Abstract
Radiologists and other diagnostic imaging specialists play a pivotal role in the management of osteoporosis, a highly prevalent condition of reduced bone strength and increased fracture risk. Bone mineral density (BMD) measurement with dual-energy X-ray absorptiometry (DXA) is a critical component of identifying individuals at high risk for fracture. Strategies to prevent fractures are consolidated in the Osteoporosis Canada clinical practice guideline which was updated in 2023. In this guideline, treatment recommendations are based upon a consideration of fracture history, 10-year major osteoporotic fracture (MOF) risk, and BMD T-score in conjunction with age. The current review aims to familiarize radiologists and other diagnostic imaging specialists with the reporting requirements needed to support implementation of this guideline using the FRAX™ risk calculation tool. Fortunately, for specialists already familiar with the Canadian Association of Radiologists and Osteoporosis Canada (CAROC) tool, the transition to FRAX-based reporting is readily accommodated in a radiology workflow.
Introduction
Radiologists and other diagnostic imaging specialists play a pivotal role in the management of osteoporosis, a highly prevalent condition of reduced bone strength and increased fracture risk, as highlighted in the recently updated practice guideline from the Canadian Association of Radiologists (CAR). 1 The consequences of osteoporosis-related fractures are substantial and includes pain, morbidity, loss of independence, increased mortality, and a large economic burden on the healthcare system and society. 2 The maxim that “fractures beget fractures” highlights the importance of primary fracture prevention to avoid the cascade of recurrent fractures. 3 Strategies to prevent fractures have been consolidated in the Osteoporosis Canada clinical practice guideline, updated in 2023. 4 The current review aims to familiarize radiologists and other diagnostic imaging specialists with the fracture risk reporting requirements needed to support implementation of this guideline. Although both FRAXTM and the Canadian Association of Radiologists and Osteoporosis Canada (CAROC) tool are acceptable fracture risk reporting tools, FRAX is recommended for clinical management in the Osteoporosis Canada guideline for reasons explained below. 4 Fortunately, for specialists already familiar with CAROC the transition to FRAX-based reporting is readily accommodated into a radiology workflow.
Evolution of Absolute Fracture Risk Prediction
Bone mineral density (BMD) measurement with dual-energy X-ray absorptiometry (DXA) is a critical component of identifying individuals at high risk for fracture. In the early 1990s the World Health Organization (WHO) Study Group proposed epidemiologic criteria for diagnosis of osteoporosis based upon a BMD of 2.5 or more standard deviations below the young adult BMD (T-score −2.5 or lower).5,6 This same threshold has been adopted into clinical practice as an operational criterion for diagnosis of osteoporosis, 7 recognizing that a clinical diagnosis of osteoporosis is appropriate in individuals with a typical fracture history including hip, vertebral, and other multiple fractures. Since the WHO report, it has been learned that only a minority of fractures occur in older individuals with BMD in the osteoporotic range. 8 Conversely, not all individuals with an osteoporotic BMD measurement are at equally high fracture risk. 9 Therefore, strategies to enhance fracture prediction that combine clinical risk factors with BMD have proven to be more successful than BMD alone. 10
Fracture Risk Prediction in Canada: Brief History of FRAX and CAROC
A shift to absolute fracture prediction through the Canadian Association of Radiologists and Osteoporosis Canada collaboration dates back to 2005. 11 The 2005 CAROC fracture risk prediction tool used the best available evidence (Swedish fracture data) to construct a semiquantitative classification of 10-year major osteoporotic fracture (MOF) risk as low (less than 10%), moderate (10%-20%), and high (greater than 20%). 12 This paradigm was prescient and anticipated subsequent development of the fracture risk assessment (FRAX™) initiative in 2008 (history reviewed in Kanis et al 13 ). FRAX was designed to accommodate the large international variations in fracture risk by calibrating each tool to fracture risk within that specific population, with currently more than 80 FRAX tools across the world.14,15 Nationwide fracture data were obtained through the Public Health Agency of Canada (PHAC) and led to the creation of a Canadian FRAX tool in 2010.16,17 The Canadian FRAX tool was independently validated in 2 large Canadian population-based cohorts.18,19 FRAX provides measures of both 10-year MOF risk and 10-year hip fracture risk, though only MOF risk is used in the Osteoporosis Canada clinical practice guideline. 4
The Canadian FRAX tool was not readily available when Osteoporosis Canada released its 2010 clinical practice guideline.20,21 Therefore, to support implementation of this guideline, the decision was made to update the CAROC tool using the same Canadian fracture data as used in the Canadian FRAX tool. 22 Pharmacologic treatment was recommended for individuals with high fracture risk (greater than 20%) in addition to those with a significant fracture history (hip, vertebral, multiple fracture episodes).20,21 Treatment was not recommended for low risk individuals (less than 10%). For individuals with moderate risk (10%-20%, including those with osteoporotic BMD and fracture risk less than 10%) an individualized approach was recommended that considered lateral spine imaging to identify vertebral fractures, additional clinical risk factors and patient preference.
In the 2010 guideline it was noted that “
software for the Canadian FRAX tool is not yet widely available on BMD machines; therefore, for purposes of reporting BMD, the 2010 CAROC tool is the only system that can be applied on a national basis at the present time. This situation may change as FRAX software becomes more widely available. Clinical practitioners need to be aware of the FRAX tool . . . which allows assessment of risk in the absence of a BMD measurement and is more accurate for patients with one or more of the additional risk factors listed above.”
