Abstract
This is a visual representation of the abstract.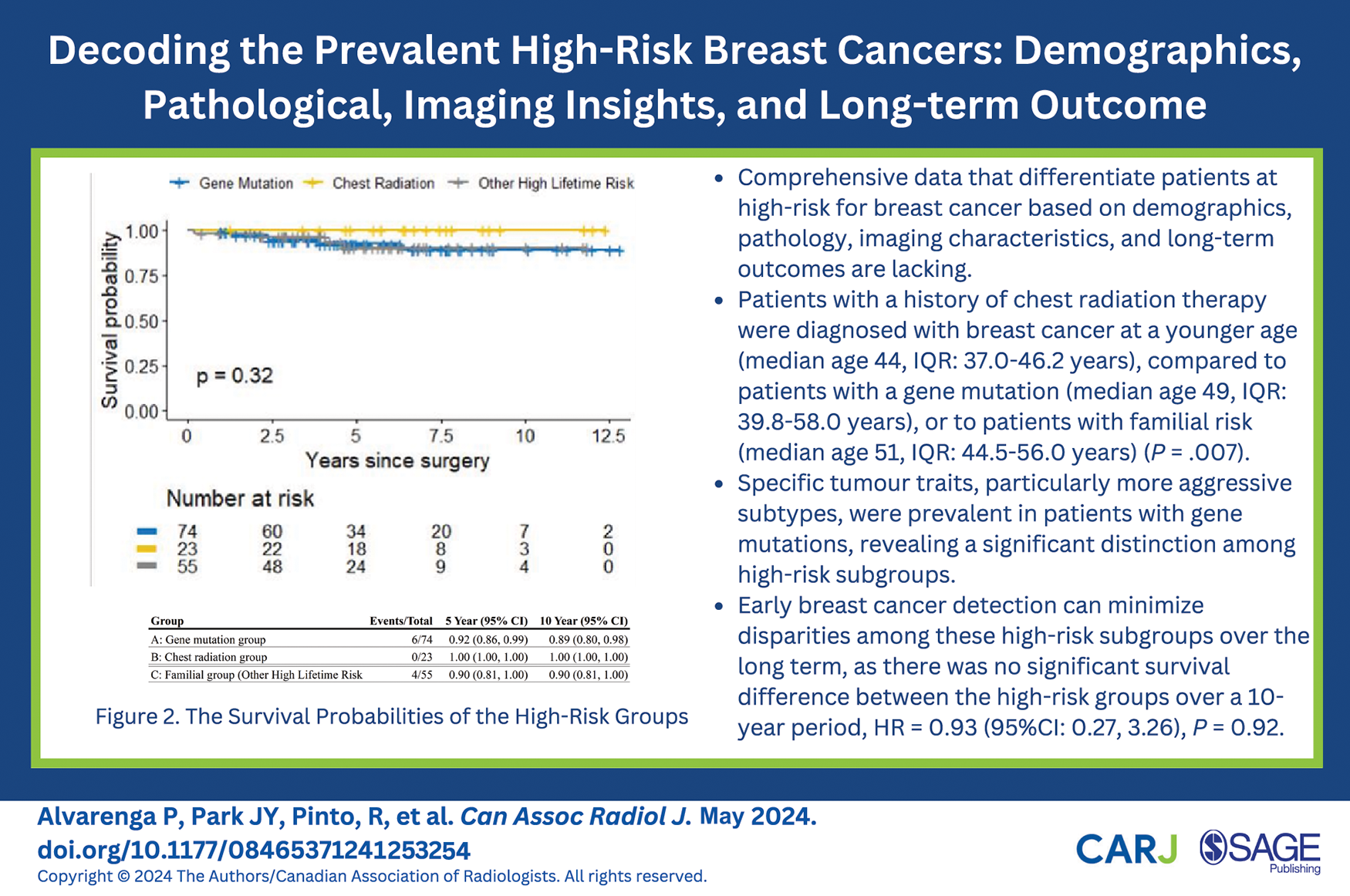
Introduction
Breast cancer (BC) constitutes the most prevalent cancer in women. 1 While the lifetime risk for an average woman to develop BC is about 13%, 1 many factors elevate this risk. Five primary higher-than-average risk groups have been identified by the American College of Radiology (ACR) 2 and a systematic review of global breast cancer screening guidelines. 3 This includes: (1) a personal history of pre-cancerous lesions or BC, (2) a family history of BC, (3) genetic predisposition, (4) prior mantle or chest radiation therapy, and (5) having dense breasts. Notably, women with a genetic risk, those whose lifetime risk exceeds 20%, and those who received chest radiation at a young age are advised by most guidelines to undergo annual mammograms and magnetic resonance imaging (MRI) screenings starting at an earlier age than women at average risk.2,3
Evidence indicates that patients at high risk, BC exhibits distinct characteristics in screening compared to those at average risk,4,8 yet the findings are not consistent. While some research shows similarities in imaging features between cancers associated with BRCA 1/2 mutations and those without,5,6,7 other studies point to unique aspects in those with BRCA 1/2 mutations, such as well-defined margins in mammography and MRI. 4 Specifically, cancers in patients harbouring BRCA-1 mutations often present with features that appear benign in imaging, 8 whereas BRCA-2 patients are more commonly associated with mammographic microcalcifications. 1 However, there’s a gap in research concerning the imaging characteristics of BC following chest radiation therapy, 9 and limited studies have directly compared imaging traits between cancers with BRCA 1/2 mutations and other high-risk categories.10,11 This study aims to investigate the features and outcomes of BC in the most prevalent high-risk subgroups.
