Abstract
The use of magnetic resonance imaging (MRI) during pregnancy is associated with concerns among patients and health professionals with regards to fetal safety. In this work, the Canadian Association of Radiologists (CAR) Working Group on MRI in Pregnancy presents recommendations for the use of MRI in pregnancy, derived from literature review as well as expert panel opinions and discussions. The working group, which consists of academic subspecialty radiologists and obstetrician-gynaecologists, aimed to provide updated, evidence-based recommendations addressing safety domains related to energy deposition, acoustic noise, and gadolinium-based contrast agent use based on magnetic field strength (1.5T and 3T) and trimester scanned, in addition to the effects of sedative use and occupational exposure.
Introduction
The use of magnetic resonance imaging (MRI) in clinical practice is increasing. In Canada, the use of MRI nearly doubled from 1 to 1.86 million from 2007-2017. 1 Because MRI can provide high quality depiction of cross-sectional anatomy and pathology without the need for ionizing radiation, it is unsurprising that its use during pregnancy has also been on the rise. 2
Several theoretical concerns exist that may cause unnecessary anxiety for patients, patient families and the health care team, potentially limiting widespread use of MRI during pregnancy. These concerns are further confounded by a lack of sufficiently powered, prospective, human studies with long-term follow-up. Many topics extrapolate data from a combination of small case studies and animal studies, as designing large-scale randomized control trials to assess the potential long-term adverse effects of MRI on fetal development is not practical. To date, the highest quality evidence relates to large public health care database studies which have helped alleviate some concerns regarding the safety of MRI and the pregnant patient.2,3
Technological advancements, primarily the increasing transition towards use of higher field strengths, stronger magnetic gradients and improved software applications directly translates into better quality and faster MRI examinations which may be used for identification and characterization of maternal and fetal pathologies. With increased utilization and availability of 3 Tesla (T) compared to 1.5 T MR systems, there are theoretical and practical improvements in signal-to-noise ratio which can increase temporal and/or spatial resolution, resulting in overall improved image quality, length of exams and diagnostic capability. 4 This is beneficial for most maternal MR imaging requirements, and particularly in the burgeoning area of fetal MRI. 4 However, the potential risks involved with fetal MRI are not well studied at 3 T relative to 1.5 T.
Given the recent changes in the field of MRI in pregnant patients and of the fetus, several societies worldwide have updated their recommendations and practice guidelines regarding the use of MRI during pregnancy. These include: the American College of Obstetricians and Gynecologists (ACOG; updated in 2017); the International Society of Ultrasound in Obstetrics and Gynecology (ISUOG; updated in 2019); and the American College of Radiology (ACR; updated in 2020). Conversely, the last practice guideline update from the Canadian Association of Radiologists (CAR) is from 2011 and does not cover many of the recent studies and technological advancements that have been made in the field since.
In this work, the CAR working group on MRI in Pregnancy provide evidence-based recommendations that are derived from literature review as well as expert panel opinions and discussions. Safety domains related to energy deposition, acoustic noise, and contrast agent use will be addressed, based on magnet strength (1.5 T and 3 T) and trimester scanned. The effects of sedative use and occupational exposure will also be explored. The aim of this work is to provide an up-to-date comprehensive source of information for patients, MRI practitioners and other health care personnel to understand the different concerns relating to safety of MRI in pregnancy, and provide contemporary, evidence-based recommendations and guidance.
