Abstract
Purpose:
To assess the diagnostic accuracy of limited chest ultrasound in detecting pneumothorax following percutaneous transthoracic needle interventions using chest X-ray (CXR) as the reference standard.
Methods:
With IRB approval, after providing consent, asymptomatic patients after percutaneous transthoracic needle interventions were enrolled to undergo limited chest ultrasound in addition to CXR. A chest Radiologist blinded to the patient’s prior imaging performed a bedside ultrasound, scanning only the first 3 anterior intercostal spaces. Pneumothorax diagnosed on CXR was categorized as small or large and on ultrasound as grades 1, 2, or 3 when detected in 1, 2, or 3 intercostal spaces, respectively.
Results:
38 patients underwent 36 biopsies (34 lungs, 1 pleura, and 1 mediastinum) and 2 coil localizations. CXR showed pneumothorax in 13 patients. Ultrasound was positive in 10 patients, with 9 true-positives, 1 false-positive, 4 false-negatives, and 24 true-negatives. The false positive results were due to apical subpleural bullae. The false-negative results occurred in 2 small apical and 2 focal pneumothoraces at the needle entry sites. Four pneumothoraces were categorized as large on CXR, all of which were categorized as grade 3 on ultrasound. Sensitivity and specificity of US for detection of pneumothorax of any size were 69.23% (95%CI 38.6%, 90.1%) and 96.0% (95%CI 79.6%, 99.9%), and for detection of large pneumothorax were 100% (95%CI 39.8%, 100%) and 100% (95%CI 89.7%, 100%).
Conclusions:
Results of this prospective study is promising. Limited chest ultrasound could potentially replace CXR in the management of postpercutaneous transthoracic needle intervention patients.
Introduction
Percutaneous transthoracic needle interventions (PTNI) are well-established diagnostic and therapeutic techniques that include, but are not limited to, fine needle aspiration, core biopsy, thermal ablation, and preoperative localization of lung nodules. Pneumothorax is the most common complication of PTNI, with reported rates ranging from 10% to 49%, with 6% to 56% requiring pleural drainage catheter insertion, depending on multiple factors, including the type and technique performed.1-14 The main risk factors for post-biopsy pneumothorax include larger needle size, a higher number of needle passes, longer procedure duration, the longer distance of nodule from the pleura, and the presence of emphysema.1-6,15 In some centers, post-PTNI patients are admitted to the hospital for continuous monitoring that includes follow-up chest X-ray (CXR) to detect pneumothorax.1,2,6,9,11,12,15 However, studies have shown that transthoracic needle biopsy performed on an outpatient basis, including outpatient management of post-procedural pneumothorax, is safe and cost-effective.3,7,10,14,16 Therefore, many institutions practice early discharge of patients within 1-4 hours after the procedure, including those with a stable small asymptomatic pneumothorax.
Traditionally, the assessment for post-procedure pneumothorax has been based on CXR performed before discharge. Large pneumothoraces are always detectable on upright posteroanterior (PA) view CXR.17,18 More recent studies have shown that ultrasound (US) has high sensitivity in the diagnosis of pneumothorax in the trauma and critical care settings19-21 as well as post-lung biopsy.22-25 Bedside US following lung biopsy has some advantages to CXR. It is radiation-free and more convenient for patients as they do not need to be transferred to the X-ray room and be repositioned. US is also fast and can be performed and interpreted by the physician at the time of post-procedural assessment and follow-ups before the discharge. We conducted this prospective study to assess the diagnostic accuracy of the limited bedside chest US in the detection of pneumothorax following PTNI, using upright PA view CXR as the reference standard.
Methods
Study Design and Patients Population
Our institute is a tertiary cancer care center with expertise in thoracic procedures, including biopsy, thermal ablation, and preoperative coil localization of lung nodules.
This prospective diagnostic accuracy study was approved by the Institutional Review Board (IRB). The study was performed from February 2019 to April 2019 inclusive. Since the US exams were to be performed in a very short period of time from the CXR, moving an US machine from the US department was not practical. Due to limited resources, an US machine dedicated to this study, stationed in the patient observation area, was only available for 3 months.