20
The Canadian FRAX and CAROC tools provide valid risk estimates when applied in routine clinical practice (Figure 1). 23 These tools showed exact agreement in risk category in 90% of individuals without previous fracture or glucocorticoid use (the clinical risk factors in CAROC), but greater disagreement (approximately 1 in 3) for individuals with these clinical risk factors.23,24 Direct comparison of these tools showed superior performance (closer agreement between predicted and observed 10-year fracture risk) using the Canadian FRAX (Figure 1), particularly among individuals with previous fracture or recent glucocorticoid use. 23
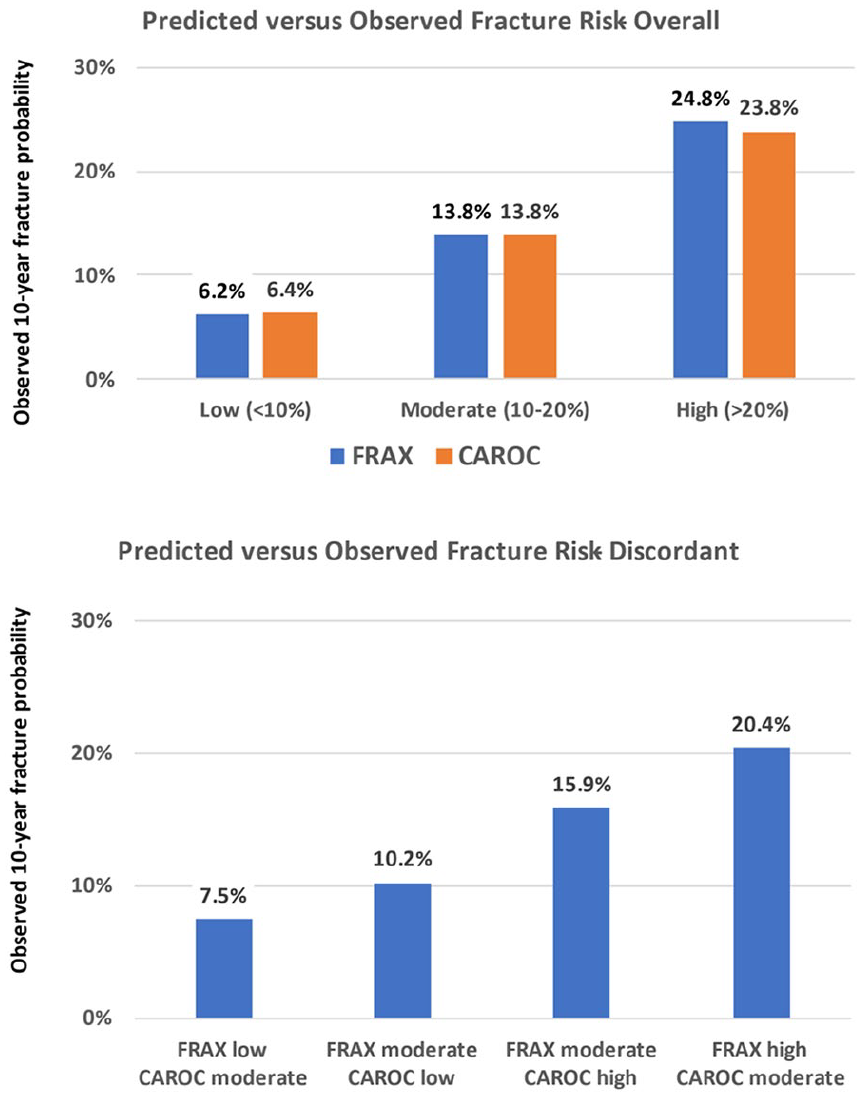
Risk stratification for individuals age ≥50 years not receiving anti-osteoporosis therapy according to FRAX and CAROC risk category: (upper) all 34 060 individuals, (lower) subgroups with risk category discordance. Adapted from Leslie et al. 23
2023 Osteoporosis Canada Guideline: Role of FRAX and CAROC
The 2023 Osteoporosis Canada clinical practice guideline update followed the Grades of Recommendation, Assessment, Development, and Evaluation (GRADE) framework. 25 Risk categories were replaced with treatment recommendations based upon a consideration of fracture history, 10-year MOF risk, and BMD T-score in conjunction with age. 4 Anti-osteoporosis pharmacologic treatment was recommended (strong recommendation) for individuals with high-risk fracture (same definition as 2010 guideline), 10-year MOF risk ≥20%, or BMD T-score in the osteoporotic range in conjunction with age ≥70 years. Treatment was suggested (conditional recommendation, also referred to as a weak or discretionary recommendation) for those with 10-year MOF risk ≥15% but <20% or BMD T-score in the osteoporotic range in conjunction with age <70 years. The reader is referred to the guideline for additional information on when to perform baseline BMD testing, repeat BMD testing, vertebral imaging for detection of undiagnosed vertebral fractures, and pharmacotherapy considerations. 4
The 2023 guideline made a conditional recommendation in favour of the Canada-specific FRAX tool as the preferred tool for fracture risk estimation, with CAROC an alternative fracture risk assessment tool. 4 It was noted that the performance of FRAX was as good or slightly better than other tools, and gave better fracture risk classification than CAROC. Moreover, the 15% threshold used in the 2023 guideline cannot be directly assessed using CAROC. FRAX is now widely available on DXA software and therefore is easier to integrate into a radiology workflow (Figure 2). It was noted that FRAX and CAROC may underestimate fracture risk in the presence of specific risk factors such as recency of fractures, recurrent falls, other comorbidities, or very low BMD at the lumbar spine and total hip sites. Many of these are being addressed as discussed below.