Energy Deposition
A safety concern with MRI in pregnancy is that energy will be deposited within the maternal and fetal tissues due to exposure to radiofrequency (RF) pulses. Fetal heating greater than 2°C can be teratogenic. 5 In practice, fetal temperature cannot be directly measured; instead, the maternal specific absorption rate (SAR) is used to estimate the absorbed RF power per unit body weight and is measured in W/kg. Although SAR can be calculated for specific regions, the most commonly reported value is whole-body average SAR. A maternal rise in SAR logically coincides with a risk for embryonic or fetal temperature increases, although models have shown that the fetus receives only approximately 40-70% of the maternal SAR. 6
Commercially available MR scanners have proprietary programs based on National Electrical Manufacturers Association standards that estimate SAR values automatically and warn the operator if regulatory limits are likely to be exceeded. 7 MR scanners have 3 operating modes with different whole-body average SAR limits as set out by the International Electrotechnical Commission: normal (2 W/kg), controlled (4 W/kg) and restricted (> 4 W/kg). Operation in normal mode limits rise of body temperature to 0.5°C. 8
SAR calculation depends on numerous factors, including but not limited to, the electrical conductivity of the tissue, patient weight and shape, field strength, flip angle and RF pulse duration. SAR is thus pulse sequence dependent. Sequences with long trains of RF pulses such as steady-state free precession (SSFP) and single-shot fast-spin echo (SSFSE) have higher SAR values, 9 yet constitute the bulk of fetal MRI as they are short and distributed throughout the exam, thus mitigating temperature increases from SAR. 10
There is considerable overlap of evidence regarding the safety of MRI in pregnancy in relation to a specific MRI safety domain. For example, a study which evaluates the safety of MRI relating to SAR will also potentially overlap with other domains such as contrast use or acoustic noise. In this guideline, evidence is presented sequentially but will be cited frequently in overlapping sections.
SAR at 1.5 T
First trimester
An observational study of 15 neonates inadvertently imaged in the first trimester did not demonstrate any attributable adverse outcomes. 11 In a population-based cohort study involving more than 1.4 million pregnancies, first-trimester MRI with or without contrast was not significantly associated with stillbirth or neonatal death, congenital anomaly, neoplasm, vision or hearing loss. However, in the cohort with MRI exposure between 5-10 weeks gestation (calculated from the first day of the last menstrual period), the risk of congenital anomalies and hearing loss was unchanged but the risk of vision loss was higher in a small group (8/416 fetuses) compared to those that did not undergo MRI. 3 No other studies have shown a similar outcome.
Second and third trimesters
Prenatal exposure to 1.5 T MRI during the second or third trimester of pregnancy in a cohort of 72 healthy fetuses was not associated with disturbances in functional outcomes or hearing impairment at preschool age. 12 Exposure had no adverse effects on birth weight, 13 long-term neurodevelopmental outcomes, 14 growth, motor functioning, social or neurological development. 15 Direct measurement of amniotic fluid temperature in animal models at 1.5 T have not shown increases >0.5°C during scanning.16,17
SAR at 3.0 T
Transition from 1.5 T to 3 T results in a quadrupling of the SAR if all other factors are kept constant. This raises concerns about fetal safety, and energy deposition in the fetus resulting in an increase in body temperature. Direct measurement of temperature rise in fetal organs and amniotic fluid in pregnant miniature pigs using a typical standard of care MR protocol was found to be < 1°C if the imaging time was < 30 minutes. 18 However, the cumulative temperature increase over 1 hour of imaging time with high SAR pulse sequences reached up to 2.5°C. Thus, caution is warranted when imaging the pregnant patient at high field strength. 18
First trimester
There is less data on fetal outcomes after exposure to 3 T compared to 1.5 T field strength. A retrospective case control study of 81 exposed pregnancies (14/81 in first trimester) showed no adverse effects on fetal growth in any trimester, including exposure during the first trimester. 19
Second and third trimesters
In addition to the previously mentioned study, 19 in a study of 12 pregnant women who underwent fetal MRI at both 1.5 T and 3 T in the second or third trimester, deliberate modifications of pulse sequence parameters resulted in a significantly lower SAR at 3 T compared to 1.5 T while maintaining similar or increased tissue contrast and conspicuity. 20 Such modifications include decreasing the flip angle of the imaging sequence and increasing the repetition time.
