Abstract
Introduction
The detection and quantification of coronary artery calcifications (CAC) has emerged as a crucial diagnostic tool in cardiovascular risk assessment due to its strong association with coronary artery disease (CAD) and adverse cardiovascular events. 1 The 2019 ACC/AHA guidelines recommend the use of CAC quantification alongside traditional risk factors in assessing cardiovascular risk. 2 Historically used for assessing obstructive coronary artery disease, CAC is now recognized as a marker of overall atherosclerotic burden, with quantitative plaque burden proving most predictive in primary prevention populations. Its utility extends further to refining preventive therapy initiation thresholds, improving shared decision-making, and enhancing patient adherence to treatment and lifestyle modifications. CAC testing is also deemed cost-effective in asymptomatic populations spanning various risk levels. 3
Efforts to detect coronary calcium using digital subtraction methods and fluoroscopy date back to the mid-1980s. While dual-exposure dual-energy chest radiography in the posteroanterior (PA) projection is used clinically,4,5 challenges remain due to motion artefacts and partial obstruction of coronary arteries from the superimposition of bones and other denser structures.5,6 One of the key limitations of dual-exposure techniques is that 2 separate images are acquired at different energy levels, which introduces the possibility of misregistration due to patient motion between exposures. This can lead to blurring or artifacts, reducing diagnostic accuracy. Besides, it increases patient’s radiation exposure. 7
In contrast, single-exposure, dual-energy X-ray imaging (DEX) overcomes these limitations by capturing both energy levels in a single acquisition, eliminating the risk of misregistration while not inherently increasing the radiation dose. 8 Thus, single-exposure DEX can potentially improve the detection of coronary artery calcium (CAC) and holds promise to better visualize valve and other vascular calcifications, a task that remains difficult on conventional X-ray. However, despite these advantages, single-exposure DEX can introduce a different challenge of increased image noise and, therefore, a potential loss of low-contrast resolution due to the computational subtraction process.
This study aimed to evaluate whether single-exposure, dual-energy chest X-ray improves the visualization of coronary artery, valve and other vascular (aortic and supra-aortic branches) calcifications compared to conventional X-ray imaging.
Materials and Methods
Patient Selection
With Institutional Research Ethics Board approval, 61 consecutive bone marrow transplant patients presenting to the medical imaging department at a large tertiary care hospital for suspected pneumonia were enrolled in this prospective cohort study. The population of bone marrow transplant patients was chosen due to their predisposition to cardiac complications.
Imaging
All patients underwent single-exposure, dual-energy X-ray (DEX, Reveal 35C, KA Imaging Inc.) in posteroanterior and lateral projections, immediately followed by an unenhanced, low-dose, standard-of-care chest non-EKG-gated CT (SOC-CT) on a wide-area detector CT (Aquilion Vision, Canon Medical Systems, Otawara, Tochigi 324-0036, Japan) with the following parameters: 135 kV, automatic exposure control, 3 mm slice thickness, coronal and sagittal reformations and sliding maximum projections with 8 mm slice thickness. Two image pairs (posteroanterior and lateral) were calculated from available dual-energy X-ray data: a composite conventional radiograph (COMP; “regular chest X-ray”) and a bone image (BI) with soft-tissue subtraction. The imaging parameters for the DEX acquisition were not changed from the institution’s standard-of-care X-ray technique and included the use of a grid, an SID of 180 cm, 120 kVp, and the defaults for AP/lateral views of 5.2/10.4 mAs for small adults (21/31 cm thickness), 6.3/12 mAs for medium adults (25/37 cm thickness), and 8.0/14.4 mAs for large adults (30/44 cm thickness).
Data Analysis
All images were independently reviewed by 2 subspecialty-trained chest radiologists with 6 and 32 years of clinical experience, respectively, on a clinical PACS workstation (CORAL v. 3.8.1.3). Initially, the COMP images (posteroanterior and lateral) were assessed for the presence or absence of coronary calcium using a confidence scale of −2 = confidently not present, −1 = probably not present, 1 = probably present, and 2 = confidently present. Valve and other vascular (aortic and supra-aortic branches) calcifications were assessed using the same scale and the same images. Subsequently, the BI pair was revealed, and the same readers re-evaluated the images using the same grading approach.
