Abstract
Chronic pelvic pain (CPP) is a debilitating condition affecting up to 26% of women worldwide. Among its many causes, pelvic venous disorders (PeVD) is increasingly recognized as an underdiagnosed contributor, often overlooked due to its non-specific presentation. PeVD results from venous reflux, or obstruction, leading to venous hypertension, congestion, and chronic pain. Advanced imaging techniques play a pivotal role in diagnosing PeVD, differentiating it from other etiologies of CPP. Ultrasound, particularly Doppler imaging, serves as the firstline modality for assessing venous reflux and dilation. Computed tomography and magnetic resonance venography provide detailed anatomical and haemodynamic evaluations, aiding in the identification of compressive syndromes and collateral pathways. Selective venography remains the gold standard, offering real-time visualization of reflux severity and guiding minimally invasive interventions such as venous embolization. Despite these advances, PeVD remains underrecognized in clinical practice, leading to delays in diagnosis and management. Increased awareness and standardized diagnostic criteria are crucial for improving patient outcomes. A multidisciplinary approach incorporating radiologists, gynecologists, and vascular specialists is essential for the comprehensive evaluation and treatment of PeVD. Emerging therapies, including endovascular techniques, offer promising options for symptom relief, reducing the need for invasive surgical procedures. This review highlights the pathophysiology, imaging modalities, and evolving management strategies for PeVD, emphasizing the importance of early recognition and intervention in patients with CPP.
Introduction
Chronic pelvic pain (CPP) affects up to 26% of women globally, significantly impacting quality of life and healthcare costs.1,2 CPP encompasses a complex spectrum of conditions, with etiologies ranging from gynecologic and gastrointestinal to musculoskeletal and urologic disorders. 2 Diagnostic radiology is essential in CPP evaluation, offering non-invasive imaging tools like ultrasound, MRI, and CT to improve diagnosis and guide treatment.1,3-6 While endometriosis, irritable bowel syndrome, and interstitial cystitis are well-recognized contributors, the emerging evidence also highlights pelvic venous disorder (PeVD) as a critical yet underdiagnosed cause of CPP.
PeVD, historically referred to by pelvic congestion, nutcracker, and May-Thurner syndromes, involves venous reflux, or obstruction, commonly in the ovarian and internal iliac veins. While May-Thurner syndrome is primarily associated with left lower extremity deep vein thrombosis due to compression of the left common iliac vein, it may also contribute to pelvic venous congestion in select cases. PeVD is often overlooked due to non-specific symptoms and disproportionately affects premenopausal women, particularly individuals with hormonal risk factors.7-9 It remains underdiagnosed, especially in marginalized communities facing healthcare inequities. Clinicians must adopt multidisciplinary and other health professionals to improve patient outcomes.
Background
Chronic Pelvic Pain
CPP is a non-cyclic pain lasting over 6 months, localized to the pelvic region, and significantly impacting quality of life. Its prevalence ranges from 5% to 26% globally, accounting for 20% of gynecological outpatient visits and posing substantial economic burdens.10-13
The pathophysiology of CPP involves a complex interplay of nociceptive, neuropathic, and nociplastic pain mechanisms, with key contributors including inflammatory processes, central sensitization, and disrupted pain signalling pathways. Pelvic somatic and autonomic nerves, such as the pudendal, iliohypogastric, and genitofemoral nerves, play a significant role in transmitting pain signals.10,11 Risk factors for CPP include psychosocial elements, such as childhood trauma, sexual abuse, and psychological conditions like depression and anxiety, as well as clinical factors like a history of pelvic surgeries or pelvic inflammatory disease.12,13
Gynecologic etiologies of CPP include endometriosis, chronic pelvic inflammatory disease (PID), and adenomyosis, which are significant contributors to pelvic pain. Endometriosis involves ectopic endometrial tissue, causing local inflammation and adhesions, often leading to persistent pain. Chronic PID results in scarring and recurrent pain due to unresolved infections. Adenomyosis, characterized by endometrial tissue invading the uterine muscle, and other conditions like ovarian cysts and post-surgical adhesions further contribute to chronic discomfort.10,14
Non-gynecologic etiologies of CPP include gastrointestinal, genitourinary, and musculoskeletal conditions, which contribute to pelvic discomfort through mechanisms such as inflammation, compression, or muscle dysfunction. Conditions like irritable bowel syndrome (IBS), inflammatory bowel disease (IBD), interstitial cystitis, urinary retention, pelvic floor myalgia, and myofascial pain commonly overlap, often presenting with urgency, referred pain, or localized discomfort.10,14
Comorbidities play a significant role in exacerbating CPP by amplifying pain perception and hindering coping mechanisms. Factors such as a history of abuse, depression, and anxiety contribute to chronic pain syndromes through mechanisms like central sensitization. Additionally, conditions such as abdominal wall pain, including myofascial and neuropathic causes, further compound the impact of CPP.12-14
Pelvic Venous Disorder
PeVD refers to a spectrum of venous disorders contributing to CPP, rooted in the anatomy and haemodynamics of the pelvic venous system. This includes the ovarian veins, internal iliac veins (IIV), and the left renal vein (LRV). The left ovarian vein is particularly prone to reflux and varicosity due to its length and perpendicular drainage angle into the LRV.1,2,15
Venous insufficiency in PeVD results from valve dysfunction, hormonal influences, and pregnancy-related changes. Due to anatomical predispositions, absent or incompetent valves in the ovarian veins elevate venous pressure and reflux, particularly on the left side. 2 Hormonal changes during pregnancy, including elevated estrogen and progesterone, further weaken vein walls, increasing venous return and exacerbating insufficiency.3,15 Pathophysiological outcomes, such as venous hypertension and nociceptor activation, lead to chronic pain and overlap with disorders like IBS and endometriosis. Vascular compression, including left iliac vein compression (May-Thurner Syndrome) and left renal vein compression (Nutcracker Syndrome), can intensify pelvic and lower extremity reflux through venous obstruction or by themselves serve as a cause of pain.1-3,15
PeVD is classified using the Symptoms-Varices-Pathophysiology (SVP) system, which evaluates symptoms, varices, and pathophysiology. This framework helps assess presentations like CPP, varicose veins, and venous abnormalities.8,16 Complementing SVP system, the Clinical-Etiologic-Anatomic-Pathophysiologic (CEAP) classification offers insights into overlapping pelvic and lower extremity conditions. Together, these systems support comprehensive assessments using patient-reported outcomes, Doppler ultrasound, and advanced imaging like MRV and CT venography.7,8,17,18
Imaging Modalities
Importance of Imaging
Imaging serves as an essential tool for objectively assessing pelvic structures, particularly when clinical findings are unclear or inconclusive. 19 By providing detailed visualization of structural, inflammatory, and vascular abnormalities, it helps refine the differential diagnosis and differentiate between gynecologic, gastrointestinal, urologic, musculoskeletal, and neurologic causes of chronic pelvic pain.10,20 Furthermore, imaging evaluates disease severity, forecasts treatment outcomes, and reduces the need for unnecessary invasive surgical interventions. 21 Table 1 compares advantages and disadvantages of various imaging modalities. Table 2 summarizes the diagnostic criteria for pelvic venous disorders.