Methods
Study Population
This observational cross-sectional study, conducted with the approval of the Research Ethics Board (REB#21-5999), analyzed data from the imaging department of a tertiary-care academic center affiliated with the University of Toronto, from January 2010 to December 2019. The requirement for obtaining patient consent was waived for this study.
The focus was consecutive high-risk patients with imaging studies presented in the radiology department with a confirmed BC diagnosis between 2010 and 2019 regardless of the availability of concurrent mammograms and MRI scans, the nature of pathology findings, and the duration of follow-up. High-risk patients were defined as 3 subgroups based on the Ontario Breast Screening Program (OBSP) high-risk eligibility criteria 12 : those with genetic mutations, patients with previous chest radiation, and familial risk groups. The familial risk was assessed using tools like the International Breast Cancer Intervention Study (Tyrer-Cuzick model) 13 and the Breast and Ovarian Analysis of Disease Incidence and Carrier Estimation Algorithm (BOADICEA) model, 14 with a required estimated lifetime risk of 25% or higher for inclusion. It is noted that the OBSP program does not include the Claus 15 and the BRCA mutation carrier prediction (BRCAPRO) models 16 for risk assessment.
Data Extraction
Patient data encompassing demographics, clinical history, imaging, and pathology reports from percutaneous biopsy and surgical procedures were extracted from the electronic patient record. Data collection concluded on March 21, 2023. We calculated the follow-up period from the surgery date to the last follow-up or the occurrence of an event, such as death. Overall survival (OS) was determined as the interval from surgery to death from any cause. In instances where death was not recorded, the data was marked as censored at the date of the last follow-up.
Studies
Mammograms were performed using full-field digital mammography (FFDM) with the Senographe 2000D system from GE Healthcare, employing standard craniocaudal and mediolateral oblique views. The dynamic contrast-enhanced MRI scans were performed on various systems: either a 1.5-T system, including the Signa Excite by GE Healthcare and Espree or Avanto by Siemens Healthcare, or a 3-T Verio system from Siemens Healthcare. All MRI scans utilized a standard, bilateral, dedicated breast coil (Sentinelle Vanguard; Sentinelle Medical, Inc., Toronto, Canada). The MRI procedures adhered to the ACR’s quality standards. 17
Reader Study
The reader study included 3 breast imaging radiologists (JYP, PA, and VF with 9, 6, and 22 years of experience, respectively. They evaluated the imaging results using the Breast Imaging Reporting and Data System (BI-RADS) lexicon, 5th edition. 18 When there were differences in their assessments, 2 of the radiologists (PA and VF) jointly reviewed the cases to achieve a consensus on the imaging descriptors. It is noteworthy that in line with guidelines for screening high-risk individuals, only mammograms and MRIs are recommended. Consequently, our analysis focused exclusively on these techniques. Radiologists did not evaluate other techniques, such as digital breast tomosynthesis and ultrasound. Despite being aware they were evaluating a cohort of cancer patients, the reviewing radiologists were unaware of the specific high-risk subgroup to which the patients belonged and also remained blind about which imaging modality revealed the cancer, as well as the specific breast and location of the malignancy. To mimic a real-world setting, where tests are typically conducted concurrently to allow for comparative analysis, no washout period was carried out between the tests, enabling the reviewer radiologists to incorporate findings from mammograms and MRIs in their assessments.
Statistical Analysis
Cohort characteristics were summarized using descriptive statistics, and differences by high-risk group were assessed using the Kruskal-Wallis and chi-square tests for continuous and categorical variables, respectively. For the reader study, Fleiss Kappa was calculated to determine the level of agreement/disagreement amongst readers. The Kaplan-Meier method was used to estimate OS and the 5 and 10-year survival probabilities for each high-risk subgroup. The log-rank test and an univariable Cox proportional hazards model were used to assess differences in OS by high-risk subgroup. All hypothesis tests were 2-sided, and P-values less than .05 were considered statistically significant. Statistical analyses were conducted using R version 4.2.1.
Results
Demographic and Tumour Characteristics
Between January 2010 and December 2019, 140 high-risk women with a BC diagnosis were identified and included in this study (1. Group of gene carriers included: BRCA-1, n = 36; BRCA-2, n = 27, Neurofibromatosis Type 1, n = 4, PALB2, n = 1, AKT1E17K, n = 1, LiFraumeni Syndrome, n = 1, Peutz-Jeghers Syndrome, n = 1; 2. mantle radiation, n = 18; and 3. familial risk, n = 50). These women accounted for a total of 155 diagnosed malignant lesions within the study period. Of these, 5 patients, accounting for 3.6% (5/140) of the cohort, were found to have contralateral synchronous BC, diagnosed within the same year as the initial breast cancer. Out of 140 patients, 67 (47.8%, 67/140) opted for prophylactic mastectomy following the initial breast cancer diagnosis. No cases of cancer were detected exclusively through prophylactic surgery without corresponding imaging evidence. Among the remaining 73 patients, 9 patients, 12% (9/73), developed subsequent BC, including 4 ipsilateral recurrences and 6 metachronous contralateral cancers. One patient (11.1%, 1/9) developed 2 additional separate malignancies, one ipsilateral recurrence, and one contralateral metachronous disease, and 8 patients (88.9%, 8/9) developed a separate ipsilateral recurrence/contralateral metachronous malignancies over time. The median interval time between primary malignancy and the development of the second recurrence/metachronous malignancy was 2 years (interquartile range [IQR]: 1-8 years). The study flowchart is in Figure 1.
