Abstract
Functional and efficient medical equipment is at the core of modern healthcare delivery, particularly in medical imaging. Growing healthcare costs and constrained budgets can delay equipment renewal. Aging equipment risks malfunction, potentially causing injury to patients and staff, and downtimes delaying patient care and impacting departmental revenue. Extensive equipment failure can lead to significant operational disruption which can compromise the delivery of timely and quality healthcare. Although extensive equipment failure is uncommon, 2 interventional radiology divisions at tertiary academic hospitals in Canada and the UK recently faced such a crisis. Their experiences of crisis and recovery inform this review of angiography equipment failure, and the principles learned. The concept of organizational resilience is introduced as a framework through which we review the crises. This concept can be split into successive and cooperative stages of anticipation, coping, and adaptation. Resilient organizations can identify potential threats, cope with unexpected crises, and recover swiftly to ensure future success. The author’s experience of critical angiography unit failure, their response, and lessons learned are reviewed. We find these principles are broadly applicable to other medical imaging divisions and relevant to any system reliant on technology for healthcare delivery.
“If you run an airline, you had better know the state of your planes.” Healthcare and aviation industry comparisons are common as they operate in high-risk environments that prioritize reliability and failure prevention. Like pilots, Interventional Radiologists (IR) are dependent on equipment to perform procedures. Consider the following scenario: Your busy IR department has 4 angiography units nearing end of life. Financial constraints have delayed plans for renewal. The systems function with an occasional re-boot. One day, you arrive to discover the re-boot has not worked. Your preferred angiography unit is down. Over the coming days, you compensate by moving patients to your other angiography units. With repairs ongoing, the remaining angiography units fail as well. You are left without a quality angiography unit. Cases are backing up. Emails and phone calls from referrers won’t stop. What do you do?
While this scenario is extreme, this review will describe the experience as it unfolded in a large tertiary academic hospital in Canada. This scenario is not unique, as it also occurred in a tertiary academic hospital in the United Kingdom within the past 5 years. Both centres experienced multisystem angiography unit failures causing severe operational disruption. Considering hospital reliance on IR, this had a grave effect on patient throughput. The high reliability model of healthcare quality supposes performance reliability and a preoccupation with failure. 1 Preparation for this scenario asks several important questions: what backup is available to your angiography unit [equipment]? Should units offer procedures without an appropriate backup available? This editorial examines our experience of angiography unit failure with the aim of highlighting awareness and providing a management strategy. As will be demonstrated, this is a complex scenario that lacks evidence-based guidance but remains a critical patient safety issue. Using a framework of organizational resilience: a concept split into successive and cooperative phases of anticipation, coping, and adaptation; specific solutions are provided. 2 Our experience of angiography unit failure is used as a specific example but we find the principles are broadly applicable to other divisions of medical imaging and to any healthcare provider reliant on technology.
Anticipation
The anticipation stage of organizational resilience requires an ability to identify and prepare for threats. 2 Equipment failure can take many forms as malfunction of any component of the angiography unit can harm patients and/or staff. This can lead to delays in diagnosis and treatment of the patient and safety problems for both the patient and staff. 3 In the digital age, the first issue that comes to mind is a system shut-down or failure to start. However, other forms of system malfunction can lead to harm; this can include table/system movement or failure to move, excessive radiation dose, and physical injury from falling equipment. Each of these examples was a reported adverse event in a recent 10-year analysis of the US Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE). 4 As expected, software problems were the most common cause of a safety event (58.7%, 364 of 651), and the majority of safety events involved intra-procedural system shutdown (38.5%, 216 of 651). Twenty-six patient deaths were associated with intra-procedure system shutdown. These rare, but likely under-reported, events highlight the dangers of malfunctioning equipment. In addition, malfunctioning equipment has a detrimental impact on staff morale, department revenue, and timely patient care.5-7 One of the key questions regarding critical equipment is: can we predict the failure mode and timing and thus prepare an appropriate backup and replacement strategy? Manufacturer and society guidelines offer direction but in truth, we do not know.8,9
Original Equipment Manufacturer (OEM) Guidelines
Original Equipment Manufacturers (OEMs) provide an expected life cycle for each model. These timelines are typically based on the availability of equipment and technician expertise for each model, rather than the performance of a particular unit. OEMs define “end of life” and “end of support” thresholds. These thresholds vary depending on the OEM, equipment model, and contract. The authors discussions with the most common OEMs in Canada show that most manufacturers offer full support until an “end of life” threshold is reached. This mark is typically ~10 years after production has ended for a particular model. At this point, support may end or could be offered on a “best effort” basis until the “end of support” mark is reached; after which no further support is available from the manufacturers. There are independent companies which will continue to provide services to upkeep the equipment after this stage, but this period is often limited due to lack of parts, technician expertise, and issues of liability for aging equipment. A higher rate of wear-out failures is expected as equipment nears end of life but there is no threshold at which equipment replacement is mandatory (Figure 1). In practice, as most departments do not employ in-house medical engineering teams, it is the loss of OEM support which can often determine the life expectancy of equipment.