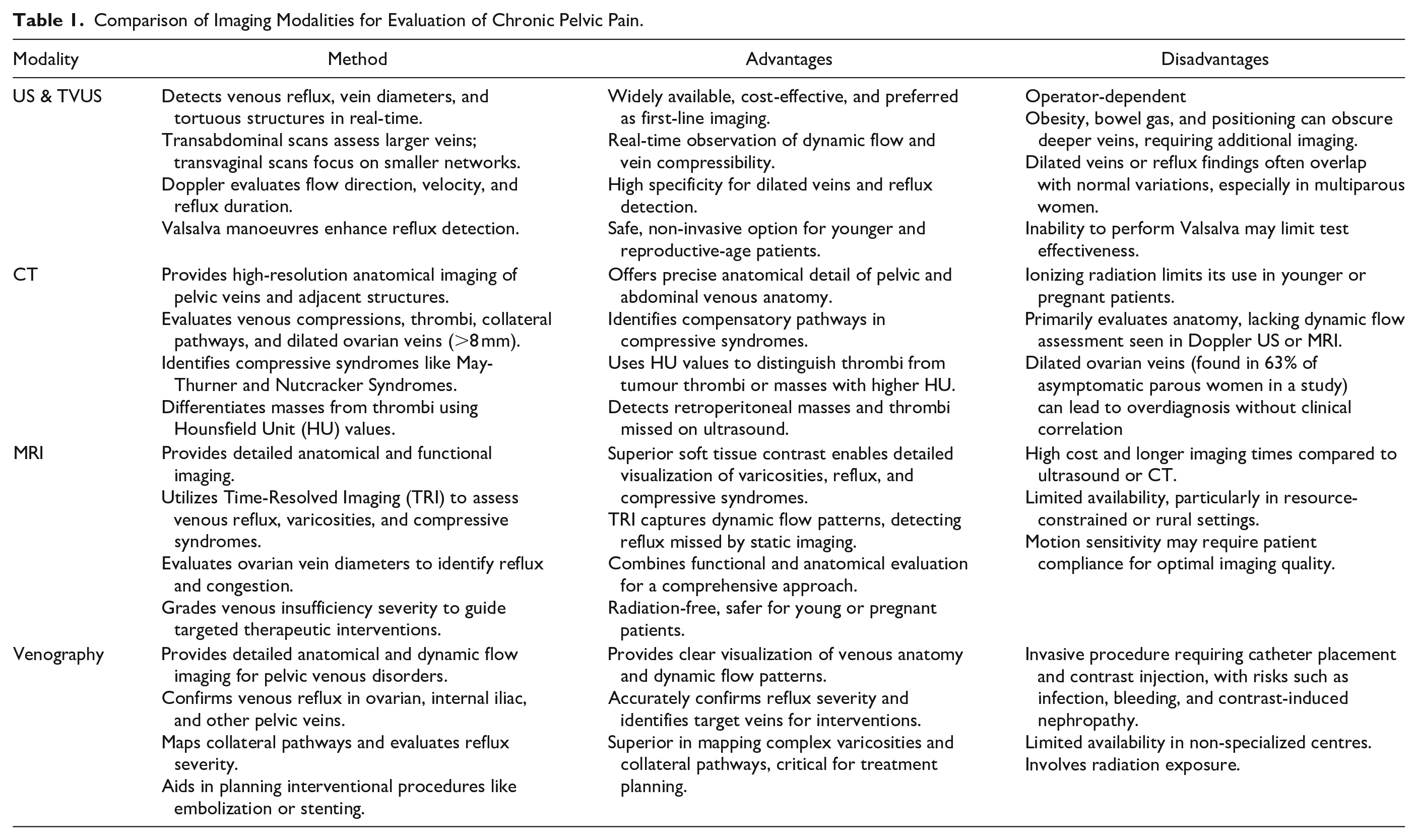
Comparison of Imaging Modalities for Evaluation of Chronic Pelvic Pain.