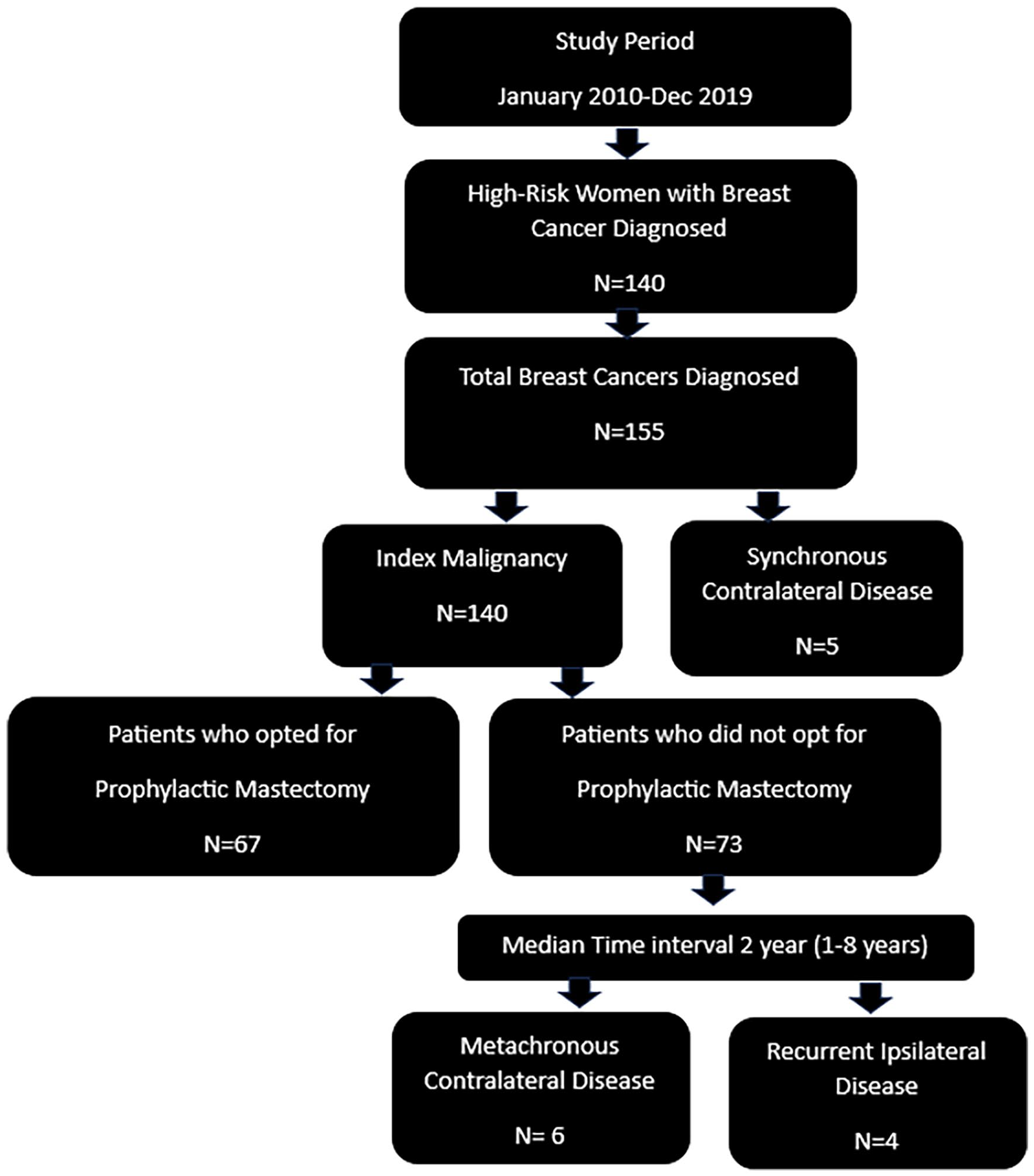
Study workflow.
The median age of diagnosis in the gene mutation group was 48.5 years (IQR: 39.8-58.0 years), 43.5 years in the chest radiation group (IQR: 37.0-46.2 years), and 51.0 years (IQR: 44.5-56.0 years) in the familial high-risk group. A statistically significant difference was observed in the age at diagnosis of breast cancer among these groups, with the chest radiation group being diagnosed at a notably younger age (P = .007).
In terms of histological subtypes within the study, invasive tumours were more common than in situ diseases as the primary form of diagnosed malignancy. However, this predominance did not show a statistically significant difference across the various high-risk subgroups (P = .48). While there were no significant disparities in pTstage (P = .25), lymphovascular invasion status (P = .53), or axillary node involvement (P = .95), notable differences were observed in other areas. Specifically, patients with gene mutations were diagnosed with smaller tumours (P = .01), higher histological grades (P = .002), and a greater incidence of triple-negative tumours. This includes higher proportions of ER-negative (P = .02), PR-negative (P = .002), and HER2-negative (P = .02) cancers.
Table 1 summarizes the patients’ demographics and tumour characteristics of the high-risk groups.
Demographics and Tumour Characteristics of the High-Risk Groups.