The calcium score was analyzed on the SOC-CT based on the Agatston score using the VScore coronary artery calcium scoring software (Vital Imaging, Florida, United States) and reported as part of the clinical reporting template. For the purpose of this study, this score was categorized according to the Coronary Artery Calcium Data and Reporting System (CAC-DRS, scale ranges from 0 to 3) and served as the reference standard, 9 valve/vascular calcification were categorized on the SOC-CT as present or absent.
Receiver operating curves (ROC) were calculated for the COMP and BI images both for detection of CAC and valve-vascular calcifications, from which the sensitivity, specificity, and accuracy were derived. The performance characteristics of COMP and BI images were evaluated by averaging the readers’ scores and statistically comparing them using the ROC’s area under the curve (AUC) with DeLong’s algorithm using a freely available Python library (https://github.com/yandexdataschool/roc_comparison). 10 This analysis was performed for patients in any CAC-DRS category (ie, categories 1-3), for patients in the CAC-DRS categories 2-3, and for patients only in the CAC-DRS category 3. The rationale for analyzing the combined categories of CAC-DRS 2-3 and CAC-DRS 3 separately was to account for increasing levels of calcification severity, and to better assess the performance of DEX in identifying calcifications in patients with more significant disease (CAC-DRS 2 and 3). The rater reliability was calculated using the Intraclass-Correlation-Coefficient (ICC).
Results
All images obtained with the single exposure, dual-energy detector were reconstructed and eligible for analysis. The age range for the 24 women and 37 men was 22 to 79 years (median, 61 years, interquartile range, 15). Twenty-seven out of 61 patients (44.3%) showed measurable coronary artery calcifications with 12 patients classified in the CAC-DRS category 1 (19.7%), 4 patients in CAC-DRS category 2 (6.6%), and 11 patients in CAC-DRS category 3 (18.0%). A summary of the patient demographics is provided in Table 1.
Patient Characteristics.

Note. IQR = interquartile range.
Detection of CAC in Any CAC-DRS Category 1-3
For the detection of CAC within any CAC-DRS category (1-3) on dual-energy X-ray (DEX) images, the sensitivity and ROC-AUC values varied between the 2 readers. Reader 1 reported a sensitivity of 48.1% using COMP images and 77.8% with BI, whereas reader 2 showed lower sensitivities of 44.4% for COMP and 74.1% for BI. The ROC-AUC was higher for BI (0.89 for reader 1 and 0.87 for reader 2) compared to COMP (0.74 for reader 1 and 0.72 for reader 2). The specificity was 100% for readers and both methods. The combined AUC for the COMP images was 0.74 (CI: 0.64-0.84) and for the BI images 0.91 (CI: 0.83-0.98), the difference was significant (P = .0003).
Detection of CAC in CAC-DRS Categories 2-3
Reader 1 showed an increased sensitivity with BI (86.7%) compared to COMP (66.7%), but the specificity was slightly lower with BI (82.6%) that with COMP (93.5%). ROC-AUC values were 0.81 for COMP and 0.90 for BI. Reader 2 also reported a higher sensitivity with BI (93.3%) compared to COMP (66.7%) and the specificity for BI was 87.0%, slightly lower than 95.7% achieved with COMP; the ROC-AUC values were 0.82 for COMP and 0.94 for BI. The combined AUC for the COMP images was 0.81 (CI: 0.68-0.94) and for the BI images 0.94 (CI: 0.86-1.00), the difference was statistically significant as well (P = .044).
Detection of CAC in CAC-DRS Category 3
For the detection of CAC confined to CAC-DRS category 3 only, reader 1 had a sensitivity of 72.7% for COMP and 81.8% BI images, and the specificity for COMP was 90.0% compared to 76.0% for BI, while ROC-AUC was equal at 0.83 for both methods. Reader 2 noted a sensitivity of 72.7% for COMP and 90.9% for BI, and a higher specificity for COMP (92.0%) compared to BI (80.0%); the ROC-AUC was slightly higher for BI (0.90) than COMP (0.84). The combined AUC for the COMP images was 0.84 (CI: 0.69-0.98) and for the BI images 0.89 (CI: 0.77-1.00), the difference was statistically not significant (P = .42).
