Abstract
This is a visual representation of the abstract.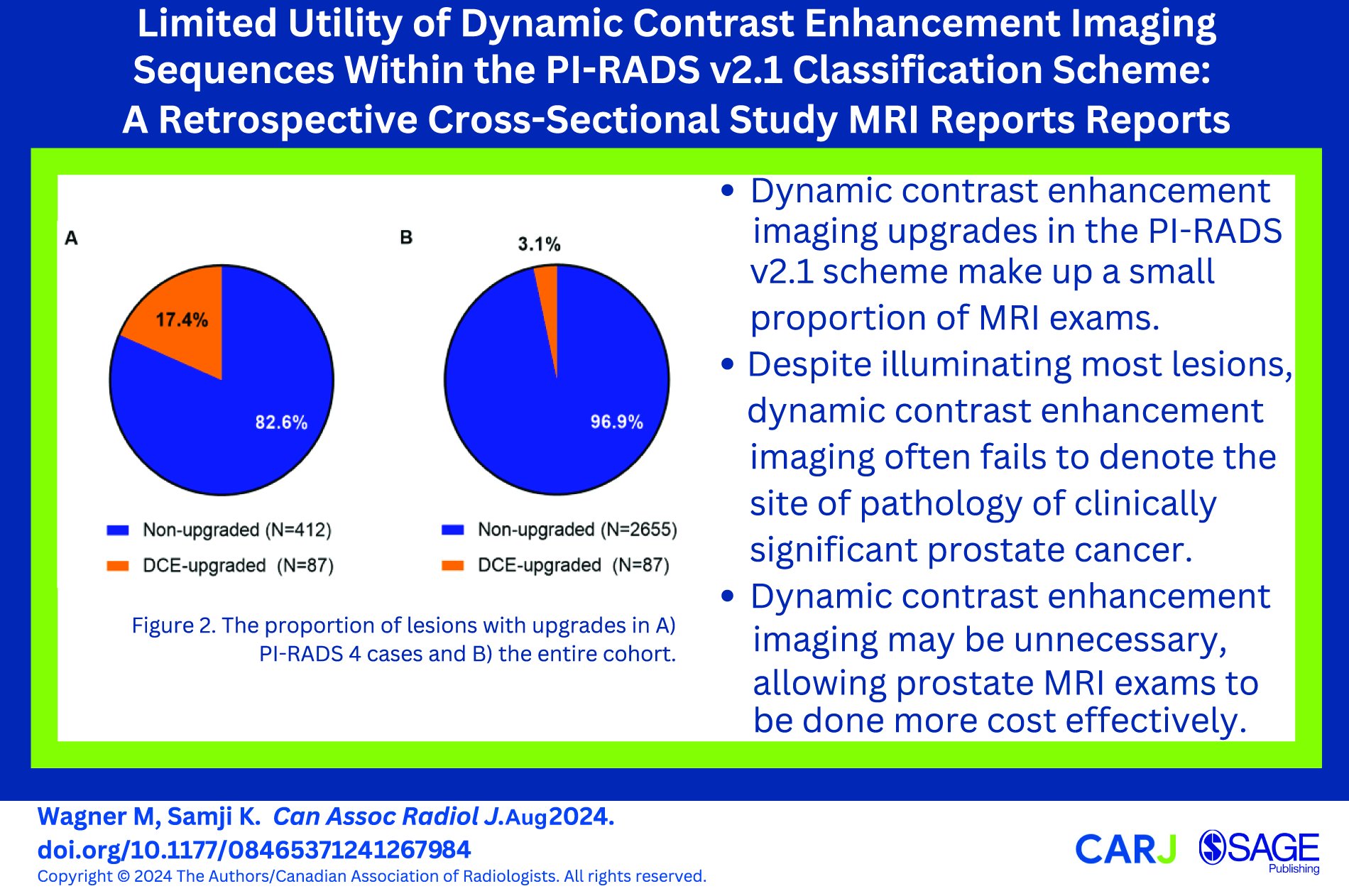
Introduction
According to the most recent Canadian Cancer Registry statistics (2022), prostate cancer is the most prevalent cancer type in males, and the second most prevalent cancer type in all of Canada, according to 25-year prevalence data. 1 Given prostate cancer’s significant imposition of morbidity and mortality amongst males, great focus has been placed on identifying robust diagnostic methods for early detection of prostate cancer. Serum prostate specific antigen (PSA) is highly sensitive, but not specific, to prostate cancer. Use of the PSA to screen for prostate cancer has been known to cause an overdiagnosis of clinically insignificant prostate cancers, leading to unnecessary biopsies and treatment. 2 Integrating prostate volume with PSA to yield the prostate density improves the diagnostic value compared to PSA alone, though is still limited: either method is unable to direct clinicians to potentially significant lesions within the prostate to biopsy. It has been shown that by using MRI to locate lesions within the prostate to biopsy, unnecessary biopsy of clinically insignificant cancers was decreased compared to standard biopsy.2,3 For this reason, the use of prostate MRI in the screening algorithm for prostate cancer has garnered much interest in recent years.