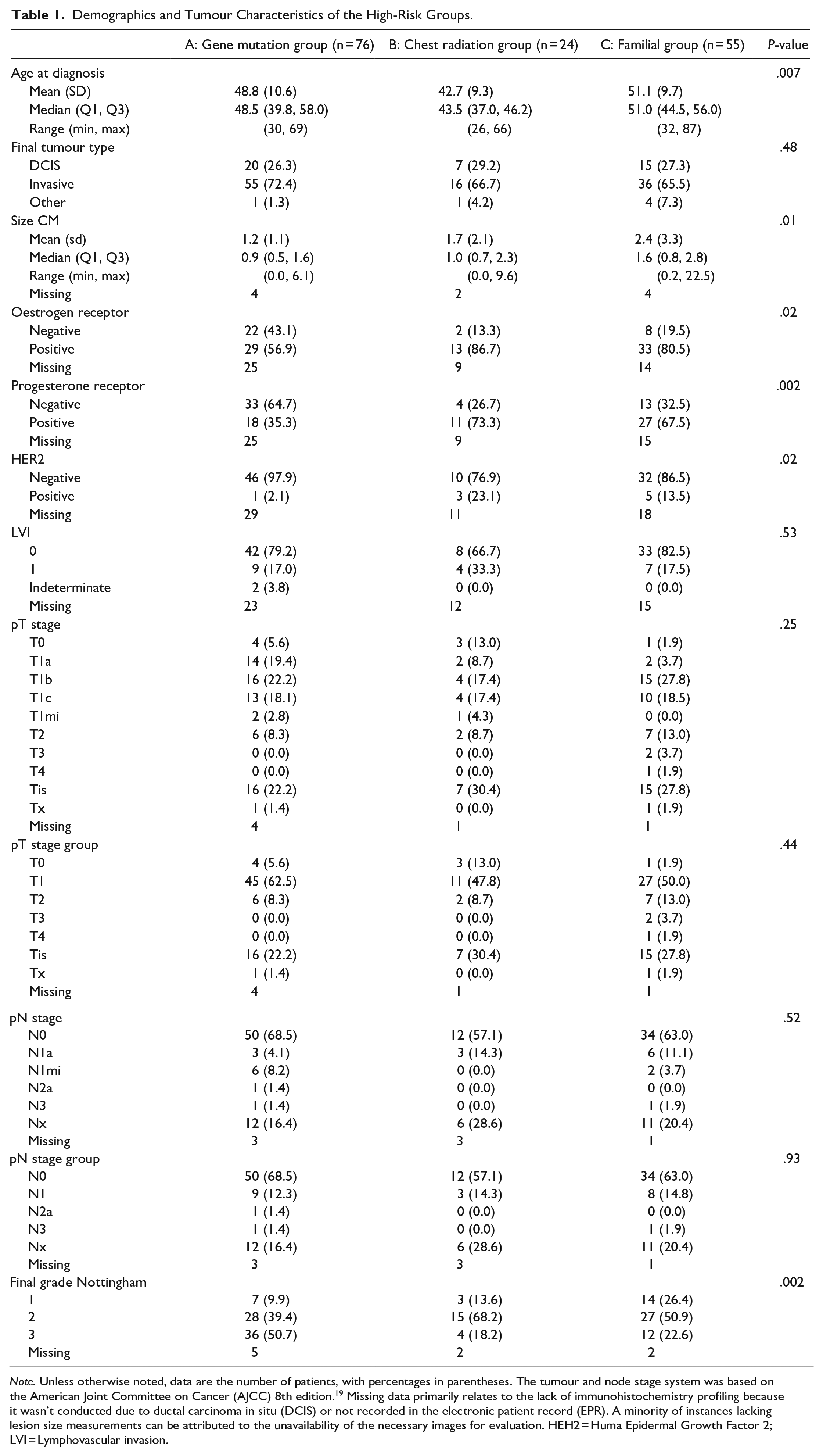
Note. Unless otherwise noted, data are the number of patients, with percentages in parentheses. The tumour and node stage system was based on the American Joint Committee on Cancer (AJCC) 8th edition. 19 Missing data primarily relates to the lack of immunohistochemistry profiling because it wasn’t conducted due to ductal carcinoma in situ (DCIS) or not recorded in the electronic patient record (EPR). A minority of instances lacking lesion size measurements can be attributed to the unavailability of the necessary images for evaluation. HEH2 = Huma Epidermal Growth Factor 2; LVI = Lymphovascular invasion.
Imaging
Out of the 155 malignant lesions identified during the study period, MRI was not performed in 9 cases, and although 2 mammograms were carried out outside, they were not accessible for review. The MRI and mammogram were performed within a 3-month interval in 129 cases (83%) and on the same day in 65 cases (42%). The median interval between mammogram and MRI was 25.5 days (range 0-264 days). In evaluating the effectiveness of imaging techniques, MRI frequently detected most of the BC across all high-risk subgroups, 72 out of 76 cases (96%) in the gene mutation group, 20 out of 24 cases (95.2%) in the chest radiation group, and 47 out of 55 cases (94%) in the familial group. Conversely, mammography often yielded negative results except in those with a history of mantle radiation therapy, 43 out of 76 cases (56.6%) in the gene mutation group, 9 out of 24 cases (39.1%) in the chest radiation group, and 36 out of 55 cases (66.7%) in the familial group. Overall, MRI yielded positive results in 139 (95.2%) of the cases, while MMG showed a positive detection rate of 65 (42.5%). MRI imaging alone depicted 85 cancers (85/155—54%), and mammograms depicted alone only 8 cancers (8/155—5%). For the 5 synchronous diseases, all in situ diseases, mammography exclusively detected one as microcalcifications, yielding an incremental detection rate (ICDR) of 0.6% (1/155). In contrast, MRI solely depicted 2 cancers with an ICDR of 1.3% (2/155). One case of an in situ disease was solely identified during the prophylactic mastectomy. Regarding imaging descriptors, none provided a statistically significant ability to differentiate between the high-risk groups. In cases where mammography identified cancer, the most frequent observation in the gene mutation group was microcalcifications, accounting for 22.4% (17/76, P = .05). However, the specific morphology and distribution of these calcifications did not aid in distinguishing malignancies among the subgroups (P = .11 and P = .91, respectively). Similarly, the shape and margins of masses identified in mammograms were not indicative of specific high-risk subgroups (P = .14 and P = .40, respectively). Although ACR breast density categories B (scattered fibroglandular densities) and C (heterogeneously dense) 18 were more prevalent, they did not vary significantly across groups (P = .38). In MRI, irregular mass lesions were commonly observed, but this finding did not significantly differ across the subgroups of high-risk patients (P = .43). The enhancement kinetics and lesion location within the breast also did not provide differentiation among the high-risk subgroups (P = .43 and P = .91, respectively). The detailed clinical and imaging features for each subgroup are compiled in Tables 2 and 3.