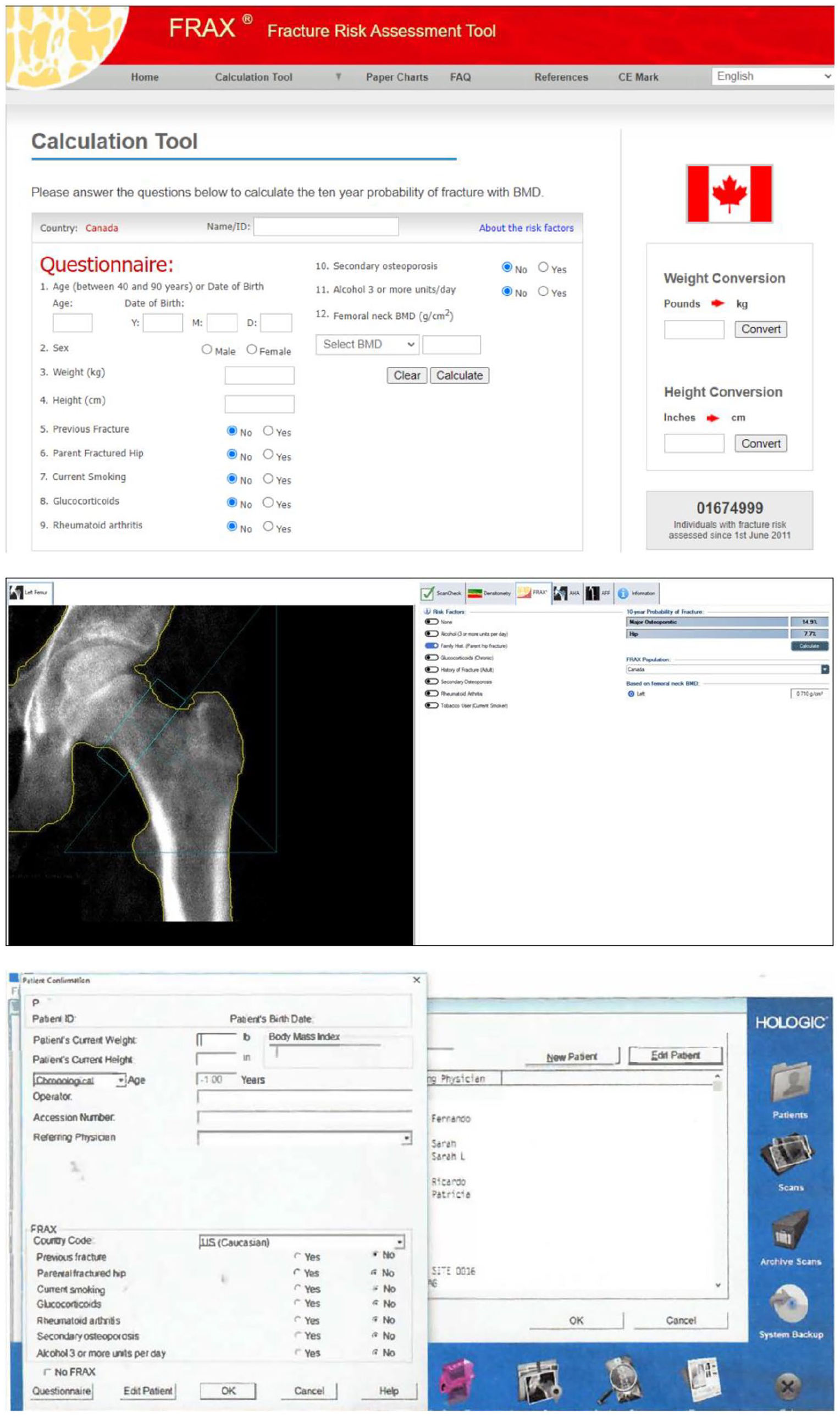
FRAX data entry pages for the website (top), GE DXA scanner (middle), and Hologic DXA scanner (bottom, courtesy Dr. Sarah L Morgan, MD, RD, CCD. University of Alabama at Birmingham).
All individuals who were considered low risk in 2010 fall within the 2023 no treatment category, and individuals considered high risk in 2010 fall within the 2023 recommend treatment category. The 2010 moderate-risk group was refined such that individuals could fall within the 2023 no treatment, suggested treatment, or recommend treatment categories. When this strategy was evaluated in a large clinical population (79 654 individuals age greater than equal 50 years undergoing baseline DXA testing), it was found that among the 33.8% of individuals with 2010 moderate risk categorization, 13.0% were assigned to no treatment, 14.4% to suggest treatment, and 6.4% to recommend treatment (Figure 3). 26 Observed 10-year fracture risk in the reclassified moderate-risk subgroups were all within the 10% to 20% range, with the highest risk being in those in the recommend treatment subgroup.