A fellowship-trained chest radiologist, who had completed formal radiology residency training, which included ultrasound and 1-year post-residency training in chest imaging and 5 years of experience, performed the post-procedure US examinations within 10 minutes of the CXR. The radiologist was available in 15 nonconsecutive days, during which 51 patients underwent image-guided PTNIs. The radiologist was scheduled on tasks such as running multidisciplinary conferences and reporting CT scans of inpatients. Therefore, the radiologist was only able to approach 39 patients, of which 38 patients agreed to have limited chest US in addition to the standard of care upright PA view CXR. One patient was reluctant to consent. The reason was not asked, and according to the study protocol, the patient was not persuaded to participate. Signed informed consent was obtained from all participating patients. Patients with symptoms, such as shortness of breath or hemoptysis, were excluded to avoid interfering with patient care and subjecting the radiologist to a potential cognitive bias. The management of post-PTNI patients was based on upright inspiratory PA view CXR findings and clinical assessment only.
Institutional PTNI and Management
The PTNIs were performed following institutional standards, after written consent, under aseptic conditions and local anesthesia. Procedures were performed under fluoroscopy or CT guidance by 4 chest radiologists and 2 chest radiology fellows under supervision. Patients were positioned according to the lesion’s location to ensure the shortest and safest path. The attempts were made to avoid crossing the interlobular fissures, large airways, and vessels.
In our institution, most PTNIs are performed as outpatient procedures. However, the procedures are sometimes performed on inpatients to complete their clinical workup in a timely fashion. Post-procedure patients are transferred onto a stretcher, moved to an observation area, and monitored by a nurse. Thirty minutes after the biopsy, a radiology technologist moves the patient to the X-ray room, where the patient is positioned for CXR. All patients were able to sit upright and hold breath to obtain an inspiratory PA view CXR. On average, it takes 6-8 minutes to obtain the CXR and bring the patient back to the observation area. The radiologist who performed the procedure reviews the CXR when it becomes available on PACS and then visits the patient before discharge. Asymptomatic patients with no pneumothorax and most patients with a small focal pneumothorax limited to the site of needle entry are discharged. Patients with large pneumothorax are managed with pleural drainage catheter insertion. Patients with a small pneumothorax are monitored and will undergo a follow-up upright inspiratory PA view CXR in 30-45 minutes. Asymptomatic patients with a stable small pneumothorax are discharged. Patients with enlarging pneumothorax receive a pleural drainage catheter. Patients discharged with a pleural catheter return in 2 days to remove the catheter, provided the pneumothorax is resolved.
We follow guidelines by The American College of Chest Physicians (ACCP) and The British Thoracic Society (BTS) to determine the size of pneumothorax on CXR. As per ACCP, when the apex-to-cupola distance is >3 cm, pneumothorax is defined as large. 26 Based on BTS guidelines, a distance of >2 cm between the lung margin and the chest wall at the level of hilum denotes a 50% loss of lung volume. 27 Patients are considered clinically stable, if according to ACCP guideline, 26 they are readily able to speak an entire sentence and have stable vital signs, including the respiratory rate < 24/min, O2 saturation ≥ 90% on room air, pulse rate between 60-120 beats/min, and normal blood pressure.
Bedside Chest Ultrasound
Limited chest US was performed with a GE Logiq E portable machine, using an 8L linear transducer (14 × 48 mm Footprint, 4-13 MHz). The radiologist performed the post-procedure US examinations within 10 minutes of the CXR. The radiologist was informed which hemithorax to scan but was blinded to the patient’s underlying lesion, the procedure, and the post-procedure CXR results. The radiologist, who was familiar with performing and reporting US examinations mainly for localizing loculated pleural effusion and US-guided biopsy of the chest wall and pleural lesions during his 5 years of practice, watched an approximately 10-minute-long online training video 28 to learn utilizing chest US for diagnosing pneumothorax. The first 3 anterior intercostal spaces in the midclavicular line were scanned with the patient sitting up on the stretcher. All patients were able to sit upright on the stretcher. The supraclavicular fossa was also scanned whenever the patient’s body habitus allowed. The decision to scan only the first 3 intercostal spaces was based on the fact that in the upright position, pneumothorax is presumably localized to the nondependent upper hemithorax, and compared to standard chest US, this protocol would take a shorter time to perform.