Mammography Findings of the High-Risk Groups.
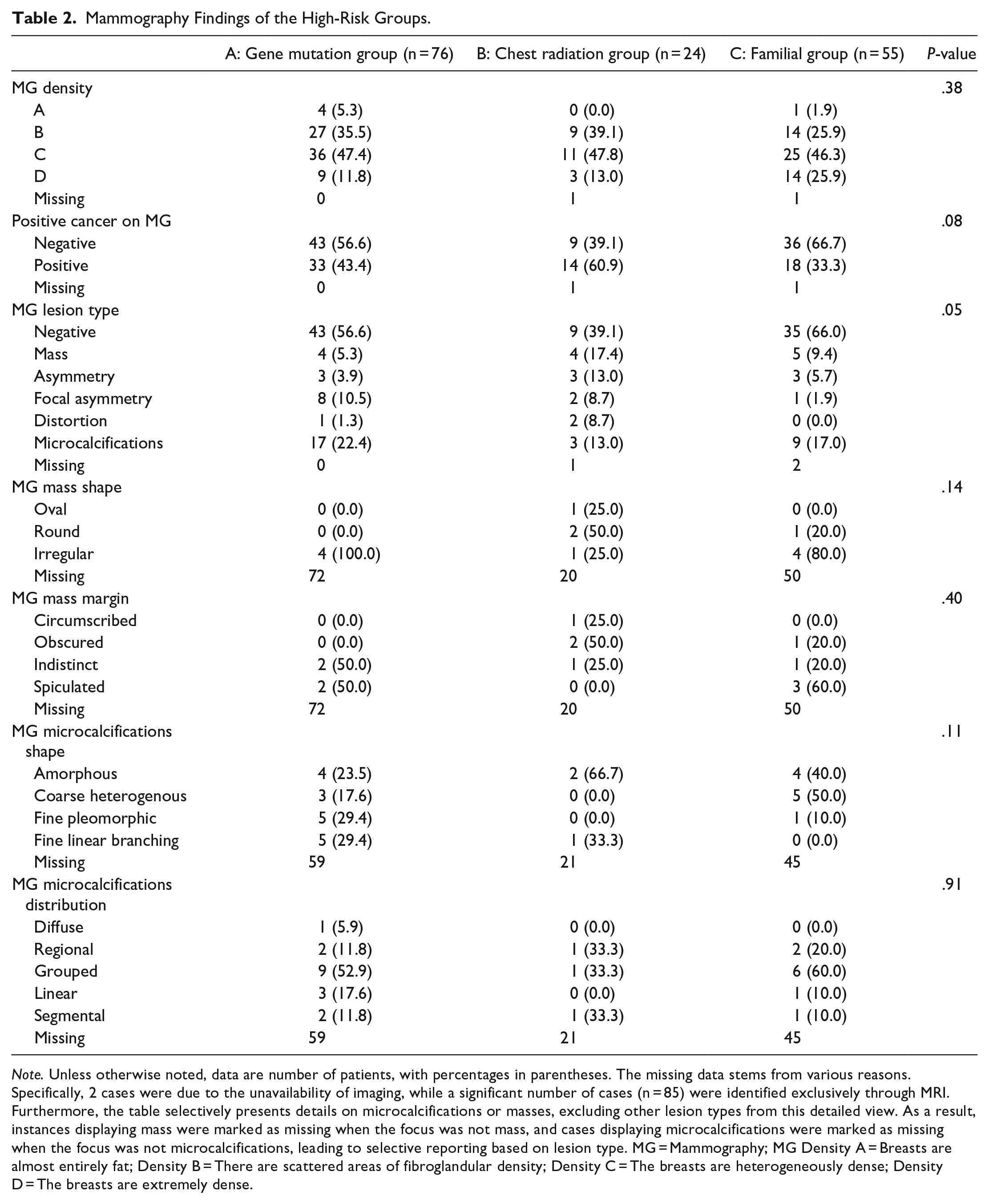
Note. Unless otherwise noted, data are number of patients, with percentages in parentheses. The missing data stems from various reasons. Specifically, 2 cases were due to the unavailability of imaging, while a significant number of cases (n = 85) were identified exclusively through MRI. Furthermore, the table selectively presents details on microcalcifications or masses, excluding other lesion types from this detailed view. As a result, instances displaying mass were marked as missing when the focus was not mass, and cases displaying microcalcifications were marked as missing when the focus was not microcalcifications, leading to selective reporting based on lesion type. MG = Mammography; MG Density A = Breasts are almost entirely fat; Density B = There are scattered areas of fibroglandular density; Density C = The breasts are heterogeneously dense; Density D = The breasts are extremely dense.
MRI Breast Cancer Findings of the High-Risk Groups.