As a non-invasive screening modality, MRI can not only localize lesions able to be biopsied, but also provide lesion characteristics which correlate to a lesion’s potential malignancy. To report this data in a structured and reproducible manner, the Prostate Imaging Reporting and Data System (PI-RADS) scheme was developed, with version 2.1 being the latest (depicted in Figure 1). The PI-RADS scheme summarizes T2-weighted (T2W) and diffusion weighted imaging (DWI) sequences into a concise score of 1 to 5 denoting the lesion’s likelihood of being a clinically significant prostate cancer (csPCa).4,5 The overall PI-RADS score is composed of individual scores of 1 to 5 for each of the T2W and DWI sequences, whereby dynamic contrast enhancement (DCE) positivity can “upgrade” a DWI score of 3 to 4 for a peripheral zone lesion.4,5 These 3 imaging sequences comprise multiparametric MRI (mpMRI) and are standard for a typical prostate cancer screening exam.
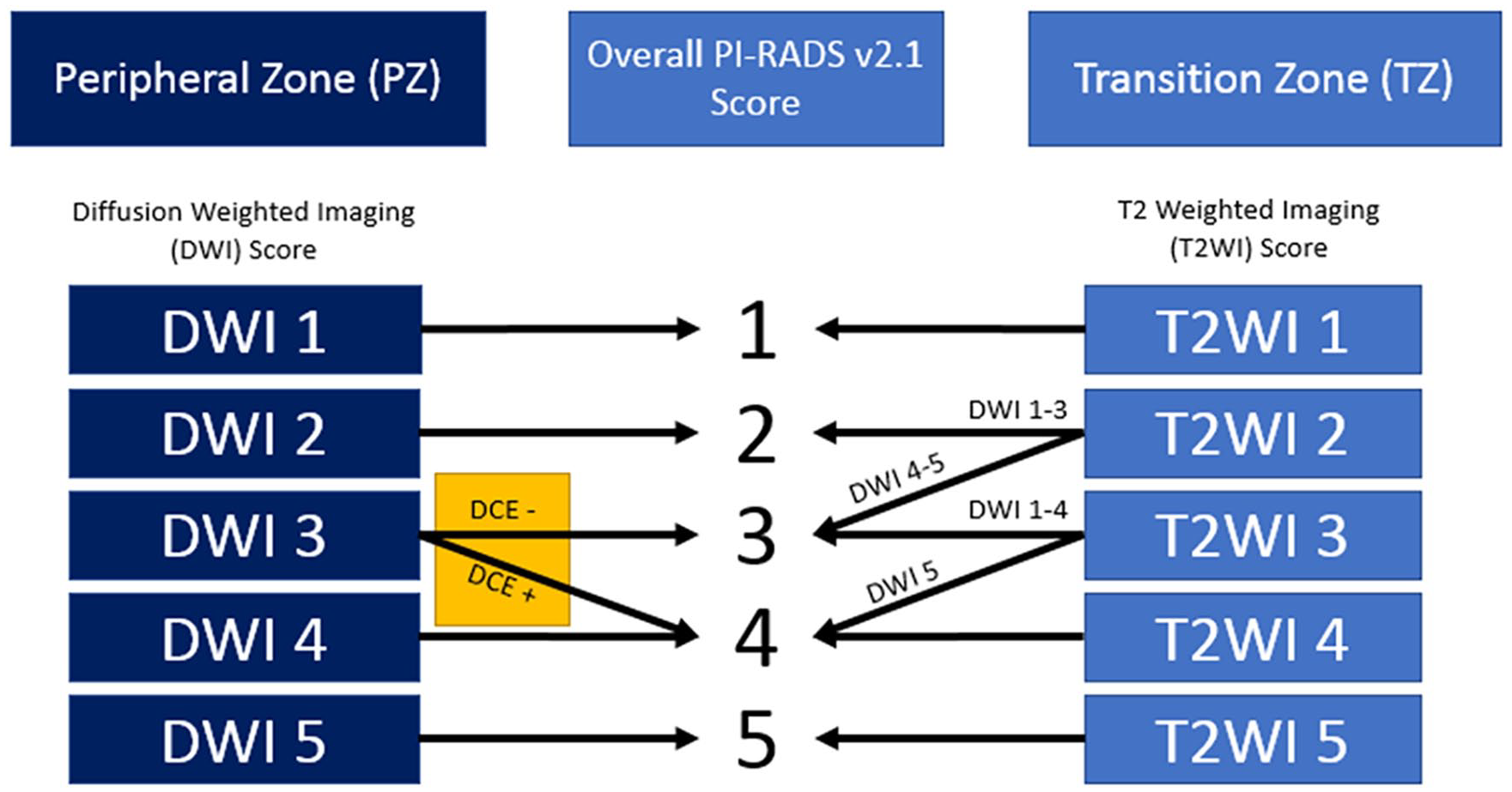
Schematic representation of the PI-RADS v2.1 categorization system for prostate lesions.