In practice, however, sequence parameters and RF pulse design are automatically adjusted by the scanner to lower SAR. A recent retrospective study found that when comparing optimized fetal MRI in the second and third trimesters at 1.5 T and 3 T, the mean SAR was statistically equivalent. 9 In addition, 99.9% of the sequences had an SAR of 2 W/kg or less (ie, the upper limit for MRI under normal operating mode) although it was noted that some higher SAR sequences such as 3 dimensional (3D) SSFP may require modification at 3 T to keep the energy delivered to the patient as low as possible. In newer systems, RF shimming with multi-channel transmit coils will be able to optimize the power, phase and waveform shape of the individual RF sources to each patient’s anatomy, thus preventing hotspots and reducing overall SAR. 21
Recommendations
MRI of pregnant patients should be performed in normal operating mode (whole body averaged SAR limit of ≤2 W/kg), which limits body temperature rise to 0.5°C. To ensure accurate SAR calculations, it is recommended that the patient’s weight be taken immediately prior to MRI.
The preponderance of studies have shown no adverse outcomes attributable to MRI in any trimester at either 1.5 T or 3 T; however, out of an abundance of caution, 1.5 T is preferred in the first trimester if both field strengths are available.
Acoustic Noise
Acoustic noise from MRI occurs primarily as a result of Lorentz forces, which are generated with rapid current alterations in the gradient coils. 22 The Lorentz forces are proportional to the main magnetic field strength and the gradient current. With increasing use of higher field strengths (> 1.5 T) and more powerful gradient systems, noise levels can exceed 110 A-weighted decibel (dB(A)).23,24 Noise levels also depend on hardware design, location in relation to the scanner, and sequences used. Noise levels are particularly loud at the bore entrance. 23 In addition, noise levels are increased during certain sequence types, such as echo planar imaging, fast gradient echo sequences or 3D sequences such Magnetization Prepared Rapid Acquisition Gradient Echo (MP-RAGE). 25 Certain sequence protocol parameters such as smaller field of view, shorter repetition time (TR) and to a lesser extent, thinner slice thickness also increase noise levels. 23
The primary concerns revolve around the fact that loud noise levels generated by the MR scanner may potentially damage fetal hearing, particularly in the early stages when the related structures are being developed. Fetuses have been reported to respond to low frequency (100-500 Hz), high intensity (> 100 dB(A)) noises by 19 weeks of gestation, 26 though it is unclear whether this timeframe was calculated based on the last menstrual period or from conception date. By 24 weeks of gestation, the auditory organs are fully developed and fetuses have been reported to respond to higher frequency tones (1000 and 3000 Hz) by 35 weeks gestation at <100 dB(A) intensity. 26 Fetuses may therefore theoretically be at risk of hair cell damage or abnormal development of hearing structures when exposed to loud noises. Sheep are commonly used as a fetal animal model to extrapolate data as they share many commonalities with humans in terms of development (in particular, for inner ear function) and transmission physics. 27 Although limited in sample size, sheep studies have demonstrated that noises <1000 Hz in utero are attenuated as little as 2-3 dB; the degree of attenuation increases with higher noise frequencies (>1000 Hz), ranging from 20-40 dB of attenuation. 28 However, how much the noise is attenuated within the maternal tissue and amniotic fluid in humans remains uncertain.
Noise at 1.5 T
First trimester
Ray et al. have presented the largest retrospective database to date, with 1,737 fetuses scanned at 1.5 T during the first trimester. These were compared to approximately 1.4 million fetuses who did not undergo MRI, and children were followed until 4 years of age. 3 No differences in hearing were detected, in agreement with previous studies that performed otoacoustic testing soon after birth.13,19,29,30 In studies with long-term follow up, Streizek et al 13 reported 44 fetuses that were scanned during the first trimester had no hearing impairments when tested at birth or 3-month follow up.
