Abstract
Acute non-traumatic pelvic pain represents a frequent and complex diagnostic dilemma in pre-menopausal females presenting to the emergency department due to overlapping symptomatology across pregnancy and non-pregnancy related causes. Radiologists play a pivotal role in expediting accurate diagnosis and guiding appropriate management in these potentially life-threatening scenarios. This review provides an approach to workup and imaging selection in these patients, emphasizing the necessity of serum β-hCG testing and the central role of transabdominal and transvaginal pelvic ultrasound. An overview of female pelvic anatomy is provided. Using a multimodality imaging approach, early pregnancy related complications such as ectopic pregnancy, retained products of conception, and gestational trophoblastic disease and non-pregnancy related causes of acute pelvic pain such as ovarian torsion, ruptured ovarian cysts, pelvic inflammatory disease, endometriosis, uterine vascular malformation, ovarian vein thrombosis, ovarian hyperstimulation syndrome, and intrauterine device complications are reviewed. Finally, we propose an algorithmic approach to imaging selection and interpretation tailored to the clinical scenario, laboratory findings (notably β-hCG status), and patient demographics. This structured framework aims to support radiologists in efficiently narrowing the differential diagnosis and optimizing patient outcomes in acute, non-traumatic pelvic emergencies.
Introduction
Acute non-traumatic pelvic pain in females is a common and diagnostically challenging presentation in the emergency department (ED). Defined as pain lasting <3 months, 1 with causes ranging from benign to life-threatening, the overlapping clinical features of the various gynecologic and pregnancy-related causes can complicate diagnosis.
This review provides an overview of pelvic anatomy to support interpretation of imaging findings. First-line imaging selection is discussed in the context of key clinical and laboratory data, with particular emphasis on serum β-human chorionic gonadotropin (β-hCG) levels. Ultrasound (US) remains the primary modality for initial assessment in most cases, with the roles of computed tomography (CT) and magnetic resonance imaging (MRI) reviewed where applicable. An imaging-focused review of early pregnancy-related and gynecologic causes of acute pelvic pain is provided (Table 1). Finally, an algorithmic approach to imaging selection and interpretation is proposed to aid radiologists in evaluation of these complex presentations.
Gynecologic and Pregnancy-Related Causes of Acute Non-Traumatic Pelvic Pain.

Not included in this imaging review.
Anatomy
The female pelvis is a confined anatomical space housing multiple organ systems in close proximity. It is functionally divided into 3 compartments: the anterior compartment, containing the urinary bladder and distal ureters; the middle (gynecologic) compartment, containing the uterus, fallopian tubes, ovaries; and the posterior compartment, containing the sigmoid colon, rectum, and anal canal.
The uterus is centrally positioned, comprised of the fundus, body, isthmus, and cervix, and supported by the broad, round, uterosacral, and cardinal ligaments. Laterally, the fallopian tubes extend from the uterine cornua toward the ovaries, which reside posterolateral to the uterus near the pelvic sidewalls. The ovaries receive dual arterial supply from the ovarian (branching directly from the abdominal aorta) and uterine arteries (branches of the internal iliac arteries), while venous drainage occurs through a complex network of ovarian, uterine, and pelvic venous plexuses.
The urinary bladder lies anterior to the uterus, while the sigmoid colon and rectum course posteriorly. The ureters descend along the lateral pelvic walls, crossing beneath the uterine arteries before inserting into the posterior bladder wall. Additionally, bowel loops frequently occupy parts of the pelvis.
The pelvic peritoneum, a serous membrane composed of visceral and parietal layers, reflects over these structures to form the pelvic peritoneal spaces. It covers the fundus of the bladder, the anterior and posterior surfaces of the uterus, and descends anterior to the rectum, creating 2 potential spaces: the uterovesical pouch (between the bladder and uterus) and the rectouterine pouch (pouch of Douglas), which is the most dependent portion of the pelvis in the supine position.
This compact arrangement of structures creates overlap in both clinical presentation and cross-sectional imaging findings when evaluating acute pelvic pain (Figure 1).
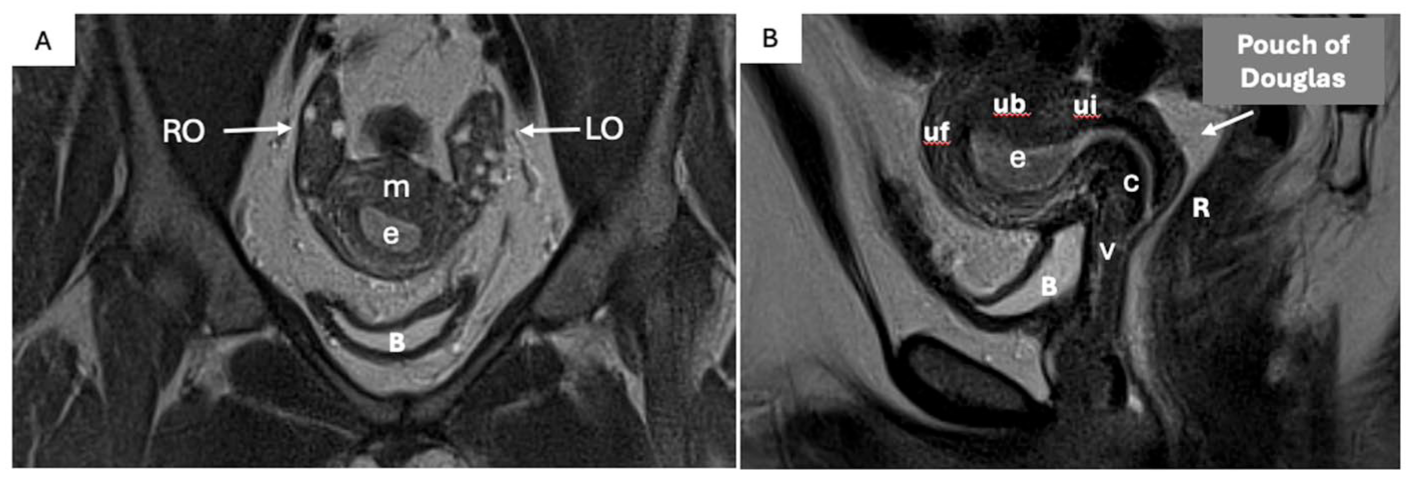
Female pelvic anatomy. Coronal (A), and sagittal (B) T2W non-FS MRI images of the pelvis. Labeled anatomy as follows; bladder (B), rectum (R), uterine fundus (uf), uterine body (ub), uterine isthmus (ui), endometrium (e), myometrium (m), left and right ovaries (LO and RO), cervix (c), and vagina (v).
