta (like 177Lu) or alpha (like 225Ac) particles once internalized, causing DNA damage and cell death. This mechanism of action aims to expand the therapeutic index by targeting delivery of radiation, and reducing toxicity to surrounding healthy tissue. Common isotopes used in the theranostic field are summarized in Table 1. Table 2 provides the agents (isotopes and ligands) that have not yet been in routine clinical use for theranostic, however, have shown promising potential in research studies and may enter the clinic in the future.
Commonly Used Radionuclides in the Field of Theranostics. 4
Currently, the diagnostic-therapeutic pairs of 68Ga/18F-177Lu are the most widely used in modern theranostics. 68Ga-labelled ligands are used in diagnostic PET imaging to identify tumours that express the molecular target, particularly in advanced prostate cancer with 68Ga-PSMA and neuroendocrine tumours with 68Ga-SSTRs (somatostatin receptors—such as DOTATATE and variants). After diagnostic PET identifies avid disease, 177Lu-labelled analogues of the ligand are administered to deliver therapeutic doses of radiation directly to the previously identified targets. 177Lu-labelled analogues can be further imaged post-therapy using SPECT. 15
Imaging Modalities
PET (Positron Emission Tomography)
PET works based on positron emissions detection after they collide with electrons and undergo annihilation. 16 It provides high-resolution, functional images that allow for a wide range of clinical applications. PET, nowadays, is combined with an anatomical imaging modality, CT (PET/CT), or less commonly MRI (PET/MRI), to provide both functional and anatomical information, enhancing diagnostic accuracy.
SPECT (Single Photon Emission Computed Tomography)
SPECT detects gamma rays emitted from radioisotopes. 17 Although SPECT generally provides lower resolution than PET, it remains essential in clinical theranostics, particularly for therapeutic monitoring. The gamma emissions from 131I and 177Lu, for instance, can be captured by SPECT to monitor the distribution of the therapeutic agent post-administration, providing important information about the effectiveness and safety of the treatment to ensure all preidentified targets received the therapy. Similar to PET, SPECT can also be combined with anatomical imaging, that is, in SPECT/CT.
Common Theranostic Targets
PSMA
PSMA is a transmembrane protein highly overexpressed in prostate cancer cells, particularly in aggressive metastatic, high-risk prostate adenocarcinoma. 18 It has become a central target in prostate cancer theranostics, with several ligands (eg, 68Ga-PSMA-11, 68Ga-PSMA-617, 18F-DCFPyL, 99mTc-PSMA-I&T, 18F-PSMA-1007) routinely used diagnostic PET imaging. The commonly used PSMA radioligands, such as 68Ga-PSMA-11, 68Ga-PSMA-617, and 18F-DCFPyL, exhibit almost identical biodistribution. These radiopharmaceuticals can be synthesized through automated methods or kit-based labelling. However, their production requires access to a germanium-68/gallium-68 (68Ge/68Ga) generator or a cyclotron, posing some logistical challenges. Additionally, the short half-life of gallium-68 (67.8 minutes) limits widespread distribution to other centres.
SSTR
NETs frequently overexpress SSTRs, particularly subtype 2 (SSTR2). 19 Radiolabelled somatostatin analogues, called DOTA agents in general, allow for both imaging and treatment of these tumours. DOTA (also known as tetraxetan) is linked to different molecules to create, that is, DOTA-TATE, DOTA-TOC, and DOTA-NOC ligands, which can be later labelled with a radioisotope to target SSTRs (different subtypes, ie, 2, 3, and 5).
How Theranostics Works in Practice
The successful implementation of theranostics in clinical practice requires a well-coordinated workflow that integrates diagnostic, therapeutic, and follow-up stages, often involving a multidisciplinary team of specialists. This section outlines the key steps in the theranostic workflow (Figure 1).