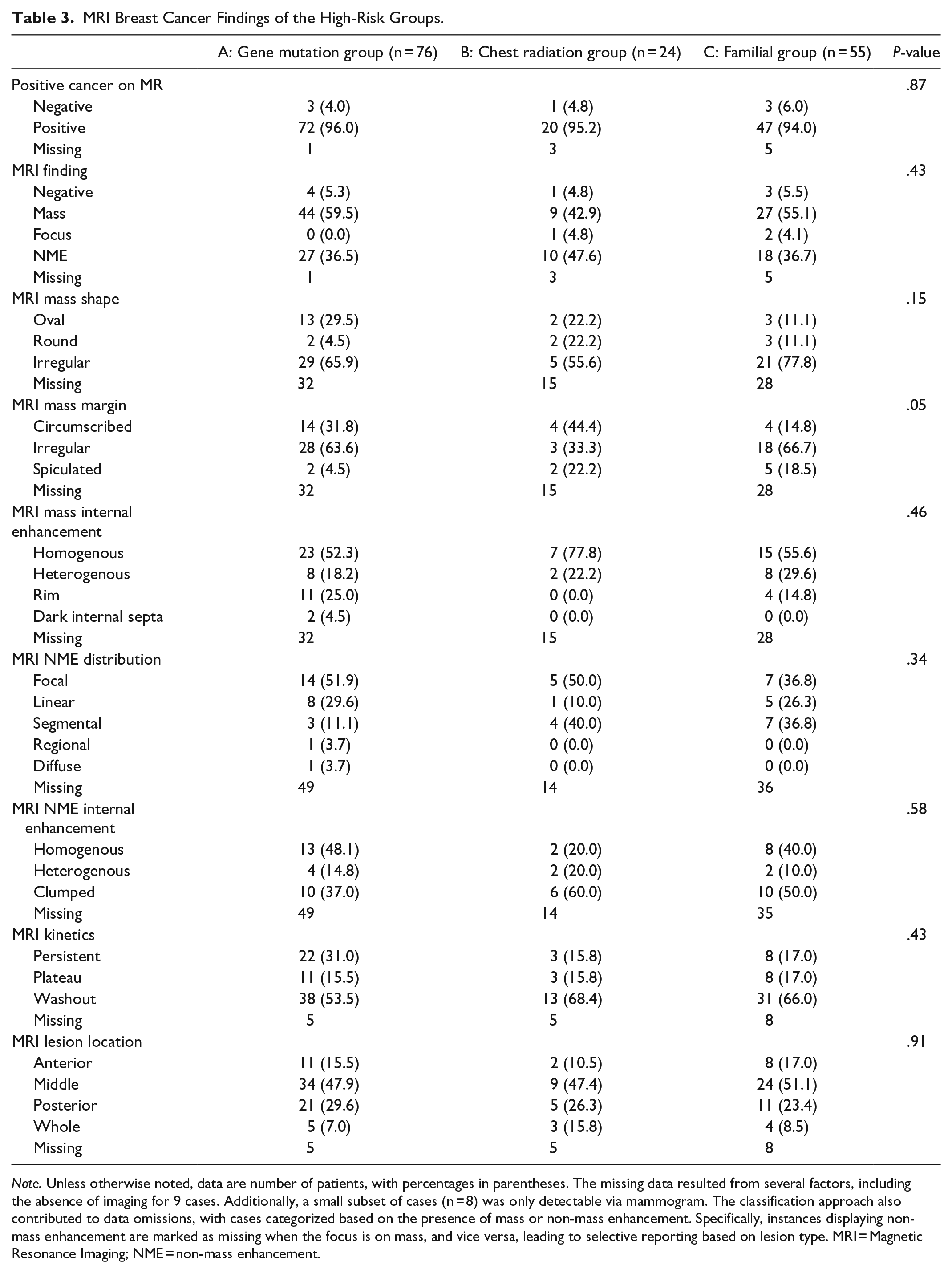
Note. Unless otherwise noted, data are number of patients, with percentages in parentheses. The missing data resulted from several factors, including the absence of imaging for 9 cases. Additionally, a small subset of cases (n = 8) was only detectable via mammogram. The classification approach also contributed to data omissions, with cases categorized based on the presence of mass or non-mass enhancement. Specifically, instances displaying non-mass enhancement are marked as missing when the focus is on mass, and vice versa, leading to selective reporting based on lesion type. MRI = Magnetic Resonance Imaging; NME = non-mass enhancement.
Interobserver Agreement
The interobserver agreement analysis revealed a substantial to near-perfect consensus among the 3 radiologists across all evaluated parameters. This included their ability to detect breast cancer using either mammography or MRI, along with all other imaging parameters. Detailed information on this agreement can be found in the Supplemental Material, specifically in Tables S1 to S16.
Long-Term Outcome
The last date of data collection was March 21, 2023. The follow-up duration was calculated from the date of surgery to the last date of follow-up or to the occurrence of the event (death), with a median of 5 years (0-13 years). Over the course of the follow-up, there were no recorded deaths among patients who had previously undergone chest radiation therapy. Additionally, when comparing patients with gene mutations to those with familial risks, no significant differences were observed in overall survival (OS), with overall 6 events out of 74 versus 4 events out of 55, respectively, with a hazard ratio (HR) of 0.93 (95% CI: 0.27, 3.26), P = .92. The survival probabilities for the high-risk group are depicted in Figure 2.