Imaging Modalities
Imaging selection in the evaluation of acute pelvic pain is guided by clinical suspicion, laboratory findings—particularly serum β-hCG levels—and the need for rapid, accurate diagnosis. According to the American College of Radiology (ACR) Appropriateness Criteria, US is the first-line imaging modality in most cases of acute, non-traumatic pelvic pain in premenopausal females. 1 Ultrasound offers several advantages: it is widely available, cost-effective, and does not expose patients to ionizing radiation, which is especially important in those of reproductive age and in early pregnancy. Furthermore, transvaginal ultrasound provides high-resolution visualization of the uterus and adnexal structures, enabling accurate detection of conditions such as ectopic pregnancy and ovarian torsion. The addition of color and spectral Doppler imaging enhances assessment of vascular flow. Real-time dynamic assessment, including evaluation for tenderness and mobility of structures, further augments diagnostic accuracy.1,2
Despite US being the initial modality of choice, CT and MRI play important complementary roles. CT is particularly useful when non-gynecologic or gastrointestinal causes of pelvic pain are suspected, such as appendicitis or diverticulitis, and may also help identify complications of gynecologic pathology, including hemorrhage or abscess formation.1-3 MRI, while less accessible in the acute setting, offers superior soft tissue contrast and is valuable in problem-solving scenarios, especially when ultrasound findings are inconclusive or further tissue characterization is required. MRI is also preferred in pregnant patients when additional imaging is necessary and ionizing radiation should be avoided. 1
Pregnancy-Related Causes of Acute Pelvic Pain
Ectopic Pregnancy
Ectopic pregnancy refers to the implantation of a fertilized ovum outside the endometrial cavity, most commonly within the fallopian tube. Accounting for ~1% to 2% of pregnancies, it remains a leading cause of first-trimester maternal mortality. 4 Risk factors include previous ectopic pregnancy, pelvic inflammatory disease, prior tubal or uterine surgery, assisted reproductive technology, and the presence of an intrauterine device. 5
US is the preferred imaging modality. The absence of an intrauterine gestational sac with a positive β-hCG should raise concern for an ectopic pregnancy. However, visualization of a gestational sac is often limited with a β-hCG of <2000 IU. 5 Classic findings include a complex adnexal mass separate from the ovary with a thick echogenic rim and peripheral hypervascularity (Figure 2). 6 A live ectopic embryo with cardiac activity is diagnostic. If no gestational sac is visualized despite a positive β-hCG, it is termed a pregnancy of unknown location. 7 The presence of hemoperitoneum is suggestive of a ruptured ectopic. 6
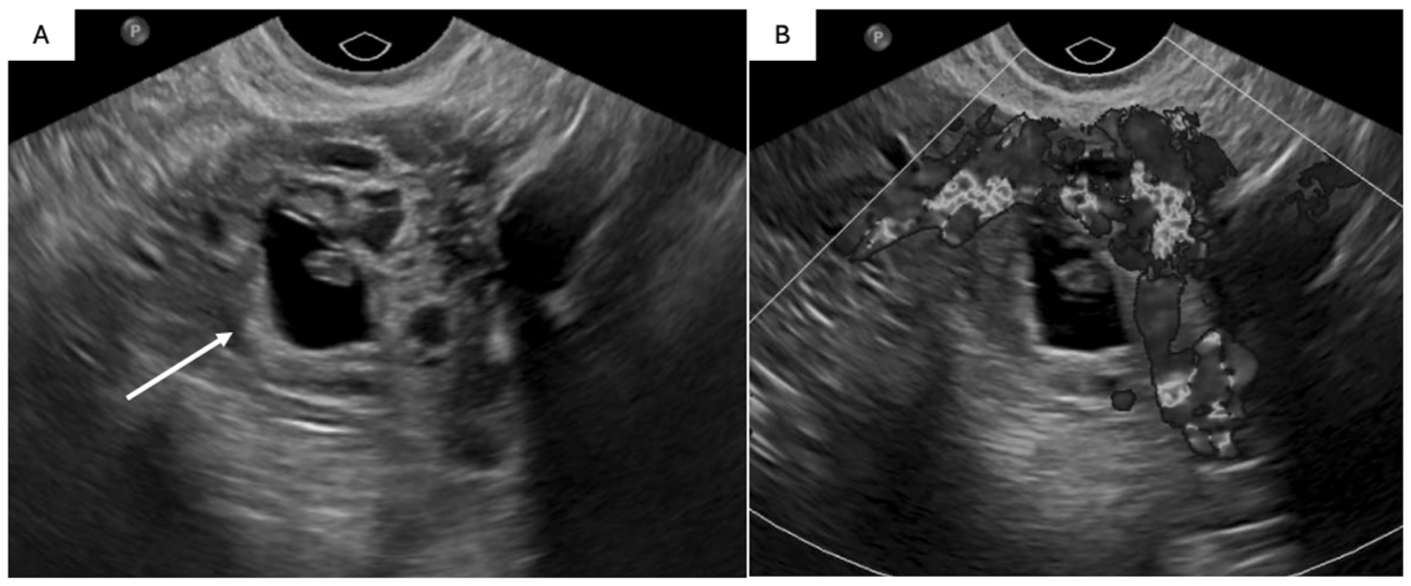
Left tubal ectopic pregnancy, pathology proven, in a 24 F with positive β-hCG and pelvic pain. Transvaginal ultrasound images of the uterus show no intrauterine gestational sac (not shown). Within the left adnexa, there is an extraovarian mass with a thick, hyperechoic rim (A; solid arrow), echogenic internal components, and (B) peripheral vascularity on Doppler assessment. With gentle probe pressure this was separate from the ovary.
Uncommon implantation sites include cesarean scar, interstitial (cornual), angular, cervical, and abdominal locations. Cesarean scar ectopic pregnancies implant at the site of prior uterine incision, appearing as gestational sacs embedded in the anterior lower uterine segment with a thin or absent myometrial mantle, typically <5 mm. 8 Interstitial pregnancies occur in the intra-myometrial portion of the fallopian tube and appear as eccentrically located gestational sacs surrounded by <5 mm of myometrium, separate from the endometrial cavity. 9 These carry a higher risk of rupture and hemorrhage due to their proximity to uterine vasculature. Angular pregnancies implant medial to the utero-tubal junction but within the endometrial cavity and imaging features can mimic an interstitial ectopic. 9 Cervical pregnancies appear as gestational sacs within the cervical canal, often with an hourglass configuration. 10 Abdominal ectopic pregnancies are rare and may implant on peritoneal surfaces, bowel, or other abdominal organs. 10 Careful assessment of the gestational sac location, myometrial mantle, and relationship to uterine landmarks is essential for accurate diagnosis.
CT is not usually appropriate for assessment of suspected ectopic pregnancy. 1 MRI is not usually appropriate in the acute setting, however, can be useful in equivocal cases or when surgical planning is required. It may delineate gestational tissue, hemorrhage, and exact implantation site, particularly in cornual and cesarean scar ectopic pregnancies.10,11
Gestational Trophoblastic Disease (GTD)
GTD encompasses a spectrum of tumors with a wide range of biologic behavior and potential for distant metastasis including hydatidiform mole (complete and partial), invasive mole, choriocarcinoma, and placental site trophoblastic tumor, 12 the last 3 of which are termed gestational trophoblastic neoplasia (GTN). GTN refers the malignant entities on the spectrum of GTD. Molar pregnancies, often diagnosed while asymptomatic through routine ultrasounds and β-hCG testing, can present with symptoms such as first-trimester bleeding, nausea, and pelvic pain if the diagnosis is missed.