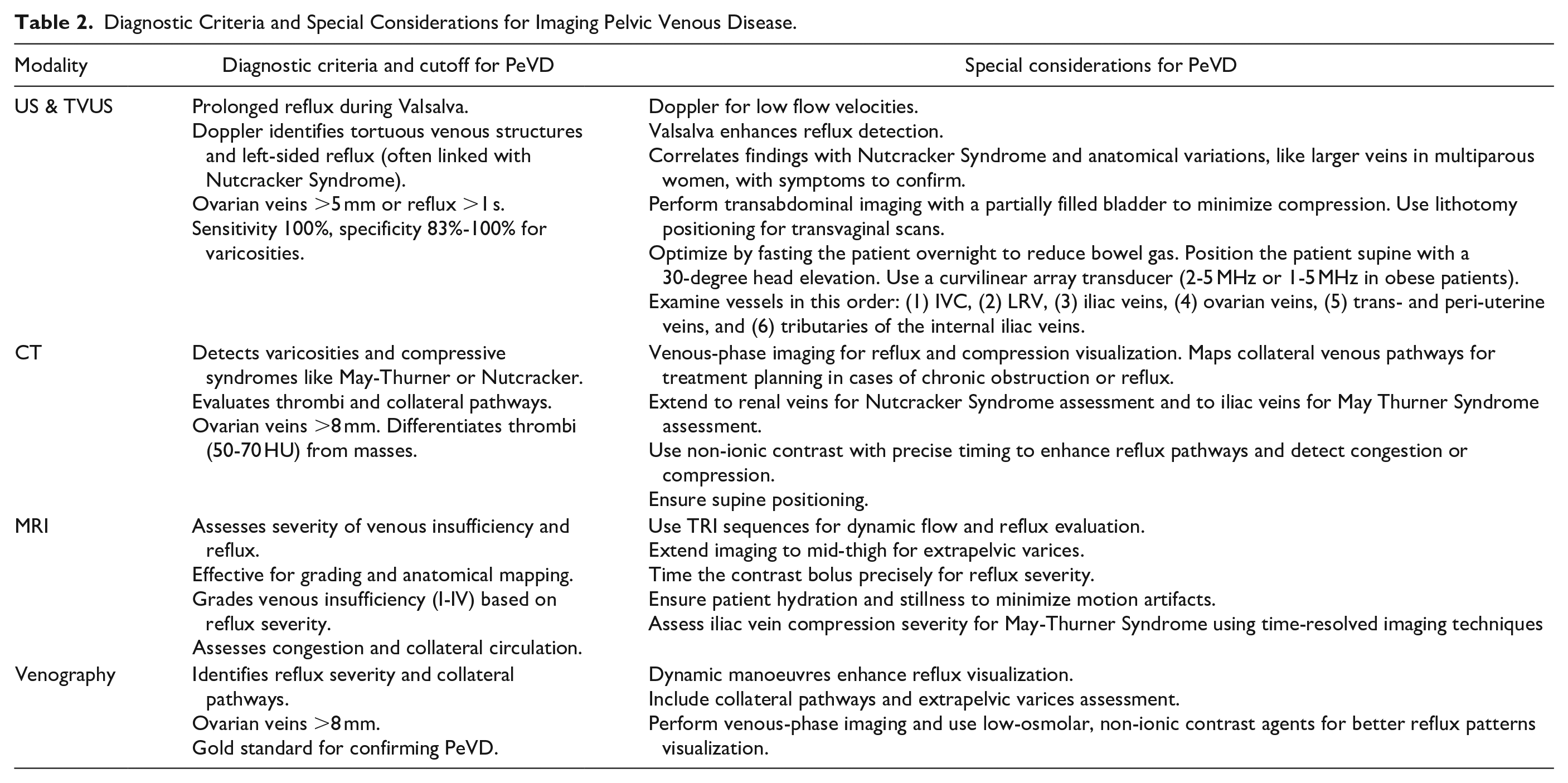
Diagnostic Criteria and Special Considerations for Imaging Pelvic Venous Disease.
Ultrasound (US) and Transvaginal Ultrasound (TVUS)
Ultrasound is the first-line imaging modality for suspected pelvic venous disorders (See Supplemental Appendix 1 for a summary of our institution’s protocol). Transabdominal ultrasound (TAUS) provides a broad overview of pelvic anatomy, while transvaginal ultrasound (TVUS) offers higher resolution for smaller structures. TAUS is often sufficient for most examinations.
US assesses vein diameters, pelvic varicosities, and reflux. TAUS is used for initial ovarian vein evaluation, with diameters >5 mm considered abnormal, as seen in Figures 1 and 2. 22 TAUS with a full bladder can provide detailed visualization of uterine, parametrial, and vaginal veins, often employing Valsalva manoeuvres to enhance reflux detection, as seen in Figure 3.
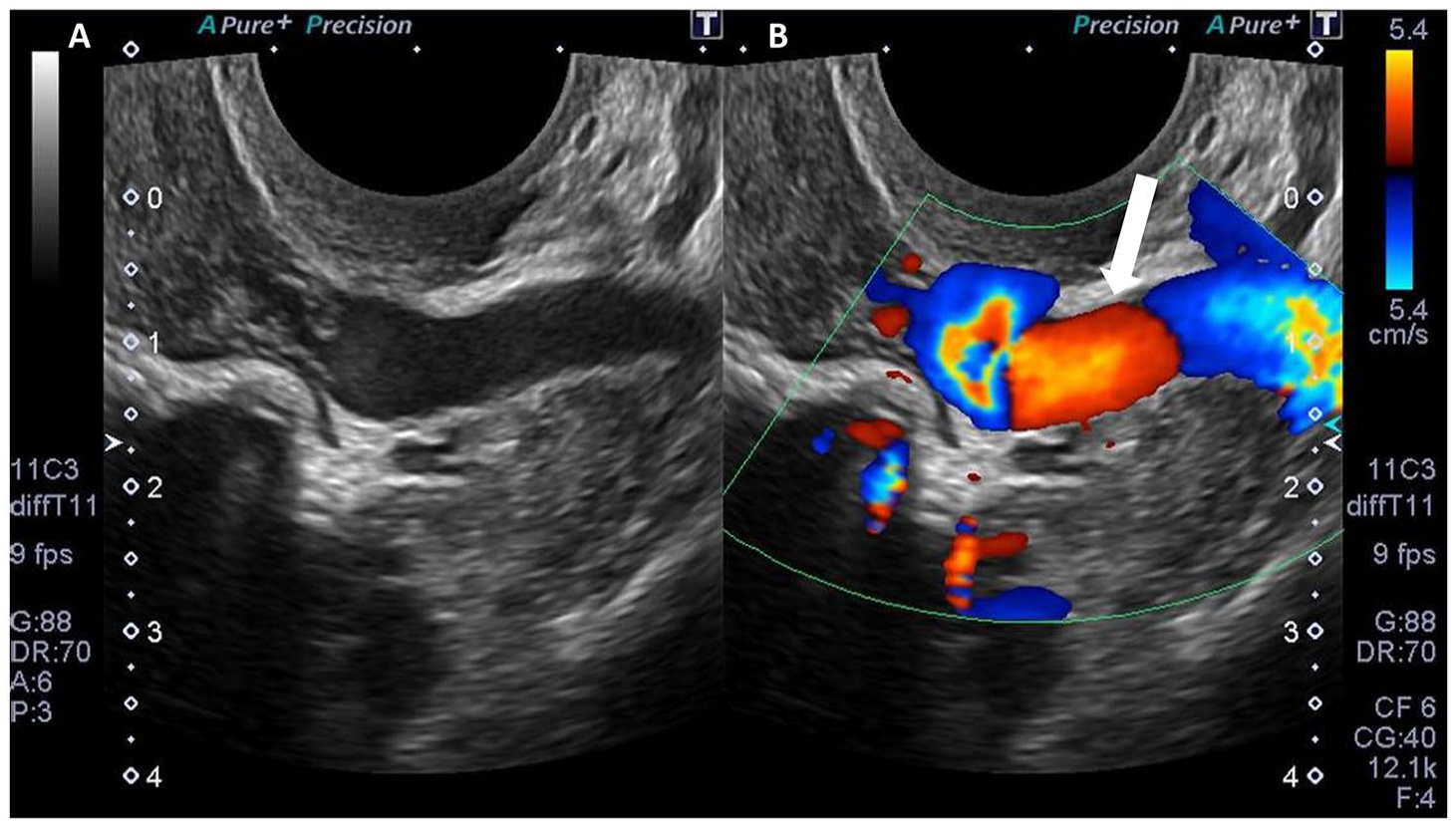
Fifty year old female with flank pain and pelvic pain not yet diagnosed. Transabdominal ultrasound imaging revealed prominent left periuterine vein (white arrow). (A) B-mode image of dilated left periuterine vein. (B) Colour doppler image of dilated left periuterine vein.