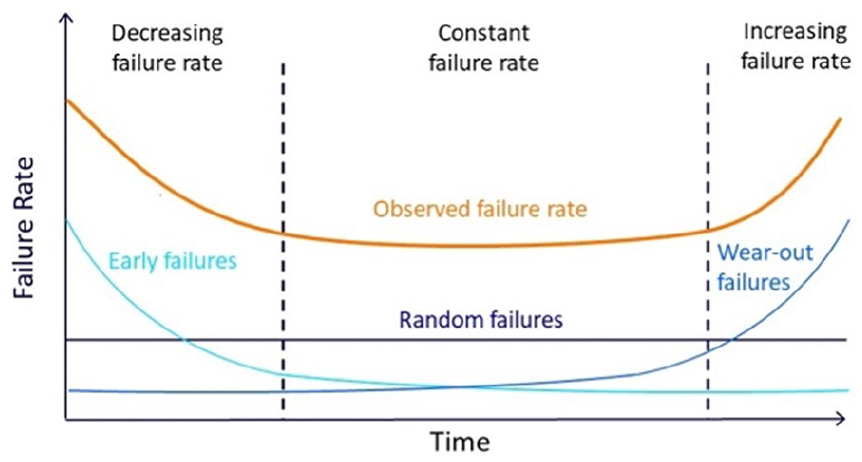
Bathtub curve illustrating the trend of equipment failures noted over the life cycle of an angiography unit.
Society Life Cycle Guidelines
Society guidelines determine life cycles for equipment renewal, typically based on a unit’s age and utilization. Table 1 summarizes the published international renewal guidelines for single/biplane angiography units as a reference. Most guidelines advise a life expectancy of 10 years for angiography units with moderate utilization (2000-4000 exams/year).3,10 Similar thresholds are available for radiology equipment used in other divisions of medical imaging.3,10,11 It must be acknowledged that the basis for the chosen numbers is often not specifically outlined. Replacement planning for any piece of medical imaging equipment over 10 years old is considered essential to avoid reaching a maximum life expectancy and clinical relevance threshold of 15 years for any technology.3,10,12
Life Expectancy Reported for Interventional Angiography Units (Single/Biplane) in International Guidelines.
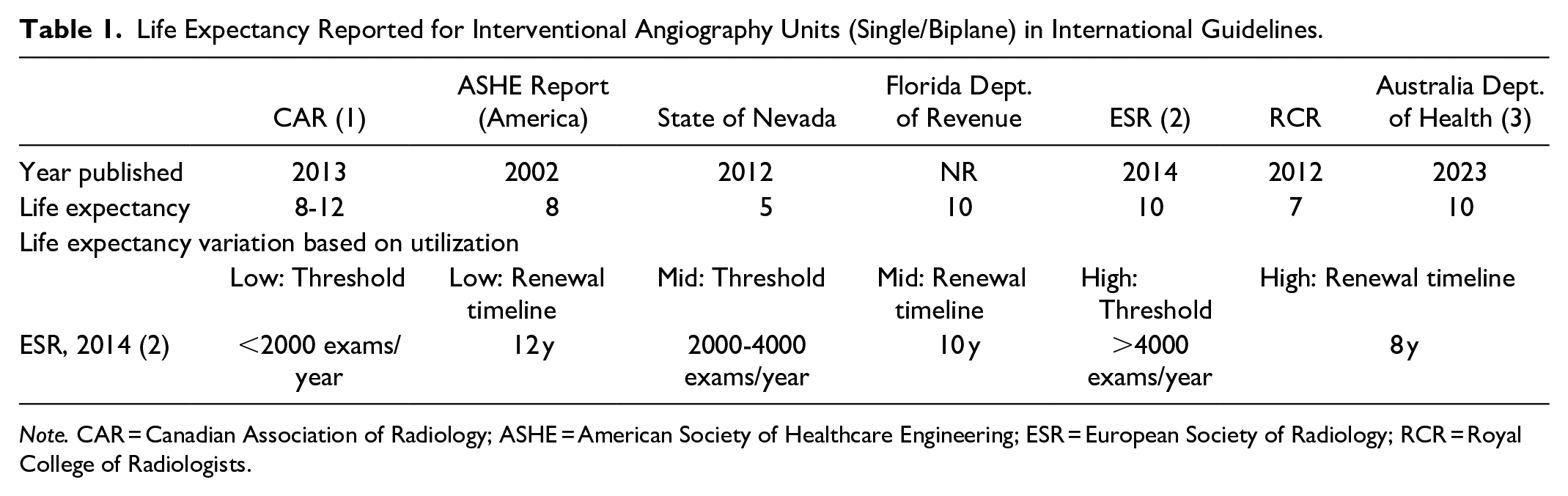
Note. CAR = Canadian Association of Radiology; ASHE = American Society of Healthcare Engineering; ESR = European Society of Radiology; RCR = Royal College of Radiologists.