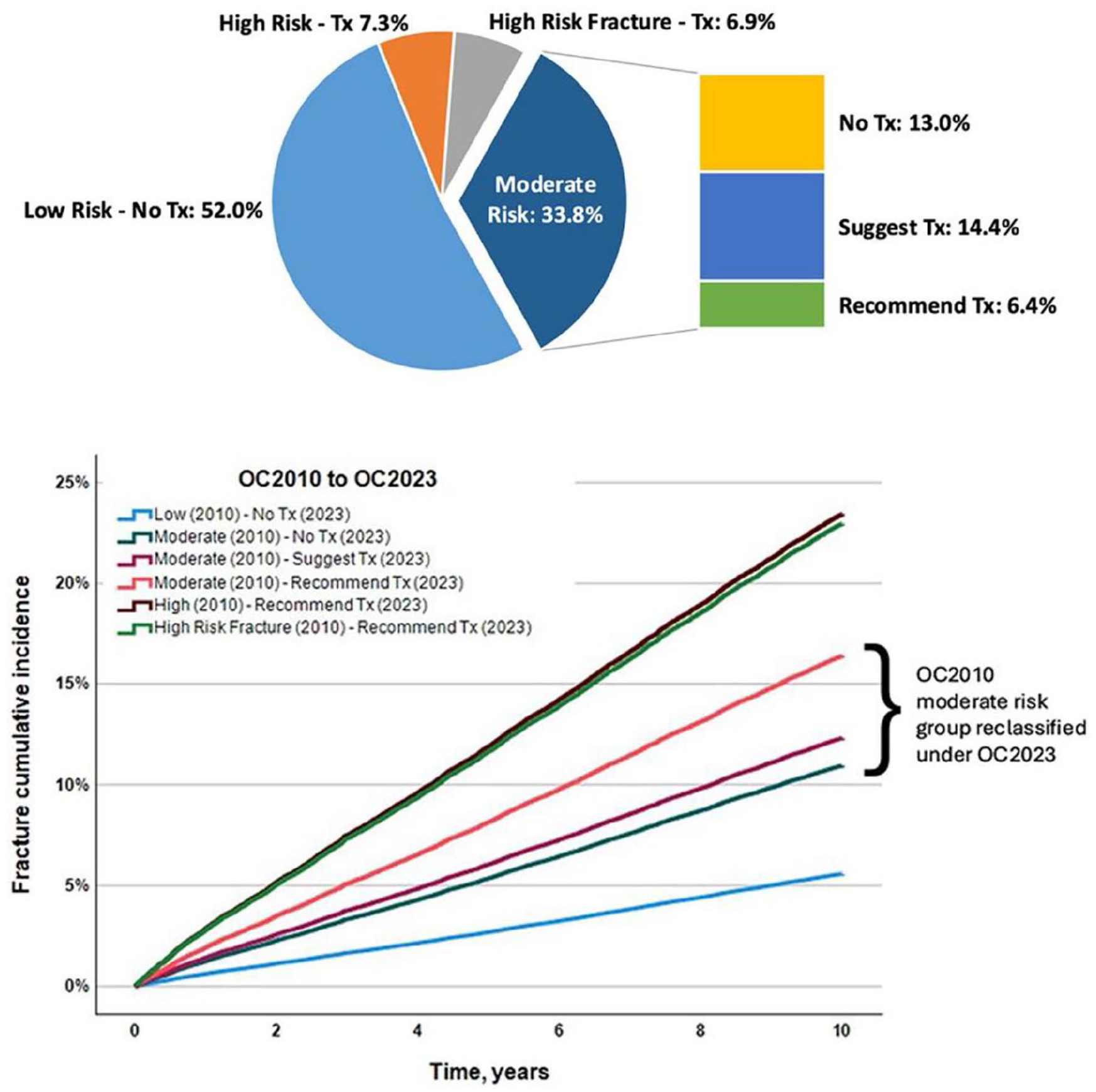
Percentage of individuals qualifying for osteoporosis treatment (upper) and 10-year major osteoporotic fracture cumulative incidence (lower), highlighting reclassification of the Osteoporosis Canada 2010 moderate-risk group. Reprinted with permission. 26
Easing Into FRAX
Relative Importance of FRAX Inputs
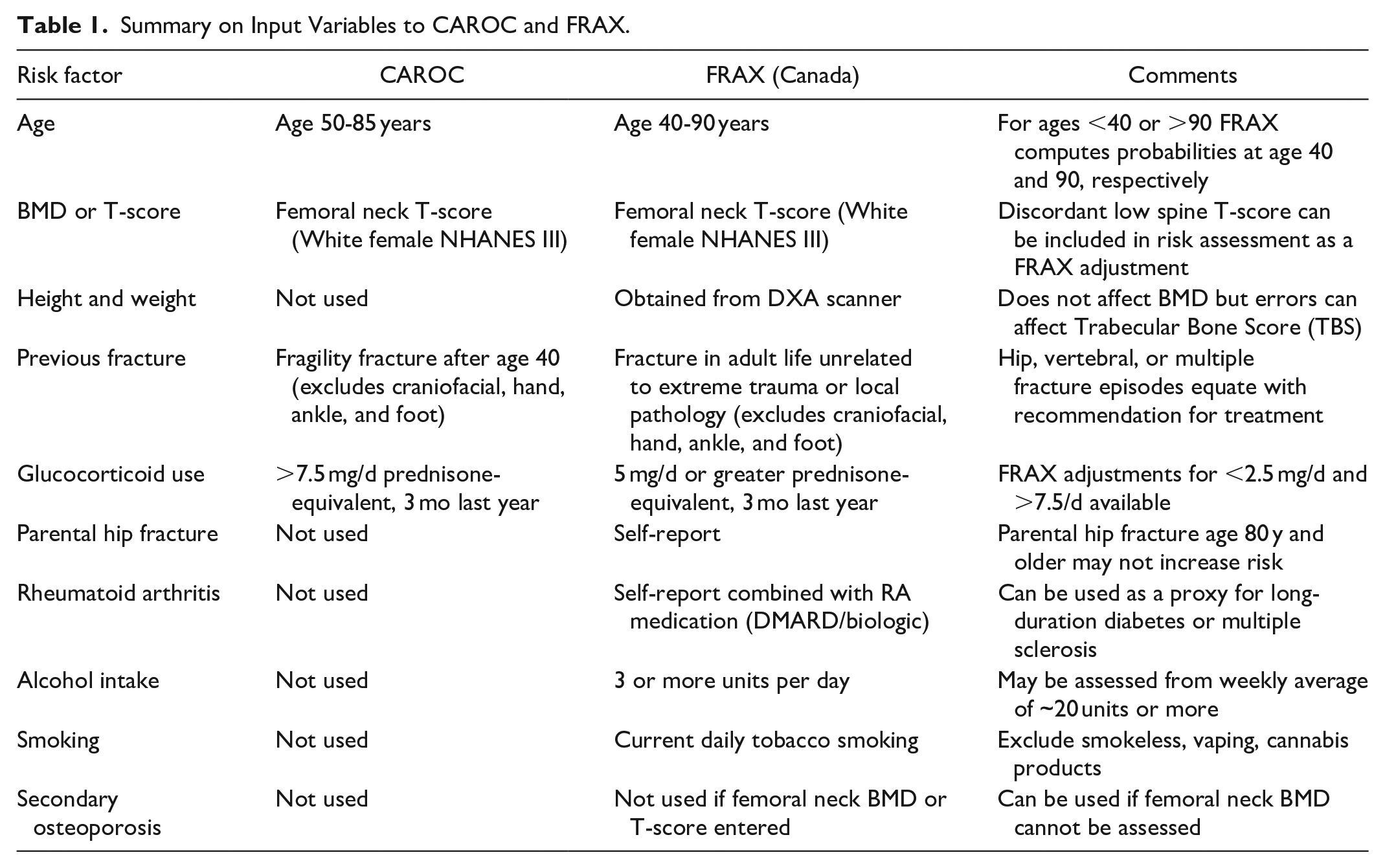
The FRAX algorithm has not changed since it was first developed in 2008. Basic inputs when performed in conjunction with DXA are age, sex, weight, height, femoral neck BMD (available through the DXA instrument), previous fracture, glucocorticoid use, parental hip fracture, rheumatoid arthritis, current smoking, and high alcohol intake (Table 1). Previous fracture and glucocorticoid use are used in CAROC and are already familiar to radiologists using this tool. Secondary osteoporosis also appears on the input page but does not affect the FRAX calculation when BMD is available since it is assumed that secondary osteoporosis mediates its affect through reduced BMD. Figure 4 shows the magnitude of the effect of the clinical risk factors on output from the Canadian FRAX tool for a