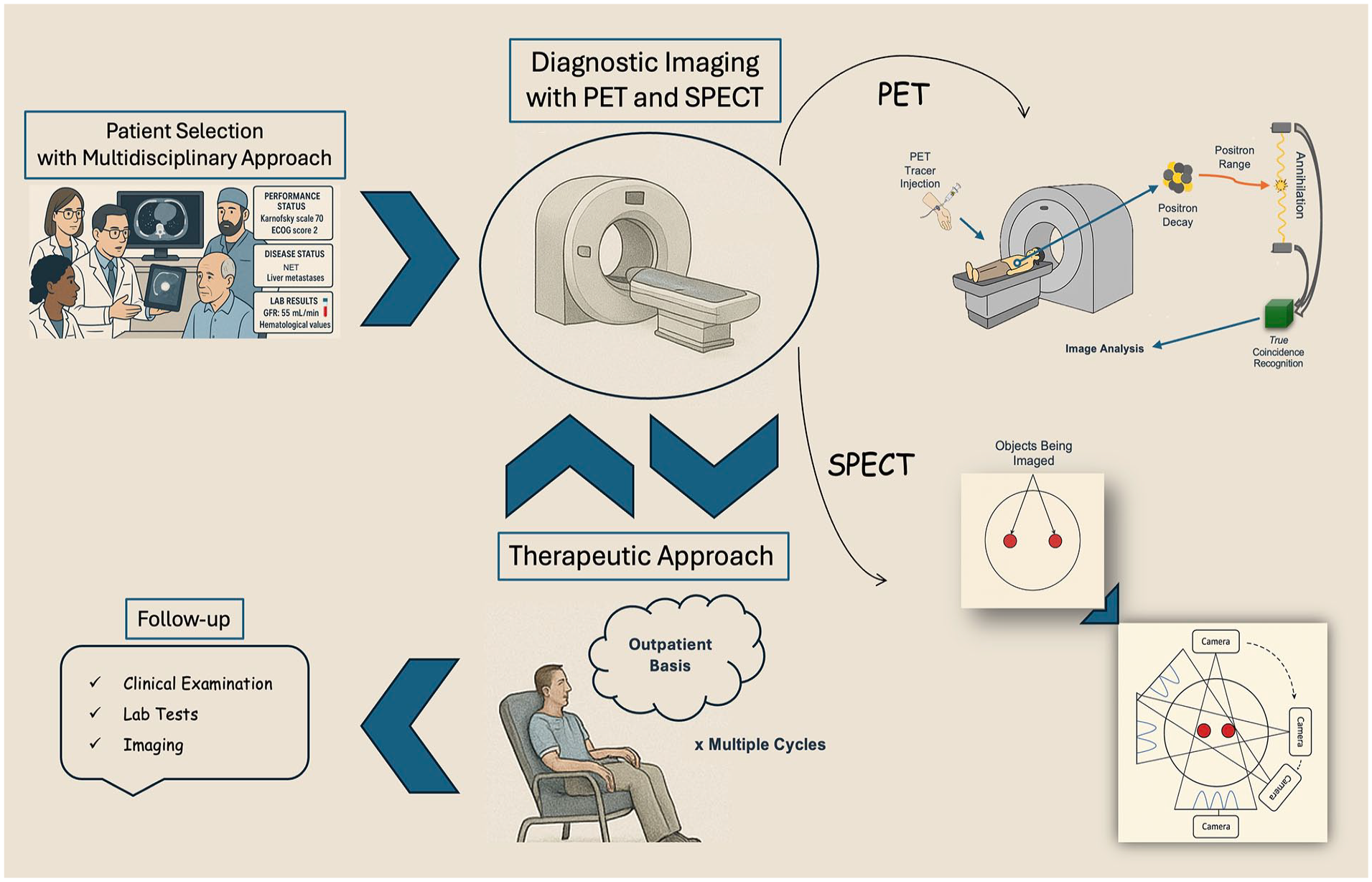
Representation of the theranostic workflow.
Patient Selection
One of the cornerstones of theranostic practice is careful patient selection. Since not all tumours express the specific molecular targets [as much as needed] for theranostic agents to be effective, it is essential to identify suitable candidates through confirmation of expression of the target on screening imaging. Patient selection typically involves a multidisciplinary team, which may include but is not limited to oncologists, nuclear medicine physicians, radiologists, surgeons, and pathologists, who collaborate to assess the patient’s eligibility for theranostic treatment.
