Abstract

Keywords
Clinical Statement
Reanalysing the Cochrane review after excluding CNBSS found no mortality benefit of .80 (P = .05) for women aged 40–49 years invited to screening mammography. Further research with modern equipment or the inclusion of large observational trials is needed for this clinical equipoise.
Introduction
Breast cancer screening using mammography is effective in reducing mortality for women in the 50-69-year-old group. Benefit in the 40-49-year-old group remains controversial. 1
The Canadian Task Force on Preventive Health Care’s (CTFPHC) 2018 guideline for breast cancer screening does not recommend routine screening for this age group. The evidence for this recommendation is largely based on a Cochrane Review of 8 randomised controlled trials. 2 Methodological flaws were identified in several of the included trials; therefore, the primary evidence was based on a sensitivity analysis of the 4 low risk of bias studies with ‘adequate randomisation’ (CNBSS1, CNBSS2, Malmo 3 , UK Ages 4 ). The benefit of screening was not significant when analyses included only adequately randomized trials (.94 [.77, 1.15] in women 50–69 years old and .87 [.73, 1.03] in women 40–49 years old) at 13-year follow-up.
The 2 Canadian National Breast Screening Study (CNBSS) trials were originally considered at a low risk for bias, and they were the only 2 of all the studies that did not demonstrate a mortality benefit for breast cancer screening. However, recent research uncovered methodological flaws in the CNBSS that were not evident at the time of the Cochrane review. These include subversion of allocation of randomisation (open-book), inclusion of women with symptoms of breast cancer and poor quality of mammography. 5 As such, the CNBSS should not be considered at a ‘low risk of bias’ and should not have been retained in the Cochrane review sensitivity analysis.
The purpose of this study is to re-evaluate the strength of the evidence of the Cochrane review for assessment of mortality benefit of breast cancer in women aged 40–49 years by reanalysing its results considering that the CNBSS are at high risk of bias.
Methods