Detection of Valve and Vascular Calcifications
Reader 1 showed a sensitivity of 52.4% with COMP and 76.2% with BI. The specificity was 97.5% for both methods, with ROC-AUC values of 0.89 for COMP and 0.98 for BI. Reader 2 also reported a higher sensitivity for BI (76.2%) compared to COMP (57.1%), and the specificity was 97.5% for both methods as well, and ROC-AUC was 0.91 for COMP and 0.98 for BI. The combined AUC for COMP images was 0.91 (CI: 0.83-0.99) and for the BI images 0.98 (CI: 0.96-1.00), without a statistical difference (P = .55).
Rater Reliability
The Intraclass-Correlation-Coefficient (ICC) for CAC on COMP and BI was 0.973 and 0.954, indicating a excellent agreement. The ICC for valve/vascular calcifications was 0.971 and 0.965, indicating excellent agreement as well.
The results are summarized in Table 2. Figures 1 and 2 provide example cases comparing DEX and CT, illustrating the improved detection of CAC using the bone image (BI). Figure 3 summarizes the combined ROC analysis (both readers) for the COMP images and BI images.
Results Summary.
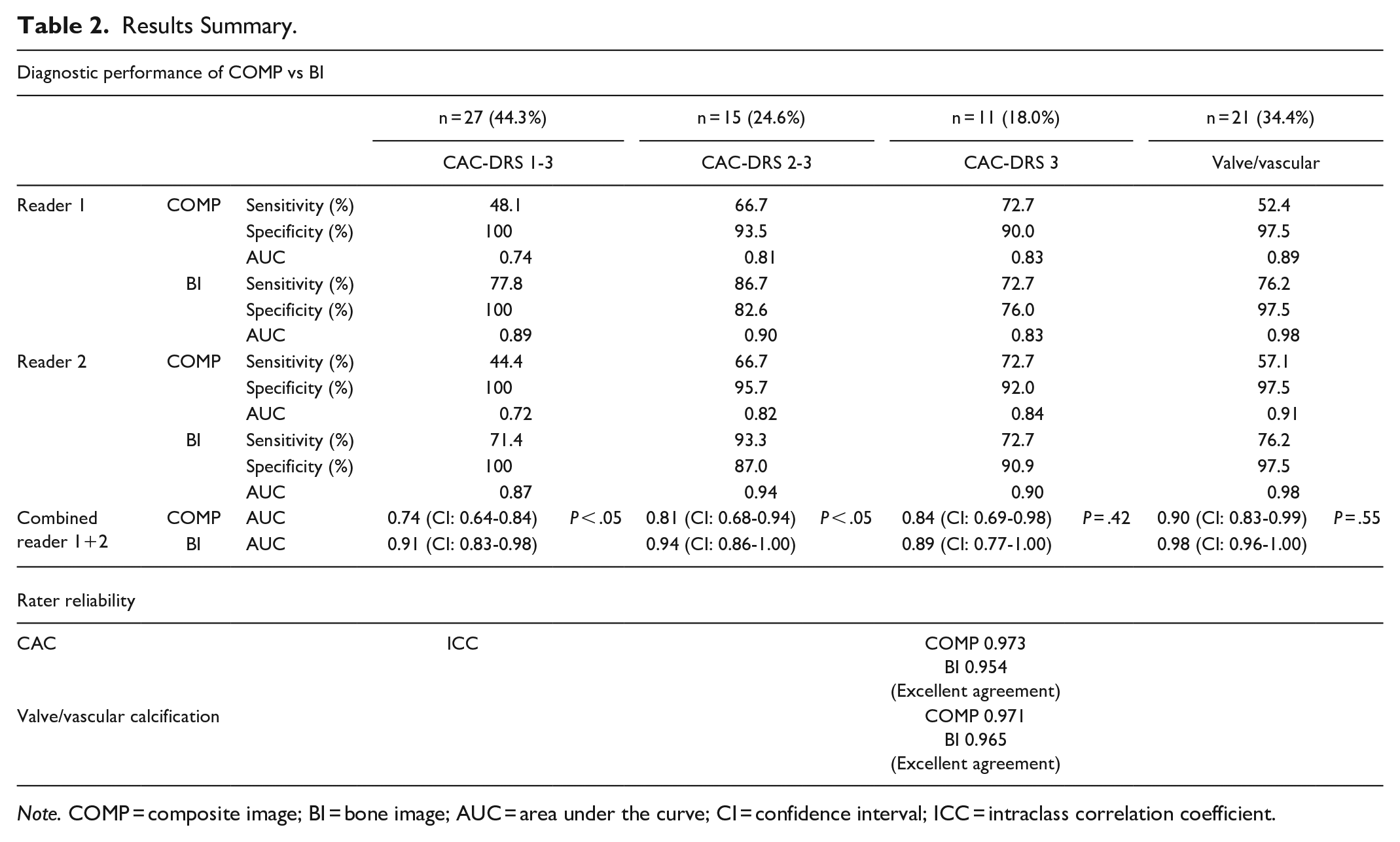
Note. COMP = composite image; BI = bone image; AUC = area under the curve; CI = confidence interval; ICC = intraclass correlation coefficient.