In patients with symptoms and β-hCG titers higher than expected for gestational age, US remains the mainstay for assessment. With complete hydatidiform mole (CHM), US commonly shows a heterogenous, echogenic mass with internal hypoechoic foci/cystic spaces (“snowstorm appearance” and/or “cluster of grapes”). Multiple large bilateral functional ovarian cysts may be seen (<20% of cases) called theca lutein cysts. With partial hydatidiform mole (PHM), US findings include; (1) an empty gestational sac or one containing amorphous echoes (fetal parts), (2) elongated or ovoid gestational sac, (3) fetal demise, anomalies such as growth restriction or oligohydramnios, (4) enlarged placenta with multiple internal cystic spaces (“Swiss cheese pattern”). 12 Treatment includes evacuation of the molar pregnancy with serial monitoring of β-hCG levels to exclude GTN.
Use of CT is typically reserved for evaluation of metastatic disease in cases of GTN. Similarly, there is little role for MRI, which is used only as a problem-solving tool when US non-diagnostic.
Retained Products of Conception (RPOC)
Defined as the persistence of placental and/or fetal tissue in the uterus following delivery, termination of pregnancy or miscarriage, RPOC usually presents as abnormal postpartum bleeding and is common, with an incidence of 3% to 5% after vaginal delivery.13,14 US is the first-line imaging modality for these patients and early diagnosis is critical for directing clinical management of bleeding and preventing complications such as infection. Treatment strategies are beyond the scope of this article, however, generally include expectant management, the use of uterotonic medications, or surgical interventions such as dilation and curettage.
The sensitivity and specificity of US is variable. The finding most suggestive for RPOC is a thickened endometrial echo complex (EEC), with cutoffs ranging in studies from 8 to 13 mm, or an endometrial mass. Color Doppler increases diagnostic confidence as blood clots are avascular and vascularity within a thickened endometrium or endometrial mass are likely to represent RPOC. In cases with significant internal vascularity, robust assessment is required to exclude the possibility of an arteriovenous malformation which is discussed later.
Gynecologic Causes of Acute Pelvic Pain
Ovarian Torsion
Torsion occurs when the ovary, and often the fallopian tube, partially or completely rotates along the axis of its vascular pedicle, leading to impaired lymphatic and venous drainage, and ultimately arterial compromise. This gynecologic emergency accounts for 2% to 3% of females who present with acute pelvic pain 15 and is more common in reproductive age. Clinical symptoms include sudden-onset, severe, unilateral lower abdominal pain, nausea, and vomiting. Ovarian enlargement increases the tendency of the ovary to twist on its ligaments, thus the presence of an ovarian lesion is the single greatest risk factor for torsion. 15 Adnexal torsion can occur in normal ovaries, particularly in pediatric or pregnant patients. Early diagnosis and surgical detorsion are essential to preserve ovarian function.
Ultrasound is the first-line imaging modality with typical US features include an enlarged ovary (>4 cm) with peripheral distribution of follicles (“string of pearls”) and heterogeneous or hyperechoic central stroma (Figure 3). The “whirlpool sign,” representing a twisted vascular pedicle, may be seen adjacent to the ovary. Color Doppler may show absent or reduced venous and arterial flow, though preserved flow does not exclude torsion due to the dual ovarian blood supply. Associated findings include free fluid in the pelvis and the presence of a lead mass, particularly simple cysts or mature cystic teratomas. 15
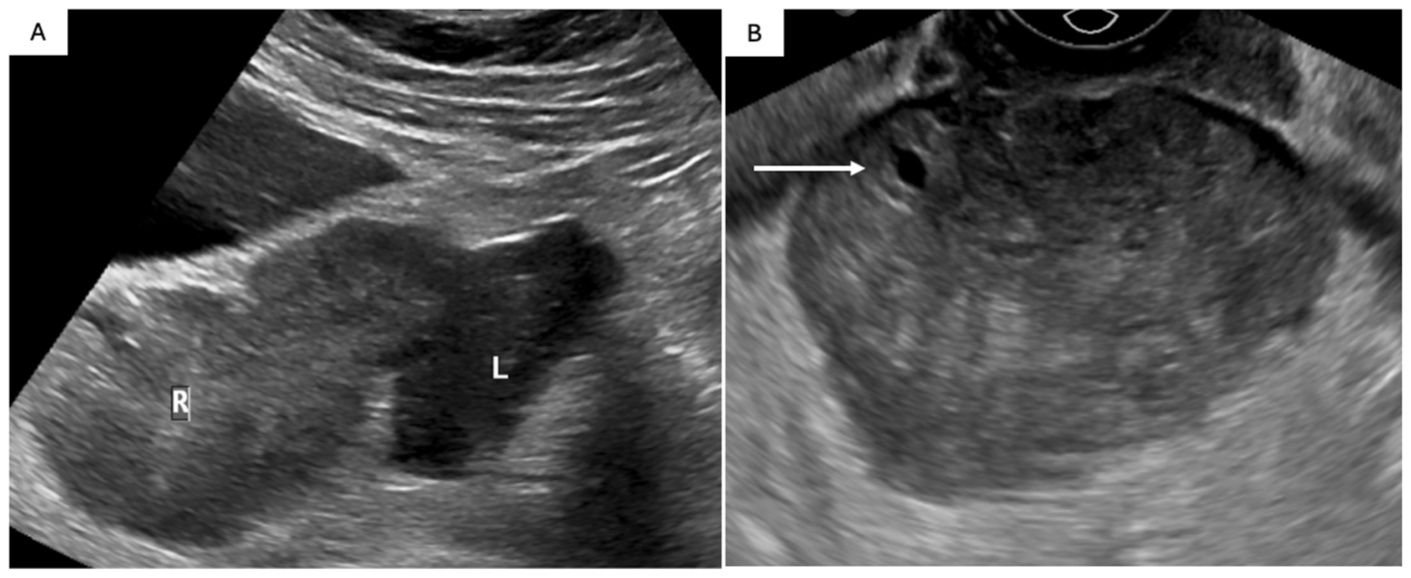
41 F prior hysterectomy with 2 days history of RLQ pain. Transabdominal (A) and transvaginal (B) sonographic images of the pelvis. On transabdominal US the right ovary (R) is enlarged and medialized (A), positioned adjacent to the left ovary. On transvaginal US, the right ovary is again enlarged with heterogenous stroma and a peripheral follicle (B; arrow). On Doppler assessment (not shown) there was no detectable internal vascularity. Patient underwent emergency laparoscopy revealing an ischemic right ovary with at least 5 twists of the vascular pedicle.
CT and MRI are not first-line imaging modalities, although CT may be the first imaging modality performed depending on the clinicians initial assessment. CT may show an enlarged, edematous, medialized ovary with surrounding fat stranding and/or small-volume free fluid, deviation of the uterus toward the torsed side, thickened fallopian tubes, and absence of contrast enhancement.15,16 A twisted pedicle may be identified as a swirling soft tissue structure adjacent to the ovary. Hemoperitoneum, when present in the context of adnexal torsion, suggests ovarian infarction and necrosis. 15 Its recognition is vital because it marks a transition from potentially reversible torsion to infarction, guiding the urgency of surgery, patient counseling, and whether ovarian conservation is feasible.