Replacement Criteria
There is limited data to identify when replacement of a unit becomes essential. Equipment life cycles can vary widely depending on maintenance and utilization. 3 Changes in medical practice may drive a need for newer types of imaging. The Canadian Association of Radiology produced a life cycle guidance document in 2013 that outlined the most important factors in medical imaging equipment replacement. The survey of Division Heads and Department leads outlined several factors deemed “most important” in deciding when to replace equipment: risk assessment, replacement criteria (safety, efficiency, etc.), and financing (summarized in Table 2). 10 Financing was reported as the single biggest factor affecting decision-making. Life cycle guidelines are considered helpful guidance in the decision process, but equipment replacement is too complex to be defined by age alone.
Factors Contributing to Medical Imaging Equipment Replacement, as Rated by a Survey of Division Heads and Department Leads Across Canada.

Source. Adapted from Canadian Association of Radiology. 10
Aging Equipment
Despite the OEM and society guidelines, there remain a large proportion of aging equipment in radiology departments worldwide. An industry age profile report on European countries identified 34% of the interventional units were greater than 10 years old. 13 Renewal of imaging equipment requires significant capital investment and several years of planning. Constrained healthcare budgets can hinder its timely replacement. Frontline staff, who use this equipment daily, must advocate for its replacement to ensure that adequate resources are allocated by the department administration. However, several factors may prevent this. A lack of a speak-out culture, feelings of futility, and previous unresponsiveness to staff concerns can lead to frustration and discourage advocacy for necessary equipment updates.
In our scenario, the interventional radiology department has 4 dedicated interventional single plane angiography units. Each was installed consecutively 12 years prior to failure. The units were regularly maintained by in-house medical engineering teams with Original Equipment Manufacturer (OEM) consultation on a case-by-case basis. Replacement planning had begun 4 years previously based on the equipment age and technical obsolescence (the units did not have cone-beam CT capability). The equipment began to fail while waiting to initiate the construction process for new equipment. A review of incidents related to angiography unit malfunction reported in the hospital safety and quality system showed a low level of reports with a notable increase in the year prior to unit failure (Figure 2), recognizing the potential for under-reporting as can be experienced with any incident system. This example illustrates the pattern seen as angiography units reach their end of life: wear out failures and downtimes increase. Delaying equipment replacement and/or purchase until it reaches end of life creates a dilemma; by the time the equipment reaches failure, the increased risk is compounded by the inevitable delay required for installation of new equipment, further heightening patient safety risks.
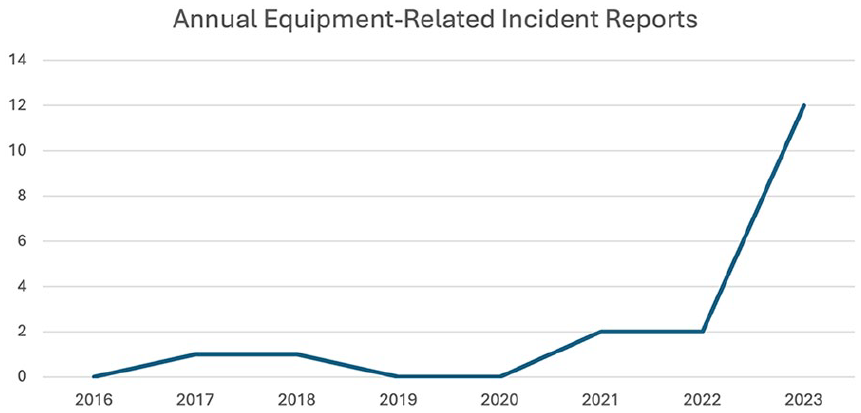
Trend in incident reports of angiography unit malfunction in the years leading up to angiography unit failure in 2023.
In summary, determining when imaging equipment should be replaced is influenced by many factors, and relying solely on numerical thresholds such as equipment age or case volume is insufficient to guide this decision. Determining when and how to replace equipment is a complex interplay of risk assessments for patients and staff, utilization patterns, maintenance history and economic considerations. Effective decision-making requires ongoing, close communication between physicians, medical engineering, industry, and medical leadership to ensure a comprehensive and safety-focused approach.
Summary: Anticipation
• Aging equipment in healthcare can pose risks to patients and staff, lead to technical obsolescence, lower staff morale, reduce department revenue, and delay patient care.