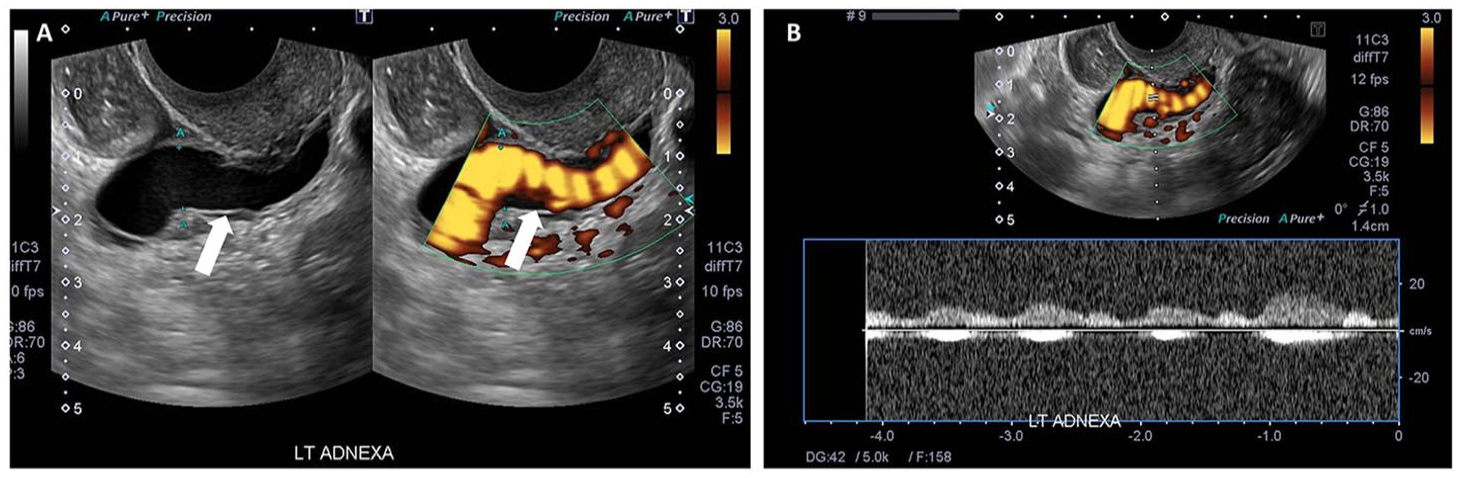
Sixty-one year old female with cyclic pelvic pain not yet diagnosed. Transabdominal ultrasound (A) with colour doppler (B) demonstrated engorged parametrial vessels (white arrows), more significant on the left side.
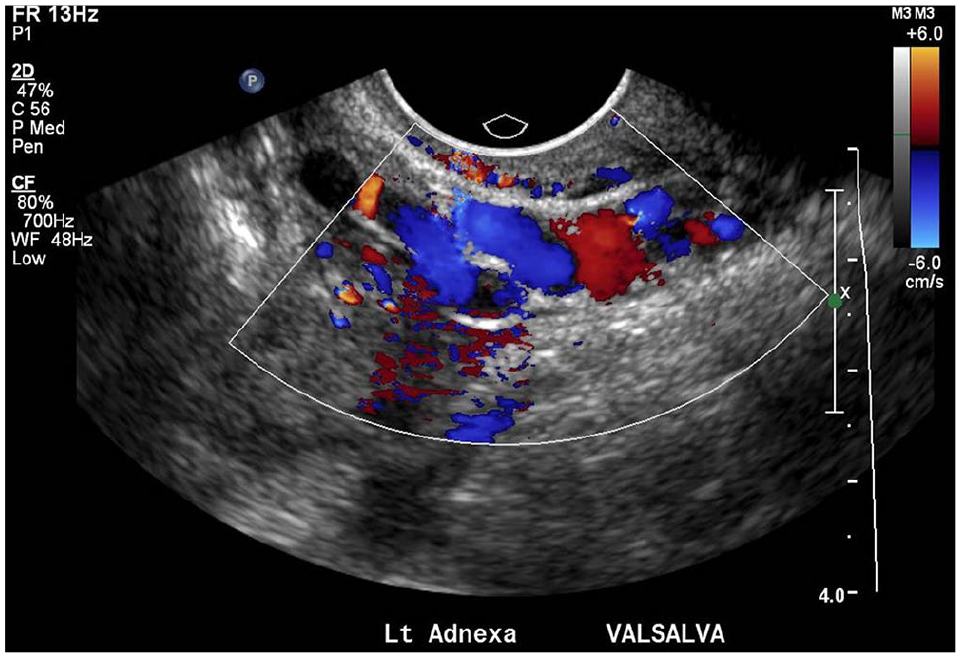
Nineteen year old female with 3 months of pelvic cramps. Transabdominal ultrasound under Valsalva manoeuvre demonstrating prominent left gonadal veins, with reversed venous flow on colour doppler.
Steenbeek et al reported sensitivity and specificity values of 83% to 100% for TVUS detecting pelvic varicosities. 22 Doppler US achieves a sensitivity of 100% for reversed caudal flow in the ovarian vein, reinforcing its diagnostic reliability. However, patient-dependent factors such as obesity or bowel gas can compromise image quality.