The extent of disease spread, tumour size, sites of metastatic involvement, and degree of target expression are critical in determining a patient’s suitability for theranostic treatment. Moreover, patients’ prior treatments (chemotherapy, radiation, surgery), overall clinical status (eg, blood count levels, renal function), ECOG performance status, alternative treatment options, and patient preferences all play a role in decisions regarding radioligand therapy.
Diagnostic Imaging
The imaging-based confirmation of the specific molecular targeting, primarily through PET/CT, is a fundamental aspect of theranostic. The target expression (ie, the uptake of the radiopharmaceutical in the tumours and metastases) on PET imaging should ideally be several times higher than the physiologic background activity (eg, liver) to ensure the efficacy of the treatment. Diagnostic imaging is crucial for assessing the presence and extent of target expression, determining a patient’s eligibility for radioligand therapy and ensuring proper disease staging.
Therapeutic Approach
Once a patient has been deemed eligible, they are assessed by the respective oncologist and discussed by a multidisciplinary team prior to administration of the therapeutic radiopharmaceutical to ensure the fact that radioligand therapy is the best treatment option for the patient. This treatment is administered in specialized nuclear medicine facilities by trained personnel. This process typically occurs through multiple cycles over several months (usually 4-6 cycles with commonly applied intervals of 6-8 weeks), depending on the type of radioligand, malignancy, the extent of disease, and the patient’s response to therapy. Dosimetry studies could also be performed to optimize therapeutic doses while minimizing potential toxicity to healthy tissues (eg, bone marrow, kidneys). This can be done using both pre- and post-therapy dosimetry, the latter being more commonly used and can be measured using post-treatment SPECT imaging.
Clinical and Imaging Follow-Up
Follow-up typically involves a combination of clinical examinations, blood tests, and imaging to assess the therapeutic response or detect probable adverse effects.
PET or SPECT imaging may be repeated after a certain number of therapy cycles to evaluate tumour response. For example, 68Ga-PSMA PET/CT or 68Ga-DOTATATE PET/CT may be used to reassess the target PSMA or SSTR expression, respectively, and measure changes in tumour burden. Also, an interim response assessment can be done to re-evaluate the disease between cycles. Functional imaging provides real-time information on how the tumour is responding to treatment, allowing for potential adjustments in therapy if necessary.
In addition to efficacy, follow-up also involves clinical monitoring for both short-term and long-term side effects. The kidneys, bone marrow, and liver are particularly susceptible to radiation toxicity from radioligand therapy. Regular blood tests, including complete blood counts, kidney function tests, and liver function tests, are performed regularly to diagnose any signs of early or late toxicities.
Clinical Examples
To provide examples from routine clinical practice, the workflow of the 2 most recognized theranostics pairs, 68Ga/177Lu-PSMA and 68Ga/177Lu-DOTATATE, are provided.
68Ga/177Lu-PSMA
177Lu-PSMA is approved for patients with PSMA-positive metastatic castration-resistant prostate cancer (mCRPC) who have failed at least one androgen-receptor pathway inhibitor (eg, enzalutamide or abiraterone) and taxane-based chemotherapy (docetaxel).20,21 Taxane-naïve mCRPC patients who progressed under at least one novel androgen axis drug (eg, enzalutamide or abiraterone) may also benefit from 177Lu-PSMA therapy.
To be eligible, patients should have positive PSMA expression on pre-therapy imaging, which is assessed by 68Ga-/ or 18F-PSMA PET. Imaging should ideally be performed within 3 months of treatment initiation or after any significant disease progression to ensure that it reflects the current tumour status. PSMA uptake of lesions, reported as standardized uptake values (SUVs), should be significantly higher than the hepatic background for this purpose. Preferably, 18FDG PET/CT can also be done to exclude any PSMA- FDG+ tumour lesions and improve overall response rates, however, it is not currently mandatory. 22 The presence of extensive PSMA-negative lesions or lesions with mixed PSMA and 18FDG uptake likely results in poorer response rates.