This study was a secondary analysis of previously published data and did not require REB approval. We did not undertake a repeat search, screen, or data extraction. We took results of the Cochrane review for women 40–49 years of age who were randomized to invitation to screening mammography vs usual care and re-analysed them based on new evidence indicating that the CNBSS is at high risk of bias. Data synthesis and analysis were modelled after the original review. 2 Data were pooled using a fixed‐effect model with the Mantel–Haenszel method. Results are presented with 95% confidence intervals and plotted in graphs as risk ratios (RR). Outcome data is reported at 7- and 13-year follow-up and the outcome of interest was breast cancer mortality. Analyses were performed in Review Manager (RevMan) [Computer program Version 5.4].
Results
Characteristics of Randomized Trials Included in the Cochrane Review. 2
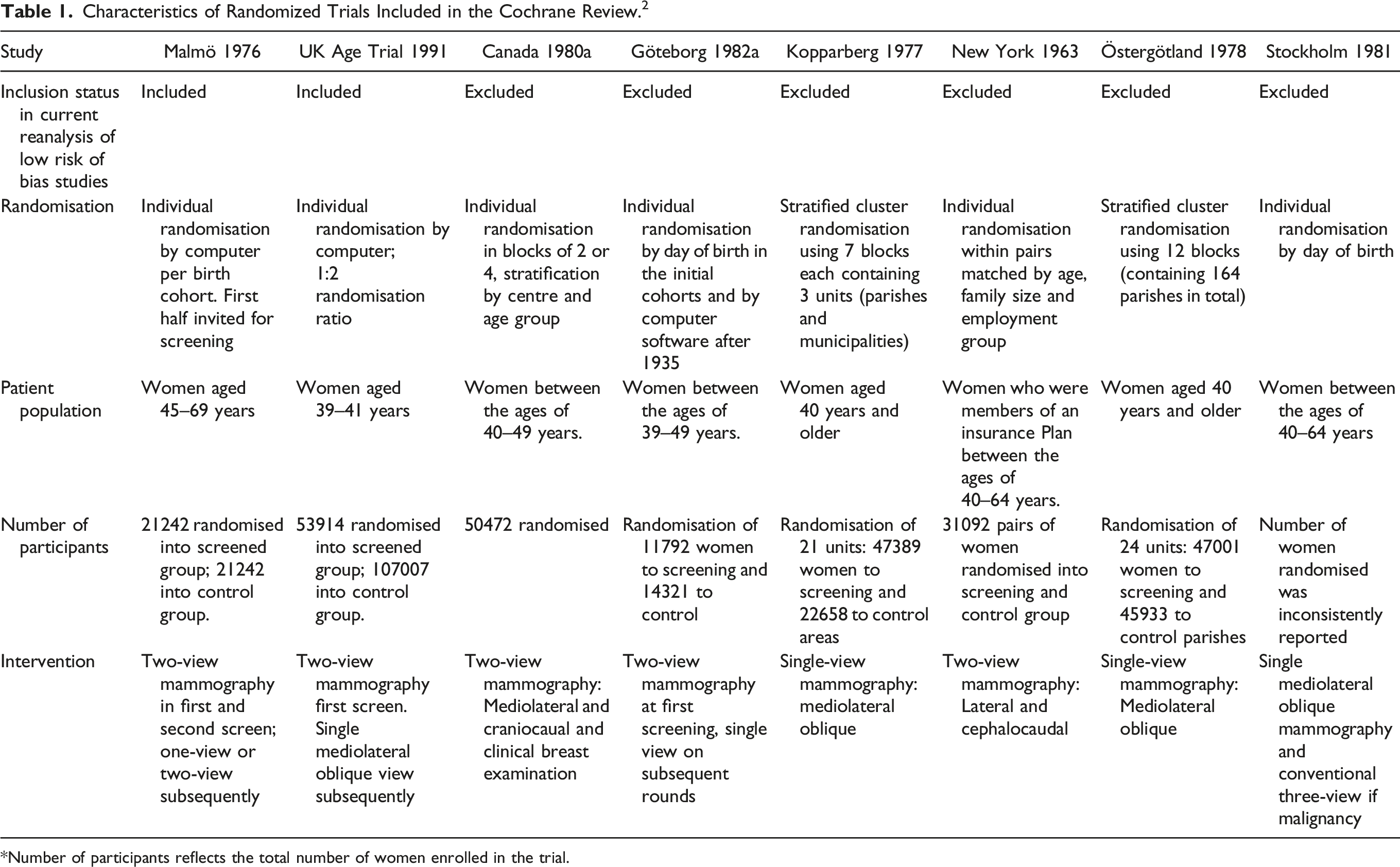
Number of participants reflects the total number of women enrolled in the trial.
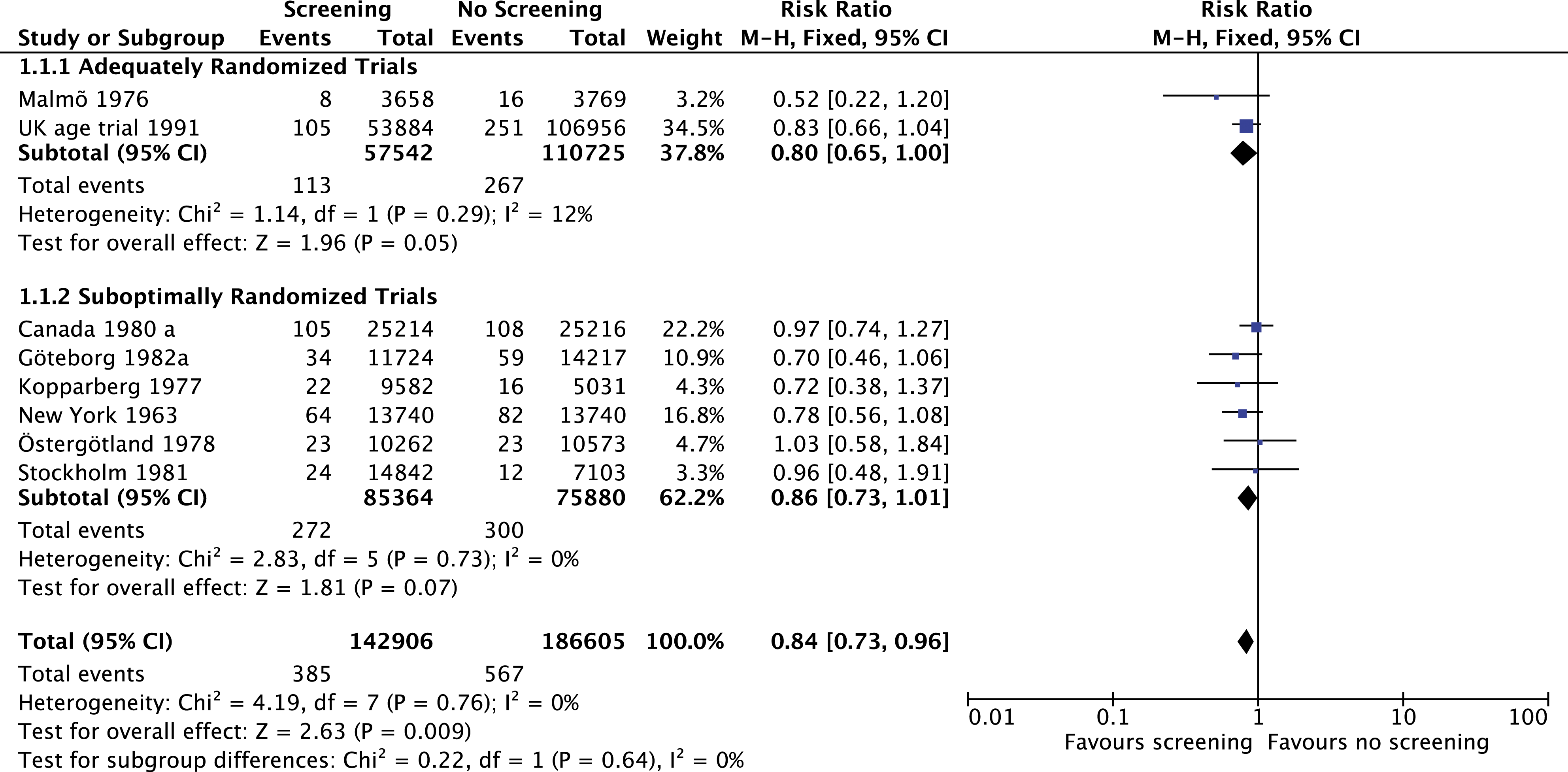
Deaths due to breast cancer at 7-year follow-up in women 40–49 years of age in the screening compared to the no screening group. The Canada 1980a study represents the Canadian National Breast Screening Study (CNBSS 1).