Ultrasound results are highly operator-dependent, with variability significantly impacting diagnostic accuracy. The main limitations of ultrasound include challenges in patients who are uncooperative due to excessive pain, large amounts of bowel gas that prevent ultrasound penetration, and patient body habitus, such as obesity, which further complicates visualization. Additionally, the need for an experienced ultrasonographer adds another layer of variability. These factors often compromise the visualization of deep pelvic veins, limiting its effectiveness in certain patients. Moreover, findings on ultrasound are not always specific, frequently necessitating complementary imaging to confirm or rule out diagnoses.23,24 Small sample sizes and selective populations in studies have introduced variability in diagnostic metrics. Rezaei-Kalantari et al noted that ultrasound findings like dilated veins >5 mm and prolonged reflux >1 second highly suggest PeVD but lack specificity for universal application. 3
Computed Tomography (CT)
CT venography offers detailed anatomical imaging, useful for cases with inconclusive US findings or suspected compressive syndromes. It identifies dilated veins, as seen in Figures 4 and 5, as well as retroperitoneal masses, and compressive syndromes such as May-Thurner or Nutcracker Syndromes. It also evaluates collateral pathways and thrombi. 25
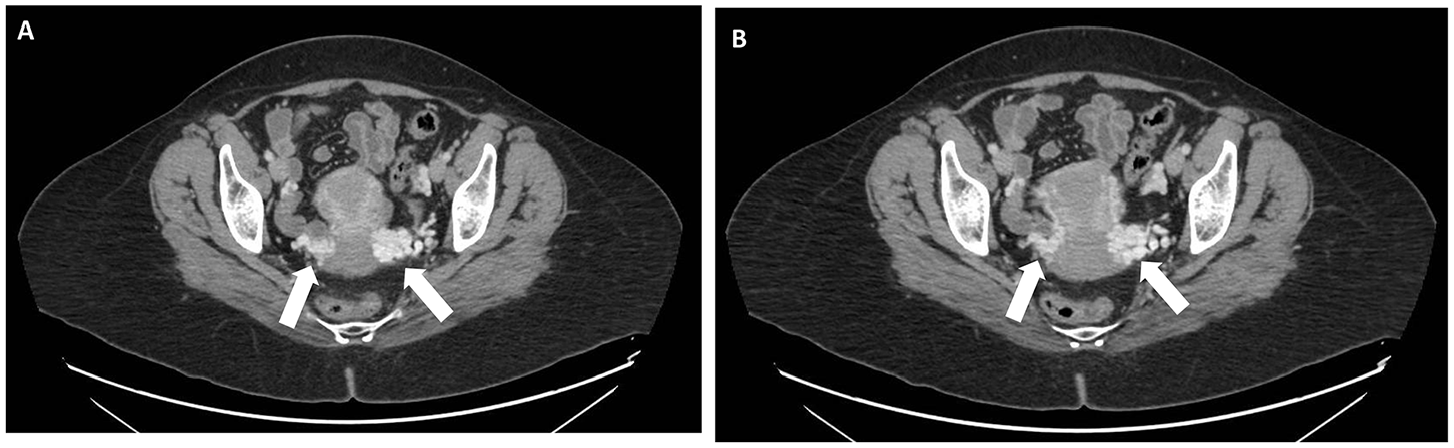
Forty-six year old female with vague lower abdominal pain, with a previously normal pelvic ultrasound. (A) CT abdomen/pelvis without contrast images revealed bilateral parametrial vein dilation (white arrows in A, B).
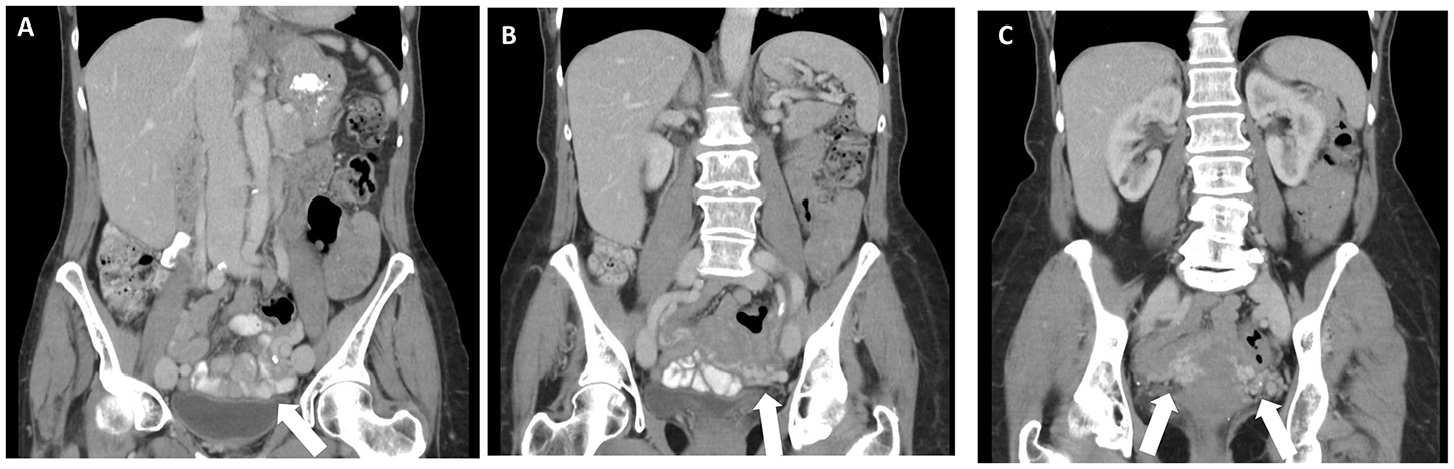
Seventy-two year old female with 1 month history of generalized pelvic pain. CT abdomen/pelvis with oral contrast demonstrating incidental finding of prominent parametrial vessels (white arrows in A, B, C).