Second and third trimester
Similar to Ray et al., a large retrospective database with 96 fetuses that were scanned during the second and third trimester found no effect on fetal hearing when compared to over 10,000 control neonates who were not exposed to fetal MRI. Similar findings were reported for 707 fetuses when tested at birth and 3-month follow up. 13 Smaller studies have also reported no long-term hearing effects related to MRI exposure.12,15
Noise at 3 T
First trimester
In a study with 14 fetuses scanned during the first trimester, and matched 1:2 with unexposed, normal babies, no significant differences in hearing impairment, when tested at birth or 1 month follow up, were reported. 19
Second and third trimesters
Jaimes et al. 30 investigated the impact of 1.5 T vs 3 T scans on neonatal hearing, with 62 neonates in each cohort. No significant differences in fail rates of transient otoacoustic emissions test (9.7% vs 6.5%, P = 0.74 for 1.5 T and 3 T, respectively) and in the auditory brainstem response test (3.2% vs 1.6%, P = 0.80 for 1.5 T and 3 T, respectively) were reported.
Recommendations
There is currently no evidence of long-term damage to fetal hearing organs from MRI performed at a field strength of 3 T or less during the second or third trimester. Although there is no evidence of damage to the fetal hearing organs during the first trimester either, the available literature is more limited; if clinically necessary, first trimester fetal MRI at a field strength of 1.5 T or below is generally deemed to be safe.
GBCA Use in Pregnancy
Gadolinium-based contrast agents (GBCAs) are vital for many MRI examinations, providing diagnostic information through enhancement of signal intensity by shortening the T1-relaxation of tissues with which they interact. It is estimated that MRI with GBCA comprise nearly half of all MRI studies performed. 31 GBCAs consist of a gadolinium molecule (which is responsible for the desired T1 shortening effects observed with MRI) bound to a chelate, which is used to mitigate the toxicity associated with unbound gadolinium in the human body. The composition of the chelated molecule determines the biochemical property of a particular GBCA, including its ability to move between body compartments, its ability to shorten T1 times and also, to some extent, its potential toxicity.
Use of GBCAs have been associated with several unintentional and harmful, or potentially harmful, effects in humans. The primary concerns in the context of fetal/maternal MRI are: 1) the potential for exposure of the fetus to gadolinium, due to its ability to cross the placental barrier into the fetal circulation and amniotic sac, where it can remain indefinitely;32-34 2) gadolinium retention. Although this is being actively investigated, the clinical implications of retained gadolinium remains unknown; and 3) the potential for nephrogenic systemic fibrosis (NSF), a systemic sclerosing condition similar to scleroderma which has no known treatment. 35 These concerns are theoretical, as no studies have reported any adverse effects when human fetuses were exposed in utero to date.
Exposure During the First Trimester
Teratogenic effects related to GBCA are thought to occur in the first trimester, during the phase of organogenesis. In humans this corresponds to the highest risk period for GBCA exposure, and also the timeframe when GBCA-enhanced MRI is most likely to inadvertently occur. 2 Fetal toxicity in humans has never been studied in a controlled fashion, and current guidelines extrapolate from observational studies, including case series and observational cohort studies. Most early case reports and cohort studies reported no adverse effects from GBCA to the fetus.36-38 One small case study with 13 fetuses had 1 fetus diagnosed with neurofibromatosis, but this was deemed unlikely to be related to GBCA exposure. 39 However, these reports were based off a small number of patients with limited long-term outcomes reported, and no control groups were used for comparison. The largest study assessing the effects of GBCA enhanced MRI in pregnancy is an epidemiological study using a health care database (Ontario, Canada). 3 Exposure to GBCA-enhanced MRI at any point during pregnancy was associated with increased risk of NSF-like conditions (HR 1.36; 95% CI, 1.09 to 1.69). Risk was highest when exposure was during the first trimester (adjusted HR 1.41; 95%, 1.11 -1.79). 3
Exposure During the Second and/or Third Trimesters