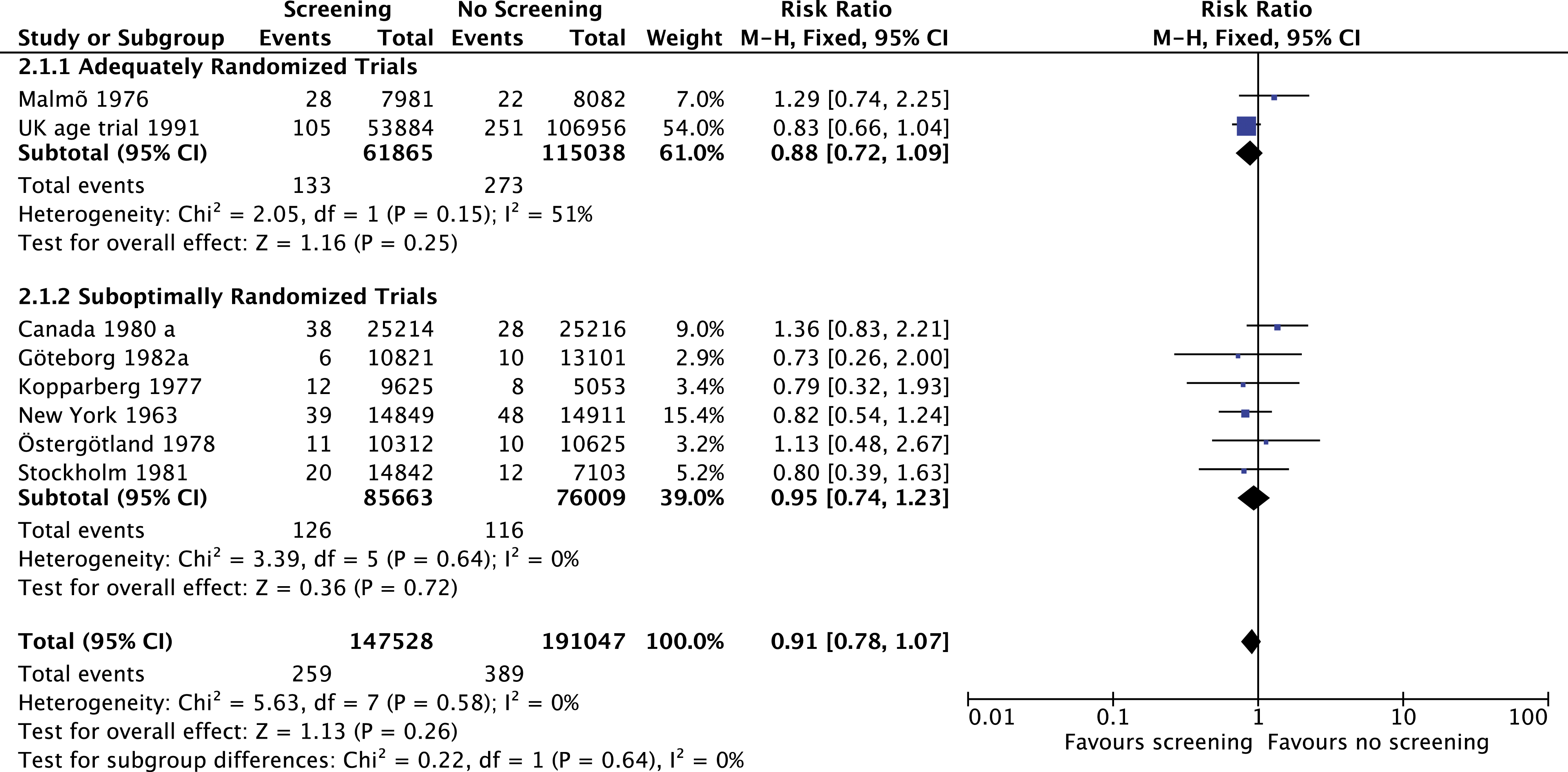
Deaths due to breast cancer at 13-year follow-up in women 40–49 years of age in the screening compared to the no screening group. The Canada 1980 a study represents the Canadian National Breast Screening Study (CNBSS 1).
Discussion
Our analysis demonstrates that re-classifying the CNBSS as high risk of bias will not have an impact on the results of the Cochrane review that informed guidelines such as the CTFPHC guideline on breast cancer screening. When the CNBSS were included in the previous review, no significant reduction in breast cancer mortality was identified (RR .87 [.73, 1.03]) in women 40–49 years old at 13-year follow-up; when excluding the CNBSS, the effect size remained non-significant at 13-year follow-up: .80 (.65–1.00) (P = .05).
Exclusion of the CNBSS trials from the sensitivity analysis does not impact the findings; there remains a lack of significant mortality benefit for the evaluated endpoints. However, the point estimate and confidence intervals indicate that this may be due to small sample size and inadequate power. These findings highlight the paucity of rigorous trials for assessing mammographic screening in women aged 40–49. Further study using contemporary medical imaging equipment and treatment techniques, as well as the consideration of results of large observational studies 6 is warranted to address this area of clinical debate.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