• Maximum life expectancy and clinical relevance for any piece of medical imaging equipment cannot be determined by age and case volume alone.
• The loss of OEM support can determine the practical life expectancy of equipment. Reliance on this alone may lead to further risks by waiting too long such that time taken to replace equipment can add to further delays.
Coping
The coping phase is similar to crisis management and can be split into 2 sub-categories: the organization’s ability to accept there is a problem and the ability to develop and implement solutions. 2
Accepting the Problem
Solutions cannot be implemented without first accepting that there is a problem. Organizations may delay responding to critical events due to initial denial. 2 Repairs are made without considering the deeper implication of a malfunction. Multidisciplinary decision making between physicians, engineers, and administrators is required. A key challenge in addressing equipment malfunction is the lack of a common vocabulary that bridges the gap between clinicians, administrative leadership, and engineering teams. The exact interpretation of what constitutes “safe” for clinical use could vary between what is clinically acceptable versus a technical standard. For example, consider the following scenario: an angiography unit has an intermittent but rare technical failure, the cause has not been diagnosed but will be monitored and the unit has been declared suitable for use. On the one hand, shutting a unit(s) down can have a significant negative impact in the face of a rising clinical workload and financial constraints. This is compounded in practice settings where alternative sites of care are not available. On the other hand, patient safety is the priority. Healthcare staff have a duty to mitigate risks to the patient and not perform a procedure with equipment that is known to be unsafe. In Canada, this is a legal requirement where the physician would be liable for damages that are sustained from the use of equipment that they know to be unsafe. Similarly, the owner of the equipment (hospital, physician group, etc.) has a duty to maintain the equipment (Canadian Medical Protection Association [CMPA] representative, personal communication, September 1, 2023). Denial can lead to ethical and legal implications, particularly if there is disagreement between stakeholders on the safety of equipment. Such a scenario can create an environment where accountability is blurred leaving clinicians uncertain how to proceed. Specific case examples of involving aging medical imaging equipment are rare, possibly due to under-reporting. A 2022 review of medico-legal cases involving radiologists closed by the CMPA did not identify equipment as a factor contributing to medico-legal risk, possibly as “until recently the CMPA data coding focused on provider factors and captured a limited number of system factors.” 14 This illustrates the limited data available in guiding physicians on the risks of aging equipment. The physician must be the ultimate decision-maker who, considering all factors and with patient safety as the primary focus, decides whether an angiography unit is safe for use.
Implementing Solutions
Maintaining timely and effective clinical care is a challenge through the months required to replace several angiography units. The broad strategy involves leveraging intra- and extra-departmental resources to maximize available equipment, followed by triage of patients to the appropriate unit.
Resource Assessment and Triage
The initial assessment identifies what facilities are fully functional, available in a limited fashion, or suspended entirely. 15 Crisis management is a fluid scenario that will require frequent re-assessment. There are many backup strategies for a suspended angiography unit(s): substitution with mobile C-arms, changing image guidance modality, and changing treatment strategies. Mobile C-arms are typically available within a radiology department through reallocation from other divisions or can be obtained quickly from OEMs. They can provide adequate backup for higher quality angiography procedures, particularly C-arms with digital subtraction angiography (DSA) capability and larger detectors (minimum 30 × 30 cm). 16 However, there may be concerns around their use where high quality angiography is needed, for example, embolization procedures and peripheral vascular disease. Alternative image guidance modalities can be used for many interventional radiology procedures. Ultrasound can provide adequate guidance for many superficial drainage tube placements for example, chest tube for pleural effusion. CT guidance can identify wire and drain position for deep abscess drainages. Use of a limited angiography unit may be enough for wire guidance and drain placement in many circumstances.
Other endovascular subspecialties can provide key resources, for example, vascular surgery, neuro-interventional radiology, and interventional cardiology. A collaborative relationship can allow sharing of their angiography units or transfer of time-sensitive procedures to available endovascular partners. Naturally, it’s unlikely that their angiography units are sitting idle. This may limit angiography unit availability to unsociable hours. Additionally, the difficulties of working outside of a “home” environment must be stressed. Smooth and efficient procedures require more than familiarity with the angiography unit itself. Basic steps of procedure preparation become more difficult in an unfamiliar environment, for example, entering the patient information in the angiography unit, locating leads and vital monitors, locating basic dressing supplies. Each healthcare team member will be affected by these challenges. The challenge is exacerbated during high-stress scenarios, for example, emergent procedures or cardiopulmonary arrest. Mitigating these stresses requires engagement from each team member for workflow improvements.