For the CT venography protocol, patients are positioned supine, and a scout scan is performed from the highest dome of the diaphragm down to the mid-thigh level. A bolus tracking technique is used, with the region of interest (ROI) placed at the abdominal aorta below the renal arteries. Intravenous contrast (100 ml of non-ionic contrast medium, such as Omnipaque 350 or Ultravist 370) is injected at a rate of 4 to 5 ml/s via a wide bore cannula inserted into the anticubital vein. The arterial phase is followed by the venous phase 10 seconds later. Reformats in coronal and sagittal planes are obtained to provide more comprehensive anatomical evaluation. For optimal assessment, images are obtained at 1.25 mm slice thickness. 26
CT findings include dilated veins and varicosities in 47% of asymptomatic women, highlighting potential overdiagnosis concerns. Rozenblit et al observed that such findings are often incidental and not always clinically significant, underscoring the need for correlation with symptoms. 25
CT imaging presents several limitations. The ionizing radiation exposure limits its utility and increases the risk of developing radiation-induced cancer, particularly in younger patients. Additionally, CT provides limited functional assessment compared to US or MRI, as it primarily offers anatomical details rather than dynamic flow evaluation. A high prevalence of incidental findings, such as dilated veins in asymptomatic individuals, can lead to diagnostic uncertainty, highlighting the need for careful clinical correlation and judicious use of this modality. Studies often lack diverse patient demographics, limiting generalizability. Incidental findings of dilated ovarian veins on CT are reported in 63% of asymptomatic parous women, necessitating caution in interpretation. 25
Magnetic Resonance Imaging (MRI)
MRI, particularly MR venography, is a modality that provides both functional and anatomical insights. Additionally, the absence of radiation exposure makes MRI particularly advantageous for younger or pregnant patients.27-31
Protocols for PeVD integrate several advanced imaging sequences to achieve a comprehensive diagnosis. T2-weighted short tau inversion recovery (STIR) imaging enhances visualization by suppressing fat and bone signals, making it highly effective for identifying varicosities and low-flow venous structures. 32 Gadolinium-enhanced T1-weighted imaging provides superior delineation of refluxing veins, stenosis, and collateral circulation by highlighting slow-flow regions and vascular abnormalities. Moreover, the phase-contrast MRI (PC-MRI) quantitatively evaluates haemodynamic parameters, including flow velocity and direction, making it useful for detecting retrograde venous flow and reflux.
For patients contraindicated for gadolinium contrast, non-contrast MRI techniques, such as triggered angiography non-contrast-enhanced (TRANCE) imaging and gated 3D turbo spin-echo STIR (TSE-STIR) offer detailed anatomical and functional assessments by leveraging cardiac cycle-based signal differences. Additionally, time-resolved MR venography captures dynamic changes in venous flow, enabling precise evaluation of reflux severity and collateral pathways.33,34
MRI evaluates ovarian vein diameters (>8 mm) as seen in Figure 6, as well as venous reflux, and compressive syndromes. Time-resolved imaging techniques enhance dynamic flow assessment, which is critical for severity grading. 35 Studies like Chennur et al demonstrate that MRI achieves superior soft tissue contrast, enabling comprehensive evaluation of pelvic varicosities, reflux patterns, and alternative etiologies. It is particularly effective in guiding interventions like embolization.35-37
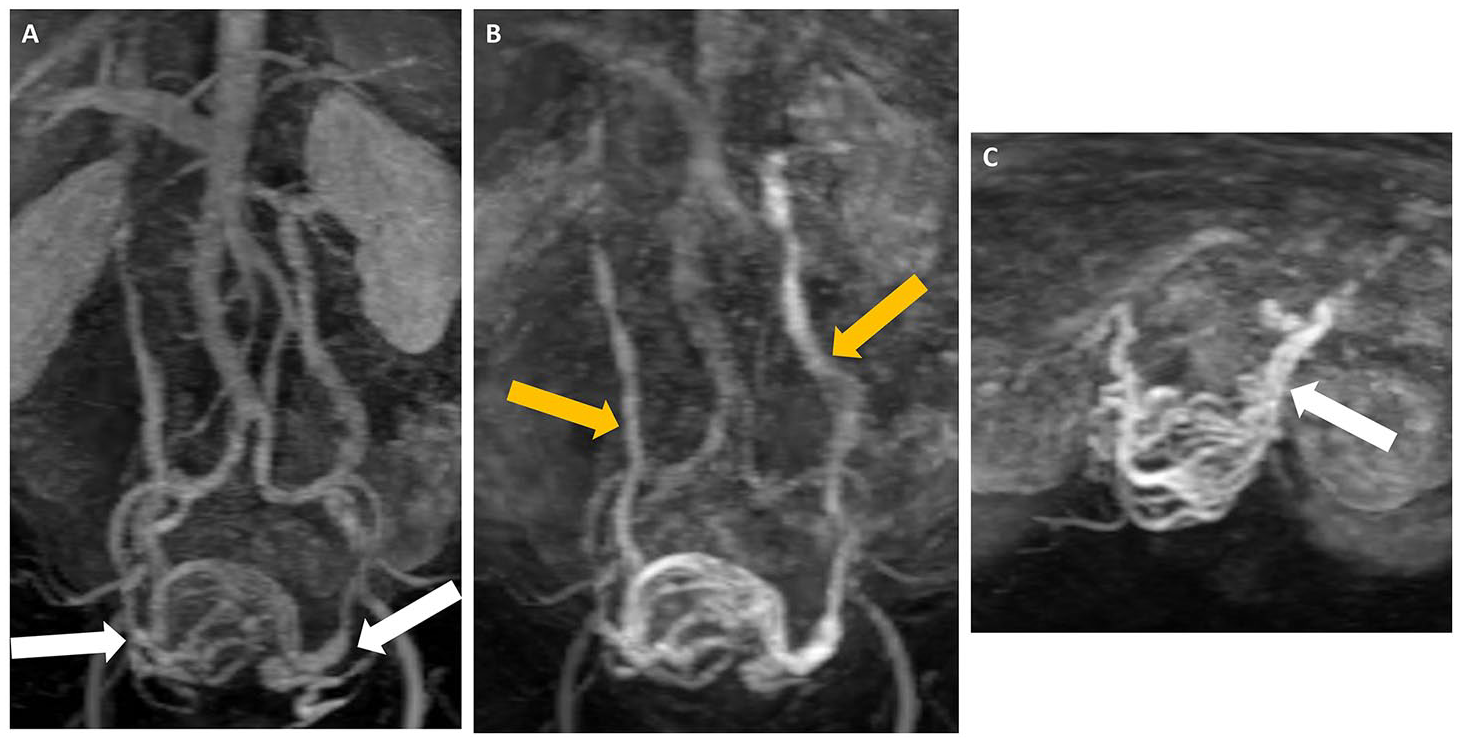
Sixty-one year old female with vague pelvic pain. (A) Coronal MRI TWIST (Time-Resolved MRA) sequence in keeping with bilateral dilated tortuous periuterine veins (white arrows), slightly more prominent in the left than right, measuring up to 8 mm in diameter. (B) Coronal MRI TWIST demonstrating dilation and tortuosity of bilateral gonadal veins (yellow arrows). (C) Axial MRI TWIST demonstrating dilated tortuous periuterine veins (white arrows).