hypothetical 70-year-old woman with femoral neck T-score −2.3. This shows that the largest increment in fracture risk is seen with previous fracture, glucocorticoid use, and parental hip fracture, with a smaller effect from rheumatoid arthritis, smoking, alcohol intake, and no effect from secondary osteoporosis. Based upon this, it follows that greater effort should be expended on collecting accurate information that has the greatest potential to affect the risk score. Conversely, there is little value in expending effort collecting information on secondary osteoporosis when femoral neck BMD is already available. The following sections are intended as general recommendations on how to operationalize these inputs to FRAX. Of course, this can never cover all patient circumstances and therefore clinical judgment by the reporting radiologist remains an essential component.
Summary on Input Variables to CAROC and FRAX.
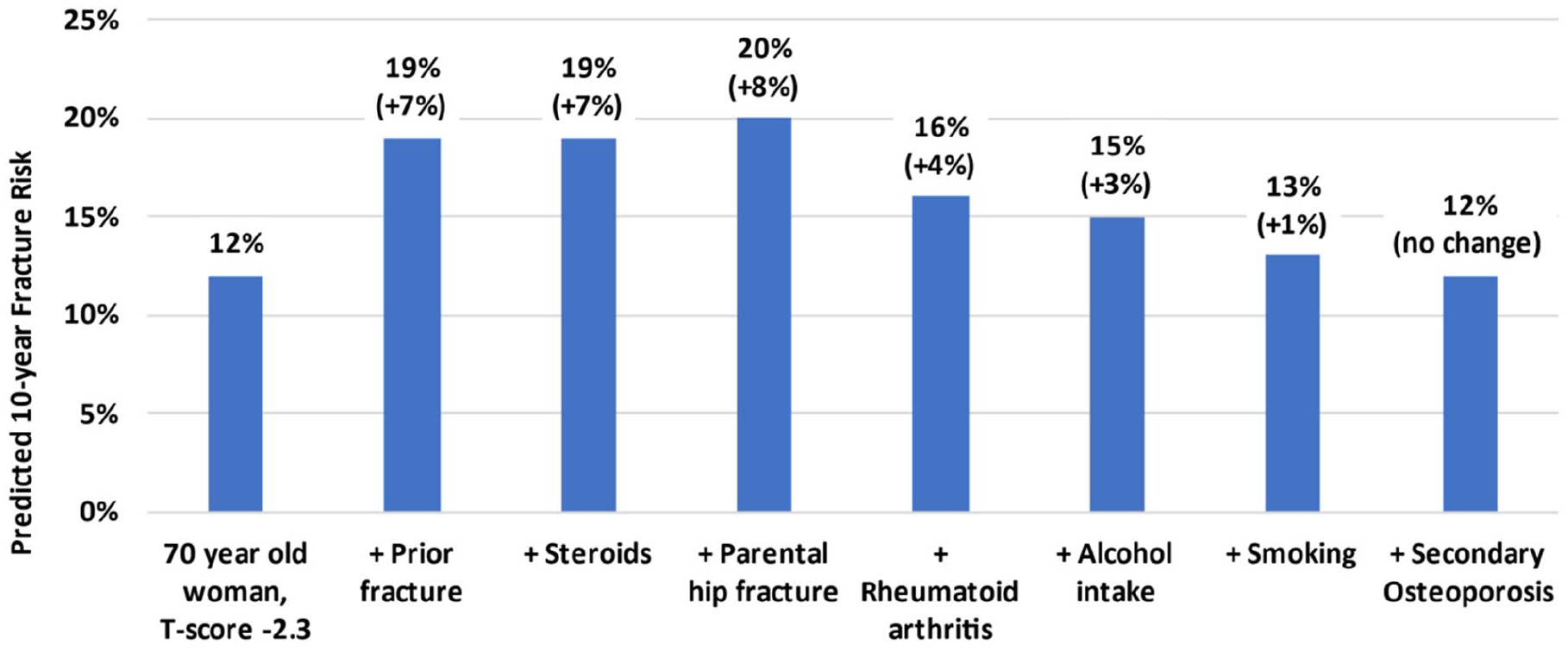
Effect of individual clinical risk factors on 10-year major osteoporotic fracture output from the Canadian FRAX tool for a hypothetical 70-year-old woman with femoral neck T-score −2.3, descending importance.