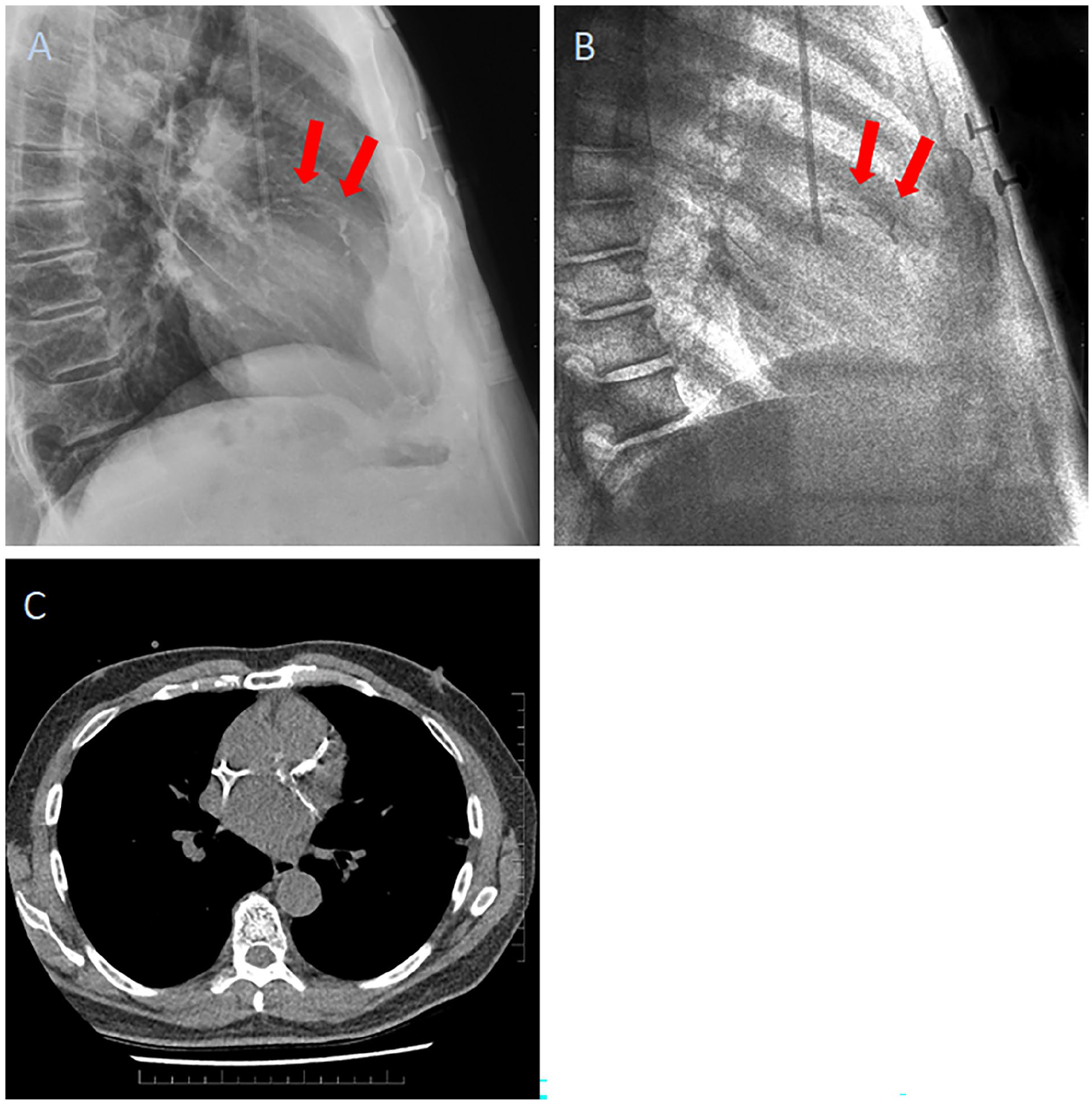
(A) Composite DEX image (COMP, lateral view) and (B) bone image (BI, lateral view); the arrows point at the CAC, better visualized on the BI image. (C) shows the corresponding CT.