Other eligibility criteria would include adequate bone marrow reserve (eg, platelet count ≥75-100/nL and hemoglobin ≥8-9 g/dL) and preserved renal (eg, GFR ≥30-40 mL/min) and liver function (eg, liver enzymes less than the fivefold upper normal limit).20,23 Also, serum PSA levels provide a marker for disease burden, and additional tests, such as lactate dehydrogenase (LDH) and alkaline phosphatase (ALP), help quantify tumour activity and metastatic spread. A comprehensive review of medical history, including prior treatments and current disease status and comorbidities, should also be done, and the ECOG scale value (commonly <3 is acceptable) should be calculated. Furthermore, a life expectancy of less than 3 months is not acceptable for therapy.
To prepare patients, hydration is recommended to protect renal function. Also, patients should be educated prior to the therapy about the therapy process, potential side effects, and radiation safety precautions . Discussions should address the possibility of temporary side effects such as fatigue and dry mouth, as well as the rare but serious risks of myelosuppression and nephrotoxicity. To administer the radiopharmaceutical, the primary agent is 177Lu-PSMA-617, which is FDA-approved and extensively studied. 20 Alternative agents, such as 177Lu-PSMA-I&T, have shown similar efficacy and are routinely used as well. The standard fixed dose is usually 7.4 GBq (200 mCi) per cycle, administered intravenously. Typical treatment involves 4 to 6 cycles at 6-week intervals. Notably, patient-adjusted (such as using dosimetry) are discussed in the literature to optimize therapy but have not been clinically implemented yet.
Post-therapeutic whole-body scintigraphy or multi-field-of-view SPECT or SPECT/CT is recommended to be performed at least 2 hours after injection to rule out extravasation and ensure proper targeting. After several cycles, 68Ga-PSMA PET may help with a more precise therapy response assessment. Response assessment includes both biochemical evaluation and imaging assessment. A PSA reduction of ≥50% is a commonly used marker of biochemical response. For imaging, changes in PSMA uptake on 68Ga-PSMA PET are measured and changes in lesion size can also be considered. In this regard, for example, Response Evaluation Criteria in PSMA (RECIP) and PSMA PET Progression (PPP) are proposed criteria for standardized response evaluation24,25 (Table 3).
Head-to-Head Comparison of the Response Evaluation Criteria in PSMA PET (RECIP; v1.0) and PSMA PET Progression (PPP) Criteria for PSMA-Ligand PET-Based Progressive Disease Interpretation.

Regarding follow-up, patients should be monitored for delayed toxicities such as myelosuppression, nephrotoxicity, xerostomia, and other common side effects. Periodic imaging and laboratory tests may be needed to assess disease progression/recurrence and organ function in the long term.
68Ga/177Lu-DOTATATE
177Lu-DOTATATE therapy has demonstrated efficacy in prolonging survival and improving quality of life in eligible NET patients, particularly those with gastroenteropancreatic NETs (GEP-NETs). It is primarily used for patients with SSTR-positive, well-differentiated NETs (routinely grades 1 and 2 with Ki-67 ≤20%), particularly those with progressive or metastatic disease refractory to first-line somatostatin analogue (SSA) therapy. 26 Recent trials have also suggested its efficacy as a first-line therapy in this specific patient population. 27 The imaging eligibility is determined similarly through positive SSTR expression demonstrated via 68Ga-DOTATATE PET, with uptake significantly exceeding liver background. However, patients with a high volume of SSTR-negative lesions, extensive liver dysfunction, severe renal impairment (glomerular filtration rate <30 mL/min), inadequate bone marrow reserve (eg, platelet count <75/nL, hemoglobin <8 g/dL, white blood cell count <3/nL), or active uncontrolled hormonal crises (eg, carcinoid syndrome) are typically excluded. 28
A similar comprehensive clinical evaluation, as outlined above, remains crucial. Laboratory evaluation may also include hormonal markers such as chromogranin A and urinary 5-HIAA for assessing functional NETs. Also, pregnancy and breastfeeding should be ruled out. Imaging plays a central role in both diagnosis and therapy planning. 68Ga-DOTATATE PET/CT confirms significant SSTR expression, assesses disease burden and identifies SSTR-negative lesions. Additionally, conventional imaging (eg, CT and MRI) can complement PET by providing detailed anatomical evaluation and surgical planning if needed.