The US machine was stationed in the observation area, and the US exam time varied between 1 and 2 minutes. The results of the US were documented as negative or positive for pneumothorax. Pneumothorax was diagnosed based on the absence of normal lung sliding or comet-tail artifact, with or without lung point 29 (Figure 1). Depending on the number of intercostal spaces where pneumothorax was detectable, positive cases were graded as 1, 2, or 3 (i.e., grade 1: seen above the clavicle or only in first intercostal space; grade 2: seen in first 2 intercostal spaces, and grade 3: seen in all 3 intercostal spaces). There was no change in the clinical assessment and management of patients based on the US results.
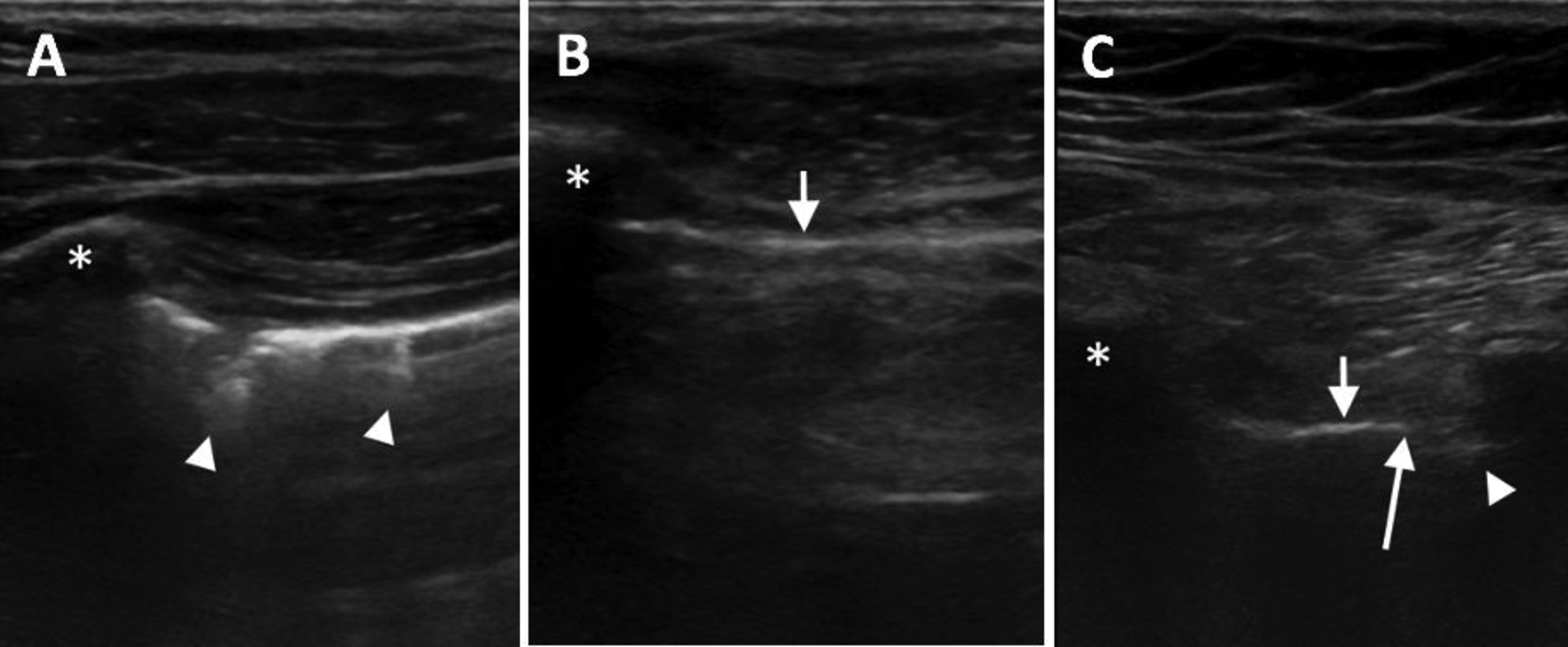
Longitudinal views of superior anterior chest US in midclavicular line in 3 patients with different chest wall thickness. Asterisks show the ribs. A, Comet-tail artifacts (arrow heads) indicate normal lung, no pneumothorax. B, Solid line (short arrow) indicates pneumothorax. C, Solid line (short arrow) indicates pneumothorax, comet-tail artifact (arrow head) indicate normal lung. The point between pneumothorax and normal lung where visceral and parietal pleura are separated is lung point (long arrow).