MRI, although not commonly required, can be useful for problem-solving and may show an enlarged midline ovary with T2 hyperintensity due to stromal edema, peripheral T2W follicles (“string of pearls”), and lack of enhancement in the context of ovarian infarction if performed with gadolinium (Figure 4). As above, a benign lead-point mass is often readily identified on MRI, and the uterus may be pulled to the involved side. A twisted pedicle may appear as a target-like or concentric flow void pattern, often with soft tissue thickening of the pedicle. If infarcted, diffusion restriction may be present.15,16
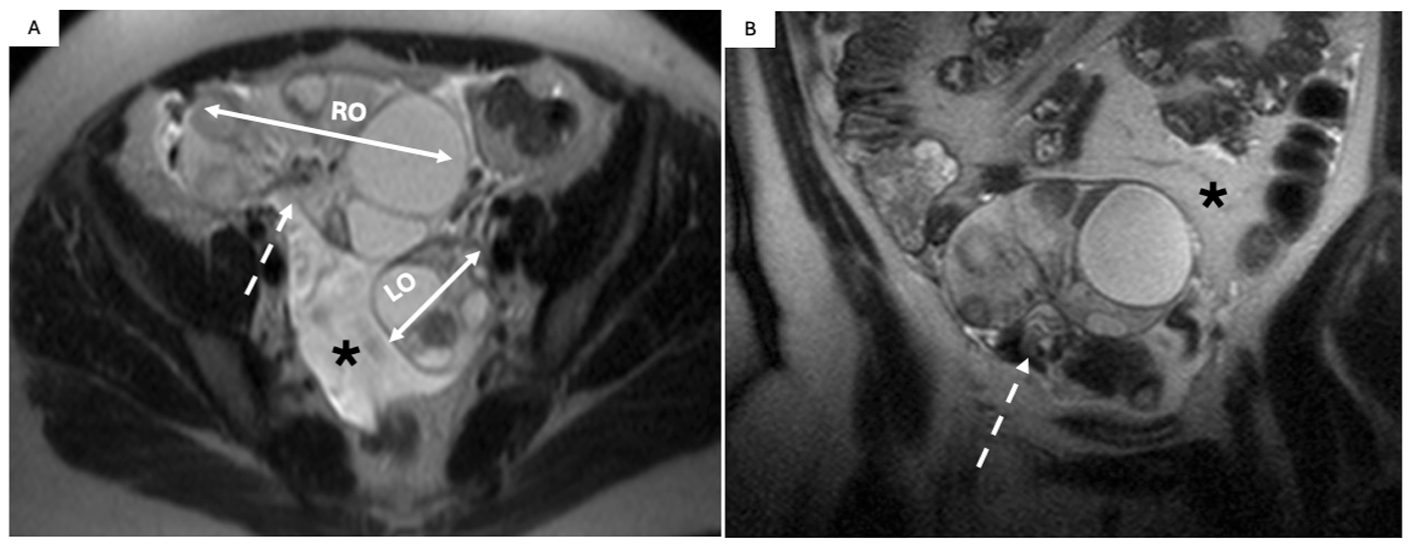
Right adnexal torsion in a 27 F undergoing fertility treatment with intrauterine pregnancy (GA 9 + 5 weeks) and ovarian hyperstimulation syndrome (OHSS) presenting with RLQ pain. Appendix not identified with US and MRI requested to rule out appendicitis and was performed without contrast in this pregnant patient. T2W non-FS MRI images in the axial (A) and coronal plane (C) show imaging findings of OHSS including ascites (*), and enlarged bilateral ovaries with a “spoke wheel” appearance. There is a notable discrepancy in ovarian size (R > L; double head arrows). The right ovary demonstrates T2 hyperintense stromal edema with peripheralization of follicles and swirling and thickening of the right vascular pedicle (A and B; dash arrow). Intraoperative findings included enlarged, purple, and edematous right ovary with >270° of torsion.
Hemorrhagic Cyst ± Rupture
Physiologic changes during the menstrual cycle lead to increased ovarian vascularity during the luteal phase which can cause ovarian cyst hemorrhage and/or rupture 17 ; the most common cause of acute pelvic pain in premenopausal females. Ultrasound is the first line imaging modality. When the clinical diagnosis is less certain or the patient is hemodynamically unstable, CT may be performed as first line imaging. MRI is not a first-line imaging tool, instead used for problem solving or follow-up where appropriate.
On US, a hemorrhagic cyst usually appears as a unilocular cyst without internal vascularity and at least one of (1) reticular pattern (fine, thin intersecting lines representing fibrin strands), (2) retractile clot (intracystic component with straight, concave, or angular margins)18–20 (Figure 5B). Management for hemorrhagic or suspected hemorrhagic cysts is clearly defined by the ACR18–20 and not covered in this review. In cases of rupture, hemoperitoneum is seen as free fluid containing low-level internal echoes. When CT is performed, a hemorrhagic cyst appears as a complex adnexal mass with mixed internal attenuation (25-100 HU), commonly with fluid-debris or fluid-fluid levels 21 (Figure 5A). With rupture, a collapsed adnexal cyst with wall discontinuity can be seen, often with surrounding hyperattenuating “sentinel clot.” High density fluid (hemoperitoneum), typically 45 to 100 HU, can be seen layering in the dependent portions of the pelvis and abdomen. 22 Occasionally active bleeding is detected on contrast enhanced images as a serpiginous blush of contrast adjacent to a complex adnexal mass.
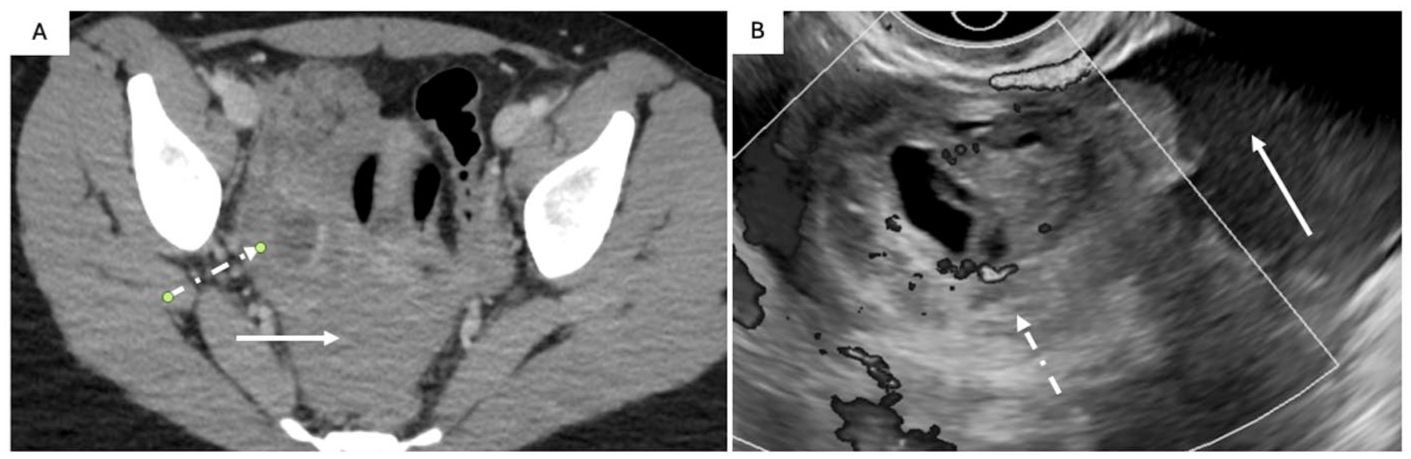
Ruptured hemorrhagic ovarian cyst in a 40 F presenting with acute onset pelvic pain and negative β-hCG. CT was selected as the first imaging modality as the differential included renal colic and appendicitis. Axial (A) contrast-enhanced CT scan shows a complex observation in the right adnexa (A; dot and dash arrow) with high attenuating fluid within the pelvis (HU 64) (A; solid arrow). Transvaginal greyscale ultrasound with Doppler (B) shows free fluid with low-level internal echoes (B; solid arrow) consistent with hemoperitoneum. The right ovary demonstrates normal Doppler color signal and a complex cystic observation with avascular echogenic component with angular/straight margins (clot) (B; dot and dash arrow) consistent with a ruptured hemorrhagic cyst.