Animal studies have demonstrated GBCAs cross the placental barrier into the fetal circulation and amniotic sac32-34 in the later stages of pregnancy.40-42 However, clearance from the fetal circulation appears to be rapid. One recent study injected 5-times the recommended equivalent dosage of gadoterate meglumine into mice during the equivalent of the third trimester of pregnancy. The highest concentration (0.08% of injected dose) was found 30 minutes after maternal injection, with undetectable fetal concentrations after 48 hours. 43 Another study in macaques, who were injected with the recommended dosage of 0.1 mmol/kg gadoteridol during the third trimester, found the highest concentration (0.016% of injected dose) between 19-21 hours, declining to 0.0007% by 45 hours. Despite rapid clearance, daily repeated, high doses (0.5-2 mmol/kg) of GBCA had multiple adverse effects in rabbits, including intrauterine fetal demise. 44
Small case studies in humans have not reported any adverse events related to GBCA use during the second or third trimesters,45-48 and are in agreement with the findings of Ray et al. who showed no evidence for adverse outcomes when performing a subgroup analysis of women exposed to GBCA during the second or third trimester. 3
The effects of gadolinium retention have not been thoroughly studied. In the group that studied the effects of gadoteridol in macaques, injected both during the equivalent of the second and third trimesters, they found gadolinium retention was consistently seen in the liver and femur of the offspring upon tissue sampling at 7 months after birth. 49 In addition, single cases of gadolinium retention were seen in the juvenile spleen, skin and brain, respectively. 49 To our knowledge, there are no human studies assessing gadolinium retention in tissues related to in utero exposure to GBCA.
GBCA use and Breast Feeding
GBCAs demonstrate minimal binding to milk and plasma protein, limiting their excretion in breast milk. 50 In a study of 20 lactating women, very small amounts (<0.04%, equivalent to 0.57 ± 0.71 µmol) of intravenously administered GBCA was excreted into breast milk. 51 It is estimated that less than 0.1% of this is absorbed by the infant through the gastrointestinal tract. 51 Hence, the estimated exposure to the breastfeeding infant is significantly lower than the recommended weight-based pediatric dose of 0.1 mmol/kg in children under 6 months. 52
Recommendations
Based on the available literature, we do not recommend GBCA administration unless absolutely necessary. Informed patient and health care team consent describing animal and human population data indicating deleterious effects to the fetus when GBCA is administered in utero is recommended.
Postnatally, following GBCA administration to the mother, it is safe to continue breastfeeding. However, if the mother chooses to do so she can discard the milk pumped within 24 hours following GBCA injection. Discarding milk beyond 24 hours is of no value.
Medication use to reduce fetal motion
Fetal movements begin between 7-8 weeks of gestation (defined as the first day of the last menstrual period), with the emergence of strong and sudden movements by 13-16 weeks onwards. 53 Numerous methods have been explored to reduce fetal motion over the duration of the MRI and improve image quality. 54 In a recent survey by the European Society of Pediatric Radiology, it was found that 24% of institutions studied still use maternal sedation (either antihistamines or benzodiazepines). 55 However, technological advancements that significantly reduced scan times, including the use of parallel-imaging techniques, prospective and retrospective motion compensation techniques and faster pulse-sequences, have largely eliminated the need for sedatives, 56 though there remains some debate on their use for fetal MRI.
One study reported 19/131 fetal MRIs where diazepam was administered and found no difference in fetal motion when compared to the control group. 57 Another study reported having difficulties in performing T1-weighted imaging, however sedation was not required to acquire T2-weighted images. 58
As an alternative to sedative use, some centres report utilizing maternal fasting or avoidance of caffeine intake in the 4 hours immediately prior to MRI in a survey study. 59 This is supported by one ultrasound study that found active fetal movements during the first and third hours after food intake. 60 However, in a prospective survey study of 228 mothers, Yen et al. recently reported no relationship between intake of high glucose or caffeinated beverages with fetal motion on MRI. 61
Ultimately, based on the limited data available, the use of sedatives and/or fasting does not appear to be necessary in most routine cases.