Statistical Analysis
The sensitivity and specificity of limited chest US in the detection of pneumothorax were calculated using the upright inspiratory PA view CXR as the reference standard. Accuracy using disease positive threshold of “all” pneumothoraces and “large” pneumothoraces were calculated. A statistical software program (MedCalc Version 19.2) was used to calculate 95% confidence intervals (CI) based on the binomial distribution for these diagnostic indices.
Results
A total of 38 patients underwent 38 PTNIs. None of the patients were symptomatic or had any other reason to be excluded. Table 1 summarizes patient demographics and the details of the PTNIs. Two biopsy cases were not lung lesions, one was a biopsy of a mediastinal mass, and the other was a biopsy of a pleural nodule. Although the rate of pneumothorax after biopsy of mediastinal and pleural lesions is expected to be less than lung lesions, these cases were not excluded since the radiologist performing the US was blinded to the location of the biopsied lesions and results of US in both of these cases were true negative.
Summary of Patient Demographics and Procedures.
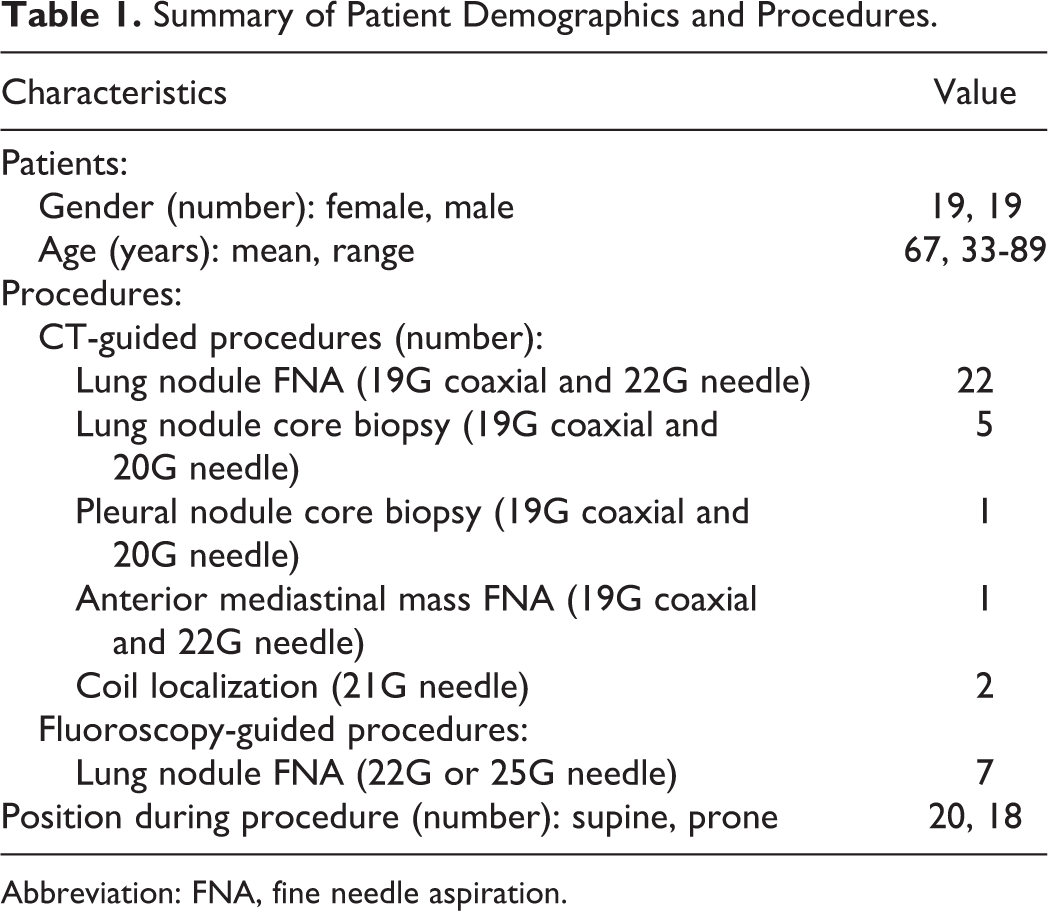
Abbreviation: FNA, fine needle aspiration.