In our scenario, the IR department contained 4 angiography units and a fifth “small procedure” room with a C-arm for tube exchanges and venous access. Equipment failure reduced the department to one limited angiography unit, 2 rooms with mobile C-arms, and one room without fluoroscopic capabilities. Ultrasound-guided or non-image guided procedures were maximized and performed in the fourth room for example, port removals, tunnelled pleural and peritoneal catheters, chest tubes, and superficial abscess drainages. Mobile C-arm rooms were used for basic angiographic procedures for example, drainage tube exchanges, nephrostomy placement. The room with a limited angiography unit was used for basic angiographic procedures, for example, fistulography. Any procedures requiring “high quality” angiography were moved to an alternative functional angiography unit based on request urgency and time of day. Emergent inpatient procedures requiring high quality angiography were performed in a hybrid OR suite during working hours and an interventional cardiology unit during “on call” hours (Figure 3). Time-sensitive outpatient requests were scheduled for evening procedures in an interventional cardiology unit or at an alternative interventional radiology department within the hospital network. The interventional cardiology unit was new to IR staff. Staff responded by creating quick reference guides for the fixed detector angiography units (patient positioning, image transfer, etc.), carts with common catheters and wires, and a nursing cart with commonly used and emergency equipment. Team members were oriented to the new angiography unit with a focus on the location of emergency equipment.
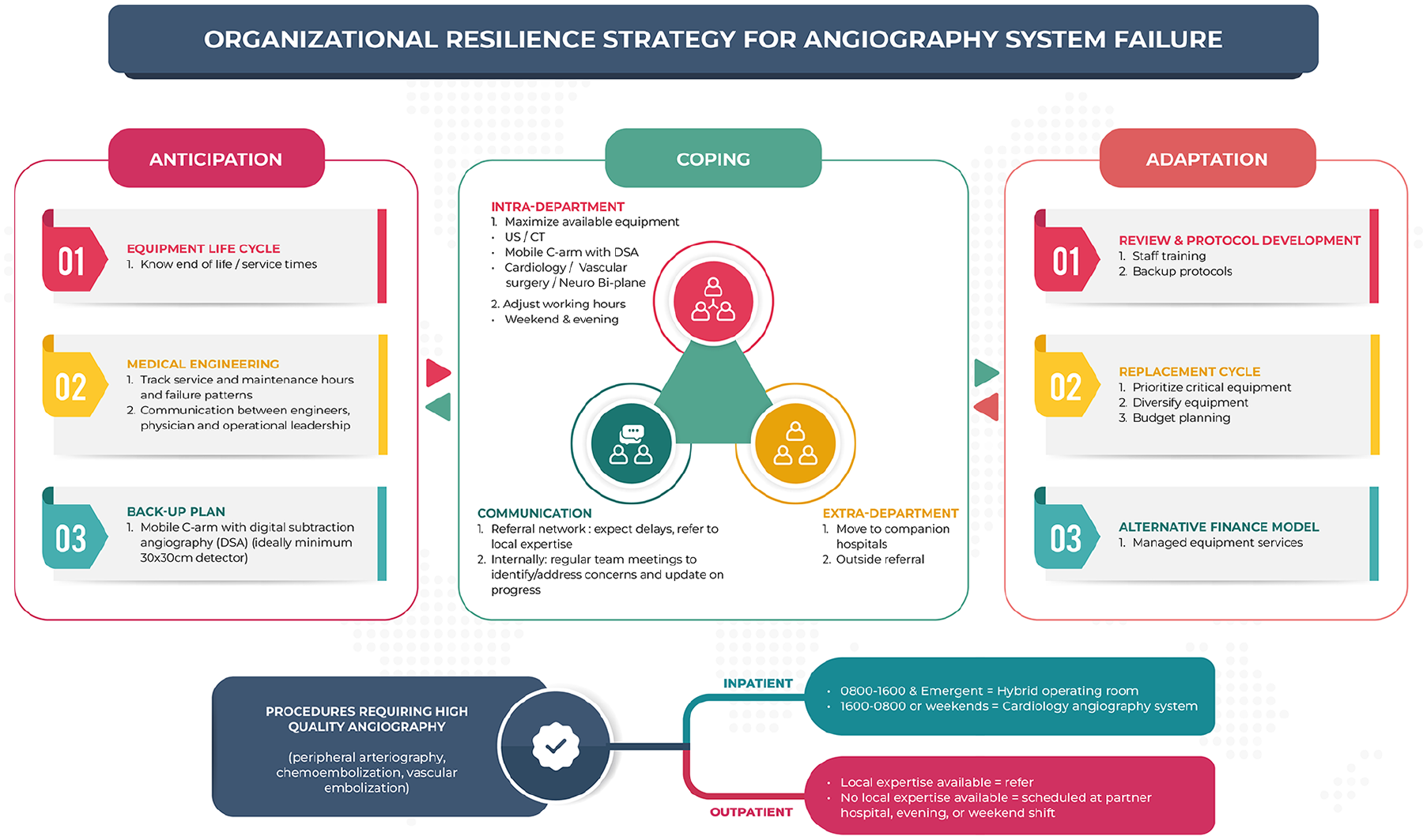
Overview of organizational resilience strategy for angiography system failure.