Recommendation
The use of medication to reduce fetal motion is not recommended.
Occupational Exposure
The concerns regarding energy deposition and acoustic noise, addressed for pregnant patients, are also applicable to pregnant MRI personnel, which includes technologists, nurses, physicians, physicists, porters, receptionists, and any other medical personnel working in or near the magnetic field of the MRI system. However, there are a few notable differences when considering exposure of pregnant MRI personnel compared to pregnant patients. These differences include: The level of exposure of the MRI personnel compared to the patient with regards to the electromagnetic fields. MRI personnel may primarily be found in ACR Zones I-III, with limited time in Zone IV (the MRI scanner room).
62
Outside of Zone IV, exposure to radiofrequency and gradient magnetic fields decline rapidly.
63
However, MRI personnel remain exposed to the static magnetic field.
64
Additionally, exposure to RF and gradient magnetic fields may still occur if MRI personnel are needed inside Zone IV during scanning. Length of exposure to the electromagnetic fields over the course of pregnancy. The exposure of the pregnant MRI personnel carries a potential risk to the fetus with no benefit to the fetus or mother (as opposed to patients for which the benefits are deemed to outweigh the risks).
Electromagnetic Field Exposures
During scanning, MRI personnel may be exposed to 3 different electromagnetic fields: time-varying magnetic fields, static magnetic fields, and pulsed RF fields. Current information does not indicate any serious health effects resulting from the acute exposure of stationary humans to static magnetic fields up to 8 T. 65 Results on humans in fields up to 8 T and on animals up to 16 T indicate that there is a substantial margin of safety remaining above the highest fields now in clinical use, which are in the range of 3-4 T. 66
A large-scale study comparing pregnant MRI workers across multiple clinical and research MRI sites in the United States with other workers and home makers did not demonstrate a correlation between working in the MR environment and offspring gender or changes in the prevalence of premature delivery, infertility, low birth weight, or spontaneous abortion. 67 There is no information yet available regarding the strength of the magnetic field versus worker exposure during a specific pregnancy and its outcome. Likewise, no reliable data are present with regards to the potential effects (if any) on different stages of pregnancy in MR personnel. Based on this data, several societies have recommended that it is safe for MR personnel to work in and around the MRI environment over the course of pregnancy, though they should avoid remaining in Zone IV during data acquisition.68-70
Acoustic Noise
The data regarding the long-term exposure of pregnant MR personnel to loud acoustic noise is not clear. Early animal studies suggest that there is potential for damage with exposure to high acoustic noise during pregnancy.71-73 In humans, one study reported no significant differences in the incidence of neonatal hearing impairment when comparing 65 women exposed to 80-85 dB(A) per day, 5 times a week for 8 hours against 2,588 controls, 74 in direct contradiction to an older paper by Lalande et al. 75 Recently, in a large cohort study that included over 1.4 million single births from 1986-2008, a slightly higher risk of neonatal hearing dysfunction was found in women were exposed to noise ≤ 84 dB(A) for 8 hours on average (adjusted hazard ratio (aHR), 1.05), and greater risk when exposure was ≥ 85 dB(A) (aHR, 1.27). Longer duration of exposure (full time vs part time vs non-working) and less leave of absence (< 20 days) were also associated with higher risk of hearing dysfunction. 76
Recommendations
Pregnant MR personnel are safe to work around and within the 1.5 T and 3 T MR imaging environment. As a precaution, it is recommended that they do not remain in the MR imaging bore or magnet room during data acquisition, though they may enter the room if necessary (i.e., to position patients; place RF coils; or in response to an emergency). 68
Discussion
Based on our literature review and through expert panel discussions, recommendations regarding energy deposition, acoustic noise, GBCA administration, medications to reduce fetal motion, and occupational exposure to 1.5 T and 3 T MRI systems have been derived. These are in line with those presented by other national and international societies (Table 1).
Comparison of the recommended guidelines from various international societies and bodies.