Pelvic Inflammatory Disease (PID)
PID refers to an ascending infection of the upper genital tract, commonly due to sexually transmitted organisms. It encompasses a clinical and imaging spectrum including endometritis, salpingitis, oophoritis, hydrosalpinx, pyosalpinx, and tubo-ovarian abscess (TOA). While imaging plays a supportive role, PID remains largely a clinical diagnosis based on history, physical exam, and laboratory findings. Clinical features include fever, lower abdominal or pelvic pain, cervical motion tenderness, vaginal discharge, and an elevated white blood cell count. Risk factors include age 15 to 25, intercourse with multiple partners, prior episode of PID, insertion of an intrauterine device (IUD), and a history of tubal ligation. 23
US findings can include enlarged ovaries and hyperemia (oophoritis), distended fluid-filled fallopian tubes (hydrosalpinx), or complex adnexal observations with internal echoes or hyperechoic debris (pyosalpinx or TOA). Nodular infoldings/projections arising from the lumen, likened to that of a cogwheel, can be seen with hydrosalpinx or pyosalpinx. Layering echogenic debris within the tube may represent pus or blood products. TOAs appear as multiloculated, tubular, complex observations inseparable from the ovary and uterus, often with thick walls and internal septations. Doppler may show peripheral or septal hyperemia. 24 Pelvic free fluid may be present, which can be simple, complex, and/or loculated.
Contrast enhanced CT (CECT) may be performed first due to a nonspecific presentation. CT findings include thickened uterosacral ligaments, pelvic inflammatory changes and fat stranding with obscuration of fascial planes, and reactive lymphadenopathy. 24 Pyosalpinx or TOA present as tubular, serpiginous rim-enhancing adnexal structures with surrounding parametrial inflammatory changes, and reactive lymphadenopathy 24 (Figure 6A).
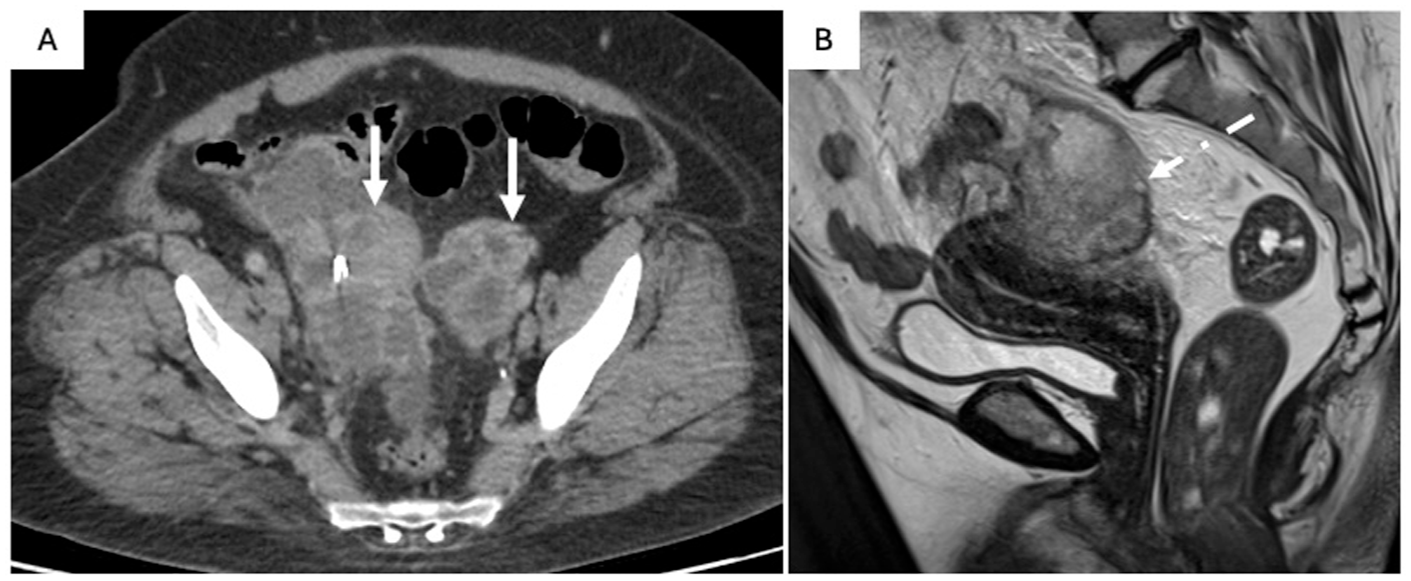
Bilateral tubo-ovarian abscesses in a 46 F presenting with pelvic pain, fever, and a negative betahCG. Axial (A) contrast-enhanced CT image show complex adnexal masses (A; solid arrow) with significant surrounding inflammatory fat stranding and reactive thickening of traversing bowel loops. Sagittal (B) T2W non-FS MRI image shows a thick-walled, complex adnexal mass with internal T2 heterogeneity (B; dash arrow).
MRI can further characterize indeterminate adnexal observations. TOA findings include T1 hypointense, T2 hyperintense multiloculated observations with thickened, enhancing walls and septa. Restricted diffusion within abscesses and enhancement of inflamed endometrial or tubal structures may be seen, often accompanied by surrounding inflammatory changes, including T2-hyperintense fluid tracking throughout the pelvis25,26 (Figure 6B).