DXA-Derived Inputs
It is axiomatic that high-quality DXA is required for high-quality fracture reporting. This includes both clinical considerations as well as rigorous attention to machine quality assurance.27,28 Among the various BMD measurements sites, femoral neck BMD is the only permissible input to FRAX. 15 This was based upon evidence of a strong relationship between hip BMD and fracture risk, particularly hip fractures, and the availability of an internationally accepted reference database, the Third National Health and Nutrition Examination Survey (NHANES III).7,29,30 The FRAX tool can accept femoral neck BMD in units of grams per centimetre squared (specific to the DXA manufacturer) or as a T-score (calibrated for White female reference NHANES III data). Although other BMD measurements sites are typically used in clinical practice, these should not be used as inputs to the FRAX tool. Lumbar spine BMD may have a slightly greater ability to stratify vertebral fracture risk, but is limited in its ability to assess non-vertebral fracture risk.31,32 Moreover, change in hip BMD is a more robust indication of antifracture effect among treated individuals.33-35 Age, sex, weight, and height do not affect the actual BMD measurement but are important to enter correctly into the DXA machine along with other demographic information. These are then available for the FRAX calculation software on the DXA machine. For users of trabecular bone score (discussed below) accurate weight and height information are critically important. 36
CAROC-Derived Inputs
Previous Fracture
Confirmed fractures under CAROC are also eligible for entry into the FRAX calculation. Fractures of the head/neck, hands, and feet (including ankle) are typically excluded as these show a weak association with future fracture risk. Patellar fractures are strongly associated with fracture risk and should not be omitted.37,38 The same applies to vertebral fractures, whether clinical or non-clinical, when diagnosis is based upon well-validated radiologic features.39,40 Lateral spine vertebral fracture assessment (VFA) with DXA has proven to be a valuable tool in routine clinical practice, strongly predicting future clinical fracture events, including hip fractures, and leading to appropriate treatment initiation.41,42 Ideally, history of fracture by self-report should be confirmed radiologically, and this is often possible through picture archiving and communication systems (PACS). PACS review may even identify fractures that the patient has omitted; 20% of confirmed hip and other fractures are not reported by the patient.43-45 Self-report is relatively poor for vertebral and rib fractures, and radiologic confirmation is important, whereas self-report for fractures of the extremities, including hip, humerus, forearm, tends to be more reliable, with positive predictive value approaching 90%.45,46 The circumstances around the fracture should be clarified whenever possible, although whether a distinction between “fragility” and “non-fragility” fractures is clinically useful has recently been questioned. 47 Large cohort studies have found that low-trauma and high-trauma fractures show a similar association with low BMD and recurrent fracture.48,49 Moreover, a pooled analysis of randomized clinical trials found that osteoporosis pharmacotherapy prevented non-vertebral fractures regardless of the degree of trauma. 50 Fractures occurring from extreme violence (eg, fall from roof, polytrauma) or local pathology (eg, metastasis) should still be excluded. Although fracture risk declines over time from the initial fracture episode, it never returns to baseline and has been shown to be a risk factor for recurrent fracture beyond 20 years. 51 More recently, there is evidence that any fracture in adulthood, regardless of age, remains a risk factor for fracture in later life, even decades later. 52
Glucocorticoid Use
Prolonged high-dose glucocorticoid use is a risk factor for BMD loss and independently increases fracture risk. 53 CAROC considers glucocorticoid use in the last year exceeding 3 months of prednisone-equivalent 7.5 mg daily.11,54 Glucocorticoid use in FRAX is based upon average exposure (average prednisone-equivalent 5 mg daily), and will therefore capture the larger number of individuals receiving this lower dose. Although the FRAX website includes glucocorticoid use at any point in the individual’s lifetime, there is strong evidence that fracture risk decreases 6 to 12 months after discontinuing glucocorticoid therapy.55,56 This supports the Canadian recommendation to limit glucocorticoid use to 3 months in the last year, information that is usually readily available from patients and electronic charts.
FRAX-Specific Risk Factors
Parental Hip Fracture
From Figure 4 it can be seen that parental hip fracture has the largest impact on fracture risk, and therefore warrants careful consideration when this is collected. Parental hip fracture is derived from self-report and is usually accurate, particularly if this occurred in the preceding 10 years. It is our practice to ask about these circumstances surrounding the fracture, age of the parent when fracture occurred, whether surgery was performed and that this involved a birth parent (not adoptive parent). A fall related fracture after age 70 requiring surgery in a birth parent clearly warrants inclusion in FRAX. When the fracture occurs in younger individuals (before age 60), did not require surgery, or was related to a non-fall mechanism (such as high trauma or malignancy) then this probably does not represent a fracture that should be included in FRAX. There is evidence from one large population-based study that included long-term fracture data from 478 792 parents and 261 705 offspring that the age of parental hip fracture was a significant risk modifier, with a large effect when the parent fracture of the hip was before age 70 years, but no significant effect when this occurred after age 80 years. 57 Although this analysis has not been replicated in other cohorts, it is the practice of one of the cooperators (WDL) to exclude parental hip fracture in a parent when this occurs after age 80 years.
Rheumatoid Arthritis
Self-report of rheumatoid arthritis is often overestimated, as patients often confused this with other arthropathies (especially osteoarthritis), fibromyalgia and polymyalgia rheumatica. The FRAX website states that reliance should not be placed on a patient’s report of rheumatoid arthritis unless this can be confirmed. Clinical and laboratory data supporting the diagnosis of rheumatoid arthritis is usually not available to radiologists. However, a review of the patient’s current medications can help to resolve uncertainty. Patients with significant rheumatoid arthritis are usually taking a disease modifying antirheumatic drug (DMARD) or biologic agent. One useful strategy for patients reporting rheumatoid arthritis is to review their current medication list. Arthropathies, including rheumatoid arthritis, that do not require a DMARD/biologic agent are unlikely to independently increase fracture risk and can be excluded from FRAX.