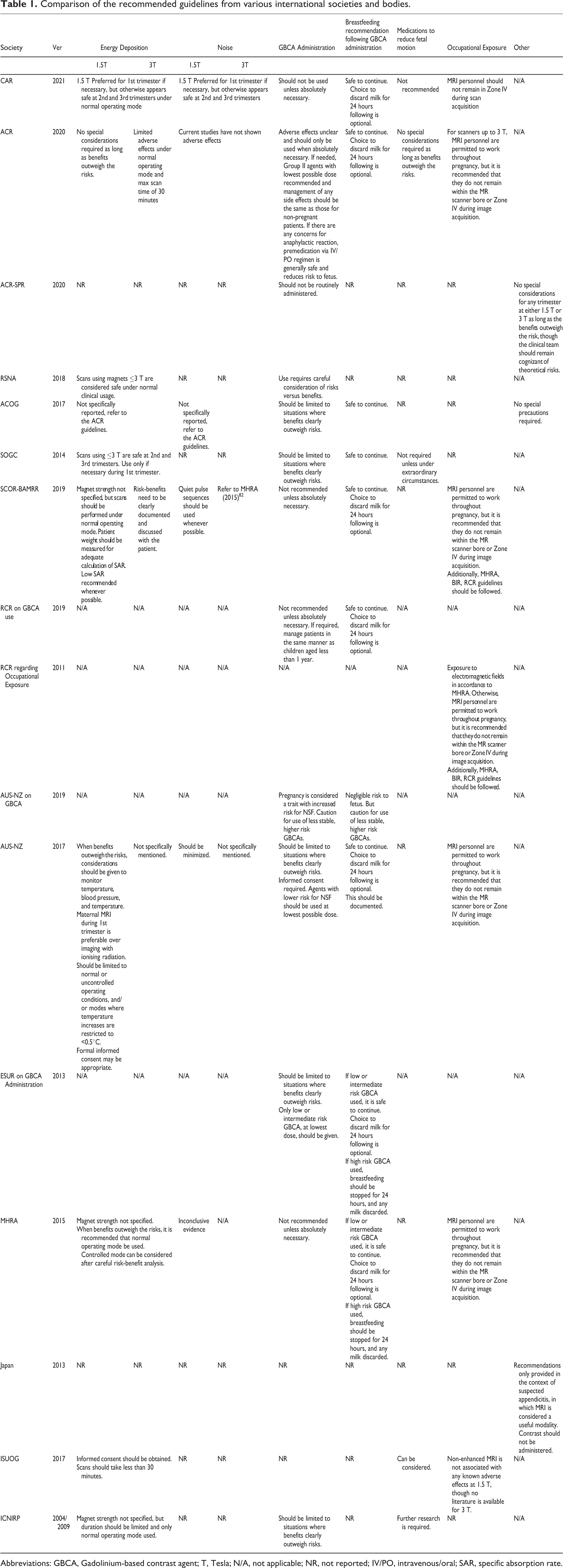
Abbreviations: GBCA, Gadolinium-based contrast agent; T, Tesla; N/A, not applicable; NR, not reported; IV/PO, intravenous/oral; SAR, specific absorption rate.
When deciding whether or not to proceed with MRI, thoughtful risk-benefit analysis is prudent. Any prenatal test, particularly one which may not be part of routine clinical care, may provoke a heightened sense of maternal, family and health care team anxiety. 77 It is recommended that a detailed explanation of the purpose, course, and risk-benefits be conveyed to the patient 78 to reduce anxiety and emphasize the medical value of MRI and how the information from MRI will directly influence a patient care decision. 79 The referring physician can do this through a leaflet provided to the patient in advance of the MRI, so that the patients have enough time to review and ask questions; 79 however, a detailed explanation regarding how MRI will be useful for a particular patient is advised. Suggested excerpts, based off the findings and recommendations presented here, have been appended in Table 2.