Staffing Schedules
Adjusting staff schedules can maximize resource use. Working unsociable hours can maximize the use of functional angiography units. This decision should not be taken lightly as new schedules further increase the strain on staff. Our response included scheduling evening and Saturday shifts to ensure time-sensitive inpatient and outpatient procedures were completed.
Patient Triage
Inpatient and outpatient referrals were triaged by staff physicians to appropriate timing and to appropriate equipment. Non-urgent referrals were delayed prioritizing time-sensitive care, for example, oncology and limb salvage. Triage required choosing the appropriate setting for procedures as staff physicians agreed on informal guidelines for which procedures were suitable to the piece of equipment available. An important consideration was whether another treatment option is more suitable, given the limited angiography equipment available. This can include somewhat equivalent treatment options, for example, urology referral for double-J stent placement instead of a nephrostomy; or may require more in-depth discussions for treatment pathways. This is a nuanced discussion that is often required on a case-by-case basis.
Communication
Communication with patient referral networks is critical. Angiography unit failure can significantly impact patient throughput and the timely delivery of patient care. Clear and continuous communication to referral bases, in and out of hospital, to inform them of potential delays is required. Multiple mediums of communication are most effective in spreading a message across an organization.17,18 Electronic communication via e-mail and other media can be simplest but should be augmented by communication through formal organizational leadership structures, and informally to individual stakeholders during department meetings or clinical multi-disciplinary team meetings. As was clear during the COVID-19 pandemic, an effective and agile crisis leadership team structure is essential for quick decision making. New workflows are often implemented quickly and with incomplete information. Rapid review after a period of implementation, with feedback from key stakeholders, allows for adjustments as needed. 18
Our case example of angiography equipment failure occurred quickly over 1 week. Once the scale of equipment malfunction was clear, this was communicated via multiple mediums throughout referral networks. An organization wide summary email was sent in SBAR (Situation, Background, Assessment, Recommendation) format (Figure 4). Key clinical stakeholders were informed during the various inter-departmental meetings. The situation was escalated to top levels of the hospital leadership, including the CEO such that communication around leveraging support from nearby hospitals could also be initiated.
Summary: Coping
• Coping is an organization’s ability to accept there is a problem and the ability to develop and implement a solution.