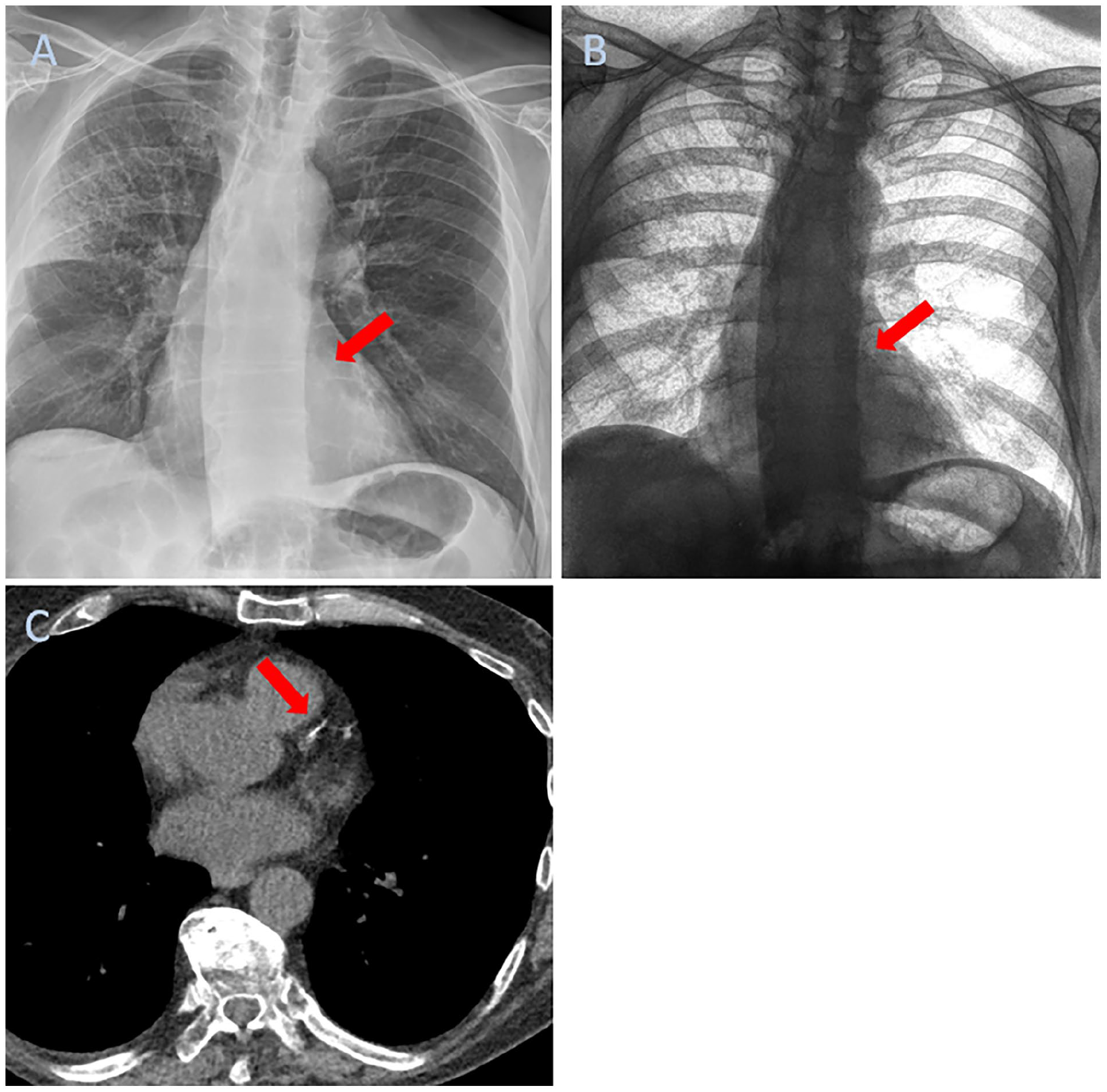
(A) Composite DEX image (COMP, AP view) and (B) bone image (BI, AP); the arrows point at the CAC in the LAD that is nearly invisible on the COMP image. (C) shows the corresponding CT with an Agatston score of 123.