Smoking and High Alcohol Intake
Current smoking and alcohol intake are assessed from self-report and cannot be independently verified. These have a relatively small effect on the MOF risk calculation, and show dose-response in terms of exposure. Casual smoking (not daily) should not be included in FRAX. This also applies to smokeless use of tobacco, vaping products and cannabis products. The effect of smoking likely attenuates after cessation but one study suggests that a recent ex-smoker continues to have an increase in fracture risk similar to current smokers. 58 Therefore, smoking should probably still be included in FRAX for individuals who are recent exposures. It is difficult to obtain accurate self-report on alcohol intake and this also can vary over time. Our practice is to ask for average number of alcoholic drinks per week, and include this in FRAX if the daily average equals or rounds up to 3.
FRAX Workflow Considerations
Just as it is important to have standard operating procedures for performing and reporting DXA,27,59 radiology departments should have clearly defined standard operating procedures for collecting, entering, and reviewing the clinical components of FRAX (Figure 5). The clinical risk factors are most commonly obtained from a patient questionnaire. This can be sent out to patients prior to their visit, providing an opportunity for patients to collect and summarize the necessary information. Alternatively, some practices may prefer to obtain this information from the referring physician. The FRAX-related information should be reviewed by the technologist performing the DXA testing, clarifying areas as outlined above where patient information may be incomplete or inconsistent, with final review by the reporting physician. When patients return for a follow-up examination, comparing questionnaire responses provides an additional level of quality assurance. Occasionally it may be necessary to return the scan to the technologist to update the FRAX clinical risk factors (eg, if a fracture is identified on PACS review that the patient has omitted, or a self-report fracture was not confirmed on PACS review). If desired, the DXA software can be configured to send the final FRAX report to the PACS. This will usually contain both 10-year MOF risk and 10-year hip fracture risk, though only MOF risk is used in the Osteoporosis Canada clinical practice guideline. 4 In most cases, an efficient workflow can be established that allows high quality FRAX reporting with minimal additional effort.
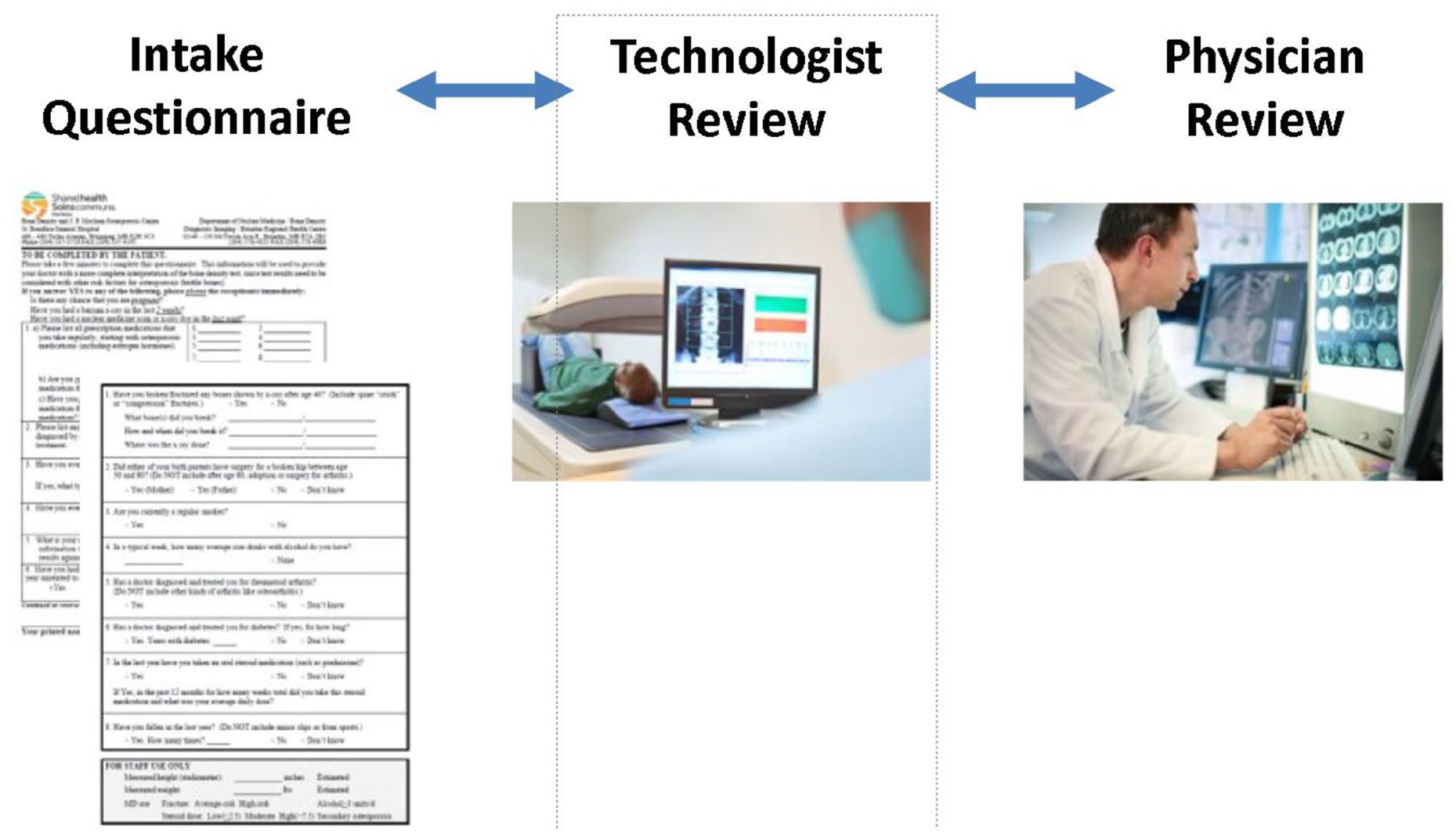
Sample workflow for FRAX reporting in a radiology environment.