Radiologic evaluation must assess for complications of PID. Possible complications include TOA rupture with peritonitis, involvement of adjacent structures, perihepatitis (Fitz-Hugh–Curtis syndrome), and ovarian vein thrombophlebitis. Peritonitis secondary to TOA rupture manifests with abdominal pain, fever, and leukocytosis. CT may reveal ascites, fat stranding, and diffuse peritoneal thickening and enhancement. 24 Ovarian vein thrombophlebitis is a rare but important complication. Perihepatitis, or Fitz-Hugh–Curtis syndrome, refers to perihepatic inflammation secondary to ascending infection and presents with right upper quadrant (RUQ) or pleuritic chest pain. US can show free fluid in the RUQ, and CT may demonstrate capsular enhancement of the liver and perihepatic free fluid. 24
Endometriosis
Endometriosis affects ~10% of reproductive-age individuals assigned female at birth in Canada. 27 Chronic or relapsing/remitting pelvic pain is the most common symptom but individuals with acute on chronic pain can present to the ED for assessment. The gold-standard for disease assessment is laparoscopy, but US and MRI are useful imaging tools. The CAR released practice statements on the use of pelvic US and MRI for endometriosis.27,28 Endometriosis primarily affects the extra-uterine and extra-ovarian structures, thus basic pelvic US is inadequate and “advanced ultrasound” is the recommended technique. This includes a structural assessment of the uterus and ovaries, and comprehensive assessment for deep endometriosis within the anterior and posterior compartments, observation of the relative positioning of the uterus and ovaries to assess for abnormal positioning due to adhesions, and real-time maneuvers such as the “uterine sliding sign.” A comprehensive review of the imaging features of endometriosis is beyond the scope of this article.
Uterine Fibroid Complications
Uterine fibroids or leiomyomas can present with acute complications including degeneration, torsion, and prolapse—each of which carries unique clinical and imaging features.
Degeneration occurs most commonly when a fibroid outgrows its blood supply and can cause acute pelvic pain. There are multiple types of degeneration, which is beyond the scope of this review. Degeneration may be spontaneous or iatrogenic, following uterine artery embolization. Pain control following this procedure can be difficult, resulting in patients presenting to the ED.
Torsion of a fibroid is rare but clinically important, with symptoms often mimicking adnexal torsion. Torsion most frequently occurs in FIGO type 7 subserosal pedunculated fibroids which are connected to the uterus by a narrow stalk, predisposing them to vascular compromise. 29 Patients present with acute lower abdominal pain. US may show a peri-uterine lesion with absent internal vascularity or “whirlpool” sign on Doppler assessment at the level of the vascular pedicle with adjacent inflammatory changes. CT and MRI, although not first-line modalities, can reveal a non-enhancing fibroid and congestion at the site of torsion of the vascular pedicle.30,31 MRI demonstrates a heterogeneous T2 hyperintense mass with absent enhancement, often associated with restricted diffusion if infarcted.31,32
Prolapse can occasionally occur through the endocervical canal into the vagina, presenting with vaginal bleeding, pelvic pressure, or abnormal discharge and is most commonly a complication of FIGO type 0 submucosal intracavitary pedunculated fibroids.29,33 US may identify a hypoechoic mass extending from the endometrial canal into the cervix or vagina. MRI is often performed for better delineation of soft tissue and can show a T2 hypointense lesion protruding through the cervical os, with a stalk seen at the endometrial origin 32 (Figure 7).
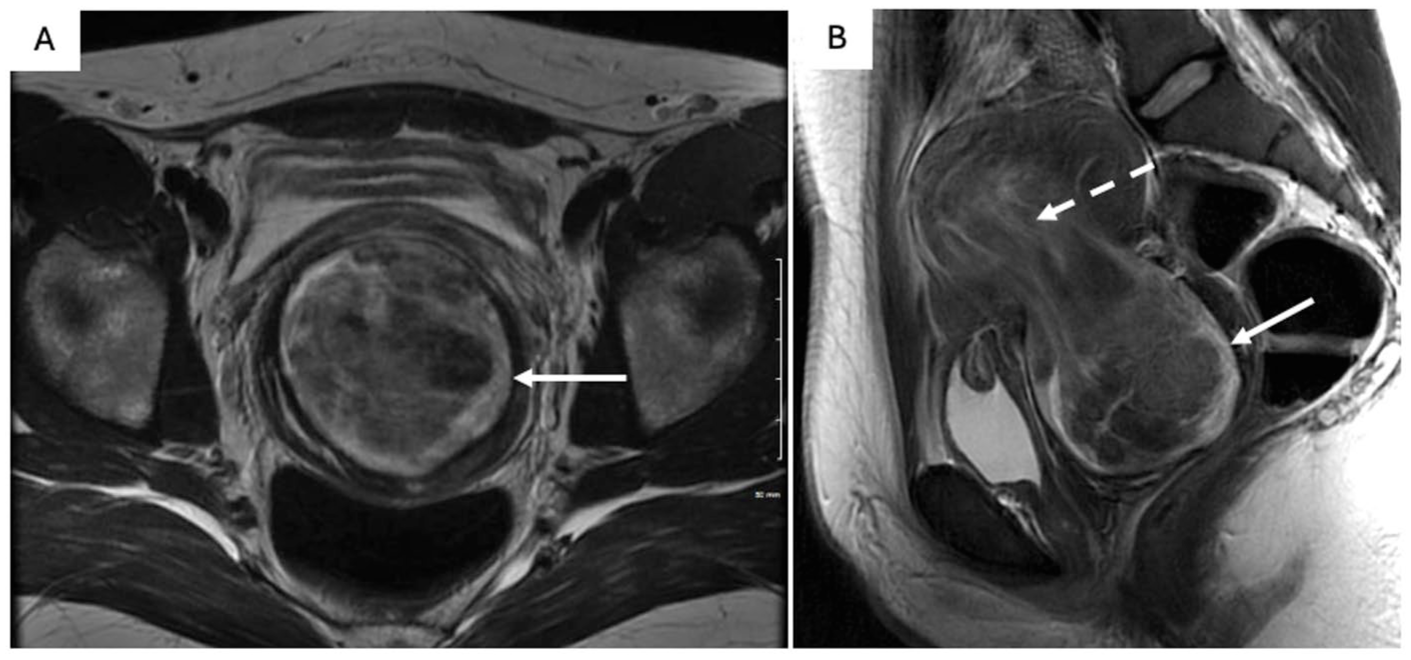
Prolapsed uterine fibroid in a 25 F with severe left lower quadrant pain and heavy vaginal bleeding. Axial (A) and sagittal (B) T2W images show a well-circumscribed predominantly T2 hypointense (A, B; solid arrow) observation which is protruding into the endocervical canal with possible extension beyond the external os into the vaginal canal via a widened cervix. A stalk (B; dash arrow) is noted along its superior-most aspect.
Uterine Vascular Malformations (UVM)
UVMs encompass a spectrum of rare anomalies, including arteriovenous malformations (AVMs), venous malformations, and mixed vascular channels. These may be congenital or acquired following interventions such as curettage or cesarean section. Clinically, patients present with abnormal uterine bleeding, often severe, which may be intermittent and refractory to medical treatment. Early and accurate identification is essential, as UVMs can result in life-threatening hemorrhage.