MRI faces several limitations, including high costs and limited availability in resource-constrained settings, which restrict its accessibility. Extended scan times compared to CT or US can also increase patient discomfort and reduce efficiency, particularly in busy clinical settings. Additionally, MRI’s sensitivity to patient motion necessitates good patient compliance during the procedure to ensure image quality. Despite these drawbacks, its superior soft tissue contrast and dynamic flow capabilities make it an indispensable tool in diagnosing complex pelvic venous pathologies. MRI studies often include small cohorts, limiting statistical power. However, its ability to integrate anatomical and functional data provides a robust diagnostic advantage, particularly in complex PeVD cases.23,34
Venography
Selective catheter venography remains the gold standard for diagnosing PeVD, providing real-time dynamic flow and anatomical details, as seen in Figures 7 and 8. The procedure typically involves venous access via the common femoral or internal jugular vein, followed by selective catheterization of the ovarian, common iliac, and/or internal iliac veins. Venography identifies reflux severity, collateral pathways, and varices in real-time, often used to guide interventional procedures like embolization or stenting. 23
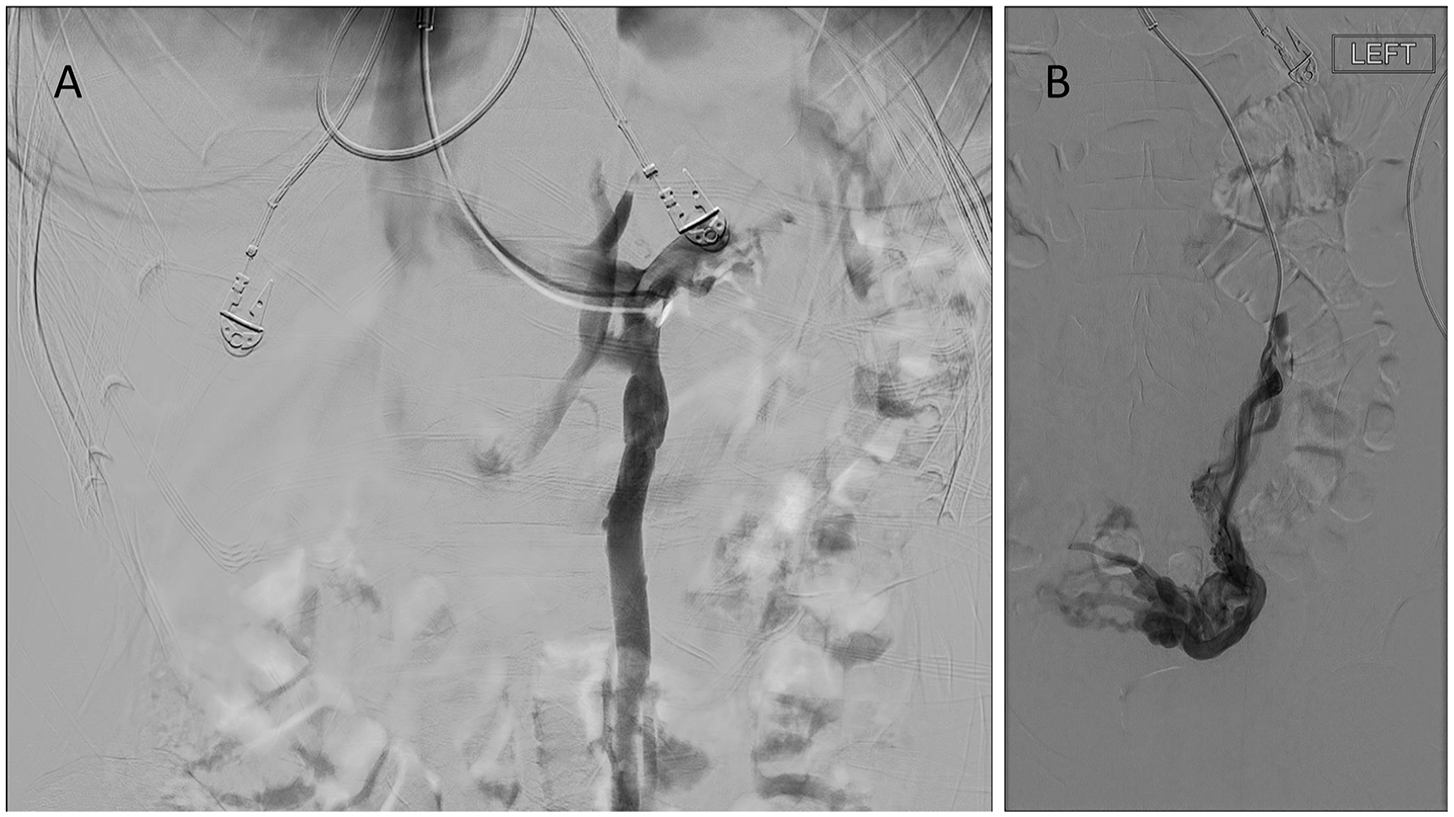
Venographic findings in PeVD. (A) Catheterization via the inferior vena cava (IVC) into the left renal vein and left ovarian vein, demonstrating a dilated and incompetent left ovarian vein with contrast reflux. (B) Catheterization of the left ovarian vein and left ovarian venous plexus, revealing dilated periuterine and pelvic veins.