The use of DCE imaging in multi-parametric MRI screening for prostate cancer has been criticized on the premise that DCE acts to further categorize what is thought to be a very small subset of patients evaluated with mpMRI. Considering that in published cohorts 9% to 46% of patients undergoing prostate MRI have a PI-RADS 3 lesion, this already represents limited applicability of DCE imaging to all patients undergoing prostate MRI. 6 Conducting the DCE imaging sequence lengthens exam times by almost 3 times that of a protocol without DCE, decreasing the cost effectiveness of mpMRI as a prostate cancer screening modality.7,8 Furthermore, gadolinium contrast agents pose a risk of adverse drug reactions, and have been reported to accumulate in the tissues, representing a potential risk of toxicity.9,10 Administration of gadolinium contrast for DCE imaging is also expensive and liable to supply-chain failure, as demonstrated by the recent worldwide shortage of iodine-based contrast. Finally, it should be noted that prostate biopsy carries risk of potentially severe sepsis and other infectious complications for which unfounded biopsy of lesions via DCE upgrades may exacerbate. 11 For these reasons, omission of DCE from the mpMRI imaging protocol, referred to as bi-parametric MRI (bpMRI), has been explored with varying conclusions regarding its diagnostic efficacy. 12
Though DCE imaging has been theorized to benefit only a small subset of patients, few studies have endeavoured to quantify the proportion of patients which would actually benefit from DCE imaging. Furthermore, quantifying the typical proportions of PI-RADS scores observed in patients screened with prostate MRI could inform future screening strategies using the classification scheme. Thus, the objective of this study was to quantify this proportion of patients with each PI-RADS score, as well as the proportion of patients whose PI-RADS scores were upgraded by DCE imaging amongst a large cohort of patients undergoing prostate MRI. Given DCE imaging’s utility in a small part of the PI-RADS grading scheme, we hypothesize that upgrades necessitated by DCE imaging will be infrequent and insufficient to justify its routine inclusion in prostate MRI protocols.
Methods
Following institutional ethics approval, a retrospective analysis of radiology reports attached to prostate MRI cases performed at 2 large teaching hospitals was conducted. All MRIs were performed on 3T MRI scanners and utilized multiparametric MRI consisting of T2WI, DWI, and DCE imaging protocols as per requirements for PI-RADS scoring. Prostate MRI cases conducted between January 1st 2017 and January 1st 2022 were included in this analysis. Any exams which were non-diagnostic, incomplete, or were missing overall PI-RADS score or lesion DCE status (for example, in the case of a known gadolinium contrast allergy), were excluded. Patient information, such as patient age at time of scanning, previous prostate biopsies, and most recent serum PSA were gathered from radiology reports. Prostate characteristics such as prostate volume (PV), were also gathered, and if a lesion was present, lesion location, individual PI-RADS T2 and DWI scores, and DCE positivity. PSA density was calculated using the most recent serum PSA and the calculated PV from MRI. The overall PI-RADS score for the patient was determined as the greatest PI-RADS score given to a focal or diffuse lesion, if any. Cases prior to the institution’s adoption of PI-RADS v2.1 (which were scored using PI-RADS v2.0) were re-scored via the PI-RADS v2.1 specifications.