US with color Doppler is the primary imaging modality. Findings include a heterogeneous, ill-defined myometrial lesion with anechoic tubular channels (Figure 8A and B). Color and spectral Doppler demonstrate variable flow patterns depending on the subtype of malformation.34,35 CT can be helpful in the acute setting, particularly in cases of heavy bleeding and findings may include a bulky uterus with serpiginous or tubular enhancing myometrial vessels34,35 ± contrast blush in cases of active hemorrhage. At MRI, UVMs appear as serpiginous flow voids on T2W sequences and demonstrate avid enhancement post-contrast following the blood pool (Figure 8C). In some cases, they may extend into the endometrial canal or cervix and in these cases, MRI is helpful in differentiating AVMs from RPOC or neoplasm, which may have overlapping features. Dynamic contrast-enhanced MRI further aids in this distinction by providing temporal resolution of enhancement patterns and can identify feeding or draining vessels.34,35
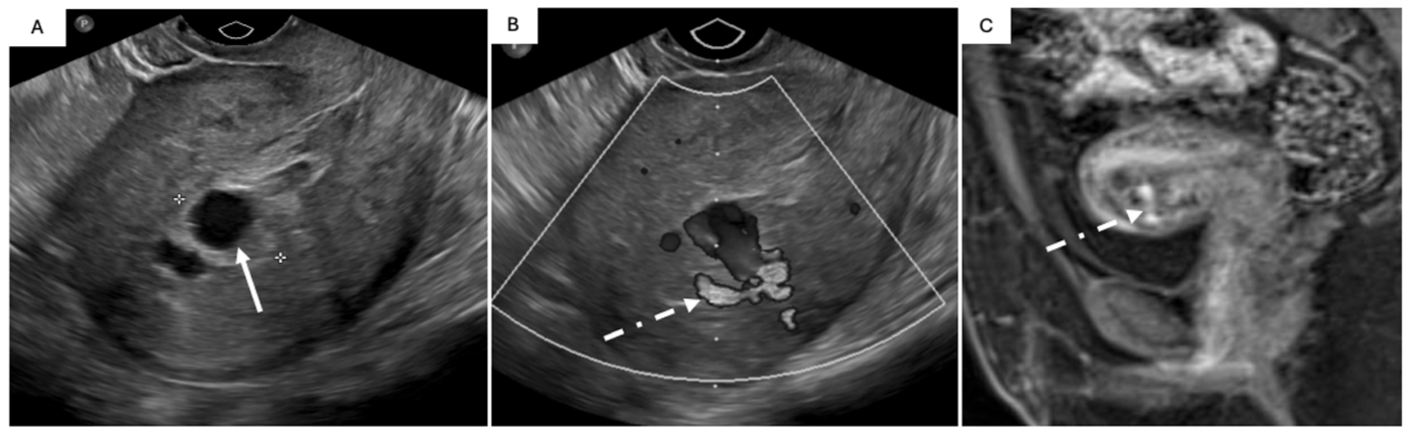
Uterine vascular malformation in a 40 F 12 days post-D&C for retained products of conception with worsening vaginal bleeding and passage of large clots (A, B) and a 32 F with history of ectopic pregnancy and D&C and US suspicious for UVM (C). Transvaginal US without (A) and with Doppler (B) shows an anechoic observation (A; solid arrow) within the endometrial cavity, continuous with a feeding vessel within the myometrium (B; dash arrow). Sagittal T1 post-contrast (C) MRI image show a serpiginous T1 hypointense (not shown) observation that follows the blood pool on post-contrast-enhanced images (C; dash arrow) traversing the anterior myometrium.
Ovarian Vein Thrombosis (OVT)
OVT may occur in the early postpartum period, following pelvic surgery, or in association with PID, malignancy, or thromboembolic disorders. 36 OVT may represent either bland thrombosis or infectious thrombophlebitis, particularly in the setting of PID. Early diagnosis is essential due to the risk of thrombus propagation and potentially life-threatening complications such as pulmonary embolism. Patients may present with nonspecific symptoms such as lower abdominal or flank pain, fever, or signs of sepsis, although many cases are asymptomatic and incidentally detected on imaging. 36
CECT is the imaging modality of choice with limited utility of US. Acute findings include an enlarged, tubular structure along the expected course of the ovarian vein, containing a central low-attenuation thrombus with possible peripheral rim hyper attenuation. 37 In the setting of thrombophlebitis, vessel wall thickening and surrounding fat stranding, reflecting inflammation, may be present. 24 It is imperative to note if the thrombus extends into the renal vein or inferior vena cava.
Ovarian Hyperstimulation Syndrome (OHSS)
OHSS is a complication related to assisted reproductive therapy representing an exaggerated response to the induction of ovulation which results in third spacing of fluids leading to edema, ascites, and pleural effusion. 38 Classification and symptoms of OHHS can range from mild (abdominal distension, pain, nausea, vomiting) to severe (hypercoagulability, hypotension, anuria). 38
Ultrasound is the mainstay for assessment, and the radiologist can provide supporting information for the severity of OHSS including ovarian size and presence of ascites and pleural effusion. In most cases, ovaries are enlarged (>5 cm) and contain follicles of varying sizes which are usually peripherally positioned, giving the “spoke-wheel” appearance 38 (Figure 4).
Intrauterine Device (IUD) Complications
IUDs are commonly used long-acting reversible contraceptives. They may be associated with complications including malposition, embedment, perforation, and infection. Risk factors for malposition or perforation include postpartum insertion, uterine anomalies, recent abortion or delivery, and operator inexperience. 39 A correctly positioned IUD should have the stem within the endometrial cavity with the arms extending laterally at the fundus. On physical exam, the IUD string should be seen through the external cervical os, typically extending into the vagina by 2.5 to 3 cm. 40
Ultrasound is the preferred modality for assessing IUD positioning. A properly positioned device appears as a linear hyperechoic structure in the endometrial canal, often with posterior acoustic shadowing. Positioning is assessed by determining the distance from the top of the IUD to the junction between the endometrium and fundal end of the uterine cavity, termed the “IUD-endometrium distance.” This measurement is best achieved in the longitudinal axis where the distance should be <3 mm. Greater distances are more commonly associated with symptoms such as bleeding and pain. The arms should extend laterally at the level of the uterine fundus parallel to the axis of the uterine cornua. Malpositioned devices may appear low in the uterine cavity, embedded in the myometrium, or oriented abnormally (Figure 9). Absent IUD visualization on US, especially with a missing string, raises concern for expulsion or migration.40,41 Hyperechoic foci with dirty posterior acoustic shadowing within the pelvis can represent pneumoperitoneum, concerning for bowel perforation as a complication.