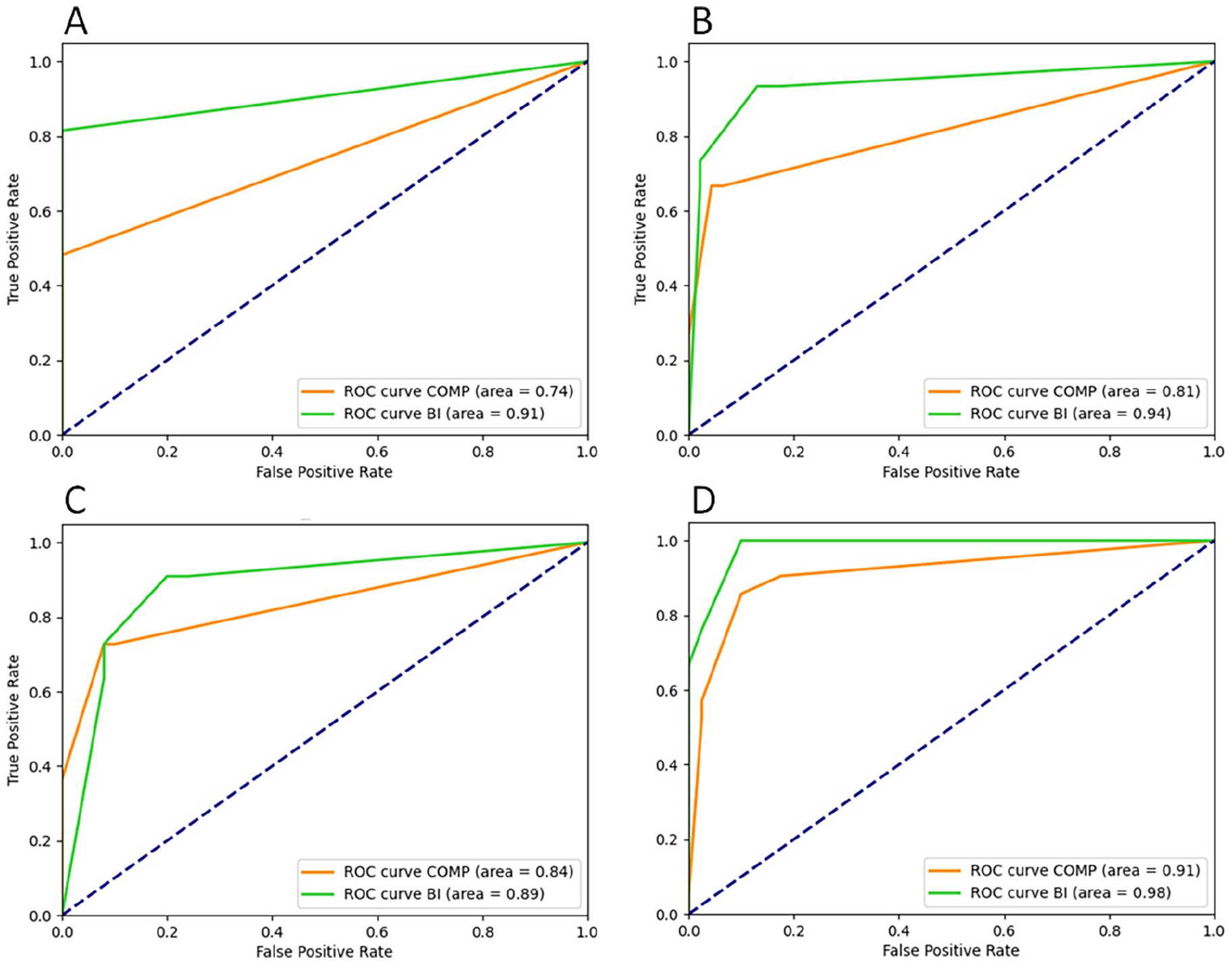
Combined ROC analysis of CAC in any CAC-DRS category 1-3 (A), in CAC-DRS categories 2-3 (B), in CAC-DRS category 3 only (C) and for valve/vascular calcifications (D). The AUC was larger for bone images (green line) but reached significance only in ROC (A) and (B).