Potential Excerpts That Can be Added to Informed Consent and/or Leaflets Regarding the Various Issues Detailed in this Work.

Based on our findings, non-GBCA enhanced 1.5 T MRI is considered safe to perform at any trimester. The panel had conflicting opinions regarding the need, type and value of patient informed consent for MRI during pregnancy (Table 3). While some panel members argued that there was no point in obtaining consent because of no documented or known/proven risk to fetus based on the available literature, others argue that consent process may provide an opportunity for the physician or their delegate to have a face-to-face conversation for verbal or written consent with the patients, and answer their questions to reduce any anxiety. Institutions are urged to decide whether informed consent in this context is required in accordance with their local institutional policy. If MRI is considered necessary, only normal operating mode should be used at 1.5 T and at 3 T. MRI at 1.5 T is recommended over 3 T if both are available, due to greater available data supporting the safety of MRI at 1.5 T in the pregnant patient although 3 T MRI is also acceptable if 1.5 T is not practical or available. Regarding fetal MRI during the first trimester, if the scan can be delayed without altering patient care, this should be considered as fetal MRI is limited due to the small fetus size and increased fetal motion; additionally, fetal pathologies may not be detectable at this time. 80
Panel Decisions Regarding the Necessity of Informed Consent in Different Scenarios for MRI.
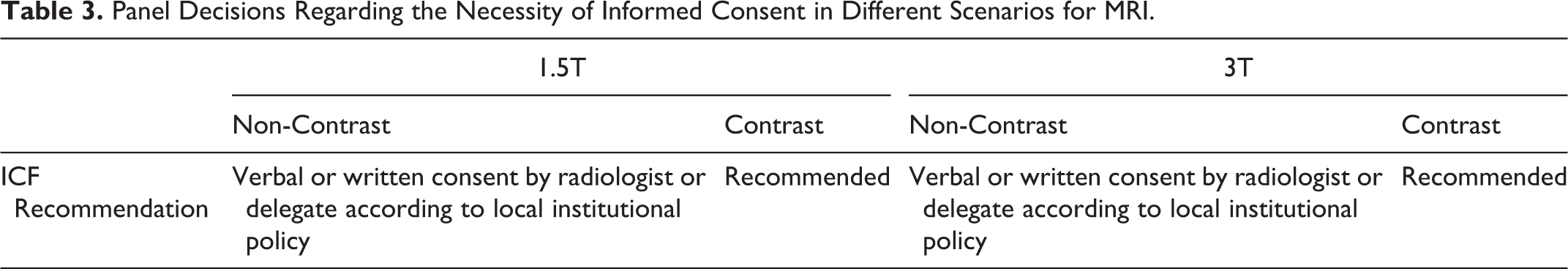
While no adverse events have been reported, due to the limited safety data available the panel unanimously agreed that informed consent should be obtained prior to use of non-GBCA-enhanced 3 T MRI during the first trimester. Considerations with regards to whether informed consent is needed when scanning at 3 T during the second or third trimesters are the same as those when scanning at 1.5 T during this timeframe.
GBCA use is considered contraindicated during pregnancy; any consideration of use requires careful scrutiny, discussion by the health care team and informed consent should be obtained and carefully documented. With regards to lactating non-pregnant patients, the panel has unanimously agreed that while it is safe to continue breastfeeding within the 24 hours after GBCA, because of very small amount of GBCA that may enter breast milk, the patients choose to discard the milk produced during this timeframe.
Footnotes
Acknowledgments
The authors thank Bahareh Moazen (MD) and Aimee Chan (MSc) for their assistance with this manuscript. The authors further thank the Diagnostic Imaging Committee of the Society of Obstetricians and Gynaecologists of Canada, who provided invaluable feedback during the review process.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Sharon Clarke has received research funding from GE Healthcare Canada. Dr. Grace Liu sits on an advisory board for Abbvie Inc, and has given a webinar for Hologic. The other authors have nothing to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