Regarding patient preparation, long-acting SSAs should be discontinued 4 to 6 weeks prior to therapy to avoid interference with SSTR binding. Short-acting SSAs can be used up to 24 hours before treatment. Importantly, amino acid infusion should be done to lessen the risk of nephrotoxicity. In this regard, pre-treatment with lysine-arginine infusions minimizes renal radiation exposure. Additionally, patients are advised to hydrate well to protect renal function and facilitate radiotracer clearance. Furthermore, prophylactic antiemetics, including 5-HT3 antagonists (eg, ondansetron), are recommended to manage nausea during amino acid infusions. Subsequently, 177Lu-DOTATATE is administered intravenously over 30 minutes, accompanied by a concurrent amino acid infusion to protect the kidneys. The standard fixed dose is 7.4 GBq (200 mCi) per cycle, administered every 6 to 8 weeks for a total of 4 cycles. 28
Similar to 177Lu-PSMA therapy, post-therapy imaging can be performed to rule out extravasation and ensure proper targeting. Individualized dosimetry can also be conducted to evaluate organ-specific radiation doses, particularly to the kidneys and bone marrow, helping to guide further therapy decisions while minimizing toxicity. To assess response, preferably 68Ga-DOTA-ligand PET, as well as potential additional conventional imaging, can be performed to assess any changes in tumour burden. Moreover, a decline in serum chromogranin A or 5-HIAA, as biochemical markers, is often indicative of a therapeutic response. Lastly, patient-reported outcomes, including symptom relief and functional status, are also key metrics of success .
Regarding follow-up, regular monitoring includes laboratory tests and imaging at 3 to 6 month intervals to assess disease control and detect potential late toxicities. Common side effects include mild hematologic suppression and nausea. 29 Long-term monitoring is essential for rare but serious toxicities, such as myelodysplastic syndrome or renal dysfunction. Also noteworthy, as maintenance therapy, continued use of long-acting SSAs is recommended for hormone control and potential anti-tumour effects.
Response Assessment
Radiologists play an important role in evaluating pre-, mid-, and post-therapy imaging. Before initiating radioligand therapy, as mentioned earlier, patients are assessed with various imaging technologies to determine the presence of disease and expression levels of the molecular target. Following treatment, radiologists monitor the therapeutic response using molecular and morphological follow-up imaging, including interim and post-therapy response assessments.
Molecular imaging is used to evaluate changes in tumour metabolic activity and receptor expression, which can provide early indicators of therapy success or failure, often before anatomical changes become evident by conventional imaging modalities. This helps to adjust treatment strategies based on real-time imaging feedback, ensuring more personalized and effective patient management.
The evaluation of response to theranostic treatments requires a nuanced approach, integrating both molecular and anatomical imaging. Each modality offers distinct insights into tumour biology, such as metabolic activity and receptor expression, as well as changes in the tumour morphology and extent. Traditional response assessment criteria, such as Response Evaluation Criteria In Solid Tumours (RECIST), classically rely on changes in tumour size to evaluate treatment efficacy. 30 However, anatomical changes can be delayed in comparison to molecular or functional changes. Thus, molecular and anatomical components provide complementary information, and a combined approach is often necessary in response assessment, as it allows for a more comprehensive evaluation of the patients. Radiologists must be adept at integrating both types of information to provide the most accurate and informative assessments possible.
To provide an example of head-to-head differences between conventional imaging-only and molecular imaging response criteria, RECIST versus PET Response Criteria in Solid Tumours (PERCIST) response assessment criteria are compared in Table 4.
Head-to-Head Comparison of the Response Evaluation Criteria in Solid Tumours (RECIST; v1.1) and Positron Emission Tomography Response Criteria in Solid Tumours (PERCIST) Criteria for Solid Tumour Response Assessment.

Safety and Side Effects
Common Side Effects
Radioligand therapies are generally well-tolerated. However, like any other treatment, they may also cause side effects. The nature of these side effects depends on the specific radiopharmaceutical used and the patient’s underlying condition.