• Intra-department responses include use of mobile C-arms, alternative imaging modalities, and adjusting staff scheduling to maximize available resources.
• Extra-department resources can include alternative angiography units (vascular surgery, interventional cardiology) or referral networks.
• Frequent reassessment and multi-medium communication through leadership and referral networks is key throughout the crisis.
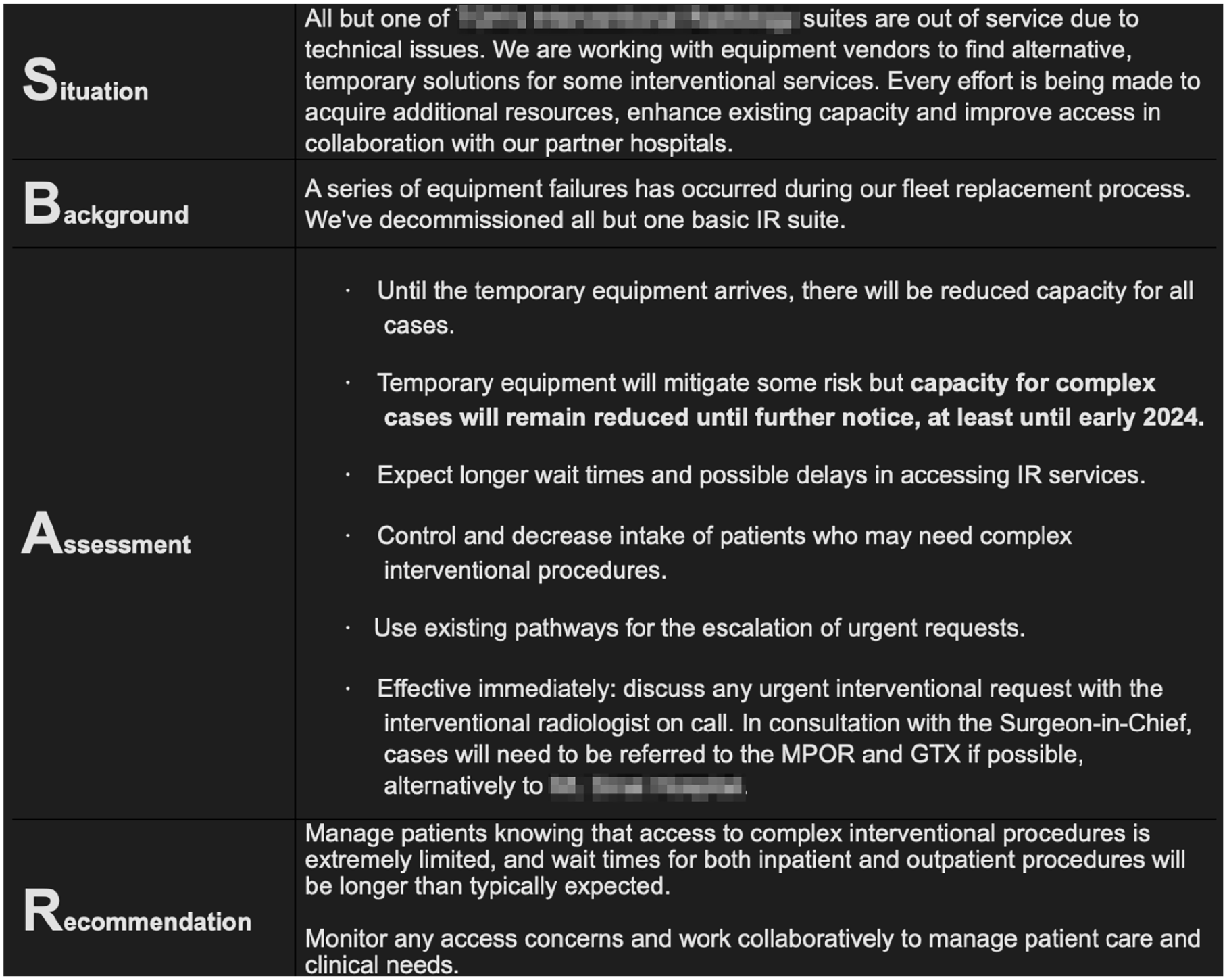
Organization-wide email informing referral networks to expect patient care delays due to angiography unit decommissioning.
Adaptation
Adaptation is defined as adjustment following a crisis that is directed toward organizational advancement. 2 The crisis does not have to occur in your own unit or organization. These principles are broadly applicable to other medical imaging divisions and relevant to any system reliant on technology for healthcare delivery.
Contingency Planning
Adaptation requires contingency plans for scenarios of equipment failure. This includes reviewing and protocolizing the workflows, communication patterns, and staff training that were required during the crisis. New service lines should include contingency planning to prepare for facility or equipment failure. An example of this is outlined by the Medicines and Healthcare products Regulatory Agency (MHRA) describing requirements for new endovascular aortic repair (EVAR) sites in the UK. 16 They outline several requirements and state that “the procedure should not go ahead if there is no backup plan.” Essential features that should be considered include the power supply to the department and the angiography unit, a backup of another static unit or a mobile C-arm (with DSA availability), and alternative injector pumps. 16
Renewal Planning
Replacing an angiography unit is a multiyear process and significant financial commitment that can be difficult to fund, particularly in nationalized healthcare systems. The European Society of Radiology advocates for a medical imaging equipment upgrade or replacement plan that should “look forward a minimum of 5 years, with annual updating.” 3 This will ensure replacement occurs in a non-emergent fashion and avoid equipment exceeding the 10 year mark where imaging equipment is no longer state-of-the-art and replacement is essential. 3 When requirements exceed available funding, prioritization is required. To facilitate objective evaluations, scoring criteria for equipment replacement are recommended to prioritize critical and/or end-of-life equipment.8,19-21 Factors common to several scoring systems include patient risk, equipment age and utilization, support status, obsolescence, and mission-criticality (Table 3). Faculty-specific criteria and weighting is encouraged to prioritize factors that are more applicable to each organization.
Common Factors in Scoring Criteria to Prioritize Equipment Replacement.