To analyze the pathology of upgraded lesions, a retrospective review of prostate biopsy results within 12 months after prostate MRI in patients with upgraded lesions was conducted. A grade group ≥2 or Gleason score ≥7 at the site of the upgraded lesion was considered clinically significant.
Results
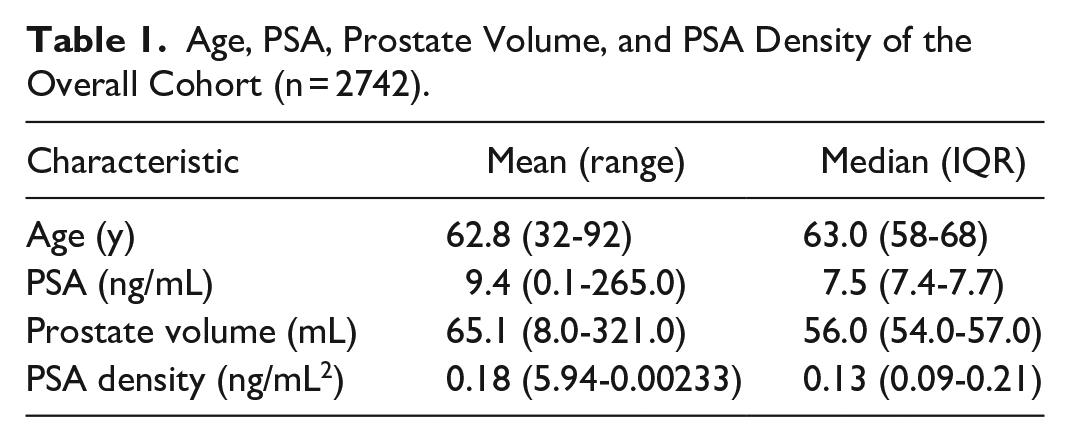
Between January 1st 2017 and January 1st 2022, a total of 2796 prostate MRI cases were available for analysis. Upon analysis, 54 of these cases were found to be non-diagnostic, or missing key data to allow assessment of PI-RADS scores (as noted above), and were excluded. This left 2742 cases to analyze. In the overall cohort, the average and median age was 62.8 and 63.0 years respectively. Mean prostate specific antigen (PSA) was 9.4 ng/mL, with a median of 7.5 ng/mL. Prostate volume had a mean of 65.1 mL with a median of 56.0 mL. Calculating each patient’s PSA density, the average was 0.18 ng/mL2, with a median value of 0.13 ng/mL2. These values are summarized in Table 1 with range and interquartile ranges (IQR).
Age, PSA, Prostate Volume, and PSA Density of the Overall Cohort (n = 2742).
Separating the cases by PI-RADS score, we found that PI-RADS 2 cases made up most of the cohort (45.8%). After PI-RADS 2, PI-RADS 5 (19.0%), PI-RADS 4 (18.2%), PI-RADS 1 (12.6%), and PI-RADS 3 (4.4%) followed in terms of frequency in the cohort, from most frequent to least frequent. The majority of examinations with focal suspicious lesions were categorized as PI-RADS 4, at 617 lesions. We observed that age, median PSA, and PSA density seemed to increase alongside PI-RADS score. We also found that median PV decreased alongside PI-RADS score (Tables 2).
Cohort Information Organized by Overall PI-RADS Score.