Acute Side Effects
Common acute side effects include fatigue, nausea, and dry mouth (xerostomia; for 177Lu-PSMA therapy), which are frequently reported following therapy. These side effects are typically mild and self-limiting, resolving without long-term consequences.
Short-Term Effects
The short-term effects of radiopharmaceuticals are generally well-managed in the clinical setting. However, patients may also experience some more serious early toxicities, such as nephrotoxicity and hematotoxicity, for which they should be monitored closely using lab tests. Furthermore, imaging studies during this period may reveal tumour flare reactions, where the initial morphological increase in tumour size or functional activity could be misinterpreted for disease progression. Radiologists must differentiate between these therapy-related changes and true disease progression.
Long-Term Effects
The long-term effects are of growing concern, particularly as more patients undergo multiple rounds of radioligand therapy. Long-term exposure to radiopharmaceuticals carries the risk of late radiation toxicity, including irreversible organ damage. For instance, patients receiving high cumulative doses over multiple cycles of 177Lu-therapy may develop renal insufficiency after several treatment cycles. Although such effects are uncommon (given that patients were selected precisely in the initial phase), they underscore the importance of patient monitoring. To minimize radiation-induced organ toxicity, carefully calculating organ-specific doses (before and after therapy) plays a key role.
Challenges and Limitations
While theranostic through RLT has dramatically changed conventional approaches to personalized medicine, it still presents challenges and limitations. These barriers span across the clinical, technological, and logistical domains, and addressing them is crucial for the widespread and equitable adoption of theranostic techniques in routine clinical practice.
Not all cancer patients are suitable candidates for theranostic approaches. The efficacy of theranostics depends on the presence of specific molecular targets. Additionally, the heterogeneous nature of tumour biology means that not every patient will express these targets at sufficient levels to benefit from therapy. Even within the same tumour type, the level of target expression can vary between patients and even between different involved sites within the same patient. This inter- and intra-patient heterogeneity poses a challenge in patient selection and underscores the need for personalized treatment planning.
Furthermore, tumours can undergo molecular changes over the course of treatment, leading to a loss of target expression that was initially present. For instance, prostate cancer patients treated with 177Lu-PSMA may develop resistance as the tumour cells down/deregulate pathways. 31 This necessitates ongoing molecular imaging to reassess target expression during the course of therapy and adjust treatment strategies accordingly .
From the cost perspective, theranostic treatments are resource-intensive. The need for specialized imaging modalities, such as PET/CT or PET/MRI, along with radioligand therapies, makes theranostics expensive for healthcare providers and patients. Additionally, the cost of developing, producing, and delivering treatments incurs substantial costs. Thus, the high price of PET imaging agents, combined with multiple cycles of therapies, can make theranostic prohibitively expensive, particularly in healthcare systems with limited insurance coverage and reimbursements. This financial burden extends to healthcare systems, which may be hesitant to adopt such costly technologies on a large scale.
Moreover, the implementation of theranostics requires significant investment in imaging equipment, radiochemistry labs, and trained personnel. Many hospitals, particularly in underserved or rural areas, may lack the necessary infrastructure to deliver these treatments. This infrastructure gap can create inequities in access to theranostics, with patients in larger academic centres having greater access to these advanced therapies.
Concluding Remarks
Theranostic represents a paradigm shift in personalized medicine, combining diagnosis and treatment, offering tailored therapies based on a patient’s unique molecular profile. The combination of precision imaging and targeted radiopharmaceutical therapies has significantly improved outcomes for patients with difficult-to-treat metastatic cancers. The dual ability to diagnose and treat in a single framework allows for real-time monitoring of therapeutic efficacy and the possibility of adjusting treatments as needed. For radiologists, theranostics represents an important area of growth. As theranostic becomes increasingly integrated into routine clinical practice, radiologists will play a pivotal role in guiding therapeutic decisions, monitoring response to treatment, and ensuring patient safety.
Moreover, with the advancements in radiopharmaceutical technology, the scope of theranostics will likely expand further to include a broader range of oncological diseases. Additionally, the integration of advanced imaging modalities, such as PET/MRI, and the growing role of artificial intelligence and quantification in image analysis and predictive modelling will further enhance the precision and effectiveness of theranostics.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.