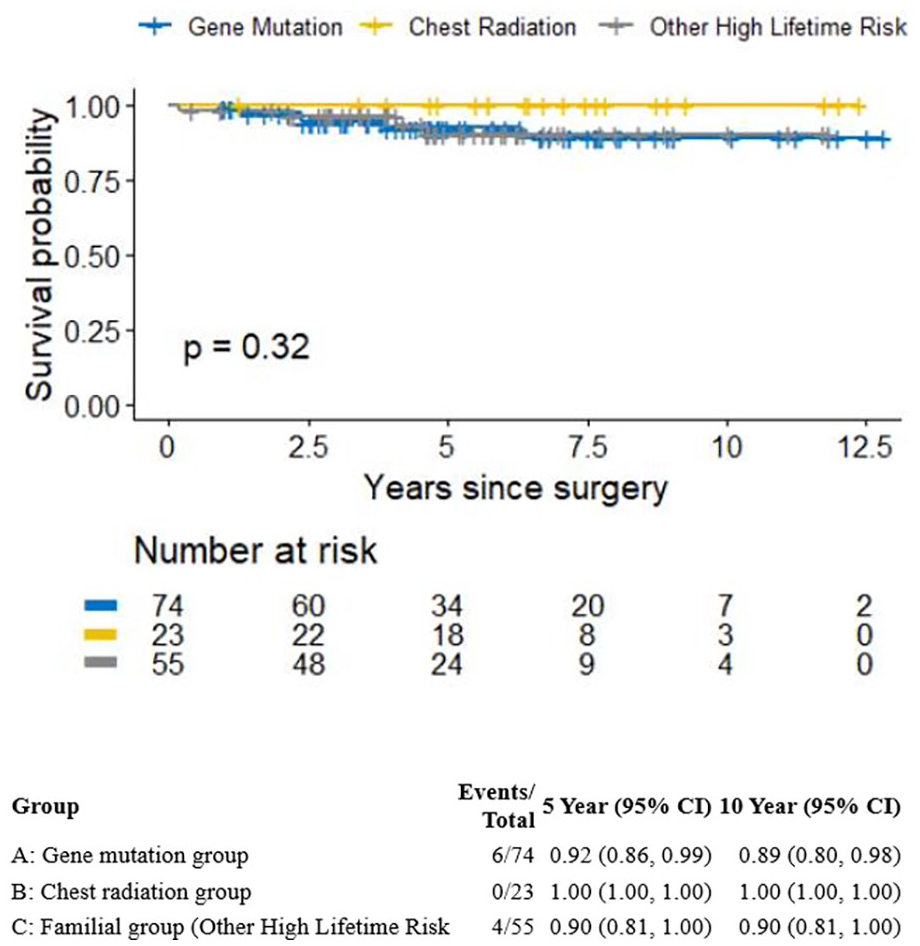
The survival probabilities of the high-risk groups.
Discussion
This study conducted a comprehensive evaluation of clinical, pathological, and imaging characteristics in breast cancer cases detected among the most prevalent high-risk groups, including those with genetic mutations, patients who had previous chest radiation, and familial risk groups, aiming to discern distinctions between these groups. The findings revealed that while all subgroups exhibited a younger age at tumour diagnosis, individuals with a history of chest radiation therapy were even more likely to be diagnosed at younger ages (P = .007). Moreover, significant variations in tumour size, histological grade, and molecular subtypes were observed among high-risk patient subgroups. Notably, smaller tumour sizes (P = .01), higher histological grades (P = .002), and a greater prevalence of triple-negative malignancies, oestrogen receptor negative (P = .02), progesterone receptor negative (P = .002), and HER2 negative (P = .02) were noted in those with gene mutations. In terms of imaging characteristics, mammography frequently yielded negative results and failed to detect malignancies (88/155—57%). Conversely, MRI consistently exhibited positive results across the high-risk groups (139/155—90%), emphasizing its importance as a screening imaging tool. Furthermore, the study exceeded expectations by demonstrating that, despite the higher occurrence of aggressive cancer subtypes in gene mutation carriers, this did not negatively impact their survival rates HR = 0.93 (95% CI: 0.27, 3.26), P = .92. This highlights the critical role of intensive screening in identifying smaller malignancies within this patient subgroup.
Consistent with our findings, overall, high-risk patients tend to be diagnosed with breast cancer at a younger age compared to women at average risk. 20 While there is limited evidence that directly compares the age of onset for breast cancer in the high-risk groups, most studies indicate an earlier diagnosis, particularly among BRCA 1/2 mutation carriers. These studies report a range of 41 to 50 years for BRCA-1 mutation carriers and 51 to 60 years for BRCA-2 mutation carriers. 21 Additionally, in line with our observations, Moskowitz et al 22 found that individuals with a history of chest radiation were diagnosed at a median age of 39 years. Therefore, our study’s findings align with existing evidence23,24 and current guidelines,2,25,26 emphasizing the importance of considering the initiation of screening at a younger age for all high-risk patients and even earlier for those with a history of chest radiation.
Regarding the pathological aspects, our study’s findings align with previous research 10 in highlighting the prevalence of invasive carcinomas over in situ disease. This observation is noteworthy, considering the uncertain clinical significance of low and intermediate in situ disease diagnoses.27,28 Moreover, within the gene mutation group, our study establishes a significant association with triple-negative malignancies and a higher frequency of tumours with elevated histological grades. These findings are consistent with those reported in other studies.20,29,30 Consequently, our study underscores the critical importance of early detection for these high-risk patients, particularly when these aggressive cancers are still small in size. This approach can lead to improved outcomes for patients, 31 as also evidenced by our study, which found no differences in overall survival between the high-risk groups.