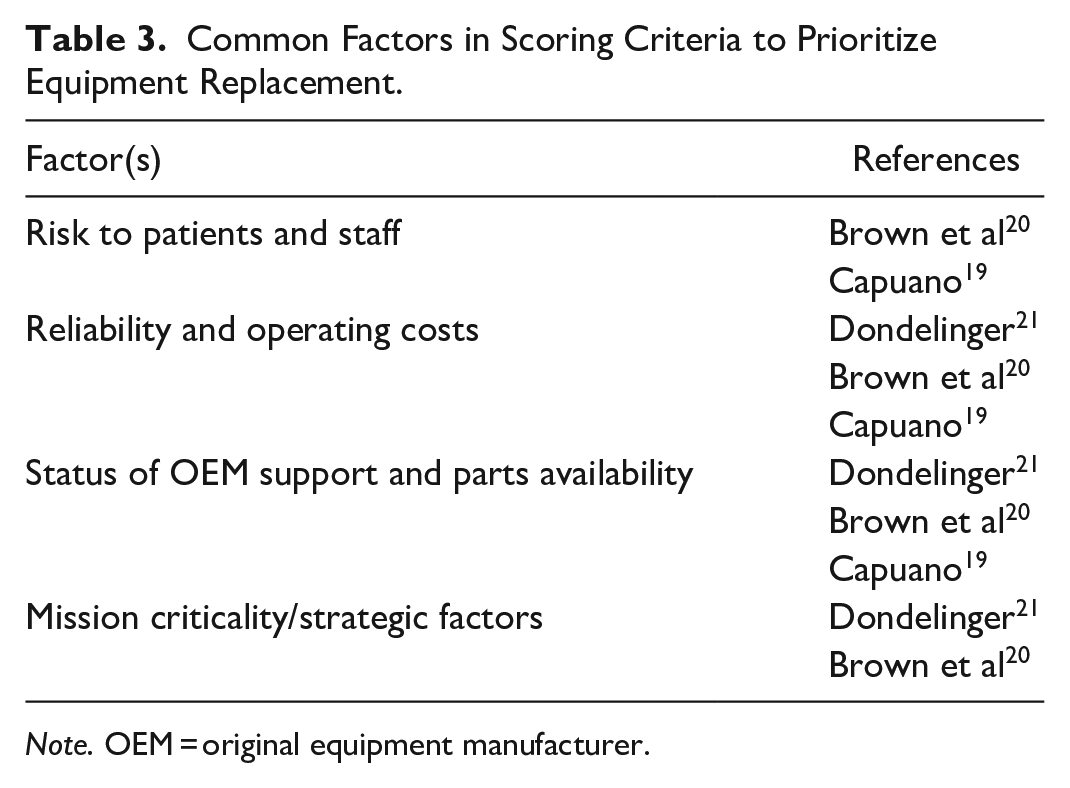
Note. OEM = original equipment manufacturer.
Financial Planning
Financing is the most influential factor in the replacement of imaging equipment. 10 Large capital expenditures require years of planning and may prove difficult in constrained budgets. Alternatives include maintenance and/or upgrade of equipment to solve performance issues and keep the equipment in service. Appropriate maintenance can shorten the life of medical equipment by up to 50%.3,22 Government influence can act as a valuable incentive for facilities to maintain upgraded and quality medical imaging equipment. Australian legislation incentivizes equipment maintenance and renewal by requiring accreditation of imaging equipment to qualify for Medicare reimbursement. Equipment that is not maintained or renewed appropriately will lose eligibility for Medicare payments. For example, diagnostic imaging services rendered using equipment that has exceeded its effective life age, or maximum extended life age for appropriately upgraded equipment, are reimbursed at 50% of the Medicare rate.12,23 In addition to penalizing aging equipment, providing additional government funding would incentivize maintenance and renewal. However, the incentives should not lie entirely on the funding side. OEMs should also be accountable to provide affordable solutions, with flexible options that cater to the varying financial capabilities of different organizations. Alternative payment models are available to help with large capital expenditures. Managed equipment service contracts offer a fixed payment plan that can include the purchase, maintenance, and long term replacement schedule. 24 The fixed payment model offers predictability in the operational budget while the OEM maintains and upgrades equipment to ensure high uptime and reliability. Similarly, public-private partnerships involve collaboration with OEMs to fund and manage medical equipment. This offers new equipment, operational support, and stable financial payments for a healthcare organization, while the OEM has a stable revenue stream, opportunity for collaboration and feedback, and risk sharing of implementing new technologies. Disadvantages include a long-term contract with a single OEM and difficulties aligning public health and private sector objectives.
Summary: Adaptation
• Adaptation should follow a crisis, either experienced in your own organization or as a lesson from another.
• Departments should include contingency plans for equipment failure in any service line.
• Medical imaging replacement should look forward a minimum of 5 years, with annual updating.
• Scoring systems for imaging equipment replacement prioritize patient risk, equipment age and utilization, support status, obsolescence, and mission-criticality; faculty-specific systems are recommended.
Conclusion
Adaptation should not require a crisis. The concept of organizational resilience and the principles learned in this review of angiography equipment failure are broadly applicable. Our hope is that Learning from incidents in similar organizations should review your own department’s preparedness. Our hope is that this article will prompt readers to review their department’s preparedness for this unfortunate but real scenario.
Footnotes
Acknowledgements
Katie Dionne, MRT(R); Carolyn Wight, MHI MRT(R) for their assistance in concept development and data collection.
Author Contributions
All authors significantly contributed to conception and writing of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