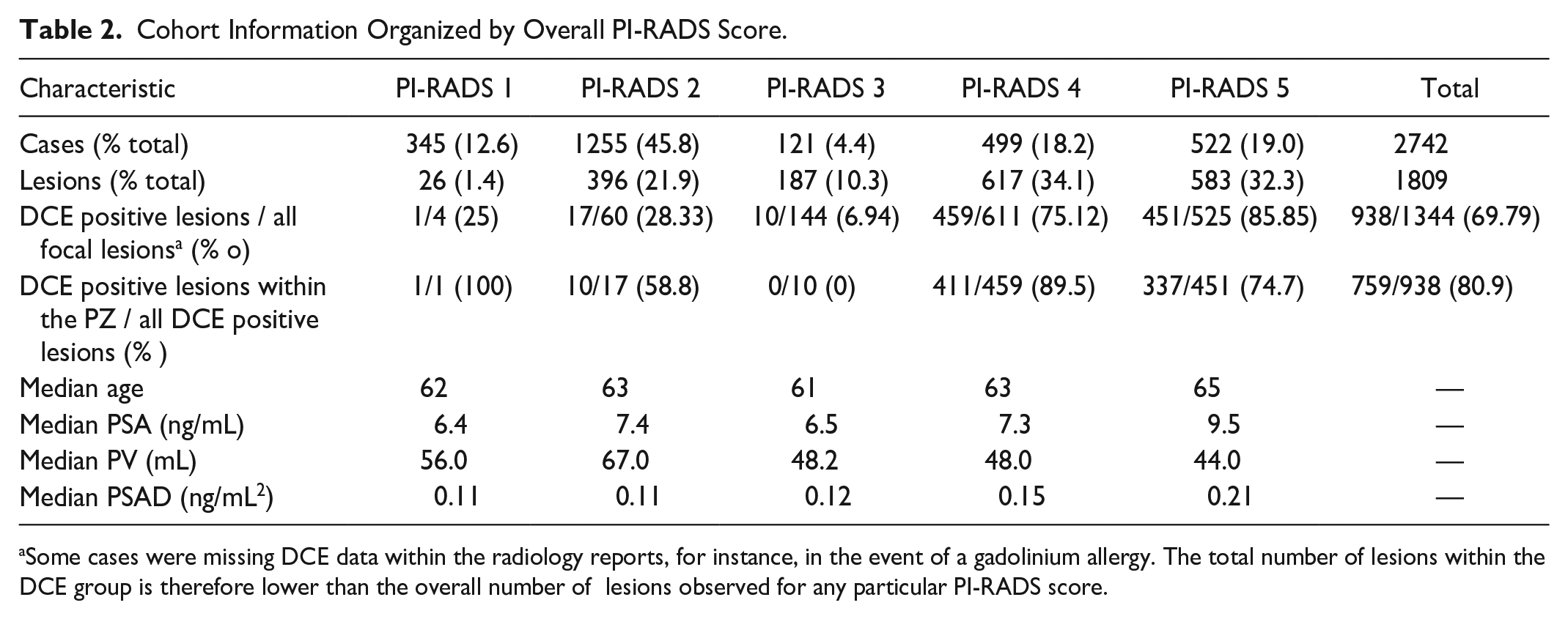
Some cases were missing DCE data within the radiology reports, for instance, in the event of a gadolinium allergy. The total number of lesions within the DCE group is therefore lower than the overall number of lesions observed for any particular PI-RADS score.
DCE positivity was found in all PI-RADS groups, with the greatest proportion of DCE+ lesions unsurprisingly in the PI-RADS 5 group (86%), followed by the PI-RADS 4 group (75%). (Table 2).
We also specifically analyzed patients with lesions which were upgraded due to DCE imaging. These upgrades were composed of lesions within the periperhal zone with a DWI score of 3, demonstrating DCE positivity, leading to an overall PI-RADS score of 4. In our cohort, this comprised 87 patients (7.4% (87/499) of PI-RADS 4 cases, and 3.2% (87/2742) of all cases within our cohort (Figure 2)).
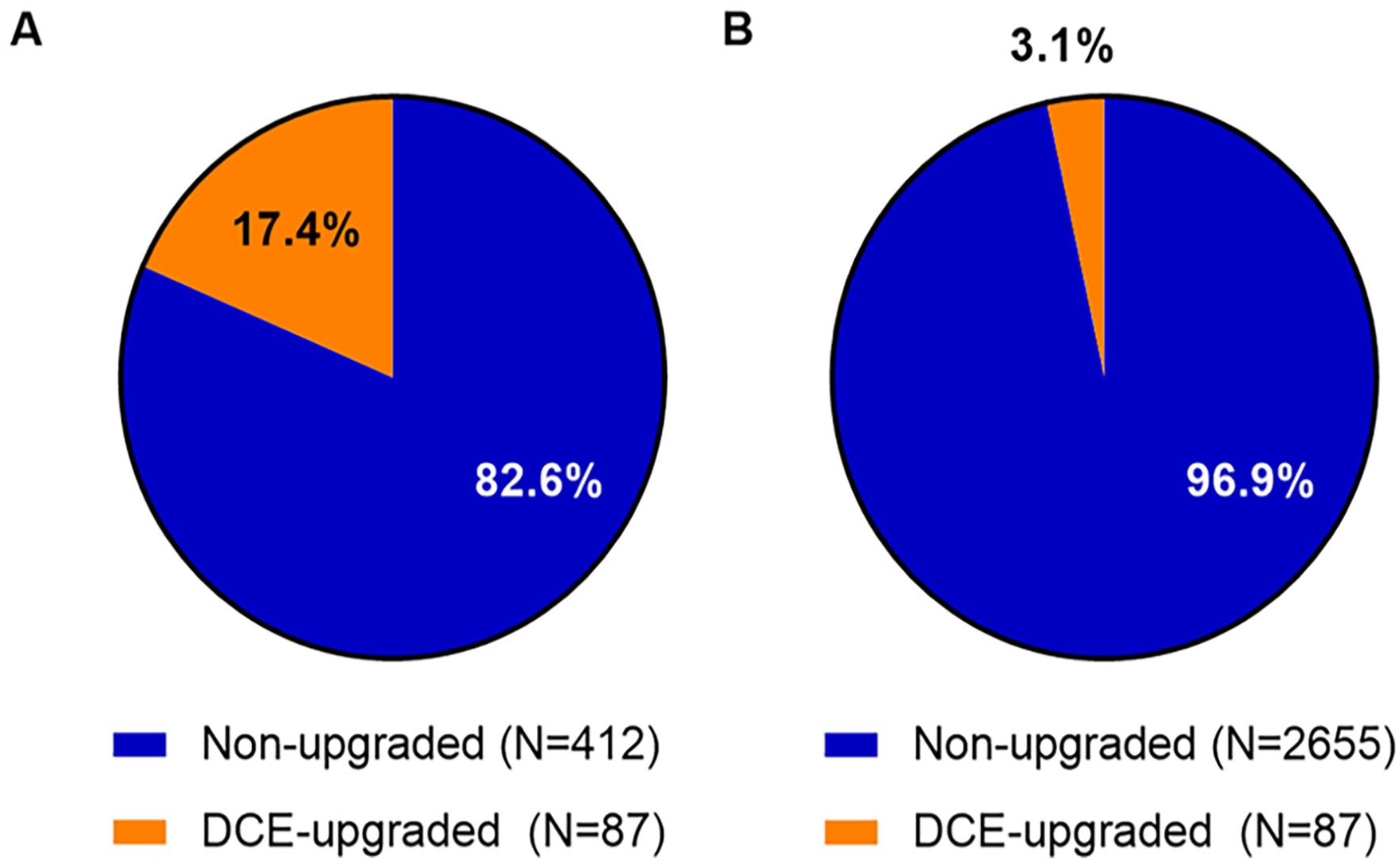
The proportion of lesions with upgrades in (A) PI-RADS 4 cases and (B) the entire cohort.
Of the 87 patients with upgraded lesions by DCE, 65 had a biopsy of the upgraded lesion within the year following their exam. Of these 65, n = 18 (20.7% of upgrades and 27.7 of upgrades with pathology) had csPCa as determined by a gleason score ≥7 or grade group ≥2 at the site of the upgraded lesion, with 47 (54.0% of upgrades and 72.3% of upgrades with pathology) being non-significant (Table 3 and Figure 3). This would suggest that in our total cohort, DCE only had an impact in patient management in 18/2742 (0.66%) of patients. Since gadolinium-based contrast was administered in all patients in the study, this suggests that 152.3 (2742 cases/18 csPCA detected) gadolinium administrations were required for the detection of one csPCA via DCE imaging. For every one csPCA detected via DCE upgrade, 2.6 benign biopsies were conducted.
Pathology Reports of N = 65 Upgraded Patients With Available Pathology Reports Within 12 Months Following Prostate MRI.