CXR demonstrated a pneumothorax in 13 (34.2%) of 38 patients, 4 were large, and 9 were small. US identified 9 (69%) of 13 pneumothoraces. All large pneumothoraces were categorized as Grade 3 on US, whereas detected small pneumothoraces were categorized as Grade 2 or Grade 1. Pneumothorax sizes for both US and CXR are summarized in Table 2.
Results of CXR and Ultrasound for Pneumothorax.
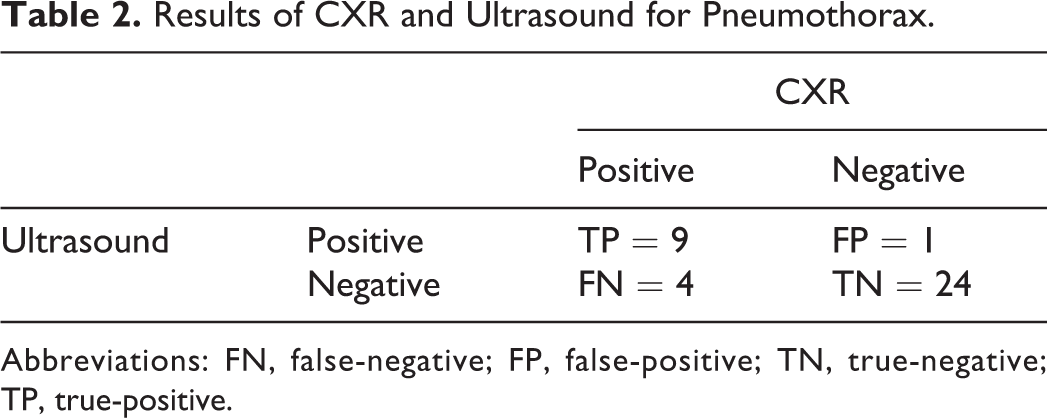
Abbreviations: FN, false-negative; FP, false-positive; TN, true-negative; TP, true-positive.
Two of the 4 pneumothoraces not detected on US were small apical pneumothoraces. The other 2 were small focal