Advanced FRAX
Since the release of FRAX, additional risk factors and quantitative adjustments have been developed that can enhance the performance of FRAX in specific circumstances, as described below. These adjustments are not directly integrated into the FRAX algorithm, which is less convenient for the reporting radiologist. However, even if not routinely used, it is worth being familiar with the existence of these as options in fracture risk reporting. The calculations are available through the FRAXplus website (https://www.fraxplus.org/). These includes recency of fracture (since a fracture in the last 2 years is associated with greater risk than a remote fracture in many but not all studies) 60 ; dose of oral glucocorticoid (with average daily prednisone-equivalent doses exceeding 7.5 mg daily or less than 2.5 mg having greater or lesser effect, respectively) 61 ; type 2 diabetes mellitus (especially when this is of long-duration) 62 ; lumbar spine BMD (especially when the spine T-score is significantly lower than the femoral neck T-score) 63 ; trabecular bone score (TBS, a grayscale texture measurement derived from the lumbar spine DXA image that increases fracture risk independent of BMD) 64 ; history of falls in the preceding year (especially if these are multiple fall episodes) 65 ; and hip axis length derived from the hip DXA image (greater values associated with significantly greater hip fracture risk independent of BMD). 66 Longstanding diabetes increases MOF risk and has an effect similar to the effect of rheumatoid arthritis. 67 Therefore, a simple adjustment is to use the rheumatoid arthritis input in FRAX as a proxy for the effect of long-duration diabetes. 62 The same approach has been shown to work with multiple sclerosis. 68
The initial FRAX algorithm was developed from approximately 60 000 individuals in 9 prospective cohorts, including the Canadian Multicentre Osteoporosis Study (CaMos). 14 The fracture team is currently working to update the FRAX algorithm using a much larger data set that comprises more than 2.1 million individuals from 64 international cohorts (2 from Canada), comprising over 20 million person-years of follow-up and more than 110 000 MOF events. 69 Those analyses are currently being conducted by the FRAX team with the original fracture risk factors. Additional analyses will explore whether including history of falls and/or diabetes improves the performance of FRAX. The release date for the new version FRAX is currently unknown.
Frequently Asked Questions (FAQs)
When Is It Necessary to Use Secondary Osteoporosis?
As noted above, when femoral neck BMD is included in the FRAX calculation then secondary osteoporosis has no effect. Including secondary osteoporosis in the FRAX calculation increases risk when femoral neck BMD is not included, and has the same weight as rheumatoid arthritis (only 1 of which can be active at the time). Computing FRAX without BMD has been suggested for population-based screening to identify individuals who warranted referral for DXA testing. 70 Indeed, FRAX without BMD performs almost as well as FRAX with BMD for fracture risk prediction.71-73 The former is not relevant in the context of radiologists reporting results in individuals who have undergone BMD testing. If femoral neck BMD cannot be assessed (such as bilateral hip arthroplasties) then the radiologist has the option of reporting FRAX-based risk without BMD (in which case secondary osteoporosis should be included in the calculation if available to avoid underestimating risk) or simply stating that FRAX cannot be reported.
How Does Ethnicity and Immigration Affect FRAX?
The Canadian population and indeed all populations are ethnically diverse and this raises the question of how this is accommodated in fracture risk assessment. Most countries have a single FRAX calculated that is applied to all individuals. A small number of countries have ethnicity/race specific FRAX calculators, including the United States, though the use of race/ethnicity in clinical algorithms has become controversial. 74 Recently, the American Society for Bone and Mineral Research (ASBMR) has suggested that ethnicity/race-specific FRAX calculated to the United States be replaced with a single population-based FRAX calculator. 75 Even where there are known differences in fracture rates between ethnic/race subgroups, these may not be homogeneous and may not reflect the effects of admixture, migration, or consequences of fracture. These complex clinical considerations are currently beyond the scope of what can be achieved with FRAX. The Osteoporosis Canada guideline assumes fracture risk derived from the Canadian risk calculators, FRAX or CAROC. The thresholds used may not be applicable to other FRAX tools, especially for countries where fracture risk is higher or lower than in Canada.76,77 Therefore, when applying the Osteoporosis Canada guideline only the Canadian tools should be used.
How Does Anti-Osteoporosis Treatment Affect FRAX?
Although anti-osteoporosis treatment typically produces an increase in femoral neck BMD, which would reduce the calculated FRAX score, treatment has no effect on other risk factors, most notably age. The age effect in FRAX score can lead to an increase in fracture risk even when femoral neck BMD improves. 78 Therefore, a decrease in FRAX score on effective treatment is not expected and a change in FRAX score has not proven to be useful for documenting anti-fracture effect of treatment. The 2010 Osteoporosis Canada guideline noted that FRAX and CAROC “reflects the theoretical risk for a hypothetical patient who is treatment-naive; it cannot be used to determine risk reduction associated with therapy.” 20 However, FRAX still provides useful risk stratification in individuals who are currently on or have recently received anti-osteoporosis treatment,79,80 and can therefore still inform clinical management regarding need for ongoing or intensified treatment.
Conclusions
The 2023 Osteoporosis Canada clinical practice guideline update provides a new framework for treatment initiation. Fracture risk assessment is integral to this procedure, with FRAX now being the preferred risk prediction tool. Radiologists involved in DXA reporting should be aware of these changes and best practices in terms of fracture risk reporting.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