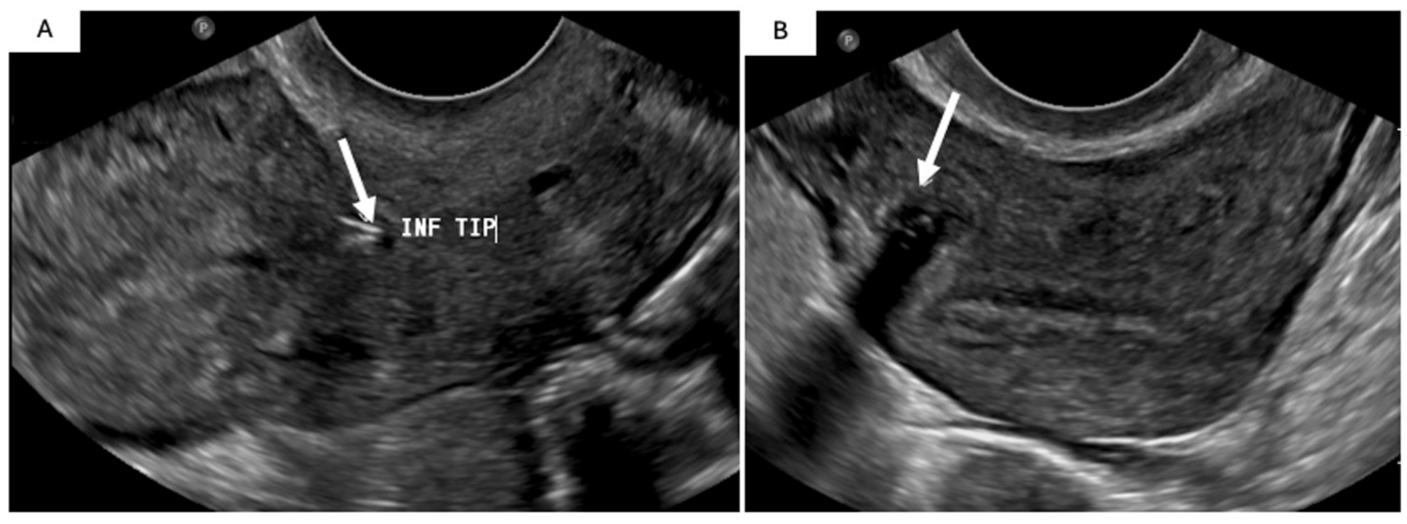
Malpositioned IUD in a 43 F presenting with a 1-month history of progressive abdominal cramping, vaginal bleeding and discharge. Transvaginal US images shows the IUD inferior tip at the level of the anterior lower uterine segment cesarean section scar (A; solid arrow). The IUD stem is embedded in the right anterolateral myometrium cranially beyond the scar toward the fundus (B; solid arrow).
Abdominal x-ray may be performed to assess for the IUDs presence if not seen on US. CT is particularly useful for identifying extrauterine IUDs and associated complications Hyperdense linear or T-shaped structures, representing the IUD, may be seen penetrating the myometrium, embedded in adjacent structures, or free within the peritoneal cavity. 41 Complications can include bowel perforation, pelvic abscess, or localized peritonitis.
Summary
An algorithmic approach to managing pelvic pain in the ED has been provided (Figure 10) and begins with a thorough clinical history and physical examination, which are critical in narrowing the differential diagnosis and guiding appropriate investigations. Key historical factors include pain onset, location, character, menstrual and sexual history, and associated symptoms such as bleeding. A pelvic and abdominal exam helps assess for tenderness, masses, or signs of peritonitis. As part of the initial workup, a urine or serum β-hCG test is essential to rule in/out pregnancy-related causes. Ultrasound is the first-line imaging modality due to its accessibility, safety, and high sensitivity for common gynecologic pathologies. Using this structured algorithm improves diagnostic accuracy, prioritizes life-threatening conditions, and supports timely, targeted treatment.
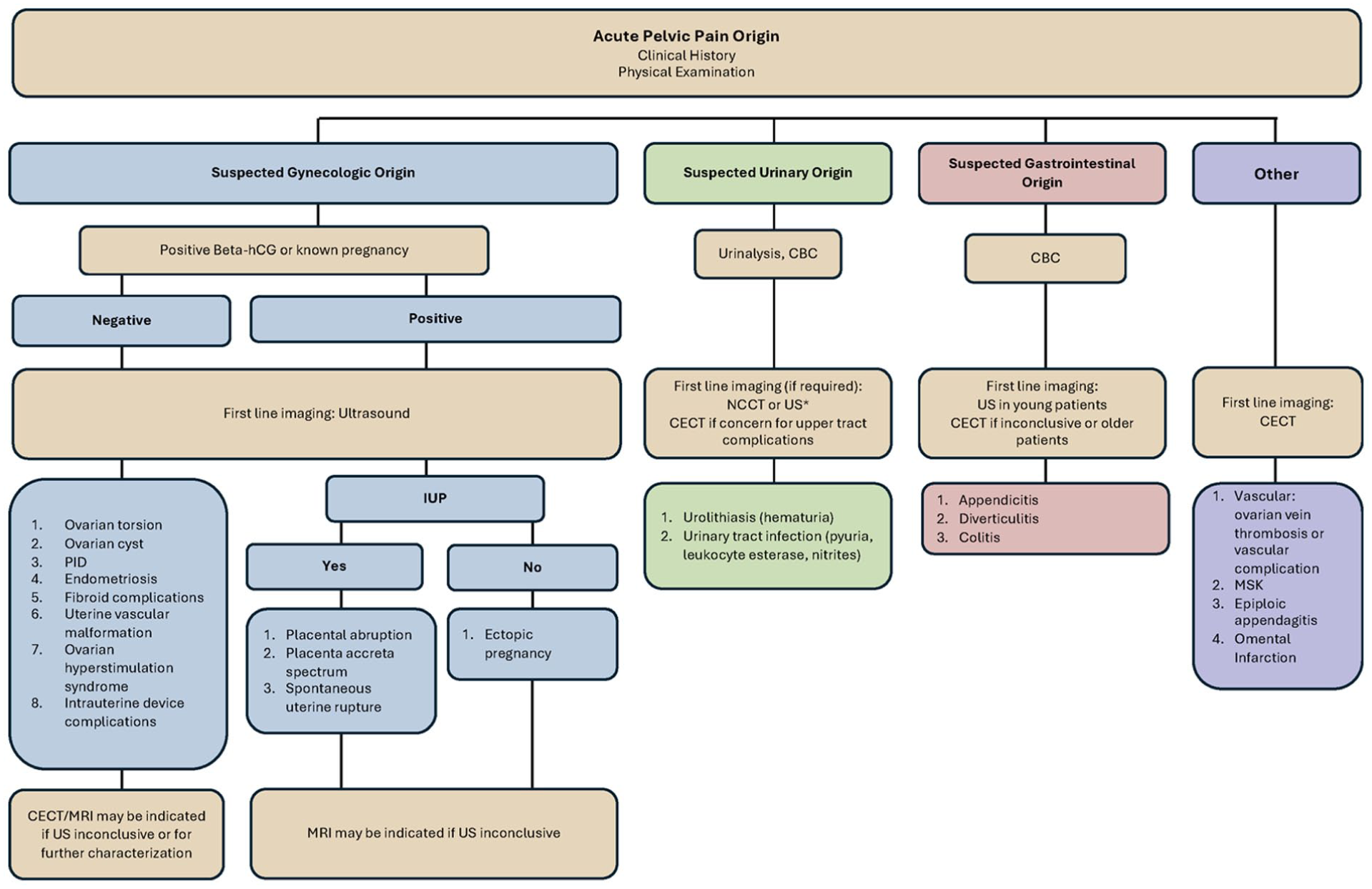
Diagnostic approach to common causes of acute non-traumatic pelvic pain.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