pneumothoraces at the needle entry site, in the area not scanned at the time of US examination (Figure 2). In one patient, US diagnosed a small (grade 1) pneumothorax, which was not present on CXR. A subsequent review of chest CT in this patient revealed apical subpleural bullae, simulating pneumothorax on US (Figure 3). In 24 patients, both CXR and US were negative for pneumothorax. US showed a sensitivity of 69.23% (95%CI 38.6% to 90.1%) and a specificity of 96.0% (95%CI 79.6% to 99.9%) in the detection of pneumothorax of any size; and a sensitivity of 100% (95%CI 39.8% to 100%) and specificity of 100% (95%CI 89.7% to 100%) in the detection of large pneumothoraces. Table 3 summarizes the size and grading of the detected pneumothoraces on CXR and US.
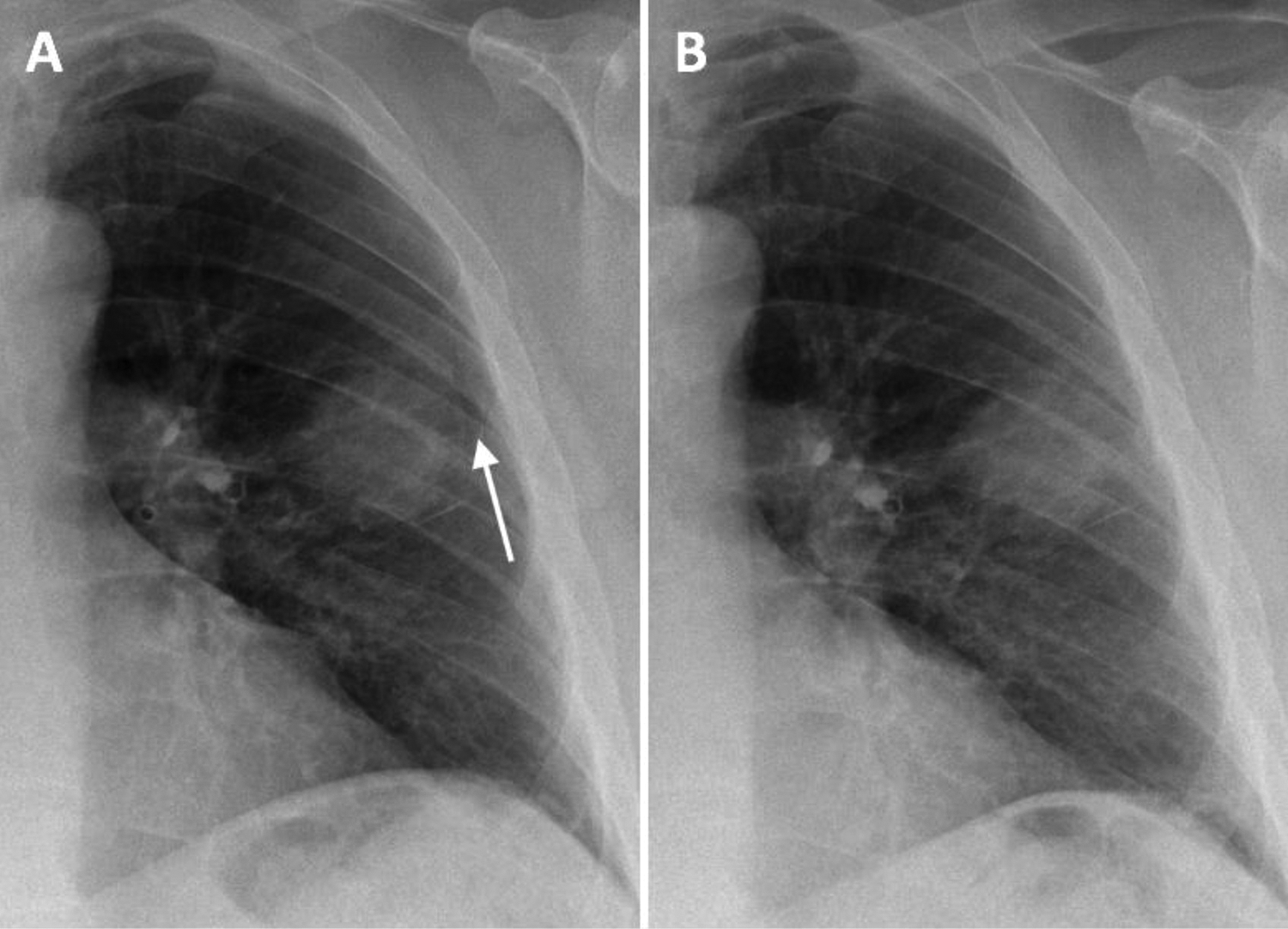
A, Focal pneumothorax at the needle entry site adjacent to the biopsied mass (Arrow). B, Follow-up CXR in 45 minutes shows resolution.
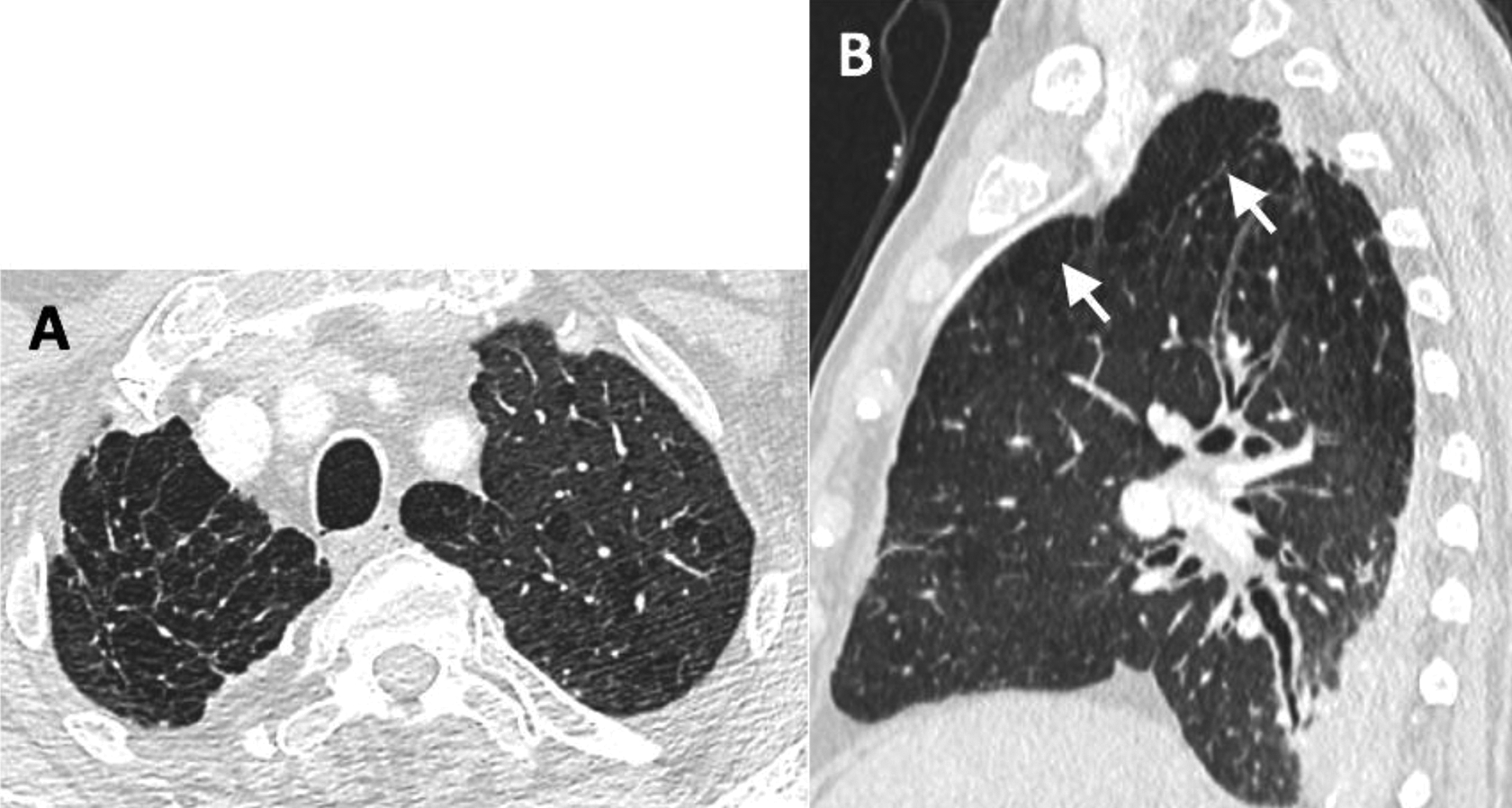
Axial (A) and sagittal (B) CT images of the chest in a patient with subpleural bullae at the right apex (arrows) mimicking pneumothorax on ultrasound.
Comparison of Size of Pneumothorax on CXR and Ultrasound.
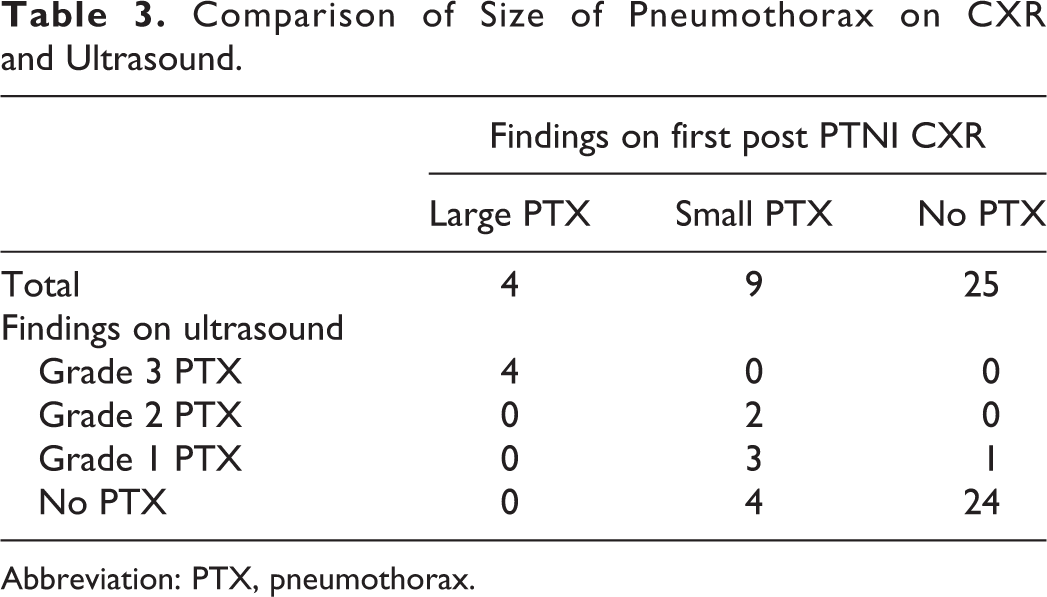
Abbreviation: PTX, pneumothorax.