Our study’s findings contribute positively to the existing body of research on the effectiveness of different imaging modalities in detecting cancer within high-risk groups. We found that MRI was notably effective, identifying the majority of malignancies, with 139 out of 155 cases (95.2%) detected, 85 of which (54%) were identified exclusively by MRI. This compares to 65 cases (42.5%) identified by mammogram, with a smaller fraction, 8 cases (5%), detected solely through mammography. These results align with previous studies, such as the one by Lo et al, 32 which, in a study of 1249 high-risk patients undergoing 3934 screening rounds, found 45 malignancies, 29 (64%) of which were detected solely by MRI and none exclusively by mammogram. Similarly, Vreemann et al’s 33 research on 2026 high-risk patients across 6553 screening rounds identified 125 screen-detected cancers, with MRI exclusively identifying 59 (47%) of these, compared to only 13 (10%) detected solely by mammogram. These findings raise the question of whether MRI alone could serve as the primary screening test, potentially allowing for the discontinuation of annual mammography for high-risk patients who have access to quality-assured MRI. This shift towards MRI as the primary screening tool could hold relevance in efforts to reduce the cumulative radiation exposure that high-risk patients face over their lifetimes. This consideration is especially crucial given the radiation sensitivity observed in certain gene mutation groups and the potential mutagenic effects of radiation on young fibroglandular tissue, particularly in cases where early screening begins at the age of 25 to 30 years. 34
Furthermore, the higher MRI incremental cancer detection rate (ICDR) for synchronous disease in our high-risk cohort, which was 1.3% compared to 0.6% for mammography, falls well within the range reported in previous studies conducted in similar settings (MRI’s ICDR range: 0.8%-4.4%).35-48 This highlights the critical role of MRI in identifying additional contralateral malignancies, potentially reducing delays in their detection, which would lead to a second round of treatment, with negative implications for both patients and the healthcare system. It is also worth noting that despite 48% of our cohort opting for prophylactic mastectomy after their first breast cancer diagnosis as a strategy to lower the risk of metachronous disease development, among the remaining 73 patients who did not opted for this procedure, we observed a rather high rate of ipsilateral recurrence/contralateral metachronous disease (12%—9/73). This rate significantly exceeds the findings of a previous study focused primarily on a non-high-risk population, which reported a metachronous disease rate of 4.5% using pre-operative MRI. 49 This underscores the continued need for rigorous clinical and imaging surveillance after the initial cancer diagnosis in high-risk populations, especially in those who have chosen not to pursue prophylactic surgery or chemoprevention as risk reduction strategies.
In contrast to previous studies10,11 that identified malignant imaging descriptors capable of distinguishing high-risk subgroups, our study did not reveal any imaging descriptors that could statistically differentiate among the high-risk groups. Additionally, while Ha et al 8 observed that, unlike in BRCA2 mutation carriers, cancers in BRCA1 mutation carriers typically developed in the breast’s posterior area (P = .034), our combined analysis of these mutations, when compared to other high-risk groups, did not indicate location as a significant determinant in distinguishing cancer in high-risk groups. These discrepancies are likely attributed to the relatively limited sample size within our study, when subdividing the high-risk groups, which may have restricted our ability to detect all significant differences in clinical and imaging features among high-risk patients. In this sense, artificial intelligence classifiers distinguishing benign and malignant lesions have been developed and hold potential significance in this context in the near future.50,51
Our study has certain limitations that need to be acknowledged. One of the primary limitations is rooted in the study’s design as an observational cross-sectional study. This design introduces the possibility of selection bias, wherein crucial parameters capable of distinguishing between the evaluated high-risk patient subgroups may remain unidentified. Additionally, the limited sample size, especially when subdividing high-risk groups, diminishes the statistical power of our study. Consequently, our study may not possess the necessary statistical robustness to detect all significant differences among high-risk patients. Furthermore, although the majority of the genetic predisposition group consisted of BRCA 1/2 gene mutation carriers, other genetic predisposition subgroups were also included in the study, which might exhibit distinct patterns of outcomes compared to the BRCA 1/2 group. However, due to the limited sample sizes of these subgroups, it was challenging to identify these distinct patterns conclusively. Moreover, due to the nature of the data collection, which was based on successive imaging studies of high-risk patients who were presented in the radiology department and had a confirmed malignant lesion, with a range of circumstances, including pregnancy, missed appointments, or choosing not to undergo imaging, some individuals eligible for high-risk screening were not imaged, we were unable to provide accurate information regarding the number of high-risk screenings during the study period to provide cancer detection rate in the study. Lastly, it is important to recognize that our study was conducted exclusively at a single academic tertiary cancer centre. Consequently, the generalizability of our results to all healthcare centres may be limited.
In conclusion, the findings indicate that the age of patients and certain tumour pathological characteristics can distinguish between the predominant high-risk groups. MRI has proven effective as a screening tool, emphasizing the importance of its use at early ages. Although gene mutation carriers show aggressive cancer traits, timely detection appears pivotal in influencing their survival outcomes. These insights, while necessitating further validation with larger studies, advocate for a move towards personalized medical care, strengthening the existing healthcare guidelines.
Supplemental Material
sj-docx-1-caj-10.1177_08465371241253254 – Supplemental material for Decoding the Prevalent High-Risk Breast Cancers: Demographics, Pathological, Imaging Insights, and Long-Term Outcome
Supplemental material, sj-docx-1-caj-10.1177_08465371241253254 for Decoding the Prevalent High-Risk Breast Cancers: Demographics, Pathological, Imaging Insights, and Long-Term Outcome by Pedro Alvarenga, Ji Yeon Park, Renata Pinto, Daniella Parente, Katherine Lajkosz, Shelley Westergard, Sandeep Ghai, Raymond Kim, Supriya Kulkarni, Frederick Au, Juliana Chamadoira and Vivianne Freitas in Canadian Association of Radiologists Journal
Footnotes
Abbreviations
ACR American College of Radiology
BC Breast Cancer
BI-RADS Breast Imaging Reporting and Data System
BOADICEA Breast and Ovarian Analysis of Disease Incidence and Carrier Estimation Algorithm model
BRCAPRO BRCA mutation carrier prediction model
FFDM Full Field Digital Mammogram
HR Hazard Ratio
ICDR Incremental Cancer Detection Rate
IQR Interquartile Range
MRI Magnetic Resonance Imaging
OBSP Ontario Breast Screening Program
OS Overall Survival
Author Contributions
All authors contributed to the study’s conception and design.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