Discussion
Our study demonstrated that single-exposure, dual-energy X-rays helped detect coronary artery calcium (CAC) and valve/vascular calcifications with higher diagnostic confidence than composite X-ray images alone. Specifically, for any severity of CAC, the bone images (BI) derived from soft-tissue subtraction in dual-energy X-ray significantly improved the detection of CAC compared to conventional composite X-ray images. Both readers showed statistically excellent agreement in their image assessment. Interestingly, the sensitivity for both readers in the BI group was lower for CAC-DRS category 3 compared to CAC-DRS categories 2-3. Besides the lower number of patients in the CAC-DRS category 3, one potential additional explanation might be that the dual-energy technique demonstrates its higher sensitivity more evidently in detecting early or moderate calcifications (as seen in patients with CAC-DRS 1 or 2) but less so when calcifications are more extensive and denser (as in CAC-DRS 3) and already sufficiently visible on the composite images. Allowing for this, the overall diagnostic benefit of single-exposure dual-energy imaging remains evident as it improves detection across varying degrees of calcifications.
It is noteworthy that the presence and amount of coronary artery calcium (CAC) are strongly associated with the overall atherosclerotic plaque burden and the risk of cardiovascular events. Studies consistently showed that individuals with detectable CAC have a significantly higher risk of myocardial infarction, ischaemic stroke, and cardiovascular mortality compared to those without CAC. 11 Additionally, the progression of CAC over time independently predicts future cardiovascular events, regardless of traditional risk factors. 12 Valve and other vascular calcifications are also important markers of cardiovascular risk 13 and may warrant to be opportunistically screened as well.
Simulation experiments have demonstrated that it is feasible to capture a detectable coronary artery calcification signal in dual-energy, double-exposure chest X-rays. 14 Previous attempts to use this technique for coronary calcium detection in clinical practice, however, have faced challenges predominantly due to inherent motion artefacts from the 2 separate exposures and the partial obstruction of coronary arteries on the PA projection by the sternum, vertebral bodies, and rib cartilage calcifications.5,6 Furthermore, the diagnostic benefit of dual-exposure, dual-energy X-ray for detecting pulmonary nodules, calcifications and determination of malignancy has been questioned. 15 Single-exposure dual-energy X-ray, on the contrary, eliminates these technical and clinical limitations and enables the acquisition of lateral dual-energy images. 16 This technology has been shown to improve lesion visibility by up to 43%, find up to 33% more cases of pneumonia, and increase diagnostic confidence in up to 57% of cases with no added radiologist reading time compared to conventional X-rays.17,18
Limitations of single-exposure, dual-energy X-rays include the risk for higher radiation doses compared to conventional radiography when the exposure parameters are adjusted to generate low-noise subtraction images. We did not modify technical exposure parameters for DEX in our study; instead, we used the institutional standard-of-care parameters. Furthermore, the processing times are notably longer compared to conventional X-rays and range between 5 and 15 seconds; however, with rapidly increasing computing power, this limitation may soon become irrelevant. Whether or not DEX and its superiority in detecting calcifications impacts the radiologists’ interpretation time remains to be investigated.