Three of 4 patients with large pneumothorax were managed with pleural catheter insertion (23.1% of total). One patient categorized as having a large pneumothorax was asymptomatic in the post-biopsy period. Pneumothorax in this patient decreased in size on the follow-up radiograph, and the patient was discharged without a pleural drainage catheter.
Discussion
In our patient cohort, bedside US correctly detected and characterized all large pneumothoraces, with no false-negative results. US was also highly specific in the detection of post-PTNI pneumothorax of any size. The only false-positive pneumothorax on US was related to apical subpleural bullae, which like severe emphysema and pleural adhesions, may mimic pneumothorax on US.30,31 Knowledge of the presence of such findings on CT when planning the procedure could eliminate such misdiagnoses. One should be cautious in diagnosing pneumothorax in patients with apical subpleural bullae. CXR may be performed to confirm pneumothorax in such cases. In this study, 4 pneumothoraces were not detected by US, including small apical pneumothoraces in 2 patients in which the supraclavicular regions were not scanned, and 2 small focal pneumothoraces localized to the site of needle entry, outside of the scanned zone (i.e. the first 3 intercostal spaces). The focal pneumothoraces at the needle entry site can be seen on CT images during or at the end of the procedure, and in our experience, they remain stable. Therefore, performing US over the needle entry site, which may require patient repositioning and removing the dressing, is of low yield in detecting a clinically significant pneumothorax.
Few studies in the literature compare the accuracy of US with CXR in the detection of pneumothorax following transthoracic lung biopsy. In a study by Chung et al, 23 using CT as the reference, US showed significantly higher sensitivity than supine or semierect CXR when the entire anterior chest wall is scanned. In another study, Sartori et al 22 showed that chest US is as accurate as upright PA view CXR in detecting pneumothorax after ultrasound-guided biopsy of peripheral lung lesions extending to the pleura. They scanned the entire chest in both supine and prone positions and used CT as the reference only when there was a discrepancy between the US and CXR. The US protocol by Sartori et al takes a considerable amount of time to perform, which may explain why it is not widely adopted. In our study, the rate of pneumothorax detected on upright inspiratory PA view CXR (34.2%) and the rate of catheter insertion in those with pneumothorax (23.1%) were comparable to previously published data.1-14 We limited the US examination to the first 3 intercostal spaces, which takes much less time to perform. Also, all patients underwent a standard upright inspiratory PA view CXR, which is widely accepted as the modality of choice to detect and quantify post-procedure pneumothorax for management decision-making.
Our study had limitations: First, all US examinations were performed by one radiologist. Therefore, we could not assess inter-observer variation. However, US examination for pneumothorax is a facile skill to learn; and the findings are easy to interpret by radiologists who have general US knowledge and skills. Therefore, we believe multiple readers would most likely achieve comparable results. Second, the sample size was small. Although the US showed good diagnostic performance in the detection of pneumothorax in our patient cohort, future studies with a larger number of patients, and more than one reader, is required to verify the validity of our findings. Third, we used a transducer with a relatively large footprint, which could hamper scanning the supraclavicular fossa in some patients depending on body habitus. However, in our experience, pneumothoraces limited to the apex are unlikely to be large and require further management. Fourth, all patients in this study were able to sit upright. Although pneumothorax can be diagnosed with US in supine position, scanning different areas of the chest (i.e., inferior anterior intercostal spaces) is required. Therefore, the results of this study cannot be applied to those who cannot sit upright.
In summary, this prospective study showed promising results. Bedside limited chest US, which takes less time to perform than standard chest US, has high accuracy for detecting pneumothoraces requiring treatment and, therefore, potentially could replace CXR. This US protocol may also be used for follow-up of small pneumothoraces in asymptomatic patients. However, further study with a larger sample size is required to validate. The radiologist who reviews the patient’s chest imaging and performs the biopsy would be the best physician to perform the US and manage the patient.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