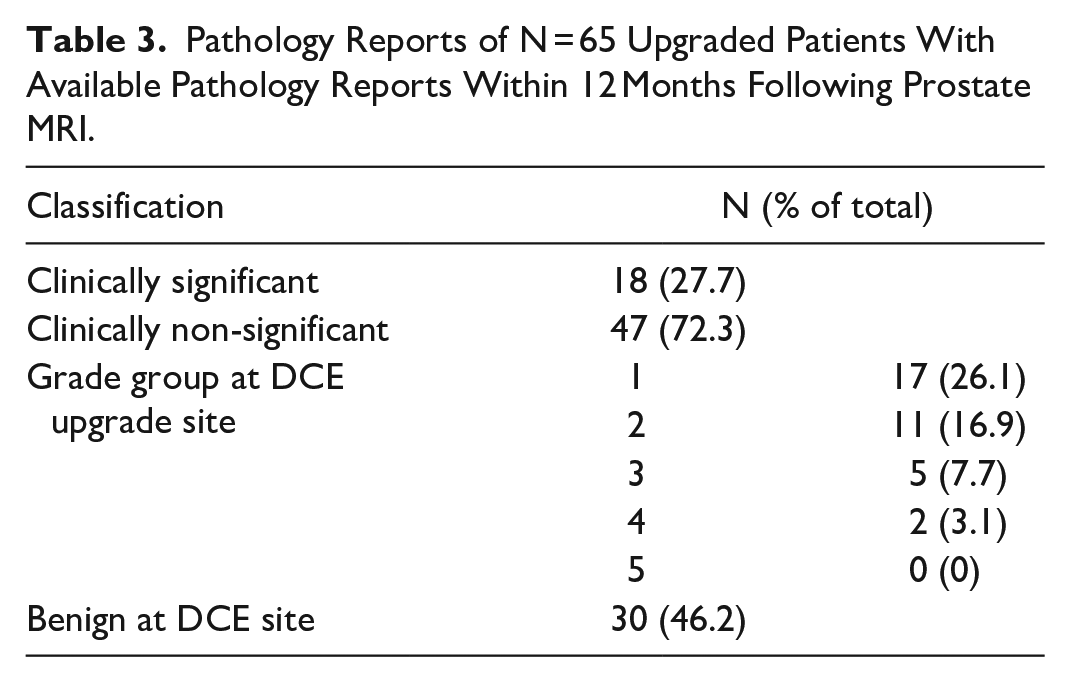

Breakdown of pathology reports for upgraded patients. Note that csPCa is based on the lesion grade group ≥2 or gleason score ≥7 at the site of the upgraded lesion on MRI.
Discussion
By reviewing the PI-RADS v2.1 classification scheme for prostate MRI, one may presume that DCE imaging plays a small role in the classification scheme. Though it further classifies peripheral zone lesions that have an equivocal probability of csPCa, this is typically a very small proportion of all patients undergoing prostate MRI. While this preposition is generally accepted, few studies have quantified in a large cohort the proportion of patients where DCE may be of benefit, as was the objective of our study.
We demonstrate that 17.4% of patients with an overall PI-RADS 4 score are due to DCE imaging upgrades, which makes up 3.2% of the entire study cohort. This is similar to another study by Ullrich et al which reported that DCE positivity within the peripheral zone which upgraded patients from an overall PI-RADS 3 to PI-RADS 4 constituted 20% of PI-RADS 4 patients in their cohort (38/193). 13 They also report similar age, PSA, prostate volume, and PSA density for their PI-RADS 4 cohort to ours. 13 In another study by Roh et al, similar cohort characteristics were shown in terms of median PSA, prostate volume, and PSA density (PSAD) and it was stated that DCE was in many cases “unnecessary”: DCE upgraded 26 of 69 (38%) of equivocal PI-RADS 3 lesions to PI-RADS 4, a particularly small proportion of the peripheral zone lesions in their cohort (8%). 14 This is similar to our study: there were a total of 93 upgraded lesions out of a possible 280 lesions (187 PI-RADS 3 lesions remaining plus 93 upgrades) for a percentage of 33% (8.4% in our cohort). In the study by Roh et al, they note that 16% of the entire cohort could potentially utilize the DCE imaging sequence to score prostate lesions using the PI-RADS v2 protocol. 14 To compare with our study, we noted that there were 87 patients (PI-RADS 4) who were upgraded by DCE imaging, and a total of 121 patients who were left with equivocal scores (PI-RADS 3), which constitutes 208 patients (7.5% of our cohort) who could potentially utilize DCE imaging for further classification. This variability amongst studies regarding how many patients can potentially utilize DCE for further classification may be reflective of more or less liberal use of prostate MRI at different centres. Despite this, studies reporting on the proportion of upgrades within a cohort agree that the number who utilize DCE for further classification is low, and DCE administration in a majority of exams seems unjustified.13,14
If not as a tool to help further classify lesions in the PI-RADS v2.1 scoring protocol, the value of DCE in prostate MRI may instead be in helping illuminate lesions for detection by the radiologist. In our study, the majority of lesions, particularly in the PI-RADS 4 and PI-RADS 5 categories, were DCE positive (Table 2). However, a meta-analysis by Tan et al on the effect of DCE-MRI either alone or in combination collected 24 articles, and concluded that combination of DCE-MRI, DWI, and T2-weighted imaging together did not significantly improve the AUC for detection of PCa in comparison with DWI alone. 15 Therefore, despite our results which show that whilst many lesions regardless of PI-RADS score will enhance via DCE, the assertion that the use of DCE in all exams has utility in aiding in lesion detection for csPCa may not necessarily be correct. This is reinforced by our data. We show in the current study that pathology reports done within 1 year of the MRI detecting the enhancing lesion more often do not reveal pathology at that site, suggesting a low specificity of DCE for enhancing clinically significant lesions in the specific case of PI-RADS 3 lesions in the peripheral zone.