Cardiac CT coronary calcium scoring is an excellent predictor of future cardiac events. 19 While dual-energy X-ray technology cannot compete with the diagnostic performance of cardiac CT calcium scoring, it allows a rapid assessment for a more widely distributed opportunistic screening for CAC at lower cost and lower radiation. 20 In clinical practice, this may not only be useful in low- and middle-income countries or other resource-constrained areas; relying solely on clinical screening through risk factors is insufficient to accurately identify all at-risk patients. Clinical guidelines alone can identify, at best, two-thirds of those at risk, leading to many at-risk individuals not qualifying for CT calcium score according to guideline recommendations. While it is important to note that the availability of DEX technology in underserved regions may not be as widespread as that of CT, its implementation can still provide a valuable alternative for early detection when CT is not accessible. Early identification of higher-risk patients has the potential to lead to earlier treatment and ultimately reduce the burden on the healthcare system imposed by heart failure, myocardial infarction, and other cardiac diseases. Notably, about $108.7 billion USD was spent on the treatment of heart disease in adults in the United States alone in 2017. 21
Particularly for patients who have undergone bone marrow transplants, early identification and treatment of cardiac disease is vital. These patients are at higher risk of numerous cardiac complications, including heart failure, arrhythmias, pericardial tamponade, cardiac arrest, cardiomyopathies, ischaemic heart disease, vascular disease, stroke, or other comorbid conditions. 22 Recent studies highlight that patients who have undergone haematopoietic stem cell transplants (HCT) face increased risks of both short- and long-term cardiovascular events and mortality. This elevated risk is partly due to cardiotoxic chemotherapy, radiation exposure, and their effects on cardiovascular reserve. Given these risks, the growing focus in cardio-oncology emphasizes the importance of early detection of cardiotoxicity and cardiovascular disease in this population, with the number of HCT survivors projected to reach 502 000 in the United States by 2030. 19 These patients routinely undergo imaging prior to hospital admission as well as during their extensive follow-up. Noting the presence of coronary or valve/vascular calcifications on routine imaging could enable more prompt and dedicated cardiovascular risk assessment and, where necessary, the initiation of appropriate treatment strategies.
Our study has three main limitations. First, we used a non-gated chest CT as the reference standard for the detection of CAC and valve and other vascular calcifications. Shin et al have shown that non-gated wide-area CT correlates highly with EKG-gated calcium-scoring CT, in particular when thinner slices are used. 23 Excellent agreement between non-gated and gated CT has also been reported when low-dose technique is applied for the non-gated CT. 24 On the other hand, CAC is slightly underestimated on non-gated CT as motion during image acquisition carries the risk that the focal higher calcium density projects onto a larger local area in the image, thus the peak density might be reduced. 25 It is therefore possible that we underestimated the true CAC scoring on our reference standard, non-gated CT, and subsequently overestimated the sensitivity on DEX. Second, we included only a limited number of patients in clinical care who were scanned for complications, mainly to rule out pneumonia, during their bone marrow transplant treatment. This patient population and the populations’ distribution of calcium scores are not representative for other screening populations, and generalizing conclusions may need to be drawn with caution. Lastly, we acknowledge that the small number of patients, particularly in category 2, limited our ability to perform independent assessments. This small sample size may impact the robustness of our findings and underscores the need for further studies with larger and more diverse populations to validate our results.
In conclusion, single-exposure dual-energy chest X-ray improves the identification of both coronary artery calcium and valve/vascular calcifications with higher diagnostic confidence when compared to conventional X-ray imaging. Our findings suggest that bone images derived from DEX may even be more diagnostically helpful in patients with lower CAC burden than in patients with heavy calcifications. Although DEX may not be suited for quantification of coronary artery calcifications, the incidental detection on chest X-ray may help with further risk stratification and potential guide to further workup if clinically warranted. Further larger-scale investigations are warranted based on these preliminary findings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jay Potipcoe, Steve Tilley, Nathan Speck, Neda Afkhami are employees of KA Imaging. Karim S. Karim is the co-founder, KA Imaging. The other authors declare no conflicting interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was internally funded.