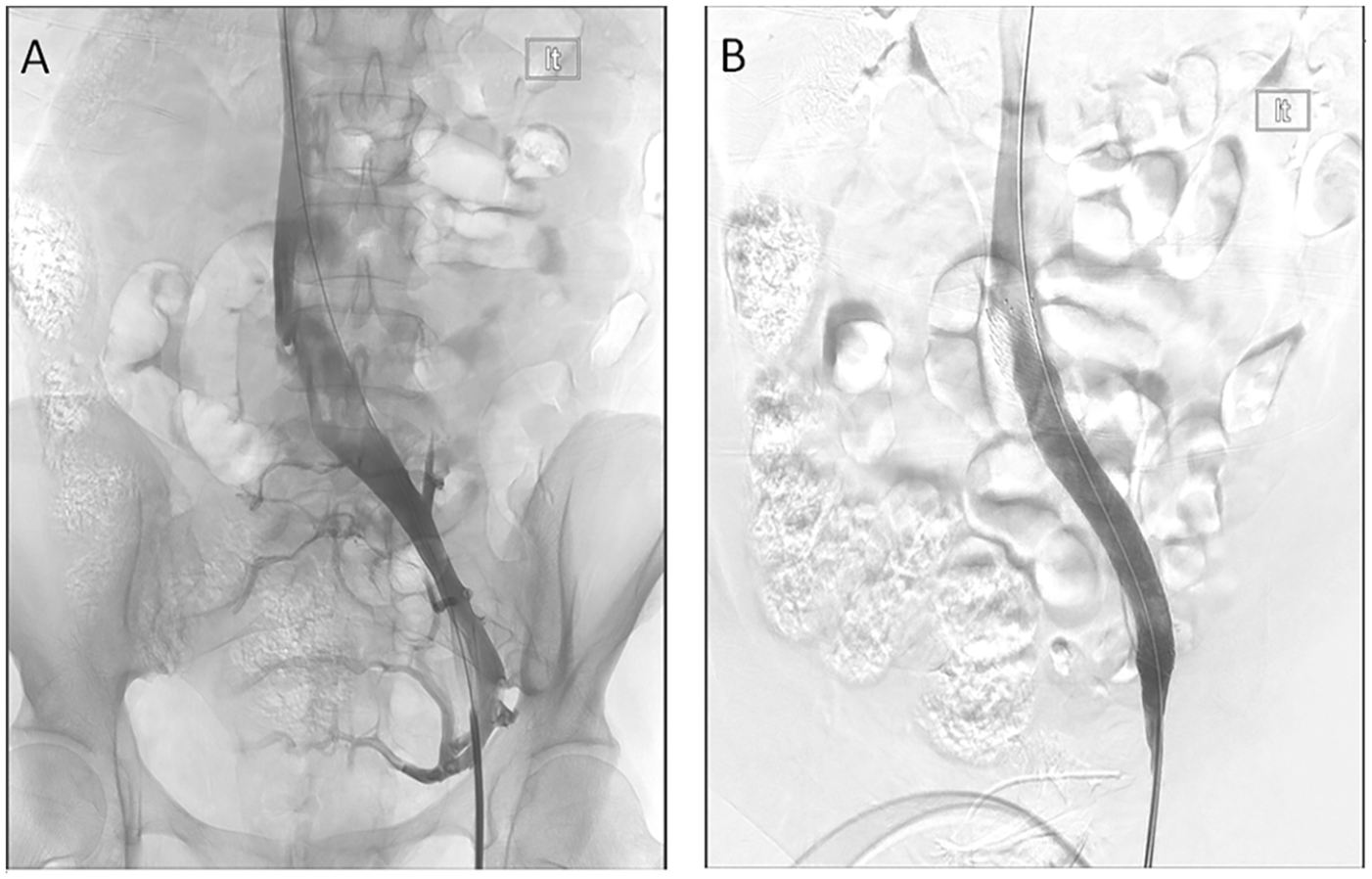
Venographic findings in left common iliac vein compression (May-Thurner syndrome). (A) Venogram from the left common femoral vein demonstrated funnel-shaped left common iliac vein, with a lucent linear compression consistent with a crossing right common iliac artery, and collateral venous drainage via the internal iliac vein and ascending lumbar vein. (B) Post stenting venogram demonstrates improvement in flow with resolution of collaterals and rapid antegrade flow.
Brahmbhatt et al underscore venography’s unmatched precision in mapping venous anatomy and confirming reflux. 38 Its dynamic imaging capabilities allow for detailed assessment of reflux patterns, collateral pathways, and venous anatomy, making it indispensable in guiding interventional procedures like embolization or stenting. Sensitivity and specificity remain consistently high across studies, validating its role as a diagnostic gold standard.
Venography is invasive, carrying risks such as infection, bleeding, and contrast-induced nephropathy, all of which require careful consideration before use. 23 It demands specialized expertise and facilities to perform accurately, limiting its availability in general clinical settings. Despite these challenges, venography’s precision in assessing dynamic venous flow and mapping complex venous anatomy makes it invaluable for guiding therapeutic interventions in cases of PeVD. The technique’s invasive nature restricts large-scale studies. Findings often highlight venography’s role as a confirmatory test rather than a first-line option.35,39
When venographic findings confirm clinically significant venous reflux or obstruction, interventional radiology (IR) techniques such as embolization and venous stenting may be pursued. Embolization involves the use of coils, plugs, or sclerosants to occlude refluxing ovarian or internal iliac veins, alleviating venous congestion and improving symptoms. It is often performed during the same session as diagnostic venography and can be done on a day-case basis. Venous stenting is reserved for cases of mechanical obstruction, such as May-Thurner syndrome, where restoring vessel patency is necessary. These minimally invasive, fluoroscopy-guided procedures have demonstrated favourable long-term outcomes and safety in appropriately selected patients. Long-term data support their safety and efficacy, with low complication and recurrence rates.6,23
Conclusion
CPP significantly lowers the quality of life for affected individuals, particularly reproductive-age women. Within this spectrum, PeVD remains an underdiagnosed yet crucial contributor, often overshadowed by more commonly recognized etiologies. Advanced imaging modalities, including ultrasound, CT, MRI, and venography, provide vital tools for identifying and characterizing PeVD, enabling targeted management strategies. Each modality has unique strengths, from the accessibility and real-time ultrasound assessment to the dynamic flow analysis of advanced MRI sequences and the precise anatomical mapping of venography. However, limitations such as incidental findings complicating clinical interpretation and resource constraints for advanced imaging highlight the importance of thoughtful application. By combining modality-specific insights with tailored procedural techniques and recognizing PeVD-specific considerations, radiologists can enhance diagnostic accuracy and treatment outcomes.
The comprehensive evaluation and management of PeVD and its associated conditions demand a truly interdisciplinary approach. Radiologists play a pivotal role in diagnosing and guiding minimally invasive interventions, while collaboration with allied health professionals, gynecologists, and vascular specialists ensures holistic care. Timely and sufficient referral pathways are essential to bridge gaps in diagnosis and treatment, particularly for patients in marginalized communities disproportionately affected by these disorders. By fostering integrated teamwork and leveraging advanced imaging modalities, healthcare providers can improve diagnostic precision, streamline therapeutic interventions, and ultimately enhance patient outcomes.
Supplemental Material
sj-docx-1-caj-10.1177_08465371251336121 – Supplemental material for Imaging-Based Approach to Venous-Origin Chronic Pelvic Pain
Supplemental material, sj-docx-1-caj-10.1177_08465371251336121 for Imaging-Based Approach to Venous-Origin Chronic Pelvic Pain by Vaneeza Moosa, Tiffany Ni, Inbar Friedman and Andrew D. Brown in Canadian Association of Radiologists Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