In the current study we were unable to assess the proportion of the entire cohort who developed csPCa, due to the large number of patients analyzed by our study. However, a study by Messina et al found that the prevalence of csPCa in upgraded patients was 14.5% compared to 53.3% in the traditional PI-RADS 4 category, 16 which further suggests a low yield of upgrades from the perspective of csPCa detection. We were able to assess biopsy records within 1 year of upgraded patients, and found similarly that only 17.2% of upgraded patients had a csPCa. Though, this comparison is limited as our analysis considered whether csPCa was present at the site of upgrade rather than present at all in the upgraded individual. Therefore, we would expect that the prevalence of PCa in our study’s upgraded individuals to be higher than this, especially when taking into account that 25.3% of the patients did not have pathology reports available for analysis. We did not analyze PI-RADS 4 patients to compare the rate of csPCA detection via DCE positivity as our study objective was to investigate upgrades only.
In terms of the whole cohort, we found that out of 2742 examinations administering gadolinium contrast, csPCA was detected in 18 of those upgraded, and the remaining 47 upgraded lesions were benign. This suggests that for every csPCA being biopsied due to DCE upgrades, roughly 2.6 unnecessary biopsies occur. On top of unpleasant complications such as haematuria and rectal bleeding, there are reports of urinary tract infections in 2% to 6% and severe post-biopsy sepsis manifesting in anywhere from 0.1% to 2.2% of performed transrectal biopsies. 11 When considering that over 2 million transrectal prostate biopsies are performed each year in North America and Europe, 17 the decreased efficiency posed by an exam period with triple the duration when utilizing DCE, and the propensity for severe complications to occur as a result of unnecessary biopsy due to lesion upgrades, are important to consider. Furthermore, we calculated that each csPCA detected would take approximately 152.3 gadolinium administrations, for which contrast, at an average per examination cost to the hospital of $43.50 (CAD) per patient, comes at a total cost of $6625 per csPCA detected via DCE upgrades. Within the study cohort, there were 2724 patients which did not make direct use of contrast from DCE imaging at an approximate total cost for contrast of $118494. These numbers describe the cost of contrast alone, and do not factor in complications directly due to contrast administration, costs of resulting biopsies and their associated complications, and the opportunity costs from lengthened exam times.
Our study is subject to limitations. This is not a true screening study, as patients who are referred for prostate MRI at our centre are due to elevated PSA, abnormal DRE, family history, or increasing PSA despite negative biopsy, local staging, or at the discretion of a urologist. This may limit the extrapolation of these results for screening populations, however the goal of our study was to investigate how often at our centre DCE contributes to the PI-RADS v2.1 classification paradigm in those with suspected PCa. Due to the anonymization of patients during data collection, repeat examinations for the same patient across the study period could have been included as unique records within the study dataset. This may skew the results, 18 however the number of patients with repeat MRIs within the study period is judged to be small. Finally, we acknowledge that our cohort featured a relatively low proportion of PI-RADS 3 patients (4.4%) compared to other cohorts (9%-46%). 6 The proportion of cases receiving an equivocal PI-RADS score should be considered when extrapolating our study results.
Conclusion
In the current study, we aimed to quantify the degree of patients whose PI-RADS classification would change due to the use of DCE imaging. We found that a very low proportion of a large cohort was impacted by DCE imaging upgrades. Further, upon review of prostate biopsy records of those patients with upgraded lesions via DCE, we found that DCE positivity within an area of the prostate seldom predicted the discovery of a clinically significant prostate cancer. These results suggest that despite being applied to each prostate MRI exam, the overall impact of DCE imaging within the PI-RADS v2.1 classification scheme is low. Exclusion of DCE imaging from the PI-RADS v2.1 protocol may be warranted to boost cost effectiveness of prostate MRI with minimal impacts on patient care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was not supported directly by any funding source. Author MJW received a research studentship from the University of Alberta Radiology Endowed Fund.
